Spider Webs and Lichens as Bioindicators of Heavy Metals: A Comparison Study in the Vicinity of a Copper Smelter (Poland)
Abstract
1. Introduction
2. Materials and Methods
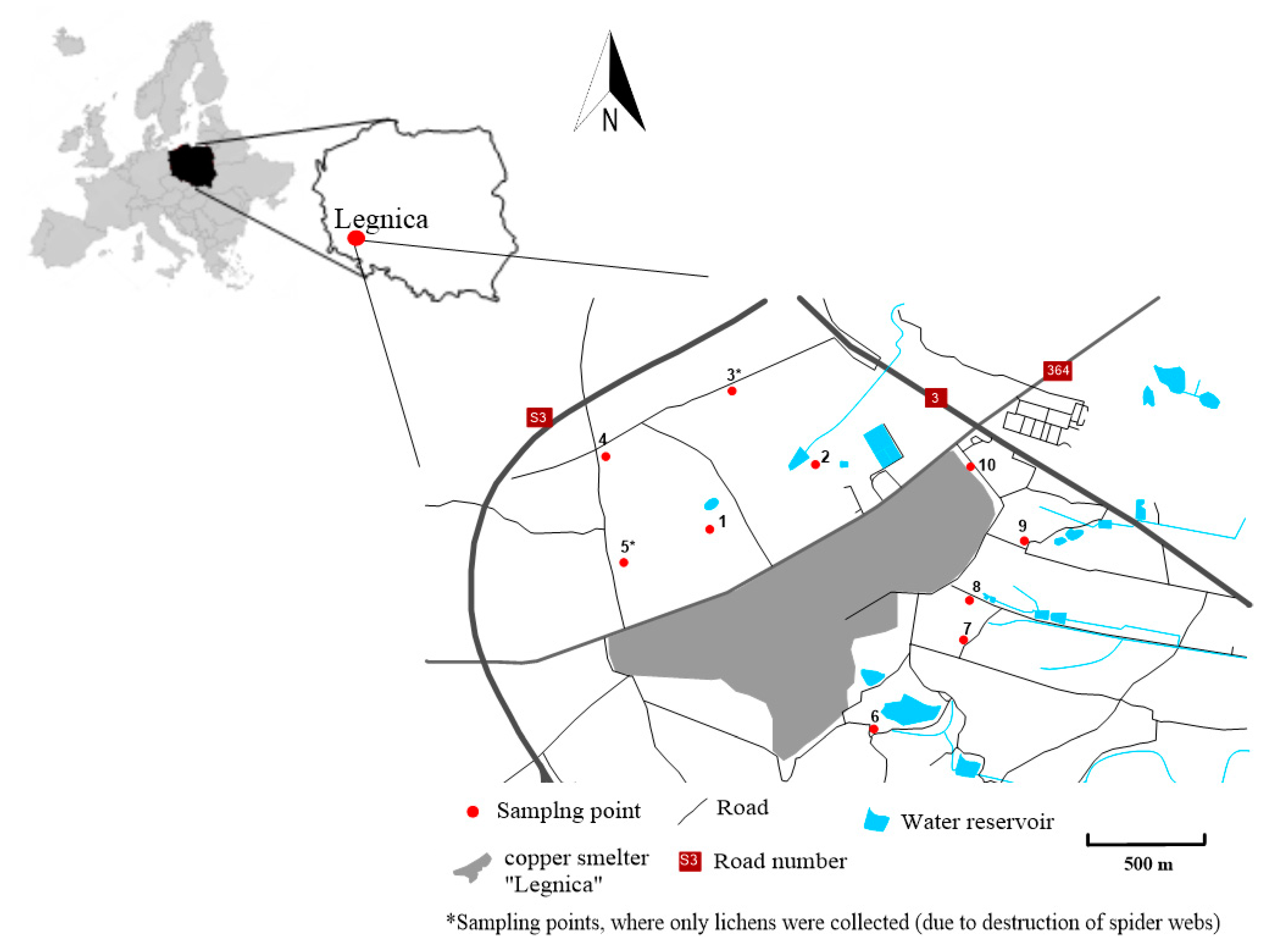
2.1. Study Area
2.2. Materials and Methods
2.3. Contamination Factor
2.4. Statistics
2.5. Meteorological/Environmental Parameters
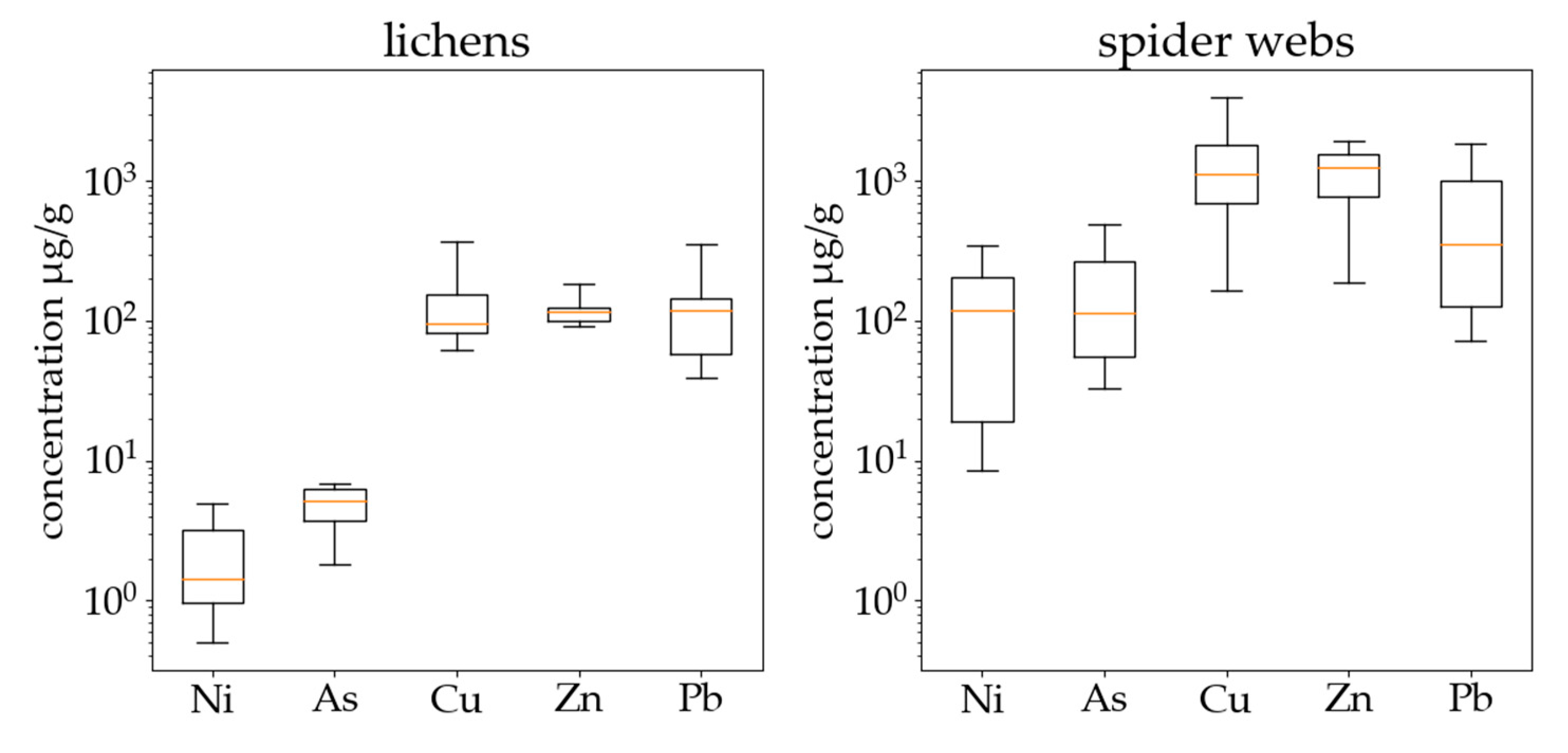
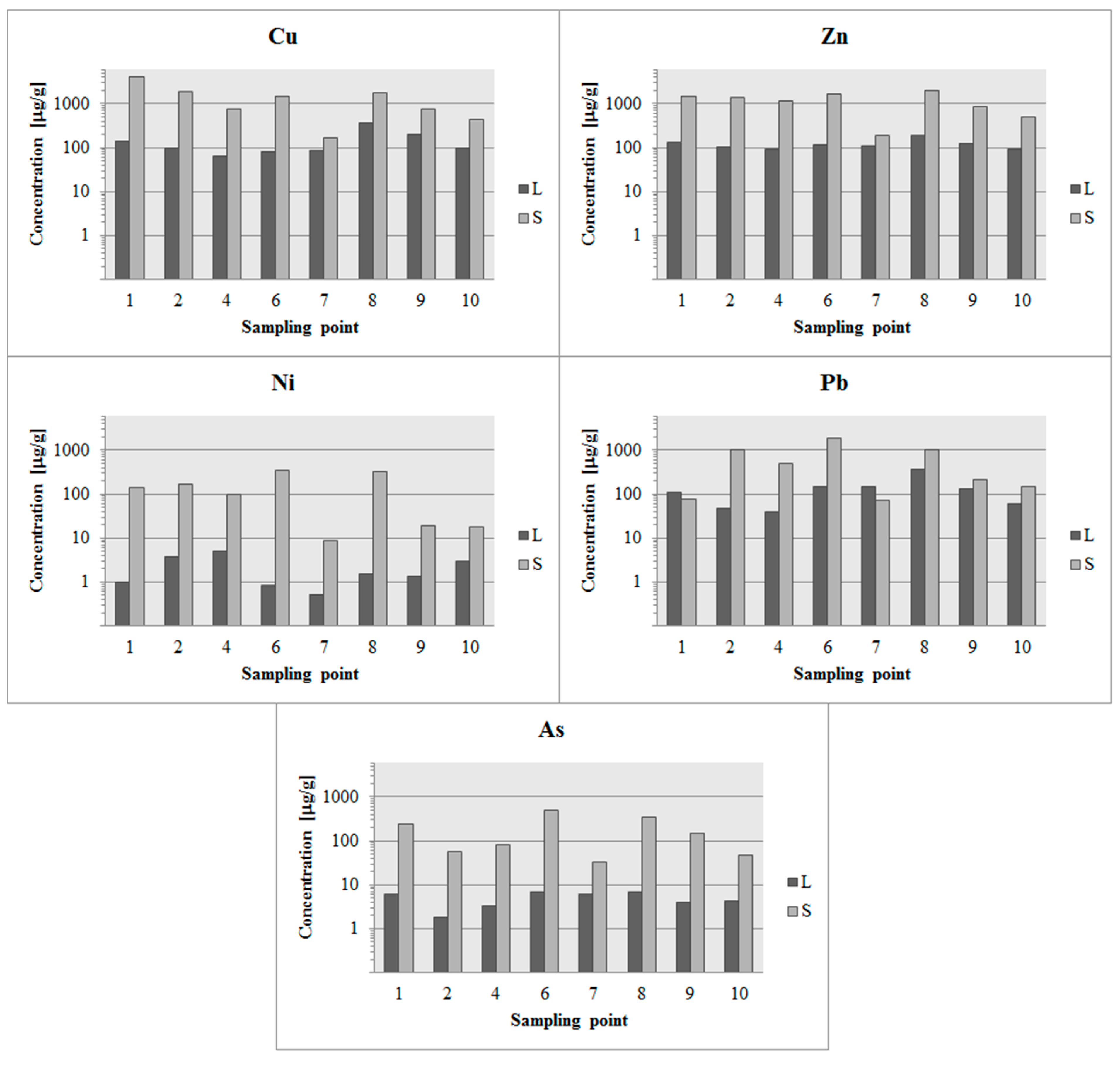
3. Results
3.1. Results of Contamination Factor (CF)
3.2. Results of the Shapiro–Wilk Test
3.3. Results of Tests Comparing Samples
3.4. Results of Cosine Similarity
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Onianwa, P.C. Monitoring atmospheric metal pollution: A review of the use of mosses as indicators. Environ. Monit. Assess. 2001, 71, 13–50. [Google Scholar] [CrossRef]
- Bargagli, R.; Monaci, F.; Borghini, F.; Bravi, F.; Agnorelli, C. Mosses and lichens as biomonitors of trace metals. A comparison study on Hypnum cupressiforme and Parmelia caperata in a former mining district in Italy. Environ. Pollut. 2002, 116, 279–287. [Google Scholar] [CrossRef]
