Microorganisms and Enzymes Used in the Biological Pretreatment of the Substrate to Enhance Biogas Production: A Review
Abstract
1. Introduction
- -
- the possible production of useful results that can of great use further
- -
- decreased development of inhibitory substances because the conditions for operating are more moderated,
- -
- the reduced application chemicals,
- -
- a reduced energy input,
- -
- lower costs for waste deposit.
2. Structure of Lignocellulosic Biomass
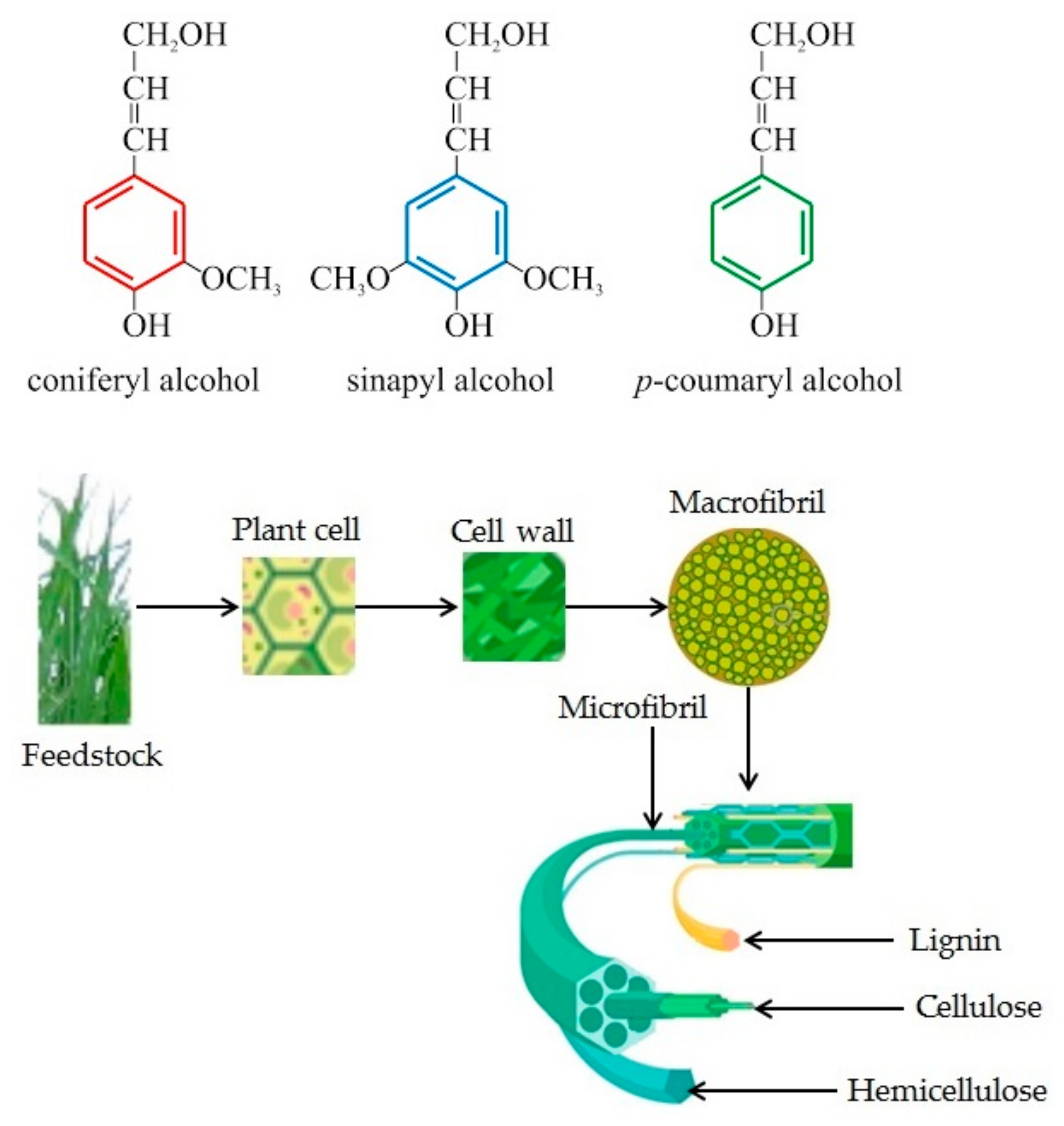
2.1. Lignocellulosic Biomass Composition
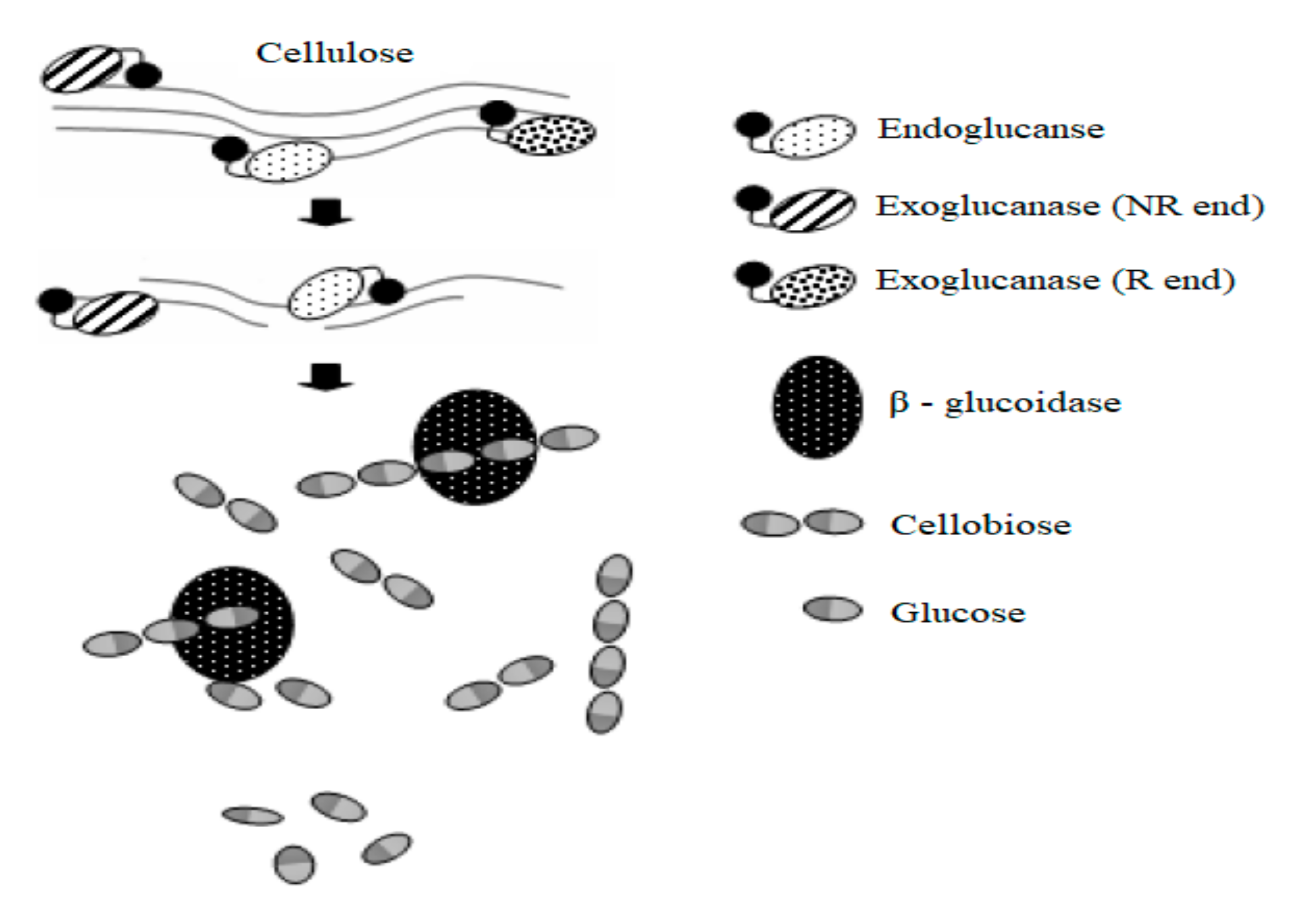
2.2. Cellulose
2.3. Hemicellulose
2.4. Lignin
2.5. Lignocellulose Structure
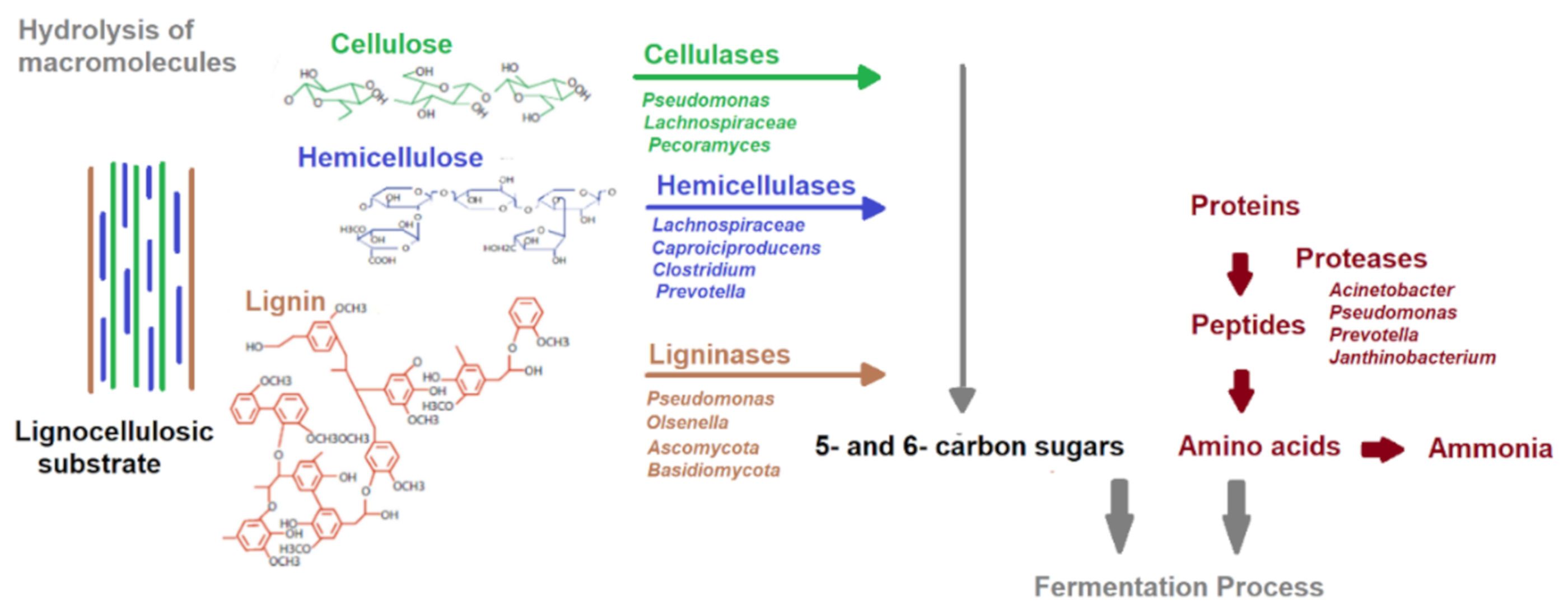
3. Biological Pretreatment
3.1. Bacterial Pretreatment
3.1.1. Microbial Consortia
3.1.2. Fluid Rumen Bacteria
3.1.3. Single Strain Bacterial Cultures
3.1.4. Pretreatment of Microalgae and Water Plants with Bacteria
3.1.5. Selection of Bacteria by Thermophilic Pretreatment
3.2. Fungal Pretreatment
3.3. Enzymatic Pretreatment
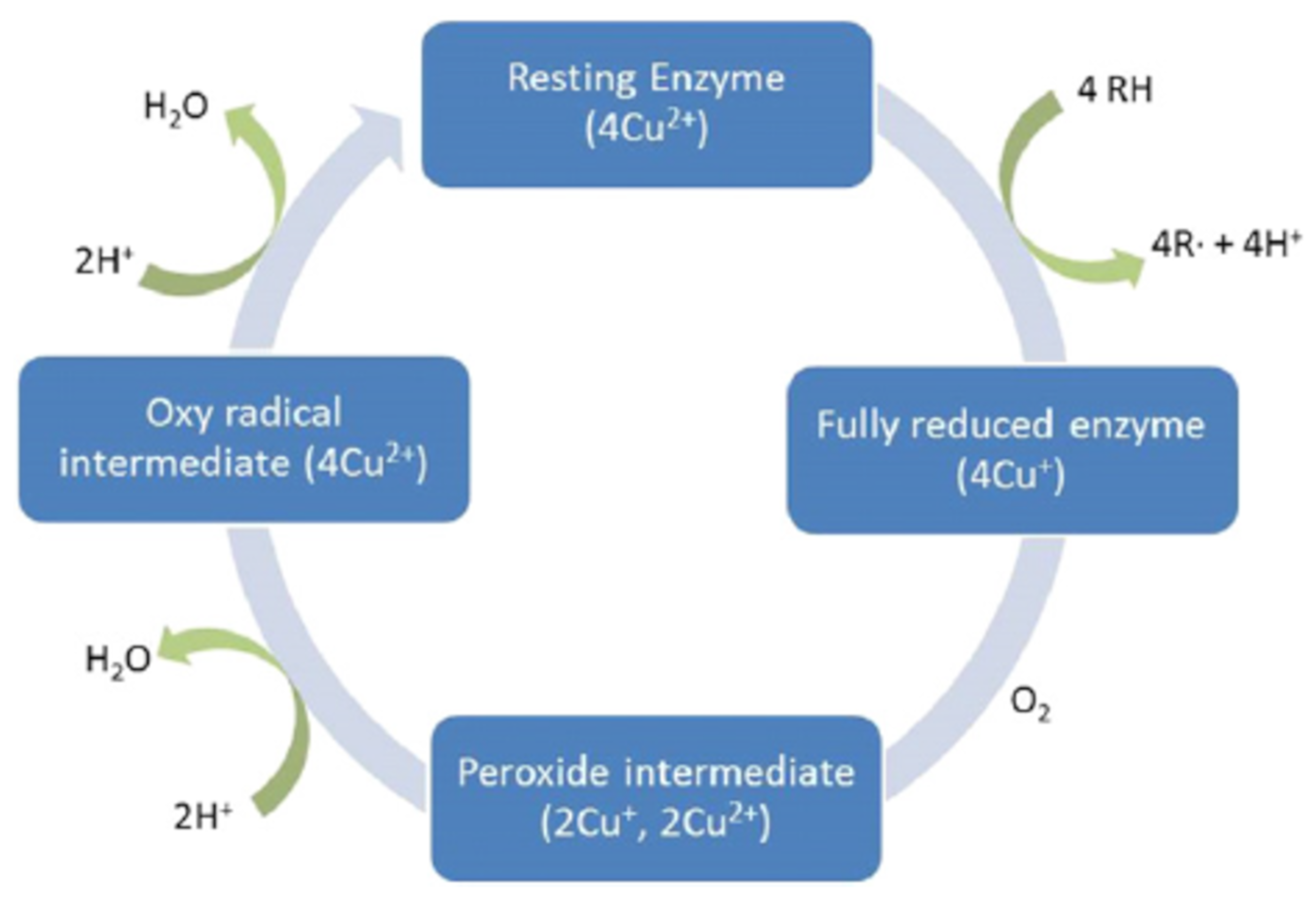
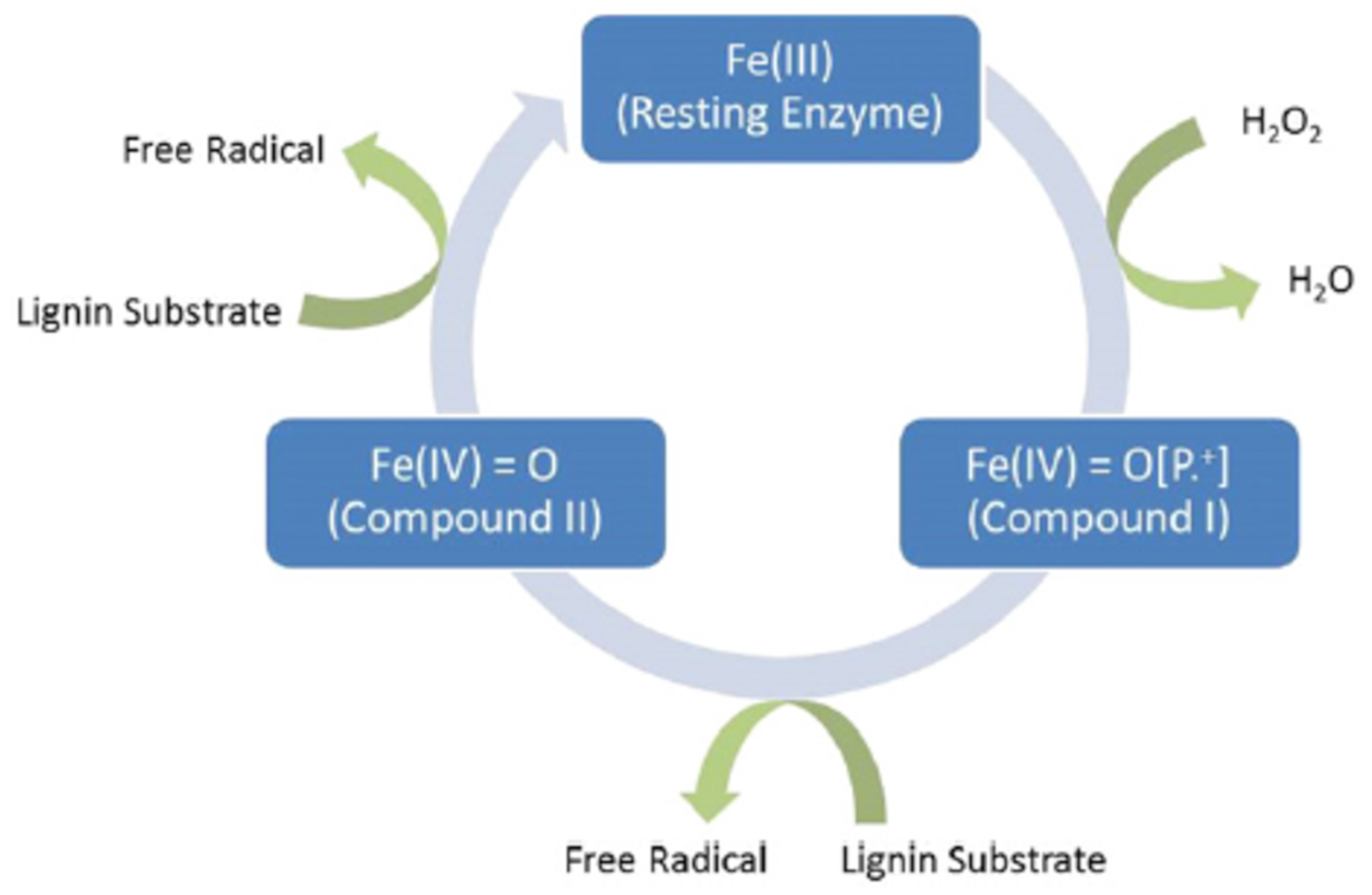
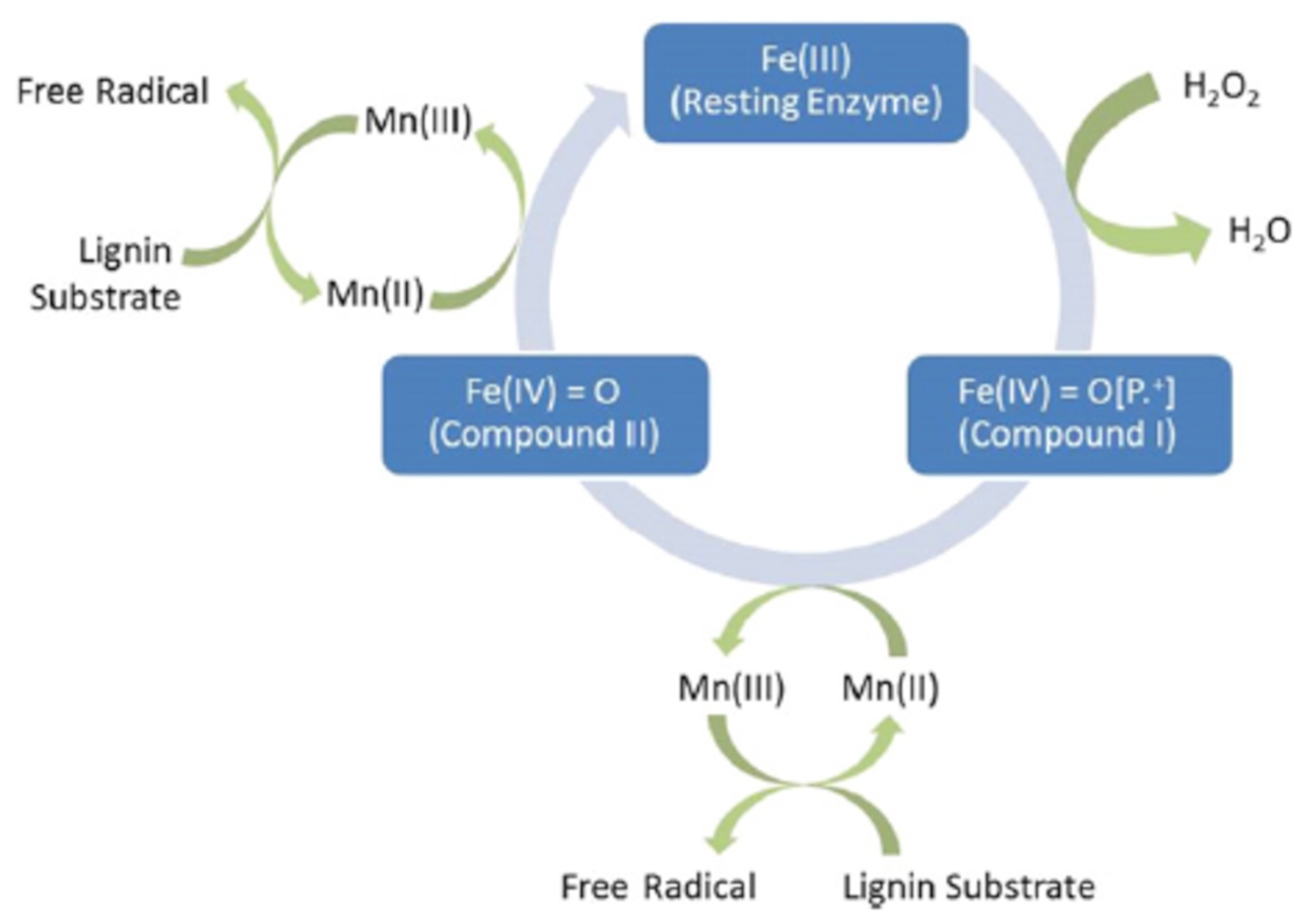
3.3.1. Cellulase and Laccase
3.3.2. Other Enzymes
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Sawatdeenarunat, C.; Surendra, K.C.; Takara, D.; Oechsner, H.; Kumar, S. Anaerobic digestion of lignocellulosic biomass: Challenges and opportunities. Bioresour. Technol. 2015, 178, 178–186. [Google Scholar] [CrossRef] [PubMed]
- Calderon, C.; Colla, M.; Jossart, J.M. Statistical Report 2020–Report biogas. Bioenergy Eur. 2020, 1–17. Available online: https://bioenergyeurope.org/statistical-report.html (accessed on 18 July 2020).
- Baruah, J.; Nath, B.K.; Sharma, R.; Kumar, S.; Deka, R.C.; Baruah, D.C.; Kalita, E. Recent trends in the pretreatment of lignocellulosic biomass for value-added products. Front. Energy Res. 2018, 6, 141. [Google Scholar] [CrossRef]
- Karuppiah, T.; Azariah, V.E. Biomass pretreatment for enhancement of biogas production. In Anaerobic Digestion; Rajesh Banu, J., Ed.; IntechOpen: London, UK, 2019. [Google Scholar] [CrossRef]
- Carrere, H.; Antonopoulou, G.; Affes, R.; Passos, F.; Battimelli, A.; Lyberatos, G.; Ferrer, I. Review of feedstock pretreatment strategies for improved anaerobic digestion: From lab-scale research to full-scale application. Bioresour. Technol. 2016, 199, 386–397. [Google Scholar] [CrossRef] [PubMed]
- Singh, J.; Suhag, M.; Dhaka, A. Augmented digestion of lignocellulose by steam explosion, acid and alkaline pretreatment methods: A review. Carbohydr. Polym. 2015, 117, 624–631. [Google Scholar] [CrossRef]
- Zheng, Y.; Zhao, J.; Xu, F.; Li, Y. Pretreatment of lignocellulosic biomass for enhanced biogas production. Prog. Energy Combust. Sci. 2014, 42, 35–53. [Google Scholar] [CrossRef]
- Otto Wagner, A.; Lackner, N.; Mutschlechner, M.; Prem, E.M.; Markt, R.; Illmer, P. Biological pretreatment strategies for second-generation lignocellulosic resources to enhance biogas production. Energies 2018, 11, 1797. [Google Scholar] [CrossRef]
- Zhang, Q.; Hu, J.; Lee, D.J. Biogas from anaerobic digestion processes: Research updates. Renew. Energy 2016, 98, 108–119. [Google Scholar] [CrossRef]
- Luca, C.; Pilu, R.; Tambone, F.; Scaglia, B.; Adani, F. New energy crop giant cane (Arundo donax L.) can substitute traditional energy crops increasing biogas yield and reducing costs. Bioresour. Technol. 2015, 191, 197–204. [Google Scholar] [CrossRef]
- Frigon, J.C.; Mehta, P.; Guiot, S.R. Impact of mechanical, chemical and enzymatic pretreatments on the methane yield from the anaerobic digestion of switchgrass. Biomass Bioenerg. 2012, 36, 1–11. [Google Scholar] [CrossRef]
- Nanda, S.; Azargohar, R.; Dalai, A.K.; Kozinski, J.A. An assessment on the sustainability of lignocellulosic biomass for biorefining. Renew. Sustain. Energy Rev. 2015, 50, 925–941. [Google Scholar] [CrossRef]
- Hernández-Beltrán, J.U.; Hernández-De Lira, I.O.; Cruz-Santos, M.M.; Saucedo-Luevanos, A.; Hernández-Terán, F.; Balagurusamy, N. Insight into Pretreatment Methods of Lignocellulosic Biomass to Increase Biogas Yield: Current State, Challenges, and Opportunities. Appl. Sci. 2019, 9, 3721. [Google Scholar] [CrossRef]
- Hendriks, A.; Zeeman, G. Pretreatments to enhance the digestibility of lignocellulosic biomass. Bioresour. Technol. 2009, 100, 10–18. [Google Scholar] [CrossRef] [PubMed]
- Moglia, E.S. Enzymatic Pre-treatment of Cellulose Rich Biomasses for Use in the Biogas Process; Swedish University of Agricultural Sciences: Uppsala, Sweden, 2008. [Google Scholar]
- Hosseini, K.E.; Dahadha, S.; Bazyar, L.A.A.; Azizi, A.; Elbeshbishy, E. Enzymatic pretreatment of lignocellulosic biomass for enhanced biomethane production-A review. J. Environ. Manag. 2019, 233, 774–784. [Google Scholar] [CrossRef] [PubMed]
- Saini, J.K.; Saini, R.; Tewari, L. Lignocellulosic agriculture wastes as biomass feedstocks for second-generation bioethanol production: Concepts and recent developments. 3 Biotech 2015, 5, 337–353. [Google Scholar] [CrossRef]
