What Is the Threshold Carbonization Temperature for Sustainable Preservation of the Good Nitrogen Supply Ability of Chicken Manure?
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Source and Preparation
2.2. Physical and Chemical Properties of the Samples
2.3. Surface Functional Group Analysis
2.4. Nitrogen Forms and Speciation
2.5. Measurement of Nitrogen Supply Ability
2.6. Statistical Data Analysis
3. Results
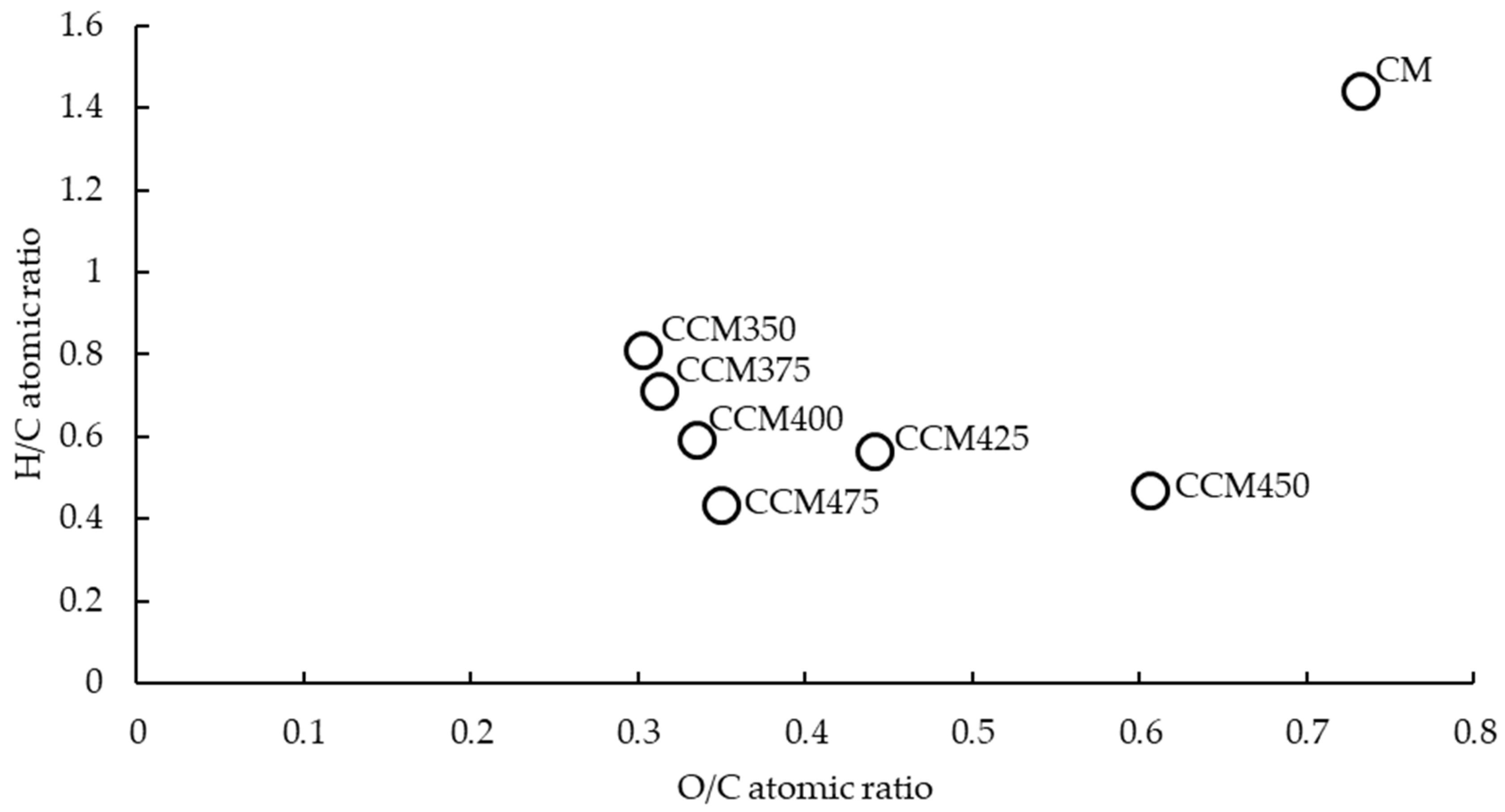
3.1. Basic Chemical Properties and Elemental Composition
3.2. Surface Functional Groups
- Decreased intensity, shifting, and subsequent loss of the broad band attributed to free amino acids mixed with O-H groups from 3500 to 2500 cm−1.
- Decreased C–H band with increasing temperature of carbonization.
- Decreased C=O band on the surface of CM with increasing temperature.
- Increase in CH2 units in biopolymer on the surface of CM with temperature increase.
- Increase in the C–O band with increasing temperature up to 450 °C.
