Project and Implementation of an Educational Large-Scale Water Distillation Unit with a Closed-Circuit Condenser
Abstract
1. Introduction
2. Materials and Methods
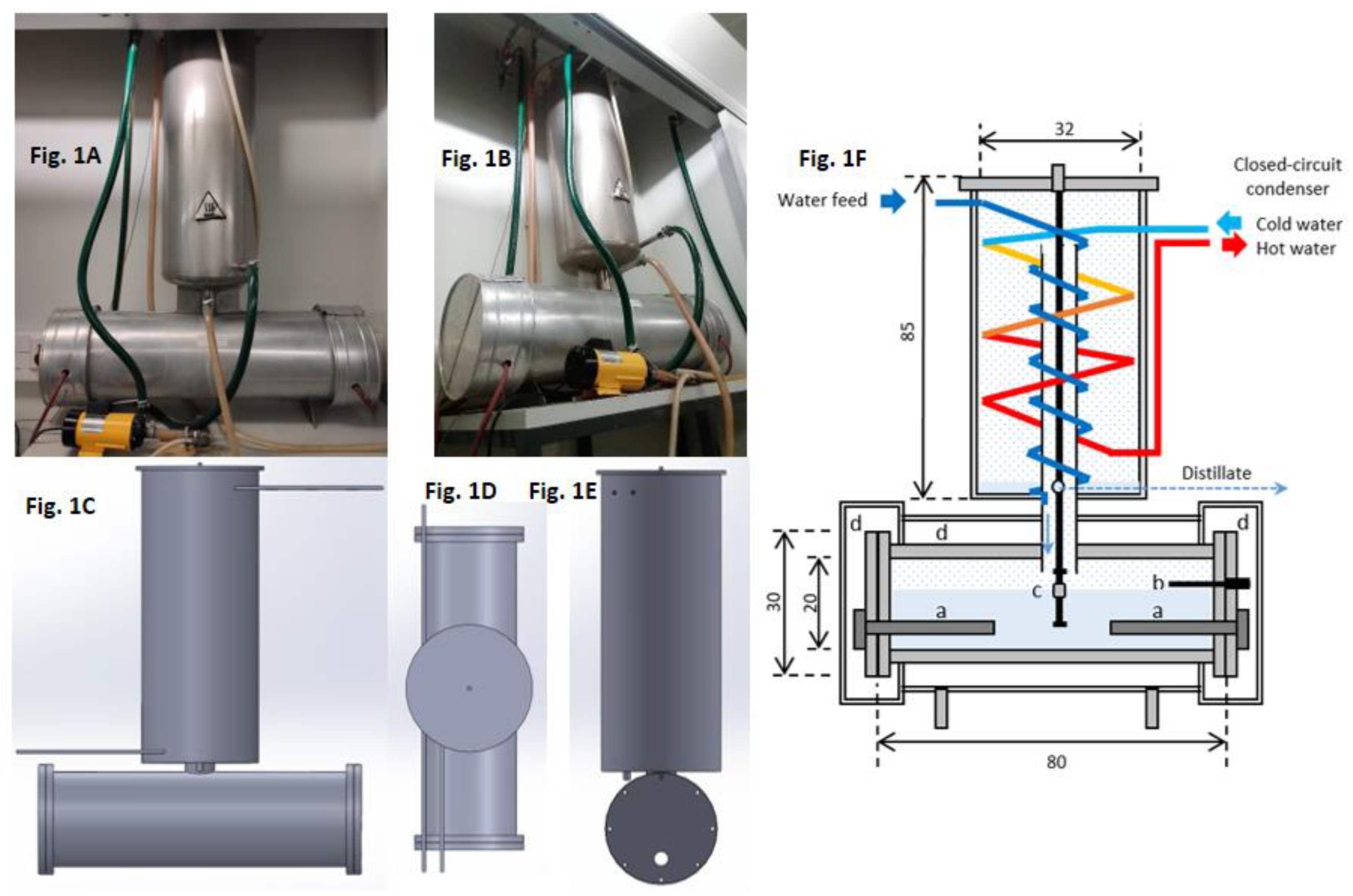
2.1. Design of the Distillation Unit
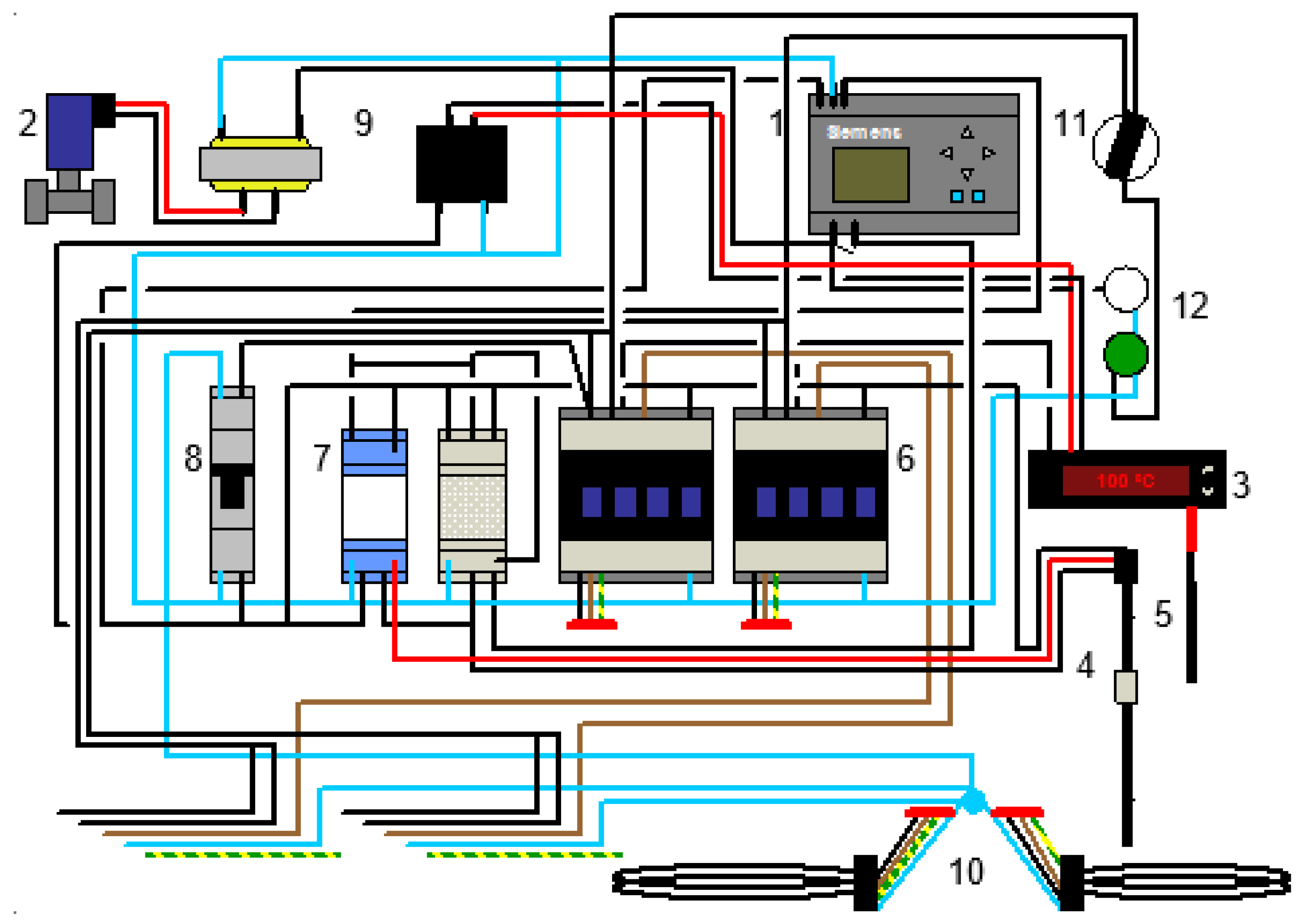
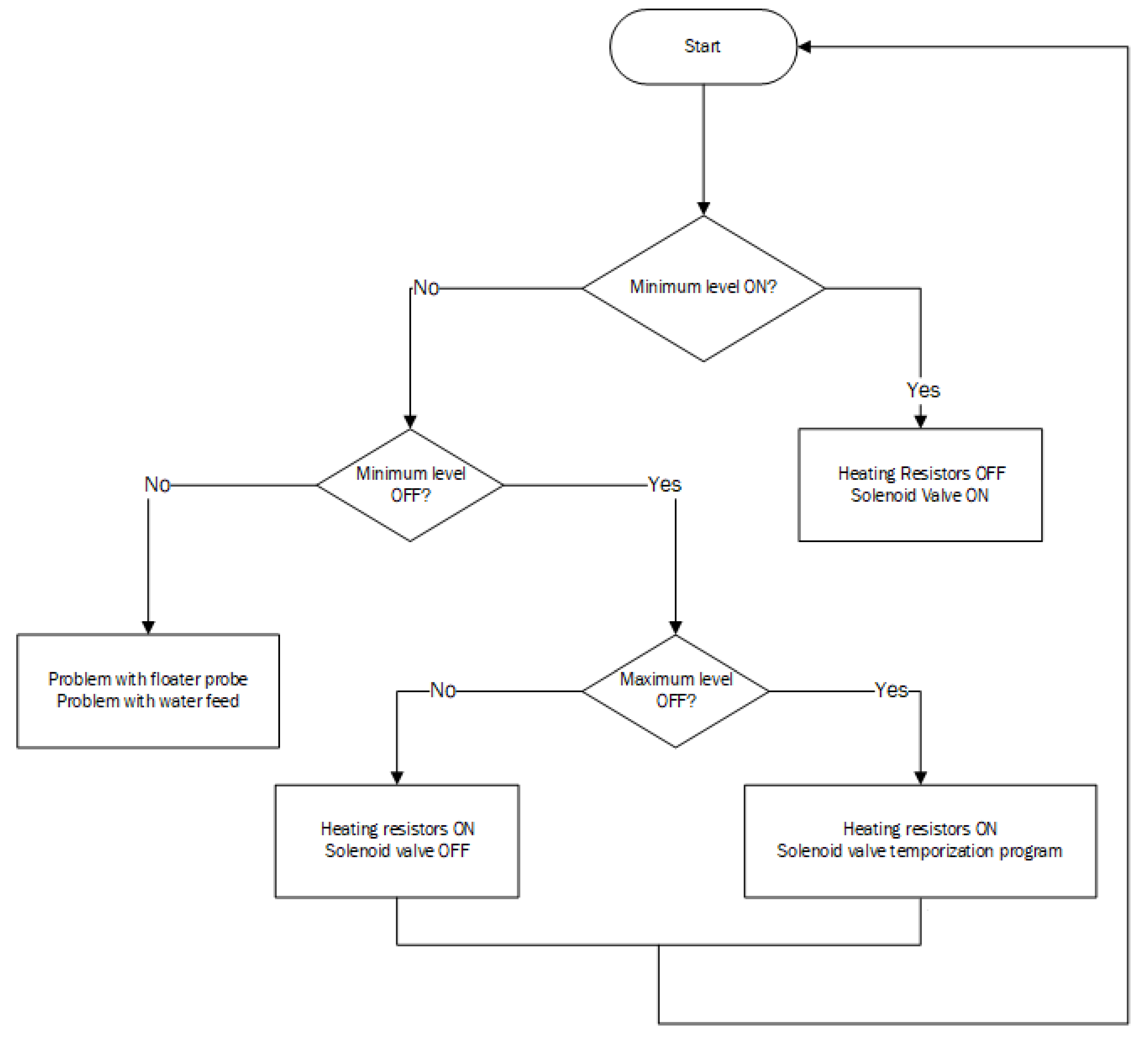
