Factors Influencing Usability of Rehabilitation Robotic Devices for Lower Limbs
Abstract
1. Introduction
1.1. Increasing Demands for Rehabilitation Robotic Devices
1.2. Usability and Users of Medical Devices
1.3. Usability Assessment of Medical Devices by SMEs
2. Materials and Methods
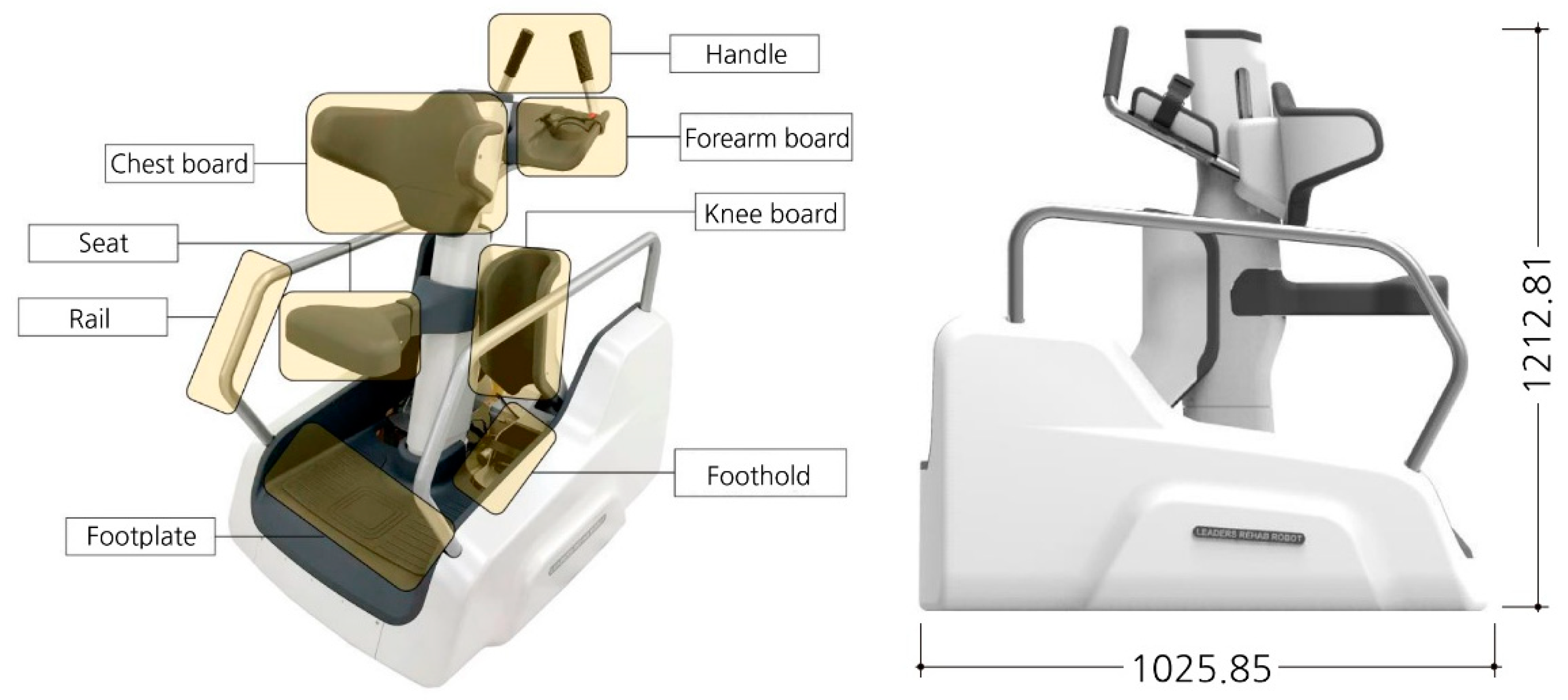
2.1. Materials and Subjects
2.2. Questionnaire Development
2.3. Survey Administration
3. Results
3.1. Characteristics of Respondents
3.2. Identifying Factors Influencing Usability
3.3. Differences in Usability Evaluation between User Groups
4. Discussion
4.1. Factors Influencing Usability of Medical Devices for Lower Limbs
4.2. Discrepancy in Usability Evaluation among User Groups
4.3. Contribution of Expected Usability to Improved Visual Design
5. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Laut, J.; Porfiri, M.; Raghavan, P. The present and future of robotic technology in rehabilitation. Curr. Phys. Med. Rehabil. Rep. 2016, 4, 312–319. [Google Scholar] [CrossRef] [PubMed]
- Kim, T. Identifying Stakeholders and Interactions in the Dementia Café in Seongju through Empathic Service Design Approaches. J. Open Innov. Technol. Mark. Complex. 2018, 4, 28. [Google Scholar] [CrossRef]
- Statistics Korea. 2018 The Statistics of the Elderly. Available online: http://kostat.go.kr/portal/korea/kor_nw/1/1/index.board?bmode=read&aSeq=370779 (accessed on 20 August 2019).
