Land Sparing Can Maintain Bird Diversity in Northeastern Bangladesh
Abstract
1. Introduction
2. Materials and Methods
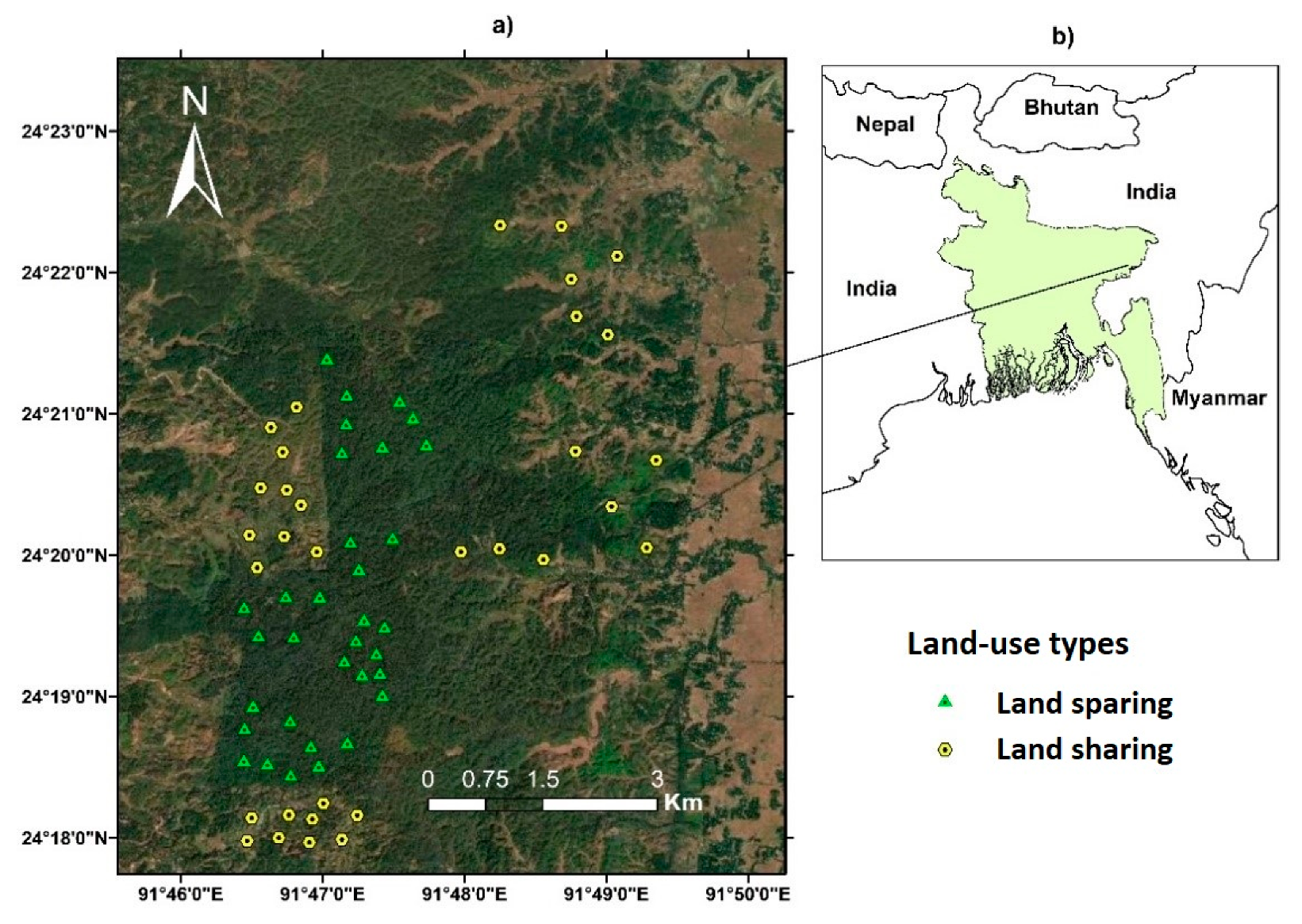
2.1. Study Area
2.2. Experimental Design
2.3. Bird Identification and Counting
2.4. Data Analysis
3. Results
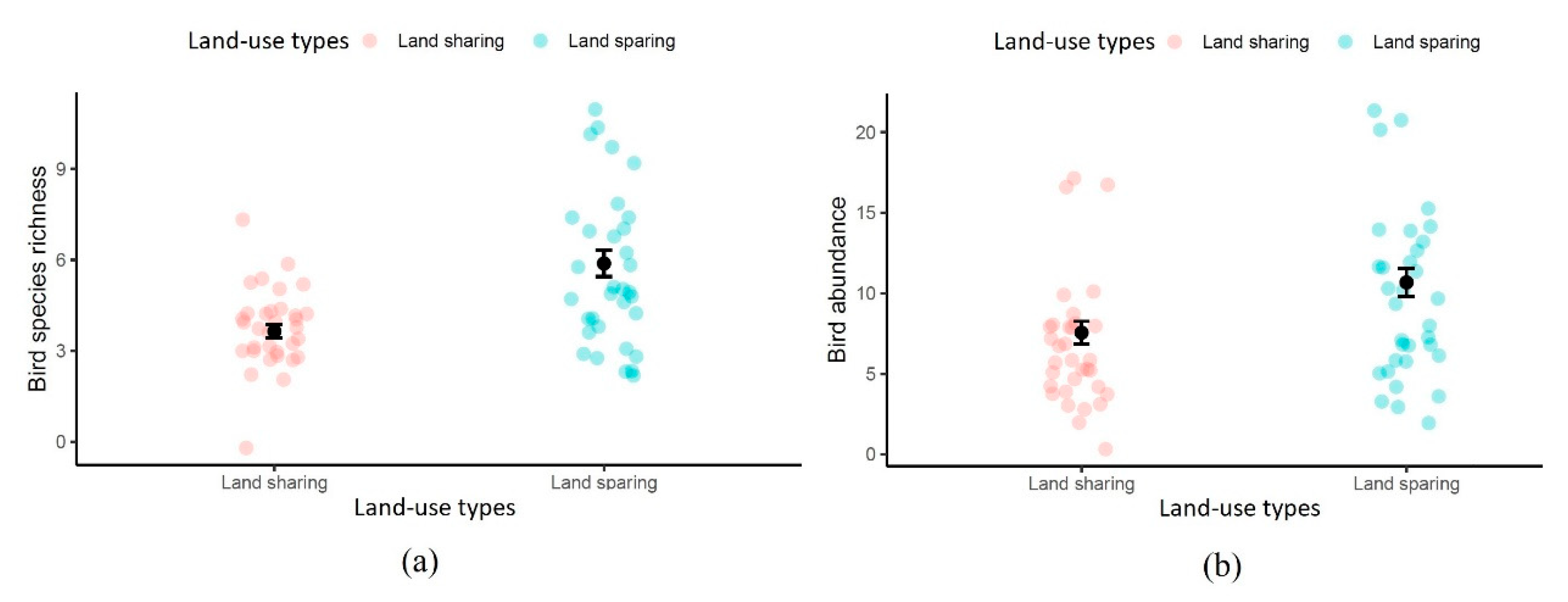
3.1. Abundance
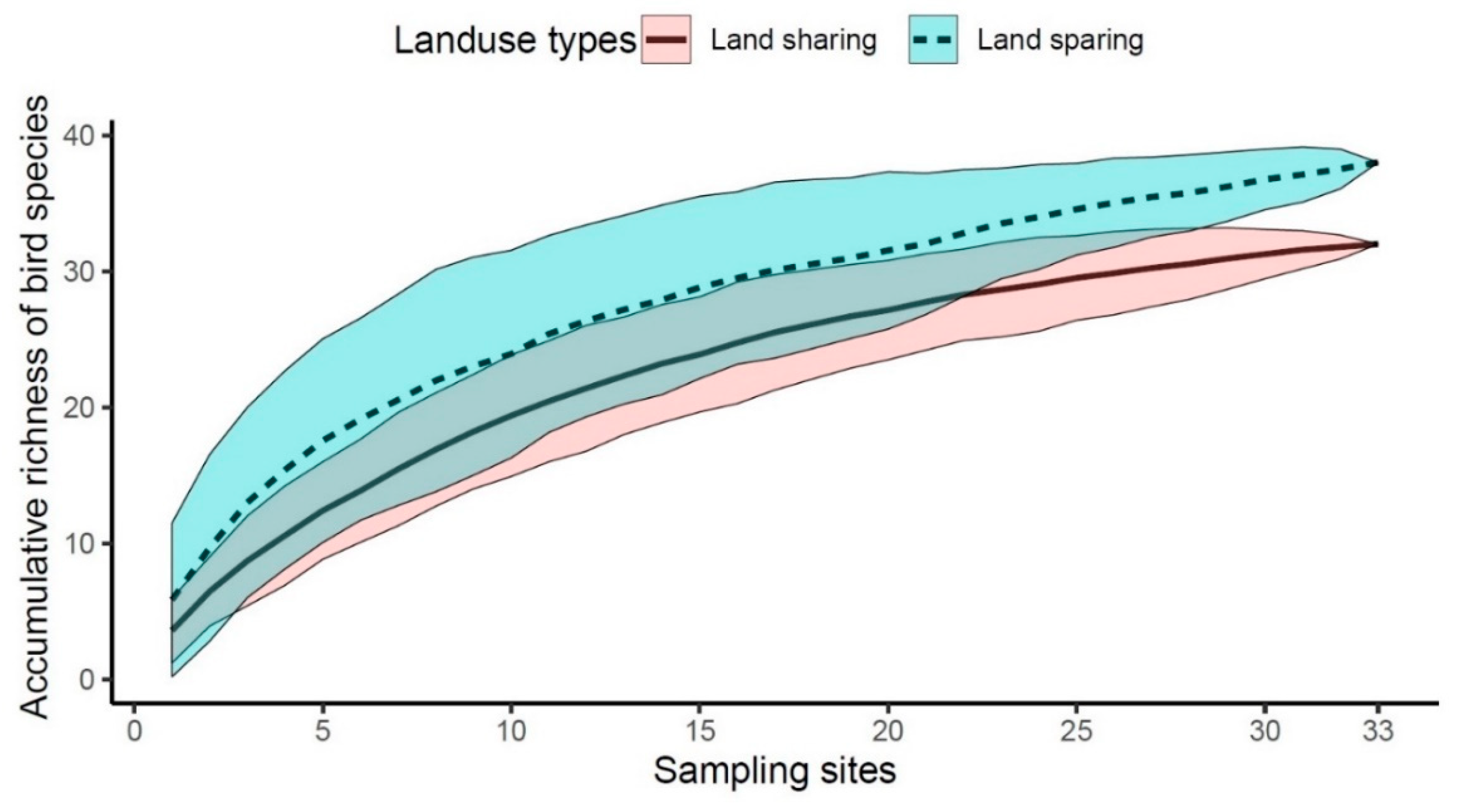
3.2. Richness and Diversity
3.3. Indicator Species
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Foley, J.; Defries, R.; Asner, G.; Barford, C.; Bonan, G.; Carpenter, S.; Chapin, F.S., III; Coe, M.; Daily, G.; Gibbs, H.; et al. Global consequences of land use. Science 2005, 309, 570–574. [Google Scholar] [CrossRef] [PubMed]
- Rands, M.; Adams, W.; Bennun, L.; Butchart, S.; Clements, A.; Coomes, D.; Entwistle, A.; Hodge, I.; Kapos, V.; Scharlemann, J.; et al. Biodiversity conservation: Challenges beyond 2010. Science 2010, 329, 1298–1303. [Google Scholar] [CrossRef] [PubMed]
- Quarrie, J. (Ed.) Earth Summit’ 92: The United Nations Conference on Environment and Development; The Regency Press: London, UK, 1992. [Google Scholar]
- Gabriel, D.; Roschewitz, I.; Tscharntke, T.; Thies, C. Beta diversity at different spatial scales: Plant communities in organic and conventional agriculture. Ecol. Appl. 2006, 16, 2011–2021. [Google Scholar] [CrossRef]
- Tscharntke, T.; Clough, Y.; Wanger, T.C.; Jackson, L.; Motzke, I.; Perfecto, I.; Vandermeer, J.; Whitbread, A. Global food security, biodiversity conservation and the future of agricultural intensification. Biol. Conserv. 2012, 151, 53–59. [Google Scholar] [CrossRef]
- Martin, M.; Girona, M.M.; Morin, H. Driving factors of conifer regeneration dynamics in eastern Canadian boreal old-growth forests. PLoS ONE 2020, 15, 0230221. [Google Scholar] [CrossRef]
- Godfray, H.C.J. Food and biodiversity. Science 2011, 333, 1231–1232. [Google Scholar] [CrossRef]
- Wilkinson, J.B. The State Role in Biodiversity Conservation. Sci. Technol. 1999, 15, 71–77. [Google Scholar]
- Frison, E.; Jeremy, C.; Hodgkin, T. Agricultural biodiversity is essential for a sustainable improvement in food and nutrition security. Sustainability 2011, 3, 238. [Google Scholar] [CrossRef]