2.2. Control and Instrumentation of the Distillation Unit
3. Results and Discussion
3.1. Distillation Unit Design
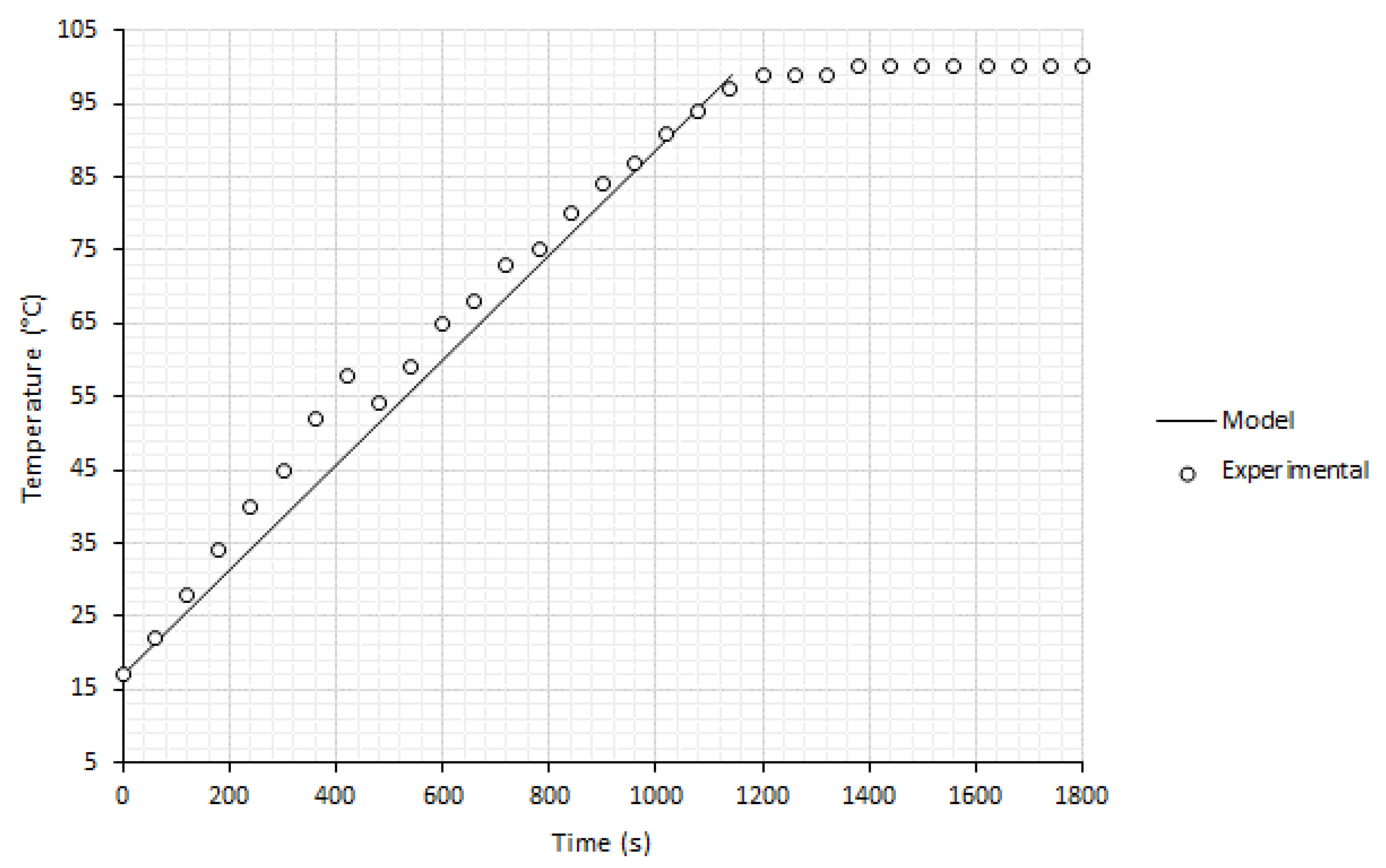
3.2. Reboiler Heating and Performance Analysis
3.3. Economic Analysis
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
Appendix A: Design of the Distillation Unit
A.1. Preheating Coil Design
A.2. Cooling Coils Design
| Constants | Values |
|---|---|
| (kg∙s−1) | 2.78∙10−3 |
| (kJ∙kg−1) | 2.257∙103 |
| (J∙kg−1∙°C−1) | 4181 |
| (kg∙m3) | 987 |
| k (W∙m−1∙°C−1) | 0.647 |
| (Pa∙s) | 5.3∙10−4 |
| (Pa∙s) | 3.186∙10−4 |
| g (m∙s−2) | 9.8 |
| (W∙m−1∙°C−1) | 16.3 |
References
- Escobar, I.C. Chapter 1 An Overview of the Global Water Situation. In Sustainability Science and Engineering; Elsevier: Amsterdam, The Netherlands, 2010; Volume 2, pp. 3–5. [Google Scholar] [CrossRef]
- Aqueduct Projected Water Stress Country Rankings | World Resources Institute. Available online: https://www.wri.org/resources/data-sets/aqueduct-projected-water-stress-country-rankings (accessed on 16 February 2020).