- Increase in the aromatic C–H band with increasing carbonization temperature
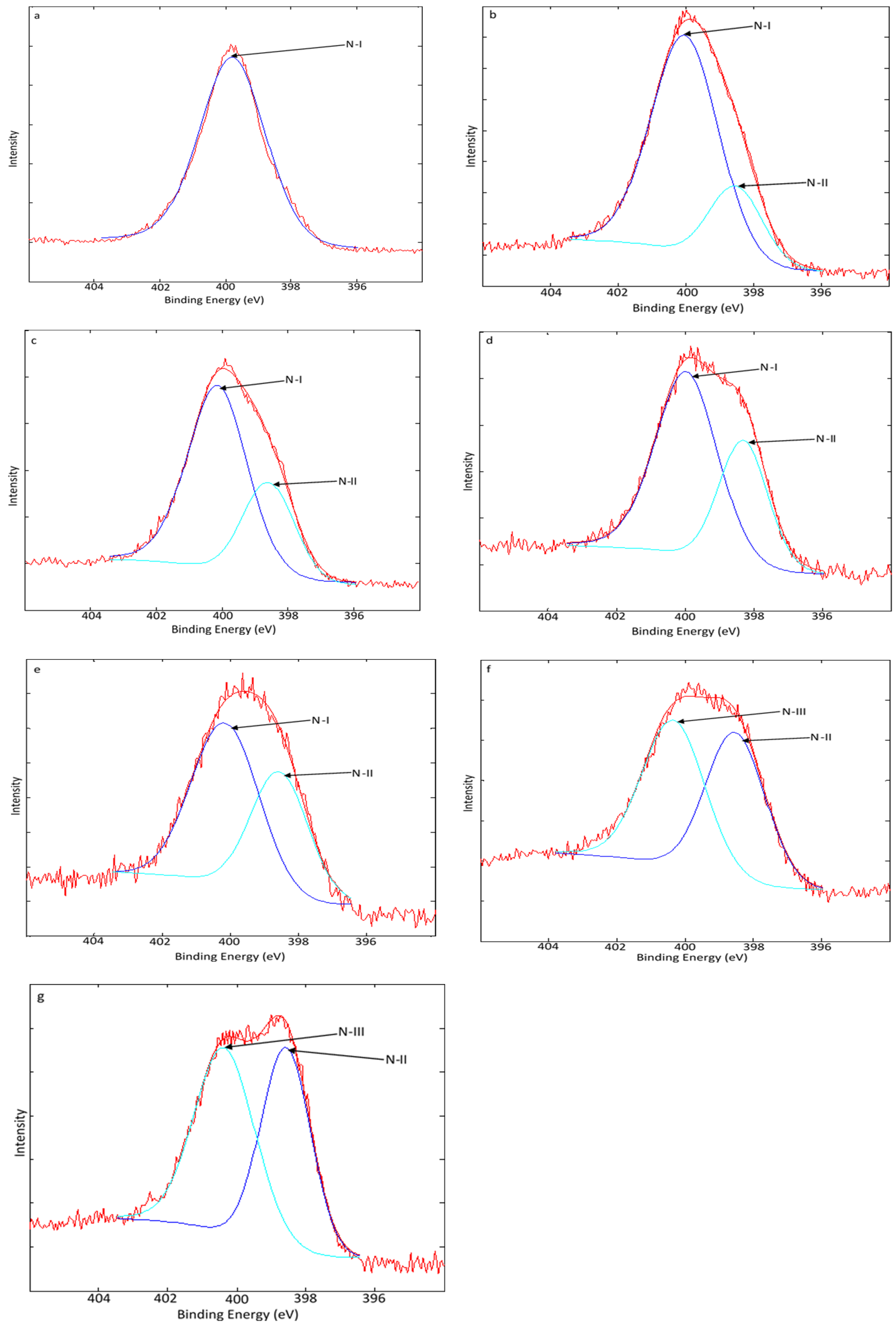
3.3. Transformation of N Species in CM During Carbonization
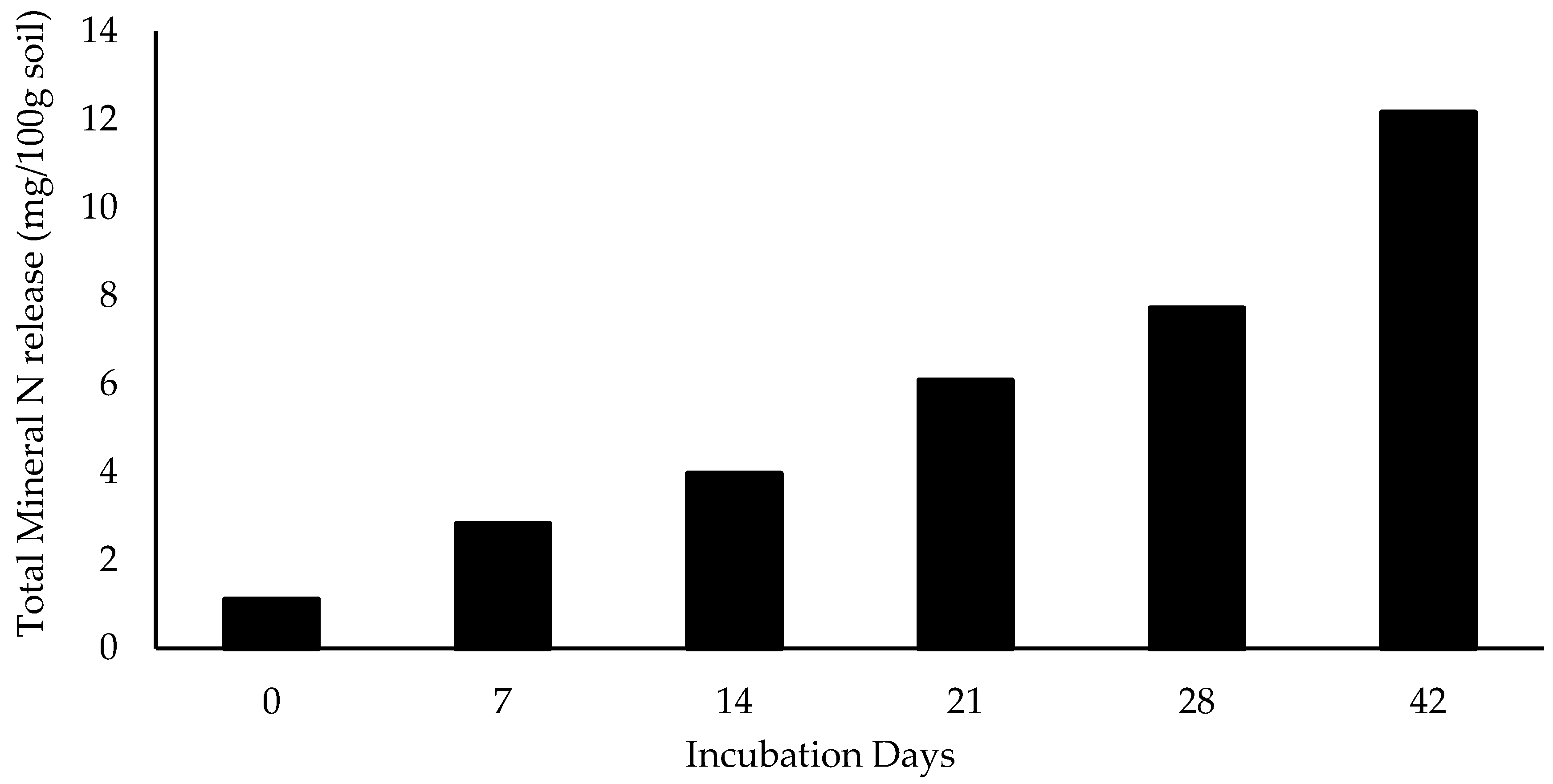
3.4. N Fertilizer Capacity of CM and CM Carbonized at Different Temperatures Over Time
3.5. Regression Analysis Between Total Mineral N Released over the 42 Days and Compositional Makeup of the CM and CCM Produced at Different Temperatures
4. Discussion
4.1. Alteration of the Chemical Properties of CM After Carbonization at Different Temperatures
4.2. Changes on the Surface Functional Group of CM After Carbonization at Different Temperatures
4.3. N Speciation Pattern During Carbonization of CM at Different Temperatures
4.4. Relationship Between Total Mineral N Release During the 42 Day Incubation Study and Compositional Makeup of CM and Carbonized CM Produced at Different Temperatures
4.5. Connection Between the Chemical Alteration and N Supply Ability of CM During Carbonization at Different Temperatures
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Parameters | Amount of Nutrient |
|---|---|
| Total N (%) | 0.1 |
| Total C (%) | 1.1 |
| C/N ratio | 9.4 |
| NH4+-N (mg/100 g) | 0.82 |
| NO3-N (mg/100 g) | 1.84 |
| Organic N (%) | 0.11 |
| pH | 6.7 |
Appendix B
References
- FAO. Agribusiness Handbook: Poultry Meat and Eggs, 1st ed.; FAO Investment Centre Division: Rome, Italy, 2010; p. 75. [Google Scholar]
- Risse, L.M.; Cabrera, M.L.; Franzluebbers, A.J.; Gaskin, J.W.; Gilley, J.E.; John, E.; Killorn, R.; Radcliffe, D.E.; Tollner, W.E.; Zhang, H. Land application of manure for beneficial reuse. In Animal Agriculture and the Environment: National Center for Manure and Animal Waste Management White Papers; American Society of Agricultural and Biological Engineers: St. Joseph, MI, USA, 2006; Available online: https://digitalcommons.unl.edu/biosysengfacpub (accessed on 18 October 2019).
- Chan, K.Y.; Zwieten, L.V.; Meszaros, I.; Downie, A.; Joseph, S. Using poultry litter biochars as soil amendments. Aust. J. Soil Res. 2008, 46, 437–444. [Google Scholar] [CrossRef]
- Cantrell, K.B.; Hunt, P.G.; Uchimiya, M.; Novak, J.M.; Ro, K.S. Impact of pyrolysis temperature and manure source on physicochemical characteristics of biochar. Bioresour. Technol. 2012, 107, 419–428. [Google Scholar] [CrossRef] [PubMed]