- Hayat, H.; Lock, R.; Murray, I. Measuring software usability. In Proceedings of the BCS Software Quality Management Conference, Loughborough University, Loughborough, UK, 30 March 2015. [Google Scholar]
- Meng, W.; Liu, Q.; Zhou, Z.; Ai, Q.; Sheng, B.; Xie, S.S. Recent development of mechanisms and control strategies for robot-assisted lower limb rehabilitation. Mechatronics 2015, 31, 132–145. [Google Scholar] [CrossRef]
- Kim, K. Policy Research for Cultivating Physiotherapists, Korea Health Personnel Licensing Examination Institute. 2017. Available online: https://rnd.kuksiwon.or.kr/ (accessed on 20 August 2019).
- Zhou, Z.; Meng, W.; Ai, Q.; Liu, Q.; Wu, X. Practical –velocity tracking control of a parallel robot based on fuzzy adaptive algorithm. Adv. Mech. Eng. 2013, 5, 574896. [Google Scholar] [CrossRef]
- Nef, T.; Mihelj, M.; Kiefer, G.; Perndl, C.; Muller, R.; Riener, R. ARM in-Exoskeleton for arm therapy in stroke patients. In Proceedings of the 2007 IEEE 10th International Conference on Rehabilitation Robotics, Noordwijk, The Netherlands, 13–15 June 2007; pp. 68–74. [Google Scholar]
- Lu, R.; Li, Z.; Su, C.Y.; Xue, A. Development and learning control of a human limb with a rehabilitation exoskeleton. IEEE Trans. Ind. Electron. 2013, 61, 3776–3785. [Google Scholar] [CrossRef]
- Song, W.K. Trends in rehabilitation robots and their translational research in National Rehabilitation Center of Korea. Biomed. Eng. Lett. 2016, 6, 1–9. [Google Scholar] [CrossRef]
- Kang, C. The Market of Rehabilitation Robotics Rapidly Growing but Asking a Critical Technology. Available online: http://e-healthnews.com/ (accessed on 24 August 2019).
- Dollar, A.M.; Herr, H. Lower extremity exoskeletons and active orthoses: Challenges and state-of-the-art. IEEE Trans. Robot. 2008, 24, 144–158. [Google Scholar] [CrossRef]
- Song, W.K. Technology trend of rehabilitation robots. J. Korean Inst. Commun. Inf. Sci. 2016, 33, 10–18. [Google Scholar]
- Lee, B.; Kim, K. Technology trend and industrial prospect of surgical robots. KEIT PD Issue Rep. 2017, 17-3, 62–76. [Google Scholar]
- Seo, G. Situation of exports of medical devices of South Korea; focusing on recent five years. KHIDI Brief. 2014, 151, 1–8. [Google Scholar]
- Ministry of Trade, Industry and Energy. Development Plan for the Robot Industry of South Korea. 2019. Available online: http://www.motie.go.kr/motie/ne/presse/press2/bbs/bbsView.do?bbs_cd_n=81&bbs_seq_n=161458 (accessed on 30 August 2019).