- UNDESA. Water scarcity | International Decade for Action ‘Water for Life’ 2005–2015. Available online: https://www.un.org/waterforlifedecade/scarcity.shtml (accessed on 9 April 2020).
- Liu, J.; Yang, H.; Gosling, S.N.; Kummu, M.; Flörke, M.; Pfister, S.; Hanasaki, N.; Wada, Y.; Zhang, X.; Zheng, C.; et al. Water scarcity assessments in the past, present, and future. Earth’s Future 2017, 5, 545–559. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Qin, Z.; Yuan, Q. The impact of eco-label on the young Chinese generation: The mediation role of environmental awareness and product attributes in green purchase. Sustainability 2019, 11, 973. [Google Scholar] [CrossRef]
- Brock, A. Education for Sustainable Development in Germany: Not Just Desired but Also Effective for Transformative Action. Sustainability 2020, 12, 2838. [Google Scholar]
- Schoeddert, A.; Babooram, K.; Pelletier, S. Reduction of Water Waste in an Organic Chemistry Laboratory Using a Low-Cost Recirculation System for Condenser Apparatus. J. Chem. Educ. 2019, 96, 180–182. [Google Scholar] [CrossRef]
- Baum, E.W.; Esteb, J.J.; Wilson, A.M. Waterless Condensers for the Teaching Laboratory: An Adaptation of Traditional Glassware. J. Chem. Educ. 2014, 91, 1087–1088. [Google Scholar] [CrossRef]
- Baum, E.W.; O’Callaghan, I.; Cinninger, L.; Esteb, J.J.; Wilson, A.M. Static fluid condensers for the containment of refluxing solvent. ACS Sustain. Chem. Eng. 2013, 1, 1502–1505. [Google Scholar] [CrossRef]
- Leal Filho, W. Living Labs for Sustainable Development: The Role of the European School of Sustainability Sciences and Research. In World Sustainability Series; Springer: Berlin/Heidelberg, Germany, 2020; pp. 3–9. [Google Scholar] [CrossRef]
- Alves, M.; Pinto, A.G.D.C.J. Two Simple Experiments for the Fluid Mechanics and Heat Transfer Laboratory Class. Chem. Eng. Educ. 1999, 33, 226–231. [Google Scholar]
- Kasumu, A.S.; Nassar, N.N.; Mehrotra, A.K. A heat-transfer laboratory experiment with shell-and-tube condenser. Educ. Chem. Eng. 2017, 19, 38–47. [Google Scholar] [CrossRef]
- Narang, A.; Ben-Zvi, A.; Afacan, A.; Sharp, D.; Shah, S.L.; Huang, B. Undergraduate design of experiment laboratory on analysis and optimization of distillation column. Educ. Chem. Eng. 2012, 7, e187–e195. [Google Scholar] [CrossRef]
- Macey, A.; Gurguis, N.; Tebboth, M.; Shah, P.S.; Chesi, C.; Shah, U.V.; Brechtelsbauer, C. Teaching reaction kinetics with chemiluminescence. Educ. Chem. Eng. 2018, 22, 53–60. [Google Scholar] [CrossRef]
- Mendes, A.; Magalhães, F.; Madeira, L. Sucrose Inversion: An Experiment on Heterogeneous Catalysis*. Int. J. Eng. Educ. 2003, 19, 893–901. [Google Scholar]
- Matos, L.C.; Martins, J.I. Analysis of an Educational Cathodic Protection System with a Single Drainage Point: Modeling and Experimental Validation in Aqueous Medium. Materials (Basel). 2018, 11, 2099. [Google Scholar] [CrossRef] [PubMed]
- Coulson, J.M.; John, M.; Richardson, J.F.; John, F. Chemical Engineering; Pergamon Press: Oxford, UK, 1979. [Google Scholar]

