The Role of Biochar in Regulating the Carbon, Phosphorus, and Nitrogen Cycles Exemplified by Soil Systems
Abstract
1. Introduction
2. Adsorption of Carbon, Nitrogen, and Phosphorus Species
2.1. Adsorption of Carbon Species
2.1.1. Inorganic Carbons
2.1.2. Organic Carbons
2.2. Adsorption of Nitrogen Species
2.2.1. Inorganic Nitrogen
2.2.2. Organic Nitrogen
2.3. Adsorption of Phosphorus Species
2.3.1. Inorganic Phosphorus
2.3.2. Organic Phosphorus
3. Biochar Mediated Degradation
3.1. Organic Carbons
3.2. Organic Nitrogen Species
3.3. Organophosphorus Species
4. Biochar Mediated Transport of Carbon, Nitrogen, and Phosphorus in Soil Systems
5. Biochar Weathering in Soil Environment
6. Outlook
7. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Minasny, B.; Malone, B.P.; McBratney, A.B.; Angers, D.A.; Arrouays, D.; Chambers, A.; Chaplot, V.; Chen, Z.S.; Cheng, K.; Das, B.S.; et al. Soil carbon 4 per mille. Geoderma 2017, 292, 59–86. [Google Scholar] [CrossRef]
- Woolf, D.; Amonette, J.E.; Street-Perrott, F.A.; Lehmann, J.; Joseph, S. Sustainable biochar to mitigate global climate change. Nat. Commun. 2010, 1, 56. [Google Scholar] [CrossRef]
- You, S.; Ok, Y.S.; Chen, S.S.; Tsang, D.C.W.; Kwon, E.E.; Lee, J.; Wang, C.H. A critical review on sustainable biochar system through gasification: Energy and environmental applications. Bioresour. Technol. 2017, 246, 242–253. [Google Scholar] [CrossRef]
- Azeem, M.; Hayat, R.; Hussain, Q.; Ahmed, M.; Pan, G.; Ibrahim Tahir, M.; Imran, M.; Irfan, M. Biochar improves soil quality and N2-fixation and reduces net ecosystem CO2 exchange in a dry land legume-cereal cropping system. Soil Tillage Res. 2019, 186, 172–182. [Google Scholar] [CrossRef]
- Oldfield, T.L.; Sikirica, N.; Mondini, C.; Lopez, G.; Kuikman, P.J.; Holden, N.M. Biochar, compost and biochar-compost blend as options to recover nutrients and sequester carbon. J. Environ. Manag. 2018, 218, 465–476. [Google Scholar] [CrossRef]
- Bi, Y.; Cai, S.; Wang, Y.; Xia, Y.; Zhao, X.; Wang, S.; Xing, G. Assessing the viability of soil successive straw biochar amendment based on a five-year column trial with six different soils: Views from crop production, carbon sequestration and net ecosystem economic benefits. J. Environ. Manag. 2019, 245, 173–186. [Google Scholar] [CrossRef] [PubMed]
- Paustian, K.; Lehmann, J.; Ogle, S.; Reay, D.; Robertson, G.P.; Smith, P. Climate-smart soils. Nature 2016, 532, 49–57. [Google Scholar] [CrossRef]
- Harter, J.; Krause, H.M.; Schuettler, S.; Ruser, R.; Fromme, M.; Scholten, T.; Kappler, A.; Behrens, S. Linking N2O emissions from biochar-amended soil to the structure and function of the n-cycling microbial community. ISME J. 2014, 8, 660–674. [Google Scholar] [CrossRef]
- Kammann, C.I.; Schmidt, H.P.; Messerschmidt, N.; Linsel, S.; Steffens, D.; Müller, C.; Stephen, J. Plant growth improvement mediated by nitrate capture in co-composted biochar. Sci. Rep. 2015, 5, 11080. [Google Scholar] [CrossRef]
- Nguyen, T.T.N.; Xu, C.Y.; Tahmasbian, I.; Che, R.; Xu, Z.; Zhou, X.; Wallace, H.M.; Bai, S.H. Effects of biochar on soil available inorganic nitrogen: A review and meta-analysis. Geoderma 2017, 288, 79–96. [Google Scholar] [CrossRef]
- Viglašová, E.; Galamboš, M.; Danková, Z.; Krivosudský, L.; Lengauer, C.L.; Hood-Nowotny, R.; Soja, G.; Rompel, A.; Matík, M.; Briančin, J. Production, characterization and adsorption studies of bamboo-based biochar/montmorillonite composite for nitrate removal. Waste Manag. 2018, 79, 385–394. [Google Scholar] [CrossRef] [PubMed]
- Viglašová, E.; Galamboš, M.; Diviš, D.; Danková, Z.; Daňo, M.; Krivosudský, L.; Lengauer, C.L.; Matik, M.; Briančin, J.; Soja, G. Engineered biochar as a tool for nitrogen pollutants removal: Preparation, characterization and sorption study. Desalination Water Treat. 2020, 191, 318–331. [Google Scholar] [CrossRef]
- Ahmad, M.; Rajapaksha, A.U.; Lim, J.E.; Zhang, M.; Bolan, N.; Mohan, D.; Vithanage, M.; Lee, S.S.; Ok, Y.S. Biochar as a sorbent for contaminant management in soil and water: A review. Chemosphere 2014, 99, 19–33. [Google Scholar] [CrossRef]
- Bai, S.H.; Reverchon, F.; Xu, C.Y.; Xu, Z.; Blumfield, T.J.; Zhao, H.; Van Zwieten, L.; Wallace, H.M. Wood biochar increases nitrogen retention in field settings mainly through abiotic processes. Soil Biol. Biochem. 2015, 90, 232–240. [Google Scholar] [CrossRef]
- Borchard, N.; Schirrmann, M.; Cayuela, M.L.; Kammann, C.; Wrage-Mönnig, N.; Estavillo, J.M.; Fuertes-Mendizábal, T.; Sigua, G.; Spokas, K.; Ippolito, J.A.; et al. Biochar, soil and land-use interactions that reduce nitrate leaching and N2O emissions: A meta-analysis. Sci. Total Environ. 2019, 651, 2354–2364. [Google Scholar] [CrossRef]
- Cayuela, M.L.; van Zwieten, L.; Singh, B.P.; Jeffery, S.; Roig, A.; Sánchez-Monedero, M.A. Biochar’s role in mitigating soil nitrous oxide emissions: A review and meta-analysis. Agric. Ecosyst. Environ. 2014, 191, 5–16. [Google Scholar] [CrossRef]
- Clough, T.J.; Condron, L.M. Biochar and the nitrogen cycle. J. Environ. Qual. 2010, 39, 1218–1223. [Google Scholar] [CrossRef] [PubMed]
- Ducey, T.F.; Ippolito, J.A.; Cantrell, K.B.; Novak, J.M.; Lentz, R.D. Addition of activated switchgrass biochar to an aridic subsoil increases microbial nitrogen cycling gene abundances. Appl. Soil Ecol. 2013, 65, 65–72. [Google Scholar] [CrossRef]
- Xiao, X.; Chen, B.; Chen, Z.; Zhu, L.; Schnoor, J.L. Insight into Multiple and Multilevel Structures of Biochars and Their Potential Environmental Applications: A Critical Review. Environ. Sci. Technol. 2018, 52, 5027–5047. [Google Scholar] [CrossRef] [PubMed]
- Daňo, M.; Viglašová, E.; Galamboš, M.; Štamberg, K.; Kujan, J. Surface complexation models of pertechnetate on biochar/montmorillonite composite—Batch and dynamic sorption study. Materials 2020, 13, 3108. [Google Scholar] [CrossRef]
- Glaser, B.; Lehr, V.I. Biochar effects on phosphorus availability in agricultural soils: A meta-analysis. Sci. Rep. 2019, 9, 9338. [Google Scholar] [CrossRef]
- Zhao, X.; Li, D.; Kong, J.; Lin, Q. Does Biochar Addition Influence the Change Points of Soil Phosphorus Leaching? J. Integr. Agric. 2014, 13, 499–506. [Google Scholar] [CrossRef]
- Arif, M.; Ilyas, M.; Riaz, M.; Ali, K.; Shah, K.; Ul Haq, I.; Fahad, S. Biochar improves phosphorus use efficiency of organic-inorganic fertilizers, maize-wheat productivity and soil quality in a low fertility alkaline soil. Field Crops Res. 2017, 214, 25–37. [Google Scholar] [CrossRef]
- Biederman, L.A.; Phelps, J.; Ross, B.J.; Polzin, M.; Harpole, W.S. Biochar and manure alter few aspects of prairie development: A field test. Agric. Ecosyst. Environ. 2017, 236, 78–87. [Google Scholar] [CrossRef]
- Cayuela, M.L.; Sanchez-Monedero, M.A.; Roig, A.; Hanley, K.; Enders, A.; Lehmann, J. Biochar and denitrification in soils: When, how much and why does biochar reduce N2O emissions? Sci. Rep. 2013, 3, 1732. [Google Scholar] [CrossRef] [PubMed]
- Cayuela, M.L.; Jeffery, S.; van Zwieten, L. The molar H:Corg ratio of biochar is a key factor in mitigating N2O emissions from soil. Agric. Ecosyst. Environ. 2015, 202, 135–138. [Google Scholar] [CrossRef]