- Ministry of Trade, Industry and Energy. Research of Competitiveness of Robot Industry. 2017. Available online: http://www.prism.go.kr/homepage/entire/retrieveEntireDetail.do?research_id=1450000-201700201 (accessed on 30 August 2019).
- Qian, Z.; Bi, Z. Recent development of rehabilitation robots. Adv. Mech. Eng. 2015, 7, 1–11. [Google Scholar] [CrossRef]
- Lemke, M.R.; Winters, J. Removing barriers to medical devices for users with impairments. Ergon. Des. Q. Hum. Factors Appl. 2008, 16, 18–25. [Google Scholar] [CrossRef]
- Brockhoff, K. Customers’ perspectives of involvement in new product development. Int. J. Technol. Manag. 2003, 26, 464–481. [Google Scholar] [CrossRef]
- Shah, S.G.S.; Robinson, I. User involvement in medical device technology development and assessment: Structured literature review. Int. J. Health Care Qual. Assur. Inc. Leadersh. Health Serv. 2006, 19, 500–515. [Google Scholar] [CrossRef]
- Gosbee, J. Human factors engineering and patient safety. Qual. Saf. Health Care 2002, 11, 352–354. [Google Scholar] [CrossRef]
- Gallivan, M.J.; Keil, M. The user–developer communication process: A critical case study. Inf. Syst. J. 2003, 13, 37–68. [Google Scholar] [CrossRef]
- Wegge, K.P.; Zimmermann, D. Accessibility, usability, safety, ergonomics: Concepts, models, and differences. In Universal Access in Human-Computer Interaction. Interaction Techniques and Environments, Proceedings of the International Conference on Universal Access in Human-Computer Interaction, Toronto, ON, Canada, 17–22 July 2016; Antona, M., Stephanidis, C., Eds.; Springer: Berlin/Heidelberger, Germany, 2016; pp. 294–301. [Google Scholar]
- Martin, J.L.; Norris, B.J.; Murphy, E.; Crowe, J.A. Medical device development: The challenge for ergonomics. Appl. Ergon. 2008, 39, 271–283. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Medical Device Regulations: Global Overview and Guiding Principles; WHO: Geneva, Switzerland, 2003; pp. 1–8. [Google Scholar]
- Samore, M.H.; Evans, R.S.; Lassen, A.; Gould, P.; Lloyd, J.; Gardner, R.M.; Abouzelof, R.; Taylor, C.; Woodbury, D.A.; Willy, M.; et al. Surveillance of medical device–related hazards and adverse events in hospitalized patients. J. Am. Med Assoc. 2004, 291, 325–334. [Google Scholar] [CrossRef]
- Kaye, R.; Crowley, J. Medical Device Use-Safety: Incorporating Human Factors Engineering into Risk Management; Food and Drug Administration, Guidance Document; Center for Devices and Radiological Health, Department of Health and Human Services, U.S.: Silver Spring, MD, USA, 2000; pp. 1–33. Available online: http://www.fda.gov/cdrh/humfac/1497.pdf (accessed on 10 March 2006).
- Department of Health. EC Medical Devices Directives: Guidance Notes for Manufacturers of Custom-Made Devices; Department of Health, Guidance Document No. 9; Medicines and Healthcare Products Regulatory Agency (MHRA): London, UK, 2003; pp. 1–12.
- Therapeutic Goods Administration. Medical Devices, TGA News-Issue; Therapeutic Goods Administration: Symonston, ACT, Australia, 2003; Volume 42 (November 2003), pp. 3–4.
