Sustainable Sesame (Sesamum indicum L.) Production through Improved Technology: An Overview of Production, Challenges, and Opportunities in Myanmar
Abstract
1. Introduction
2. Major Constraints on Sustainable Sesame Production
3. Research Achievements through Improved Technologies in Sesame
4. Overview of Myanmar Sesame Production
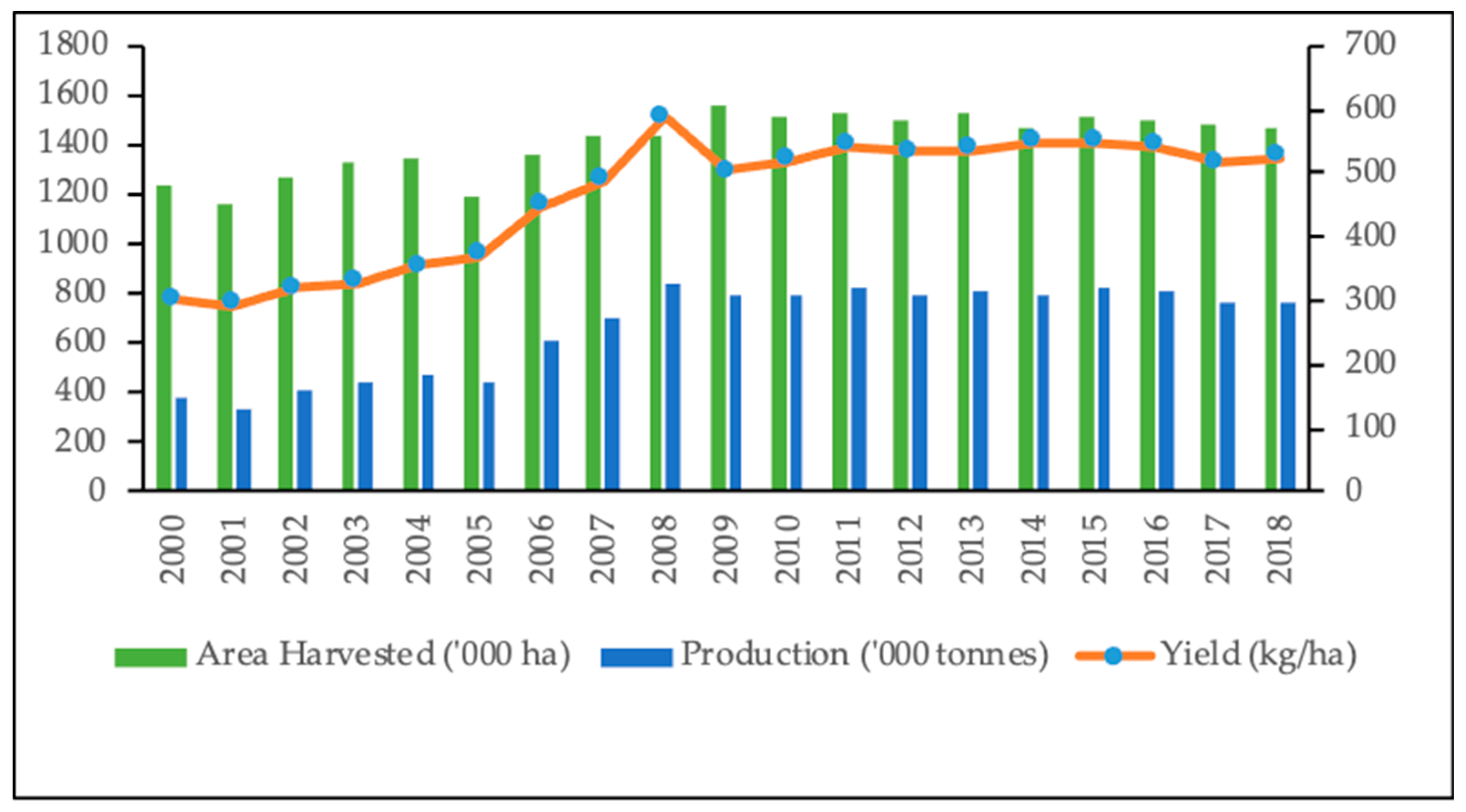
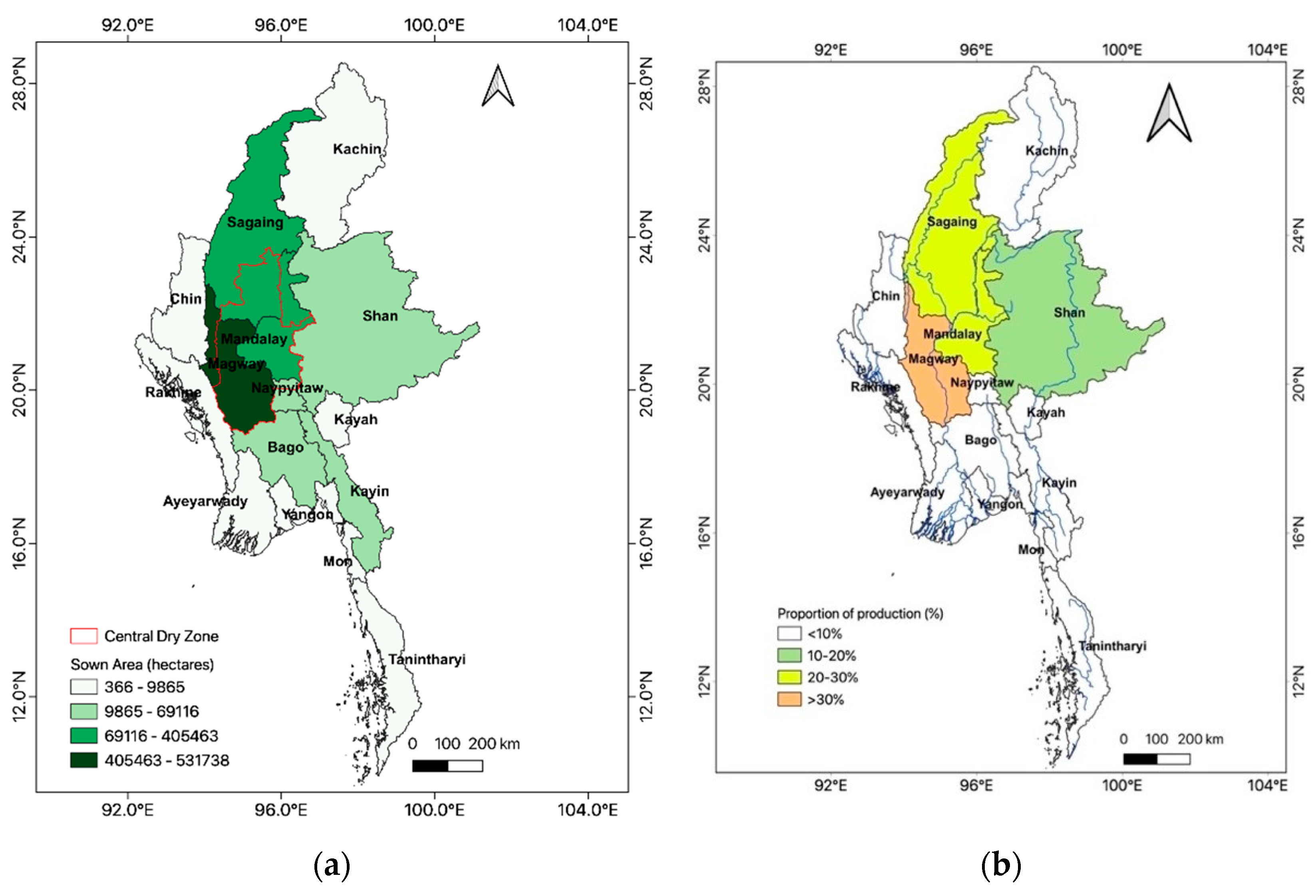
4.1. Past and Present Status of Sesame Production in Myanmar
4.2. Sesame Genetic Resources and Research Activities
4.3. Myanmar’s Sesame Export and Quality
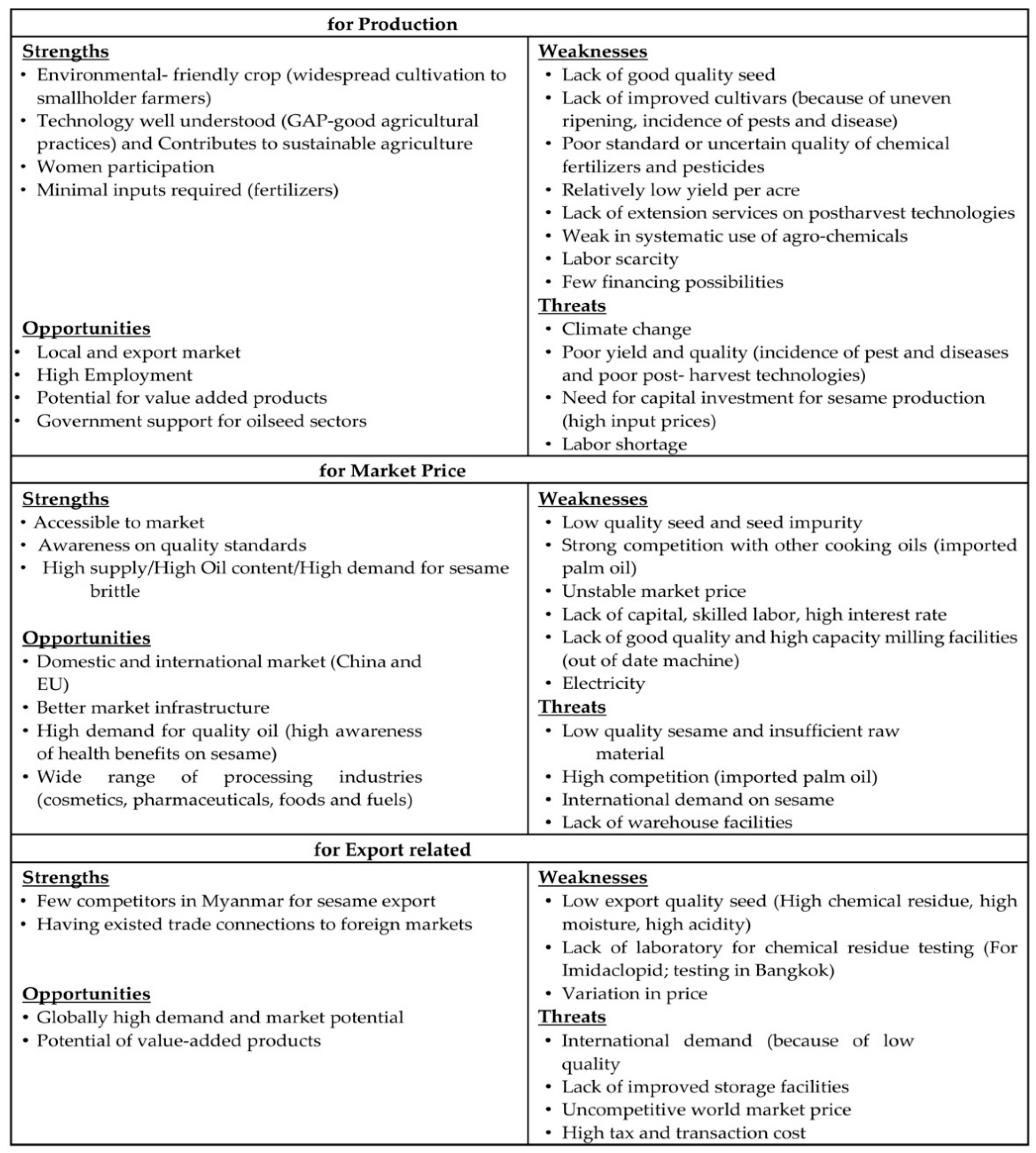
4.4. Constraints and Challenges of Myanmar’s Sesame Production
5. Future Opportunities, Vision, and Strategies Proposed for Enhancing Sesame Production
- Genetic enhancement and conservation of local germplasm
- Institutionalization of quality seed production and distribution mechanisms
- Development of varieties with suitable traits (high oil content, tolerant to drought, waterlogging, phyllody, and sesame black stem rot)
- Development of area-specific production technology packages (integrated crop, soil, and pest management)
- Strong coordination and linkages with research, extension, and private organizations for the effective implementation of scaling up the oilseed production technology
