Nanoparticles from the Cosmetics and Medical Industries in Legal and Environmental Aspects
Abstract
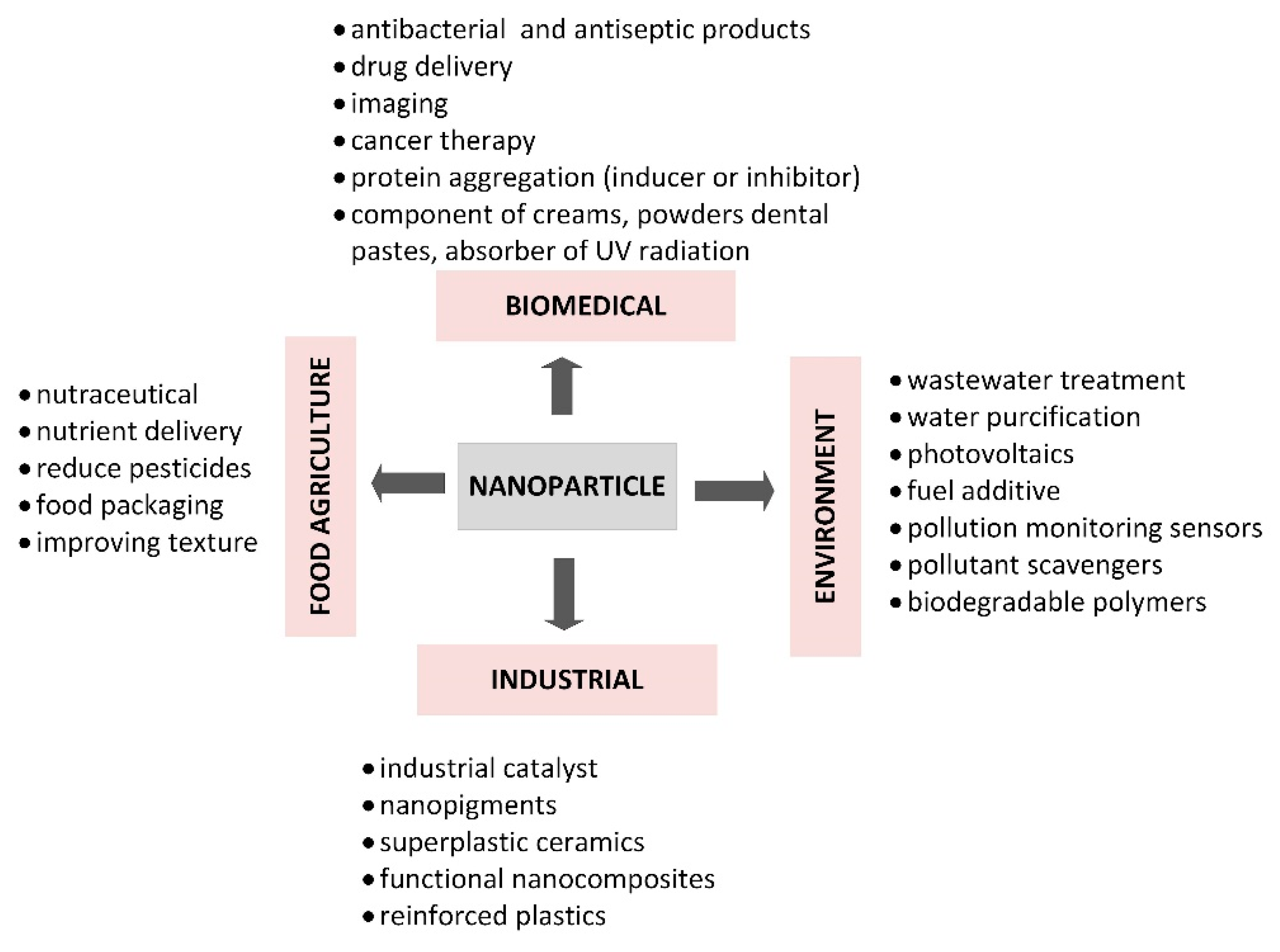
1. Application and Role of Nanoparticles in Cosmetology and Medicine
- the “top-down” method—in which the size of large structures is reduced to a nanometer scale by reducing the size (grinding) of materials to nanoparticles (products of the first generation);
- disruption of the electrical potentials of the cell membrane, nucleus and mitochondria (in bacteria);
- water management disorders (in the case of mushrooms);
- depriving the ability of catalytic degradation of the lipid-protein base (in viruses).