- FAO. Sustainable Agriculture for Biodiversity; FAO: Rome, Italy, 2018. [Google Scholar]
- Fischer, J.; Abson, D.J.; Butsic, V.; Chappell, M.J.; Ekroos, J.; Hanspach, J.; Von Wehrden, H. Land sparing versus land sharing: Moving forward. Conserv. Lett. 2014, 7, 149–157. [Google Scholar] [CrossRef]
- Green, R.; Cornell, S.; Scharlemann, J.; Balmford, A. Farming and the fate of wild nature. Science 2005, 307, 550–555. [Google Scholar] [CrossRef]
- Phalan, B.; Onial, M.; Balmford, A.; Green, R.E. Reconciling food production and biodiversity conservation: Land sharing and land sparing compared. Science 2011, 333, 1289–1291. [Google Scholar] [CrossRef] [PubMed]
- Trisurat, Y.; Shirakawa, H.; Johnston, J.M. Land-use/land-cover change from socio-economic drivers and their impact on biodiversity in Nan province, Thailand. Sustainability 2019, 11, 649. [Google Scholar] [CrossRef]
- Kremen, C. Reframing the land-sparing/land-sharing debate for biodiversity conservation. Annal. N. Y. Acad. Sci. 2015, 1355. [Google Scholar] [CrossRef] [PubMed]
- Steffan-Dewenter, I.; Kessler, M.; Barkmann, J.; Bos, M.M.; Buchori, D.; Erasmi, S.; Faust, H.; Gerold, G.; Glenk, K.; Gradstein, S.R.; et al. Tradeoffs between income, biodiversity, and ecosystem functioning during tropical rainforest conversion and agroforestry intensification. Proc. Natl. Acad. Sci. USA 2007, 104, 4973–4978. [Google Scholar] [CrossRef] [PubMed]
- Mastrangelo, M.E.; Gavin, M.C. Trade-Offs between Cattle Production and Bird Conservation in an Agricultural Frontier of the Gran Chaco of Argentina. Conserv. Biol. 2012, 26, 1040–1051. [Google Scholar] [CrossRef] [PubMed]
- Wade, A.S.I.; Asase, A.; Hadley, P.; Mason, J.; Ofori-Frimpong, K.; Preece, D.; Spring, N.; Norris, K. Management strategies for maximizing carbon storage and tree species diversity in cocoa-growing landscapes. Agric. Ecosyst. Environ. 2010, 138, 324–334. [Google Scholar] [CrossRef]