- Popov, V.; Itoh, H.; Brebbia, C.A.; Kungoles, A. Waste Management and the Environment, 2nd ed.; WIT Press: Ashurst Lodge Southampton, UK, 2004; p. 696. [Google Scholar]
- Shinogi, Y.; Yoshida, H.; Koizumi, T.; Yamaoka, M.; Saito, T. Basic characteristics of low temperature carbon products from waste sludge. Adv. Environ. Res. 2003, 7, 661–665. [Google Scholar] [CrossRef]
- Cantrell, K.; Ro, K.; Mahajan, D.; Anjom, M.; Hunt, P.G. Role of thermochemical conversion in livestock waste-to-energy treatments: Obstacles and opportunities. Ind. Eng. Chem. Res. 2007, 46, 8918–8927. [Google Scholar] [CrossRef]
- Song, W.; Guo, M. Quality variations of poultry litter biochar generated at different pyrolysis temperatures. J. Anal. Appl. Pyrolysis 2012, 94, 138–145. [Google Scholar] [CrossRef]
- Rehrah, D.; Reddy, M.R.; Novak, J.M.; Bansode, R.R.; Schimmel, K.A.; Yu, J.; Watts, D.W.; Ahmedn, M. Production and characterization of biochars from agricultural by-products for use in soil quality enhancement. J. Anal. Appl. Pyrolysis 2014, 108, 301–309. [Google Scholar] [CrossRef]
- Tagoe, S.O.; Horiuchi, T.; Matsui, T. Effects of carbonized and dried chicken manures on the growth, yield, and N content of soybean. Plant. Soil 2008, 306, 211–220. [Google Scholar] [CrossRef]
- Ishimori, T.; Takahashi, Y.; Sato, H.; Hassan, A.; Iwamoto, Y.; Pandian, G.N.; Hori, H. Low temperature carbonization of chicken manure to char and its effect on growth of Oryza sativa L. Koshihikari and Brassica rapa komatsuna. Euro-Mediterr J. Environ. Integr. 2017, 2, 10. [Google Scholar] [CrossRef]
- Steiner, C.; Harris, K.; Gaskin, J.; Das, K.C. The nitrogen contained in carbonized poultry litter is not plant available. Open Agric. 2018, 3, 284–290. [Google Scholar] [CrossRef]
- Clough, T.; Condron, L.; Kammann, C.; Müller, C. A review of biochar and soil nitrogen dynamics. Agronomy 2013, 3, 275–293. [Google Scholar] [CrossRef]
- Knicker, H. “Black nitrogen”—An important fraction in determining the recalcitrance of charcoal. Org. Geochem. 2010, 41, 947–950. [Google Scholar] [CrossRef]
- Almendros, G.; Knicker, H.; Gonzalez-Vila, F.J. Rearrangement of carbon and nitrogen forms in peat after progressive thermal oxidation as determined by solid-state 13C- and 15N-NMR spectroscopy. Org. Geochem. 2003, 34, 1559–1568. [Google Scholar] [CrossRef]
