Variability of Hordeum vulgare L. Cultivars in Yield, Antioxidant Potential, and Cholinesterase Inhibitory Activity
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. Extraction
2.2.2. Growth Rate
2.2.3. Reaction with the Folin–Ciocalteu Reagent
2.2.4. Polyphenolic Acids Determination
2.2.5. Chelating Activity
2.2.6. Flavonols Composition
2.2.7. Antiradical Capacity
2.2.8. Cholinesterases Inhibition
2.3. Statistical Analysis
3. Results
3.1. Yield Size
3.2. Active Compounds and Antiradical Properties
3.3. Flavonols Analysis
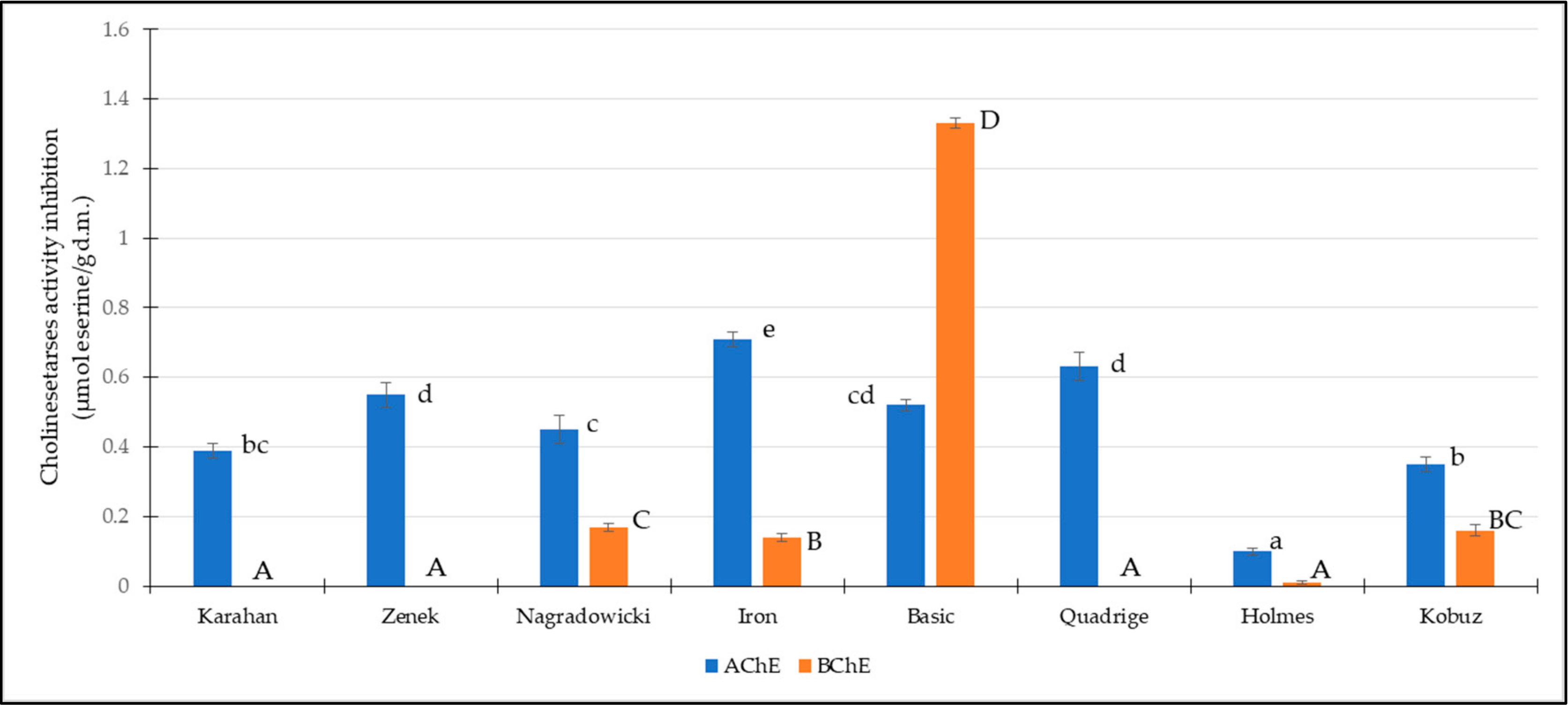
3.4. Cholinesterases Inhibition
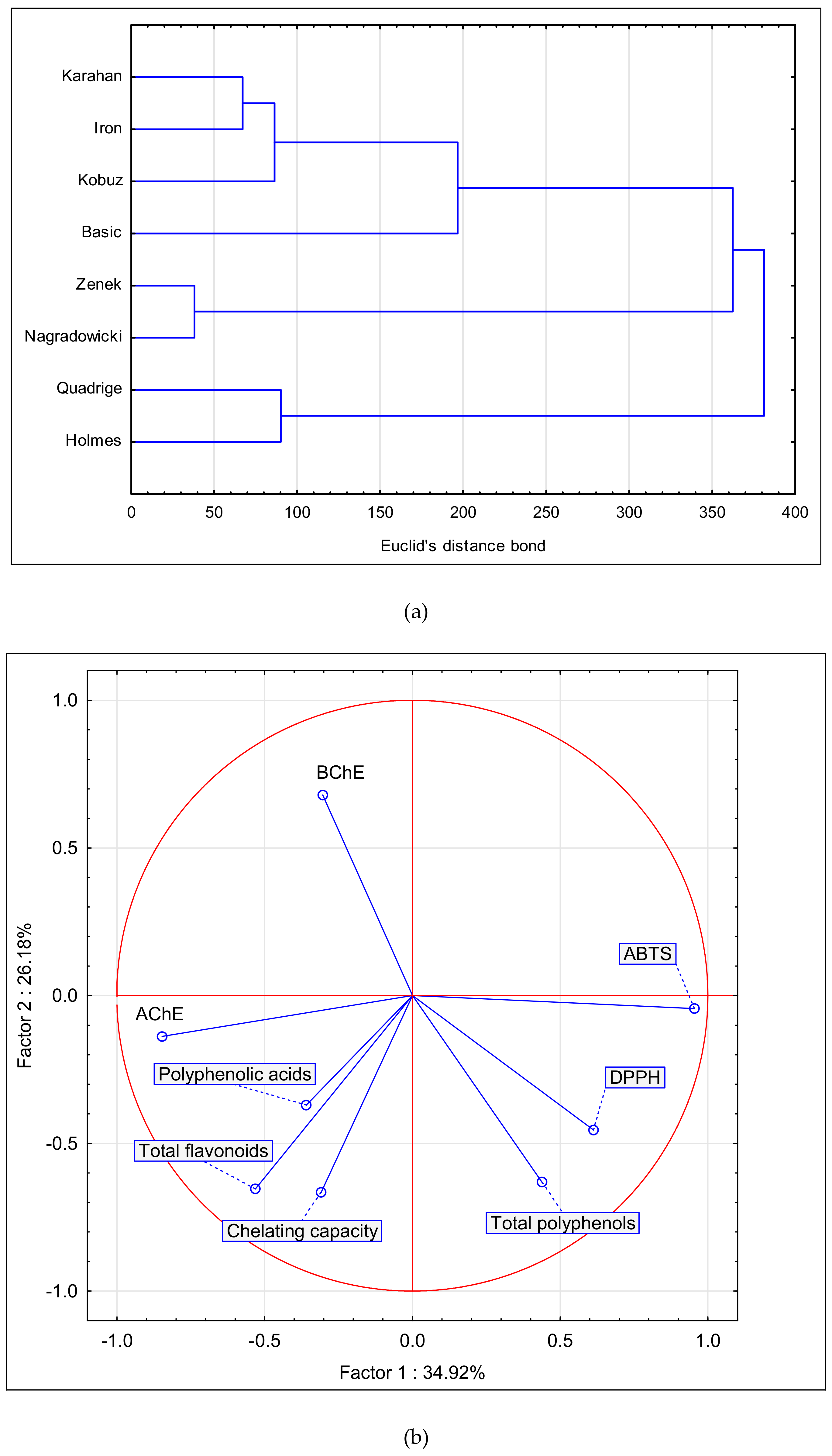
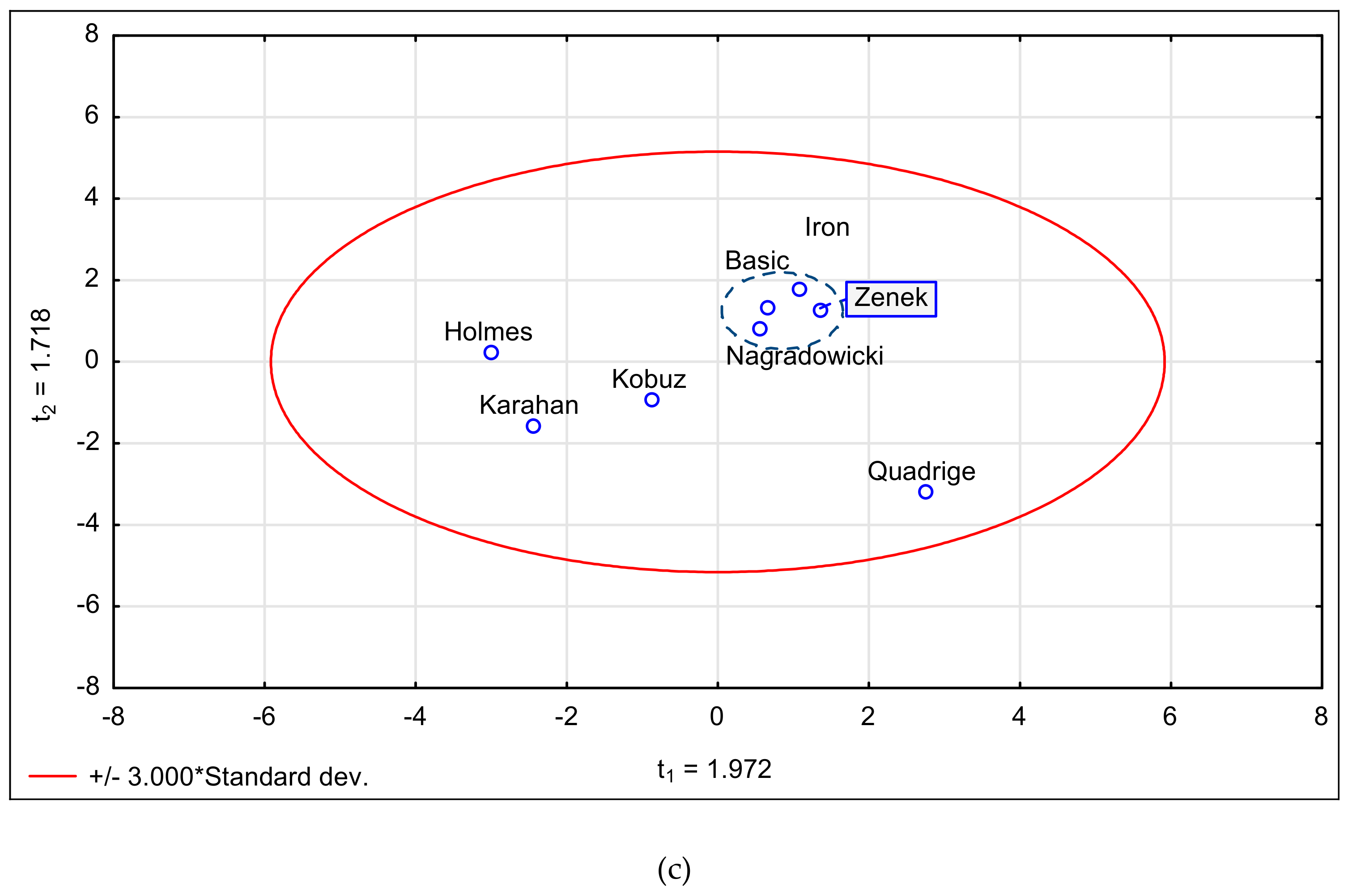
3.5. Statistical Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Lahouar, L.; El Arem, A.; Ghrairi, F.; Chahdoura, H.; Ben Salem, H.; El Felah, M.; Achour, L. Phytochemical content and antioxidant properties of diverse varieties of whole barley (Hordeum vulgare L.) grown in Tunisia. Food Chem. 2014, 145, 578–583. [Google Scholar] [CrossRef]
- Kamiyama, M.; Shibamoto, T. Flavonoids with Potent Antioxidant Activity Found in Young Green Barley Leaves. J. Agric. Food Chem. 2012, 60, 6260–6267. [Google Scholar] [CrossRef] [PubMed]