- Hulme, M.F.; Vickery, J.A.; Green, R.E.; Phalan, B.; Chamberlain, D.E.; Pomeroy, D.E.; Nalwanga, D.; Mushabe, D.; Katebaka, R.; Bolwig, S.; et al. Conserving the Birds of Uganda’s Banana-Coffee Arc: Land Sparing and Land Sharing Compared. PLoS ONE 2013, 8, e54597. [Google Scholar] [CrossRef]
- Da Silva, T.W.; Dotta, G.; Fontana, C.S. Structure of avian assemblages in grasslands associated with cattle ranching and soybean agriculture in the Uruguayan savanna ecoregion of Brazil and Uruguay. Condor 2015, 117, 53–63. [Google Scholar] [CrossRef]
- Gilroy, J.J.; Woodcock, P.; Edwards, F.A.; Wheeler, C.; Uribe, C.A.M.; Haugaasen, T.; Edwards, D.P. Optimizing carbon storage and biodiversity protection in tropical agricultural landscapes. Glob. Change Biol. 2014, 20, 2162–2172. [Google Scholar] [CrossRef]
- Edwards, D.P.; Gilroy, J.J.; Woodcock, P.; Edwards, F.A.; Larsen, T.H.; Andrews, D.J.R.; Derhe, M.A.; Docherty, T.D.S.; Hsu, W.W.; Mitchell, S.L.; et al. Land-sharing versus land-sparing logging: Reconciling timber extraction with biodiversity conservation. Glob. Change Biol. 2014, 20, 183–191. [Google Scholar] [CrossRef]
- Mainuddin, M.; Kirby, M. National food security in Bangladesh to 2050. Food Secur. 2015, 7. [Google Scholar] [CrossRef]
- United Nations Department of Economic and Social Affairs Population Division. World Population Prospects 2019; UN: New York, NY, USA, 2019. [Google Scholar]
- UN Population Division. World Population Prospects: The 2012 Revision, Economic & Social Affairs; UN: New York, NY, USA, 2013. [Google Scholar] [CrossRef]
- Mukul, S.; Biswas, S.; Rashid, A.Z.M. Biodiversity in Bangladesh. Global Biodevirsity; Apple Academic Press: Palm Bey, FL, USA, 2018. [Google Scholar] [CrossRef]
- Mittermeier, R.A.; Myers, N.; Thomsen, J.B.; Da Fonseca, G.A.; Olivieri, S. Biodiversity hotspots and major tropical wilderness areas: Approaches to setting conservation priorities. Conserv. Biol. 1998, 12, 516–520. [Google Scholar] [CrossRef]
- Khan, M.T. The Nishorgo Support Project, the Lawachara National Park, and the Chevron seismic survey: Forest conservation or energy procurement in Bangladesh? J. Polit. Ecol. 2018. [Google Scholar] [CrossRef]
- Huq, S. Climate change and Bangladesh. Science 2001, 294, 1617. [Google Scholar] [CrossRef]