- Shah, S.G.S.; Robinson, I. Medical device technologies: Who is the user? Int. J. Healthc. Technol. Manag. 2008, 9, 181–197. [Google Scholar] [CrossRef]
- Weinger, M.B.; Gardner-Bonneau, D.J.; Wiklund, M.E. General Principles. In Handbook of Human Factors in Medical Device Design; Weinger, M.B., Wiklund, M.E., Gardner-Bonneau, D.J., Eds.; CRC Press: Boca Raton, FL, USA, 2010; pp. 1–22. [Google Scholar]
- National Research Council. The Role of Human Factors in Home Health Care: Workshop Summary; National Academies Press: Washington, DC, USA, 2010; pp. 9–28. [Google Scholar]
- Baker, G.R.; Norton, P.G.; Flintoft, V.; Blais, R.; Brown, A.; Cox, J.; Etchells, E.; Ghali, W.A.; Hebert, P.; Majumdar, S.R.; et al. The Canadian adverse events study: The incidence of adverse events among hospital patients in Canada. Can. Med Assoc. J. 2004, 170, 1678–1686. [Google Scholar] [CrossRef]
- Martin, J.L.; Craven, M.P.; Norris, B.J. MATCH: A new industry-focused approach to medical device development. In Proceedings of the International Conference of Healthcare systems Ergonomics and Patient Safety, Florence, Italy, 30 March–2 April 2005; Tartaglia, R., Bagnara, S., Bellandi, T., Albolino, S., Eds.; Taylor & Francis: London, UK, 2005; pp. 294–297. [Google Scholar]
- Hass, C.; Berlin, D. Usability testing medical devices: A practical guide to minimizing risk and maximizing success. In Proceedings of the International Conference of Design, User Experience, and Usability; Springer: Berlin/Heidelberg, Germany, 2013; pp. 407–416. [Google Scholar]
- Choi, S. The Integrated Framework for Developing Design of and Evaluating Usability of Medical Devices. Ph.D. Thesis, Chung-Ang University, Ansan, Korea, 2018. [Google Scholar]
- Dumas, J.S.; Redish, J.C. A Practical Guide to Usability Testing; Intellect: Portland, OR, USA, 1993; pp. 119–134. [Google Scholar]
- Evans, G. Accessibility and user needs in transport design. In Proceedings of the International Conference on Inclusive Design, Royal College of Art, London, UK, 5–8 April 2005; pp. 32–44. [Google Scholar]
- Reed, D.; Monk, A. Inclusive design: Beyond capabilities towards context of use. Univers. Access Inf. Soc. 2011, 10, 295–305. [Google Scholar] [CrossRef]
- Amoore, J.; Ingram, P. Learning from adverse incidents involving medical devices. BMJ Nurs. Stand. 2002, 32517, 27241–27546. [Google Scholar]
- Bevan, N. Measuring usability as quality of use. Softw. Qual. J. 1995, 4, 115–130. [Google Scholar] [CrossRef]
- Cho, J.S.; Kwon, T.K.; Hong, J.P. A study of evaluation index development of healthcare rehabilitation device design. Korean J. Sci. Emot. Sensib. 2014, 17, 129–142. [Google Scholar] [CrossRef]
- Brooke, J. SUS: A retrospective. J. Usability Stud. 2013, 8, 29–40. [Google Scholar]
- Nielsen, J.; Landauer, T.K. A mathematical model of the finding of usability problems. In Proceedings of the INTERACT’93 and CHI’93 Conference on Human Factors in Computing Systems, Amsterdam, The Netherlands, 24–29 April 1993; ACM: New York, NY, USA, 1993; pp. 206–213. [Google Scholar]
- ISO DIS 9241-11 Ergonomic Requirements for Office Work with Visual Display Terminals (VDT)s—Part 11 Guidance on Usability, International Organization for Standardization. Available online: https://isotc.iso.org/livelink/livelink?func=ll&objId=4230456&objAction=browse&sort=subtype (accessed on 10 March 2006).