- Gao, T.; Zhang, M.; Fang, Z.; Zhong, Q. Optimization of microwave-assisted extraction of flavonoids from young barley leaves. Int. Agrophysics 2017, 31, 45–52. [Google Scholar] [CrossRef]
- Havlíková, L.; Šatínský, D.; Opletal, L.; Solich, P. A Fast Determination of Chlorophylls in Barley Grass Juice Powder Using HPLC Fused-Core Column Technology and HPTLC. Food Anal. Methods 2014, 7, 629–635. [Google Scholar] [CrossRef]
- Paulíčková, I.; Ehrenbergerová, J.; Fiedlerová, V.; Gabrovská, D.; Havlová, P.; Holasová, M.; Kopáček, J.; Ouhrabková, J.; Pinkrová, J.; Rysová, J.; et al. Evaluation of barley grass as a potential source of some nutritional substances. Czech. J. Food Sci. 2007, 25, 65–72. [Google Scholar] [CrossRef]
- Březinová Belcredi, N.; Ehrenbergerová, J.; Fiedlerová, V.; Běláková, S.; Vaculová, K. Antioxidant Vitamins in Barley Green Biomass. J. Agric. Food Chem. 2010, 58, 11755–11761. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Zeng, Y.; Yang, X.; Pu, X.; Du, J. Utilization of Barley Functional Foods for Preventing Chronic Diseases in China. Agric. Sci. Technol. 2016, 17, 2195–2204. [Google Scholar]
- Kiewlicz, J. Evaluation of Total Phenolic Content and Antioxidant Properties of the Water Extract of the Powdered Barley Grass (Hordeum vulgare L.). Towaroznawcze Problemy Jakości 2016, 47, 29–37. [Google Scholar] [CrossRef]