- Gao, J.; Chen, L.; Yuan, K.; Huang, H.; Yan, Z. Ionic liquid pretreatment to enhance the anaerobic digestion of lignocellulosic biomass. Bioresour. Technol. 2013, 150, 352–358. [Google Scholar] [CrossRef]
- Brandt, A.; Gräsvik, J.; Halletta, J.P.; Welton, T. Deconstruction of lignocellulosic biomass with ionic liquids. Green Chem. 2013, 15, 550–583. [Google Scholar] [CrossRef]
- Kulkarni, M.B.; Ghanegaonkar, P.M. Pretreatment methods in anaerobic digestion for biogas generation: A review. Int. J. New Innov. Eng. Technol. 2015, 4, 14–18. [Google Scholar]
- Laureano-Perez, L.; Teymouri, F.; Alizadeh, H.; Dale, B.E. Understanding factors that limit enzymatic hydrolysis of biomass: Characterization of pretreated corn stover. Appl. Biochem. Biotechnol. 2005, 24, 1081–1099. [Google Scholar] [CrossRef]
- Chandra, R.; Takeuchi, H.; Hasegawa, T. Methane production from lignocellulosic agricultural crop wastes: A review in context to second generation of biofuel production. Renew. Sustain. Energy Rev. 2012, 16, 1462–1476. [Google Scholar] [CrossRef]
- Muranaka, Y.; Nakagawa, H.; Hasegawa, I.; Maki, T.; Hosokawa, J.; Ikuta, J.; Mae, K. Lignin-based resin production from lignocellulosic biomass combining acidic saccharification and acetone-water treatment. Chem. Eng. J. 2017, 308, 754–759. [Google Scholar] [CrossRef]
- Yu, J.; Paterson, N.; Blamey, J.; Millan, M. Cellulose, xylan and lignin interactions during pyrolysis of lignocellulosic biomass. Fuel 2017, 191, 140–149. [Google Scholar] [CrossRef]
- Khan, A.S.; Man, Z.; Bustam, M.A.; Nasrullah, A.; Ullah, Z.; Sarwono, A.; Shah, F.U.; Muhammad, N. Efficient conversion of lignocellulosic biomass to levulinic acid using acidic ionic liquids. Carbohydr. Polym. 2017, 181, 208–214. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Chen, Y.; Yang, H.; Xia, M.; Li, K.; Chen, X.; Chen, H. Co-pyrolysis of lignocellulosic biomass and microalgae: Products characteristics and interaction effect. Bioresour. Technol. 2017, 245A, 860–868. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.-F.; Chiueh, P.-T.; Lo, S.-L. A review on microwave pyrolysis of lignocellulosic biomass. Sustain. Environ. Res. 2016, 26, 103–109. [Google Scholar] [CrossRef]
- Raud, M.; Tutt, M.; Olt, J.; Kikas, T. Dependence of the hydrolysis efficiency on the lignin content in lignocellulosic material. Int. J. Hydrogen Energy 2016, 41, 16338–16343. [Google Scholar] [CrossRef]
- De Caprariis, B.; De Filippis, P.; Petrullo, A.; Scarsella, M. Hydrothermal liquefaction of biomass: Influence of temperature and biomass composition on the bio-oil production. Fuel 2017, 208, 618–625. [Google Scholar] [CrossRef]
- Monlau, F.; Barakat, A.; Trably, E.; Dumas, C.; Steyer, J.P.; Carrère, H. Lignocellulosic materials into biohydrogen and biomethane: Impact of structural features and pretreatment. Crit. Rev. Environ. Sci. Technol. 2013, 43, 260–322. [Google Scholar] [CrossRef]
- Monlau, F.; Barakat, A.; Steyer, J.P.; Carrere, H. Comparison of seven types of thermo-chemical pretreatments on the structural features and anaerobic digestion of sunflower stalks. Bioresour. Technol. 2012, 120, 241–247. [Google Scholar] [CrossRef]
- Subhedar, P.B.; Ray, P.; Gogate, P.R. Intensification of delignification and subsequent hydrolysis for the fermentable sugar production from lignocellulosic biomass using ultrasonic irradiation. Ultrason. Sonochem. 2017, 40, 140–150. [Google Scholar] [CrossRef]
- Álvarez, A.; Cachero, S.; González-Sánchez, C.; Montejo-Bernardo, J.; Pizarro, C.; Bueno, J.L. Novel method for holocellulose analysis of non-woody biomass wastes. Carbohydr. Polym. 2018, 189, 250–256. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Ahn, Y.; Kwak, S.-Y. Comparing the influence of acetate and chloride anions on the structure of ionic liquid pretreated lignocellulosic biomass. Biomass Bioenergy 2016, 93, 243–253. [Google Scholar] [CrossRef]
- Karthikeyan, O.P.; Visvanathan, C. Bio-energy recovery from high-solid organic substrates by dry anaerobic bio-conversion processes: A review. Rev. Environ. Sci. Bio. Technol. 2012, 12, 257–284. [Google Scholar] [CrossRef]
- Daza Serna, L.V.; Orrego Alzate, C.E.; Alzate, C.A.C. Supercritical fluids as a green technology for the pretreatment of lignocellulosic biomass. Bioresour. Technol. 2016, 199, 113–120. [Google Scholar] [CrossRef]
- Liu, X.; Hiligsmann, S.; Gourdon, R.; Bayard, R. Anaerobic digestion of lignocellulosic biomasses pretreated with Ceriporiopsis subvermispora. J. Environ. Manag. 2017, 193, 154–162. [Google Scholar] [CrossRef]
- Ye, J.; Li, D.; Sun, Y.; Wang, G.; Yuan, Z.; Zhen, F.; Wang, Y. Improved biogas production from rice straw by co-digestion with kitchen waste and pig manure. Waste Manag. 2013, 33, 2653–2658. [Google Scholar] [CrossRef]
- Brown, D.; Shi, J.; Li, Y. Comparison of solid-state to liquid anaerobic digestion of lignocellulosic feedstocks for biogas production. Bioresour. Technol. 2012, 124, 379–386. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, R.; He, Y.; Zhang, C.; Liu, X.; Chen, C.; Liu, G. Anaerobic co-digestion of chicken manure and corn stover in batch and continuously stirred tank reactor (CSTR). Bioresour. Technol. 2014, 156, 342–347. [Google Scholar] [CrossRef]
