Regulation of Microclimatic Conditions inside Native Beehives and Its Relationship with Climate in Southern Spain
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Type and Study Units
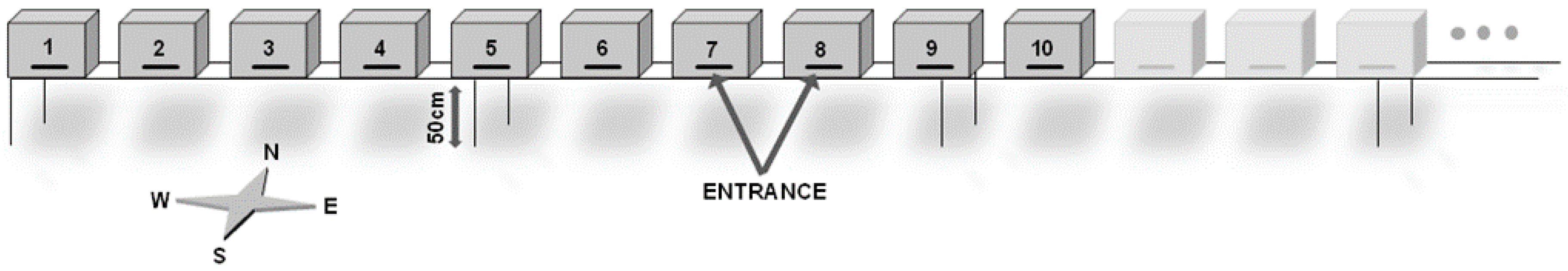
2.2. Hive Type, Location and Spatial Configuration
2.3. Colony Creation, Maintenance and Monitoring
2.4. Data Collection Computerized System
2.5. Statistical Analysis
2.5.1. Within Hive Temperature, Humidity and Weight Benchmark Values
2.5.2. Effects of Conditioning Factors on Hive Temperature, Humidity and Weight Evolution
2.5.3. Model Complexity Dimensionality Reduction and Predictive Regression Models
3. Results
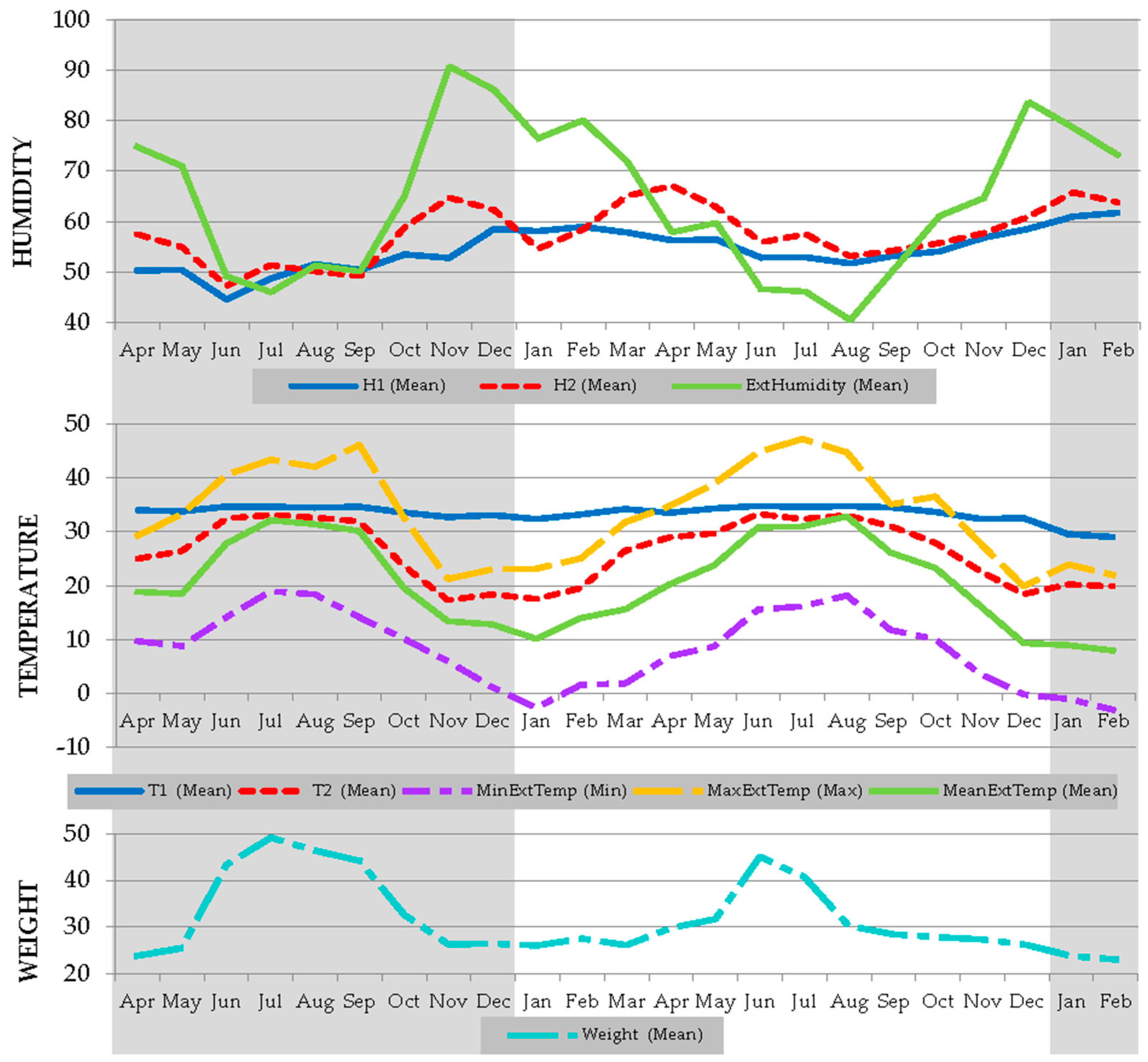
3.1. Within Hive Temperature, Humidity and Weight Benchmark Values for Iberian Honeybees
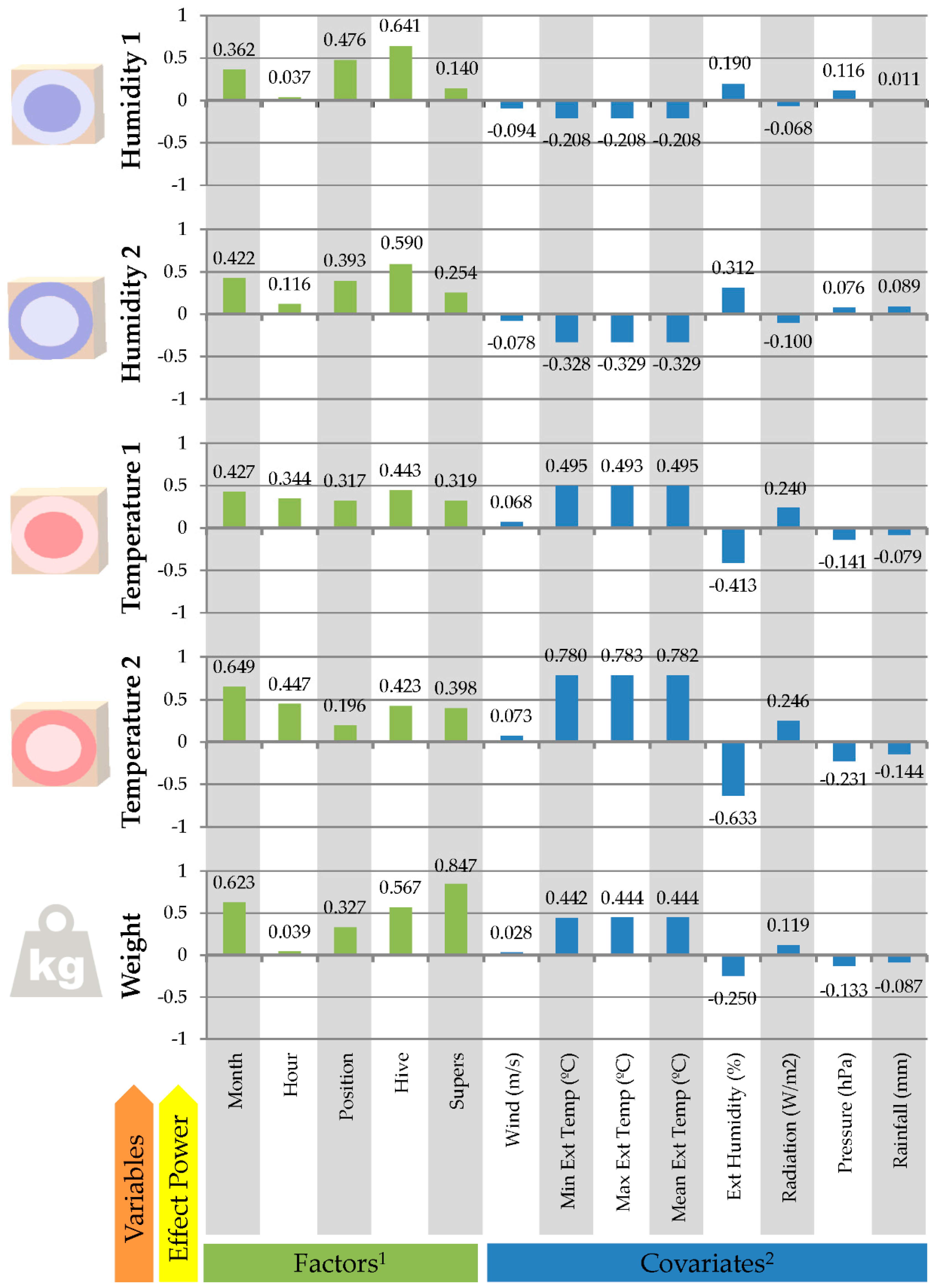
3.2. Effects of Conditioning Factors on Hive Temperature, Humidity and Weight Evolution
3.3. Model Complexity Dimensionality Reduction
3.4. Predictive Regression Models
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Huber, F. New Observations on the Natural History of Bees, 1st ed.; Printed for J. Anderson; Longman, Hurst, Rees, and Orme: London, UK, 1806; pp. 9–300. [Google Scholar]
- Oertel, E. Relative humidity and temperature within the beehive. J. Econ. Entomol. 1949, 42, 528–531. [Google Scholar] [CrossRef]
- Dunham, W.E. The influence of external temperature on the hive temperatures during the summer. J. Econ. Entomol. 1929, 22, 798–801. [Google Scholar] [CrossRef]
- Atmowidjojo, A.H.; Wheeler, D.E.; Erickson, E.H.; Cohen, A.C. Temperature tolerance and water balance in feral and domestic honey bees, Apis mellifera L. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 1997, 118, 1399–1403. [Google Scholar] [CrossRef]
- Ohashi, M.; Okada, R.; Kimura, T.; Ikeno, H. Observation system for the control of the hive environment by the honeybee (Apis mellifera). Behav. Res. Methods 2009, 41, 782–786. [Google Scholar] [CrossRef] [PubMed]
- Altun, A.A. Remote Control of the Temperature-Humidity and Climate in the Beehives with Solar-Powered Thermoelectric System. Control Eng. Appl. Inform. 2012, 14, 93–99. [Google Scholar]
