Plant Diversity and Agroecosystem Function in Riparian Agroforests: Providing Ecosystem Services and Land-Use Transition
Abstract
1. Introduction
2. Materials and Methods
2.1. Site Description and Study Design
2.2. Plant Functional Traits
2.3. Plant Diversity Metrics
2.4. Ecosystem Processes
2.5. Statistical Analysis
3. Results
3.1. Taxonomic and Phylogenetic Diversity of Plant Communities among Riparian Systems
3.2. Functional Diversity of Plant Communities among Riparian Systems
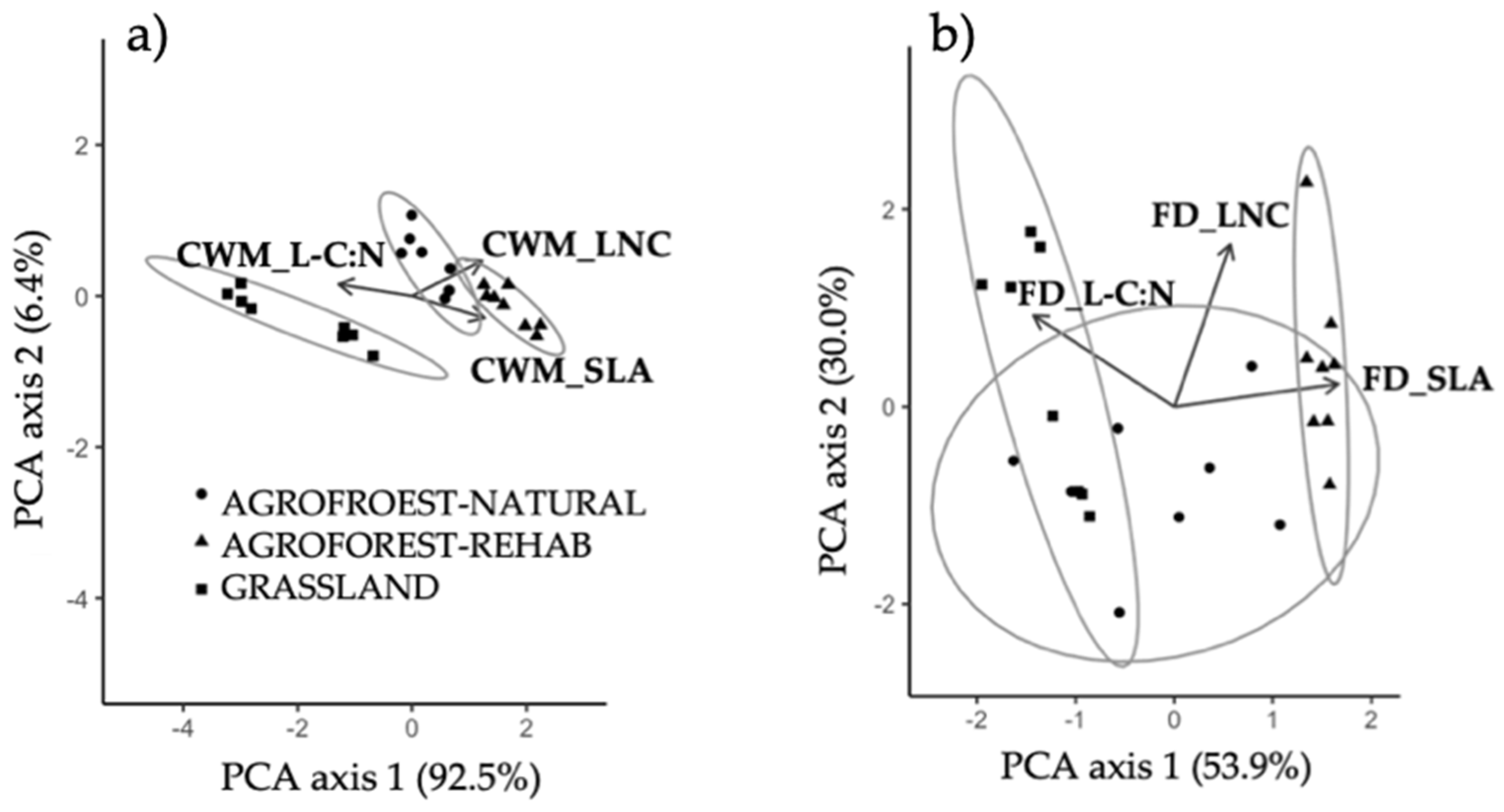
3.3. Multivariate Plant Community Variation
3.4. Relationships between Plant Community Diversity and Soil C and N Processes
4. Discussion
4.1. Taxonomic and Functional Biodiversity in Riparian Agroforestry Systems
4.2. Riparian System Functional Diversity
4.3. Biodiversity and Ecosystem Processes in Riparian Agroforestry Systems
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Jose, S. Agroforestry for ecosystem services and environmental benefits: An overview. Agrofor. Syst. 2009, 76, 1–10. [Google Scholar] [CrossRef]
- Tscharntke, T.; Klein, A.M.; Kruess, A.; Steffan-Dewenter, I.; Thies, C. Landscape perspectives on agricultural intensification and biodiversity—Ecosystem service management. Ecol. Lett. 2005, 8, 857–874. [Google Scholar] [CrossRef]
- Maseyk, F.J.F.; Dominati, E.J.; White, T.; Mackay, A.D. Farmer perspectives of the on-farm and off-farm pros and cons of planted multifunctional riparian margins. Land Use Policy 2017, 61, 160–170. [Google Scholar] [CrossRef]
- Sullivan, W.C.; Anderson, O.M.; Lovell, S.T. Agricultural buffers at the rural-urban fringe: An examination of approval by farmers, residents, and academics in the Midwestern United States. Landsc. Urban Plan. 2004, 69, 299–313. [Google Scholar] [CrossRef]
- Osborne, L.L.; Kovacic, D.A. Riparian vegetated buffer strips in water-quality restoration and stream management. Freshw. Biol. 1993, 29, 243–258. [Google Scholar] [CrossRef]
- Zhang, X.; Liu, X.; Zhang, M.; Dahlgren, R.A.; Eitzel, M. A review of vegetated buffers and a meta-analysis of their mitigation efficacy in reducing nonpoint source pollution. J. Environ. Qual. 2010, 39, 76–84. [Google Scholar] [CrossRef]
- Mayer, P.M.; Reynolds, S.K.; McCutchen, M.D.; Canfield, T.J. Meta-analysis of nitrogen removal in riparian buffers. J. Environ. Qual. 2007, 36, 1172–1180. [Google Scholar] [CrossRef]
- Dosskey, M.G.; Vidon, P.; Gurwick, N.P.; Allan, C.J.; Duval, T.P.; Lowrance, R. The role of riparian vegetation in protecting and improving chemical water quality in streams. J. Am. Water Resour. Assoc. 2010, 46, 261–277. [Google Scholar] [CrossRef]
- Hazlett, P.; Broad, K.; Gordon, A.; Sibley, P.; Buttle, J.; Larmer, D. The importance of catchment slope to soil water N and C concentrations in riparian zones: Implications for riparian buffer width. Can. J. For. Res. 2008, 38, 16–30. [Google Scholar] [CrossRef]