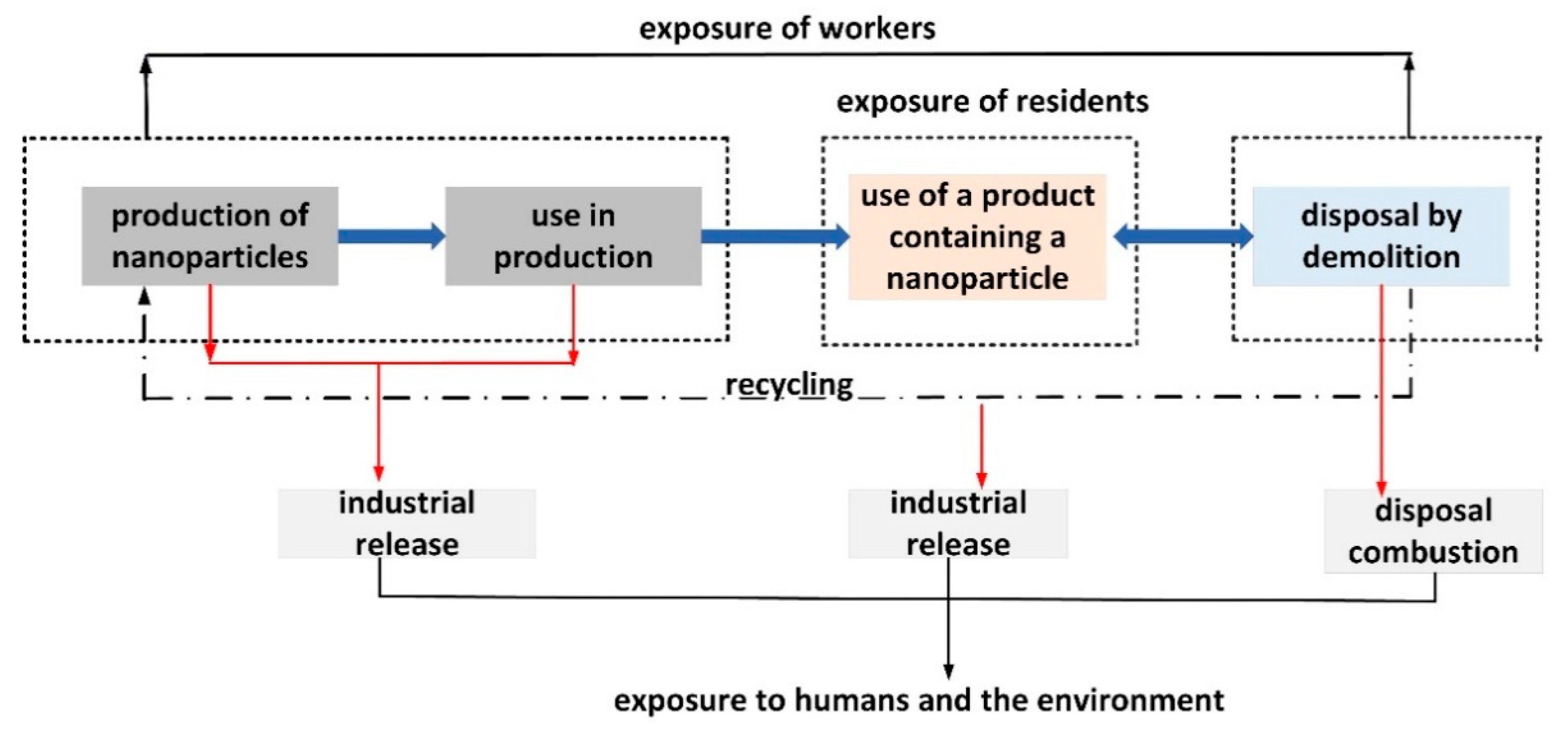
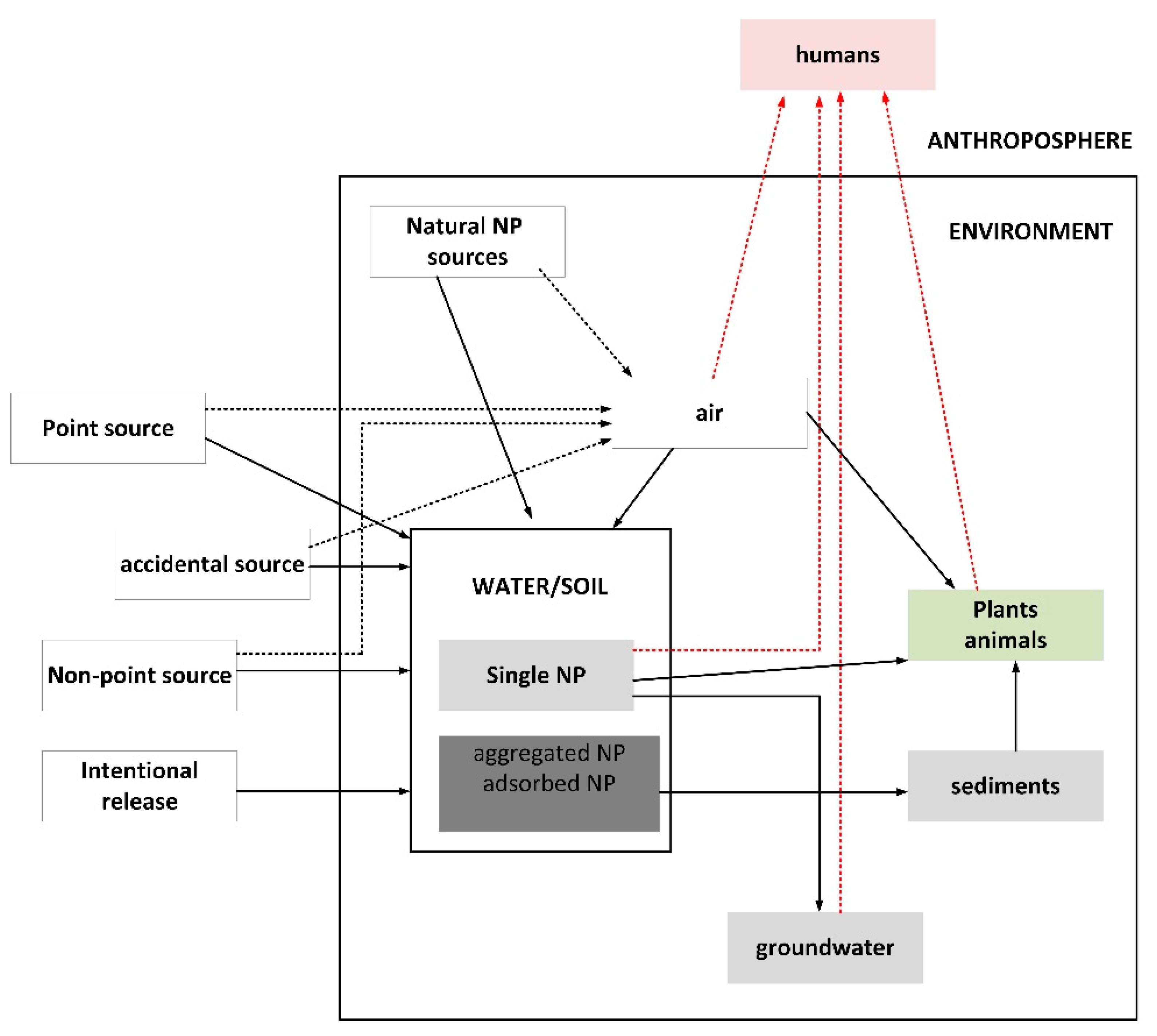
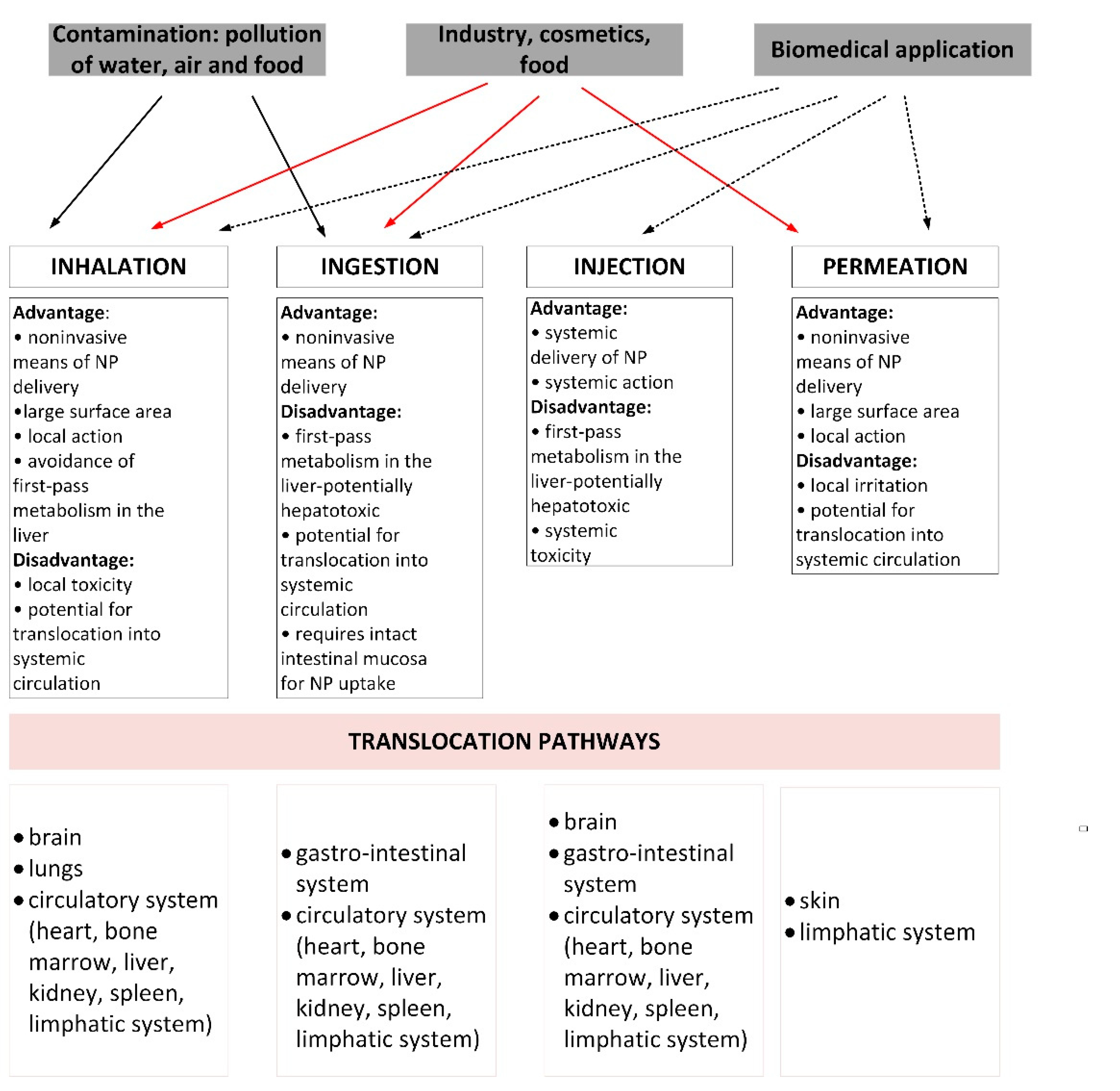
2. Environmental and Health Risk Resulting from the Use of Nanoparticles
- extremely toxic < 0.1 mg/L,
- very toxic 0.1–1 mg/L,
- toxic 1–10 mg/L,
- harmful 10–100 mg/L,
- non-toxic > 100 mg/L.
3. Legal and Environmental Regulations in the Field of Nanotechnology
- ISO/TS 10798:2011-Nanotechnologies—Characterization of single-wall carbon nanotubes using scanning electron microscopy and energy dispersive X-ray spectrometry analysis
- ISO/TS 10797:2012-Nanotechnologies—Characterization of single-wall carbon nanotubes using transmission electron microscopy
- ISO/TS 10868:2017-Nanotechnologies—Characterization of single-wall carbon nanotubes using ultraviolet-visible-near infrared (UV-Vis-NIR) absorption spectroscopy
- ISO/TR 11251:2019-Nanotechnologies—Characterization of volatile components in single-wall carbon nanotube samples using evolved gas analysis/gas chromatograph-mass spectrometry
- ISO/TS 11308:2020-Nanotechnologies—Characterization of carbon nanotube samples using thermogravimetric analysis
- ISO/TS 13278:2017-Nanotechnologies—Determination of elemental impurities in samples of carbon nanotubes using inductively coupled plasma mass spectrometry
- ISO/TS 18827:2017-Nanotechnologies—Electron spin resonance (ESR) as a method for measuring reactive oxygen species (ROS) generated by metal oxide nanomaterials
- ISO/TS 19590:2017-Nanotechnologies—Size distribution and concentration of inorganic nanoparticles in aqueous media via single particle inductively coupled plasma mass spectrometry
- ISO/TS 19807-1:2019-Nanotechnologies—Magnetic nanomaterials—Part 1: Specification of characteristics and measurements for magnetic nanosuspensions
- ISO/TS 21356-1:2021-Nanotechnologies—Structural characterization of graphene—Part 1: Graphene from powders and dispersions.