- Montoro Girona, M.; Morin, H.; Lussier, J.M.; Ruel, J.C. Post-cutting mortality following experimental silvicultural treatments in unmanaged boreal forest stands. Front. For. Glob. Change 2019, 2, 4. [Google Scholar] [CrossRef]
- Lavoie, J.; Montoro Girona, M.; Morin, H. Vulnerability of conifer regeneration to spruce budworm outbreaks in the Eastern Canadian boreal forest. Forests 2019, 10, 850. [Google Scholar] [CrossRef]
- Labrecque-Foy, J.P.; Morin, H.; Girona, M.M. Dynamics of territorial occupation by North American beavers in Canadian boreal forests: A novel dendroecological approach. Forests 2020, 11, 221. [Google Scholar] [CrossRef]
- Montoro Girona, M.; Navarro, L.; Morin, H. A secret hidden in the sediments: Lepidoptera scales. Front. Ecol. Evol. 2018, 6, 2. [Google Scholar] [CrossRef]
- Navarro, L.; Morin, H.; Bergeron, Y.; Girona, M.M. Changes in spatiotemporal patterns of 20th century spruce budworm outbreaks in eastern Canadian boreal forests. Front. Plant Sci. 2018, 9, 1905. [Google Scholar] [CrossRef]
- Seidl, R.; Fernandes, P.M.; Fonseca, T.F.; Gillet, F.; Jönsson, A.M.; Merganičová, K.; González-Olabarria, J.R. Modelling natural disturbances in forest ecosystems: A review. Ecol. Model. 2011, 222, 903–924. [Google Scholar] [CrossRef]
- Reid Bell, A.; Bryan, E.; Ringler, C.; Ahmed, A. Rice productivity in Bangladesh: What are the benefits of irrigation? Land Use Policy 2015, 48, 1–12. [Google Scholar] [CrossRef]
- FAO; CIFOR. FAO Framework Methodology for Climate Change Vulnerability Assessments of Forests and Forest Dependent People; FAO: Rome, Italy, 2019. [Google Scholar]
- Pachauri, R.K.; Reisinger, A. Climate Change 2007: Synthesis Report; Contribution of Working Groups I, II and III to the Fourth Assessment Report of the Intergovernmental Panel on Climate; IPCC: Geneva, Switzerland, 2007. [Google Scholar]
- Padoa-Schioppa, E.; Baietto, M.; Massa, R.; Bottoni, L. Bird communities as bioindicators: The focal species concept in agricultural landscapes. Ecol. Indic. 2006, 6, 83–93. [Google Scholar] [CrossRef]