- Paneque, M.; Knicker, H.; Kern, J.; De la Rosa, J.M. Hydrothermal Carbonization and Pyrolysis of Sewage Sludge: Effects on Lolium perenne Germination and Growth. Agronomy 2019, 9, 363. [Google Scholar] [CrossRef]
- Bruun, S.; Harmer, S.L.; Bekiaris, G.; Christel, W.; Zuin, L.; Hu, Y.; Jensen, L.S.; Lombi, E. The effect of different pyrolysis temperatures on the speciation and availability in soil of P in biochar produced from the solid fraction of manure. Chemosphere 2017, 169, 377–386. [Google Scholar] [CrossRef]
- Singh, B.; Singh, B.P.; Cowie, A.L. Characterisation and evaluation of biochars for their application as a soil amendment. Aust. J. Soil Res. 2010, 48, 516–525. [Google Scholar] [CrossRef]
- Pascual, J.A.; Garcia, C.; Hernendez, T. Comparison of fresh and composted organic waste in their efficacy for the improvement of arid soil quality. Bioresour. Technol. 1999, 68, 255–264. [Google Scholar] [CrossRef]
- Cataldo, D.A.; Haroon, M.; Schrader, L.E.; Youngs, V.L. Rapid colorimetric determination of nitrate in plant tissue by nitration of salicylic acid. Commun. Soil Sci. Plant. Anal. 1975, 6, 71–80. [Google Scholar] [CrossRef]
- Logah, V.; Safo, E.Y.; Quansah, C. Evaluation of Nitrogen Mineralization Dynamics Following Amendments Application under Cropping Systems on a Ferric Acrisol in Ghana. Int. J. Environ. Sci. Dev. 2011, 2, 133–137. [Google Scholar] [CrossRef]
- De la Rosa, J.M.; Paneque, M.; Miller, A.Z.; Knicker, H. Relating physical and chemical properties of four different biochars and their application rate to biomass production of Lolium perenne on a Calcic Cambisol during a pot experiment of 79 days. Sci. Total Environ. 2014, 499, 175–184. [Google Scholar] [CrossRef]
- Bernier, M.H.; Levy, G.J.; Fine, P.; Borisover, M. Organic matter composition in soils irrigated with treated wastewater: FT-IR spectroscopic analysis of bulk soil samples. Geoderma 2013, 209–210, 233–240. [Google Scholar] [CrossRef]
- Paneque, M.; De la Rosa, J.M.; Kern, J.; Reza, M.T.; Knicker, H. Hydrothermal carbonization and pyrolysis of sewage sludges: What happen to carbon and nitrogen? J. Anal. Appl. Pyrolysis 2017, 128, 314–323. [Google Scholar] [CrossRef]