- Improved specialized laboratory testing for residues, enhancing awareness training on postharvest management and pesticides use, farm machinery, and quality assurance systems to meet the international standards.
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Johnson, L.A.; Suleiman, T.M.; Lusas, E.W. Sesame Protein: A Review and Prospectus. J. Am. Oil Chem. Soc. 1979, 56, 463–468. [Google Scholar] [CrossRef] [PubMed]
- Miyake, Y.; Fukumoto, S.; Okada, M.; Sakaida, K.; Nakamura, Y.; Osawa, T. Antioxidative catechol lignans converted from Sesamin and Sesamino lTriglucoside by culturing with Aspergillus. J. Agric. Food Chem. 2005, 53, 22–27. [Google Scholar] [CrossRef] [PubMed]
- Bedigian, D. Sesame: The genus Sesamum, 1st ed.; Taylor & Francis: Boca Raton, FL, USA, 2011; ISBN 978-0-8493-3538-9. [Google Scholar]
- Lyon, C.K. Sesame: Current knowledge of composition and use. J. Am. Oil Chem. Soc. 1972, 49, 245–249. [Google Scholar] [CrossRef]
- Bedigian, D.; Seigler, D.S.; Harlan, J.R. Sesamin, sesamolin and the origin of sesame. Biochem. Syst. Ecol. 1985, 13, 133–139. [Google Scholar] [CrossRef]
- Mordor Intelligence Home Page. Global Sesame Seeds Market-Segmented by Geography-Growth, Trends, and Forescast (2020–2025). Available online: https://www.mordorintellignece.com/industry-reports (accessed on 11 November 2019).