- Kowalczewski, P.Ł.; Radzikowska, D.; Ivanišová, E.; Szwengiel, A.; Kačániová, M.; Sawinska, Z. Influence of Abiotic Stress Factors on the Antioxidant Properties and Polyphenols Profile Composition of Green Barley (Hordeum vulgare L.). Int. J. Mol. Sci. 2020, 21, 397. [Google Scholar] [CrossRef]
- Yamaura, K.; Nakayama, N.; Shimada, M.; Bi, Y.; Fukata, H.; Ueno, K. Antidepressant-like effects of young green barley leaf (Hordeum vulgare L.) in the mouse forced swimming test. Pharmacogn. Res. 2012, 4, 22–26. [Google Scholar] [CrossRef]
- Yamaura, K.; Tanaka, R.; Bi, Y.; Fukata, H.; Oishi, N.; Sato, H.; Mori, C.; Ueno, K. Protective effect of young green barley leaf (Hordeum vulgare L.) on restraint stress-induced decrease in hippocampal brain-derived neurotrophic factor in mice. Pharmacogn. Mag. 2015, 11, S86. [Google Scholar] [CrossRef] [PubMed]
- Yoshizawa, M.; Yokoyama, K.; Nakano, Y.; Nakamura, H. Protective Effects of Barley and Its Hydrolysates on Gastric Stress Ulcer in Rats. Yakugaku Zasshi 2004, 124, 571–575. [Google Scholar] [CrossRef][Green Version]