- Smidt, E.; Parravicini, V. Effect of sewage sludge treatment and additional aerobic post-stabilization revealed by infrared spectroscopy and multivariate data analysis. Bioresour. Technol. 2009, 100, 1775–1780. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.; Johnson, E.J.; Chefetz, B.; Zhu, L.; Xing, B. Sorption of polar and nonpolar aromatic organic contaminants by plant cuticular materials: The role of polarity and accessibility. Environ. Technol. 2005, 39, 6138–6146. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.; Wang, Y.; Hu, D. Biosorption and biodegradation of polycyclic aromatic hydrocarbons in aqueous solutions by a consortium of white rot fungi. J. Hazard. Mater. 2010, 179, 845–851. [Google Scholar] [CrossRef] [PubMed]
- Hossain, M.K.; Strezov, V.; Chan, K.Y.; Ziolkowski, A.; Nelson, P.F. Influence of pyrolysis temperature on production and nutrient properties of wastewater sludge biochar. J. Environ. Manag. 2011, 92, 223–228. [Google Scholar] [CrossRef] [PubMed]
- Wei, L.; Wen, L.; Yang, T.; Zhang, N. Nitrogen Transformation during Sewage Sludge pyrolysis. Energ. Fuels 2015, 29, 5088–5094. [Google Scholar] [CrossRef]
- Wang, Z.; Li, Q.; Lin, Z.; Whiddon, R.; Qiu, K.; Kuang, M.; Cen, K. Transformation of nitrogen and sulphur impurities during hydrothermal upgrading of low quality coals. Fuel 2016, 164, 254–261. [Google Scholar] [CrossRef]
- Wojtowicz, M.A.; Pels, J.R.; Moulijn, J.A. The fate of nitrogen functionalities in coal during pyrolysis and combustion. Fuel 1995, 74, 507–516. [Google Scholar] [CrossRef]
- Jindo, K.; Mizumoto, H.; Sawada, Y.; Sanchez-Monedero, M.A.; Sonoki, T. Physical and chemical characterization of biochars derived from different agricultural residues. Biogeosciences 2014, 11, 6613–6621. [Google Scholar] [CrossRef]
- Wang, Y.; Lin, Y.; Chiu, P.C.; Imhoff, P.T.; Guo, M. Phosphorus release behaviors of poultry litter biochar as a soil amendment. Sci. Total Environ. 2015, 512–513, 454–463. [Google Scholar] [CrossRef]
- Hadroug, S.; Jellali, S.; Leahy, J.J.; Kwapinska, M.; Jeguirim, M.; Hamdi, H.; Kwapinski, W. Pyrolysis Process as a Sustainable Management Option of Poultry Manure: Characterization of the Derived Biochars and Assessment of their Nutrient Release Capacities. Water 2019, 11, 2271. [Google Scholar] [CrossRef]