- Morris, J.B. Food, industrial, nutraceutical, and pharmaceutical uses of Sesame genetic resources. In Trends in News Crops and New Uses; Janick, J., Whipkey, A., Eds.; ASHS Press: Alexandria, Egypt, 2002; pp. 153–156. [Google Scholar]
- Agritech. Sesame Seed Consumption Continues to Increase. Available online: https://www.agriorbit.com/sesame-seed-consumption-continues-to-increase/ (accessed on 20 November 2019).
- Food and Agriculturre Organization Statistical Databases (FAOSTAT). Available online: http://faostat.fao.org/ (accessed on 5 February 2020).
- Troncoso-Ponce, M.A.; Kilaru, A.; Cao, X.; Durrett, T.P.; Fan, J.; Jensen, J.K.; Thrower, N.A.; Pauly, M.; Wilkerson, C.; Ohlrogge, J.B. Comparative deep transcriptional profiling of four developing oilseeds: Transcriptional profiling of oilseeds. Plant J. 2011, 68, 1014–1027. [Google Scholar] [CrossRef]
- Bedigian, D.; Harlan, J.R. Evidence for cultivation of sesame in the ancient world. Econ. Bot. 1986, 40, 137–154. [Google Scholar] [CrossRef]
- Boureima, S.; Oukarroum, A.; Diouf, M.; Cisse, N.; Van Damme, P. Screening for drought tolerance in mutant germplasm of sesame (Sesamum indicum L.) probing by chlorophyll a fluorescence. Environ. Exp. Bot. 2012, 81, 37–43. [Google Scholar] [CrossRef]
- Islam, F.; Gill, R.A.; Ali, B.; Farooq, M.A.; Xu, L.; Najeeb, U.; Zhou, W.J.; Mirza, M.Y.; Jilani, G.; Mubashir, A.K.; et al. Sesame. In Technological Innovations in Major World Oil Crops; Gupta, S.K., Ed.; Springer: New York, NY, USA, 2012; Volume 1, pp. 131–145. [Google Scholar]
- Tripathy, S.K.; Kar, J.; Sahu, D. Advances in Sesame (Sesamum indicum L.) Breeding. In Advances in Plant Breeding Strategies: Industrial and Food Crops; Al-Khayri, J.M., Jain, S.M., Johnson, D.V., Eds.; Springer: Cham, Switzerland, 2019; pp. 577–635. [Google Scholar]
- Uçan, K.; Kıllı, F.; Gençoğlan, C.; Merdun, H. Effect of irrigation frequency and amount on water use efficiency and yield of sesame (Sesamum indicum L.) under field conditions. F. Crop. Res. 2007, 101, 249–258. [Google Scholar] [CrossRef]
- Weiss, E.A. Sesame. Oilseed Crops, 2nd ed.; Blackwell Science: London, UK, 2000. [Google Scholar]
- Weiss, E.A. Castor, Sesame and Safflower; Barnes & Noble: New York, NY, USA, 1971. [Google Scholar]
- Langham, D.R.; Wiemers, T. Progress in Mechanizing Sesame in the US through Breeding. In Trends in New Crops and New Uses; Janick, A., Whipkey, A., Eds.; ASHS Press: Alexandria, Egypt, 2002; pp. 157–173. [Google Scholar]
- Gebretsadik, D.; Haji, J.; Tegegne, B. Sesame post-harvest loss from small-scale producers in Kafta Humera District, Ethiopia. J. Dev. Agric. Econ. 2019, 11, 33–42. [Google Scholar]
- National Action Plan for Agriculture (NAPA). Post-Harvest and Agro-Industry, Formulation and Operation of National Action Plan for Poverty Alleviation and Rural Development through Agriculture; Government of the republic of the Union of Myanmar: Yangon, Myanmar, 2016.
- Hickey, L.T.; Hafeez, A.N.; Robinson, H.; Jackson, S.A.; Leal-Bertioli, S.C.M.; Tester, M.; Gao, C.; Godwin, I.D.; Hayes, B.J.; Wulff, B.B.H. Breeding crops to feed 10 billion. Nat. Biotechnol. 2019, 37, 744–754. [Google Scholar] [CrossRef] [PubMed]
- Sebillotte, C.; Messéan, A. Foresight on the Oilseed Competitiveness in the Future. OCL 2002, 10, 3. [Google Scholar]
- Dossa, K.; Diouf, D.; Wang, L.; Wei, X.; Zhang, Y.; Niang, M.; Fonceka, D.; Yu, J.; Mmadi, M.A.; Yehouessi, L.W.; et al. The Emerging Oilseed Crop Sesamum indicum Enters the “Omics” Era. Front. Plant Sci. 2017, 8, 1154. [Google Scholar] [CrossRef] [PubMed]
- Langham, D.G. Genetics of Sesame III: “Open sesame” and mottled lea. J. Heredity 1946, 37, 149–152. [Google Scholar] [CrossRef]
- Stamatov, S.; Velcheva, N.; Deshev, M. Introduced sesame accessions as donors of useful qualities for breeding of Introduced sesame accessions as donors of useful qualities for breeding of mechanized harvesting cultivars. Bulg. J. Agric. Sci. 2018, 24, 820–824. [Google Scholar]
- IPGRI; NBPGR. Report of an International Workshop on Okra Genetic Resources; International Crop Network Series 5; International Plant genetic Resources Institute: Rome, Italy; National Bureau of Indian Plant Genetic Resources: New Delhi, India, 2004; p. 61.