- Chen, B.; Zhou, D.; Zhu, L. Transitional adsorption and partition on nonpolar and polar aromatic contaminants by biochars of pine needles with different pyrolytic temperatures. Environ. Sci. Technol. 2008, 42, 5137–5143. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Guo, H.; Shen, F.; Yang, G.; Zhang, Y.; Zeng, Y.; Wang, L.; Xiao, H.; Deng, S. Biochar produced from oak sawdust by Lanthanum (La)-involved pyrolysis for adsorption of ammonium (NH4+), nitrate (NO3−), and phosphate (PO43-). Chemosphere 2015, 119, 646–653. [Google Scholar] [CrossRef]
- Yang, J.; Li, H.; Zhang, D.; Wu, M.; Pan, B. Limited role of biochars in nitrogen fixation through nitrate adsorption. Sci. Total Environ. 2017, 592, 758–765. [Google Scholar] [CrossRef]
- Dong, C.D.; Chen, C.W.; Hung, C.M. Synthesis of magnetic biochar from bamboo biomass to activate persulfate for the removal of polycyclic aromatic hydrocarbons in marine sediments. Bioresour. Technol. 2017, 245, 188–195. [Google Scholar] [CrossRef] [PubMed]
- Dong, C.D.; Chen, C.W.; Kao, C.M.; Chien, C.C.; Hung, C.M. Wood-biochar-supported magnetite nanoparticles for remediation of PAH-contaminated estuary sediment. Catalysts 2018, 8, 73. [Google Scholar] [CrossRef]
- Dong, C.D.; Chen, C.W.; Tsai, M.L.; Chang, J.H.; Lyu, S.Y.; Hung, C.M. Degradation of 4-nonylphenol in marine sediments by persulfate over magnetically modified biochars. Bioresour. Technol. 2019, 281, 143–148. [Google Scholar] [CrossRef] [PubMed]
- Dong, C.D.; Chen, C.W.; Nguyen, T.B.; Huang, C.P.; Hung, C.M. Degradation of phthalate esters in marine sediments by persulfate over Fe–Ce/biochar composites. Chem. Eng. J. 2020, 384, 123301. [Google Scholar] [CrossRef]
- Hung, C.M.; Huang, C.P.; Hsieh, S.L.; Tsai, M.L.; Chen, C.W.; Dong, C.D. Biochar derived from red algae for efficient remediation of 4-nonylphenol from marine sediments. Chemosphere 2020, 254, 126919. [Google Scholar] [CrossRef] [PubMed]
- Hung, C.M.; Huang, C.P.; Chen, C.W.; Wu, C.H.; Lin, Y.L.; Dong, C.D. Activation of percarbonate by water treatment sludge–derived biochar for the remediation of PAH-contaminated sediments. Environ. Pollut. 2020, 265, 114914. [Google Scholar] [CrossRef]
- Hung, C.M.; Huang, C.P.; Chen, C.W.; Dong, C.D. The degradation of di-(2-ethylhexyl) phthalate, DEHP, in sediments using percarbonate activated by seaweed biochars and its effects on the benthic microbial community. J. Clean. Prod. 2021, 292, 126108. [Google Scholar] [CrossRef]
- Hung, C.M.; Huang, C.P.; Cheng, C.W.; Chen, C.W.; Dong, C.D. Production and characterization of a high value-added seaweed-derived biochar: Optimization of pyrolysis conditions and evaluation for sediment treatment. J. Anal. Appl. Pyrolysis 2021, 155, 105071. [Google Scholar] [CrossRef]
- Haider, G.; Steffens, D.; Müller, C.; Kammann, C.I. Standard extraction methods may underestimate nitrate stocks captured by field-aged biochar. J. Environ. Qual. 2016, 45, 1196–1204. [Google Scholar] [CrossRef]
- Pillai, M.G.; Simha, P.; Gugalia, A. Recovering urea from human urine by bio-sorption onto microwave activated carbonized coconut shells: Equilibrium, kinetics, optimization and field studies. J. Environ. Chem. Eng. 2014, 2, 46–55. [Google Scholar] [CrossRef]
- Sanford, J.R.; Larson, R.A.; Runge, T. Nitrate sorption to biochar following chemical oxidation. Sci. Total Environ. 2019, 669, 938–947. [Google Scholar] [CrossRef]
- Ventura, M.; Sorrenti, G.; Panzacchi, P.; George, E.; Tonon, G. Biochar reduces short-term Nitrate leaching from a horizon in an apple orchard. J. Environ. Qual. 2013, 42, 76–82. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Lin, K.; Hou, Z.; Richardson, B.; Gan, J. Sorption of the herbicide terbuthylazine in two New Zealand forest soils amended with biosolids and biochars. J. Soils Sediments 2009, 10, 283–289. [Google Scholar] [CrossRef]
- Yang, X.Y.; Chang, K.H.; Kim, Y.J.; Zhang, J.; Yoo, G. Effects of different biochar amendments on carbon loss and leachate characterization from an agricultural soil. Chemosphere 2019, 226, 625–635. [Google Scholar] [CrossRef] [PubMed]
- Yao, Y.; Gao, B.; Zhang, M.; Inyang, M.; Zimmerman, A.R. Effect of biochar amendment on sorption and leaching of nitrate, ammonium, and phosphate in a sandy soil. Chemosphere 2012, 89, 1467–1471. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.; Zhang, Q.; Sun, K.; Liu, X.; Zheng, W.; Zhao, Y. Sorption of simazine to corn straw biochars prepared at different pyrolytic temperatures. Environ. Pollut. 2011, 159, 2594–2601. [Google Scholar] [CrossRef]
- Dong, C.D.; Chen, C.W.; Hung, C.M. Persulfate activation with rice-husk-based magnetic biochar for degrading PAEs in marine sediments. Environ. Sci. Pollut. Res. 2019, 26, 33781–33790. [Google Scholar] [CrossRef]
- Hung, C.M.; Huang, C.P.; Chen, C.W.; Hsieh, S.L.; Dong, C.D. Effects of biochar on catalysis treatment of 4-nonylphenol in estuarine sediment and associated microbial community structure. Environ. Pollut. 2021, 268, 115673. [Google Scholar] [CrossRef]
- Mohan, D.; Sarswat, A.; Ok, Y.S.; Pittman, C.U., Jr. Organic and inorganic contaminants removal from water with biochar, a renewable, low cost and sustainable adsorbent—A critical review. Bioresour. Technol. 2014, 160, 191–202. [Google Scholar] [CrossRef]
- Solanki, A.; Boyer, T. Pharmaceutical removal in synthetic human urine using biochar. Environ. Sci. Water Res. Technol. 2017, 3, 553–565. [Google Scholar] [CrossRef]
- Jin, Y.; Liang, X.; He, M.; Liu, Y.; Tian, G.; Shi, J. Manure biochar influence upon soil properties, phosphorus distribution and phosphatase activities: A microcosm incubation study. Chemosphere 2016, 142, 128–135. [Google Scholar] [CrossRef]
- Pratiwi, E.P.A.; Hillary, A.K.; Fukuda, T.; Shinogi, Y. The effects of rice husk char on ammonium, nitrate and phosphate retention and leaching in loamy soil. Geoderma 2016, 277, 61–68. [Google Scholar] [CrossRef]
- Bruun, E.W.; Ambus, P.; Egsgaard, H.; Hauggaard-Nielsen, H. Effects of slow and fast pyrolysis biochar on soil C and N turnover dynamics. Soil Biol. Biochem. 2012, 46, 73–79. [Google Scholar] [CrossRef]
- Bargmann, I.; Martens, R.; Rillig, M.C.; Kruse, A.; Kücke, M. Hydrochar amendment promotes microbial immobilization of mineral N. J. Plant. Nutr. Soil Sci. 2014, 177, 59–67. [Google Scholar] [CrossRef]
- Mao, J.D.; Johnson, R.L.; Lehmann, J.; Olk, D.C.; Neves, E.G.; Thompson, M.L.; Schmidt-Rohr, K. Abundant and stable char residues in soils: Implications for soil fertility and carbon sequestration. Environ. Sci. Technol. 2012, 46, 9571–9576. [Google Scholar] [CrossRef] [PubMed]
- Conte, P.; Marsala, V.; Pasquale, C.D.; Bubici, S.; Valagussa, M.; Pozzi, A.; Alonzo, G. Nature of water-biochar interface interactions. Glob. Chang. Biol. Bioenergy 2013, 5, 116–121. [Google Scholar] [CrossRef]
- Qian, L.; Chen, B. Interactions of aluminum with biochars and oxidized biochars: Implications for the biochar aging process. J. Agric. Food Chem. 2014, 62, 373–380. [Google Scholar] [CrossRef]
- Uchimiya, M.; Bannon, D.I.; Wartelle, L.H. Retention of heavy metals by carboxyl functional groups of biochars in small arms range soil. J. Agric. Food Chem. 2012, 60, 1798–1809. [Google Scholar] [CrossRef]
- Xu, R.; Zhao, A.; Yuan, J.; Jiang, J. pH buffering capacity of acid soils from tropical and subtropical regions of China as influenced by incorporation of crop straw biochars. J. Soils Sediments 2012, 12, 494–502. [Google Scholar] [CrossRef]