- Adamo, P.; Giordano, S.; Vingiani, S.; Cobianchi, R.C.; Violante, P. Trace element accumulation by moss and lichen exposed in bags in the city of Naples (Italy). Environ. Pollut. 2003, 22, 91–103. [Google Scholar] [CrossRef]
- Giordano, S.; Adamo, P.; Spagnuolo, V.; Tretiach, M.; Bargagli, R. Accumulation of airborne trace elements in mosses, lichens and synthetic materials exposed at urban monitoring stations: Towards a harmonisation of the moss-bag technique. Chemosphere 2013, 90, 292–299. [Google Scholar] [CrossRef] [PubMed]
- Goodman, G.T.; Roberts, T.M. Plants and soils as indicators of metals in the air. Nature 1971, 231, 287–292. [Google Scholar] [CrossRef]
- Temple, P.J.; McLaughlin, D.L.; Linzon, S.N.; Wills, R. Moss bags as monitors of atmospheric deposition. J. Air Pollut. Control Assoc. 1981, 31, 668–670. [Google Scholar] [CrossRef]
- Garty, J.; Karary, Y.; Harel, J. The impact of air pollution on the integrity of cell membranes and chlorophyll in the lichen Ramalina duriaei (de not.) bagl. transplanted to industrial sites in Israel. Arch. Environ. Contam. Toxicol. 1993, 24, 455–460. [Google Scholar] [CrossRef]
- Hose, G.C.; James, J.M.; Gray, M.R. Spider webs as environmental indicators. Environ. Pollut. 2002, 120, 725–733. [Google Scholar] [CrossRef]
- Rutkowski, R.; Rybak, J.; Rogula-Kozłowska, W.; Bełcik, M.; Piekarska, K.; Jureczko, I. Mutagenicity of indoor air pollutants adsorbed on spider webs. Ecotoxicol. Environ. Saf. 2019, 171, 549–557. [Google Scholar] [CrossRef]
- Rybak, J. Accumulation of major and trace elements in spider webs. Water Air Soil Pollut. 2015, 226, 105. [Google Scholar] [CrossRef]
- Rybak, J.; Sówka, I.; Zwozdziak, A.; Fortuna, M.; Trzepla-Nabagło, K. Evaluation of the usefulness of spider webs as an air quality monitoring tool for heavy metals. Ecol. Chem. Eng. S 2015, 22, 389–400. [Google Scholar] [CrossRef]
- Xiao-Li, S.; Yu, P.; Hose, G.C.; Jian, C.; Feng-Xiang, L. Spider webs as indicators of heavy metal pollution in air. Bull. Environ. Contam. Toxicol. 2006, 76, 271–277. [Google Scholar] [CrossRef] [PubMed]
- Strzelec, Ł.; Niedźwiecka, W. Stan środowiska naturalnego wrejonie oddziaływania hut miedzi. Kierunki zmian. Med. Środowiskowa Environ. Med. 2012, 15, 21–31. [Google Scholar]
- Tyszka, R.; Pietranik, A.; Kierczak, J.; Ettler, V.; Mihaljevič, M.; Medyńska-Juraszek, A. Lead isotopes and heavy minerals analyzed as tools to understand the distribution of lead and other potentially toxic elements in soils contaminated by Cu smelting (Legnica, Poland). Environ. Sci. Pollut. Res. 2016, 23, 24350–24363. [Google Scholar] [CrossRef] [PubMed]
- KGHM Cuprum Sp.z.o.o. Research Center. Monograph of KGHM Polish Copper Company; KGHM Cuprum Sp.z.o.o. Research Center: Wrocław, Poland, 2007. (In Polish) [Google Scholar]
- Wojewódzki Inspektorat Ochrony Środowiska. Informacja o Jakości Powietrza na Obszarze miasta Legnica. 2017. Available online: https://www.wroclaw.pios.gov.pl/pliki/powietrze/powietrze_legnica_2016_wios.pdf (accessed on 25 September 2020).