- Zeven, A.C.; Zhukovskiĭ, P.M. Dictionary of Cultivated Plants and Their Centres of Diversity Excluding Ornamentals, Forest Trees, and Lower Plants; Centre for Agricultural Publishing and Documentation: Wageningen, The Netherlands, 1975; ISBN 978-90-220-0549-1. [Google Scholar]
- Hodgkin, T.; Brown, A.H.D.; van Hintum, T.J.; Morales, E. Core Collections of Plant Genetic Resources; IPGRI Technical Bulletin No.3; International Plant Genetic Resources Institute: Rome, Italy, 1995. [Google Scholar]
- Bisht, I.S.; Mahajan, R.K.; Loknathan, T.R.; Agrawal, R.C. Diversity in Indian sesame collection and stratification of germplasm accessions in different diversity groups. Genet. Resour. Crop Evol. 1998, 45, 325–335. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, X.; Che, Z.; Wang, L.; Wei, W.; Li, D. Genetic diversity assessment of sesame core collection in China by phenotype and molecular markers and extraction of a mini-core collection. BMC Genet. 2012, 13, 102. [Google Scholar] [CrossRef]
- Park, J.; Suresh, S.; Raveendar, S.; Baek, H.; Kim, C.; Lee, S. Development and evaluation of core collection using qualitative and quantitative trait descriptor in sesame (Sesamum indicum L.) Germplasm. Korean J. Crop Sci. 2015, 60, 75–84. [Google Scholar] [CrossRef]
- Baskaran, P.J.N. In Vitro mass propagation and diverse callus orientation on Sesamum indicum L.—An important oil plant. J. Agric. Technol. 2006, 2, 259–269. [Google Scholar]
- Jeya Mary, R.; Jayabalan, N. Influence of growth regulators on somatic embryogenesis in sesame. Plant Cell. Tissue Organ Cult. 1997, 49, 67–70. [Google Scholar] [CrossRef]
- Shashidhara, N.; Ravikumar, H.; Ashoka, N.; Santosh, D.T.; Pawa, P.; Lokesha, R.; Janagoudar, B.S. Callus induction and sub-culturing in sesame (Sesamum indium L.). Int. J. Agric. Environ. Biotechnol. 2011, 4, 153–156. [Google Scholar]
- Zimik, M.; Arumugam, N. Induction of shoot regeneration in cotyledon explants of the oilseed crop Sesamum indicum L. J. Genet. Eng. Biotechnol. 2017, 15, 303–308. [Google Scholar] [CrossRef] [PubMed]
- Debnath, A.J.; Gangopadhyay, G.; Basu, D.; Sikdar, S.R. An efficient protocol for in vitro direct shoot organogenesis of Sesamum indicum L. using cotyledon as explant. 3 Biotech 2018, 8, 146. [Google Scholar]
- Burni, T.; Muhammad, A. In Vitro shoots multiplication from nodal explants of Sesame (Sesamum indicum L.). Pure Appl. Biol. 2020, 9, 303–308. [Google Scholar]
- Yadav, M.; Chaudhary, D.; Sainger, M.; Jaiwal, P.K. Agrobacterium tumefaciens-mediated genetic transformation of sesame (Sesamum indicum L.). Plant Cell Tissue Organ Cult. 2010, 103, 377–386. [Google Scholar] [CrossRef]
- Parveen, S.; Mazumder, M.; Bhattacharya, A.; Mukhopadhyay, S.; Saha, U.; Mukherjee, A.; Mondal, B.; Debnath, A.J.; Das, S.; Sikdar, S.; et al. Identification of Anther-Specific Genes from Sesame and Functional Assessment of the Upstream Region of a Tapetum-Specific β-1, 3-glucanase Gene. Plant Mol. Biol. Report. 2017, 36, 149–161. [Google Scholar] [CrossRef]
- Chowdhury, S.; Basu, A.; Kundu, S. A new high-frequency Agrobacterium-mediated transformation technique for Sesamum indicum L. using de-embryonated cotyledon as explant. Protoplasma 2014, 251, 1175–1190. [Google Scholar] [CrossRef]
- Khan, A.W.; Garg, V.; Roorkiwal, M.; Golicz, A.A.; Edwards, D.; Varshney, R.K. Super-Pangenome by integrating the wild side of a species for accelerated crop improvement. Trends Plant Sci. 2020, 25, 148–158. [Google Scholar] [CrossRef]
- Zhang, H.; Miao, H.; Wang, L.; Qu, L.; Liu, H.; Wang, Q.; Yue, M. Genome sequencing of the important oilseed crop Sesamum indicum L. Genome Biol. 2013, 14, 401. [Google Scholar] [CrossRef]
- Wang, L.; Yu, S.; Tong, C.; Zhao, Y.; Liu, Y.; Song, C.; Zhang, Y.; Zhang, X.X.X.; Wang, Y.; Hua, W.; et al. Genome sequencing of the high oil crop sesame provides insight into oil biosynthesis. Genome Biol. 2014, 15, R39. [Google Scholar] [CrossRef]
- Wei, X.; Liu, K.; Zhang, Y.; Feng, Q.; Wang, L.; Zhao, Y.; Li, D.; Zhao, Q.; Zhu, X.X.X.; Zhu, X.X.X.; et al. Genetic discovery for oil production and quality in sesame. Nat. Commun. 2015, 6, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Kitts, P.A.; Church, D.M.; Thibaud-Nissen, F.; Choi, J.; Hem, V.; Sapojnikov, V.; Smith, R.G.; Tatusova, T.; Xiang, C.; Zherikov, A.; et al. Assembly: A resource for assembled genomes at NCBI. Nucleic Acids Res. 2016, 44, D73–D80. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Golicz, A.A.; Lu, K.; Dossa, K.; Zhang, Y.; Chen, J.; Wang, L.; You, J.; Fan, D.; Edwards, D.; et al. Insight into the evolution and functional characteristics of the pan-genome assembly from sesame landraces and modern cultivars. Plant Biotechnol. J. 2019, 17, 881–892. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Wang, L.; Xin, H.; Li, D.; Ma, C.; Ding, X.; Hong, W.; Zhang, X. Construction of a high-density genetic map for sesame based on large scale marker development by specific length amplified fragment (SLAF) sequencing. BMC Plant Biol. 2013, 13, 141. [Google Scholar] [CrossRef]
- Wang, L.; Yu, J.; Li, D.; Zhang, X. Sinbase: An Integrated Database to Study Genomics, Genetics and Comparative Genomics in Sesamum indicum L. Plant Cell Physiol. 2015, 56, e2. [Google Scholar] [CrossRef]
- Yu, J.; Dossa, K.; Wang, L.; Zhang, Y.; Wei, X.; Liao, B.; Zhang, X. PMDBase: A database for studying microsatellite DNA and marker development in plants. Nucleic Acids Res. 2017, 45, D1046–D1053. [Google Scholar] [CrossRef]
- Wei, X.; Gong, H.; Yu, J.; Liu, P.; Wang, L.; Zhang, Y.; Zhang, X. Sesame FG: An integrated database for the functional genomics of sesame. Sci. Rep. 2017, 7, 1–10. [Google Scholar]
- Ke, T.; Yu, J.; Dong, C.; Mao, H.; Hua, W.; Liu, S. ocsESTdb: A database of oil crop seed EST sequences for comparative analysis and investigation of a global metabolic network and oil accumulation metabolism. BMC Plant Biol. 2015, 15, 19. [Google Scholar] [CrossRef]
- Yu, J.; Ke, T.; Tehrim, S.; Sun, F.; Liao, B.; Hua, W. PTGBase: An integrated database to study tandem duplicated genes in plants. Database 2015, 2015, bav017. [Google Scholar] [CrossRef]