- Peres, S.C.; Pham, T.; Phillips, R. Validation of the system usability scale (SUS) SUS in the wild. In Proceedings of the Human Factors and Ergonomics Society Annual Meeting, San Diego, CA, USA, 30 September–4 October 2013; SAGE Publications: Los Angeles, CA, USA; pp. 192–196. [Google Scholar]
- Schmidt, H.; Werner, C.; Bernhardt, R.; Hesse, S.; Krüger, J. Gait rehabilitation machines based on programmable footplates. J. Neuroeng. Rehabil. 2007, 4, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Carr, J.H.; Shepherd, R.B. A Motor Relearning Programme for Stroke; Aspen Publishers: New York, NY, USA, 1987. [Google Scholar]
- Chan, D.Y.L.; Chan, C.C.H.; Au, D.K.S. Motor relearning programme for stroke patients: A randomized controlled trial. Clin. Rehabil. 2006, 20, 191–200. [Google Scholar] [CrossRef] [PubMed]
- Sharples, S.; Martin, J.; Lang, A.; Craven, M.; O’Neill, S.; Barnett, J. Medical device design in context: A model of user–device interaction and consequences. Displays 2012, 33, 221–232. [Google Scholar] [CrossRef]
- Díaz, I.; Gil, J.J.; Sánchez, E. Lower-limb robotic rehabilitation: Literature review and challenges. J. Robot. 2011, 2011, 1–11. [Google Scholar] [CrossRef]
- Nielsen, J. Guerilla HCI: Using discount usability engineering to penetrate the intimidation barrier. In Cost—Justifying Usability; Bias, R.G., Mayhew, D.J., Eds.; Academic Press: Boston, MA, USA, 1994; pp. 242–272. [Google Scholar]
- Donahue, G. Usability and the bottom line. IEEE Softw. 2001, 18, 31–37. [Google Scholar] [CrossRef]
- Siegel, D. The business case for user-centered design: Increasing your power of persuasion. Interactions 2003, 10, 30–36. [Google Scholar] [CrossRef]
- Virzi, R.A. Refining the test phase of usability evaluation: How many subjects is enough? Hum. Factors 1992, 34, 457–468. [Google Scholar] [CrossRef]
- Nielsen, J. Estimating the number of subjects needed for a thinking aloud test. Int. J. Hum.-Comput. Stud. 1994, 41, 385–397. [Google Scholar] [CrossRef]
- Spool, J.; Schroeder, W. Testing web sites: Five users is nowhere near enough. In Proceedings of the Extended abstracts on Human Factors in Computing Systems (CHI 2001), Washington, DC, USA, 31 March–5 April 2001; ACM Press: New York, NY, USA, 2001; pp. 285–286. [Google Scholar]
- Faulkner, L. Beyond the five-user assumption: Benefits of increased sample sizes in usability testing. Behavior Research Methods. Instrum. Comput. 2003, 35, 379–383. [Google Scholar] [CrossRef]
- Woolrych, A.; Cockton, G. Why and when five test users aren’t enough. In Proceedings of the IHM-HCI 2001 Conference, Lille, France, 10–14 September 2001; Cépaduès Editions: Toulouse, FR, USA, 2001; pp. 105–108. [Google Scholar]
- Glen, J.M.W.; Lord, M. New product development processes within the UK medical device industry. Med. Eng. Phys. 1996, 18, 670–676. [Google Scholar] [CrossRef]
- Lehoux, P.; Saint-Arnaud, J.; Richard, L. The use of technology at home: What patient manuals say and sell vs. what patients face and fear. Sociol. Health Illn. 2004, 26, 617–644. [Google Scholar] [CrossRef]