- Keast, D.; Newsholme, E.A. Effect of mitogens on the maximum activities of hexokinase, lactate dehydrogenase, citrate synthase and glutaminase in rat mesenteric lymph node lymphocytes and splenocytes during the early period of culture. Int. J. Biochem. 1990, 22, 133–136. [Google Scholar] [CrossRef]
- Versluijs, M.; Hjältén, J.; Roberge, J.M. Ecological restoration modifies the value of biodiversity indicators in resident boreal forest birds. Ecol. Indic. 2019, 98, 104–111. [Google Scholar] [CrossRef]
- Koutsos, E.A.; Matson, K.D.; Klasing, K.C. Nutrition of birds in the order Psittaciformes: A review. J. Avian Med. Surg. 2001. [Google Scholar] [CrossRef]
- Phalan, B. What have we learned from the land sparing-sharing model? Sustainability 2018, 10, 1760. [Google Scholar] [CrossRef]
- Rahman, S.C.; Rashid, S.M.A.; Datta, R.; Mro, P.; Roy, C. Status, exploitation, and conservation of freshwater turtles and tortoises in Chittagong Hill Tracts, Bangladesh. Chelonian Conserv. Biol. 2015, 14, 130–135. [Google Scholar] [CrossRef]
- Feeroz, M.M.; Islam, M.A. Primates of the West Bhanugach Forest Reserve: Major threats and management plan. In Bangladesh Environment 2000; Feeroz Ahmed, M., Ed.; BAPA: Dhaka, Bangladesh, 2000; pp. 239–253. [Google Scholar]
- Halim, M.A.; Shahid, A.; Chowdhury, M.S.H.; Nahar, M.N.; Sohel, M.S.I.; Nuruddin, M.J.; Koike, M. Evaluation of land-use pattern change in West Bhanugach Reserved Forest, Bangladesh, using remote sensing and GIS techniques. J. For. Res. 2008, 19, 193. [Google Scholar] [CrossRef]
- Alam, M.K. Annotated Checlist of Woody Flora of Sylhet Forests. Bulletin 5; Plant Taxonomy Series; Bangladesh Forest Research Institute: Chattogram, Bangladesh, 1998. [Google Scholar]
- Mehedi, M.A.H.; Kundu, C.; Chowdhury, M.Q. Patterns of tree buttressing at Lawachara National Park, Bangladesh. J. For. Res. 2012, 23, 461–466. [Google Scholar] [CrossRef]
- Hossain, M.K.; Islam, Q.N.; Islam, S.A.; Tarafdar, M.A.; Zashimuddin, M.; Ahmed, A. Assistance to the Second Agricultural Project-Bangladesh, Status Report on the Activities of the Silviculture Research Division; BFRI: Chittagong, Bangladesh, 1989.