- Romano, R.T.; Zhang, R.; Teter, S.; McGarvey, J.A. The effect of enzyme addition on anaerobic digestion of Jose Tall Wheat Grass. Bioresour. Technol. 2009, 100, 4564–4571. [Google Scholar] [CrossRef]
- Appels, L.; Baeyens, J.; Degreve, J.; Dewil, R. Principles and potential of the anaerobic digestion of waste-activated sludge. Prog. Energy Combust. Sci. 2008, 34, 755–781. [Google Scholar] [CrossRef]
- Schroyen, M.; Vervaeren, H.; Vandepitte, H.; Van Hulle, S.W.; Raes, K. Effect of enzymatic pretreatment of various lignocellulosic substrates on production of phenolic compounds and biomethane potential. Bioresour. Technol. 2015, 192, 696–702. [Google Scholar] [CrossRef] [PubMed]
- Teghammar, A.; Yngvesson, J.; Lundin, M.; Taherzadeh, M.J.; Horváth, I.S. Pretreatment of paper tube residuals for improved biogas production. Bioresour. Technol. 2010, 101, 1206–1212. [Google Scholar] [CrossRef] [PubMed]
- Magalhães, A.I., Jr.; de Carvalho, J.C.; de Melo Pereira, G.V.; Karp, S.G.; Câmara, M.C.; Medina, J.D.C.; Soccol, C. R-Lignocellulosic biomass from agro-industrial residues in South America: Current developments and perspectives. Biofuels Bioprod. Biorefin. 2019, 13, 1505–1519. [Google Scholar] [CrossRef]
- Nunes, C.S.; Kunamneni, A. Laccases—properties and applications. In Enzymes in Human and Animal Nutrition; Academic Press, Elsevier: Amsterdam, The Netherlands, 2018; pp. 133–161. [Google Scholar] [CrossRef]
- Michalska, K.; Bizukojć, M.; Ledakowicz, S. Pretreatment of energy crops with sodium hydroxide and cellulolytic enzymes to increase biogas production. Biomass Bioenergy 2015, 80, 213–221. [Google Scholar] [CrossRef]
- Elbeshbishy, E.; Dhar, B.R.; Nakhla, G.; Lee, H.S. A critical review on inhibition of dark biohydrogen fermentation. Renew. Sustain. Energy Rev. 2017, 79, 656–668. [Google Scholar] [CrossRef]
- Parawira, W. Enzyme research and applications in biotechnological intensification of biogas production. Crit. Rev. Biotechnol. 2012, 32, 172–186. [Google Scholar] [CrossRef]
- Bhardwaj, N.; Kumar, B.; Agarwal, K.; Chaturvedi, V.; Verma, P. Purification and characterization of a thermo-acid/alkali stable xylanases from Aspergillus oryzae LC1 and its application in Xylo-oligosaccharides production from lignocellulosic agricultural wastes. Int. J. Biol. Macrom. 2018, 122, 1191–1202. [Google Scholar] [CrossRef]
- Kumar, B.; Bhardwaj, N.; Agrawal, K.; Chaturvedi, V.; Verma, P. Current perspective on pretreatment technologies using lignocellulosic biomass: An emerging biorefinery concept. Fuel Process. Technol. 2020, 199, 106244. [Google Scholar] [CrossRef]
- Sindhu, R.; Binod, P.; Pandey, A. Biological pretreatment of lignocellulosic biomass–An overview. Bioresour. Technol. 2016, 199, 76–82. [Google Scholar] [CrossRef]
- Zhang, Q.; He, J.; Tian, M.; Mao, Z.; Tang, L.; Zhang, J.; Zhang, H. Enhancement of methane production from cassava residues by biological pretreatment using a constructed microbial consortium. Bioresour. Technol. 2011, 102, 8899–8906. [Google Scholar] [CrossRef]
- Singh, P.; Suman, A.; Tiwari, P.; Arya, N.; Gaur, A.; Shrivastava, A.K. Biological pretreatment of sugarcane trash for its conversion to fermentable sugars. World J. Microbiol. Biotechnol. 2008, 24, 667–673. [Google Scholar] [CrossRef]
- Lopez, M.J.; Vargas-Garcia, M.D.; Suarez-Estrella, F.; Nichols, N.N.; Dien, B.S.; Moreno, J.J. Lignocellulose-degrading enzymes produced by the ascomycete Coniochaeta ligniaria and related species: Application for a lignocellulosic substrate treatment. Enzyme Microb. Technol. 2007, 40, 794–800. [Google Scholar] [CrossRef]
- Galbe, M.; Zacchi, G. Pretreatment of lignocellulosic materials for efficient bioethanol production. Biofuels 2007, 108, 41–65. [Google Scholar] [CrossRef]
- Sankaran, R.; Parra Cruz, R.A.; Pakalapati, H.; Show, P.L.; Ling, T.C.; Chen, W.; Tao, Y. Recent advances in the pretreatment of microalgal and lignocellulosic biomass: A comprehensive review. Bioresour. Technol. 2020, 298, 122476. [Google Scholar] [CrossRef]
- Ali, S.S.; Mustafa, A.M.; Kornaros, M.; Manni, A.; Sun, J.; Khalil, M.A. Construction of novel microbial consortia CS-5 and BC-4 valued for the degradation of catalpa sawdust and chlorophenols simultaneously with enhancing methane production. Bioresour. Technol. 2020. [Google Scholar] [CrossRef]
- Kong, X.; Du, J.; Ye, X.; Xi, Y.; Jin, H.; Zhang, M.; Guo, D. Enhanced methane production from wheat straw with the assistance of lignocellulolytic microbial consortium TC-5. Bioresour. Technol. 2018, 263, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Ali, S.S.; Abomohra, A.E.; Sun, J. Effective bio-pretreatment of sawdust waste with a novel microbial consortium for enhanced biomethanation. Bioresour. Technol. 2017, 238, 425–432. [Google Scholar] [CrossRef] [PubMed]
- Ali, S.S.; Al-Tohamy, R.; Manni, A.; Luz, F.C.; Elsamahy, T.; Sun, J. Enhanced digestion of bio-pretreated sawdust using a novel bacterial consortium: Microbial community structure and methane-producing pathways. Fuel 2019, 254, 115604. [Google Scholar] [CrossRef]