- Ward, J.R.; Clarkson, J.P. An analysis of medical device-related errors: Prevalence and possible solutions. J. Med. Eng. Technol. 2004, 28, 2–21. [Google Scholar] [CrossRef]
- Kessner, M.; Wood, J.; Dillon, R.F.; West, R.L. On the reliability of usability testing. In Proceedings of the Extended Abstracts on Human Factors in Computing Systems(CHI’01), Seattle, WA, USA, 31 March–5 April 2001; ACM Press: New York, NY, USA, 2001; pp. 97–98. [Google Scholar]
- Walker, M.; Takayama, L.; Landay, J.A. High-fi51delity or low-fidelity, paper or computer? Choosing attributes when testing web prototypes. In Proceedings of the Human Factors and Ergonomics Society Annual Meeting, Baltimore, MD, USA, 30 September–4 October 2013; Sage Publications: Los Angeles, CA, USA, 2002; pp. 661–665. [Google Scholar]
- Van Schaik, P.; Ling, J. Modelling user experience with web sites: Usability, hedonic value, beauty and goodness. Interact. Comput. 2008, 20, 419–432. [Google Scholar] [CrossRef]
- Kim, H.; Fesenmaier, D.R. Persuasive design of destination web sites: An analysis of first impression. J. Travel Res. 2008, 47, 3–13. [Google Scholar] [CrossRef]
- Lindgaard, G.; Dudek, C.; Sen, D.; Sumegi, L.; Noonan, P. An exploration of relations between visual appeal, trustworthiness and perceived usability of homepages. ACM Trans. Comput.-Hum. Interact. 2011, 18, 1–30. [Google Scholar] [CrossRef]
- Ben-Bassat, T.; Meyer, J.; Tractinsky, N. Economic and subjective measures of the perceived value of aesthetics and usability. ACM Trans. Comput.-Hum. Interact. 2006, 13, 210–234. [Google Scholar] [CrossRef]
- Tractinsky, N.; Katz, A.S.; Ikar, D. What is beautiful is usable. Interact. Comput. 2000, 13, 127–145. [Google Scholar] [CrossRef]
- Lee, S.; Koubek, R.J. Understanding user preferences based on usability and aesthetics before and after actual use. Interact. Comput. 2010, 22, 530–543. [Google Scholar] [CrossRef]
- Katz, A. Aesthetics, usefulness and performance in user—Search-engine interaction. J. Appl. Quant. Methods 2010, 5, 424–445. [Google Scholar]
- Lindgaard, G.; Dillon, R.; Trbovich, P.; White, R.; Fernandes, G.; Lundahl, S.; Pinnamaneni, A. User Needs Analysis and requirements engineering: Theory and practice. Interact. Comput. 2006, 18, 47–70. [Google Scholar] [CrossRef]
- Sonderegger, A.; Sauer, J. The influence of design aesthetics in usability testing: Effects on user performance and perceived usability. Appl. Ergon. 2010, 41, 403–410. [Google Scholar] [CrossRef] [PubMed]
- Tuch, A.N.; Roth, S.P.; Hornbæk, K.; Opwis, K.; Bargas-Avila, J.A. Is beautiful really usable? Toward understanding the relation between usability, aesthetics, and affect in HCI. Comput. Hum. Behav. 2012, 28, 1596–1607. [Google Scholar] [CrossRef]
- Mugge, R.; Schoormans, J.P. Product design and apparent usability. The influence of novelty in product appearance. Appl. Ergon. 2012, 43, 1081–1088. [Google Scholar] [CrossRef]
- Dion, K.; Berscheid, E.; Walster, E. What is beautiful is good. J. Personal. Soc. Psychol. 1972, 24, 285–290. [Google Scholar] [CrossRef]
- Gupta, A.K.; Dey, A.R.; Shinde, C.; Mahanta, H.; Patel, C.; Patel, R. Theory of open inclusive innovation for reciprocal, responsive and respectful outcomes: Coping creatively with climatic and institutional risks. J. Open Innov. Technol. Mark. Complex. 2016, 2, 16. [Google Scholar] [CrossRef]