- Edwards-Murphy, F.; Magno, M.; Whelan, P.M.; O’Halloran, J.; Popovici, E.M. b plus WSN: Smart beehive with preliminary decision tree analysis for agriculture and honey bee health monitoring. Comput. Electron. Agric. 2016, 124, 211–219. [Google Scholar] [CrossRef]
- Gil-Lebrero, S.; Quiles-Latorre, F.J.; Ortiz-Lopez, M.; Sanchez-Ruiz, V.; Gamiz-Lopez, V.; Luna-Rodriguez, J.J. Honey Bee Colonies Remote Monitoring System. Sensors 2017, 17, 55. [Google Scholar] [CrossRef]
- Zacepins, A.; Meitalovs, J.; Stalidzans, E. Temperature Control System for Risk Minimization in Honey Bee Wintering Building. In Proceedings of the 4th International Scientific Conference on Applied Information and Communication Technologies, Jelgava, Latvia, 22–23 April 2010; p. 309. [Google Scholar]
- Abou-Shaara, H.F.; Al-Ghamdi, A.A.; Mohamed, A.A. Honey Bee Colonies Performance Enhance By Newly Modified Beehives. J. Apic. Sci. 2013, 57, 45–57. [Google Scholar] [CrossRef]
- Zacepins, A. Precision Computer Control of the Biosystem in Closed Environment. Balt. J. Mod. Comput. 2013, 1, 131–138. [Google Scholar]
- Kviesis, A.; Zacepins, A. System Architectures for Real-time Bee Colony Temperature Monitoring. In Proceedings of the ICTE in Regional Development, Valmiera, Latvia, December 2014; pp. 86–94. [Google Scholar]
- Kviesis, A.; Zacepins, A.; Riders, G. Honey bee colony monitoring with implemented decision support system. In Proceedings of the 14th International Scientific Conference: Engineering for Rural Development, Jelgava, Latvia, 20–22 May 2015; pp. 446–451. [Google Scholar]
- Kviesis, A.; Zacepins, A. Application of Neural Networks for Honey Bee Colony State Identification. In Proceedings of the 2016 17th International Carpathian Control Conference (ICCC), Tatranská Lomnika, Slovakia, 29 May–1 June 2016; pp. 413–417. [Google Scholar]
- Mitchell, D. Ratios of colony mass to thermal conductance of tree and man-made nest enclosures of Apis mellifera: Implications for survival, clustering, humidity regulation and Varroa destructor. Int. J. Biometeorol. 2016, 60, 629–638. [Google Scholar] [CrossRef]
- Stalidzans, E.; Zacepins, A.; Kviesis, A.; Brusbardis, V.; Meitalovs, J.; Paura, L.; Bulipopa, N.; Liepniece, M. Dynamics of Weight Change and Temperature of Apis mellifera (Hymenoptera: Apidae) Colonies in a Wintering Building With Controlled Temperature. J. Econ. Entomol. 2017, 110, 13–23. [Google Scholar] [PubMed]