- Kato, S.; Haruta, S.; Cui, Z.J.; Ishii, M.; Igarashi, Y. Stable coexistence of five bacterial strains as a cellulose-degrading community. Appl. Environ. Microbiol. 2005, 71, 7099–7106. [Google Scholar] [CrossRef]
- Kato, S.; Haruta, S.; Cui, Z.J.; Ishii, M.; Igarashi, Y. Effective cellulose degradation by a mixed-culture system composed of a cellulolytic Clostridium and aerobic non-cellulolytic bacteria. FEMS Microbiol. Ecol. 2004, 51, 133–142. [Google Scholar] [CrossRef]
- Yuan, X.; Ma, L.; Wen, B.; Zhou, D.; Kuang, M.; Yang, W.; Cui, Z. Enhancing anaerobic digestion of cotton stalk by pretreatment with a microbial consortium (MC1). Bioresour. Technol. 2016, 207, 293–301. [Google Scholar] [CrossRef] [PubMed]
- Hua, B.; Dai, J.; Liu, B.; Zhang, H.; Yuan, X.; Wang, X.; Cui, Z. Pretreatment of non-sterile, rotted silage maize straw by the microbial community MC1 increases biogas production. Bioresour. Technol. 2016, 216, 699–705. [Google Scholar] [CrossRef] [PubMed]
- Zhong, W.; Zhang, Z.; Luo, Y.; Sun, S.; Qiao, W.; Xiao, M. Effect of biological pretreatments in enhancing corn straw biogas production. Bioresour. Technol. 2011, 102, 11174–11182. [Google Scholar] [CrossRef] [PubMed]
- Suksong, W.; Kongjan, P.; Prasertsan, P.; O-Thong, S. Thermotolerant cellulolytic Clostridiaceae and Lachnospiraceae rich consortium enhanced biogas production from oil palm empty fruit bunches by solid-state anaerobic digestion. Bioresour. Technol. 2019, 291, 121851. [Google Scholar] [CrossRef]
- Tantayotai, P.; Pornwongthong, P.; Muenmuang, C.; Phusantisampan, T.; Sriariyanun, M. Effect of Cellulase-producing Microbial Consortium on Biogas Production from Lignocellulosic Biomass. Energ. Proc. 2017, 141, 180–183. [Google Scholar] [CrossRef]
- Vervaeren, H.; Hostyn, K.; Ghekiere, G.; Willems, B. Biological ensilage additives as pretreatment for maize to increase the biogas production. Renew. Energ. 2010, 35, 2089–2093. [Google Scholar] [CrossRef]
- Zhao, Y.; Xu, C.; Ai, S.; Wang, H.; Gao, Y.; Yan, L.; Mei, Z.; Wang, W. Biological pretreatment enhances the activity of functional microorganisms and the ability of methanogenesis during anaerobic digestion. Bioresour. Technol. 2019, 290, 121660. [Google Scholar] [CrossRef]
- Xing, B.; Han, Y.; Wang, X.C.; Wen, J.; Cao, S.; Zhang, K.; Li, Q.; Yuan, H. Persistent action of cow rumen microorganisms in enhancing biodegradation of wheat straw by rumen fermentation. Sci. Total Environ. 2020, 715, 136529. [Google Scholar] [CrossRef]
- Li, F.; Zhang, P.; Zhang, G.; Tang, X.; Wang, S.; Jin, S. Enhancement of corn stover hydrolysis with rumen fluid pretreatment at different solid contents: Effect, structural changes and enzymes participation. Int. Biodeterior. Biodegrad. 2017, 119, 405–412. [Google Scholar] [CrossRef]
- Takizawa, S.; Baba, Y.; Tada, C.; Fukuda, Y.; Nakai, Y. Pretreatment with rumen fluid improves methane production in the anaerobic digestion of paper sludge. Waste Manag. 2018, 78, 379–384. [Google Scholar] [CrossRef]
- Jin, W.; Xu, X.; Yang, F.; Li, C.; Zhou, M. Performance enhancement by rumen cultures in anaerobic co-digestion of corn straw with pig manure. Biomass Bioenergy 2018, 115, 120–129. [Google Scholar] [CrossRef]
- Baba, Y.; Matsuki, Y.; Mori, Y.; Suyama, Y.; Tada, C.; Fukuda, Y.; Saito, M.; Nakai, Y. Pretreatment of lignocellulosic biomass by cattle rumen fluid for methane production: Bacterial flora and enzyme activity analysis. J. Biosci. Bioeng. 2017, 123, 489–496. [Google Scholar] [CrossRef] [PubMed]
- Lee, G.L.; Baba, Y.; Asano, R.; Fukuda, Y.; Tada, C.; Nakai, Y. Identification of bacteria involved in the decomposition of lignocellulosic biomass treated with cow rumen fluid by metagenomic analysis. J. Biosci. Bioeng. 2020, 130, 137–141. [Google Scholar] [CrossRef] [PubMed]
- Nagler, M.; Kozjek, K.; Etemadi, M.; Insam, H.; Podmirseg, S.M. Simple yet effective: Microbial and biotechnological benefits of rumen liquid addition to lignocellulose-degrading biogas plants. J. Biotechnol. 2019, 300, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Takizawa, S.; Baba, Y.; Tada, C.; Fukuda, Y.; Nakai, Y. Preservation of rumen fluid for the pretreatment of waste paper to improve methane production. Waste Manag. 2019, 87, 672–678. [Google Scholar] [CrossRef]
- Forgacs, G.; Alinezhad, S.; Mirabdollah, A.; Feuk-Lagerstedt, E.; Horvath, I.S. Biological treatment of chicken feather waste for improved biogas production. J. Environ. Sci. 2011, 23, 1747–1753. [Google Scholar] [CrossRef]
- Yadav, M.; Paritosh, K.; Pareek, N.; Vivekanand, V. Coupled treatment of lignocellulosic agricultural residues for augmented biomethanation. J. Clean. Prod. 2019, 213, 75–88. [Google Scholar] [CrossRef]
- Kavitha, S.; Subbulakshmi, P.; Rajesh Banu, J.; Gobi, M.; Yeom, I.T. Enhancement of biogas production from microalgal biomass through cellulolytic bacterial pretreatment. Bioresour. Technol. 2017, 233, 34–43. [Google Scholar] [CrossRef]
- Bernaerts, T.M.M.; Gheysen, L.; Foubert, I.; Hendrickx, M.E.; Van Loey, A.M. The potential of microalgae and their biopolymers as structuring ingredients in food: A review. Biotechnol. Adv. 2019, 107419. [Google Scholar] [CrossRef]