- Clough, T.J.; Bertram, J.E.; Ray, J.L.; Condron, L.M.; O’Callaghan, M.; Sherlock, R.R.; Wells, N.S. Unweathered wood biochar impact on nitrous oxide emissions from a bovine-urine-amended pasture soil. Soil Sci. Soc. Am. J. 2010, 74, 852–860. [Google Scholar] [CrossRef]
- Clough, T.; Condron, L.; Kammann, C.; Müller, C. A review of biochar and soil nitrogen dynamics. Agronomy 2013, 3, 275–293. [Google Scholar] [CrossRef]
- Joseph, S.; Kammann, C.I.; Shepherd, J.G.; Conte, P.; Schmidt, H.; Hagemann, N.; Graber, E.R. Microstructural and associated chemical changes during the composting of a high temperature biochar: Mechanisms for nitrate, phosphate and other nutrient retention and release. Sci. Total Environ. 2018, 618, 1210–1223. [Google Scholar] [CrossRef]
- Fang, Q.; Chen, B.; Lin, Y.; Guan, Y. Aromatic and hydrophobic surfaces of wood-derived biochar enhance perchlorate adsorption via hydrogen bonding to oxygen-containing organic groups. Environ. Sci. Technol. 2014, 48, 279–288. [Google Scholar] [CrossRef] [PubMed]
- El-Naggar, A.H.; Usman, A.R.; Al-Omran, A.; Ok, Y.S.; Ahmad, M.; Al-Wabel, M.I. Carbon mineralization and nutrient availability in calcareous sandy soils amended with woody waste biochar. Chemosphere 2015, 138, 67–73. [Google Scholar] [CrossRef] [PubMed]
- Luo, X.; Wang, L.; Liu, G.; Wang, X.; Wang, Z.; Zheng, H. Effects of biochar on carbon mineralization of coastal wetland soils in the Yellow River Delta, China. Ecol. Eng. 2016, 94, 329–336. [Google Scholar] [CrossRef]
- El-Naggar, A.; El-Naggar, A.H.; Shaheen, S.M.; Sarkar, B.; Chang, S.X.; Tsang, D.C.W.; Rinklebe, J.; Ok, Y.S. Biochar composition-dependent impacts on soil nutrient release, carbon mineralization, and potential environmental risk: A review. J. Environ. Manag. 2019, 241, 458–467. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Wang, X.; Zhang, R. Decomposition temperature sensitivity of biochars with different stabilities affected by organic carbon fractions and soil microbes. Soil Tillage Res. 2019, 186, 322–332. [Google Scholar] [CrossRef]
- Jung, S.; Park, Y.K.; Kwon, E.E. Strategic use of biochar for CO2 capture and sequestration. J. Co2 Util. 2019, 32, 128–139. [Google Scholar] [CrossRef]
- Rittl, T.F.; Butterbach-Bahl, K.; Basile, C.M.; Pereira, L.A.; Alms, V.; Dannenmann, M.; Couto, E.G.; Cerri, C.E.P. Greenhouse gas emissions from soil amended with agricultural residue biochars: Effects of feedstock type, production temperature and soil moisture. Biomass Bioenergy 2018, 117, 1–9. [Google Scholar] [CrossRef]
- Wu, M.; Han, X.; Zhong, T.; Yuan, M.; Wu, W. Soil organic carbon content affects the stability of biochar in paddy soil. Agric. Ecosyst. Environ. 2016, 223, 59–66. [Google Scholar] [CrossRef]
- Doetterl, S.; Stevens, A.; Six, J.; Merckx, R.; Van Oost, K.; Casanova Pinto, M.; Casanova-Katny, A.; Muñoz, C.; Boudin, M.; Zagal Venegas, E.; et al. Soil carbon storage controlled by interactions between geochemistry and climate. Nat. Geosci. 2015, 8, 780. [Google Scholar] [CrossRef]
- Doetterl, S.; Berhe, A.A.; Nadeu, E.; Wang, Z.; Sommer, M.; Fiener, P. Erosion, deposition and soil carbon: A review of process-level controls, experimental tools and models to address C cycling in dynamic landscapes. Earth Sci. Rev. 2016, 154, 102–122. [Google Scholar] [CrossRef]
- El-Naggar, A.; Lee, S.S.; Awad, Y.M.; Yang, X.; Ryu, C.; Rizwan, M.; Rinklebe, J.; Tsang, D.C.W.; Ok, Y.S. Influence of soil properties and feedstocks on biochar potential for carbon mineralization and improvement of infertile soils. Geoderma 2018, 332, 100–108. [Google Scholar] [CrossRef]
- Yuan, H.Z.; Zhu, Z.K.; Wei, X.M.; Liu, S.I.; Peng, P.Q.; Gunina, A.; Shen, J.I.; Kuzyakov, Y.; Ge, T.D.; Wu, J.S.; et al. Straw and biochar strongly affect functional diversity of microbial metabolism in paddy soils. J. Integr. Agric. 2019, 18, 1474–1485. [Google Scholar] [CrossRef]
- Dong, X.; Hao, Q.; Li, G.; Lin, Q.; Zhao, X. Contrast effect of long-term fertilization on SOC and SIC stocks and distribution in different soil particle-size fractions. J. Soils Sediments 2016, 17, 1054–1063. [Google Scholar] [CrossRef]
- Namoi, N.; Pelster, D.; Rosenstock, T.S.; Mwangi, L.; Kamau, S.; Mutuo, P.; Barrios, E. Earthworms regulate ability of biochar to mitigate CO2 and N2O emissions from a tropical soil. Appl. Soil Ecol. 2019, 140, 57–67. [Google Scholar] [CrossRef]
- Wiesmeier, M.; Urbanski, L.; Hobley, E.; Lang, B.; von Lützow, M.; Marin-Spiotta, E.; van Wesemael, B.; Rabot, E.; Ließ, M.; Garcia-Franco, N.; et al. Soil organic carbon storage as a key function of soils—A review of drivers and indicators at various scales. Geoderma 2019, 333, 149–162. [Google Scholar] [CrossRef]
- Peng, X.; Tong, X.; Hao, L.; Wu, F. Applicability of biochar for limiting interrill erosion and organic carbon export of sloping cropland in a semi-arid area of China. Agric. Ecosyst. Environ. 2019, 280, 68–76. [Google Scholar] [CrossRef]
- Gascó, G.; Paz-Ferreiro, J.; Cely, P.; Plaza, C.; Méndez, A. Influence of pig manure and its biochar on soil CO2 emissions and soil enzymes. Ecol. Eng. 2016, 95, 19–24. [Google Scholar] [CrossRef]
- Pandit, N.R.; Mulder, J.; Hale, S.E.; Zimmerman, A.R.; Pandit, B.H.; Cornelissen, G. Multi-year double cropping biochar field trials in Nepal: Finding the optimal biochar dose through agronomic trials and cost-benefit analysis. Sci. Total Environ. 2018, 637, 1333–1341. [Google Scholar] [CrossRef]
- Sandhu, S.S.; Ussiri, D.A.N.; Kumar, S.; Chintala, R.; Papiernik, S.K.; Malo, D.D.; Schumacher, T.E. Analyzing the impacts of three types of biochar on soil carbon fractions and physiochemical properties in a corn-soybean rotation. Chemosphere 2017, 184, 473–481. [Google Scholar] [CrossRef]
- Butnan, S.; Deenik, J.L.; Toomsan, B.; Vityakon, P. Biochar properties affecting carbon stability in soils contrasting in texture and mineralogy. Agric. Nat. Resour. 2017, 51, 492–498. [Google Scholar] [CrossRef]
- Hansen, V.; Müller-Stöver, D.; Munkholm, L.J.; Peltre, C.; Hauggaard-Nielsen, H.; Jensen, L.S. The effect of straw and wood gasification biochar on carbon sequestration, selected soil fertility indicators and functional groups in soil: An incubation study. Geoderma 2016, 269, 99–107. [Google Scholar] [CrossRef]
- Wang, C.; Liu, J.; Shen, J.; Chen, D.; Li, Y.; Jiang, B.; Wu, J. Effects of biochar amendment on net greenhouse gas emissions and soil fertility in a double rice cropping system: A 4-year field experiment. Agric. Ecosyst. Environ. 2018, 262, 83–96. [Google Scholar] [CrossRef]
- Zhang, X.; Chen, C.; Chen, X.; Tao, P.; Jin, Z.; Han, Z. Persistent effects of biochar on soil organic carbon mineralization and resistant carbon pool in upland red soil, China. Environ. Earth Sci. 2018, 77, 177. [Google Scholar] [CrossRef]
- Dong, X.; Singh, B.P.; Li, G.; Lin, Q.; Zhao, X. Biochar application constrained native soil organic carbon accumulation from wheat residue inputs in a long-term wheat-maize cropping system. Agric. Ecosyst. Environ. 2018, 252, 200–207. [Google Scholar] [CrossRef]
- Huang, R.; Tian, D.; Liu, J.; Lv, S.; He, X.; Gao, M. Responses of soil carbon pool and soil aggregates associated organic carbon to straw and straw-derived biochar addition in a dryland cropping mesocosm system. Agric. Ecosyst. Environ. 2018, 265, 576–586. [Google Scholar] [CrossRef]