- Muzaffar, S.B.; Islam, M.; Feeroz, M.; Begum, S.; Mahmud, M.; Chakma, S.; Hasan, M. Habitat characteristics of the endangered hoolock gibbons of Bangladesh: The role of plant species richness. Biotropica 2007, 39, 539–545. [Google Scholar] [CrossRef]
- Uddin, M. Angiosperm diversity of Lawachara National Park (Bangladesh): A preliminary assessment. Bangladesh J. Plant Taxon. 2010, 17. [Google Scholar] [CrossRef]
- Deb, J.C.; Roy, A.; Wahedunnabi, M. Structure and composition of understory treelets and overstory trees in a protected area of Bangladesh. For. Sci. Technol. 2015. [Google Scholar] [CrossRef]
- Uddin, M.B.; Steinbauer, M.J.; Jentsch, A.; Mukul, S.A.; Beierkuhnlein, C. Do environmental attributes, disturbances and protection regimes determine the distribution of exotic plant species in Bangladesh forest ecosystem? For. Ecol. Manag. 2013. [Google Scholar] [CrossRef]
- Reza, A.; Perry, G. Herpetofaunal species richness in the tropical forests of Bangladesh. Asian J. Conserv. Biol. 2015, 4, 100–108. [Google Scholar]
- Rahman, S.; Rashid, S.M.A.; Das, K.; Jenkins, C.; Luiselli, L. Monsoon does matter: Annual activity patterns in a snake assemblage from Bangladesh. Herpetol. J. 2013, 23, 203–208. [Google Scholar]
- Khan, M.; Monirul, H. Protected Areas of Bangladesh: A Guide to Wildlife. Nishorgo Program, Wildlife Management and Nature Conservation Circle; Bangladesh Forest Department: Dhaka, Bangladesh, 2008.
- IUCN Bangladesh. Red list of Bangladesh: Summary; International Union for Conservation of Nature, Bangladesh Country Office: Dhaka, Bangladesh, 2015; Volume 1, p. 122. [Google Scholar]
- Bibby, C.J.; Burgess, N.D.; Hill, D.A. Description and measurement of bird habitat. Bird Census Tech. 1992. [Google Scholar] [CrossRef]
- Haselmayer, J.; Quinn, J.S. A comparison of point counts and sound recording as bird survey methods in Amazonian Southeast Peru. Condor 2000, 102, 887–893. [Google Scholar] [CrossRef]
- Scholes, E., III. Macaulay Library Audio and Video Collection; Cornell Lab of Ornithology: Ithaca, NY, USA, 2015. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Core Team: Vienna, Austria, 2013. [Google Scholar]
- Oksanen, J.; Blanchet, F.G.; Friendly, M.; Kindt, R.; Legendre, P.; McGlinn, D.; Minchin, P.R.; O’Hara, R.B.; Simpson, G.L.; Solymos, P.; et al. Vegan: Community Ecology Package. Available online: https://CRAN.R-project.org/package=vegan (accessed on 26 July 2019).
- De Cáceres, M.; Legendre, P. Associations between species and groups of sites: Indices and statistical inference. Ecology 2009, 90, 3566–3574. [Google Scholar] [CrossRef]
- Wickham, H. ggplot2. WIREs Comp. Stat. 2011, 3, 180–185. [Google Scholar] [CrossRef]
- Benayas, J.M.R.; Bullock, J.M. Restoration of biodiversity and ecosystem services on agricultural land. Ecosystems 2012, 15, 883–899. [Google Scholar] [CrossRef]
- Ahmed, A. Issues in social forestry in Bangladesh: The betagi-pomora experiment. J. Rural Dev. 1988, 7, 263–274. [Google Scholar]
- Kamp, J.; Urazaliev, R.; Balmford, A.; Donald, P.F.; Green, R.E.; Lamb, A.J.; Phalan, B. Agricultural development and the conservation of avian biodiversity on the Eurasian steppes: A comparison of land-sparing and land-sharing approaches. J. Appl. Ecol. 2015. [Google Scholar] [CrossRef]