- Chen, C.-Y.; Bai, M.-D.; Chang, J.-S. Improving microalgal oil collecting efficiency by pretreating the microalgal cell wall with destructive bacteria. Biochem. Eng. J. 2013, 81, 170–176. [Google Scholar] [CrossRef]
- He, S.; Fan, X.; Katukuri, N.R.; Yuan, X.; Wang, F.; Guo, R. Enhanced methane production from microalgal biomass by anaerobic bio-pretreatment. Bioresour. Technol. 2016, 204, 145–151. [Google Scholar] [CrossRef] [PubMed]
- Barua, V.B.; Goud, V.V.; Kalamdhad, A.S. Microbial pretreatment of water hyacinth for enhanced hydrolysis followed by biogas production. Renew. Energy 2018, 126, 21–29. [Google Scholar] [CrossRef]
- Barua, V.B.; Kalamdhad, A.S. Anaerobic biodegradability test of water hyacinth after microbial pretreatment to optimise the ideal F/M ratio. Fuel 2018, 217, 91–97. [Google Scholar] [CrossRef]
- Tapadia-Maheshwari, S.; Pore, S.; Engineer, A.; Shetty, D.; Dagar, S.S.; Dhakephalkar, P.K. Illustration of the microbial community selected by optimized process and nutritional parameters resulting in enhanced biomethanation of rice straw without thermo-chemical pretreatment. Bioresour. Technol. 2019, 289, 121639. [Google Scholar] [CrossRef]
- Fu, S.F.; He, S.; Shi, X.S.; Katukuri, N.R.; Dai, M.; Guo, R.B. The chemical properties and microbial community characterization of the thermophilic microaerobic pretreatment process. Bioresour. Technol. 2015, 198, 497–502. [Google Scholar] [CrossRef] [PubMed]
- Merlin Christy, P.; Gopinath, L.R.; Divya, D. A review on anaerobic decomposition and enhancement of biogas production through enzymes and microorganisms. Renew. Sust. Energ. Rev. 2014, 34, 167–173. [Google Scholar] [CrossRef]
- Najiah, N.; Nur Liyana, I.; Azlan Shah, H. Fungal pretreatment of lignocellulosic materials. In Biomass for Bioenergy-Recent Trends and Future Challenges; Abd El-Fatah, A., Ed.; IntechOpen: London, UK, 2019. [Google Scholar] [CrossRef]
- Dashtban, M.; Schraft, H.; Syed, T.A.; Qin, W. Fungal biodegradation and enzymatic modification of lignin. Int. J. Biochem. Mol. Biol. 2010, 1, 36–50. [Google Scholar]
- Mishra, S.; Singh, P.K.; Dash, S.; Pattnaik, R. Microbial pretreatment of lignocellulosic biomass for enhanced biomethanation and waste management. 3 Biotec. 2018, 8, 458. [Google Scholar] [CrossRef]
- Sanchez, C. Lignocellulosic residues: Biodegradation and bioconversion by fungi. Biotechnol. Adv. 2009, 27, 185–194. [Google Scholar] [CrossRef]
- Tian, S.Q.; Zhao, R.Y.; Chen, Z.C. Review of the pretreatment and bioconversion of lignocellulosic biomass from wheat straw materials. Renew. Sust. Energ. Rev. 2018, 91, 483–489. [Google Scholar] [CrossRef]
- Rouches, E.; Herpoel-Gimbert, I.; Steyer, J.P.; Carrere, H. Improvement of anaerobic degradation by white-rot fungi pretreatment of lignocellulosic biomass: A review. Renew. Sust. Energ. Rev. 2016, 59, 179–198. [Google Scholar] [CrossRef]
- Ravindran, R.; Jaiswal, A.K. A comprehensive review on pre-treatment strategy for lignocellulosic food industry waste: Challenges and opportunities. Bioresour Technol. 2016, 199, 92–102. [Google Scholar] [CrossRef] [PubMed]
- Tabatabaei, M.; Aghbashlo, M.; Valijanian, E.; Panahi, H.K.S.; Nizami, A.S.; Ghanavati, H.; Sulaiman, A.; Mirmohamadsadeghi, S.; Karimi, K. A comprehensive review on recent biological innovations to improve biogas production, Part 1: Upstream strategies. Renew. Energ. 2020, 146, 1204–1220. [Google Scholar] [CrossRef]
- Janusz, G.; Pawlik, A.; Sulej, J.; Swiderska-Burek, U.; Jarosz-Wilkolazka, A.; Paszczynski, A. Lignin degradation: Microorganisms, enzymes involved, genomes analysis and evolution. FEMS Microbiol. Rev. 2017, 41, 941–962. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Zheng, Y.; Li, Y. Fungal pretreatment of yard trimmings for enhancement of methane yield from solid-state anaerobic digestion. Bioresour. Technol. 2014, 156, 176–181. [Google Scholar] [CrossRef]
- Mustafa, A.M.; Poulsen, T.G.; Sheng, K. Fungal pretreatment of rice straw with Pleurotus ostreatus and Trichoderma reesei to enhance methane production under solid-state anaerobic digestion. Appl. Energy 2016, 180, 661–671. [Google Scholar] [CrossRef]
- Da Silva Machado, A.; Ferraz, A. Biological pretreatment of sugarcane bagasse with basidiomycetes producing varied patterns of biodegradation. Bioresour. Technol. 2017, 225, 17–22. [Google Scholar] [CrossRef]
- Nkemka, V.N.; Gilroyed, B.; Yanke, J.; Gruninger, R.; Vedres, D.; McAllister, T.; Hao, X. Bioaugmentation with an anaerobic fungus in a two-stage process for biohydrogen and biogas production using corn silage and cattail. Bioresour. Technol. 2015, 185, 79–88. [Google Scholar] [CrossRef]
- Lopez, M.J.; Suarez-Estrella, F.; Vargas-Garcia, M.C.; Lopez-Gonzales, J.A.; Verstichel, S.; Debeer, L.; Wierinck, I.; Moreno, J. Biodelignification of agricultural and forest wastes: Effect on anaerobic digestion. Biomass Bioenergy 2013, 58, 343–349. [Google Scholar] [CrossRef]