- Gupta, A.; Dey, A.; Singh, G. Connecting corporations and communities: Towards a theory of social inclusive open innovation. J. Open Innov. Technol. Mark. Complex. 2017, 3, 17. [Google Scholar] [CrossRef]
| Approach | Dimensions and Factors to Measure Usability |
|---|---|
| QUIS | Overall satisfaction, interface (screen, terminology and system feedback, learning factors, system capabilities, technical manuals, online tutorials, multimedia, teleconferencing, and software installation) |
| SUMI | Affect, efficiency, learnability, helpfulness, control |
| PSSUQ | System usefulness, information quality, interface quality, overall satisfaction |
| SUS | ease of use (learnability and usability) |
| Question Categories | Attributes | Number of Questions |
|---|---|---|
| Demographic Information | Age | 4 |
| Gender | ||
| Period of career | ||
| Prior experience | ||
| Generality across devices | Effectiveness | 3 |
| Efficiency | 4 | |
| Attractiveness | 3 | |
| Satisfaction | 5 | |
| Device-specific questions | Rail | 5 |
| Seat | 6 | |
| Chest board | 6 | |
| Knee Board | 3 | |
| Handle | 3 | |
| Handle strap | 2 | |
| Armrest | 4 | |
| Footrest | 4 | |
| Footrest strap | 2 | |
| Footplate | 4 |
| Classification | UX Design Professionals | Rehab Professionals | Lay People | Total Users | |
|---|---|---|---|---|---|
| Age | 21–30 | 15 | 15 | 33 | 63 (32.14%) |
| 31–40 | 24 | 15 | 29 | 68 (34.69%) | |
| 41–50 | 12 | 14 | 15 | 41 (20.9%) | |
| ≥51–60 | 0 | 7 | 9 | 16 (8.16%) | |
| ≥61 | 0 | 0 | 8 | 8 (4.08%) | |
| Total | 51 | 51 | 94 | 196 (100%) | |
| Sex | Male | 23 | 36 | 24 | 83 (42.35%) |
| Female | 28 | 15 | 70 | 113 (57.65%) | |
| Total | 51 | 51 | 94 | 196 (100%) | |
| Years of professional experience | 1–5 years | 24 | 20 | N/A | N/A |
| 6–10 years | 23 | 10 | N/A | N/A | |
| ≥10 years | 4 | 21 | N/A | N/A | |
| Total | 51 | 51 | N/A | N/A | |
| Factor Interpretation | Variable No. | Factor Loading | Communality | Eigen Value | % of Variance Explained | Cronbach’s Alpha |
|---|---|---|---|---|---|---|
| Piggybacking | 42 | 0.688 | 0.664 | 6.446 | 13.429 | 0.934 |
| 29 | 0.622 | 0.733 | ||||
| 44 | 0.608 | 0.782 | ||||
| 30 | 0.605 | 0.721 | ||||
| 43 | 0.584 | 0.497 | ||||
| 36 | 0.565 | 0.607 | ||||
| 31 | 0.547 | 0.642 | ||||
| 25 | 0.539 | 0.632 | ||||
| 26 | 0.527 | 0.712 | ||||
| 38 | 0.524 | 0.542 | ||||
| 24 | 0.524 | 0.646 | ||||
| Transferring | 48 | 0.801 | 0.800 | 5.297 | 11.034 | 0.905 |
| 47 | 0.769 | 0.782 | ||||
| 50 | 0.694 | 0.731 | ||||
| 51 | 0.681 | 0.789 | ||||
| 40 | 0.629 | 0.649 | ||||
| 49 | 0.598 | 0.591 | ||||
| 46 | 0.511 | 0.497 | ||||
| 39 | 0.452 | 0.542 | ||||
| Supporting | 19 | 0.742 | 0.731 | 5.000 | 10.417 | 0.911 |
| 22 | 0.731 | 0.739 | ||||
| 20 | 0.715 | 0.727 | ||||
| 18 | 0.658 | 0.674 | ||||
| 21 | 0.578 | 0.634 | ||||
| 23 | 0.545 | 0.732 | ||||
| 37 | 0.441 | 0.607 | ||||
| Sitting on/off | 01 | 0.613 | 0.645 | 3.713 | 7.736 | 0.886 |
| 15 | 0.591 | 0.641 | ||||
| 16 | 0.539 | 0.734 | ||||
| 17 | 0.536 | 0.732 | ||||
| 03 | 0.515 | 0.599 | ||||