- Jóźwiak, M. Kumulacja metali ciężkich i zmiany morfologiczne porostu Hypogymnia physodes (L.) Nyl. Monit. Środowiska Przyr. 2007, 8, 51–56. [Google Scholar]
- Kłos, A.; Ziembik, Z.; Rajfur, M.; Dołhańczuk-Śródka, A.; Bochenek, Z.; Bjerke, J.W.; Tømmervik, H.; Zagajewski, B.; Ziółkowski, D.; Jerz, D.; et al. Using moss and lichens in biomonitoring of heavy-metal contamination of forest areas in southern and north-eastern Poland. Sci. Total Environ. 2018, 627, 438–449. [Google Scholar] [CrossRef]
- Ciężka, M.; Górka, M.; Modelska, M.; Tyszka, R.; Samecka-Cymerman, A.; Lewińska, A.; Łubek, A.; Widory, D. The coupled study of metal concentrations and electron paramagnetic resonance (EPR) of lichens (Hypogymnia physodes) from the Świętokrzyski National Park—Environmental implications. Environ. Sci. Pollut. Res. 2018, 25, 25348–25362. [Google Scholar] [CrossRef]
- Koroleva, Y.; Revunkov, V. Air pollution monitoring in the south-east Baltic using the epiphytic lichen Hypogymnia physodes. Atmosphere 2017, 8, 119. [Google Scholar] [CrossRef]
- Poličnik, H.; Franc, B.; Cvetka, R.L. Monitoring of short-term heavy metal deposition by accumulation in epiphytic lichens (Hypogymnia physodes (L.) Nyl.). J. Atmos. Chem. 2004, 49, 223–230. [Google Scholar] [CrossRef]
- Balabanova, B.; Stafilov, T.; Šajn, R.; Baèeva, K. Characterisation of heavy metals in lichen species Hypogymnia physodes and Evernia prunastri due to biomonitoring of air pollution in the vicinity of copper mine. Int. J. Environ. Res. 2012, 6, 779–792. [Google Scholar]
- Foelix, R.F. Biology of Spiders; Oxford University Press: New York, NY, USA, 1996. [Google Scholar]
- Rybak, J.; Olejniczak, T. Accumulation of polycyclic aromatic hydrocarbons (PAHs) on the spider webs in the vicinity of road traffic emissions. Environ. Sci. Pollut. Res. 2014, 21, 2313–2324. [Google Scholar] [CrossRef] [PubMed]
- Darnajoux, R.; Lutzoni, F.; Miadlikowska, J.; Bellenger, J.P. Determination of elemental baseline using peltigeralean lichens from Northeastern Canada (Québec): Initial data collection for long term monitoring of the impact of global climate change on boreal and subarctic area in Canada. Sci. Total Environ. 2015, 533, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Fernández, J.A.; Carballeira, A. Evaluation of contamination by different elements in terrestrial mosses. Arch. Environ. Contam. Toxicol. 2001, 40, 461–468. [Google Scholar] [CrossRef]
- Shapiro, S.S.; Wilk, M.B. An analysis of variance test for normality (complete samples). Biometrika 1965, 52, 591–611. [Google Scholar] [CrossRef]
- Razali, N.M.; Wah, Y.B. No Power comparisons of Shapiro-Wilk, Kolmogorov-Smirnov, Lilliefors and Anderson-Darling tests. J. Stat. Model. Anal. 2011, 2, 21–33. [Google Scholar]