- in the hazard class “carcinogenicity” category 1A or 1B in accordance with the CLP Regulation;
- in the hazard class “germ cell mutagenicity” category 1A or 1B in accordance with the CLP Regulation;
- in the hazard class “reproductive toxicity”, including reproductive function and fertility, or on development of categories 1A or 1B in accordance with the CLP Regulation;
- persistent and very persistent substances, bioaccumulative and toxic according to the criteria described in REACH;
- other endocrine disruptors, the substances listed above and others for which there is scientific evidence of likely serious effects on human health or the environment.
4. Problems and Challenges in the Application of Nanotechnology in Environmental Engineering
- chemical properties: high chemical reactivity, increased corrosion resistance, diversity of chemical and phase composition;
- physical properties: small size, and at the same time a high tendency towards aggregation and/or agglomeration, diffusivity, large surface area compared to volume, which results in the appearance of strong sorption properties (adsorption and absorption) and an increase in the catalytic activity of nanomaterials;
- mechanical properties: hardness, abrasion resistance, superelasticity phenomenon occurring as a result of reducing the grain size of intermetallic cluster connections to the order of nanometers;
- biological properties: strong antibacterial properties, penetration through biological barriers, large range of impact due to dimensions and diffusivity.
5. Summary and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- American Society for Testing and Materials (ASTM). Standard terminology relating to nanotechnology. ASTM E 2006, 6, 2456–2506. [Google Scholar]
- Commission, E.E. Commission recommendation of 18 October 2011 on the definition of nanomaterial. Off. J. Eur. Union 2011, 50, 38–40. [Google Scholar]
- Pulit-Prociach, J.; Banach, M. Silver Nanoparticles—A Material of the Future...? Open Chem. 2016, 14, 76–91. [Google Scholar] [CrossRef]
- Madeła, M.; Neczaj, E.; Worąg, M.; Grosser, A. Zagrożenia środowiskowe nanocząsteczkami. Przemysł Chem. 2015, 94, 2138–2141. (In Polish) [Google Scholar]
- Świdwińska-Gajewska, A.M. Nanocząsteczki—produkt nowoczesnej technologii i nowe zagrożenie w środowisku pracy. Med. Pract. 2007, 58, 243–251. (In Polish) [Google Scholar]
- Nowack, B.; Bucheli, T.D. Occurrence, behavior and effects of nanoparticles in the environment. Environ. Pollut. 2007, 150, 5–22. [Google Scholar] [CrossRef]
- Verma, A.; Mehata, M.S. Controllable synthesis of silver nanoparticles using Neem leaves and their antimicrobial activity. J. Radiat. Res. Appl. Sci. 2016, 9, 109–115. [Google Scholar] [CrossRef]
- Ahmed, S.; Annu; Ikram, S.; Salprima, S.Y. Biosynthesis of gold nanoparticles: A green approach. J. Photochem. Photobiol. B Biol. 2016, 161, 141–153. [Google Scholar] [CrossRef]
- Waszkiewicz-Robak, B.; Świderski, F. Nanotechnologia-korzyści i zagrożenia zdrowotne. Bromat. Chem. Toksykol. 2008, 3, 202–208. (In Polish) [Google Scholar]
- Patil, M.P.; Kim, G.-D. Marine microorganisms for synthesis of metallic nanoparticles and their biomedical applications. Colloids Surf. B Biointerfaces 2018, 172, 487–495. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Xu, H.; Chen, Z.-S.; Chen, G. Biosynthesis of Nanoparticles by Microorganisms and Their Applications. J. Nanomater. 2011, 2011, 270974. [Google Scholar] [CrossRef]
- Shah, M.; Fawcett, D.; Sharma, S.; Tripathy, S.K.; Poinern, G.E.J. Green Synthesis of Metallic Nanoparticles via Biological Entities. Materials 2015, 8, 7278–7308. [Google Scholar] [CrossRef] [PubMed]
- Khan, I.; Saeed, K.; Khan, I. Nanoparticles: Properties, applications and toxicities. Arab. J. Chem. 2019, 12, 908–931. [Google Scholar] [CrossRef]
- Hasan, S. A review on nanoparticles: Their synthesis and types. Res. J. Recent Sci. 2015, 4, 2502. [Google Scholar]
- Liufu, S.-C.; Xiao, H.-N.; Li, Y.-P. Adsorption of cationic polyelectrolyte at the solid/liquid interface and dispersion of nanosized silica in water. J. Colloid Interface Sci. 2005, 285, 33–40. [Google Scholar] [CrossRef]
- Auffan, M.; Rose, J.; Bottero, J.-Y.; Lowry, G.V.; Jolivet, J.-P.; Wiesner, M.R. Towards a definition of inorganic nanoparticles from an environmental, health and safety perspective. Nat. Nanotechnol. 2009, 4, 634–641. [Google Scholar] [CrossRef] [PubMed]
- OECD. Guidance document for the testing of dissolution and dispersion stability of nanomaterials and the use of the data for further environmental testing and assessment strategies. Ser. Test. Assess. 2020, 318, 9. [Google Scholar]
- Ramirez, L.; Gentile, S.R.; Zimmermann, S.; Stoll, S. Behavior of TiO2 and CeO2 Nanoparticles and Polystyrene Nanoplastics in Bottled Mineral, Drinking and Lake Geneva Waters. Impact of Water Hardness and Natural Organic Matter on Nanoparticle Surface Properties and Aggregation. Water 2019, 11, 721. [Google Scholar] [CrossRef]
- Langauer-Lewowicka, H.; Pawlas, K. Nanocząstki, nanotechnologia—Potencjalne zagrożenia środowiskowe i zawodowe. Environ. Med. 2014, 17, 7–14. (In Polish) [Google Scholar]
- Salata, O.V. Applications of nanoparticles in biology and medicine. J. Nanobiotechnol. 2004, 2, 3. [Google Scholar] [CrossRef]
- Parlament Europejski. Rozporządzenie WE nr 1907/2006 z Dnia 18 Grudnia 2006 r. w Sprawie Rejestracji Oceny, Udzielania Zezwoleń i Stosowanych Ograniczeń w Zakresie Chemikaliów; Parlament Europejski: Bruksela, Belgia, 2006. (In Polish)
- Ministerstwa Gospodarki. 2013. Available online: www. reach-info.pl, punkt konsultacyjny do spraw REACH i CLP (accessed on 14 May 2021).