- Bhat, K.V.; Babrekar, P.P.; Lakhanpaul, S. Study of genetic diversity in Indian and exotic sesame (Sesamum indicum L.) germplasm using random amplified polymorphic DNA (RAPD) markers. Euphytica 1999, 110, 21–33. [Google Scholar] [CrossRef]
- Laurentin, H.; Karlovsky, P. AFLP fingerprinting of sesame (Sesamum indicum L.) cultivars: Identification, genetic relationship and comparison of AFLP informativeness parameters. Genet. Resour. Crop Evol. 2007, 54, 1437–1446. [Google Scholar] [CrossRef]
- Kim, D.H.; Zur, G.; Danin-Poleg, Y.; Lee, S.W.; Shim, K.B.; Kang, C.W.; Kashi, Y. Genetic relationships of sesame germplasm collection as revealed by inter-simple sequence repeats. Plant Breed. 2002, 121, 259–262. [Google Scholar] [CrossRef]
- Wei, W.; Qi, X.; Wang, L.; Zhang, Y.; Hua, W.; Li, D.; Lv, H.; Zhang, X. Characterization of the sesame (Sesamum indicum L.) global transcriptome using Illumina paired-end sequencing and development of EST-SSR markers. BMC Genom. 2011, 12, 451. [Google Scholar] [CrossRef] [PubMed]
- Badri, J.; Yepuri, V.; Ghanta, A.; Siva, S.; Siddiq, E.A. Development of microsatellite markers in sesame (Sesamum indicum L.). Turkish J. Agric. For. 2014, 38, 603–614. [Google Scholar] [CrossRef]
- Sehr, E.M.; Okello-Anyanga, W.; Hasel-Hohl, K.; Burg, A.; Gaubitzer, S.; Rubaihayo, P.R.; Okori, P.; Vollmann, J.; Gibson, P.; Fluch, S. Assessment of genetic diversity amongst Ugandan sesame (Sesamumindicum L.) landraces based on agromorphological traits and genetic markers. J. Crop Sci. Biotechnol. 2016, 19, 117–124. [Google Scholar] [CrossRef]
- Spandana, B.; Reddy, V.P.; Prasanna, G.J. Development and Characterization of Microsatellite Markers (SSR) in Sesamum (Sesamum indicum L.) Species. Appl. Biochem. Biotechnol. 2012, 168, 1594–1607. [Google Scholar] [CrossRef]
- Wang, L.; Zhang, Y.; Qi, X.; Gao, Y.; Zhang, X. Development and characterization of 59 polymorphic cDNA-SSR markers for the edible oil crop Sesamum indicumL. (pedaliaceae). Am. J. Bot. 2012, 99, 394–398. [Google Scholar] [CrossRef]
- DIXIT, A.; Jin, M.-H.H.; Chung, J.-W.W.; Yu, J.-W.W.; Chung, H.-K.K.; Ma, K.-H.H.; Park, Y.-J.J.; CHO, E.-G.G. Development of polymorphic microsatellite markers in sesame (Sesamum indicum L.). Mol. Ecol. Notes 2005, 5, 736–738. [Google Scholar] [CrossRef]
- Wei, X.; Wang, L.; Zhang, Y.; Qi, X.; Wang, X.; Ding, X.; Zhang, J.; Zhang, X. Development of simple sequence repeat (SSR) markers of sesame (Sesamum indicum L.) from a genome survey. Molecules 2014, 19, 5150–5162. [Google Scholar] [CrossRef]
- Dossa, K. A physical map of important QTLs, functional markers and genes available for sesame breeding programs. Physiol. Mol. Biol. Plants 2016, 22, 613–619. [Google Scholar] [CrossRef]
- Wu, K.; Liu, H.; Yang, M.; Tao, Y.; Ma, H.; Wu, W.; Zuo, Y.; Zhao, Y. High-density genetic map construction and QTLs analysis of grain yield-related traits in Sesame (Sesamum indicum L.) based on RAD-Seq techonology. BMC Plant Biol. 2014, 14, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Xia, Q.; Zhang, Y.; Zhu, X.X.; Zhu, X.X.; Li, D.; Ni, X.; Gao, Y.; Xiang, H.; Wei, X.; et al. Updated sesame genome assembly and fine mapping of plant height and seed coat color QTLs using a new high-density genetic map. BMC Genom. 2016, 17, 1–13. [Google Scholar] [CrossRef]
- Zhang, H.; Miao, H.; Li, C.; Wei, L.; Duan, Y.; Ma, Q.; Kong, J.; Xu, F.; Chang, S. Ultra-dense SNP genetic map construction and identification of SiDt gene controlling the determinate growth habit in Sesamum indicum L. Sci. Rep. 2016, 6, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Uncu, A.O.; Frary, A. High-throughput single nucleotide polymorphism (SNP) identification and mapping in the sesame (Sesamum indicum L.) genome with genotyping by sequencing (GBS) analysis. Mol. Breed. 2016, 36, 173. [Google Scholar] [CrossRef]
- Mei, H.; Liu, Y.; Du, Z.; Wu, K.; Cui, C.; Jiang, X. High-density Genetic map construction and gene Mapping of basal branching habit and flowers per leaf axil in Sesame plant materials and trait investigation. Front. Plant Sci. 2017, 8, 636. [Google Scholar] [CrossRef] [PubMed]
- Wei, X.; Zhu, X.; Yu, J.; Wang, L.; Zhang, Y.; Li, D.; Zhou, R.; Zhang, X. Identification of Sesame Genomic Variations from Genome Comparison of Landrace and Variety. Front. Plant Sci. 2016, 7, 1–12. [Google Scholar] [CrossRef]
- Zhao, Y.; Yang, M.; Wu, K.; Liu, H.; Wu, J.; Liu, K. Characterization and genetic mapping of a novel recessive genic male sterile gene in sesame (Sesamum indicum L.). Mol. Breed. 2013, 32, 901–908. [Google Scholar] [CrossRef]
- Li, C.; Miao, H.; Wei, L.; Zhang, T.; Han, X.; Zhang, H. Association Mapping of Seed Oil and Protein Content in Sesamum indicum L. Using SSR Markers. PLoS ONE 2014, 9, e105757. [Google Scholar] [CrossRef]
- Yan-xin, Z.; Lin-hai, W.; Dong-hua, L.I.; Yuan, G.A.O.; Hai-xia, L.Ü.; Xiu-rong, Z. Mapping of Sesame Waterlogging Tolerance QTL and Identification of Excellent Waterlogging Tolerant Germplasm. Sci. Agric. Sin. 2014, 47, 422–430. [Google Scholar]
- Wang, L.; Li, D.; Zhang, Y.; Gao, Y.; Yu, J.; Wei, X.; Zhang, X. Tolerant and susceptible sesame genotypes reveal waterlogging stress response patterns. PLoS ONE 2016, 11, 1–18. [Google Scholar] [CrossRef]
- Komivi, D.; Mareme, N.; Achille, E.A.; Ndiaga, C.; Diaga, D. Whole genome homology-based identification of candidate genes for drought tolerance in sesame (Sesamum indicum L.). African J. Biotechnol. 2016, 15, 1464–1475. [Google Scholar] [CrossRef]
- Dossa, K.; Diouf, D.; Cissé, N. Genome-Wide investigation of Hsf genes in Sesame reveals their segmental duplication expansion and their active role in drought stress response. Front. Plant Sci. 2016, 7, 1522. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, S.; Basu, A.; Kundu, S. Overexpression of a new Osmotin-Like protein gene (SindOLP) confers tolerance against biotic and abiotic stresses in sesame. Front. Plant Sci. 2017, 8, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Ministry of Agriculture, Livesock and Irrigation (MOALI). Myanmar Agrciulture in Brief; Ministry of Agriculture, Livesock and Irrigation (MOALI): Naypyidaw, Myanmar, 2018.