- Ministerio de Agricultura, Pesca y Alimentación. El sector apícola en números: Principales indicadores económicos. Government of Spain. Available online: https://www.mapa.gob.es/es/ganaderia/estadisticas/indicadoreseconomicossectordelamiel2018comentarios_tcm30-419675.pdf (accessed on 22 December 2019).
- Chavez-Galarza, J.; Henriques, D.; Johnston, J.S.; Azevedo, J.C.; Patton, J.C.; Munoz, I.; De La Rua, P.; Pinto, M.A. Signatures of selection in the Iberian honey bee (Apis mellifera iberiensis) revealed by a genome scan analysis of single nucleotide polymorphisms. Mol. Ecol. 2013, 22, 5890–5907. [Google Scholar] [CrossRef] [PubMed]
- Hatjina, F.; Costa, C.; Buchler, R.; Uzunov, A.; Drazic, M.; Filipi, J.; Charistos, L.; Ruottinen, L.; Andonov, S.; Meixner, M.D.; et al. Population dynamics of European honey bee genotypes under different environmental conditions. J. Apic. Res. 2014, 53, 233–247. [Google Scholar] [CrossRef]
- Sanchez, V.; Gil, S.; Flores, J.M.; Quiles, F.J.; Ortiz, M.A.; Luna, J.J. Implementation of an electronic system to monitor the thermoregulatory capacity of honeybee colonies in hives with open-screened bottom boards. Comput. Electron. Agric. 2015, 119, 209–216. [Google Scholar] [CrossRef]
- REDIAM. El Clima de Andalucía en el siglo XXI: Escenarios Locales de Cambio Climático de Andalucía. Resultados. Consejería de Medio Ambiente y Ordenación del Territorio de la Junta de Andalucía. Available online: http://www.juntadeandalucia.es/medioambiente/portal_web/web/temas_ambientales/clima/actuaciones_cambio_climatico/adaptacion/escenarios/elaboracion_escenarios/clima.pdf (accessed on 22 December 2019).
- Cramer, W.; Guiot, J.; Fader, M.; Garrabou, J.; Gattuso, J.P.; Iglesias, A.; Lange, M.A.; Lionello, P.; Llasat, M.C.; Paz, S.; et al. Climate change and interconnected risks to sustainable development in the Mediterranean. Nat. Clim. Chang. 2018, 8, 972–980. [Google Scholar] [CrossRef]
- Aemet, S.S.M.A. Weather Summary of 2017 in Spain. Available online: http://www.aemet.es/es/noticias/2018/01/Resumen_climatico_2017 (accessed on 9 June 2019).