- Parlament Europejski. Rozporządzenie (WE) nr 1272/2008 z Dnia 16 Grudnia 2008 r. W Sprawie Klasyfikacji, Oznakowania i Pakowania Substancji i Mieszanin; Parlament Europejski: Bruksela, Belgia, 2006. (In Polish)
- Rzeszutek, J.; Matysiak, M.; Czajka, M.; Sawicki, K.; Rachubik, P.; Kruszewski, M.; Kapka-Skrzypczak, L. Zastosowanie nanocząstek i nanomateriałów w medycynie, Hygeia. Public Health 2014, 49, 49–457. (In Polish) [Google Scholar]
- Chaudhary, P.; Fatima, F.; Kumar, A. Relevance of Nanomaterials in Food Packaging and its Advanced Future Prospects. J. Inorg. Organomet. Polym. Mater. 2020, 30, 5180–5192. [Google Scholar] [CrossRef]
- Shil, H.; Magaye, R.; Castranova, V.; Zhao, J. Titanium dioxide nanoparticles: A review of current toxicological data. Part. Fibre Toxicol. 2013, 10, 15. [Google Scholar] [CrossRef]
- Khan, R.H.; Ahmad, E.; Zaman, M.; Qadeer, A.; Rabbani, G. Nanoparticles in relation to peptide and protein aggregation. Int. J. Nanomed. 2014, 9, 899–912. [Google Scholar] [CrossRef]
- Snopczyński, T.; Góralczyk, K.; Czajka, K.; Struciński, P.; Hernik, A.; Korcz, W.; Ludwicki, J.K. Nanotechnologia- możliwości i zagrożenia. Rocz. PZK 2009, 60, 101–111. (In Polish) [Google Scholar]
- Schaumann, G.E.; Philippe, A.; Bundschuh, M.; Metreveli, G.; Klitzke, S.; Rakcheev, D.; Grün, A.; Kumahor, S.K.; Kühn, M.; Baumann, T.; et al. Understanding the fate and biological effects of Ag- and TiO2-nanoparticles in the environment: The quest for advanced analytics and interdisciplinary concepts. Sci. Total Environ. 2015, 535, 3–19. [Google Scholar] [CrossRef]
- Smijs, T.G.; Pavel, S. Titanium dioxide and zinc oxide nanoparticles in sunscreens: Focus on their safety and effectiveness. Nanotechnol. Sci. Appl. 2011, 4, 95–112. [Google Scholar] [CrossRef]
- Bilal, M.; Iqbal, H.M.N. New Insights on Unique Features and Role of Nanostructured Materials in Cosmetics. Cosmetics 2020, 7, 24. [Google Scholar] [CrossRef]
- Bilal, M.; Mehmood, S.; Iqbal, H.M.N. The Beast of Beauty: Environmental and Health Concerns of Toxic Components in Cosmetics. Cosmetics 2020, 7, 13. [Google Scholar] [CrossRef]
- Effiong, D.; Uwah, T.; Jumbo, E.; Akpabio, A. Nanotechnology in Cosmetics: Basics, Current Trends and Safety Concerns—A Review. Adv. Nanopart. 2020, 9, 1–22. [Google Scholar] [CrossRef]
- Pavel, S. Light therapy (with UVA-1) for SLE patients: Is it a good or bad idea? Rheumatology 2006, 45, 653–655. [Google Scholar] [CrossRef] [PubMed]
- Dransfield, G. Inorganic Sunscreens. Radiat. Prot. Dosim. 2000, 91, 271–273. [Google Scholar] [CrossRef]
- Antoniou, C.; Kosmadaki, M.; Stratigos, A.; Katsambas, A. Sunscreens—What’s important to know. J. Eur. Acad. Dermatol. Venereol. 2008, 22, 1110–1119. [Google Scholar] [CrossRef]
- Sharma, V.K. Aggregation and toxicity of titanium dioxide nanoparticles in aquatic environment—A Review. J. Environ. Sci. Health Part A 2009, 44, 1485–1495. [Google Scholar] [CrossRef]
- Zhao, J.; Lin, M.; Wang, Z.; Cao, X.; Xing, B. Engineered nanomaterials in the environment: Are they safe? Crit. Rev. Environ. Sci. Technol. 2020, 1–36. [Google Scholar] [CrossRef]
- Nanotechnology Products Database. 2021. Available online: https://product.statnano.com/industry/47/more (accessed on 14 May 2021).
- Raj, S.; Sumod, U.S.; Jose, S.; Sabitha, M. Nanotechnology in cosmetics: Opportunities and challenges. J. Pharm. Bioallied Sci. 2012, 4, 186–193. [Google Scholar] [CrossRef]
- Jackson, T.C.; Patani, B.O.; Ekpa, D.E. Nanotechnology in Diagnosis: A Review. Adv. Nanopart. 2017, 6, 93–102. [Google Scholar] [CrossRef]
- Beauty Industry Backs High Risk Small Particles: Controversial Nano-Ingredients Found in Big Name Brands. 2021. Available online: http://emergingtech.foe.org.au/wp-content/uploads/2014/08/Background-briefing-nanoparticles-in-cosmetics-November-2009.pdf (accessed on 14 May 2021).