- Zornoza, R.; Moreno-Barriga, F.; Acosta, J.A.; Munoz, M.A.; Faz, A. Stability, nutrient availability and hydrophobicity of biochars derived from manure, crop residues, and municipal solid waste for their use as soil amendments. Chemosphere 2016, 144, 122–130. [Google Scholar] [CrossRef] [PubMed]
- Bavariani, M.Z.; Ronaghi, A.; Ghasemi, R. Influence of pyrolysis temperatures on FTIR analysis, nutrient bioavailability, and agricultural use of poultry manure biochars. Commun. Soil Sci. Plant Anal. 2019, 50, 402–411. [Google Scholar] [CrossRef]
- Cao, W.; Cao, C.; Guo, L.; Jin, H.; Dargusch, M.; Bernhardt, D.; Yao, X. Hydrogen production from supercritical water gasification of chicken manure. Int. J. Hydrog. Energy 2016, 41, 22722–22731. [Google Scholar] [CrossRef]
- Sun, K.; Qiu, M.; Han, L.; Jin, J.; Wang, Z.; Pan, Z.; Xing, B. Speciation of phosphorus in plant- and manure-derived biochars and its dissolution under various aqueous conditions. Sci. Total Environ. 2018, 634, 1300–1307. [Google Scholar] [CrossRef]
- Xiao, R.; Wang, J.J.; Gaston, L.A.; Zhou, B.; Park, J.H.; Li, R.; Dodla, S.K.; Zhang, Z. Biochar produced from mineral salt-impregnated chicken manure: Fertility properties and potential for carbon sequestration. Waste Manag. 2018, 78, 802–810. [Google Scholar] [CrossRef]
- Jin, J.; Li, Y.; Zhang, J.; Wu, S.; Cao, Y.; Liang, P.; Zhang, J.; Wong, M.H.; Wang, M.; Shan, S. Influence of pyrolysis temperature on properties and environmental safety of heavy metals in biochars derived from municipal sewage sludge. J. Hazard. Mater. 2016, 320, 417–426. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, J.; Sheng, C.; Chen, J.; Liu, Y.; Zhao, L.; Xie, F. X-ray Photoelectron Spectroscopy (XPS) Investigation of Nitrogen Functionalities during Coal Char Combustion in O2/CO2 and O2/Ar Atmospheres. Energy Fuels 2011, 25, 240–245. [Google Scholar] [CrossRef]
- Graff, R.A.; Brandes, S.D. Modification of coal by subcritical steam: Pyrolysis and extraction yields. Energy Fuels 1987, 1, 84–88. [Google Scholar] [CrossRef]
- Mubarak, A.R.; Gali, E.A.M.; Mohamed, A.G.; Steffens, D.; Awadelkarim, A.H. Nitrogen Mineralization from Five Manures as Influenced by Chemical Composition and Soil Type. Commun Soil Plan. 2010, 41, 1903–1920. [Google Scholar] [CrossRef]
- Baldocka, J.A.; Smernik, R.J. Chemical composition and bioavailability of thermally altered Pinus resinosa (Red pine) wood. Org. Geochem. 2002, 33, 1093–1109. [Google Scholar] [CrossRef]