- Jacinthe, P.A.; Vidon, P. Hydro-geomorphic controls of greenhouse gas fluxes in riparian buffers of the White River watershed, IN (USA). Geoderma 2017, 301, 30–41. [Google Scholar] [CrossRef]
- Mander, Ü.; Maddison, M.; Soosaar, K.; Teemusk, A.; Kanal, A.; Uri, V.; Truu, J. The impact of a pulsing groundwater table on greenhouse gas emissions in riparian grey alder stands. Environ. Sci. Pollut. Res. 2015, 22, 2360–2371. [Google Scholar] [CrossRef] [PubMed]
- Fortier, J.; Truax, B.; Gagnon, D.; Lambert, F. Biomass carbon, nitrogen and phosphorus stocks in hybrid poplar buffers, herbaceous buffers and natural woodlots in the riparian zone on agricultural land. J. Environ. Manag. 2015, 154, 333–345. [Google Scholar] [CrossRef] [PubMed]
- Cadotte, M.W.; Carscadden, K.; Mirotchnick, N. Beyond species: Functional diversity and the maintenance of ecological processes and services. J. Appl. Ecol. 2011, 48, 1079–1087. [Google Scholar] [CrossRef]
- de Bello, F.; Lavorel, S.; Díaz, S.; Harrington, R.; Cornelissen, J.H.C.; Bardgett, R.D.; Berg, M.P.; Cipriotti, P.; Feld, C.K.; Hering, D.; et al. Towards an assessment of multiple ecosystem processes and services via functional traits. Biodivers. Conserv. 2010, 19, 2873–2893. [Google Scholar] [CrossRef]
- Díaz, S.; Lavorel, S.; de Bello, F.; Quétier, F.; Grigulis, K.; Robson, T.M. Incorporating plant functional diversity effects in ecosystem service assessments. Proc. Natl. Acad. Sci. USA 2007, 104, 20684–20689. [Google Scholar] [CrossRef] [PubMed]
- Wright, I.J.; Reich, P.B.; Westoby, M.; Ackerly, D.D.; Baruch, Z.; Bongers, F.; Cavender-bares, J.; Chapin, T.; Cornelissen, J.H.C.; Diemer, M.; et al. The worldwide leaf economics spectrum. Nature 2004, 428, 821–827. [Google Scholar] [CrossRef]
- Violle, C.; Navas, M.L.; Vile, D.; Kazakou, E.; Fortunel, C.; Hummel, I.; Garnier, E. Let the concept of trait be functional! Oikos 2007, 116, 882–892. [Google Scholar] [CrossRef]
- Grime, J. Befits of plant diversity to ecosystems: Immediate, filter and founder effects. J. Ecol. 1998, 86, 902–910. [Google Scholar] [CrossRef]
- Wasof, S.; Lenoir, J.; Hattab, T.; Jamoneau, A.; Gallet-Moron, E.; Ampoorter, E.; Saguez, R.; Bennsadek, L.; Bertrand, R.; Valdès, A.; et al. Dominance of individual plant species is more important than diversity in explaining plant biomass in the forest understorey. J. Veg. Sci. 2018, 29, 521–531. [Google Scholar] [CrossRef]
- Petchey, O.L.; Gaston, K.J. Functional diversity (FD), species richness and community composition. Ecol. Lett. 2002, 5, 402–411. [Google Scholar] [CrossRef]
- Oelbermann, M.; Gordon, A.M. Retention of leaf litter in streams from riparian plantings in southern Ontario, Canada. Agrofor. Syst. 2001, 53, 323–331. [Google Scholar] [CrossRef]