- Guan, S.; Liu, S.J.; Liu, R.Y.; Zhang, J.J.; Ren, J.; Cai, H.G.; Lin, X.X. Soil organic carbon associated with aggregate-size and density fractions in a Mollisol amended with charred and uncharred maize straw. J. Integr. Agric. 2019, 18, 1496–1507. [Google Scholar] [CrossRef]
- Zhao, X.; Wang, J.; Wang, S.; Xing, G. Successive straw biochar application as a strategy to sequester carbon and improve fertility: A pot experiment with two rice/wheat rotations in paddy soil. Plant. Soil 2014, 378, 279–294. [Google Scholar] [CrossRef]
- Munda, S.; Bhaduri, D.; Mohanty, S.; Chatterjee, D.; Tripathi, R.; Shahid, M.; Kumar, U.; Bhattacharyya, P.; Kumar, A.; Adak, T.; et al. Dynamics of soil organic carbon mineralization and C fractions in paddy soil on application of rice husk biochar. Biomass Bioenergy 2018, 115, 1–9. [Google Scholar] [CrossRef]
- Ghorbani, M.; Asadi, H.; Abrishamkesh, S. Effects of rice husk biochar on selected soil properties and nitrate leaching in loamy sand and clay soil. Int. Soil Water Conserv. Res. 2019, 7, 258–265. [Google Scholar] [CrossRef]
- Ghorbani, M.; Amirahmadi, E. Effect of rice husk Biochar (RHB) on some of chemical properties of an acidic soil and the absorption of some nutrients. J. Appl. Sci. Environ. Manag. 2018, 22, 313–317. [Google Scholar] [CrossRef]
- Dong, X.; Singh, B.P.; Li, G.; Lin, Q.; Zhao, X. Biochar increased field soil inorganic carbon content five years after application. Soil Tillage Res. 2019, 186, 36–41. [Google Scholar] [CrossRef]
- Jiang, L.I.; Han, G.M.; Lan, Y.; Liu, S.N.; Gao, J.P.; Yang, X.; Meng, J.; Chen, W.F. Corn cob biochar increases soil culturable bacterial abundance without enhancing their capacities in utilizing carbon sources in Biolog Eco-plates. J. Integr. Agric. 2017, 16, 713–724. [Google Scholar] [CrossRef]
- Pei, J.; Zhuang, S.; Cui, J.; Li, J.; Li, B.; Wu, J.; Fang, C. Biochar decreased the temperature sensitivity of soil carbon decomposition in a paddy field. Agric. Ecosyst. Environ. 2017, 249, 156–164. [Google Scholar] [CrossRef]
- Li, Y.; Li, Y.; Chang, S.X.; Yang, Y.; Fu, S.; Jiang, P.; Luo, Y.; Yang, M.; Chen, Z.; Hu, S.; et al. Biochar reduces soil heterotrophic respiration in a subtropical plantation through increasing soil organic carbon recalcitrancy and decreasing carbon-degrading microbial activity. Soil Boil. Biochem. 2018, 122, 173–185. [Google Scholar] [CrossRef]
- Jiang, X.; Tan, X.; Cheng, J.; Haddix, M.L.; Cotrufo, M.F. Interactions between aged biochar, fresh low molecular weight carbon and soil organic carbon after 3.5 years soil-biochar incubations. Geoderma 2019, 333, 99–107. [Google Scholar] [CrossRef]
- Brassard, P.; Godbout, S.; Palacios, J.H.; Jeanne, T.; Hogue, R.; Dubé, P.; Limousy, L.; Raghavan, V. Effect of six engineered biochars on GHG emissions from two agricultural soils: A short-term incubation study. Geoderma 2018, 327, 73–84. [Google Scholar] [CrossRef]
- Glaser, B.; Lehmann, J.; Zech, W. Ameliorating physical and chemical properties of highly weathered soils in the tropics with charcoal—A review. Biol. Fertil. Soils 2002, 35, 219–230. [Google Scholar] [CrossRef]
- Lehmann, J.; da Silva, J.P., Jr.; Steiner, C.; Nehls, T.; Zech, W.; Glaser, B. Nutrient availability and leaching in an archaeological Anthrosol and a Ferralsol of the Central Amazon basin: Fertilizer, manure and charcoal amendments. Plant. Soil 2003, 249, 343–357. [Google Scholar] [CrossRef]
- Mukherjee, A.; Zimmerman, A.R.; Harris, W. Surface chemistry variations among a series of laboratory-produced biochars. Geoderma 2011, 163, 247–255. [Google Scholar] [CrossRef]
- Hagemann, N.; Joseph, S.; Schmidt, H.P.; Kammann, C.; Harter, J.; Borch, T.; Young, R.B.; Varga, K.; Taherymoosavi, S.; Elliott, K.W.; et al. Organic coating on biochar explains its nutrient retention and stimulation of soil fertility. Nat. Commun. 2017, 8, 1089. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, A.; Zimmerman, A.R. Organic carbon and nutrient release from a range of laboratory-produced biochars and biochar–soil mixtures. Geoderma 2013, 193, 122–130. [Google Scholar] [CrossRef]
- Schneider, F.; Haderlein, S.B. Potential effects of biochar on the availability of phosphorus—Mechanistic insights. Geoderma 2016, 277, 83–90. [Google Scholar] [CrossRef]
- Li, J.; Li, B.; Huang, H.; Lv, X.; Zhao, N.; Guo, G.; Zhang, D. Removal of phosphate from aqueous solution by dolomite-modified biochar derived from urban dewatered sewage sludge. Sci. Total Environ. 2019, 687, 460–469. [Google Scholar] [CrossRef]
- Cui, X.; Dai, X.; Khan, K.Y.; Li, T.; Yang, X.; He, Z. Removal of phosphate from aqueous solution using magnesium-alginate/chitosan modified biochar microspheres derived from Thalia dealbata. Bioresour. Technol. 2016, 218, 1123–1132. [Google Scholar] [CrossRef] [PubMed]
- Takaya, C.A.; Fletcher, L.A.; Singh, S.; Okwuosa, U.C.; Ross, A.B. Recovery of phosphate with chemically modified biochars. J. Environ. Chem. Eng. 2016, 4, 1156–1165. [Google Scholar] [CrossRef]
- Trazzi, P.A.; Leahy, J.J.; Hayes, M.H.B.; Kwapinski, W. Adsorption and desorption of phosphate on biochars. J. Environ. Chem. Eng. 2016, 4, 37–46. [Google Scholar] [CrossRef]
- Zhang, H.; Chen, C.; Gray, E.M.; Boyd, S.E.; Yang, H.; Zhang, D. Roles of biochar in improving phosphorus availability in soils: A phosphate adsorbent and a source of available phosphorus. Geoderma 2016, 276, 1–6. [Google Scholar] [CrossRef]
- Uchimiya, M. Influence of pH, Ionic Strength, and Multidentate Ligand on the Interaction of CdII with Biochars. Acs Sustain. Chem. Eng. 2014, 2, 2019–2027. [Google Scholar] [CrossRef]
- Karunanayake, A.G.; Navarathna, C.M.; Gunatilake, S.R.; Crowley, M.; Anderson, R.; Mohan, D.; Perez, F.; Pittman, C.U.; Mlsna, T. Fe3O4 Nanoparticles Dispersed on Douglas Fir Biochar for Phosphate Sorption. Acs Appl. Nano Mater. 2019, 2, 3467–3479. [Google Scholar] [CrossRef]
- Novais, S.V.; Zenero, M.D.O.; Barreto, M.S.C.; Montes, C.R.; Cerri, C.E.P. Phosphorus removal from eutrophic water using modified biochar. Sci. Total Environ. 2018, 633, 825–835. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Wang, B.; Wester, A.E.; Chen, J.; He, F.; Chen, H.; Gao, B. Reclaiming phosphorus from secondary treated municipal wastewater with engineered biochar. Chem. Eng. J. 2019, 362, 460–468. [Google Scholar] [CrossRef]
- Lustosa Filho, J.F.; Barbosa, C.F.; da Silva Carneiro, J.S.; Melo, L.C.A. Diffusion and phosphorus solubility of biochar-based fertilizer: Visualization, chemical assessment and availability to plants. Soil Tillage Res. 2019, 194, 104298. [Google Scholar] [CrossRef]
- Mia, S.; Dijkstra, F.A.; Singh, B. Aging induced changes in biochar’s functionality and adsorption behavior for phosphate and ammonium. Environ. Sci. Technol. 2017, 51, 8359–8367. [Google Scholar] [CrossRef]
- Wang, Y.; Lin, Y.; Chiu, P.C.; Imhoff, P.T.; Guo, M. Phosphorus release behaviors of poultry litter biochar as a soil amendment. Sci. Total Environ. 2015, 512–513, 454–463. [Google Scholar] [CrossRef]
- Xu, M.; Gao, P.; Yang, Z.; Su, L.; Wu, J.; Yang, G.; Zhang, X.; Ma, J.; Peng, H.; Xiao, Y. Biochar impacts on phosphorus cycling in rice ecosystem. Chemosphere 2019, 225, 311–319. [Google Scholar] [CrossRef]