- Favre, R.; Myint, K. An Analysis of the Myanmar Edible Oil Crops Sub-Sector; Food and Agriculture Organization of the United Nations (FAO): Rome, Italy, 2009. [Google Scholar]
- Tun, T.; Than, M. Myanmar: Country Report to the FAO International Technical Conference on Plant. In Proceedings of the International Technical conference on Plant Genetic Resources, Leipzig, Germany, 17–23 June 1996. [Google Scholar]
- Nyein, S.L. Sesame in Myamar; Department of Agricultural Planning, Ministry of Agriculture and Irrigation: Yangon, Myanmar, 2007; 107p.
- Aye, M.; Khaning, T.T.; Hom, N.H. Morphological characterization and genetic divergence in Myanmar sesame 9Sesamum indicum L.) germplasm. Int. J. Adv. Res. 2018, 6, 297–307. [Google Scholar] [CrossRef]
- Wai, K.P.P. Pycnidium Formation In Vitro of Macrophomina phaseolina (Rhizpctoniabataticola), Causal Agent of Stem and Root Rot of Sesame (Sesamum indicum L.) and Reaction of Some Sesame Varieties to the Disease; Plant Pathology Department, Yezin Agricultural University: Nay Pyi Taw, Myanmar, 2007. [Google Scholar]
- Kyu, N.; Win, K.; Back, C.; Jung, H. Phyllody Phytoplasma infecting Sesame (Sesamum indicumL.) in Myanmar. Trop. Plant Pathol. 2010, 35, 310–313. [Google Scholar]
- Myint, T.; Mon, E.; Kyaw, T. Assessment of supply chain management of sesame seed in Pakokku Township, Magway Region, Myanmar. Int. J. 2019, 6, 215–224. [Google Scholar]
- World Food Program (WFP). Food Security Focus in Dry Zone, Regional Integrated Multi-Harzard Early Warning System (RIMES). In Proceedings of the 9th Monsoon Forum, Nay Pyi Taw, Myanmar, 26 October 2012. [Google Scholar]
- Nyo, H.T.; Htwe, N.N.; Win, K.K.; Myint, T.; Thu, M.K. Current postharvest management of sesame farmers in selected area of Myanmar. Int. J. Environ. Rural Dev. 2018, 9, 76–81. [Google Scholar]
- Linn, T. Value Chain Analysis of Sesame in Magway Township; Mekong Institute, Research Working Paper Series; Human Resources Department of Mekong Institute: KhonKaen, Thailand, 2013. [Google Scholar]
- Min, Y.Y.; Toyota, K. Occurrence of Different Kinds of Diseases in Sesame Cultivation in Myanmar and Their Impact to Sesame Yield. J. Exp. Agric. Int. 2019, 38, 1–9. [Google Scholar] [CrossRef]
- Japan Association for International Collaboration of Agriculture and Forestry (JAICAF). Technical Cooperation Project for Agricultural Productivity and Quality Improvement in Myanmar; Project Report; JAICAF: Akasaka, Japan, 2018. [Google Scholar]
- Tun, K.K.K.; Shrestha, R.; Datta, A.; Using, P.; Sensing, R. Assessment of land degradation and its impact on crop production in the Dry Zone of Myanmar Assessment of land degradation and its impact on crop production in the Dry Zone of Myanmar. Int. J. Sustain. Dev. World Ecol. 2015, 22, 533–544. [Google Scholar] [CrossRef]
- Wijnands, J.H.M.; Biersteker, J.; Hagedoorn, L.F.; Louisse, J. Business Opportunities and Food Safety of the Myanmar Edible Oil Sector; LEI Wageningen UR Research Report: Wageningen, The Netherlands, 2014. [Google Scholar]
- Wang, D.; Zhang, L.; Huang, X.; Wang, X.; Yang, R.; Mao, J.; Wang, X.; Wang, X.; Zhang, Q.; Li, P. Identification of Nutritional Components in Black Sesame Determined by Widely Targeted Metabolomics and Traditional Chinese Medicines. Molecules 2018, 23, 1180. [Google Scholar] [CrossRef]
- Ortiz, R.; Geleta, M.; Gustafsson, C.; Lager, I.; Hofvander, P.; Löfstedt, C.; Cahoon, E.B.; Minina, E.; Bozhkov, P.; Stymne, S.; et al. ScienceDirect Oil crops for the future. Curr. Opin. Plant Biol. 2020, 1–9. [Google Scholar] [CrossRef]
- Pandey, B.; Ragit, S.S.; Kumar, S. Optimization of biodiesel production from Sesamum indicum L. oil by Taguchi’s Technique. Int. J. Appl. Agric. Res. 2017, 12, 255–265. [Google Scholar]
- Barati, M. Catalytic Supercritical Process for Biodiesel Production from Sesame Oil; International Congress and Exhibition of Sciences and Innovative Technologies; Babol Noshirvani University of Technology: Babol, Iran, 2018. [Google Scholar]
- Eryilmaz, T.; Yesilyurt, M.K.; Cesur, C.; Gokdogan, O. Biodiesel production potential from oil seeds in Turkey. Renew. Sustain. Energy Rev. 2016, 58, 842–851. [Google Scholar] [CrossRef]
- Habibullah, M.; Masjuki, H.H.; Kalam, M.A.; Rahman, S.M.A.; Mofijur, M.; Mobarak, H.M.; Ashraful, A.M. Potential of biodiesel as a renewable energy source in Bangladesh. Renew. Sustain. Energy Rev. 2015, 50, 819–834. [Google Scholar] [CrossRef]
- Dawodu, F.A.; Ayodele, O.O.; Bolanle-Ojo, T. Biodiesel production from Sesamum indicum L. seed oil: An optimization study. Egypt. J. Pet. 2014, 23, 191–199. [Google Scholar] [CrossRef]
- Mujtaba, M.A.; Muk Cho, H.; Masjuki, H.H.; Kalam, M.A.; Ong, H.C.; Gul, M.; Harith, M.H.; Yusoff, M.N.A.M. Critical review on sesame seed oil and its methyl ester on cold flow and oxidation stability. Energy Rep. 2020, 6, 40–54. [Google Scholar] [CrossRef]
- Myanmar Agriculture Development Strategy and Investment Plan (2018-19 ~ 2022-23); Agriculture, Livestock and Irrigation: Nay Pyi Taw, Myanmar, 2018.