- Tisma, M.; Planinic, M.; Bucic-Kojic, A.; Panjicko, M.; Zupancic, G.D. Corn silage fungal-based solid-state pretreatment for enhanced biogas production in anaerobic co-digestion with cow manure. Bioresour. Technol. 2018, 253, 220–226. [Google Scholar] [CrossRef]
- Yadav, M.; Vivekanand, V. Biological treatment of lignocellulosic biomass by Curvularia lunata for biogas production. Bioresour. Technol. 2020, 306, 123151. [Google Scholar] [CrossRef] [PubMed]
- Akyol, C.; Ince, O.; Bozan, M.; Ozbayram, E.G.; Ince, B. Biological pretreatment with Trametes versicolor to enhance methane production from lignocellulosic biomass: A metagenomic approach. Ind. Crops Prod. 2019, 140, 111659. [Google Scholar] [CrossRef]
- Yildirim, E.; Ince, O.; Aydin, S.; Ince, B. Improvement of biogas potential of anaerobic digesters using rumen fungi. Renew. Energy 2017, 109, 346–353. [Google Scholar] [CrossRef]
- Kainthola, J.; Kalamdhad, A.S.; Goud, V.V.; Goel, R. Fungal pretreatment and associated kinetics of rice straw hydrolysis to accelerate methane yield from anaerobic digestion. Bioresour. Technol. 2019, 286, 121368. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Zheng, Z.; Cai, Y.; Zhao, Y.; Zhang, Y.; Gao, Y.; Cui, Z.; Wang, X. Accelerated biomethane production from lignocellulosic biomass: Pretreated by mixed enzymes secreted by Trichoderma viride and Aspergillus sp. Bioresour. Technol. 2020, 309, 123378. [Google Scholar] [CrossRef]
- Fang, W.; Zhang, X.; Zhang, P.; Morera, X.C.; van Lier, J.B.; Spanjers, H. Evaluation of white rot fungi pretreatment of mushroom residues for volatile fatty acid production by anaerobic fermentation: Feedstock applicability and fungal function. Bioresour. Technol. 2020, 297, 122447. [Google Scholar] [CrossRef]
- Shirkavand, E.; Baroutian, S.; Gapes, D.J.; Young, B.R. Combination of fungal and physicochemical processes for lignocellulosic biomass pretreatment–A review. Renew. Sustain. Energy Rev. 2016, 54, 217–234. [Google Scholar] [CrossRef]
- Ali, S.S.; Sun, J. Physico-chemical pretreatment and fungal biotreatment for park wastes and cattle dung for biogas production. SpringerPlus 2015, 4, 712. [Google Scholar] [CrossRef]
- Alexandropoulou, M.; Antonopoulou, G.; Fragkou, F.; Ntaikou, I.; Lyberatos, G. Fungal pretreatment of willow sawdust and its combination with alkaline treatment for enhancing biogas production. J. Environ. Manag. 2017, 203, 704–713. [Google Scholar] [CrossRef]
- Mustafa, A.M.; Poulsen, T.G.; Xia, Y.; Sheng, K. Combinations of fungal and milling pretreatments for enhancing rice straw biogas production during solid-state anaerobic digestion. Bioresour. Technol. 2017, 224, 174–182. [Google Scholar] [CrossRef]
- Wei, S. The application of biotechnology on the enhancing of biogas production from lignocellulosic waste. Appl. Microbiol. Biotechnol. 2016, 100, 9821–9836. [Google Scholar] [CrossRef] [PubMed]
- Ometto, F.; Quiroga, G.; Pšenička, P.; Whitton, R.; Jefferson, B.; Villa, R. Impacts of microalgae pre-treatments for improved anaerobic digestion: Thermal treatment, thermal hydrolysis, ultrasound and enzymatic hydrolysis. Water Res. 2014, 65, 350–361. [Google Scholar] [CrossRef] [PubMed]
- Silva, C.E.F. Avaliacao Do Potencial De Uso De Residuos Do Processamento De Frutas Na Producao De Etanol 2G. Master’s Thesis, Universida de Federal De Alagoas, Maceio, Brazil, 2014. [Google Scholar]
- Hansen, G.H.; Lubeck, M.; Frisvad, J.C.; Lubeck, P.S.; Andersen, B. Production of cellulolytic enzymes from ascomycetes: Comparison of solid state and submerged fermentation. Process Biochem. 2015, 50, 1327–1341. [Google Scholar] [CrossRef]
- Chinedu, S.N.; Okochi, V.I.; Omidiji, O. Cellulase Production By Wild Strains Of Aspergillus Niger Penicillium Chrysogenum and Trichoderma Harzianum Grown On Waste Cellulosic Materials. J. Sci. 2011, 13, 57–62. [Google Scholar]
- Mathew, G.M.; Sukumaran, R.K.; Singhania, R.R.; Pandey, A. Progress in research on fungal cellulases for lignocellulose degradation. J. Sci. Ind. Res. 2008, 67, 898–907. [Google Scholar]
- Zieminski, K.; Romanowska, I.; Kowalska, M. Enzymatic pretreatment of lignocellulosic wastes to improve biogas production. Waste Manag. Res. 2012, 32, 1131–1137. [Google Scholar] [CrossRef]
- Silva, A.F.V.; Santos, L.A.; Valença, R.B.; Porto, T.S.; Da Motta Sobrinho, M.A.; Gomes, G.J.C.; Jucá, J.F.T.; Santos, A.F.M.S. Cellulase production to obtain biogas from passion fruit (Passiflora edulis) peel waste hydrolysate. J. Environ. Chem. Eng. 2019, 7, 103510. [Google Scholar] [CrossRef]
- Edwiges, T.; Frare, L.; Mayer, B.; Lins, L.; Triolo, J.M.; Flotats, X.; Costa, M.S.S.M. Influence of chemical composition on biochemical methane potential of fruit and vegetable waste. Waste Manag. 2018, 71, 618–625. [Google Scholar] [CrossRef]
- Wang, X.; Li, Z.; Zhou, X.; Wang, Q.; Wu, Y.; Saino, M.; Bai, X. Study on the bio-methane yield and microbial community structure in enzyme enhanced anaerobic co-digestion of cow manure and corn straw. Bioresour. Technol. 2016, 219, 150–157. [Google Scholar] [CrossRef]
- Shraddha; Shekher, R.; Sehgal, S.; Kamthania, M.; Kumar, A. Laccase: Microbial Sources, Production, Purification, and Potential Biotechnological Applications-Review Article. Enzyme Res. 2011, 11. [Google Scholar] [CrossRef]