- Jakubczyk, E. Nanotechnologia w technologii żywności. Przemysł Spożywczy 2007, 4, 15–22. (In Polish) [Google Scholar]
- Mrowiec, B. Nanomateriały—nowe zagrożenie środowiska. Ecol. Eng. 2017, 18, 105–110. (In Polish) [Google Scholar]
- Patil, M.P.; Kim, G.-D. Eco-friendly approach for nanoparticles synthesis and mechanism behind antibacterial activity of silver and anticancer activity of gold nanoparticles. Appl. Microbiol. Biotechnol. 2017, 101, 79–92. [Google Scholar] [CrossRef]
- Bystrzejewska-Piotrowska, G.; Golimowski, J.; Urban, P.L. Nanoparticles: Their potential toxicity, waste and environmental management. Waste Manag. 2009, 29, 2587–2595. [Google Scholar] [CrossRef] [PubMed]
- Bakand, S.; Hayes, A. Finance Dechsakulthorn Nanoparticles: A review of particle toxicology following inhalation exposure. Inhal. Toxicol. 2012, 24, 125–135. [Google Scholar] [CrossRef]
- Part, F.; Zecha, G.; Causon, T.; Sinner, E.-K.; Huber-Humer, M. Current limitations and challenges in nanowaste detection, characterisation and monitoring. Waste Manag. 2015, 43, 407–420. [Google Scholar] [CrossRef] [PubMed]
- Holder, A.L.; Vejerano, E.P.; Zhou, X.; Marr, L.C. Nanomaterial disposal by incineration. Environ. Sci. Process. Impacts 2013, 15, 1652–1664. [Google Scholar] [CrossRef]
- Elsaesser, A.; Howard, C.V. Toxicology of nanoparticles. Adv. Drug Deliv. Rev. 2012, 64, 129–137. [Google Scholar] [CrossRef] [PubMed]
- Lowry, G.V.; Gregory, K.B.; Apte, S.C.; Lead, J.R. Transformations of Nanomaterials in the Environment. Environ. Sci. Technol. 2012, 46, 6893–6899. [Google Scholar] [CrossRef]
- Mustafa, F.; Andreescu, S. Nanotechnology-based approaches for food sensing and packaging applications. RSC Adv. 2020, 10, 19309–19336. [Google Scholar] [CrossRef]
- Oberdörster, G. Toxicokinetics and effects of fibrous and nonfibrous particles. Inhal. Toxicol. 2002, 14, 29–56. [Google Scholar] [CrossRef]
- Crisponi, G.; Nurchi, V.M.; Lachowicz, J.I.; Peana, M.; Medici, S.; Zoroddu, M.A. Toxicity of Nanoparticles: Etiology and Mechanisms. Antimicrob. Nanoarchitecton. 2017, 511–546. [Google Scholar] [CrossRef]
- Lewinski, N.; Colvin, V.; Drezek, R. Cytotoxicity of nanoparticles. Small 2008, 4, 26–49. [Google Scholar] [CrossRef]
- Yokel, R.A.; MacPhail, R.C. Engineered nanomaterials: Exposures, hazards, and risk prevention. J. Occup. Med. Toxicol. 2011, 6, 7. [Google Scholar] [CrossRef]
- Nile, S.H.; Baskar, V.; Selvaraj, D.; Nile, A.; Xiao, J.; Kai, G. Nanotechnologies in Food Science: Applications, Recent Trends, and Future Perspectives. Nano-Micro Lett. 2020, 12, 1–34. [Google Scholar] [CrossRef]
- Kirchner, C.; Liedl, T.; Kudera, S.; Pellegrino, T.; Javier, A.M.; Gaub, H.E.; Stölzle, S.; Fertig, A.N.; Parak, W.J. Cytotoxicity of Colloidal CdSe and CdSe/ZnS Nanoparticles. Nano Lett. 2005, 5, 331–338. [Google Scholar] [CrossRef]
- Zoroddu, M.A.; Peana, M.; Medici, S.; Potocki, S.; Kozlowski, H. Ni(ii) binding to the 429–460 peptide fragment from human Toll like receptor (hTLR4): A crucial role for nickel-induced contact allergy? Dalton Trans. 2014, 43, 2764–2771. [Google Scholar] [CrossRef] [PubMed]
- Peters, A.; Veronesi, B.; Calderón-Garcidueñas, L.; Gehr, P.; Chen, L.C.; Geiser, M.; Reed, W.; Rothen-Rutishauser, B.; Schürch, S.; Schulz, H. Translocation and potential neurological effects of fine and ultrafine particles a critical update. Part. Fibre Toxicol. 2006, 3, 13. [Google Scholar] [CrossRef] [PubMed]
- Kao, Y.-Y.; Cheng, T.-J.; Yang, D.-M.; Wang, C.-T.; Chiung, Y.-M.; Liu, P.-S. Demonstration of an Olfactory Bulb–Brain Translocation Pathway for ZnO Nanoparticles in Rodent Cells In Vitro and In Vivo. J. Mol. Neurosci. 2012, 48, 464–471. [Google Scholar] [CrossRef] [PubMed]
- Moller, P.; Folkmann, J.K.; Danielsen, P.H.; Jantzen, K.; Loft, S. Oxidative stress generated damage to DNA by gastrointestinal exposure to insoluble particles. Curr. Mol. Med. 2012, 12, 732–745. [Google Scholar] [CrossRef]
- Tang, Y.; Wang, F.; Jin, C.; Liang, H.; Zhong, X.; Yang, Y. Mitochondrial injury induced by nanosized titanium dioxide in A549 cells and rats. Environ. Toxicol. Pharmacol. 2013, 36, 66–72. [Google Scholar] [CrossRef]
- Dunford, R.; Salinaro, A.; Cai, L.; Serpone, N.; Horikoshi, S.; Hidaka, H.; Knowland, J. Chemical oxidation and DNA damage catalysed by inorganic sunscreen ingredients. FEBS Lett. 1997, 418, 87–90. [Google Scholar] [CrossRef]
- Lemos, C.N.; Pereira, F.; Dalmolin, L.F.; Cubayachi, C.; Ramos, D.N.; Lopez, R.F. Nanoparticles influence in skin penetration of drugs. In Nanostructures for the Engineering of Cells, Tissues and Organs; Elsevier: Amsterdam, The Netherlands, 2018; pp. 187–248. [Google Scholar]
- Horikoshi, S.; Serpone, N. Chapter 1- Introduction to Nanoparticles, Microwaves in Nanoparticle Synthesis, Fundamentals and Applications; Wiley: Hoboken, NJ, USA, 2011. [Google Scholar]
- Soto, K.; Garza, K.; Murr, L. Cytotoxic effects of aggregated nanomaterials. Acta Biomater. 2007, 3, 351–358. [Google Scholar] [CrossRef]
- Nadaroglu, H.; Gungur, A.A.; Ince, S. Synthesis of Nanoparticles by Green Synthesis Method. Int. J. Innov. Res. Rev. 2017, 1, 6–9. [Google Scholar]
- Manufactured Nanoparticles in the Workplace. Available online: www.osha.europa.eu (accessed on 10 May 2021).