- Feng, C.; Wang, H.; Lu, N.; Chen, T.; He, H.; Lu, Y.; Tu, X.M. Log-transformation and its implications for data analysis. Shanghai Arch. Psychiatry 2014, 26, 105–109. [Google Scholar] [CrossRef]
- Welch, B.L. 1.T.g.o.“Student’s” problem when several different population variances are involved. Biometrika 2013, 34, 28–35. [Google Scholar] [CrossRef]
- Wilcoxon, F. Individual comparisons by ranking methods. Biom. Bull. 1945, 1, 80–83. [Google Scholar] [CrossRef]
- Milton, R.C. An extended table of critical values for the Mann-Whitney (Wilcoxon) two-sample statistic. J. Am. Stat. Assoc. 1964, 59, 925–934. [Google Scholar] [CrossRef]
- Jones, W.P.; Furnas, G.W. Pictures of relevance: A geometric analysis of similarity measures. J. Am. Soc. Inf. Sci. 1987, 38, 420–442. [Google Scholar] [CrossRef]
- McKinney, W. Data structures for statistical computing in Python. In Proceedings of the 9th Python in Science Conference, Austin, TX, USA, 28 June–3 July 2010. [Google Scholar]
- Virtanen, P.; Gommers, R.; Oliphant, T.E.; Haberland, M.; Reddy, T.; Cournapeau, D.; Burovski, E.; Peterson, P.; Weckesser, W.; Bright, J.; et al. SciPy 1.0: Fundamental algorithms for scientific computing in Python. Nat. Methods 2020, 17, 261–272. [Google Scholar] [CrossRef] [PubMed]
- Hunter, J.D. Matplotlib: A 2D graphics environment. Comput. Sci. Eng. 2007, 9, 90–95. [Google Scholar] [CrossRef]
- Instytut Meteorologii i Gospodarki Wodnej Państwowy Instytut Badawczy. Rocznik Meteorologiczny. 2019. Available online: https://danepubliczne.imgw.pl/data/dane_pomiarowo_obserwacyjne/Roczniki/Rocznik%20meteorologiczny/Rocznik%20Meteorologiczny%202019.pdf (accessed on 25 September 2020).
- Główny Inspektorat Ochrony Środowiska, Roczna Ocena Jakości Powietrza w Województwie Dolnośląskim. 2019. Available online: https://www.wroclaw.pios.gov.pl/pliki/powietrze/ocena_roczna_2019.pdf (accessed on 25 September 2020).
- Konarski, P.; Ćwil, M.; Iwanejko, I.; Mierzejewska, A.; Diduszko, R. Morphology of micro- and nanoparticles emitted by copper plants in Western Poland. Thin Solid Films 2004, 459, 86–89. [Google Scholar] [CrossRef]
- Sternbeck, J.; Sjödin, Å.; Andréasson, K. Metal emissions from road traffic and the influence of resuspension—Results from two tunnel studies. Atmos. Environ. 2002, 36, 4735–4744. [Google Scholar] [CrossRef]
- Goix, S.; Resongles, E.; Point, D.; Oliva, P.; Duprey, J.L.; de la Galvez, E.; Ugarte, L.; Huayta, C.; Prunier, J.; Zouiten, C.; et al. Transplantation of epiphytic bioaccumulators (Tillandsia capillaris) for high spatial resolution biomonitoring of trace elements and point sources deconvolution in a complex mining/smelting urban context. Atmos. Environ. 2013, 80, 330–341. [Google Scholar] [CrossRef]
- GDDKiA: 2015. General Measurement of Traffic. Available online: https://www.gddkia.gov.pl/userfiles/articles/g/generalny-pomiar-ruchu-w-2015_15598//SYNTEZA/WYNIKI_GPR2015_DW.pdf (accessed on 4 April 2020).