- Arlettaz, R. The importance of habitat heterogeneity at multiple scales for birds in European agricultural landscapes. In Birds and Habitat: Relationships in Changing Landscapes; Cambridge University Press: Cambridge, UK, 2012; p. 177. [Google Scholar]
- Tilman, D.; Fargione, J.; Wolff, B.; D’Antonio, C.; Dobson, A.; Howarth, R.; Swackhamer, D. Forecasting agriculturally driven global environmental change. Science 2001, 292, 281–284. [Google Scholar] [CrossRef] [PubMed]
- Krauss, J.; Bommarco, R.; Guardiola, M.; Heikkinen, R.K.; Helm, A.; Kuussaari, M.; Steffan-Dewenter, I. Habitat fragmentation causes immediate and time-delayed biodiversity loss at different trophic levels. Ecol. Lett. 2010, 13, 597–605. [Google Scholar] [CrossRef]
- Mineau, P.; Whiteside, M. Pesticide acute toxicity is a better correlate of U.S. grassland bird declines than agricultural intensification. PLoS ONE 2013, 8, e57457. [Google Scholar] [CrossRef]
- Bujoczek, M.; Rybicka, J.; Bujoczek, L. Effects of disturbances in a subalpine forest on its structural indicators and bird diversity. Ecol. Indic. 2020, 112. [Google Scholar] [CrossRef]
- Quiroga, M.; Leon, E.; Beltzer, A.; Olguin, P. Diet of black-crowned night-herons (Nycticorax nycticorax) in a Wetland of the Parana River’s Alluvial Valley. Ekoloji 2013, 22, 43–50. [Google Scholar] [CrossRef]
- Zawadzka, D. Feeding habits of the Black Kite Milvus migrans, Red Kite Milvus milvus, White-tailed Eagle Haliaeetus albicilla and Lesser Spotted Eagle Aquila pomarina in Wigry National Park (NE Poland). Acta Ornithologica 1999, 34, 65–75. [Google Scholar]
- Noss, R. Protected areas: How much is enough? In National Parks and Protected Areas: Their Role in Environmental Protection; Wright, R.G., Ed.; Blackwell Science: Cambridge, MA, USA, 1996. [Google Scholar]
- Martino, D. Buffer zones around protected areas: A brief literature review. Electron. Green J. 2001. [Google Scholar] [CrossRef]
- Liu, A.-Z.; Li, D.-Z.; Wang, H.; Kress, W.J. Ornithophilous and chiropterophilous pollination in Musa itinerans (musaceae), a pioneer species in tropical rain forests of Yunnan, Southwestern China. Biotropica 2002, 34, 254–260. [Google Scholar] [CrossRef]
- Daniels, R.J.R.; Malati, H.; Madhav, G. Birds of man-made ecosystems: The plantations. Proc. Animal Sci. 1990, 99, 79–89. [Google Scholar] [CrossRef]
- Nature Conservation Management (NACOM). Co-Management of Tropical Forest Resources of Bangladesh: Secondary Data Collection for Pilot Protected Areas: Lawachara National Park; USAID/Bangladesh Ministry of Environment and Forest: Dhaka, Bangladesh, 2003. [Google Scholar]
- Santharam, V. Display behavior in woodpeckers. Newslett. Birdwatchers 1997, 37, 98–99. [Google Scholar]
- Winkler, H.; Christie, D.A. Rufous woodpecker (Micropternus brachyurus). In Handbook of the Birds of the World Alive; Del Hoyo, J., Elliott, A., Sargatal, J., Christie, D.A., De Juana, E., Eds.; Lynx Edicions: Barcelona, Spain, 2020. [Google Scholar]
- Rasmussen, P.C.; Anderton, J.C. Birds of South Asia: The Ripley Guide; Smithsonian Institution and Lynx Edicions: Barcelona, Spain, 2005; pp. 395–396. ISBN 8487334679. [Google Scholar]
- Sarker, S.; Deb, J.C.; Halim, M.A. A diagnosis of existing logging bans in Bangladesh. Int. For. Rev. 2011, 13, 461–475. [Google Scholar] [CrossRef]
- Younus, M.A.F.; Harvey, N. Community-based flood vulnerability and adaptation assessment: A case study from Bangladesh. J. Environ. Assess. Policy Manag. 2013. [Google Scholar] [CrossRef]