- Dickinson, T.; Metsger, D.; Bull, J.; Dickinson, R. Wildflowers of Ontario; Royal Ontario Museum: Toronto, ON, Canada, 2004. [Google Scholar]
- Chambers, B.; Legasy, K.; Bentley, C. V Forest Plants of Central Ontario; Lone Pine Publishing: Edmonton, Canada, 1996. [Google Scholar]
- Abràmofff, M.D.; Magalhães, P.J.; Ram, S.J. Image processing with ImageJ: Part II. Biophotonics Int. 2004, 11, 36–42. [Google Scholar]
- Webb, C.O.; Donoghue, M.J. Phylomatic: Tree assembly for applied phylogenetics. Mol. Ecol. Notes 2005, 5, 181–183. [Google Scholar] [CrossRef]
- Webb, C.O.; Ackerly, D.D.; Kembel, S.W. Phylocom: Software for the analysis of phylogenetic community structure and trait evolution. Bioinformatics 2008, 24, 2098–2100. [Google Scholar] [CrossRef] [PubMed]
- Wikstrom, N.; Savolainen, V.; Chase, M.W. Evolution of the angiosperms: Calibrating the family tree. Proc. Biol. Sci. 2001, 2668, 221–2220. [Google Scholar] [CrossRef] [PubMed]
- Kembel, S.W.; Cowan, P.D.; Helmus, M.R.; Cornwell, W.K.; Morlon, H.; Ackerly, D.D.; Blomberg, S.P.; Webb, C.O. Picante: R tools for integrating phylogenies and ecology. 26: 1463–1464. Bioinformatics 2010, 26, 1463–1464. [Google Scholar] [CrossRef]
- De Bello, F.; Lavorel, S.; Albert, C.H.; Thuiller, W.; Grigulis, K.; Dolezal, J.; Janeček, Š.; Lepš, J. Quantifying the relevance of intraspecific trait variability for functional diversity. Methods Ecol. Evol. 2011, 2, 163–174. [Google Scholar] [CrossRef]
- Gordon, A.; Tallas, M.; Van Cleve, K. Soil incubation in polyethylene bags: Effect of bag thickness and temperature on nitrogen transformation and CO2 permeability. Can. J. Soil Sci. 1987, 67, 65–75. [Google Scholar] [CrossRef]
- Dray, S.; Dufour, A.-B. The ade4 package: Implementing the duality diagram for ecologists. J. Stat. Softw. 2007, 22, 1–20. [Google Scholar] [CrossRef]
- Chang, C.; HilleRisLambers, J. Integrating succession and community assembly perspectives. F1000Res. 2016, 5, 1–10. [Google Scholar] [CrossRef]
- Gordon, A.M.; Simpson, J.; Faller, K.; Kaushiik, N.; Williams, P.; Mallory, E. Ten-year changes in the Riparian environment of a rehabilitated agricultural stream: Effects on fish and wildlife habitat. In OASIS Final Report; OASIS: Burlington, MA, USA, 1996; pp. 124–190. [Google Scholar]
- Bober, M.; Donnelly, M. Progress Report on the Effects of Riparian Reforestation in an Agricultural Watershed; Submitted as part of a thesis to the University of Guelph; University of Guelph: Guelph, ON, Canada, 1989; p. 62. [Google Scholar]
- Rey Benayas, J.M.; Martins, A.; Nicolau, J.M.; Schulz, J.J. Abandonment of agricultural land: An overview of drivers and consequences. CAB Rev. Perspect. Agric. Vet. Sci. Nutr. Nat. Resour. 2007, 2, 1–14. [Google Scholar] [CrossRef]