- Li, L.; Li, S. Nitrogen Mineralization from Animal Manures and Its Relation to Organic N Fractions. J. Integr. Agric. 2014, 13, 2040–2048. [Google Scholar] [CrossRef]
- Zheng, H.; Wang, Z.; Deng, X.; Zhao, J.; Luo, Y.; Novak, J.; Herbert, S.; Xing, B. Characteristics and nutrient values of biochars produced from giant reed at different temperatures. Bioresour. Technol. 2013, 130, 463–471. [Google Scholar] [CrossRef] [PubMed]
- Christel, W.; Bruun, S.; Magid, J.; Jensen, L.S. Phosphorus availability from the solid fraction of pig slurry is altered by composting or thermal treatment. Bioresour. Technol. 2014, 169, 543–551. [Google Scholar] [CrossRef]
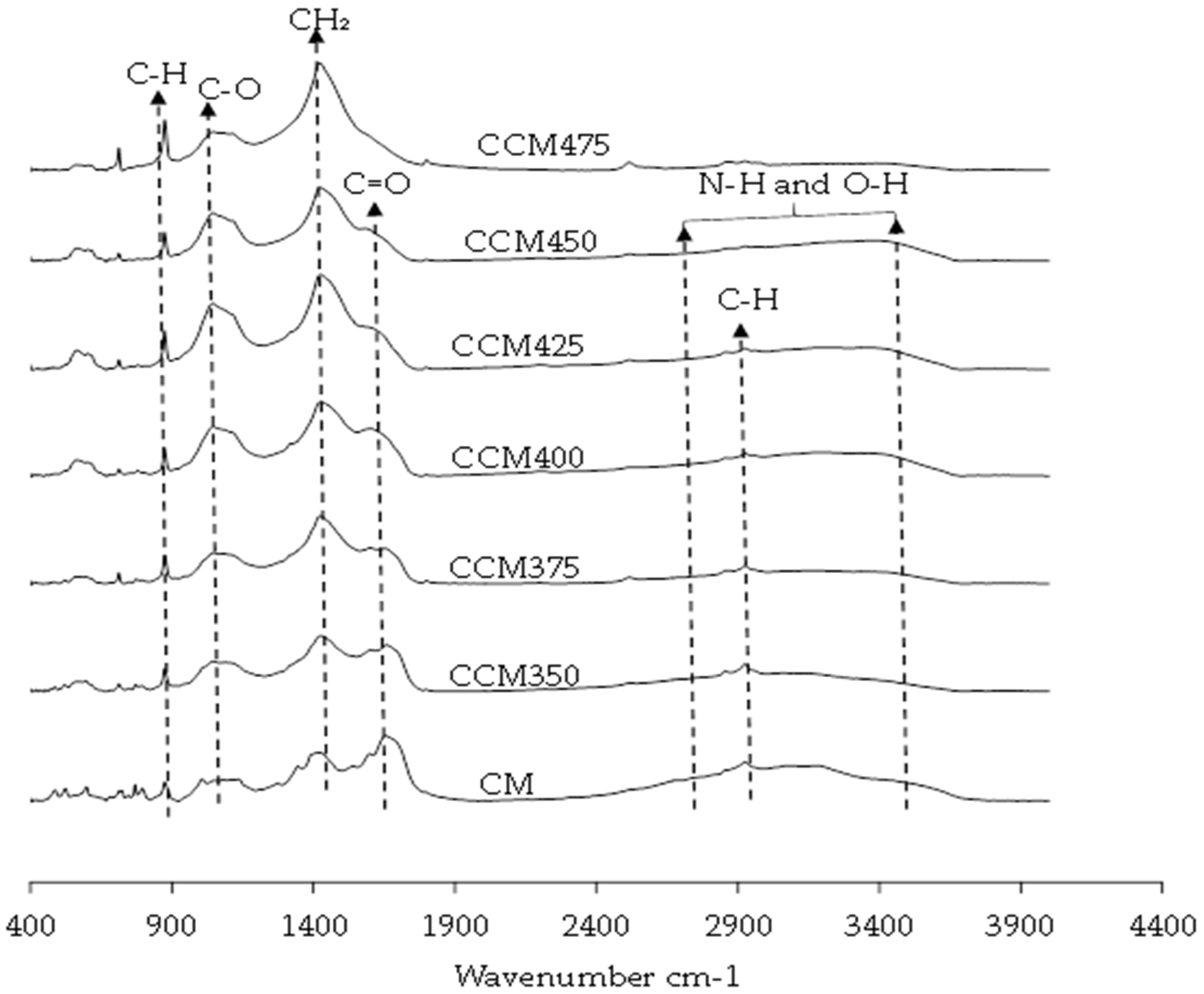
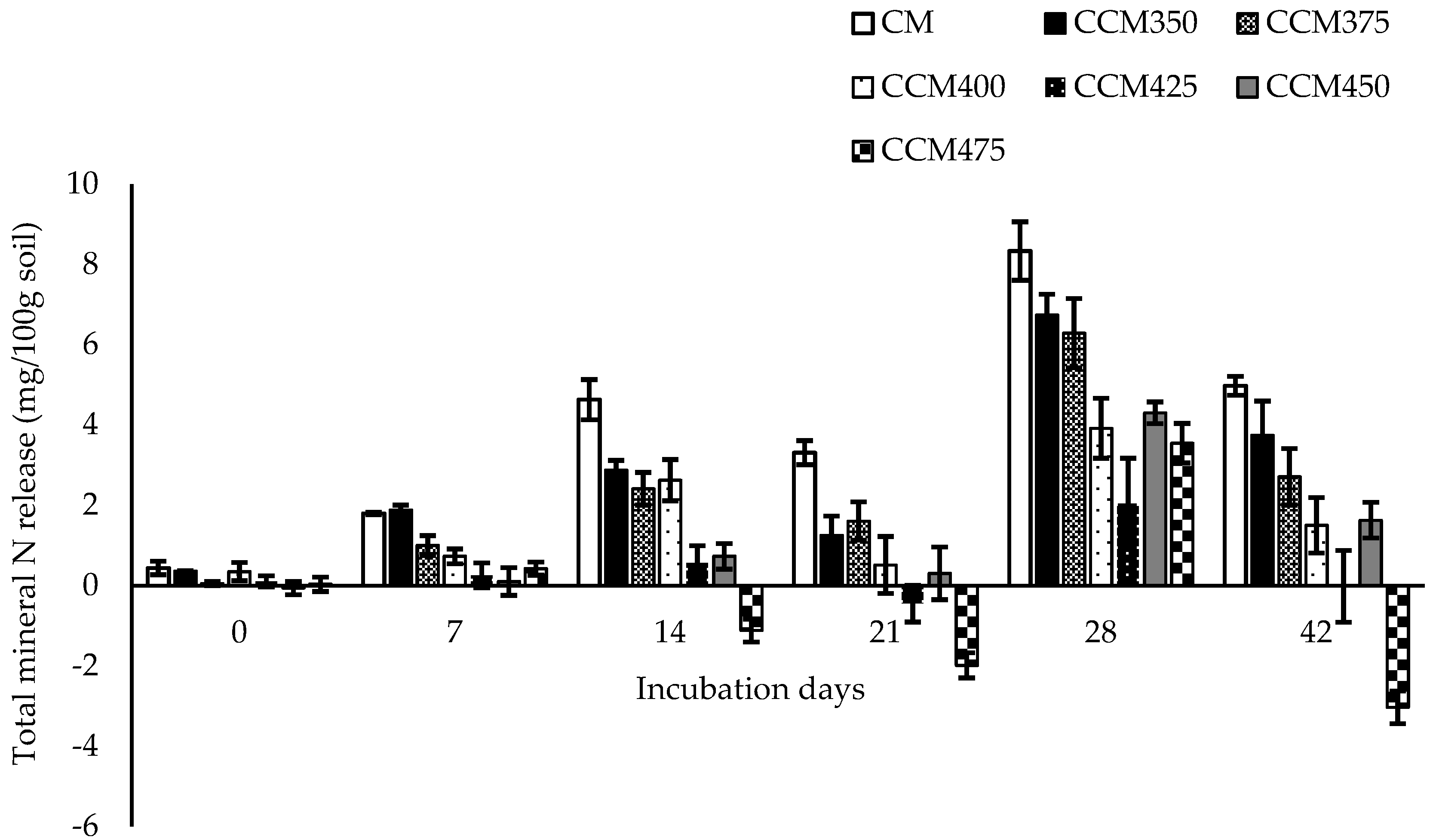
 represent chicken manure and chicken manure carbonized at 350, 375, 400, 425, 450, and 475 °C, respectively.
represent chicken manure and chicken manure carbonized at 350, 375, 400, 425, 450, and 475 °C, respectively.
 represent chicken manure and chicken manure carbonized at 350, 375, 400, 425, 450, and 475 °C, respectively.
represent chicken manure and chicken manure carbonized at 350, 375, 400, 425, 450, and 475 °C, respectively.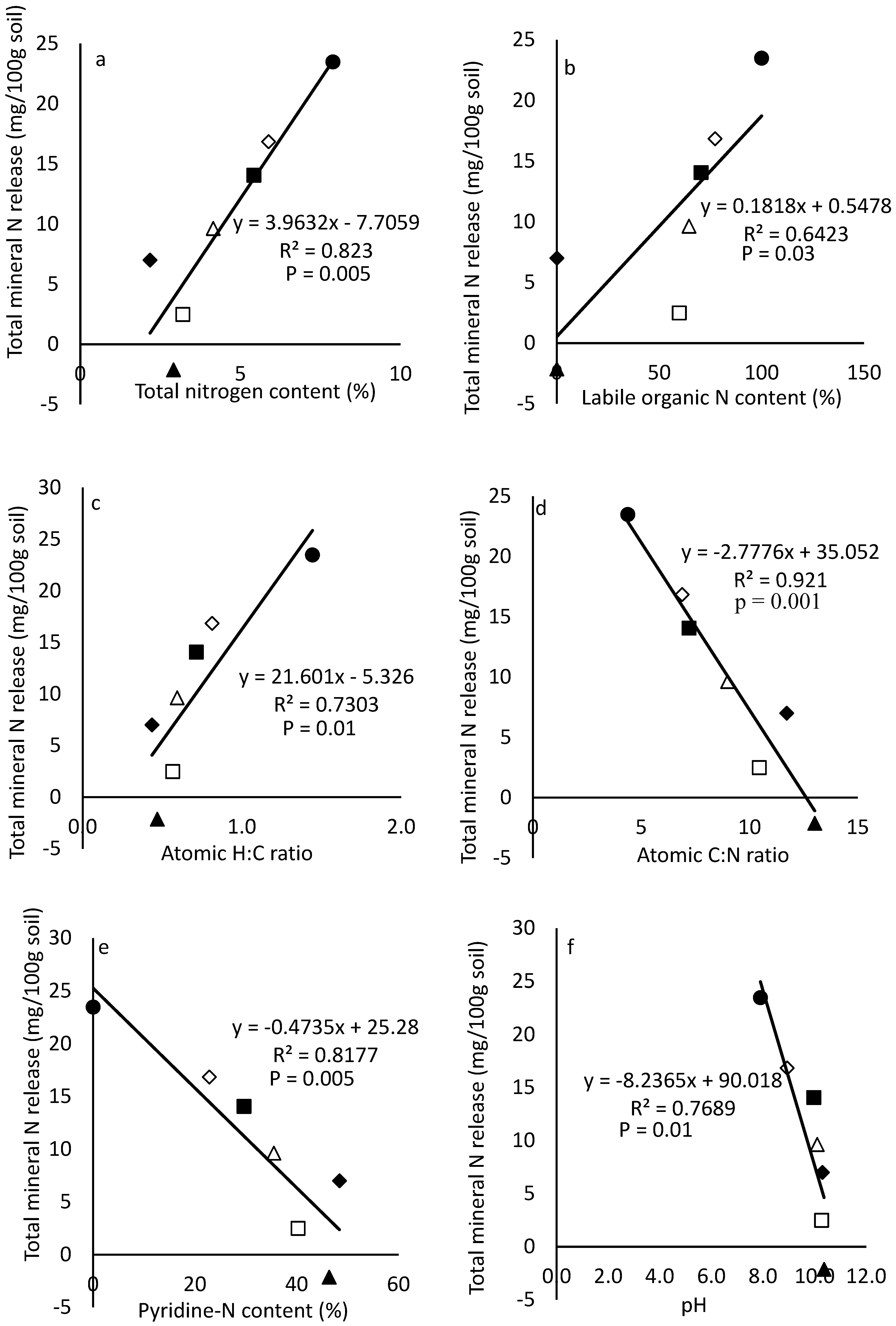
| Elemental Composition (%) | Atomic Ratios | Recovery with Respect to the Un-Carbonized CM | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Samples | Yield (%) | Ash (%) | N | C | H | O a | C:N | H:C | O:C | N (%) | C (%) | H (%) | O (%) |
| CM | 100.00 | 30.07 | 7.89 | 29.60 | 3.56 | 28.88 | 4.38 | 1.44 | 0.73 | 100.00 | 100.00 | 100.00 | 100.00 |
| CCM350 | 59.08 | 43.08 | 5.88 | 34.70 | 2.35 | 13.99 | 6.88 | 0.81 | 0.30 | 44.03 | 69.26 | 39.00 | 28.62 |
| CCM375 | 56.35 | 45.17 | 5.42 | 33.47 | 1.99 | 13.95 | 7.20 | 0.71 | 0.31 | 38.71 | 63.72 | 31.50 | 27.23 |
| CCM400 | 53.48 | 48.03 | 4.15 | 31.97 | 1.58 | 14.27 | 8.99 | 0.59 | 0.33 | 28.13 | 57.76 | 23.73 | 26.42 |
| CCM425 | 50.99 | 49.95 | 3.20 | 28.66 | 1.35 | 16.84 | 10.45 | 0.57 | 0.44 | 20.68 | 49.37 | 19.34 | 29.73 |
| CCM450 | 49.05 | 52.92 | 2.91 | 29.22 | 1.06 | 13.88 | 11.71 | 0.44 | 0.36 | 18.09 | 48.42 | 14.60 | 23.59 |
| CCM475 | 48.20 | 53.18 | 2.18 | 24.31 | 0.95 | 19.38 | 13.01 | 0.47 | 0.60 | 13.32 | 39.59 | 12.86 | 32.34 |
| Samples | pH | EC | NH4+-N (mg/100 g) | NO3−-N (mg/100 g) | Total Mineral N (mg/100 g) |