- ISO/TR 12885: 2018 Nanotechnologies—Health and Safety Practices in Occupational Settings. ASTM E2535—07(2018) Standard Guide for Handling Unbound Engineered Nanoscale Particles in Occupational Settings. Available online: https://www.iso.org05.2019 (accessed on 11 May 2021).
- ISO-ISO/TC 229. Nanotechnologies; BSI: London, UK, 2021. [Google Scholar]
- Buffle, J. The Key Role of Environmental Colloids/Nanoparticles for the Sustainability of Life. Environ. Chem. 2006, 3, 155–158. [Google Scholar] [CrossRef]
- Rezolucja Parlamentu Europejskiego. Aspekty regulacyjne nanomateriałów. Dz. Urz. UE C 2010, 184, 82. (In Polish) [Google Scholar]
- Jurewicz, M. Nanotechnologia a Ochrona Środowiska—Perspektywa Prawna, Ruch Prawniczy, Ekonomiczny i Socjologiczny; Wydzial Prawa i Administracji UAM: Poznan, Polska, 2018. (In Polish) [Google Scholar]
- Komunikat Komisji do Parlamentu Europejskiego, Rady i Europejskiego Komitetu Ekonomiczno-Społecznego. Drugi Przegląd Regulacyjny Poświęcony Nanomateriałom; Komisja Europejska: Bruksela, Belgia, 2012. (In Polish) [Google Scholar]
- Baran, A. Prawne aspekty nanotechnologii w kontekście ochrony środowiska. Ekon. Sr. 2015, 1, 28–40. (In Polish) [Google Scholar]
- Danish Ministry of the Environment. Guideline for the Danish Inventory of Nanoproducts; Danish Ministry of the Environment: Copenhagen, Denmark, 2014.
- The European Union Observatory for Nanomaterials. A Step Forward? National Institute for Public Health and the Evironment; Ministry of Health Welfare and Sport: Bilthoven, The Netherlands, 2017.
- Standards for Nanotechnology. Available online: www.nano.gov (accessed on 10 May 2021).
- Government of Canada. New Substances Program Advisory Note 2014-02, Assessment of Nanomaterials Under the New Substances Notification Regulations (Chemicals and Polymers); Environment Canada: Ottawa, ON, Canada, 2014.
- Parlament Europejski. Decyzja Komisji 2011/381/UE Ustanawiająca Kryteria Ekologiczne Przyznawania Oznakowania Ekologicznego UE; Parlament Europejski: Bruksela, Belgia, 2011.
- U.S. Food and Drug Administration. Nanotechnology—A Report of the U.S. Food and Drug Administration Nanotechnology Task Force; U.S. Food and Drug Administration: College Park, MD, USA, 2007.
- U.S. Food and Drug Administration. Guidance for Industry Safety of Nanomaterials in Cosmetic Products; U.S. Department of Health and Human Services Food and Drug Administration, Center for Food Safety and Applied Nutrition; U.S. Food and Drug Administration: College Park, MD, USA, 2014.
- U.S. Food and Drug Administration. Guidance for Industry Use of Nanomaterials in Food for Animals; U.S. Department of Health and Human Services Food and Drug Administration Center for Veterinary Medicine: College Park, MD, USA, 2015.
- U.S. Food and Drug Administration. Nanotechnology-Over a Decade of Progress and Innovation, A Report by the U.S. Food and Drug Administration; U.S. Food and Drug Administration: College Park, MD, USA, 2020.