- Işık, M.; Beydemir, Ş.; Yılmaz, A.; Naldan, M.E.; Aslan, H.E.; Gülçin, İ. Oxidative stress and mRNA expression of acetylcholinesterase in the leukocytes of ischemic patients. Biomed. Pharmacother. 2017, 87, 561–567. [Google Scholar] [CrossRef] [PubMed]
- Akhondzadeh, S.; Noroozian, M.; Mohammadi, M.; Ohadinia, S.; Jamshidi, A.H.; Khani, M. Melissa officinalis extract in the treatment of patients with mild to moderate Alzheimer’s disease: A double blind, randomised, placebo controlled trial. J. Neurol. Neurosurg. Psychiatry 2003, 74, 863–866. [Google Scholar] [CrossRef] [PubMed]
- Szewczyk, B. Zinc homeostasis and neurodegenerative disorders. Front. Aging Neurosci. 2013, 5. [Google Scholar] [CrossRef] [PubMed]
- Kobus-Cisowska, J.; Szymanowska, D.; Szczepaniak, O.; Kmiecik, D.; Przeor, M.; Gramza-Michałowska, A.; Cielecka-Piontek, J.; Smuga-Kogut, M.; Szulc, P. Humulus lupulus L. hops as a potent antioxidant: Implications for neurodegenerative disorders and antimicrobial effect. Nutrients 2019, 11, 1377. [Google Scholar] [CrossRef]
- Cheung, L.M.; Cheung, P.C.K.; Ooi, V.E.C. Antioxidant activity and total phenolics of edible mushroom extracts. Food Chem. 2003, 81, 249–255. [Google Scholar] [CrossRef]
- Office for Registration of Medicinal Products Medical Devices and Biocidal Products Farmakopea Polska; PT Farm: Warsaw, Poland, 2002.