|---|---|---|---|---|---|
| CM | 7.9 | 4.5 | 67.5 | 8.4 | 75.9 |
| CCM350 | 8.9 | 2.8 | 0.75 | 0.5 | 1.2 |
| CCM375 | 10.0 | 4.2 | 1.08 | 0.7 | 1.8 |
| CCM400 | 10.1 | 5.6 | 2.12 | 1.0 | 3.2 |
| CCM425 | 10.3 | 6.0 | 0.85 | 0.6 | 1.5 |
| CCM450 | 10.3 | 5.9 | 0.56 | 0.4 | 1.0 |
| CCM475 | 10.4 | 6.0 | 0.43 | 0.4 | 0.8 |
| Peak Positions cm−1 | Proposed Assignments | References |
|---|---|---|
| 3600–2500 | Amino acid hydrohalides (N-H vibration) or OH bond in water, carboxyl and hydroxyl groups | [22,23] |
| 2925 | Aliphatic methylene band | [24] |
| 1665–1650 | Amide band I (C=O vibration) | [25] |
| 1435–1412 | CH2 units in biopolymer | [26] |
| 1060–1000 | Polysaccharides (C–O vibration) | [27] |
| 800 and 600 | Aromatic C–H out of plane deformation | [28] |
| Nitrogen Forms | Symbol | Binding Energy | References |
|---|---|---|---|
| Protein-N, amino-N, amide-N, nitrile | N-I | 399.7 ± 0.3 | [29] |
| Pyridine-N | N-II | 398.7 ± 0.4 | [30] |
| Pyrrolic-N | N-III | 400.3 ± 0.1 | [31] |
| Sample | N-I (%) | N-II (%) | N-III (%) |
|---|---|---|---|
| CM | 100 | 0 | 0 |
| CM350 | 77.19 | 22.81 | 0 |
| CM375 | 70.36 | 29.64 | 0 |
| CM400 | 64.51 | 35.49 | 0 |
| CM425 | 59.75 | 40.25 | 0 |
| CM450 | 0 | 48.38 | 51.62 |
| CM475 | 0 | 46.35 | 53.65 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Daramy, M.A.; Kawada, R.; Oba, S. What Is the Threshold Carbonization Temperature for Sustainable Preservation of the Good Nitrogen Supply Ability of Chicken Manure? Sustainability 2020, 12, 3306. https://doi.org/10.3390/su12083306
Daramy MA, Kawada R, Oba S. What Is the Threshold Carbonization Temperature for Sustainable Preservation of the Good Nitrogen Supply Ability of Chicken Manure? Sustainability. 2020; 12(8):3306. https://doi.org/10.3390/su12083306
Chicago/Turabian StyleDaramy, Moses Ahmed, Ryoka Kawada, and Shinya Oba. 2020. "What Is the Threshold Carbonization Temperature for Sustainable Preservation of the Good Nitrogen Supply Ability of Chicken Manure?" Sustainability 12, no. 8: 3306. https://doi.org/10.3390/su12083306
APA StyleDaramy, M. A., Kawada, R., & Oba, S. (2020). What Is the Threshold Carbonization Temperature for Sustainable Preservation of the Good Nitrogen Supply Ability of Chicken Manure? Sustainability, 12(8), 3306. https://doi.org/10.3390/su12083306