- Hernando, M.D.; Gamiz, V.; Gil-Lebrero, S.; Rodriguez, I.; Garcia-Valcarcel, A.I.; Cutillas, V.; Fernandez-Alba, A.R.; Flores, J.M. Viability of honeybee colonies exposed to sunflowers grown from seeds treated with the neonicotinoids thiamethoxam and clothianidin. Chemosphere 2018, 202, 609–617. [Google Scholar] [CrossRef]
- Flores, J.M.; Gil-Lebrero, S.; Gamiz, V.; Rodriguez, M.I.; Ortiz, M.A.; Quiles, F.J. Effect of the climate change on honey bee colonies in a temperate Mediterranean zone assessed through remote hive weight monitoring system in conjunction with exhaustive colonies assessment. Sci. Total Environ. 2019, 653, 1111–1119. [Google Scholar] [CrossRef]
- Mann, C. Observational research methods. Research design II: Cohort, cross sectional, and case-control studies. Emerg. Med. J. 2003, 20, 54–60. [Google Scholar] [CrossRef]
- Aggarwal, R.; Ranganathan, P. Study designs: Part 2–Descriptive studies. Perspect. Clin. Res. 2019, 10, 34. [Google Scholar] [CrossRef]
- Jean-Prost, P.; Medori, P. Apicultura: Conocimiento de la Abeja, Manejo de la Colmena, 7th ed.; Mundi-Prensa: Madrid, Spain, 1981; pp. 551–553. [Google Scholar]
- Mann, C.J. Observational research methods—Cohort studies, cross sectional studies, and case–control studies. Afr. J. Emerg. Med. 2012, 2, 38–46. [Google Scholar] [CrossRef]
- Laerd Statistics, L. Friedman Test in SPSS Statistics; Lund Research Ltd.: Nottingham, UK, 2013; Volume 1. [Google Scholar]
- Theodorsson-Norheim, E. Friedman and Quade tests: BASIC computer program to perform nonparametric two-way analysis of variance and multiple comparisons on ranks of several related samples. Compit. Biol. Med. 1987, 17, 85–99. [Google Scholar] [CrossRef]
- IBM Corp. IBM SPSS Statistics for Windows, 25.0; IBM Corp: Armonk, NY, USA, 2017. [Google Scholar]
- Aguinis, H.; Beaty, J.C.; Boik, R.J.; Pierce, C.A. Effect size and power in assessing moderating effects of categorical variables using multiple regression: A 30-year review. J. Appl. Psychol. 2005, 90, 94. [Google Scholar] [CrossRef] [PubMed]
- Murphy, K.R.; Myors, B.; Wolach, A. Statistical Power Analysis: A Simple and General Model for Traditional and Modern Hypothesis, 4th ed.; Routledge: New York, NY, USA, 2014; pp. 1–156. [Google Scholar]
- Fritz, C.O.; Morris, P.E.; Richler, J.J. Effect size estimates: Current use, calculations, and interpretation. J. Exp. Psychol. Gen. 2012, 141, 2. [Google Scholar] [CrossRef] [PubMed]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences, 2nd ed.; Lawrence Earlbaum Associates. Inc.: New York, NY, USA, 1988; pp. 1–552. [Google Scholar]
- Coolican, H. Research Methods and Statistics in Psychology, 4th ed.; Routledge, Ed.; Hodder Education: New York, NY, USA, 2009; pp. 18–595. [Google Scholar]
- Profillidis, V.A.; Botzoris, G.N. Chapter 5—Statistical Methods for Transport Demand Modeling. In Modeling of Transport Demand; Profillidis, V.A., Botzoris, G.N., Eds.; Elsevier: Amsterdam, The Netherlands, 2019. [Google Scholar]
- Çamdevýren, H.; Demýr, N.; Kanik, A.; Keskýn, S. Use of principal component scores in multiple linear regression models for prediction of Chlorophyll-a in reservoirs. Ecol. Mod. 2005, 181, 581–589. [Google Scholar] [CrossRef]
- Ting Lee, M.L. Analysis of Microarray Gene Expression Data; Springer: New York, NY, USA, 2007; pp. 45–64. [Google Scholar]
- Welch, S.; Comer, J. Quantitative Methods for Public Administration: Techniques and Applications; Houghton Mifflin Harcourt P: Boston, MA, USA, 1988; pp. 1–362. [Google Scholar]
- Hagell, P. Testing rating scale unidimensionality using the principal component analysis (PCA)/t-test protocol with the Rasch model: The primacy of theory over statistics. Open J. Stat. 2014, 4, 456–465. [Google Scholar] [CrossRef]
- George, D.; Mallery, P. Reliability analysis. In SPSS for Windows, Step by Step: A Simple Guide and Reference, 14th ed.; Allyn & Bacon: Boston, MA, USA, 2003; pp. 222–232. [Google Scholar]
- Nunnally, J.C.; Bernstein, I. Psychometric Theory, 3rd ed.; McGraw-Hill Education: New York, NY, USA, 1994. [Google Scholar]
- Brborović, H.; Šklebar, I.; Brborović, O.; Brumen, V.; Mustajbegović, J. Development of a Croatian version of the US Hospital Survey on Patient Safety Culture questionnaire: Dimensionality and psychometric properties. Postgrad. Méd. J. 2014, 90, 125–132. [Google Scholar] [CrossRef]
- Salkind, N.J. Encyclopedia of Research Design; SAGE Publishing: Thousand Oaks, CA, USA, 2010. [Google Scholar]
- Jollife, I.T. Principal Component Analysis; Springer: Secaucus, NJ, USA, 2002. [Google Scholar]
- King, J.R.; Jackson, D.A. Variable selection in large environmental data sets using principal components analysis. Environmetrics 1999, 10, 67–77. [Google Scholar] [CrossRef]
- Al-Kandari, N.M.; Jolliffe, I.T. Variable selection and interpretation of covariance principal components. Commun. Stat. Simul. Comput. 2001, 30, 339–354. [Google Scholar] [CrossRef]
- Jackson, J.E. A User’s Guide to Principal Components; John Wiley & Sons: Hoboken, NJ, USA, 2005; Volume 587. [Google Scholar]
- Ul-Saufie, A.; Yahya, A.; Ramli, N. Improving multiple linear regression model using principal component analysis for predicting PM10 concentration in Seberang Prai, Pulau Pinang. Int. J. Environ. Sci. 2011, 2, 403–410. [Google Scholar]