- Cramer, V.A.; Hobbs, R.J.; Standish, R.J. What’s new about old fields? Land abandonment and ecosystem assembly. Trends Ecol. Evol. 2008, 23, 104–112. [Google Scholar] [CrossRef] [PubMed]
- Levine, J.M.; D’Antonio, C.M. Elton revisited: A review of evidence linking diversity and invasibility. Oikos 1999, 87, 15–26. [Google Scholar] [CrossRef]
- Kennedy, T.A.; Naeem, S.; Howe, K.M.; Knops, J.; Tilman, D.; Reich, P. Biodiversity as a barrier to ecological invasion. Nature 2002, 417, 636–638. [Google Scholar] [CrossRef]
- Mason, N.W.H.; Carswell, F.E.; Richardson, S.J.; Lawrence, E. Leaf palatability and decomposability increase during a 200-year-old post-cultural woody succession in New Zealand. J. Veg. Sci. 2011, 22, 6–17. [Google Scholar] [CrossRef]
- Ordoñez, J.C.; Van Bodegom, P.M.; Witte, J.M.; Wright, I.J.; Reich, P.B.; Aerts, R. A global study of relationships between leaf traits, climate and soil measures of nutrient fertility. Glob. Ecol. Biogeogr. 2009, 18, 137–149. [Google Scholar] [CrossRef]
- Poorter, H.; Niklas, K.J.; Reich, P.B.; Oleksyn, J.; Poot, P.; Mommer, L. Biomass allocation to leaves, stems and roots: Meta-analysis of interspecific variation and environmental control. New Phytol. 2012, 193, 30–50. [Google Scholar] [CrossRef]
- Novara, A.; Rühl, J.; La Mantia, T.; Gristina, L.; La Bella, S.; Tuttolomondo, T. Litter contribution to soil organic carbon in the processes of agriculture abandon. Solid Earth 2015, 6, 425–432. [Google Scholar] [CrossRef]
- Tilman, D.; Tilman, D.; Reich, P.B.; Knops, J.; Wedin, D.; Mielke, T.; Lehman, C. Diversity and productivity in a long-term grassland experiment. Science 2001, 294, 843–845. [Google Scholar] [CrossRef]
- Lavorel, S.; Grigulis, K.; Lamarque, P.; Colace, M.P.; Garden, D.; Girel, J.; Pellet, G.; Douzet, R. Using plant functional traits to understand the landscape distribution of multiple ecosystem services. J. Ecol. 2011, 99, 135–147. [Google Scholar] [CrossRef]
- Butterfield, B.J.; Suding, K.N. Single-trait functional indices outperform multi-trait indices in linking environmental gradients and ecosystem services in a complex landscape. J. Ecol. 2013, 101, 9–17. [Google Scholar] [CrossRef]
- Grigulis, K.; Lavorel, S.; Krainer, U.; Legay, N.; Baxendale, C.; Dumont, M.; Kastl, E.; Arnoldi, C.; Bardgett, R.D.; Poly, F.; et al. Relative contributions of plant traits and soil microbial properties to mountain grassland ecosystem services. J. Ecol. 2013, 101, 47–57. [Google Scholar] [CrossRef]
- Conti, G.; Díaz, S. Plant functional diversity and carbon storage—An empirical test in semi-arid forest ecosystems. J. Ecol. 2013, 101, 18–28. [Google Scholar] [CrossRef]
- Guo, C.; Cornelissen, J.H.C. Functional evenness of N-to-P ratios of evergreen-deciduous mixtures predicts positive non-additive effect on leaf litter decomposition. Plant Soil 2019, 436, 299–309. [Google Scholar] [CrossRef]