- Sun, K.; Qiu, M.; Han, L.; Jin, J.; Wang, Z.; Pan, Z.; Xing, B. Speciation of phosphorus in plant- and manure-derived biochars and its dissolution under various aqueous conditions. Sci. Total Environ. 2018, 634, 1300–1307. [Google Scholar] [CrossRef]
- Jin, Z.; Chen, X.; Chen, C.; Tao, P.; Han, Z.; Zhang, X. Biochar impact on nitrate leaching in upland red soil, China. Environ. Earth Sci. 2016, 75, 1109. [Google Scholar] [CrossRef]
- Khadem, A.; Raiesi, F. Response of soil alkaline phosphatase to biochar amendments: Changes in kinetic and thermodynamic characteristics. Geoderma 2019, 337, 44–54. [Google Scholar] [CrossRef]
- Jian, X.; Uchimiya, M.; Orlov, A. Particle Size- and Crystallinity-Controlled Phosphorus Release from Biochars. Energy Fuels 2019, 33, 5343–5351. [Google Scholar] [CrossRef]
- Uchimiya, M.; Wartelle, L.H.; Boddu, V.M. Sorption of Triazine and Organophosphorus Pesticides on Soil and Biochar. J. Agric. Food Chem. 2012, 60, 2989–2997. [Google Scholar] [CrossRef] [PubMed]
- Cai, D.; Wang, L.; Zhang, G.; Zhang, X.; Wu, Z. Controlling pesticide loss by natural porous micro/nano composites: Straw ash-based biochar and biosilica. Acs Appl. Mater. Interfaces 2013, 5, 9212–9216. [Google Scholar] [CrossRef]
- Tang, X.Y.; Huang, W.D.; Guo, J.J.; Yang, Y.; Tao, R.; Feng, X. Use of Fe-impregnated biochar to efficiently sorb chlorpyrifos, reduce uptake by Allium fistulosum L., and enhance microbial community diversity. J. Agric. Food Chem. 2017, 65, 5238–5243. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.B.; Ying, G.G.; Peng, P.A.; Wang, L.; Zhao, J.L.; Zhang, L.J.; Yuan, P.; He, H.P. Influence of Biochars on Plant Uptake and Dissipation of Two Pesticides in an Agricultural Soil. J. Agric. Food Chem. 2010, 58, 7915–7921. [Google Scholar] [CrossRef]
- Zhen, M.; Song, B.; Liu, X.; Chandankere, R.; Tang, J. Biochar-mediated regulation of greenhouse gas emission and toxicity reduction in bioremediation of organophosphorus pesticide-contaminated soils. Chin. J. Chem. Eng. 2018, 26, 2592–2600. [Google Scholar] [CrossRef]
- Lu, X.; Li, Y.; Wang, H.; Singh, B.P.; Hu, S.; Luo, Y.; Li, J.; Xiao, Y.; Cai, X.; Li, Y. Responses of soil greenhouse gas emissions to different application rates of biochar in a subtropical Chinese chestnut plantation. Agric. Meteorol. 2019, 271, 168–179. [Google Scholar] [CrossRef]
- Yang, X.; Meng, J.; Lan, Y.; Chen, W.; Yang, T.; Yuan, J.; Liu, S.; Han, J. Effects of maize stover and its biochar on soil CO2 emissions and labile organic carbon fractions in Northeast China. Agric. Ecosyst. Environ. 2017, 240, 24–31. [Google Scholar] [CrossRef]
- Wang, J.; Xiong, Z.; Kuzyakov, Y. Biochar stability in soil: Meta-analysis of decomposition and priming effects. Glob. Chang. Biol. Bioenergy 2016, 8, 512–523. [Google Scholar] [CrossRef]
- Luo, Y.; Zang, H.; Yu, Z.; Chen, Z.; Gunina, A.; Kuzyakov, Y.; Xu, J.; Zhang, K.; Brookes, P.C. Priming effects in biochar enriched soils using a three-source-partitioning approach: 14C labelling and 13C natural abundance. Soil Boil. Biochem. 2017, 106, 28–35. [Google Scholar] [CrossRef]
- Sagrilo, E.; Jeffery, S.; Hoffland, E.; Kuyper, T.W. Emission of CO2 from biochar-amended soils and implications for soil organic carbon. Glob. Chang. Biol. Bioenergy 2015, 7, 129–1304. [Google Scholar]
- Pignatello, J.J.; Mitch, W.A.; Xu, W. Activity and Reactivity of Pyrogenic Carbonaceous Matter toward Organic Compounds. Environ. Sci. Technol. 2017, 51, 8893–8908. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Zeng, G.; Huang, D.; Lai, C.; Chen, M.; Cheng, M.; Tang, W.; Tang, L.; Dong, H.; Huang, B.; et al. Biochar for environmental management: Mitigating greenhouse gas emissions, contaminant treatment, and potential negative impacts. Chem. Eng. J. 2019, 373, 902–922. [Google Scholar] [CrossRef]
- Liu, C.; Wang, H.; Li, P.; Xian, Q.; Tang, X. Biochar’s impact on dissolved organic matter (DOM) export from a cropland soil during natural rainfalls. Sci. Total. Environ. 2019, 650, 1988–1995. [Google Scholar] [CrossRef] [PubMed]
- Meschewski, E.; Holm, N.; Sharma, B.K.; Spokas, K.; Minalt, N.; Kelly, J.J. Pyrolysis biochar has negligible effects on soil greenhouse gas production, microbial communities, plant germination, and initial seedling growth. Chemosphere 2019, 228, 565–576. [Google Scholar] [CrossRef]
- Jaffe, R.; Ding, Y.; Niggemann, J.; Vahatalo, A.V.; Stubbins, A.; Spencer, R.G.; Campbell, J.; Dittmar, T. Global charcoal mobilization from soils via dissolution and riverine transport to the oceans. Science 2013, 340, 345–347. [Google Scholar] [CrossRef] [PubMed]
- Ameloot, N.; Sleutel, S.; Das, K.C.; Kanagaratnam, J.; de Neve, S. Biochar amendment to soils with contrasting organic matter level: Effects on N mineralization and biological soil properties. Glob. Chang. Biol. Bioenergy 2013, 7, 135–144. [Google Scholar] [CrossRef]
- Sánchez-García, M.; Alburquerque, J.A.; Sánchez-Monedero, M.A.; Roig, A.; Cayuela, M.L. Biochar accelerates organic matter degradation and enhances N mineralisation during composting of poultry manure without a relevant impact on gas emissions. Bioresour. Technol. 2015, 192, 272–279. [Google Scholar] [CrossRef]
- Nelissen, V.; Rütting, T.; Huygens, D.; Staelens, J.; Ruysschaert, G.; Boeckx, P. Maize biochars accelerate short-term soil nitrogen dynamics in a loamy sand soil. Soil Biol. Biochem. 2012, 55, 20–27. [Google Scholar] [CrossRef]
- Nelissen, V.; Rutting, T.; Huygens, D.; Ruysschaert, G.; Boeckx, P. Temporal evolution of biochar’s impact on soil nitrogen processes—a 15N tracing study. Glob. Chang. Biol. Bioenergy 2015, 7, 635–645. [Google Scholar] [CrossRef]
- Kuzyakov, Y.; Subbotina, I.; Chen, H.; Bogomolova, I.; Xu, X. Black carbon decomposition and incorporation into soil microbial biomass estimated by 14C labeling. Soil Biol. Biochem. 2009, 41, 210–219. [Google Scholar] [CrossRef]
- Mia, S.; Singh, B.; Dijkstra, F.A. Aged biochar affects gross nitrogen mineralization and recovery: A 15N study in two contrasting soils. Glob. Chang. Biol. Bioenergy 2017, 9, 1196–1206. [Google Scholar] [CrossRef]
- Zimmerman, A.R.; Gao, B.; Ahn, M.Y. Positive and negative carbon mineralization priming effects among a variety of biochar-amended soils. Soil Biol. Biochem. 2011, 43, 1169–1179. [Google Scholar] [CrossRef]
- Gao, S.; DeLuca, T.H.; Cleveland, C.C. Biochar additions alter phosphorus and nitrogen availability in agricultural ecosystems: A meta-analysis. Sci. Total Environ. 2019, 654, 463–472. [Google Scholar] [CrossRef]
- Liu, Y.; Lonappan, L.; Brar, S.K.; Yang, S. Impact of biochar amendment in agricultural soils on the sorption, desorption, and degradation of pesticides: A review. Sci. Total Environ. 2018, 645, 60–70. [Google Scholar] [CrossRef] [PubMed]
- Gul, S.; Whalen, J.K. Biochemical cycling of nitrogen and phosphorus in biochar-amended soils. Soil Biol. Biochem. 2016, 103, 1–15. [Google Scholar] [CrossRef]
- Béghin-Tanneau, R.; Guérin, F.; Guiresse, M.; Kleiber, D.; Scheiner, J.D. Carbon sequestration in soil amended with anaerobic digested matter. Soil Tillage Res. 2019, 192, 87–94. [Google Scholar] [CrossRef]