- Tang, S.; Kerry, J.; Sheehan, D.; Buckley, D. Antioxidative mechanisms of tea catechins in chicken meat systems. Food Chem. 2002, 76, 45–51. [Google Scholar] [CrossRef]
- Kobus, J.; Flaczyk, E.; Siger, A.; Nogala-Kałucka, M.; Korczak, J.; Pegg, R.B. Phenolic compounds and antioxidant activity of extracts of Ginkgo leaves. Eur. J. Lipid Sci. Technol. 2009, 111, 1150–1160. [Google Scholar] [CrossRef]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Amarowicz, R.; Zegarska, Z.; Pegg, R.B.; Karamac, M.; Kosinska, A. Antioxidant and radical scavenging activities of a barley crude extract and its fraction. Czech. J. Food Sci. 2008, 25, 73–80. [Google Scholar] [CrossRef]
- Ellman, G.L.; Courtney, K.D.; Andres, V.; Featherstone, R.M. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem. Pharmacol. 1961, 7, 88–95. [Google Scholar] [CrossRef]
- Talbot, M. Yield variability of crop varieties in the U.K. J. Agric. Sci. 1984, 102, 315–321. [Google Scholar] [CrossRef]
- Sumińska, P.; Berton, A.; Dietz, W. Subcritical water extraction (SWE) of barley (Hordeum vulgare) straw as method of antimicrobial and antioxidant additives production. World Sci. News 2017, 81, 169–183. [Google Scholar]
- Chomchan, R.; Siripongvutikorn, A.P.D.S.; Puttarak, D.P.; Rattanapon, M.R. Investigation of Phytochemical Constituents, Phenolic Profiles and Antioxidant Activities of Ricegrass Juice compared to Wheatgrass Juice. Funct. Foods Heal. Dis. 2016, 6, 822. [Google Scholar] [CrossRef]
- Koga, R.; Tsubata, M.; Ikeguchi, M.; Takagaki, K.; Irino, N.; Kondo, R. Hypercholesterolemia-reducing Effect of Young Barley Leaf Powder. Nippon Shokuhin Kagaku Kogaku Kaishi 2013, 60, 19–24. [Google Scholar] [CrossRef]
- Choe, J.-H.; Jang, A.; Choi, J.-H.; Choi, Y.-S.; Han, D.-J.; Kim, H.-Y.; Lee, M.-A.; Kim, H.-W.; Kim, C.-J. Antioxidant activities of lotus leaves (Nelumbo nucifera) and barley leaves (Hordeum vulgare) extracts. Food Sci. Biotechnol. 2010, 19, 831–836. [Google Scholar] [CrossRef]
- Khanthapok, P.; Muangprom, A.; Sukrong, S. Antioxidant activity and DNA protective properties of rice grass juices. Sci. Asia 2015, 41, 119. [Google Scholar] [CrossRef]
- Rattanapon, R.; Siripongvutikorn, S.; Usawakesmanee, W.; Thongraung, C. Changes of nutritional value, bioactive compounds and antioxidant activity of primed white rice, Chainat 1, during seedling. Int. Food Res. J. 2017, 24, 2563–2571. [Google Scholar]
- Anwar, F.; Qayyum, H.M.A.; Hussain, A.I.; Iqbal, S. Antioxidant activity of 100% and 80% methanol extracts from barley seeds (Hordeum vulgare L.): Stabilization of sunflower oil. Grasas y Aceites 2010, 61, 237–243. [Google Scholar] [CrossRef]