- Zhang, T.; Yang, B. Big data dimension reduction using PCA. In Proceedings of the 2016 IEEE International Conference on Smart Cloud (SmartCloud), New York, NY, USA, 18–20 November 2016; pp. 152–157. [Google Scholar]
- Derksen, S.; Keselman, H.J. Backward, forward and stepwise automated subset selection algorithms: Frequency of obtaining authentic and noise variables. Br. J. Math. Stat. Psychol. 1992, 45, 265–282. [Google Scholar] [CrossRef]
- Farkas, O.; Héberger, K. Comparison of ridge regression, partial least-squares, pairwise correlation, forward-and best subset selection methods for prediction of retention indices for aliphatic alcohols. J. Chem. Inf. Model. 2005, 45, 339–346. [Google Scholar] [CrossRef]
- Jiang, W.; Simon, R. A comparison of bootstrap methods and an adjusted bootstrap approach for estimating the prediction error in microarray classification. Stat. Med. 2007, 26, 5320–5334. [Google Scholar] [CrossRef] [PubMed]
- Hoaglin, D.C.; Iglewicz, B. Fine-tuning some resistant rules for outlier labeling. J. Am. Stat. Assoc. 1987, 82, 1147–1149. [Google Scholar] [CrossRef]
- Williams, R. Multicollinearity. University of Notre Dame. 2015. Available online: https://www.nd.edu/~rwilliam/stats2/l11.pdf (accessed on 12 August 2015).
- O’brien, R.M. A caution regarding rules of thumb for variance inflation factors. Qual. Quant. 2007, 41, 673–690. [Google Scholar] [CrossRef]
- Tavakol, M.; Dennick, R. Making sense of Cronbach’s alpha. Int. J. Med. Educ. 2011, 2, 53. [Google Scholar] [CrossRef]
- Field, A. Discovering Statistics; Sage Publications: London, UK, 2011. [Google Scholar]
- Human, H.; Nicolson, S.W.; Dietemann, V. Do honeybees, Apis mellifera scutellata, regulate humidity in their nest? Naturwissenschaften 2006, 93, 397–401. [Google Scholar] [CrossRef] [PubMed]
- Ellis, M.B.; Nicolson, S.W.; Crewe, R.M.; Dietemann, V. Hygropreference and brood care in the honeybee (Apis mellifera). J. Insect Physiol. 2008, 54, 1516–1521. [Google Scholar] [CrossRef] [PubMed]
- Doull, K.M. The effects of different humidities on the hatching of the eggs of honeybees. Apidologie 1976, 7, 61–66. [Google Scholar] [CrossRef]
- Southwick, E.E.; Heldmaier, G. Temperature control in honey-bee colonies. Bioscience 1987, 37, 395–399. [Google Scholar] [CrossRef]
- Kleinhenz, M.; Bujok, B.; Fuchs, S.; Tautz, J. Hot bees in empty broodnest cells: Heating from within. J. Exp. Biol. 2003, 206, 4217–4231. [Google Scholar] [CrossRef]
- Medrzycki, P.; Sgolastra, F.; Bortolotti, L.; Bogo, G.; Tosi, S.; Padovani, E.; Porrini, C.; Sabatini, A.G. Influence of brood rearing temperature on honey bee development and susceptibility to poisoning by pesticides. J. Apic. Res. 2010, 49, 52–59. [Google Scholar] [CrossRef]
- Switanek, M.; Crailsheim, K.; Truhetz, H.; Brodschneider, R. Modelling seasonal effects of temperature and precipitation on honey bee winter mortality in a temperate climate. Sci. Total Environ. 2017, 579, 1581–1587. [Google Scholar] [CrossRef] [PubMed]
- Erdős, N. Report on Prospects and Challenges for the EU Apiculture Sector (2017/2115(INI)); European Parliament: Brussels, Belgium, 2018. [Google Scholar]
- Traynor, K.S.; Mondet, F.; de Miranda, J.R.; Techer, M.; Kowallik, V.; Oddie, M.A.Y.; Chantawannakul, P.; McAfee, A. Varroa destructor: A Complex Parasite, Crippling Honey Bees Worldwide. Trends Parasitol. 2020, 36, 592–606. [Google Scholar] [CrossRef] [PubMed]
- Brodschneider, R.; Gray, A.; Adjlane, N.; Ballis, A.; Brusbardis, V.; Charrière, J.D.; Chlebo, R.; Coffey, M.F.; Dahle, B.; de Graaf, D.C. Multi-country loss rates of honey bee colonies during winter 2016/2017 from the COLOSS survey. J. Apic. Res. 2018, 57, 452–457. [Google Scholar] [CrossRef]
- Ministerio para la Transición Ecológica Agencia Estatal de Meteorología. AEMET Informe Anual 2018; Agencia Estatal de Meteorología: Madrid, Spain, 2019; p. 72. [Google Scholar]
- Mardan, M.; Kevan, P.G. Critical temperatures for survival of brood and adult workers of the giant honeybee, Apis dorsata (Hymenoptera: Apidae). Apidologie 2002, 33, 295–301. [Google Scholar] [CrossRef]
- Bastiaansen, R.; Doelman, A.; Van Langevelde, F.; Rottschafer, V. Modeling Honey Bee Colonies in Winter Using a Keller--Segel Model With a Sign-Changing Chemotactic Coefficient. SIAM J. Appl. Math. 2020, 80, 839–863. [Google Scholar] [CrossRef]
- Jones, J.C.; Myerscough, M.R.; Graham, S.; Oldroyd, B.P. Honey bee nest thermoregulation: Diversity promotes stability. Science 2004, 305, 402–404. [Google Scholar] [CrossRef]
- Chacon-Almeida, V.M.L.; Simões, Z.L.P.; Bitondi, M.M.G. Induction of heat shock proteins in the larval fat body of Apis mellifera L. bees. Apidologie 2000, 31, 487–501. [Google Scholar] [CrossRef]
- Severson, D.; Erickson, E.; Williamson, J.; Aiken, J. Heat stress induced enhancement of heat shock protein gene activity in the honey bee (Apis mellifera). Experientia 1990, 46, 737–739. [Google Scholar] [CrossRef]
- Abou-Shaara, H.; Al-Ghamdi, A. Studies on wings symmetry and honey bee races discrimination by using standard and geometric morphometrics. J. Anim. Sci. Biotechnol. 2012, 28, 575–584. [Google Scholar] [CrossRef]
- Adam, B. In search of the best strains of bee third journey: The iberian peninsula. Bee World 1961, 42, 123–131. [Google Scholar] [CrossRef]
- Milner, A. An Introduction to Understanding Honeybees, Their Origins, Evolution and Diversity. Available online: https://bibba.com/honeybee-origins/ (accessed on 22 December 2019).