- Białońska, D.; Dayan, F.E. Chemistry of the lichen Hypogymnia physodes transplanted to an industrial region. J. Chem. Ecol. 2005, 31, 2975–2991. [Google Scholar] [CrossRef]
| C1 | CF < 1 | no contamination |
| C2 | 1 < CF < 2 | suspected contamination |
| C3 | 2 < CF < 3.5 | slight contamination |
| C4 | 3.5 < CF < 8 | moderate contamination |
| C5 | 8 < CF < 27 | severe contamination |
| C6 | CF > 27 | extreme contamination |
| Metal | CF | Category |
|---|---|---|
| Pb | 276.3 | C6 |
| Cu | 25.7 | C5 |
| Ni | 2.0 | C3 |
| Zn | 2.2 | C3 |
| As | - | - |
| Lichens | Spider Webs | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| element | Cu | Zn | Ni | Pb | As | Cu | Zn | Ni | Pb | As |
| W-stat. value | 0.75 | 0.82 | 0.87 | 0.79 | 0.91 | 0.85 | 0.96 | 0.87 | 0.84 | 0.86 |
| p | 0.008 | 0.05 | 0.17 | 0.03 | 0.37 | 0.11 | 0.87 | 0.16 | 0.08 | 0.12 |
| Cu | Zn | Ni | Pb | As | |
|---|---|---|---|---|---|
| Test | Wilcoxon | Welch’s t-test | Welch’s t-test | Welch’s t-test | Welch’s t-test |
| statistics | 0.0 | –4.8070 | –2.9284 | –2.1046 | –3.0060 |
| p-value | <0.02 | 0.0019 | 0.0221 | 0.0715 | 0.0198 |
| DOF | Not applicable | >7 | >7 | >7 | >7 |
| Point 1 | Point 2 | Point 4 | Point 6 | Point 7 | Point 8 | Point 9 | Point 10 | |
|---|---|---|---|---|---|---|---|---|
| cos(θ) | 0.81 | 0.98 | 1.00 | 0.98 | 0.85 | 0.89 | 0.90 | 0.97 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stojanowska, A.; Rybak, J.; Bożym, M.; Olszowski, T.; Bihałowicz, J.S. Spider Webs and Lichens as Bioindicators of Heavy Metals: A Comparison Study in the Vicinity of a Copper Smelter (Poland). Sustainability 2020, 12, 8066. https://doi.org/10.3390/su12198066
Stojanowska A, Rybak J, Bożym M, Olszowski T, Bihałowicz JS. Spider Webs and Lichens as Bioindicators of Heavy Metals: A Comparison Study in the Vicinity of a Copper Smelter (Poland). Sustainability. 2020; 12(19):8066. https://doi.org/10.3390/su12198066
Chicago/Turabian StyleStojanowska, Agnieszka, Justyna Rybak, Marta Bożym, Tomasz Olszowski, and Jan Stefan Bihałowicz. 2020. "Spider Webs and Lichens as Bioindicators of Heavy Metals: A Comparison Study in the Vicinity of a Copper Smelter (Poland)" Sustainability 12, no. 19: 8066. https://doi.org/10.3390/su12198066
APA StyleStojanowska, A., Rybak, J., Bożym, M., Olszowski, T., & Bihałowicz, J. S. (2020). Spider Webs and Lichens as Bioindicators of Heavy Metals: A Comparison Study in the Vicinity of a Copper Smelter (Poland). Sustainability, 12(19), 8066. https://doi.org/10.3390/su12198066