- Akhtar, M.N.; Lam, K.W.; Abas, F.; Maulidiani Ahmad, S.; Shah, S.A.A.; Atta-ur-Rahman; Choudhary, M.I.; Lajis, N.H. New class of acetylcholinesterase inhibitors from the stem bark of Knema laurina and their structural insights. Bioorg. Med. Chem. Lett. 2011, 21, 4097–4103. [Google Scholar] [CrossRef] [PubMed]
- Kobus-Cisowska, J.; Szymanowska, D.; Maciejewska, P.; Kmiecik, D.; Gramza-Michałowska, A.; Kulczyński, B.; Cielecka-Piontek, J. In vitro screening for acetylcholinesterase and butyrylcholinesterase inhibition and antimicrobial activity of chia seeds (Salvia hispanica). Electron. J. Biotechnol. 2019, 37, 1–10. [Google Scholar] [CrossRef]
- Wszelaki, N. Plants as a source of acetylcholinesterase and butyrylcholinesterase inhibitors. Postępy Fitoterapii 2009, 10, 24–38. [Google Scholar]
- Szwajgier, D.; Borowiec, K. Phenolic acids from malt are efficient acetylcholinesterase and butyrylcholinesterase inhibitors. J. Inst. Brew. 2012, 118, 40–48. [Google Scholar] [CrossRef]
- Szwajgier, D.; Wydrych, M.; Wiecław, E.; Targoński, Z. Anticholinesterase and antioxidant activities of commercial preparations from Ginkgo biloba leaves. Acta Sci. Pol. Hortorum Cultus 2013, 12, 111–125. [Google Scholar]
| Cultivar | After 2 Days | After 4 Days | After 6 Days | After 8 Days | After 10 Days |
|---|---|---|---|---|---|
| Karahan | 0.50a ± 0.01 | 6.80a ± 0.04 | 10.05a ± 0.16 | 14.00b ± 0.14 | 17.84c ± 0.16 |
| Zenek | 0.10a ± 0.01 | 6.10a ± 0.10 | 12.85b ± 0.08 | 16.74c ± 0.17 | 18.30c ± 0.19 |
| Nagradowicki | 0.20a ± 0.01 | 5.86a ± 0.20 | 12.60b ± 0.17 | 14.54b ± 0.23 | 17.37c ± 0.16 |
| Iron | 0.20a ± 0.01 | 8.02d ± 0.04 | 14.37c ± 0.10 | 16.84c ± 0.14 | 18.43c ± 0.14 |
| Basic | 0.80b± 0.02 | 5.43a ± 0.10 | 12.32b ±0.16 | 15.38bc ± 0.19 | 18.4c ± 0.19 |
| Quadrige | 0.30a ± 0.02 | 6.20a ± 0.22 | 13.02b ± 0.08 | 16.27c ± 0.23 | 19.98c ± 0.20 |
| Holmes | 1.02c ± 0.01 | 7.76a ± 0.08 | 10.08a ± 0.23 | 11.87a ± 0.17 | 13.48a ± 0.22 |
| Kobuz | 1.25c ± 0.02 | 7.24a ± 0.13 | 12.76b ± 0.20 | 13.74b ± 0.22 | 15.93b ± 0.14 |
| Cultivar | Total Polyphenols mg QE/g d.w. | Polyphenolic Acids mg CA/g d.w. | ChA % | DPPH mg TE/g d.w. | TEAC mg TE/g d.w. |
|---|---|---|---|---|---|
| Karahan | 37.35c ± 3.33 | 6.95a ± 0.07 | 87.33c ± 0.37 | 8.66c± 1.17 | 12.32c ± 1.63 |
| Zenek | 25.32a ± 0.87 | 6.80a ± 0.25 | 92.23c ± 0.52 | 5.74b± 0.66 | 6.79a ± 0,77 |
| Nagradowicki | 31.17b ± 2.41 | 6.76a ± 0.11 | 78.65bc ± 0.75 | 5.32b± 0.87 | 6.64a± 0.84 |
| Iron | 33.82b ± 3.87 | 6.30a ± 0.22 | 88.54c ± 1.04 | 4.08a ± 0.93 | 7.09b ± 0.76 |