- Abou-Shaara, H.F.; Owayss, A.A.; Ibrahim, Y.Y.; Basuny, N.K. A review of impacts of temperature and relative humidity on various activities of honey bees. Insectes Soc. 2017, 64, 455–463. [Google Scholar] [CrossRef]
- EFSA. Guidance on the risk assessment of plant protection products on bees (Apis mellifera, Bombus spp. and solitary bees). EFSA J. 2013, 11, 3295. [Google Scholar]
- EPPO. Efficacy evaluation of plant protection products. In Evaluation Biologique des Produits Phytosanitaires. Side-Effects on Honeybees; OEPP/EPPO, Ed.; Bulletin OEPP/EPPO, Bulletin 40; EPPO: Paris, France, 2010; pp. 313–319. [Google Scholar]
- Salamanca, G.; Osorio, T.; Rodríguez, A. Phoretic presence and incidence of Varroa destructor A. (Mesostigmata: Varroidae) in honey bee colonies of Apis mellifera (Hymenptera: Apidae), in Colombia. Zootec. Trop. 2012, 30, 183–195. [Google Scholar]
- Gonell, F.; Gómez-Pajuelo, A.; Bota, G.; Giralt, D.; Sardá, F. Study of the impact of the bee-eater, Merops apiaster, in beehives, in Lleida, 2015. In Proceedings of the VIII National Congress of Apiculture, Granada, Spain, 3–5 November 2016; pp. 35–36. [Google Scholar]
- Stanimirovic, Z.; Glavinic, U.; Ristanic, M.; Aleksic, N.; Jovanovic, N.; Vejnovic, B.; Stevanovic, J. Looking for the causes of and solutions to the issue of honey bee colony losses. Acta Vet. 2019, 69, 1–31. [Google Scholar] [CrossRef]
- Becher, M.A.; Grimm, V.; Thorbek, P.; Horn, J.; Kennedy, P.J.; Osborne, J.L. BEEHAVE: A systems model of honeybee colony dynamics and foraging to explore multifactorial causes of colony failure. J. Appl. Ecol. 2014, 51, 470–482. [Google Scholar] [CrossRef]
- Humphreys, A.M.; Govaerts, R.; Ficinski, S.Z.; Nic Lughadha, E.; Vorontsova, M.S. Global dataset shows geography and life form predict modern plant extinction and rediscovery. Nat. Ecol. Evol. 2019, 3, 1043–1047. [Google Scholar] [CrossRef]
- Bermig, S.; Odemer, R.; Gombert, A.J.; Frommberger, M.; Rosenquist, R.; Pistorius, J. Experimental validation of an electronic counting device to determine flight activity of honey bees (Apis mellifera L.). J. Kult. 2020, 72, 132–140. [Google Scholar]
- Colin, T.; Bruce, J.; Meikle, W.G.; Barron, A.B. The development of honey bee colonies assessed using a new semi-automated brood counting method: CombCount. PLoS ONE 2018, 13, e0205816. [Google Scholar] [CrossRef]
| Factor | Level | Test | Effect Size |
|---|---|---|---|
| Year | 2016–2017 | Mann–Whitney U | Rank biserial correlation (r) |
| Month | January to December | Kruskal–Wallis H | Cohen’s f |
| Hour | 24 h | Kruskal–Wallis H | Cohen’s f |
| Position | 1 to 10 | Kruskal–Wallis H | Cohen’s f |
| Colony | A–Y (25 colonies) | Kruskal–Wallis H | Cohen’s f |
| Supers | 0, 1 and 2 supers | Kruskal–Wallis H | Cohen’s f |
| Wind (m/s) | 0–12.4 | Pearson correlation analysis | Pearson’s r |
| Min Ext Temp (°C) | −2.6–46.4 | Pearson correlation analysis | Pearson’s r |
| Max Ext Temp (°C) | −0.8–47.3 | Pearson correlation analysis | Pearson’s r |
| Mean Ext Temp (°C) | −1.8–46.8 | Pearson correlation analysis | Pearson’s r |
| Ext Humidity (%) | 16–100 | Pearson correlation analysis | Pearson’s r |
| Radiation (W/m2) | 0–323 | Pearson correlation analysis | Pearson’s r |
| Pressure (hPa) | 613–1033 | Pearson correlation analysis | Pearson’s r |
| Rainfall (mm) | 0–11.5 | Pearson correlation analysis | Pearson’s r |
| Principal Components, Blocks or Subsets | Cronbach’s Alpha | Eigenvalues |
|---|---|---|
| PC1 | 0.870 | 5.193 |
| PC2 | 0.634 | 2.434 |
| PC3 | 0.449 | 1.715 |
| PC4 | 0.263 | 1.323 |
| PC5 | −0.158 | 0.872 |
| Total | 0.984 | 11.537 |
| Dimension | Principal Components, Blocks or Subsets | |||||