- Wang, J.; Wang, S. Preparation, modification and environmental application of biochar: A review. J. Clean. Prod. 2019, 227, 1002–1022. [Google Scholar] [CrossRef]
- Llorach-Massana, P.; Lopez-Capel, E.; Pena, J.; Rieradevall, J.; Montero, J.I.; Puy, N. Technical feasibility and carbon footprint of biochar co-production with tomato plant residue. Waste Manag. 2017, 67, 121–130. [Google Scholar] [CrossRef]
- Chen, M.; Wang, D.; Yang, F.; Xu, X.; Xu, N.; Cao, X. Transport and retention of biochar nanoparticles in a paddy soil under environmentally-relevant solution chemistry conditions. Environ. Pollut. 2017, 230, 540–549. [Google Scholar] [CrossRef] [PubMed]
- Ishizuka, J. Trends in biological nitrogen fixation research and application. Plant. Soil 1992, 141, 197–209. [Google Scholar] [CrossRef]
- Mia, S.; Van Groenigen, J.W.; Van De Voorde, T.F.J.; Oram, N.J.; Bezemer, T.M.; Mommer, L.; Jeffery, S. Biochar application rate affects biological nitrogen fixation in red clover conditional on potassium availability. Agric. Ecosyst. Environ. 2014, 191, 83–91. [Google Scholar] [CrossRef]
- Abujabhah, I.S.; Doyle, R.B.; Bound, S.A.; Bowman, J.P. Assessment of bacterial community composition, methanotrophic and nitrogen-cycling bacteria in three soils with different biochar application rates. J. Soils Sediments 2018, 18, 148–158. [Google Scholar] [CrossRef]
- Wang, Z.; Zong, H.; Zheng, H.; Liu, G.; Chen, L.; Xing, B. Reduced nitrification and abundance of ammonia-oxidizing bacteria in acidic soil amended with biochar. Chemosphere 2015, 138, 576–583. [Google Scholar] [CrossRef]
- Ball, P.N.; MacKenzie, M.D.; DeLuca, T.H.; Montana, W.E.H. Wildfire and charcoal enhance nitrification and ammonium-oxidizing bacterial abundance in dry montane forest soils. J. Environ. Qual. 2010, 39, 1243–1253. [Google Scholar] [CrossRef]
- Kleiblinger, K.M.; Zehetner, F.; Mentler, A.; Zechmeister-Boltenstern, S. Biochar application increases sorption of nitrification inhibitor 3,4-dimethylpyrazole phosphate in soil. Environ. Sci. Pollut. Res. 2018, 25, 11173–11177. [Google Scholar] [CrossRef] [PubMed]
- Fuertes-Mendizábal, T.; Huérfano, X.; Vega-Mas, I.; Torralbo, F.; Menéndez, S.; Ippolito, J.A.; Kammann, C.; Wrage-Mönnig, N.; Cayuela, M.L.; Borchard, N.; et al. Biochar reduces the efficiency of nitrification inhibitor 3,4-dimethylpyrazole phosphate (DMPP) mitigating N2O emissions. Sci. Rep. 2019, 9, 2346. [Google Scholar] [CrossRef] [PubMed]
- Thakur, I.S.; Medhi, K. Nitrification and denitrification processes for mitigation of nitrous oxide from waste water treatment plants for biovalorization: Challenges and opportunities. Bioresour. Technol. 2019, 282, 502–513. [Google Scholar] [CrossRef]
- Braker, G.; Conrad, R. Diversity, structure, and size of N2O producing microbial communities in soils-what matters for their functioning? Adv. Appl. Microbiol. 2011, 75, 33–70. [Google Scholar]
- Kappler, A.; Wuestner, M.L.; Ruecker, A.; Harter, J.; Halama, M.; Behrens, S. Biochar as an electron shuttle between bacteria and Fe(III) minerals. Environ. Sci. Technol. Lett. 2014, 1, 339–344. [Google Scholar] [CrossRef]
- Wu, Z.S.; Xu, F.; Yang, C.; Su, X.X.; Guo, F.C.; Xu, Q.Y.; Peng, G.; He, Q.; Chen, Y. Highly efficient nitrate removal in a heterotrophic denitrification system amended with redoxactive biochar: A molecular and electrochemical mechanism. Bioresour. Technol. 2019, 275, 297–306. [Google Scholar] [CrossRef]
- Ribas, A.; Mattana, S.; Llurba, R.; Debouk, H.; Sebastià, M.T.; Domene, X. Biochar application and summer temperatures reduce N2O and enhance CH4 emissions in a Mediterranean agroecosystem: Role of biologically-induced anoxic microsites. Sci. Total Environ. 2019, 685, 1075–1086. [Google Scholar] [CrossRef] [PubMed]
- Anderson, C.R.; Condron, L.M.; Clough, T.J.; Fiers, M.; Stewart, A.; Hill, R.A.; Sherlock, R.R. Biochar induced soil microbial community change: Implications for biogeochemical cycling of carbon, nitrogen and phosphorus. Pedobiologia 2011, 54, 309–320. [Google Scholar] [CrossRef]
- Schmalenberger, A.; Fox, A. Chapter Three—Bacterial Mobilization of Nutrients from Biochar-Amended Soils. Adv. Appl. Microbiol. 2016, 94, 109–159. [Google Scholar]
- Ma, L.; Lv, X.; Cao, N.; Wang, Z.; Zhou, Z.; Meng, Y. Alterations of soil labile organic carbon fractions and biological properties under different residue-management methods with equivalent carbon input. Appl. Soil Ecol. 2021, 161, 103821. [Google Scholar] [CrossRef]
- Song, D.; Chen, L.; Zhang, S.; Zheng, Q.; Ullah, S.; Zhou, W.; Wang, X. Combined biochar and nitrogen fertilizer change soil enzyme and microbial activities in a 2-year field trial. Eur. J. Soil Biol. 2020, 99, 103212. [Google Scholar] [CrossRef]
- Liu, Y.; Yang, H.; Li, X.; Xing, Z. Effects of biological soil crusts on soil enzyme activities in revegetated areas of the Tengger Desert, China. Appl. Soil Ecol. 2014, 80, 6–14. [Google Scholar] [CrossRef]
- Yang, H.; Liu, C.; Liu, Y.; Xing, Z. Impact of human trampling on biological soil crusts determined by soil microbial biomass, enzyme activities and nematode communities in a desert ecosystem. Eur. J. Soil Biol. 2018, 87, 61–71. [Google Scholar] [CrossRef]
- Li, S.; Liang, C.; Shangguan, Z. Effects of apple branch biochar on soil C mineralization and nutrient cycling under two levels of N. Sci. Total Environ. 2017, 607–608, 109–119. [Google Scholar] [CrossRef] [PubMed]
- Qian, T.; Yang, Q.; Jun, D.C.F.; Dong, F.; Zhou, Y. Transformation of phosphorus in sewage sludge biochar mediated by a phosphate-solubilizing microorganism. Chem. Eng. J. 2019, 359, 1573–1580. [Google Scholar] [CrossRef]
- Masto, R.E.; Kumar, S.; Rout, T.K.; Sarkar, P.; George, J.; Ram, L.C. Biochar from water hyacinth (Eichornia crassipes) and its impact on soil biological activity. Catena 2013, 111, 64–71. [Google Scholar] [CrossRef]
- Saifullah; Dahlawi, S.; Naeem, A.; Rengel, Z.; Naidu, R. Biochar application for the remediation of salt-affected soils: Challenges and opportunities. Sci. Total Environ. 2018, 625, 320–335. [Google Scholar] [CrossRef] [PubMed]
- Mansoor, S.; Kour, N.; Manhas, S.; Zahid, S.; Wani, O.A.; Sharma, V.; Wijaya, L.; Alyemeni, M.N.; Alsahli, A.A.; El-Serehy, H.A.; et al. Biochar as a tool for effective management of drought and heavy metal toxicity. Chemosphere 2021, 271, 129458. [Google Scholar] [CrossRef] [PubMed]
- Liang, J.; Li, Y.; Si, B.; Wang, Y.; Chen, X.; Wang, X.; Chen, H.; Wang, H.; Zhang, F.; Bai, Y.; et al. Optimizing biochar application to improve soil physical and hydraulic properties in saline-alkali soils. Sci. Total Environ. 2021, 771, 144802. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Lu, H.; Chu, L.; Shao, H.; Shi, W. Biochar applied with appropriate rates can reduce N leaching, keep N retention and not increase NH3 volatilization in a coastal saline soil. Sci. Total Environ. 2017, 575, 820–825. [Google Scholar] [CrossRef] [PubMed]
- Xu, G.; Zhang, Y.; Sun, J.; Shao, H. Negative interactive effects between biochar and phosphorus fertilization on phosphorus availability and plant yield in saline sodic soil. Sci. Total Environ. 2016, 568, 910–915. [Google Scholar] [CrossRef]
- Mohanty, S.K.; Boehm, A.B. Effect of weathering on mobilization of biochar particles and bacterial removal in a stormwater biofilter. Water Res. 2015, 85, 208–215. [Google Scholar] [CrossRef]