| 08 | 0.477 | 0.695 | ||||
| 10 | 0.460 | 0.585 | ||||
| Holding the body | 35 | 0.723 | 0.725 | 3.274 | 6.822 | 0.857 |
| 28 | 0.721 | 0.710 | ||||
| 27 | 0.564 | 0.609 | ||||
| 32 | 0.506 | 0.639 | ||||
| 34 | 0.490 | 0.714 | ||||
| Visual pertinence | 02 | 0.771 | 0.729 | 3.233 | 6.736 | 0.806 |
| 07 | 0.756 | 0.733 | ||||
| 11 | 0.683 | 0.708 | ||||
| Use confidence | 06 | 0.808 | 0.804 | 3.184 | 6.634 | 0.851 |
| 04 | 0.730 | 0.754 | ||||
| 05 | 0.631 | 0.709 | ||||
| 09 | 0.459 | 0.617 | ||||
| Safety | 12 | 0.765 | 0.771 | 2.592 | 5.399 | 0.793 |
| 13 | 0.680 | 0.740 | ||||
| 14 | 0.596 | 0.637 |
| Factors | Groups | N | Mean | SD | F | p | |
|---|---|---|---|---|---|---|---|
| General Qualities | Use confidence | UX professionals (a) | 51 | 3.73 | 0.82 | 2.34 | 0.099 |
| Rehab professionals (b) | 51 | 3.66 | 0.90 | ||||
| Lay people (c) | 94 | 3.43 | 0.74 | ||||
| Total | 196 | 3.63 | 0.83 | ||||
| Safety | UX professionals (a) | 51 | 3.88 | 0.81 | 0.12 | 0.885 | |
| Rehab professionals (b) | 51 | 3.83 | 0.87 | ||||
| Lay people (c) | 94 | 3.91 | 0.75 | ||||
| Total | 196 | 3.87 | 0.80 | ||||
| Visual pertinence | UX professionals (a) | 94 | 3.93 | 0.80 | 5.62 | 0.004 | |
| Rehab professionals (b) | 51 | 3.66 | 0.82 | ||||
| Lay people (c) | 51 | 3.48 | 0.71 | ||||
| Total | 196 | 3.74 | 0.80 | ||||
| Device Specifics | Piggybacking | UX professionals (a) | 51 | 3.75 | 0.7475 | 2.74 | 0.067 |
| Rehab professionals (b) | 51 | 3.66 | 0.7172 | ||||
| Lay people (c) | 94 | 3.46 | 0.7401 | ||||
| Total | 196 | 3.65 | 0.7444 | ||||
| Transferring | UX professionals (a) | 51 | 3.38 | 0.8951 | 3.11 | 0.047 | |
| Rehab professionals (b) | 51 | 3.28 | 0.8058 | ||||
| Lay people (c) | 94 | 3.03 | 0.6787 | ||||
| Total | 196 | 3.26 | 0.8296 | ||||
| Supporting the body | UX professionals (a) | 94 | 4.04 | 0.7026 | 1.83 | 0.164 | |
| Rehab professionals (b) | 51 | 3.95 | 0.6808 | ||||
| Lay people (c) | 51 | 3.81 | 0.7032 | ||||
| Total | 196 | 3.96 | 0.7001 | ||||
| Sitting on/off | UX professionals (a) | 51 | 3.50 | 0.8525 | 1.91 | 0.150 | |
| Rehab professionals (b) | 51 | 3.50 | 0.8448 | ||||
| Lay people (c) | 94 | 3.24 | 0.6811 | ||||
| Total | 196 | 3.43 | 0.8133 | ||||
| Holding the Body | UX professionals (a) | 51 | 3.73 | 0.8371 | 6.72 | 0.002 | |
| Rehab professionals (b) | 51 | 3.62 | 0.7876 | ||||
| Lay people (c) | 94 | 3.24 | 0.6227 | ||||
| Total | 196 | 3.58 | 0.7962 | ||||
© 2020 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, T. Factors Influencing Usability of Rehabilitation Robotic Devices for Lower Limbs. Sustainability 2020, 12, 598. https://doi.org/10.3390/su12020598
Kim T. Factors Influencing Usability of Rehabilitation Robotic Devices for Lower Limbs. Sustainability. 2020; 12(2):598. https://doi.org/10.3390/su12020598
Chicago/Turabian StyleKim, Taesun. 2020. "Factors Influencing Usability of Rehabilitation Robotic Devices for Lower Limbs" Sustainability 12, no. 2: 598. https://doi.org/10.3390/su12020598
APA StyleKim, T. (2020). Factors Influencing Usability of Rehabilitation Robotic Devices for Lower Limbs. Sustainability, 12(2), 598. https://doi.org/10.3390/su12020598