|---|---|---|---|---|---|---|
| Factor | 1 | 2 | 3 | 4 | 5 | |
| Year | −0.325 | −0.027 | 0.595 | −0.319 | −0.445 | |
| Month | 0.489 | 0.120 | −0.655 1 | −0.025 | 0.133 | |
| Hour | 0.620 1 | −0.068 | 0.568 | −0.001 | 0.139 | |
| Position | 0.128 | 0.988 1 | −0.032 | 0.015 | 0.054 | |
| Colony | 0.120 | 1.002 1 | −0.011 | 0.005 | 0.036 | |
| Supers | 0.148 | −0.652 1 | −0.331 | 0.068 | 0.146 | |
| Wind (m/s) | 0.439 | −0.061 | 0.515 | 0.261 | 0.271 | |
| Min Ext Temp (°C) | 0.992 1 | −0.028 | −0.107 | −0.009 | −0.086 | |
| Max Ext Temp (°C) | 0.996 1 | −0.028 | −0.106 | −0.024 | −0.091 | |
| Mean Ext Temp (°C) | 0.997 1 | −0.029 | −0.106 | −0.016 | −0.092 | |
| Ext Humidity (%) | −0.901 1 | 0.047 | −0.165 | 0.169 | 0.143 | |
| Radiation (W/m2) | 0.552 | −0.036 | 0.371 | −0.125 | 0.314 | |
| Pressure (hPa) | −0.297 | −0.009 | 0.021 | −0.721 1 | 0.570 | |
| Rainfall (mm) | −0.211 | 0.019 | 0.191 | 0.764 1 | 0.263 | |
| Pairwise Comparisons | Test Statistic | Degree of Freedom | Asymptotic p-Values (2-Sided Test) |
|---|---|---|---|
| Mean Ext Temp, Temperature 2 | 121,936.97 | 1 | 0.001 |
| Mean Ext Temp, Temperature 1 | 227,059.06 | 1 | 0.001 |
| Temperature 1, Temperature 2 | 351,630.06 | 1 | 0.001 |
| Ext Humidity, Humidity 2 | 68.43 | 0.001 | |
| Ext Humidity, Humidity 1 | 1399.96 | 1 | 0.001 |
| Humidity 1, Humidity 2 | 21,455.82 | 1 | 0.001 |
| General Model Regression Equation | Legend | ||
|---|---|---|---|
| Z’yh1h2t1t2w = βmonth * Zmonth + βhour * Zhour + βposition * Zposition + βcolony * Zcolony + βsupers * Zsupers + βminexttemp * Zminexttemp + βmaxexttemp * Zmaxexttemp + βmeanexttemp * Zmeanexttemp + βexthumidity * Zexthumidity + βpressure * Zpressure + βrainfall * Zrainfall | Z’yh1h2t1t2w = Z score for each variable (humidity 1, humidity 3, temperature 1, temperature 3, weight). | ||
| β = standardized coefficient for each of the factors appearing in the subindex. | |||
| Z = Z scores for each of the factors appearing in the subindex. | |||
| Specific Regression Equations | Legend | ||
| Humidity 1 (%). Brood nest | Z’yh 1 = 0.214 (Zmonth) − 0.021(Zhour) + 0.560 (Zposition) + 0.289(Zcolony) + 0.053(Zsupers) + 0.028(Zminexttemp) + 0.110(Zmaxexttemp) + 0.039(Zmeanexttemp) + 0.192(Zexthumidity) − 22120.007(Zpressure) − 0.007(Zrainfall) | Z’yh1 = Z score for Humidity 1 variable. | |
| βmonthZmonth = 0.214(Zmonth) | |||
| βhourZhour = −0.021 (Zhour) | |||
| βpositionZposition = 0.560 (Zposition) | |||
| βcolonyZcolony = 0.289 (Zcolony) | |||
| βsupersZsupers = 0.053 (Zsupers) | |||
| βminexttempZminexttemp = 0.028 (Zminexttemp) | |||
| βmaxexttempZmaxexttemp = 0.110 (Zmaxexttemp) | |||
| βmeanexttempZmeanexttemp = 0.039 (Zmeanexttemp) | |||
| βexthumidityZexthumidity = 0.192 (Zexthumidity) | |||
| βpressureZpressure = −0.007 (Zpressure) | |||
| βrainfallZrainfall = −0.007 (Zrainfall) | |||
| R Square | Adjusted R Square | p-Value | |
| 0.470 | 0.470 | 0.001 | |
| Humidity 2 (%), food area | Z’yh2 = 0.285(Zmonth) − 0.026(Zhour) + 0.568(Zposition) + 0.331(Zcolony) + 0.571(Zsupers) + 0.015(Zminexttemp) + 0.061(Zmaxexttemp) + 0.028(Zmeanexttemp) + 0.170 (Zexthumidity) + 0.024(Zpressure) + 0.005(Zrainfall) | Z’yh2 = Z score for Humidity 2 variable. | |
| βmonthZmonth = 0.285 (Zmonth) | |||
| βhourZhour = −0.026 (Zhour) | |||
| βpositionZposition = 0.568 (Zposition) | |||
| βcolonyZcolony = 0.331 (Zcolony) | |||
| βsupersZsupers = 0.571 (Zsupers) | |||
| βminexttempZminexttemp = 0.015 (Zminexttemp) | |||
| βmaxexttempZmaxexttemp = 0.061 (Zmaxexttemp) | |||
| βmeanexttempZmeanexttemp = 0.028 (Zmeanexttemp) | |||
| βexthumidityZexthumidity = 0.170 (Zexthumidity) | |||
| βpressureZpressure = 0.024 (Zpressure) | |||