- Spokas, K.A. Impact of biochar field aging on laboratory greenhouse gas production potentials. Glob. Chang. Biol. Bioenergy 2013, 5, 165–176. [Google Scholar] [CrossRef]
- Kookana, R.S. The role of biochar in modifying the environmental fate, bioavailability, and efficacy of pesticides in soils: A review. Soil Res. 2010, 48, 627–637. [Google Scholar] [CrossRef]
- Yang, Y.; Sheng, G. Pesticide adsorptivity of aged particulate matter arising from crop residue burns. J. Agric. Food Chem. 2003, 51, 5047–5051. [Google Scholar] [CrossRef]
- Trigo, C.; Spokas, K.A.; Cox, L.; Koskinen, W.C. Influence of soil biochar aging on sorption of the herbicides MCPA, nicosulfuron, terbuthylazine, indaziflam, and fluoroethyldiaminotriazine. J. Agric. Food Chem. 2014, 62, 10855–10860. [Google Scholar] [CrossRef]
- Yao, F.X.; Arbestain, M.C.; Virgel, S.; Blanco, F.; Arostegui, J.; Macia-Agullo, J.A.; Macias, F. Simulated geochemical weathering of a mineral ash-rich biochar in a modified Soxhlet reactor. Chemosphere 2010, 80, 724–732. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Sotoudehniakarani, F.; Yu, Z.; Morrell, J.J.; Cappellazzi, J.; McDonald, A.G. Evaluation of corrugated cardboard biochar as reinforcing fiber on properties, biodegradability and weatherability of wood-plastic composites. Polym. Degrad. Stab. 2019, 168, 108955. [Google Scholar] [CrossRef]
- Kim, H.B.; Kim, J.G.; Kim, T.; Alessi, D.S.; Baek, K. Interaction of biochar stability and abiotic aging: Influences of pyrolysis reaction medium and temperature. Chem. Eng. J. 2021, 411, 128441. [Google Scholar] [CrossRef]
- Purakayasth, T.J.; Bera, T.; Bhaduri, D.; Sarkar, B.; Mandal, S.; Wade, P.; Kumari, S.; Biswas, S.; Menon, M.; Pathak, H.; et al. A review on biochar modulated soil condition improvements and nutrient dynamics concerning crop yields: Pathways to climate change mitigation and global food security. Chemosphere 2019, 227, 345–365. [Google Scholar] [CrossRef]
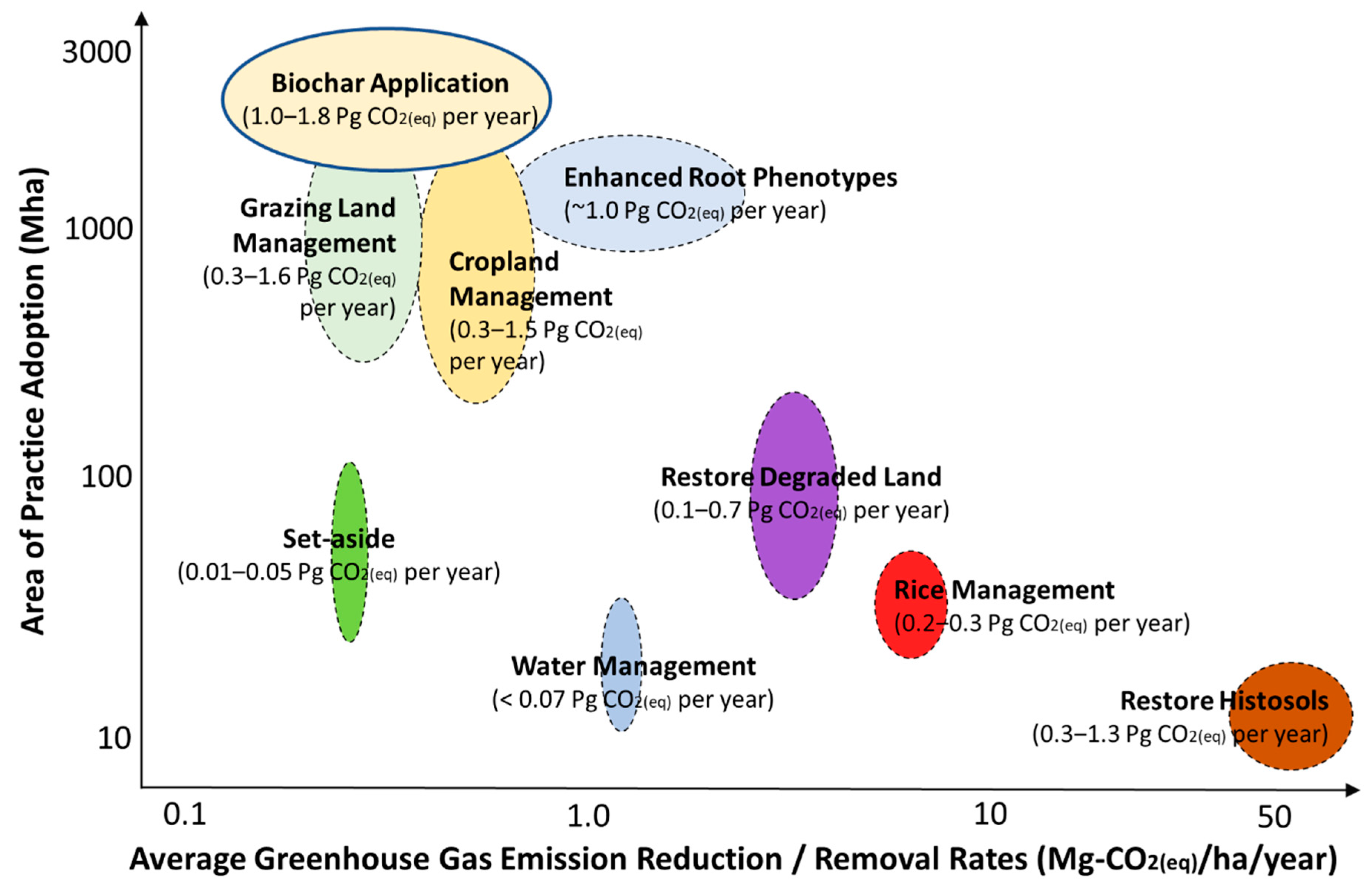
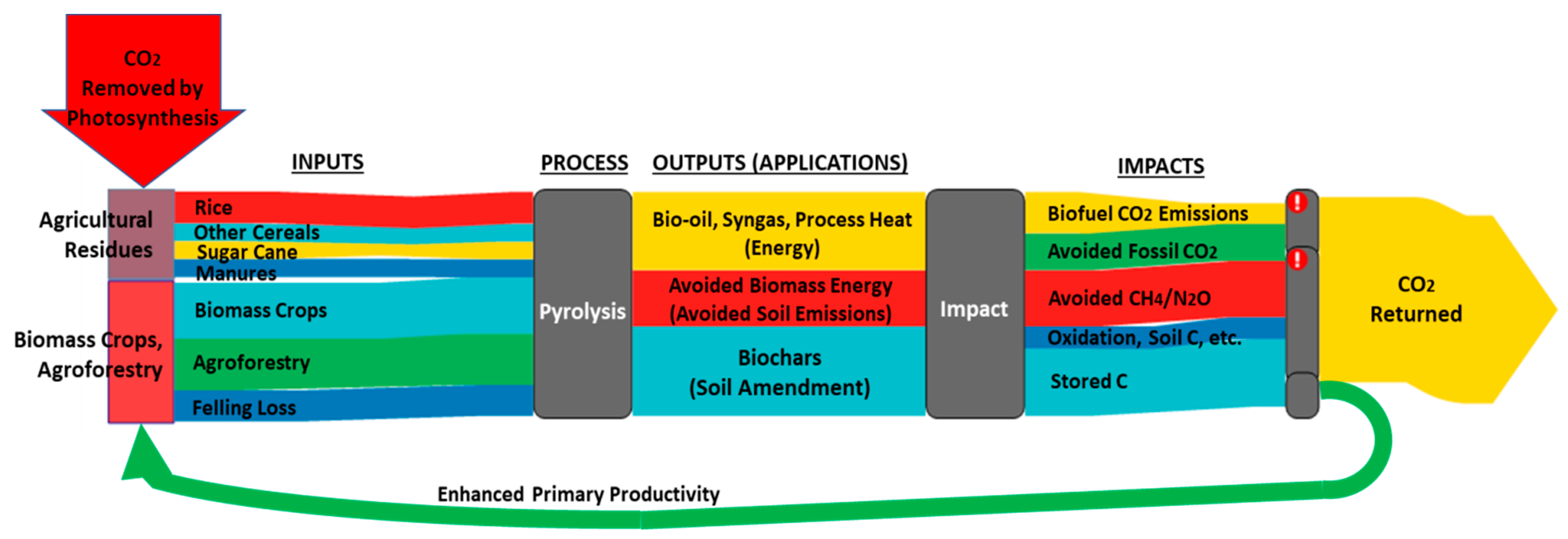
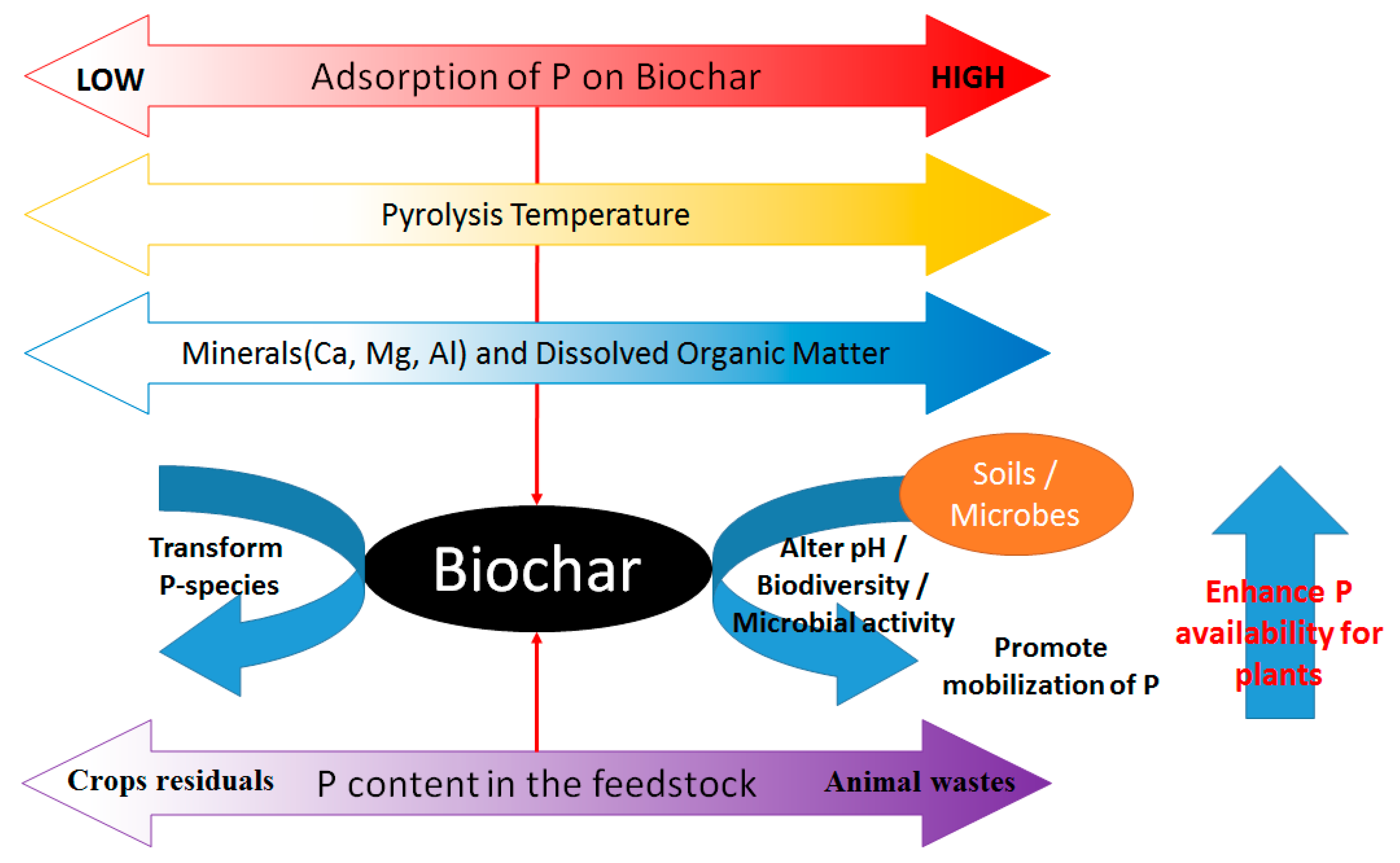
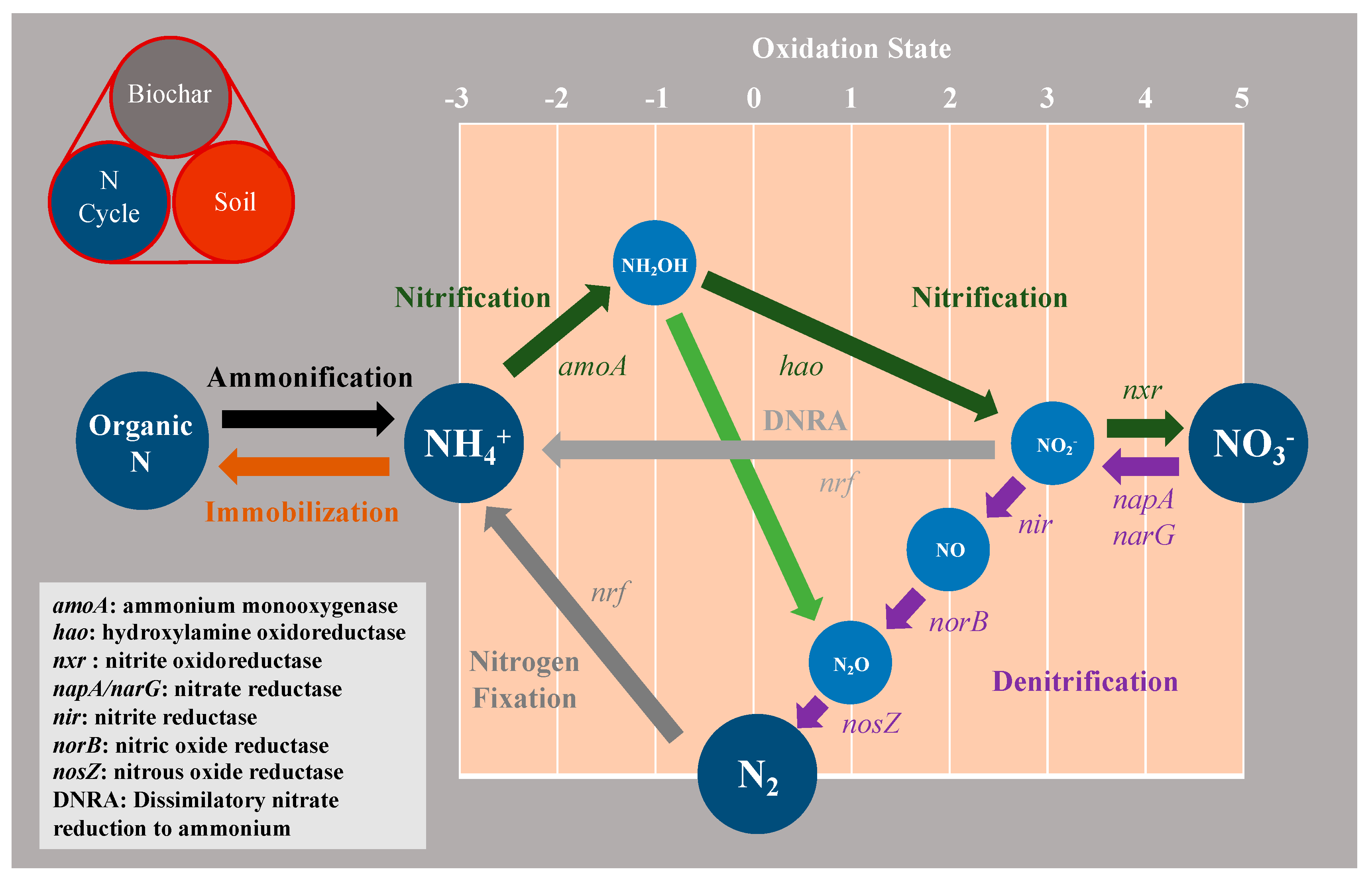
| Feedstock | Pyrolysis Temp. (°C) | Yield (%) | pH | C (%) | H (%) | O (%) | N (%) | BET Surface Area (m2g−1) | Pore Volume (cm3g−1) | Adsorbate | Adsorption Capacity (mg g-Biochar−1) | References |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Pine needles | 100 | 91.2 | ‒ | 50.9 | 6.15 | 42.3 | 0.71 | 0.7 | ‒ | naphthalene | 0 | [27] |
| nitrobenzene | 0 | |||||||||||
| m-dinitrobenzene | 0 | |||||||||||
| 200 | 75.3 | ‒ | 57.1 | 5.71 | 36.3 | 0.88 | 6.2 | ‒ | naphthalene | 0.329 | ||
| nitrobenzene | 5.908 | |||||||||||
| m-dinitrobenzene | 5.655 | |||||||||||
| 250 | 56.1 | ‒ | 61.2 | 5.54 | 32.4 | 0.86 | 9.5 | ‒ | naphthalene | 1.724 | ||
| nitrobenzene | 10.08 | |||||||||||
| m-dinitrobenzene | 7.535 | |||||||||||
| 300 | 48.6 | ‒ | 68.9 | 4.31 | 25.8 | 1.08 | 19.9 | ‒ | naphthalene | 4.006 | ||
| nitrobenzene | 51.14 | |||||||||||
| m-dinitrobenzene | 17.09 | |||||||||||
| 400 | 30.0 | ‒ | 77.9 | 2.95 | 18.0 | 1.16 | 112.4 | 0.044 | naphthalene | 25.69 | ||
| nitrobenzene | 79.71 | |||||||||||
| m-dinitrobenzene | 60.01 | |||||||||||
| 500 | 26.1 | ‒ | 81.7 | 2.26 | 15.0 | 1.11 | 236.4 | 0.095 | naphthalene | 27.40 | ||
| nitrobenzene | 96.63 | |||||||||||
| m-dinitrobenzene | 55.23 | |||||||||||
| 600 | 20.4 | ‒ | 85.4 | 1.85 | 11.8 | 0.98 | 206.7 | 0.076 | naphthalene | 15.14 | ||
| nitrobenzene | 91.99 | |||||||||||
| m-dinitrobenzene | 35.83 | |||||||||||
| 700 | 14.0 | ‒ | 86.5 | 1.28 | 11.1 | 1.13 | 490.8 | 0.186 | naphthalene | 136.8 | ||
| nitrobenzene | 181.2 | |||||||||||
| m-dinitrobenzene | 208.0 | |||||||||||
| Wood chip shavings | 550 | ‒ | ‒ | 83.1 | <1.1 | 11.2 | 0.63 | ‒ | ‒ | ‒ | ‒ | [38] |
| Wood chip | 700 | ‒ | 9.5 | 75.8 | 1.31 | 6.2 | 0.43 | 144 | ‒ | NO3− | 0.15 | [9] |
| Wood chip-compost | 600–750 | ‒ | ‒ | ‒ | ‒ | ‒ | ‒ | ‒ | ‒ | NO3− | 5.2 | |
| Coconut shell | 500 | ‒ | ‒ | ‒ | ‒ | ‒ | ‒ | 700.3 | 0.2018 | urea | 256.41 | [39] |
| Woody chips | 700 | ‒ | 9.4 | 85.7 | 7.60 | 5.30 | 0.20 | ‒ | ‒ | ‒ | ‒ | [40] |
| Woody chips-AW a | 700 | ‒ | 6.3 | 84.4 | 7.30 | 6.70 | 0.40 | ‒ | ‒ | NO3− | 0.83 | |
| Woody chips-N50, AW b | 700 | ‒ | 3.2 | 77.1 | 6.90 | 9.00 | 0.40 | ‒ | ‒ | NO3− | 3.53 | |
| Woody chips-N50, TEMPO, AW | 700 | ‒ | 3.0 | 78.1 | 7.00 | 8.20 | 0.40 | ‒ | ‒ | NO3− | 3.68 | |
| Birch wood | 700 | ‒ | 9.6 | 90.7 | 7.60 | 0.50 | 0.20 | ‒ | ‒ | ‒ | ‒ | |
| Birch wood- AW | 700 | ‒ | 7.6 | 88.6 | 7.00 | 1.90 | 0.20 | ‒ | ‒ | NO3− | 0.49 | |
| Birch wood- N50, AW | 700 | ‒ | 3.6 | 80.7 | 6.60 | 7.45 | 0.20 | ‒ | ‒ | NO3− | 3.97 | |
| fruit trees pruning wood | 500 | ‒ | 9.8 | 34.2 | ‒ | ‒ | 0.50 | 98 | ‒ | ‒ | ‒ | [41] |
| Biosolids–DBS c | ‒ | ‒ | ‒ | 37.3 | ‒ | ‒ | 48.2 | ‒ | ‒ | terbuthylazine | Kd = 2.56 L/kg g | [42] |
| Biosolids–UBS d | ‒ | ‒ | ‒ | 6.42 | ‒ | ‒ | 5.13 | ‒ | ‒ | terbuthylazine | Kd = 3.22 L/kg | |
| Charcoal | 350 | ‒ | ‒ | 83.4 | ‒ | ‒ | 0.21 | ‒ | ‒ | terbuthylazine | Kd = 6.69 L/kg | |
| Sawdust | 700 | ‒ | ‒ | 89.4 | ‒ | ‒ | 0.17 | ‒ | ‒ | terbuthylazine | Kd = 115.6 L/kg | |
| Oak sawdust | 300 | 76.17 | 6.8 | 50.2 | 5.78 | 41.6 | 0.27 | ‒ | ‒ | NH4+ | 5.31 | [28] |
| Oak sawdust | 500 | 24.90 | 8.3 | 76.5 | 3.42 | 16.6 | 0.39 | ‒ | ‒ | ‒ | ‒ | [28] |
| Oak sawdust | 600 | 21.60 | 8.4 | 80.4 | 3.12 | 12.2 | 0.43 | ‒ | ‒ | NO3− | 8.94 | [28] |
| Oak sawdust-LaCl3 | 300 | 73.07 | 5.4 | 40.3 | 5.48 | 40.2 | 0.28 | ‒ | ‒ | NH4+ | 10.10 | [28] |
| Oak sawdust-LaCl3 | 500 | 30.50 | 7.8 | 69.8 | 3.37 | 23.2 | 0.46 | ‒ | ‒ | ‒ | ‒ | [28] |
| Oak sawdust-LaCl3 | 600 | 29.13 | 7.9 | 75.7 | 2.64 | 17.6 | 0.48 | ‒ | ‒ | NO3− | 100.0 | [28] |
| Lignin | RT | ‒ | ‒ | 43.7 | 4.50 | 31.1 | 0.12 | 2.01 | 0.00624 | NO3− | 0.189 | [43] |
| 200 | ‒ | ‒ | 54.4 | 4.89 | 32.0 | 0.12 | 1.84 | 0.00578 | NO3− | 0.143 | ||
| 350 | ‒ | ‒ | 62.6 | 3.27 | 22.5 | 0.16 | 0.710 | 0.00292 | NO3− | 0.408 | ||
| 500 | ‒ | ‒ | 72.1 | 2.03 | 14.9 | 0.26 | 208 | 0.0855 | NO3− | 0.505 | ||
| 700 | ‒ | ‒ | 63.8 | 0.91 | 19.8 | 0.42 | 201 | 0.0976 | NO3− | 0.785 | ||
| Lignin-TA e | 500 | ‒ | ‒ | 85.8 | 2.45 | 9.30 | 0.39 | 3.65 | 0.00595 | NO3− | 0.357 | |
| Sugarcane bagasse | 300 | ‒ | 7.2 | 69.5 | 4.20 | 24.5 | 0.90 | 5.2 | ‒ | NH4+ | 0.175% h | [44] |
| NO3− | −0.361% | |||||||||||
| 450 | ‒ | 7.9 | 78.6 | 3.5 | 15.5 | 0.9 | 15.3 | ‒ | NH4+ | −0.100% | ||
| NO3− | −0.585% | |||||||||||
| 600 | ‒ | 7.9 | 76.5 | 2.9 | 18.3 | 0.8 | 4.2 | ‒ | NH4+ | 0.090% | ||
| NO3− | 0.636% | |||||||||||
| Peanut hull | 300 | ‒ | 7.8 | 73.9 | 3.9 | 19.1 | 1.6 | 0.8 | ‒ | NH4+ | 0.550% | |
| NO3− | −0.619% | |||||||||||
| 450 | ‒ | 8.2 | 81.5 | 2.9 | 13.0 | 1.0 | 21.8 | ‒ | NH4+ | −1.00% | ||
| NO3− | −0.430% | |||||||||||
| 600 | ‒ | 8.0 | 86.4 | 1.4 | 10.0 | 0.9 | 27.1 | ‒ | NH4+ | 0.500% | ||
| NO3− | 0.0344% | |||||||||||
| 300 (HT) f | ‒ | 6.8 | 56.4 | 5.6 | 36.7 | 0.9 | 5.6 | ‒ | NH4+ | −0.15% | ||
| NO3− | −1.290% | |||||||||||
| Brazilian pepperwood | 300 | ‒ | 6.6 | 59.3 | 5.2 | 34.1 | 0.3 | 81.1 | ‒ | NH4+ | 0.190% | |
| NO3− | −0.722% | |||||||||||
| 450 | ‒ | 7.3 | 75.6 | 3.6 | 17.2 | 0.3 | 0.7 | ‒ | NH4+ | 0.785% | ||
| NO3− | −0.791% | |||||||||||
| 600 | ‒ | 9.1 | 77.0 | 2.2 | 17.7 | 0.1 | 234.7 | ‒ | NH4+ | 0.595% | ||