- Liew, Y.X.; Chan, Y.J.; Manickam, S.; Chong, M.F.; Chong, S.; Tiong, T.J.; Lim, J.W.; Pan, G.T. Enzymatic pretreatment to enhance anaerobic bioconversion of high strength wastewater to biogas: A review. Sci. Total Environ. 2020, 713, 136373. [Google Scholar] [CrossRef] [PubMed]
- Yaver, D.S.; Berka, R.M.; Brown, S.H.; Xu, F. The Presymposium on Recent Advances in Lignin Biodegradation and Biosynthesis, Vikki Biocentre; University of Helsinki: Helsinki, Finland, 2001. [Google Scholar]
- Patel, N.; Shahane, S.; Shivam; Majumdar, R.; Mishra, U. Mode of Action, Properties, Production, and Application of Laccase: A Review. Recent Pat. Biotechnol. 2019, 13, 19–32. [Google Scholar] [CrossRef] [PubMed]
- Couto, R.S.; Herrera, T.J.L. Industrial and biotechnological applications of laccases: A review. Biotechnol. Adv. 2006, 24, 500–513. [Google Scholar] [CrossRef] [PubMed]
- Kudanga, T.; Le Roes-Hill, M. Laccase applications in biofuels production: Current status and future prospects. Appl. Microbiol. Biotechnol. 2014, 98, 6525–6542. [Google Scholar] [CrossRef]
- Schroyen, M.; Vervaeren, H.; Van Hulle, S.W.H.; Raes, K. Impact of enzymatic pretreatment on corn stover degradation and biogas production. Bioresour. Technol. 2014, 173, 59–66. [Google Scholar] [CrossRef]
- Howard, R.L.; Abotsi, E.L.J.R.; van Rensburg, E.J.; Howard, S. Lignocellulose biotechnology: Issues of bioconversion and enzyme production. Afr. J. Biotechnol. 2003, 2, 602–619. [Google Scholar] [CrossRef]
- Hammel, K.; Cullen, D. Role of fungal peroxidases in biological ligninolysis. Curr. Opin. Plant Biol. 2008, 11, 349–355. [Google Scholar] [CrossRef]
- Martínez, A.T.; Speranza, M.; Ruiz-Dueñas, F.J.; Ferreira, P.; Camarero, S.; Guillén, F.; Martínez, M.J.; Gutiérrez, A.; del Río, J.C. Biodegradation of lignocellulosics: Microbial, chemical, and enzymatic aspects of the fungal attack of lignin. Int. Microbiol. 2005, 8, 195–204. [Google Scholar]
- Jayasinghe, P.A.; Hettiaratchi, J.P.A.; Mehrotra, A.K.; Kumar, S. Effect of enzyme additions onmethane production and lignin degradation of landfilled sample of municipal solid waste. Bioresour. Technol. 2011, 102, 4633–4637. [Google Scholar] [CrossRef]
- Hasan, F.; Ali Shah, A.; Hameed, A. Industrial applications of microbial lipases. Enzym. Microb. Technol. 2006, 39, 235–251. [Google Scholar] [CrossRef]
- Eijkman, C.U. Ber Enzyme bei bakterien und Schimmelpilzen. Cbl Bakt Parasitenk Infektionskr 1901, 29, 841–848. [Google Scholar]
- Donoso-Bravo, A.; Fdz-Polanco, M. Anaerobic co-digestion of sewage sludge and grease trap: Assessment of enzyme addition. Process Biochem. 2013, 48, 936–940. [Google Scholar] [CrossRef]
- Dors, G.; Mendes, A.A.; Pereira, E.B.; Castro, H.F.; Furigo, A. Simultaneous enzymatic hydrolysis and anaerobic biodegradation of lipid-rich wastewater from poultry industry. Appl. Water Sci. 2013, 3, 343–349. [Google Scholar] [CrossRef]
- Kameswari, K.S.B.; Kalyanaraman, C.; Porselvam, S.; Thanasekaran, K. Enhancement of Biogas Generation by Addition of Lipase in the Co-Digestion of Tannery Solid Wastes. Clean (Weinh) 2011, 39, 781–786. [Google Scholar] [CrossRef]
- Xie, B.; Cheng, J.; Zhou, J.; Song, W.; Liu, J.; Cen, K. Production of hydrogen andmethane from potatoes by two-phase anaerobic fermentation. Bioresour. Technol. 2008, 99, 5942–5946. [Google Scholar] [CrossRef]
- Bonilla, S.; Choolaei, Z.; Meyer, T.; Edwards, E.A.; Yakunin, A.F.; Grant, A.D. Evaluating the effect of enzymatic pretreatment on the anaerobic digestibility of pulp and paper biosludge. Biotechnol. Rep. 2018, 17, 77–85. [Google Scholar] [CrossRef]
- Müller, L.; Kretzschmar, J.; Pröter, J.; Liebetrau, J.; Nelles, M.; Scholwin, F. Does the addition of proteases affect the biogas yield from organic material in anaerobic. Bioresour. Technol. 2016, 203, 267–271. [Google Scholar] [CrossRef]
- Sutaryo, S.; Ward, A.J.; Moller, H.B. The effect of mixed-enzyme addition in anaerobic digestion on methane yield of dairy cattle manure. Environ. Technol. 2014, 35, 2476–2482. [Google Scholar] [CrossRef]
- Lin, Y.; Liang, J.; Zeng, C.; Wang, D.; Lin, H. Anaerobic digestion of pulp and paper mill sludge pretreated by microbial consortium OEM1 with simultaneous degradation of lignocellulose and chlorophenols. Renew. Energy 2017, 108, 108–115. [Google Scholar] [CrossRef]
- Passos, F.; Hom-Diaz, A.; Blanquez, P.; Vicent, T.; Ferrer, I. Improving biogas production from microalgae by enzymatic pretreatment. Bioresour. Technol. 2016, 199, 347–351. [Google Scholar] [CrossRef]
- Al-Zuhair, S.; Ahmed, K.; Abdulrazak, A.; El-Naas, M.H. Synergistic effect of pretreatment and hydrolysis enzymes on the production of fermentable sugars from date palm lignocellulosic waste. J. Ind. Eng. Chem. 2013, 19, 413–415. [Google Scholar] [CrossRef]
- Lee, J.-W.; Gwak, K.-S.; Park, J.-Y.; Park, M.-J.; Choi, D.-H.; Kwon, M.; Choi, I.-G. Biological pretreatment of softwood Pinus densiflora by three white rot fungi. J. Microbiol. 2007, 45, 485–491. [Google Scholar] [PubMed]