| βrainfallZrainfall = 0.005 (Zrainfall) | |||
| R Square | Adjusted R Square | p-Value | |
| 0.480 | 0.480 | 0.001 | |
| Temperature 1 (ºC), brood nest | Z’yt1 = −0.289(Zmonth) + 0.121(Zhour) + 0.436(Zposition) + 0.178(Zcolony) + 0.170(Zsupers) + 0.156(Zminexttemp) + 0.180 (Zmaxexttemp) – 0.135(Zmeanexttemp) − 0.025 (Zexthumidity) + 0.007(Zpressure) − 0.014(Zrainfall) | Z’yt1 = Z score for Temperature 1 variable. | |
| βmonthZmonth = −0.289 (Zmonth) | |||
| βhourZhour = 0.121 (Zhour) | |||
| βpositionZposition = 0.436 (Zposition) | |||
| βcolonyZcolony = 0.178 (Zcolony) | |||
| βsupersZsupers = 0.170 (Zsupers) | |||
| βminexttempZminexttemp = 0.156 (Zminexttemp) | |||
| βmaxexttempZmaxexttemp = 0.180 (Zmaxexttemp) | |||
| βmeanexttempZmeanexttemp = −0.135 (Zmeanexttemp) | |||
| βexthumidityZexthumidity = −0.025 (Zexthumidity) | |||
| βpressureZpressure = 0.007 (Zpressure) | |||
| βrainfallZrainfall = −0.014 (Zrainfall) | |||
| R Square | Adjusted R Square | p-Value | |
| 0.358 | 0.358 | 0.001 | |
| Temperature 2 (ºC), food area | Z’yt2 = −0.224(Zmonth) + 0.280(Zhour) + 0.206(Zposition) + 0.057(Zcolony) + 0.044(Zsupers) + 0.358(Zminexttemp) − 0.024 (Zexthumidity) − 0.033(Zpressure) − 0.037(Zrainfall) | Z’yt2 = Z score for Temperature 2 variable. | |
| βmonthZmonth = −0.224 (Zmonth) | |||
| βhourZhour = 0.280 (Zhour) | |||
| βpositionZposition = 0.206 (Zposition) | |||
| βcolonyZcolony = 0.057 (Zcolony) | |||
| βsupersZsupers = 0.044 (Zsupers) | |||
| βminexttempZminexttemp = 0.358 (Zminexttemp) | |||
| βexthumidityZexthumidity = −0.024 (Zexthumidity) | |||
| βpressureZpressure = −0.033 (Zpressure) | |||
| βrainfallZrainfall = −0.037 (Zrainfall) | |||
| R Square | Adjusted R Square | p-Value | |
| 0.460 | 0.460 | 0.001 | |
| Weight (kg) | Z’yw = −0.302(Zmonth) − 0.030(Zhour) + 0.887(Zposition) + 0.349(Zcolony) + 0.963(Zsupers) + 0.032(Zmaxexttemp) − 0.005 (Zexthumidity) + 0.007(Zpressure) | Z’yw = Z score for Weight variable. | |
| βmonthZmonth = 0.285 (Zmonth) | |||
| βhourZhour = −0.026 (Zhour) | |||
| βpositionZposition = 0.568 (Zposition) | |||
| βcolonyZcolony = 0.331 (Zcolony) | |||
| βsupersZsupers = 0.571 (Zsupers) | |||
| βmaxexttempZmaxexttemp = 0.061 (Zmaxexttemp) | |||
| βexthumidityZexthumidity = 0.170 (Zexthumidity) | |||
| βpressureZpressure = 0.024 (Zpressure) | |||
| R Square | Adjusted R Square | p-Value | |
| 0.743 | 0.743 | 0.001 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gil-Lebrero, S.; Navas González, F.J.; Gámiz López, V.; Quiles Latorre, F.J.; Flores Serrano, J.M. Regulation of Microclimatic Conditions inside Native Beehives and Its Relationship with Climate in Southern Spain. Sustainability 2020, 12, 6431. https://doi.org/10.3390/su12166431
Gil-Lebrero S, Navas González FJ, Gámiz López V, Quiles Latorre FJ, Flores Serrano JM. Regulation of Microclimatic Conditions inside Native Beehives and Its Relationship with Climate in Southern Spain. Sustainability. 2020; 12(16):6431. https://doi.org/10.3390/su12166431
Chicago/Turabian StyleGil-Lebrero, Sergio, Francisco Javier Navas González, Victoria Gámiz López, Francisco Javier Quiles Latorre, and José Manuel Flores Serrano. 2020. "Regulation of Microclimatic Conditions inside Native Beehives and Its Relationship with Climate in Southern Spain" Sustainability 12, no. 16: 6431. https://doi.org/10.3390/su12166431
APA StyleGil-Lebrero, S., Navas González, F. J., Gámiz López, V., Quiles Latorre, F. J., & Flores Serrano, J. M. (2020). Regulation of Microclimatic Conditions inside Native Beehives and Its Relationship with Climate in Southern Spain. Sustainability, 12(16), 6431. https://doi.org/10.3390/su12166431