| Basic | 28.82a ± 1.31 | 7.69b ± 0.38 | 68.91b ± 2.09 | 4.99a ± 0.51 | 7.11b ± 0.41 |
| Quadrige | 37.94c ± 4.53 | 8.88c ± 0.34 | 90.12c ± 6.51 | 5.75b ± 0.56 | 5.42a ± 0.65 |
| Holmes | 35.58c ± 4.66 | 7.20b ± 0.37 | 62.41a ± 2.16 | 9.01c ± 0.67 | 12.28c ± 0.94 |
| Kobuz | 35.67c ± 2.58 | 6.57a ± 0.28 | 93.26c ± 1.57 | 11.22d ± 1.26 | 12.12c ± 1.61 |
| Cultivar | Catechin | Epicatechin | Quercetin | Rutin | Kaempferitrin | Total |
|---|---|---|---|---|---|---|
| Karahan | 1.05a ± 0.01 | 1.23a ± 0.05 | 212.26b ± 0.88 | 41.07a ± 0.14 | 65.84c ± 4.11 | 464.28a ± 3.60 |
| Zenek | 5.57c ± 0.78 | 27.70c ± 0.14 | 119.75a ± 0.60 | 458.39c ± 17.32 | 12.55b ± 0.25 | 1354.74d ± 92.72 |
| Nagradowicki | 6.35c ± 0.24 | 2.48b ± 0.09 | 134.63a ± 5.09 | 439.57c ± 0.89 | 17.27b ± 2.42 | 486.22ab ± 157.90 |
| Iron | 1.11a ± 0.01 | 1.30a ± 0.05 | 225.82b ± 0.94 | 43.27a ± 0.15 | 0.64a ± 0.01 | 424.53a ± 6.64 |
| Basic | 1.94b ± 0.01 | 2.29b ± 0.09 | 395.99c ± 1.65 | 75.88b ± 0.25 | 122.84d ± 7.68 | 866.16c ± 6.71 |
| Quadrige | 14.02d ± 0.32 | 60.18e ± 0.30 | 260.18b ± 1.31 | 846.81d± 32.00 | 27.26c ± 0.55 | 2943.36e ± 201.45 |
| Holmes | 11.73d ± 0.44 | 1.39a ± 0.05 | 240.77b ± 1.00 | 812.04d ± 1.66 | 74.69c ± 4.67 | 526.64b ± 4.08 |
| Kobuz | 2.05b ± 0.01 | 36.59d ± 0.18 | 158.19a ± 0.79 | 79.93b ± 0.27 | 16.57b ± 0.33 | 1789.62d ± 122.48 |
| Variables | Catechin | Epicatechin | Quercetin | Rutin | Kaempferatrin | Total Flavonoids | Total Polyphenols | Polyphenolic Acids | Chelating Capacity | DPPH | ABTS | AChE | BChE |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Catechin | 1.000000 | 0.486199 | −0.004053 | 0.986254 | −0.047441 | 0.554473 | 0.249931 | 0.677620 | −0.246055 | −0.071314 | −0.218560 | −0.213750 | −0.356166 |
| Epicatechin | 0.486199 | 1.000000 | −0.158716 | 0.419432 | −0.383136 | 0.913389 | 0.216069 | 0.569446 | 0.575383 | −0.011370 | −0.278077 | 0.276822 | −0.289807 |
| Quercetin | −0.004053 | −0.158716 | 1.000000 | −0.094495 | 0.797041 | −0.146031 | 0.092078 | 0.529698 | −0.536696 | −0.158787 | −0.119698 | 0.090814 | 0.765542 |
| Rutin | 0.986254 | 0.419432 | −0.094495 | 1.000000 | −0.075974 | 0.482866 | 0.139708 | 0.584420 | −0.264983 | −0.069538 | −0.208789 | −0.254053 | −0.397344 |
| Kaempferatrin | −0.047441 | −0.383136 | 0.797041 | −0.075974 | 1.000000 | −0.344702 | −0.041234 | 0.355644 | −0.731360 | 0.229379 | 0.242325 | −0.380820 | 0.705555 |
| Total flavonoids | 0.554473 | 0.913389 | −0.146031 | 0.482866 | −0.344702 | 1.000000 | 0.158945 | 0.630658 | 0.428766 | −0.090900 | −0.441781 | 0.282724 | −0.173784 |