- Pérez-Rodríguez, N.; García-bernet, D.; Domínguez, J.M. Extrusion and enzymatic hydrolysis as pretreatments on corn cob for biogas production. Renew. Energy 2017, 107, 597–603. [Google Scholar] [CrossRef]
- Domingues, R.F.; Sanches, T.; Silva, G.S.; Bueno, B.E.; Ribeiro, R.; Kamimura, E.S.; Franzolin Neto, R.; Tommaso, G. Effect of enzymatic pretreatment on the anaerobic digestion of milk fat for biogas production. Food Res. Int. 2015, 73, 26–30. [Google Scholar] [CrossRef]
| Source | Cellulose | Hemicellulose | Lignin | References |
|---|---|---|---|---|
| Hardwood | 40–55 | 24–40 | 18–25 | [22] |
| Eucalyptus | 44.9 | 28.9 | 26.2 | [23] |
| Oak | 43.2 | 21.9 | 35.4 | [24] |
| Rubber wood | 39.56 | 28.42 | 27.58 | [25] |
| Softwood | ||||
| Pine | 45.6 | 24 | 26.8 | [24] |
| Japanese cedar | 52.7 | 13.8 | 33.5 | [23] |
| Grasses | ||||
| Bamboo | 46.5 | 18.8 | 25.7 | [26] |
| Bamboo leaves | 31.14 | 25.55 | 35.03 | [27] |
| Bamboo stem | 43.04 | 22.13 | 27.14 | [13] |
| Amur silver-grass | 42 | 30.15 | 7 | [28] |
| Natural hay | 44.9 | 31.4 | 12 | [29] |
| Hemp | 53.86 | 10.6 | 8.76 | [28] |
| Rye | 42.83 | 27.86 | 6.51 | [28] |
| Rye straw | 38 | 36.9 | 17.6 | [30] |
| Reed | 49.40 | 31.5 | 8.74 | [28] |
| Stalk of giant reed | 33.1 | 18.5 | 24.5 | [31] |
| Leaves of giant reed | 20.9 | 17.7 | 25.4 | [31] |
| Sunflower | 34.06 | 5.18 | 7.72 | [28] |
| Sunflower stalk | 31 | 15.6 | 29.2 | [31] |
| Silage | 39.27 | 25.96 | 9.02 | [28] |
| Agro-industrial waste | ||||
| Walnut shell | 23.3 | 20.4 | 53.5 | [29] |
| Groundnut shell | 37 | 18.7 | 28 | [32] |
| Pistachio shell | 15.2 | 38.2 | 29.4 | [32] |
| Almond shell | 27 | 30 | 36 | [33] |
| Pine nut shell | 31 | 25 | 38 | [33] |
| Hazelnut shell | 30 | 23 | 38 | [33] |
| Nut shells | 25–30 | 25–30 | 30–40 | [22] |
| Coconut coir | 44.2 | 22.1 | 32.8 | [32] |
| Cotton stalk | 67 | 16 | 13 | [34] |
| Hemp stalk | 52 | 25 | 17 | [34] |
| Acacia pruning | 49 | 13 | 32 | [34] |
| Sugarcane peel | 41.11 | 26.4 | 24.31 | [27] |
| Sugarcane | 25 | 17 | 12 | [35] |
| Rice husk | 40 | 16 | 26 | [36] |
| Barley straw | 35.4 | 28.7 | 13.1 | [37] |
| 37.5 | 25.3 | 26.1 | [30] | |
| Maize straw | 38.33 | 29.76 | 3.82 | [13] |
| Rice straw | 38.14 | 31.12 | 26.35 | [27] |
| 32 | 24 | 13 | [38] | |
| Wheat straw | 38.2 | 21.2 | 23.4 | [39] |
| 43.4 | 26.9 | 22.2 | [13] | |
| Corn stover | 43.97 | 28.94 | 21.82 | [27] |
| 37.5 | 22.4 | 17.6 | [40] | |
| Miscanthus | 36.3 | 22.16 | 22.55 | [13] |
| Switchgrass | 31–45 | 20–31 | 12–18 | [39] |
| Sugarcane | 25 | 17 | 12 | [35] |
| Sorghum straw | 26.93 | 32.57 | 10.16 | [13] |
| Willow sawdust | 35.6 | 21.5 | 28.7 | [13] |
| Substrate Category | Enzyme | Enzyme Origin | Conditions of Pretreatment | Effects | Reference |
|---|---|---|---|---|---|
| Pulp and paper sludge | Endoglucanase Laccase | Pleurotus ostreatus | 37 °C for 4 h | +34% CH4 yield improvement | [145] |
| Sugar beet pulp and spent hops | Celustar XL | Trichoderma longibrachiatum | 8 days at 50 °C | +13–19% CH4 yield improvement | [121] |
| Microalgae | Cellulase Enzyme mix | / | 37 °C for 6 h | +8–15% CH4 yield improvement | [146] |
| Willow | Laccase | Trametes versicolor | 30 °C for 6–24 h | +33% CH4 yield improvement | [43] |
| Palm tree stalk | Laccase | Acquired from Sigma Aldrich Canada Ltd. | 45 °C for a number of 24 h | A rise of reducing sugar concentration from 5.6 to 60% | [147] |
| Pinus densiflora | Laccase MnP | Stereum hirsutum | 30 °C for 8 weeks | +14.5% lignin decomposition | [148] |
| Fleshings | Lipase | Purchased from Sisco Research laboratories | A residence time of 42 days in anaerobic condition | +15% CH4 volume improvement control | [140] |
| Manure | Laccase | Commercial Novozyme 51003 | / | 19.8 ± 0.4 m3 CH4 (t WW)−1) yield improvement | [130] |
| Corn cobs | Cellulolytic enzymatic cocktail | Novozymes | 3 h at 40 °C | +14% CH4 yield improvement | [149] |
| Butter | Lipase | Candida rugosa | 16 h at 40 °C | +84% CH4 yield improvement | [150] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ferdeș, M.; Dincă, M.N.; Moiceanu, G.; Zăbavă, B.Ș.; Paraschiv, G. Microorganisms and Enzymes Used in the Biological Pretreatment of the Substrate to Enhance Biogas Production: A Review. Sustainability 2020, 12, 7205. https://doi.org/10.3390/su12177205
Ferdeș M, Dincă MN, Moiceanu G, Zăbavă BȘ, Paraschiv G. Microorganisms and Enzymes Used in the Biological Pretreatment of the Substrate to Enhance Biogas Production: A Review. Sustainability. 2020; 12(17):7205. https://doi.org/10.3390/su12177205
Chicago/Turabian StyleFerdeș, Mariana, Mirela Nicoleta Dincă, Georgiana Moiceanu, Bianca Ștefania Zăbavă, and Gigel Paraschiv. 2020. "Microorganisms and Enzymes Used in the Biological Pretreatment of the Substrate to Enhance Biogas Production: A Review" Sustainability 12, no. 17: 7205. https://doi.org/10.3390/su12177205
APA StyleFerdeș, M., Dincă, M. N., Moiceanu, G., Zăbavă, B. Ș., & Paraschiv, G. (2020). Microorganisms and Enzymes Used in the Biological Pretreatment of the Substrate to Enhance Biogas Production: A Review. Sustainability, 12(17), 7205. https://doi.org/10.3390/su12177205