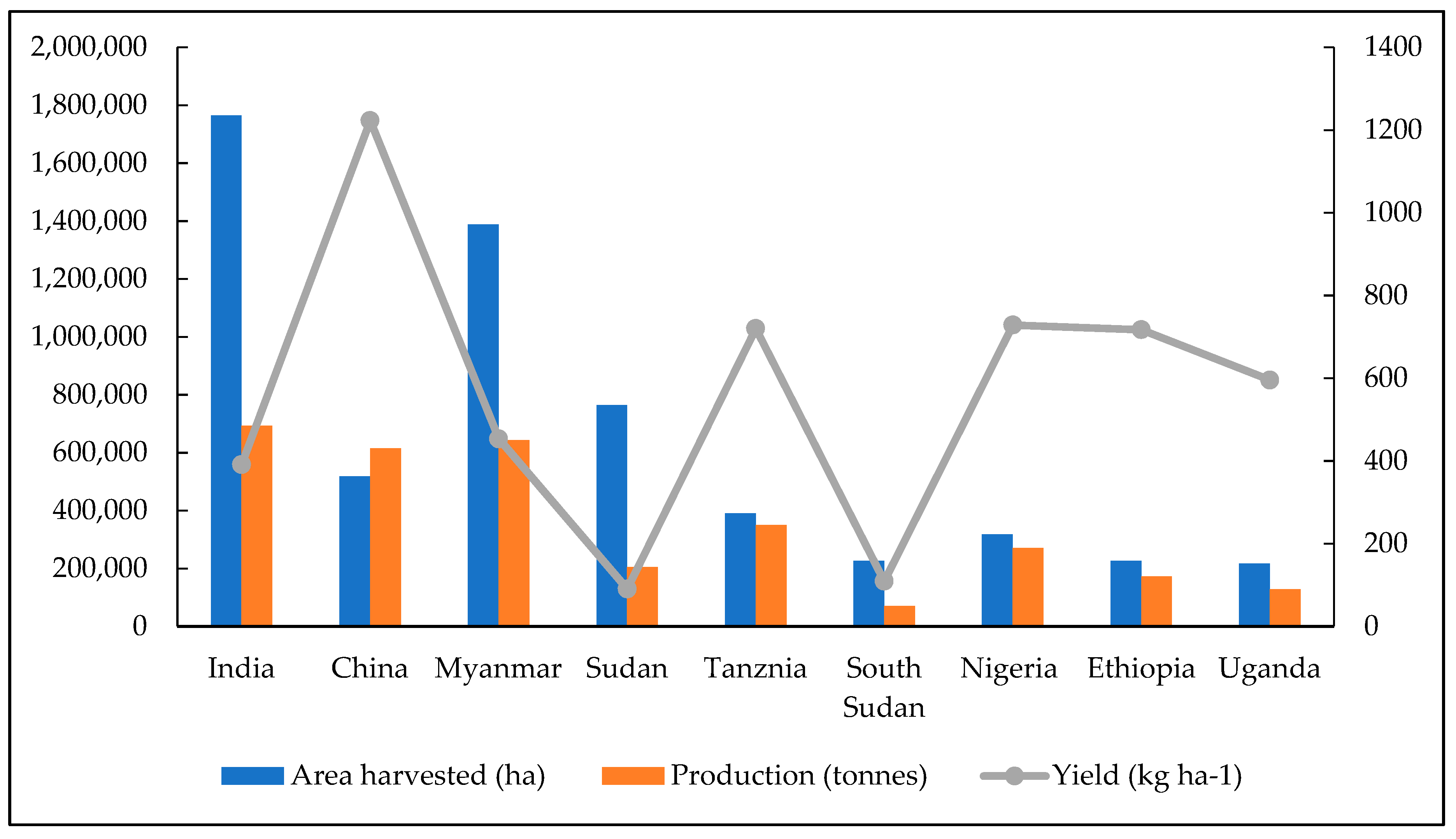

| Area (‘000 ha) | Yield (kgha−1) | Production (‘000 MTha−1) | % of World Production | |
|---|---|---|---|---|
| Top producing countries | ||||
| India | 1730 | 431 | 746 | 12.40 |
| China | 311 | 1393 | 433 | 7.20 |
| Myanmar | 1463 | 525 | 769 | 12.78 |
| Sudan | 3480 | 282 | 981 | 9.33 |
| Tanzania | 800 | 701 | 561 | 14.56 |
| South Sudan | 618 | 334 | 207 | 3.43 |
| Nigeria | 539 | 1063 | 573 | 9.52 |
| Ethiopia | 415 | 726 | 301 | 5.01 |
| Uganda | 210 | 667 | 140 | 2.33 |
| Regions | ||||
| Asia | 3906 | 578 | 2257 | 37.52 |
| Africa | 7549 | 474 | 3575 | 59.42 |
| America | 288 | 636 | 183 | 3.04 |
| Europe | 1 | 975 | 1 | 0.02 |
| World | 11,743 | 512 | 6016 | - |
| Database Name | Website | Ref. |
|---|---|---|
| The sesame genome project | http://www.sesamegenome.org | [47] |
| Sinbase | http://www.ocri-genomics.org/Sinbase/index.html | [48] |
| SesameHapMap | http://202.127.18.228/SesameHapMap/ | [44] |
| PMDBase | http://www.sesame-bioinfo.org/PMDBase | [49] |
| SesameFG | http://www.ncgr.ac.cn/SesameFG | [50] |
| Sesame Germplasm Resource Information Database | http://www.sesame-bioinfo.org/phenotype/index.html | - |
| ocsESTdb | http://www.ocri-genomics.org/ocsESTdb/index.html | [51] |
| PTGBase | http://www.ocri-genomics.org/PTGBase/index.html | [52] |
| SisatBase | http://www.sesame-bioinfo.org/SisatBase/ | - |
| Traits | QTLs/Genes | Markers Type | Marker Numbers | Mapping Population | Parent of the Cross | Ref. |
|---|---|---|---|---|---|---|
| Production enhancement | ||||||
| Grain yield | Qgn-1, Qgn-6, | SLAF | 9378 | 150 BC1 | Yuzhi 4 × Bengal Small-seed | [68] [47] [65] |
| Grain number per capsule | Qgn-12 | |||||
| Thousand grain weight | Qtgw-11 | |||||
| Seed coat color | QTL-1, QTL11-1, QTL11-2, QTL13-1 | |||||
| Seed coat color | qSCa-8.2, qSCb-4.1, qSCb-8.1, qSCb-11.1, qSCl-4.1, qSCl-8.1, qSCl-11.1, qSCa-4.1 and qSCa-8.1 | SLAF SNP | 1233 | 107 F2 430 Recombinant inbred lines (RILS, F8) | Zhongzhi No.13 ×Jiaxiang Sesame Zhongzhi No.13 × Semi-dwarf ZZM 2748 | |
| Seed coat color | SiPPO (SIN_1016759) | SSR | 400 | 500 RILs (F6) | Zhongzhi 13 × Mishuozhima | [69] |
| Plant height | Qph-6 and Qph-12 | SNP | 1,800,000 | 705 worldwide accessions | [44] | |
| Semi-dwarf sesame plant phenotype | QTL (qPH-3.3), Gene [SiGA20ox1(SIN_1002659)] | SNP SSR | 400 | 430 RILS (F8) 500 RILs (F6) | Zhongzhi No.13 × Semi-dwarf ZZM 2748 Zhongzhi 13 × Mishuozhima | [65] [69] |
| Plant height | SiDFL1 (SIN_1014512) andSiILR1 (SIN_1018135) | SNP | 1,800,000 | 705 worldwide accessions | [44] | |
| Capsule number per plant | Qcn-11 | SNP SSR InDels | 1190 22 18 | 224 (RIL), F8:9 | Miaoqianzhima × Zhongzhi 14 | [64] |
| First capsule height | Qfch-4, Qfch-11, and Qfch-12 | |||||
| Capsule axis length | Qcal-5 and Qcal-9 | |||||
| Capsule length | Qcl-3, Qcl-4, Qcl-7, Qcl-8, and Qcl-12 | |||||
| Number of capsules per axil | SiACS (SIN_1006338) | SNP | 1,800,000 | 705 worldwide accessions | [44] | |
| Mono flower vs. triple flower | SiFA | SLAF (Marker58311, Marker34507, Marker36337) | 9378 | 150 BC1 | Yuzhi-4 × Bengal Small-seed | [68] |
| Flowering time | SiDOG1 (SIN_1022538) and SiIAA14 (SIN_1021838) | SNP | 705 sesame accessions | [44] | ||