| Total polyphenols | 0.249931 | 0.216069 | 0.092078 | 0.139708 | −0.041234 | 0.158945 | 1.000000 | 0.265507 | 0.082034 | 0.377237 | 0.453683 | −0.240852 | −0.404985 |
| Polyphenolic acids | 0.677620 | 0.569446 | 0.529698 | 0.584420 | 0.355644 | 0.630658 | 0.265507 | 1.000000 | −0.142638 | 0.079976 | −0.367766 | 0.135633 | 0.174840 |
| Chelating capacity | −0.246055 | 0.575383 | −0.536696 | −0.264983 | −0.731360 | 0.428766 | 0.082034 | −0.142638 | 1.000000 | 0.149800 | −0.173299 | 0.564674 | −0.456827 |
| DPPH | −0.071314 | −0.011370 | −0.158787 | −0.069538 | 0.229379 | −0.090900 | 0.377237 | 0.079976 | 0.149800 | 1.000000 | 0.564579 | −0.382954 | −0.319621 |
| ABTS | −0.218560 | −0.278077 | −0.119698 | −0.208789 | 0.242325 | −0.441781 | 0.453683 | −0.367766 | −0.173299 | 0.564579 | 1.000000 | −0.798283 | −0.228740 |
| AChE | −0.213750 | 0.276822 | 0.090814 | −0.254053 | −0.380820 | 0.282724 | −0.240852 | 0.135633 | 0.564674 | −0.382954 | −0.798283 | 1.000000 | 0.146023 |
| BChE | −0.356166 | −0.289807 | 0.765542 | −0.397344 | 0.705555 | −0.173784 | −0.404985 | 0.174840 | −0.456827 | −0.319621 | −0.228740 | 0.146023 | 1.000000 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kobus-Cisowska, J.; Szulc, P.; Szczepaniak, O.; Dziedziński, M.; Szymanowska, D.; Szymandera-Buszka, K.; Goryńska-Goldmann, E.; Gazdecki, M.; Telichowska, A.; Ligaj, M. Variability of Hordeum vulgare L. Cultivars in Yield, Antioxidant Potential, and Cholinesterase Inhibitory Activity. Sustainability 2020, 12, 1938. https://doi.org/10.3390/su12051938
Kobus-Cisowska J, Szulc P, Szczepaniak O, Dziedziński M, Szymanowska D, Szymandera-Buszka K, Goryńska-Goldmann E, Gazdecki M, Telichowska A, Ligaj M. Variability of Hordeum vulgare L. Cultivars in Yield, Antioxidant Potential, and Cholinesterase Inhibitory Activity. Sustainability. 2020; 12(5):1938. https://doi.org/10.3390/su12051938
Chicago/Turabian StyleKobus-Cisowska, Joanna, Piotr Szulc, Oskar Szczepaniak, Marcin Dziedziński, Daria Szymanowska, Krystyna Szymandera-Buszka, Elżbieta Goryńska-Goldmann, Michał Gazdecki, Aleksandra Telichowska, and Marta Ligaj. 2020. "Variability of Hordeum vulgare L. Cultivars in Yield, Antioxidant Potential, and Cholinesterase Inhibitory Activity" Sustainability 12, no. 5: 1938. https://doi.org/10.3390/su12051938
APA StyleKobus-Cisowska, J., Szulc, P., Szczepaniak, O., Dziedziński, M., Szymanowska, D., Szymandera-Buszka, K., Goryńska-Goldmann, E., Gazdecki, M., Telichowska, A., & Ligaj, M. (2020). Variability of Hordeum vulgare L. Cultivars in Yield, Antioxidant Potential, and Cholinesterase Inhibitory Activity. Sustainability, 12(5), 1938. https://doi.org/10.3390/su12051938