- Wanjari, R.H.; Mandal, K.G.; Ghosh, P.K.; Adhikari, T.; Rao, N.H. Rice in India: Present status and strategies to boost its production through hybrids. J. Sustain. Agric. 2006. [Google Scholar] [CrossRef]

| Land Sparing | Land Sharing | |||||
|---|---|---|---|---|---|---|
| Sampling Sites | Abundance | Species Richness | Species Diversity (H) | Abundance | Species Richness | Species Diversity (H) |
| 1 | 14 | 3 | 0.9 | 10 | 4 | 1.4 |
| 2 | 7 | 4 | 1.3 | 3 | 2 | 1.1 |
| 3 | 10 | 5 | 1.5 | 4 | 3 | 1 |
| 4 | 7 | 4 | 1.3 | 3 | 3 | 1.1 |
| 5 | 2 | 2 | 0.7 | 5 | 2 | 0.7 |
| 6 | 21 | 10 | 2.1 | 8 | 4 | 1.3 |
| 7 | 4 | 3 | 1 | 17 | 7 | 1.8 |
| 8 | 3 | 2 | 0.6 | 7 | 4 | 1.3 |
| 9 | 8 | 5 | 1.5 | 4 | 4 | 1.3 |
| 10 | 14 | 7 | 1.8 | 2 | 2 | 0.7 |
| 11 | 8 | 5 | 1.5 | 16 | 5 | 1.4 |
| 12 | 13 | 7 | 1.7 | 8 | 3 | 0.9 |
| 13 | 5 | 4 | 1.3 | 8 | 4 | 1.2 |
| 14 | 5 | 2 | 0.5 | 10 | 3 | 0.8 |
| 15 | 10 | 7 | 1.9 | 17 | 6 | 1.7 |
| 16 | 13 | 9 | 2.1 | 5 | 4 | 1.3 |
| 17 | 12 | 6 | 1.7 | 5 | 3 | 1.1 |
| 18 | 7 | 5 | 1.5 | 4 | 4 | 1.4 |
| 19 | 14 | 7 | 1.9 | 10 | 4 | 1.3 |
| 20 | 14 | 8 | 1.9 | 7 | 5 | 1.5 |
| 21 | 9 | 6 | 1.7 | 8 | 3 | 1 |
| 22 | 10 | 5 | 1.6 | 8 | 3 | 1.1 |
| 23 | 15 | 10 | 2.2 | 7 | 3 | 1.1 |
| 24 | 21 | 10 | 2.1 | 8 | 4 | 1.3 |
| 25 | 20 | 11 | 2.3 | 6 | 4 | 1.3 |
| 26 | 4 | 3 | 1 | 4 | 4 | 1.4 |
| 27 | 6 | 4 | 1.3 | 5 | 3 | 1 |
| 28 | 6 | 5 | 1.6 | 3 | 3 | 1.1 |
| 29 | 7 | 5 | 1.5 | 3 | 3 | 1.1 |
| 30 | 6 | 4 | 1.3 | 4 | 4 | 1.4 |
| 31 | 7 | 6 | 1.7 | 5 | 5 | 1.6 |
| 32 | 12 | 7 | 1.9 | 4 | 4 | 1.4 |
| 33 | 3 | 3 | 1.1 | 6 | 5 | 1.6 |
| Average ± SD | 9.6 ± 5.4 | 5.6 ± 2.5 | 1.52 ± 0.45 | 6.8 ± 4.2 | 3.7 ± 1.2 | 1.23 ± 0.27 |
| Bird Species | Mean Abundance in Land Sparing | Mean Abundance in Land Sharing | p-Value (Welch’s t-Tests) | Photographs of the Species |
|---|---|---|---|---|
| Arachnothera longirostra | 15 ± 5.8 | 4 ± 2.7 | 0.007 |  |
| Micropternus brachyurus | 18 ± 5.6 | 0 ± 2.6 | 0.013 |  |
| Copsychus malabaricus | 20 ± 5.3 | 4 ± 2.7 | 0.050 |  |
| Hemixos flavala | 11 ± 8.4 | 2 ± 3.5 | 0.088 |  |
| Megalaima asiatica | 10 ± 8.6 | 2 ± 3.5 | 0.158 |  |
| Corvus macrorhynchos | 0 ± 2.1 | 3 ± 1.9 | 0.208 |  |
| Upupa epops | 0 ± 2.1 | 2 ± 2 | 0.227 |  |
| Milvus migrans | 0 ± 2.0 | 2 ± 2 | 0.477 |  |
| Orthotomus atrogularis | 2 ± 2.1 | 7 ± 2.6 | 0.480 |  |
| Dicrurus leucophaeus | 0 ± 2.1 | 2 ± 2 | 0.490 |  |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hasan, A.; Girona, M.M.; Grosbois, G.; Saha, N.; Halim, M.A. Land Sparing Can Maintain Bird Diversity in Northeastern Bangladesh. Sustainability 2020, 12, 6472. https://doi.org/10.3390/su12166472
Hasan A, Girona MM, Grosbois G, Saha N, Halim MA. Land Sparing Can Maintain Bird Diversity in Northeastern Bangladesh. Sustainability. 2020; 12(16):6472. https://doi.org/10.3390/su12166472
Chicago/Turabian StyleHasan, Akib, Miguel Montoro Girona, Guillaume Grosbois, Narayan Saha, and Md Abdul Halim. 2020. "Land Sparing Can Maintain Bird Diversity in Northeastern Bangladesh" Sustainability 12, no. 16: 6472. https://doi.org/10.3390/su12166472
APA StyleHasan, A., Girona, M. M., Grosbois, G., Saha, N., & Halim, M. A. (2020). Land Sparing Can Maintain Bird Diversity in Northeastern Bangladesh. Sustainability, 12(16), 6472. https://doi.org/10.3390/su12166472