- Kaul, S.; Gulati, N.; Verma, D.; Mukherjee, S.; Nagaich, U. Role of Nanotechnology in Cosmeceuticals: A Review of Recent Advances. J. Pharm. 2018, 2018, 3420204. [Google Scholar] [CrossRef]
- Dhull, K.; Swagat, T.; Harish, D. Cosmetics: Regulatory Scenario in USA, EU and India. J. Pharm. Technol. Res. Manag. 2015, 3, 127–139. [Google Scholar]
- Włodarczyk, R.; Kwarciak-Kozłowska, A. Analiza możliwości sorpcji nanocząstek srebra pochodzenia antropogenicznego z wykorzystaniem biowęgla. Przemysł Chem. 2019, 1, 115–118. [Google Scholar] [CrossRef]
- Wlodarczyk, R. Nanomaterials in Low-Temperatures Fuel Cells—The Latest Reports. Mater. Sci. Appl. 2019, 10, 643–664. [Google Scholar] [CrossRef]
- Ram Prasad Editor. Plant Nanobionics. Approaches in Nanoparticles, Biosynthesis, and Toxicity, Nanotechnology in the Life Sciences; Springer: Berlin/Heidelberg, Germany, 2019; Volume 2, pp. 377–398. [Google Scholar]
- Bakare, A.A.; Udoakang, A.J.; Anifowoshe, A.T.; Fadoju, O.M.; Ogunsuyi, O.I.; Alabi, O.A.; Alimba, C.G.; Oyeyemi, I.T. Genotoxicity of Titanium Dioxide Nanoparticles using the Mouse Bone Marrow Micronucleus and Sperm Morphology Assays. J. Pollut. Eff. Control 2016, 4, 1–7. [Google Scholar] [CrossRef]
- Manzer, H.; Siddiqui, M.H.; Al-Whaibi, F.M. Nanotechnology and Plant Sciences, Nanoparticles and Their Impact on Plants; Springer: Cham, Switzerland, 2015; pp. 101–119. [Google Scholar]
- Fytianos, G.; Rahdar, A.; Kyzas, G.Z. Nanomaterials in Cosmetics: Recent Updates. Nanomaterials 2020, 10, 979. [Google Scholar] [CrossRef]
- Panariello, L.; Vannozzi, A.; Morganti, P.; Coltelli, M.B. Biobased and Eco-Comptatible Beauty Films Coated with Chitin Nanofibrils, Nanolignin and Vitamin E. Cosmetics 2021, 8, 27. [Google Scholar] [CrossRef]

| Nanomaterials | Type of Cosmetics | Manufacturer |
|---|---|---|
| Zinc oxide, aluminium oxide, iron oxide and titanium dioxide | Mineral Foundation | By Terry Max Factor The Body Shop |
| Foundation | Christian Dior L’Oreal Clarins | |
| Concealer | Clinique Lancôme Paris Revlon Yves Saint Laurent | |
| UV protection | ColoreScience Dermatone Procter& Gamble Boots | |
| Fullerenes and fullersomes | Night and eye cream | Dr. Brandt Sircuit cosmeceuticals Bellapelle skin studio |
| Nanoemulsions | moisture mist Calming nanoemulsion | Chanel La prairie |
| Nanocapsules | skin cream | Dr. Brandt Lancome Enprani |
| Novasomes | Linia Neutrogena Renutriv range, resilience range | Johnson& Johnson Estee lauder |
| Nano silicon dioxide | lift makeup | Lancome |
| Country | Type of Activities | New Activities |
|---|---|---|
| Denmark’s | Environmental Protection Agency decided that nanomaterials should be registered | Guideline for the Danish Inventory of Nanoproducts-2014 |
| France | In 2013, it introduced a decree on the content and conditions for submitting annual declarations covering substances in the form of nanoscale | Not fund |
| Belgium | A project is being developed, based on which reporting will be introduced in line with the quantitative limits of nanomaterials | Royal Decree amending the Royal Decree of May 27th 2014 concerning the placing on the market of substances produced in nanoparticle state-2017 |
| Canada | A review of the chemical law is underway to adapt it to the use of nanomaterials, the first standard for workplace nanotechnology has been developed based on ISO/TR 12885 [70] | New Substances Program Advisory Note-2014 |
| Netherlands | Introduced proposals for risk assessment and setting acceptable levels as part of the work of the National Institute of Public Health and the Environment (Rijksinstituut voor Volksgezondheid en Milieu RIVM) | The European Union Observatory for Nanomaterials–National Institute for Public Health and the Environment-2017 |
| USA | National Institute for Occupational Safety and Health—NIOSH | National Nanotechnology Coordination Office (NNCO) |
| Japan | The project of the Organization for the Development of New Energy and Industrial Technologies in Japan (NEDO) concerns the risk assessment of manufactured nano-objects: titanium dioxide, fullerene and carbon nanotubes | Not found |
| Toxicity | NRV Hazard Class | Reference Values | Type of Nanomaterial | Example |
|---|---|---|---|---|
| high | class 1 | 0.01 fibers cm−3 | rigid carbon nanofibers, metal oxide fibers | SWCNT (single-walled carbon nanotubes) or MWCNT (multiwalled carbon nanotubes), fullerenes |
| medium or low | class 2a | 20,000 particles cm−3 | granular nanomaterials (non-fibrous), stable in the environment, with a density greater than 6 g cm−3 | particles Ag, Au, CeO2, COO, Fe, FexOy, La, Pb, Sb2O5, or SnO2 |
| medium or low | class 2b | 40,000 particles cm−3 | granular nanomaterials and nanofibers, stable in the environment, with a density above 6 g cm−3 | particles Al2O3, SiO2, TiO2, ZnO, CaCO3, layered aluminosilicate, carbon black, C60, dendrimers, polystyrene or nanofibers |
| low | class 3 | OEL values * | granular nanomaterials, unstable or soluble in water (solubility above 100 mg L−1) | NaCl, lipid particles, flour, sucrose. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Włodarczyk, R.; Kwarciak-Kozłowska, A. Nanoparticles from the Cosmetics and Medical Industries in Legal and Environmental Aspects. Sustainability 2021, 13, 5805. https://doi.org/10.3390/su13115805
Włodarczyk R, Kwarciak-Kozłowska A. Nanoparticles from the Cosmetics and Medical Industries in Legal and Environmental Aspects. Sustainability. 2021; 13(11):5805. https://doi.org/10.3390/su13115805
Chicago/Turabian StyleWłodarczyk, Renata, and Anna Kwarciak-Kozłowska. 2021. "Nanoparticles from the Cosmetics and Medical Industries in Legal and Environmental Aspects" Sustainability 13, no. 11: 5805. https://doi.org/10.3390/su13115805
APA StyleWłodarczyk, R., & Kwarciak-Kozłowska, A. (2021). Nanoparticles from the Cosmetics and Medical Industries in Legal and Environmental Aspects. Sustainability, 13(11), 5805. https://doi.org/10.3390/su13115805