| NO3− | 0.0206% | |||||||||||
| Bamboo | 300 | ‒ | 6.7 | 66.2 | 4.7 | 27.7 | 0.4 | 1.3 | ‒ | NH4+ | 0.200% | |
| NO3− | −0.224% | |||||||||||
| 450 | ‒ | 5.2 | 76.9 | 3.6 | 18.1 | 0.2 | 18.2 | ‒ | NH4+ | −0.650% | ||
| NO3− | −0.344% | |||||||||||
| 600 | ‒ | 7.9 | 80.9 | 2.4 | 14.9 | 0.2 | 470.4 | ‒ | NH4+ | 0.200% | ||
| NO3− | 0.430% | |||||||||||
| Corn straw | 100 | ‒ | ‒ | 47.5 | 6.23 | 0.36 | 46.0 | 2.14 | ‒ | simazine | Kd = 124 L/kg | [45] |
| 200 | ‒ | ‒ | 53.8 | 5.71 | 0.66 | 39.9 | 2.15 | ‒ | simazine | Kd = 574 L/kg | ||
| 300 | ‒ | ‒ | 66.9 | 4.14 | 1.24 | 27.7 | 6.14 | ‒ | simazine | Kd = 2669 L/kg | ||
| 400 | ‒ | ‒ | 76.5 | 3.99 | 1.27 | 18.2 | 32.4 | 0.003 | simazine | Kd = 1618 L/kg | ||
| 500 | ‒ | ‒ | 82.0 | 3.36 | 1.03 | 13.6 | 245.3 | 0.0345 | simazine | Kd = 3133 L/kg | ||
| 600 | ‒ | ‒ | 84.3 | 2.60 | 1.16 | 12.0 | 329.0 | 0.0839 | simazine | Kd = 4054 L/kg |
| Biochar Source | Property of Soil a | Property of Biochar b | Performance of Carbon Sequestration c | Reference | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| pH | CEC | TIC | TOC | pH | CEC | TIC | TOC | dp | L | t | SOC0 | SOCL | SIC0 | SICL | η | ||
| Forest shrub | 4.6 | 6.4 | ‒ | 16 | 9.3 | 72 | ‒ | 700 | ‒ | 40 | 2.5 | 1.81 ± 0.01 | 4.99 ± 0.01 | ‒ | ‒ | 175 | [79] |
| Wood residue | 5.5 | ‒ | ‒ | 34 | 9.3 | 9 | ‒ | 550 | ‒ | 10 | 2 | 3.45–3.86 | 4.61–4.86 | ‒ | ‒ | 41 | [80] |
| Wood residue | 4.8 | 3.6 | ‒ | 4.1 | ‒ | ‒ | ‒ | 815 | ‒ | 4% | 0.5 | 4.1 | 32.7 | ‒ | ‒ | 698 | [81] |
| Wood residue | 5.2 | 17.8 | ‒ | 15.3 | ‒ | ‒ | ‒ | 815 | ‒ | 4% | 0.5 | 15.3 | 46.2 | ‒ | ‒ | 202 | [81] |
| Wheat straw | 7.9 | ‒ | ‒ | 15.2 | 11.6 | ‒ | ‒ | 484 | 0–1 | 5% | 1.8 | ~15.2 | ~41.0 | ‒ | ‒ | 170 | [82] |
| Wheat straw | 8.1 | 22.1 | ‒ | 10.3 | 10.4 | 21.8 | ‒ | 465 | <0.25 | 3 | 3 | 12.79 ± 0.21 | 17.49 ± 0.43 | ‒ | ‒ | 37 | [77] |
| Wheat straw | 5.1 | 8.0 | ‒ | 17.5 | 9.3 | 28.6 | ‒ | 418 | ‒ | 48 | 4 | 17.6 ± 0.1 | 28.4 ± 0.4 | ‒ | ‒ | 61 | [83] |
| Wheat straw | 4.5 | 15.2 | ‒ | 9.5 | 10.4 | 21.7 | ‒ | 467 | ‒ | 40 | 5 | 8.80 ± 0.02 | 13.31 ± 0.11 | ‒ | ‒ | 51 | [84] |
| Wheat straw | 8.0 | 10 | ‒ | 4.3 | 10.6 | 12.5 | ‒ | 491 | ‒ | 30 | 5 | 4.32 | 6.96 | ‒ | ‒ | 61 | [85] |
| Wheat straw | 8.1 | 22.1 | ‒ | 10.3 | 10.4 | 21.8 | ‒ | 465 | ‒ | 30 | 3 | 10.32 ± 0.09 | 13.71 ± 0.23 | ‒ | ‒ | 33 | [77] |
| Wheat straw & peanut shell | 7.0 | ‒ | ‒ | 12.4 | 10.2 | ‒ | ‒ | ‒ | 0.3–2 | 8 | 2 | ‒ | ‒ | ‒ | ‒ | 56 * | [72] |
| Rape straw | 6.7 | ‒ | ‒ | 10.0 | 8.9 | ‒ | ‒ | 626 | 10 | 5.3 | ‒ | 11.82 ± 0.47 | 20.76 ± 0.89 | ‒ | ‒ | 76 | [86] |
| Maize straw | 5.7 | ‒ | ‒ | 16.0 | 9.4 | ‒ | ‒ | 507 | ‒ | 6.3 | 5 | 17.71 | 21.86 | ‒ | ‒ | 23 | [87] |
| Rice straw | 6.0 | 10.9 | ‒ | 16.6 | 9.2 | 18.9 | ‒ | 620 | ‒ | 9 | 2 | ~24 | ~36 | ‒ | ‒ | 50 | [88] |
| Rice straw | 5.2 | 11.3 | ‒ | 21.8 | 10.4 | 19.4 | ‒ | 725 | ‒ | 11.3 | 5 | 45.1 ± 0.4 | 126.0 ± 0.2 | ‒ | ‒ | 179 | [6] |
| Rice husks | 6.8 | 24.0 | ‒ | 7.1 | 8.9 | 48.9 | ‒ | 500 | ‒ | 10 | 6 | 12.1 | 32.6 | ‒ | ‒ | 169 | [89] |
| Rice husks | 4.3 | 12.3 | ‒ | 5.3 | 9.2 | 17.6 | ‒ | 478 | ‒ | 3% | 0.8 | 0.53 ± 0.02 | 1.42 ± 0.03 | ‒ | ‒ | 79 | [90] |
| Rice husks | 6.9 | 5.7 | ‒ | 11.0 | 9.2 | 17.6 | ‒ | 478 | ‒ | 3% | 0.8 | 1.10 ± 0.03 | 1.87 ± 0.04 | ‒ | ‒ | 70 | [90] |
| Rice husks | 5.8 | 31.5 | ‒ | 4.6 | 9.2 | ‒ | ‒ | 491 | ‒ | 4% | 0.25 | 0.26 | 0.63 | ‒ | ‒ | 142 | [91] |
| Rice husks & cotton seed | 8.0 | 10 | 2.1 | 4.3 | 10.6 | 12.5 | 10.0 | 491 | 0.5–5 | 90 | 5 | ‒ | ‒ | 0.62 | 0.79 | 27 | [92] |
| Corn cob | 6.0 | ‒ | ‒ | 12.3 | 9.8 | ‒ | ‒ | 803 | ‒ | 40 | 0.13 | ~14 | ~18 | ‒ | ‒ | 29 | [93] |
| Corn cob | 7.9 | ‒ | ‒ | 8.2 | 9.6 | ‒ | ‒ | 572 | ‒ | 100 | 0.64 | 8.23 ± 0.25 | 24.00 ± 0.26 | ‒ | ‒ | 192 | [94] |
| Corn stover | 5.4 | ‒ | ‒ | 34 | 10.0 | 24 | ‒ | 480 | ‒ | 10 | 2 | 3.70–3.86 | 6.65–4.21 | ‒ | ‒ | 80 | [80] |
| Bamboo leaf | 4.5 | ‒ | ‒ | 18.6 | 9.4 | ‒ | ‒ | 667 | ‒ | 15 | 1 | ~0.15 | ~0.19 | ‒ | ‒ | 27 | [95] |
| Bluestem grass | 7.8 | ‒ | ‒ | 6.2 | 9.7 | ‒ | ‒ | 500 | ‒ | 10% | 3.5 | ‒ | 5.50 ± 0.28 | ‒ | ‒ | 39 | [96] |
| Bluestem grass | 7.7 | ‒ | ‒ | 5.8 | 9.7 | ‒ | ‒ | 500 | ‒ | 10% | 3.5 | ‒ | 5.70 ± 0.23 | ‒ | ‒ | 99 | [96] |
| Sweetgrass | 4.9 | ‒ | ‒ | 16 | 10.8 | 19 | ‒ | 550 | ‒ | 10 | 2 | 2.24–2.34 | 3.64–5.41 | ‒ | ‒ | 131 | [80] |
| Pig manure | 4.9 | ‒ | ‒ | ‒ | 8.6 | ‒ | ‒ | 474 | ‒ | 2% | 0.13 | 0.062 | 0.108 | ‒ | ‒ | 74 | [97] |
| Feedstock | Pyrolysis Temp. (°C) | Yield (%) | pH | C (%) | H (%) | O (%) | N (%) | C/N | BET Surface Area (m2g−1) | Pore Volume (cm3g−1) | Soil Organic Level | Net Mineralization (mg N g-soil−1) | References |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Pine chip | 400 | ‒ | ‒ | 74.4 | 4.06 | 14.6 | 0.25 | 298 | 0.22 | 0.00179 | High soil organic matter | −0.0066 | [136] |
| Low soil organic matter | 0.0003 | ||||||||||||
| 500 | ‒ | ‒ | 81.7 | 3.10 | 8.76 | 0.22 | 371 | 22.77 | 0.0253 | High soil organic matter | −0.0107 | ||
| Low soil organic matter | −0.0074 | ||||||||||||
| Poultry litter | 400 | ‒ | ‒ | 41.9 | 2.43 | 16.2 | 4.29 | 10 | 4.85 | 0.0269 | High soil organic matter | 0.032 | |
| Low soil organic matter | 0.0257 | ||||||||||||
| 500 | ‒ | ‒ | 44.4 | 1.64 | 12.2 | 4.02 | 11 | 6.55 | 0.0317 | High soil organic matter | 0.0196 | ||
| Low soil organic matter | 0.0094 | ||||||||||||
| Wheat straw | 525 | ‒ | 10 | 69.6 | 2.10 | 7.1 | 1.50 | 46 | 0.6 | ‒ | Slow pyrolysis | 0.0028 | [52] |
| 525 | ‒ | 6.8 | 49.3 | 3.70 | 24.1 | 1.20 | 41 | 1.6 | ‒ | Fast pyrolysis | −0.0206 | ||
| Blue mallee wood | 500 | ‒ | 9.6 | 54.9 | ‒ | ‒ | 1.40 | 39.5 | ‒ | ‒ | Dermosol + Phosphorus | −0.0018 | [114] |
| Tenosol + Phosphorus | 0.0014 | ||||||||||||
| Maize | 350 | ‒ | 8.4 | 72.1 | ‒ | ‒ | 1.7 | 43 | ‒ | ‒ | ‒ | 0.00207 a | [138] |
| 500 | ‒ | 9.8 | 69.1 | ‒ | ‒ | 1.4 | 49 | ‒ | ‒ | ‒ | 0.00173 a | [138] | |
| Maize | 480 | ‒ | 8.6 | 68.1 | 1.5 | ‒ | 0.4 | 164 | ‒ | ‒ | Year 1 | 0.0098 a | [139] |
| 480 | ‒ | 8.6 | 68.1 | 1.5 | ‒ | 0.4 | 164 | ‒ | ‒ | Year 2 | 0.00755 a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pan, S.-Y.; Dong, C.-D.; Su, J.-F.; Wang, P.-Y.; Chen, C.-W.; Chang, J.-S.; Kim, H.; Huang, C.-P.; Hung, C.-M. The Role of Biochar in Regulating the Carbon, Phosphorus, and Nitrogen Cycles Exemplified by Soil Systems. Sustainability 2021, 13, 5612. https://doi.org/10.3390/su13105612
Pan S-Y, Dong C-D, Su J-F, Wang P-Y, Chen C-W, Chang J-S, Kim H, Huang C-P, Hung C-M. The Role of Biochar in Regulating the Carbon, Phosphorus, and Nitrogen Cycles Exemplified by Soil Systems. Sustainability. 2021; 13(10):5612. https://doi.org/10.3390/su13105612
Chicago/Turabian StylePan, Shu-Yuan, Cheng-Di Dong, Jenn-Fang Su, Po-Yen Wang, Chiu-Wen Chen, Jo-Shu Chang, Hyunook Kim, Chin-Pao Huang, and Chang-Mao Hung. 2021. "The Role of Biochar in Regulating the Carbon, Phosphorus, and Nitrogen Cycles Exemplified by Soil Systems" Sustainability 13, no. 10: 5612. https://doi.org/10.3390/su13105612
APA StylePan, S.-Y., Dong, C.-D., Su, J.-F., Wang, P.-Y., Chen, C.-W., Chang, J.-S., Kim, H., Huang, C.-P., & Hung, C.-M. (2021). The Role of Biochar in Regulating the Carbon, Phosphorus, and Nitrogen Cycles Exemplified by Soil Systems. Sustainability, 13(10), 5612. https://doi.org/10.3390/su13105612