- Hao, M.H.; Zhang, C.; Zhao, X.; von Gadow, K. Functional and phylogenetic diversity determine woody productivity in a temperate forest. Ecol. Evol. 2018, 8, 2395–2406. [Google Scholar] [CrossRef]
- Hisano, M.; Searle, E.B.; Chen, H.Y.H. Biodiversity as a solution to mitigate climate change impacts on the functioning of forest ecosystems. Biol. Rev. 2018, 93, 439–456. [Google Scholar] [CrossRef]
- Cornwell, W.K.; Cornelissen, J.H.C.; Amatangelo, K.; Dorrepaal, E.; Eviner, V.T.; Godoy, O.; Hobbie, S.E.; Hoorens, B.; Kurokawa, H.; Pérez-Harguindeguy, N.; et al. Plant species traits are the predominant control on litter decomposition rates within biomes worldwide. Ecol. Lett. 2008, 11, 1065–1071. [Google Scholar] [CrossRef]
- Smith, K.A.; Ball, T.; Conen, F.; Dobbie, K.E.; Massheder, J.; Rey, A. Exchange of greenhouse gases between soil and atmosphere: Interactions of soil physical factors and biological processes. Eur. J. Soil Sci. 2018, 69, 10–20. [Google Scholar] [CrossRef]
- Abalos, D.; van Groenigen, J.W.; De Deyn, G.B. What plant functional traits can reduce nitrous oxide emissions from intensively managed grasslands? Glob. Chang. Biol. 2018, 24, e248–e258. [Google Scholar] [CrossRef]
- Mitsch, W.J.; Bernal, B.; Nahlik, A.M.; Jørgensen, S.E.; Brix, H. Wetlands, carbon, and climate change. Landsc. Ecol. 2013, 28, 583–597. [Google Scholar] [CrossRef]
- Lee, K.; Isenhart, T.; Schultz, R. Sediment and nutrient removal in an established multi-species riparian buffer. J. Soil Water Conserv. 2003, 58, 1–8. [Google Scholar]
- Gurr, G.M.; Wratten, S.D.; Luna, J.M. Multi-function agricultural biodiversity: Pest management and other benefits. Basic Appl. Ecol. 2003, 4, 107–116. [Google Scholar] [CrossRef]
- Cole, L.J.; Brocklehurst, S.; Robertson, D.; Harrison, W.; Mccracken, D.I. Riparian buffer strips: Their role in the conservation of insect pollinators in intensive grassland systems. Agric. Ecosyst. Environ. 2015, 211, 207–220. [Google Scholar] [CrossRef]
- Williams, N.M. Restoration of nontarget species: Bee communities and pollination function in riparian forests. Restor. Ecol. 2011, 19, 450–459. [Google Scholar] [CrossRef]
| Diversity Indice | Riparian Buffer Type | F (p-Value) | |||
|---|---|---|---|---|---|
| GRASS | AGROFOREST-REHAB | AGROFOREST-NATURAL | |||
| Taxonomic diversity | Species richness | 7.13 ± 0.52 a | 8.88 ± 1.06 a | 3.63 ± 0.32 b | 14.36 (<0.001) |
| Shannon’s diversity index | 1.53 ± 0.08 a | 1.50 ± 0.07 a | 0.97 ± 0.09 b | 15.17 (<0.001) | |
| Phylogenetic diversity | Faith’s phylogenetic diversity | 537.84 ± 36.3 a | 763.81 ± 75.53 a | 735.14 ± 126.97 a | 1.18 (0.33) |
| Rao quadratic entropy | 78.35 ± 3.62 a | 82.03 ± 3.91 a | 175.39 ± 28.45 b | 10.83 (<0.001) | |
| Diversity Indice | Riparian Buffer Type | F (p-Value) | |||
|---|---|---|---|---|---|
| GRASS | AGROFOREST-REHAB | AGROFOREST-NATURAL | |||