| Determinate trait in sesame | gene SiDt (DS899s00170.023) | SNP | 30,193 | 120 F2 | Yuzhi 11 (indeterminate, Dt) × Yuzhi DS 899 (determinate dt1) | [66] |
| Branching habit | SiBH | SLAF (Marker129539, Marker41538, Marker31462) | 9378 | 150 BC1 | Yuzhi-4 × Bengal Small- seed | [68] |
| Recessive GMS | recessive GMS geneSiMs1 | AFLP markers P01MC08, P06MG04, P12EA14 | 237 NILs (Near-Isogenic Lines) | Sib mating between 95ms-5A and 95ms-5B | [70] | |
| dominant GMS geneMs | SBM298 and GB50 | SSR | 1500 | Noval GMS line W1098A (Backcrossing and sib-mating; BC2F6 | Conventional variety Ezhi 1 × wild germplasm Yezhi2 | [71] |
| Stress related | ||||||
| Water logging tolerance | qEZ09ZCL13, qWH09CHL15, qEZ10ZCL07, qWH10ZCL09, qEZ10CHL07, and qWH10CHL09 | SSR (ZM428) closely linked toqWH10CHL09 | 113 | 206 RIL F6 | Zhongzhi No.13 (high tolerance to waterlogging) × Yiyangbai (extremely sensitive to waterlogging) | [72,73] |
| Drought tolerance | TFs (Transcription Factors) families (AP2/ERF and HSF) | - | - | - | - | [74,75] |
| Drought, salinity, oxidative stresses, charcoal rot | Osmotin-like gene (SindOLP) | - | - | - | - | [76] |
| Gene for Oil traits | ||||||
| Sesamin production | SiDIR (SIN_1015471), SiPSS (SIN_1025734) | SNP | 1,800,000 | 705 worldwide accessions | [44] | |
| Oil content | SIN_1003248, SIN_1013005, SIN_1019167, SIN_1009923 SiPPO (SIN_1016759) SiNST1 (SIN_1005755) | |||||
| Fatty Acid composition | SiKASI (SIN_1001803), SiKASII (SIN_1024652), SiACNA (SIN_1005440), SiDGAT2 (SIN_1019256), SiFATA (SIN_1024296), SiFATB (SIN_1022133), SiSAD (SIN_1008977), SiFAD2 (SIN_1009785) | |||||
| Sesamin and sesamolin content | SiNST1 (SIN_1005755) | |||||
| Protein content | SiPPO (SIN_1016759) | |||||
| Years | Sesame Seed | Sesame Oil | ||
|---|---|---|---|---|
| Export Quantity (tons) | Export Value (US$) | Export Quantity (tons) | Export Value (US$) | |
| 2010 | 53,700 | 53,348 | 800 | 450 |
| 2011 | 36,500 | 30,000 | 800 | 470 |
| 2012 | 38,200 | 37,500 | 800 | 480 |
| 2013 | 33,300 | 32,000 | 800 | 500 |
| 2014 | 25,679 | 44,220 | 161 | 631 |
| 2015 | 19,814 | 31,944 | 65 | 292 |
| 2016 | 24,506 | 38,410 | 44 | 190 |
| 2017 | 33,500 | 47,447 | 13 | 88 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Myint, D.; Gilani, S.A.; Kawase, M.; Watanabe, K.N. Sustainable Sesame (Sesamum indicum L.) Production through Improved Technology: An Overview of Production, Challenges, and Opportunities in Myanmar. Sustainability 2020, 12, 3515. https://doi.org/10.3390/su12093515
Myint D, Gilani SA, Kawase M, Watanabe KN. Sustainable Sesame (Sesamum indicum L.) Production through Improved Technology: An Overview of Production, Challenges, and Opportunities in Myanmar. Sustainability. 2020; 12(9):3515. https://doi.org/10.3390/su12093515
Chicago/Turabian StyleMyint, Daisy, Syed A. Gilani, Makoto Kawase, and Kazuo N. Watanabe. 2020. "Sustainable Sesame (Sesamum indicum L.) Production through Improved Technology: An Overview of Production, Challenges, and Opportunities in Myanmar" Sustainability 12, no. 9: 3515. https://doi.org/10.3390/su12093515
APA StyleMyint, D., Gilani, S. A., Kawase, M., & Watanabe, K. N. (2020). Sustainable Sesame (Sesamum indicum L.) Production through Improved Technology: An Overview of Production, Challenges, and Opportunities in Myanmar. Sustainability, 12(9), 3515. https://doi.org/10.3390/su12093515