| Coil Diameter (m) | Horizontal Tube | Vertical Tube | |||||
|---|---|---|---|---|---|---|---|
| Length (m) | No. of Turns | Length (m) | No. of Turns | Semi-Length (m) | Total Length (m) | No. of Turns | |
| 0.08 | 2.20 | 9.0 | 2.22 | 10.0 | 0.13 | 2.21 | 9.0 |
| 0.10 | 2.29 | 7.5 | 2.32 | 8.5 | 0.16 | 2.31 | 8.0 |
| 0.12 | 2.36 | 6.5 | 2.40 | 7.5 | 0.19 | 2.38 | 7.0 |
| 0.15 | 2.44 | 5.5 | 2.47 | 6.0 | 0.24 | 2.45 | 6.0 |
| 0.18 | 2.49 | 5.0 | 2.49 | 5.5 | 0.28 | 2.51 | 5.0 |
| 0.21 | 2.52 | 4.5 | 2.52 | 5.0 | 0.33 | 2.55 | 4.5 |
| 0.24 | 2.55 | 4.0 | 2.55 | 4.5 | 0.38 | 2.58 | 4.0 |
| 0.27 | 2.58 | 3.5 | 2.58 | 4.0 | 0.42 | 2.60 | 4.0 |
| 0.30 | 2.60 | 3.0 | 2.65 | 3.5 | 0.47 | 2.62 | 3.5 |
| Coil Diameter (m) | Horizontal Tubes | Vertical Tubes | |||||
|---|---|---|---|---|---|---|---|
| Length (m) | No. of Turns | Length (m) | No. of Turns | Semi-Length (m) | Total Length (m) | No. of Turns | |
| 0.08 | 2.08 | 4.5 | 2.13 | 6.5 | 0.13 | 2.10 | 5.0 |
| 0.10 | 2.23 | 4.0 | 2.30 | 5.5 | 0.16 | 2.27 | 4.5 |
| 0.12 | 2.35 | 3.5 | 2.44 | 4.5 | 0.19 | 2.40 | 4.0 |
| 0.15 | 2.50 | 3.0 | 2.60 | 4.0 | 0.24 | 2.55 | 3.5 |
| 0.18 | 2.60 | 2.5 | 2.73 | 3.5 | 0.28 | 2.66 | 3.0 |
| 0.21 | 2.69 | 2.5 | 2.83 | 3.0 | 0.33 | 2.76 | 2.5 |
| 0.25 | 2.78 | 2.0 | 2.93 | 3.0 | 0.39 | 2.85 | 2.5 |
| 0.28 | 2.83 | 2.0 | 3.00 | 2.5 | 0.44 | 2.91 | 2.0 |
| 0.31 | 2.87 | 1.5 | 3.05 | 2.0 | 0.49 | 2.96 | 2.0 |
| Service | Subtotal (€) |
|---|---|
| Distillation unit with closed-circuit condenser | 1908 |
| Automation and control | 269 |
| Total | 2177 |
| Service | Subtotal (€) |
|---|---|
| Water 1 | 23 |
| Electricity 2 | 2249 |
| Maintenance 3 | 109 |
| Total | 2381 |
| Service | Subtotal (€) |
|---|---|
| Water 1 | 1646 |
| Electricity 2 | 3327 |
| Maintenance 3 | 168 |
| Total | 5140 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Matos, L.C.; Eulálio, A.; Antunes, T.; Loureiro, J.M.; Ferreira, A.; Mendes, A. Project and Implementation of an Educational Large-Scale Water Distillation Unit with a Closed-Circuit Condenser. Sustainability 2020, 12, 3239. https://doi.org/10.3390/su12083239
Matos LC, Eulálio A, Antunes T, Loureiro JM, Ferreira A, Mendes A. Project and Implementation of an Educational Large-Scale Water Distillation Unit with a Closed-Circuit Condenser. Sustainability. 2020; 12(8):3239. https://doi.org/10.3390/su12083239
Chicago/Turabian StyleMatos, Luís Carlos, Ana Eulálio, Tiago Antunes, José Miguel Loureiro, Alexandre Ferreira, and Adélio Mendes. 2020. "Project and Implementation of an Educational Large-Scale Water Distillation Unit with a Closed-Circuit Condenser" Sustainability 12, no. 8: 3239. https://doi.org/10.3390/su12083239
APA StyleMatos, L. C., Eulálio, A., Antunes, T., Loureiro, J. M., Ferreira, A., & Mendes, A. (2020). Project and Implementation of an Educational Large-Scale Water Distillation Unit with a Closed-Circuit Condenser. Sustainability, 12(8), 3239. https://doi.org/10.3390/su12083239