| Community weighted means | SLA | 0.02 ± 0.001 a | 0.05 ± 0.001 b | 0.04 ± 0.001 c | 229.91 (<0.001) |
| L-C:N | 17.93 ± 1.14 a | 11.74 ± 0.27 b | 13.75 ± 0.29 c | 24.74 (<0.001) | |
| LNC | 25.23 ± 1.08 a | 35.85 ± 0.55 b | 29.69 ± 0.68 c | 43.92 (<0.001) | |
| Functional diversity | SLA | 0.005 ± 0.0003 a | 0.02±0.002 b | 0.01 ± 0.004 a | 16.37 (<0.001) |
| L-C:N | 45.74 ± 11.31 a | 6.20 ± 0.56 b | 13.02 ± 2.23 b | 15.85 (<0.001) | |
| LNC | 55.72 ± 6.73 a,b | 74.47 ± 7.42 a | 38.93 ± 3.96 b | 8.18 (0.002) | |
| Ecosystem Process | Riparian Buffer Type | F (p-Value) | ||
|---|---|---|---|---|
| GRASS | AGROFOREST-REHAB | AGROFOREST-NATURAL | ||
| N mineralization (mg kg−1 28 d−1) | 8.5 ± 4.6 a | 6.3 ± 2.7 a | −6.3 ± 2.3 b | 5.66 (0.01) |
| CO2 efflux (mg m−2 h−1) | 423.7 ± 67.7 a | 190.5 ± 30.9 b | 182.0 ± 42.5 b | 6.04 (<0.01) |
| N2O efflux (μg m−2 h−1) | 19.0 ± 3.2 a | 32.9 ± 11.5 a | 24.8 ± 9.9 a | 0.72 (0.49) |
| CH4 efflux (μg m−2 h−1) | −92.9 ± 45.7 a | -0.9 ± 26.4 a | 613.5 ± 164.7 b | 12.65 (<0.01) |
| Multivariate Plant Community Variation | Ecosystem Process | Slope Estimate | p-Value | Cond. r2 |
|---|---|---|---|---|
| Community weighted mean (CWM, 92.5% explained variation) | N Mineralization | −0.43 | 0.70 | 0.01 |
| CO2 efflux | −0.22 | 0.01 | 0.26 | |
| N2O efflux | 0.01 | 0.94 | 0.30 | |
| CH4 efflux | 0.35 | 0.05 | 0.22 | |
| Functional diversity (FD, 53.9% explained variation) | N Mineralization | 0.42 | 0.78 | 0.004 |
| CO2 efflux | −0.22 | 0.05 | 0.19 | |
| N2O efflux | −0.01 | 0.96 | 0.30 | |
| CH4 efflux | 0.17 | 0.48 | 0.15 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Buchanan, S.W.; Baskerville, M.; Oelbermann, M.; Gordon, A.M.; Thevathasan, N.V.; Isaac, M.E. Plant Diversity and Agroecosystem Function in Riparian Agroforests: Providing Ecosystem Services and Land-Use Transition. Sustainability 2020, 12, 568. https://doi.org/10.3390/su12020568
Buchanan SW, Baskerville M, Oelbermann M, Gordon AM, Thevathasan NV, Isaac ME. Plant Diversity and Agroecosystem Function in Riparian Agroforests: Providing Ecosystem Services and Land-Use Transition. Sustainability. 2020; 12(2):568. https://doi.org/10.3390/su12020568
Chicago/Turabian StyleBuchanan, Serra W., Megan Baskerville, Maren Oelbermann, Andrew M. Gordon, Naresh V. Thevathasan, and Marney E. Isaac. 2020. "Plant Diversity and Agroecosystem Function in Riparian Agroforests: Providing Ecosystem Services and Land-Use Transition" Sustainability 12, no. 2: 568. https://doi.org/10.3390/su12020568
APA StyleBuchanan, S. W., Baskerville, M., Oelbermann, M., Gordon, A. M., Thevathasan, N. V., & Isaac, M. E. (2020). Plant Diversity and Agroecosystem Function in Riparian Agroforests: Providing Ecosystem Services and Land-Use Transition. Sustainability, 12(2), 568. https://doi.org/10.3390/su12020568




