How to Improve the Functional Capacity of Frail and Pre-Frail Elderly People? Health, Nutritional Status and Exercise Intervention. The EXERNET-Elder 3.0 Project
Abstract
1. Introduction
2. Materials and Methods
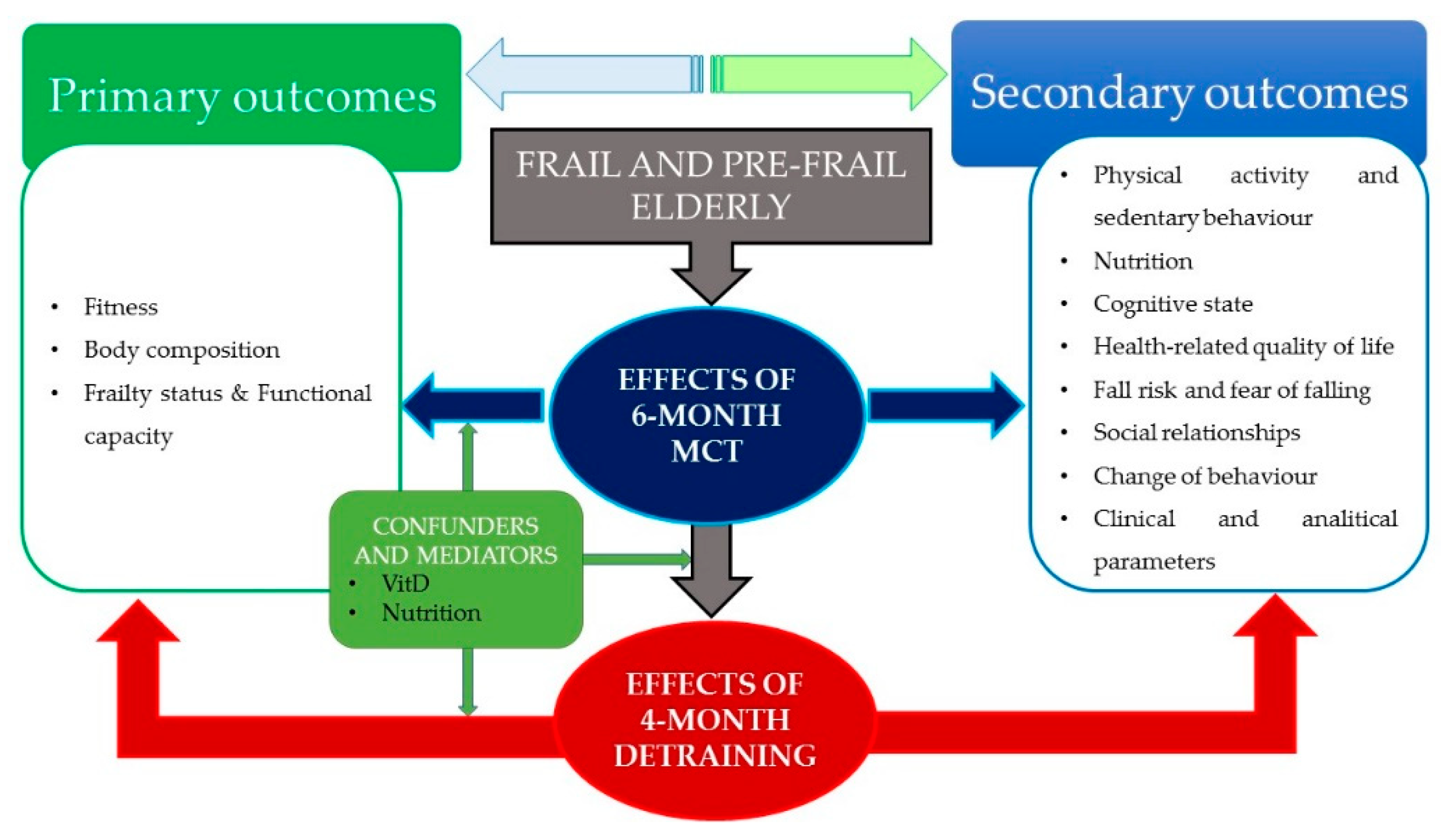
2.1. Conceptual Framework
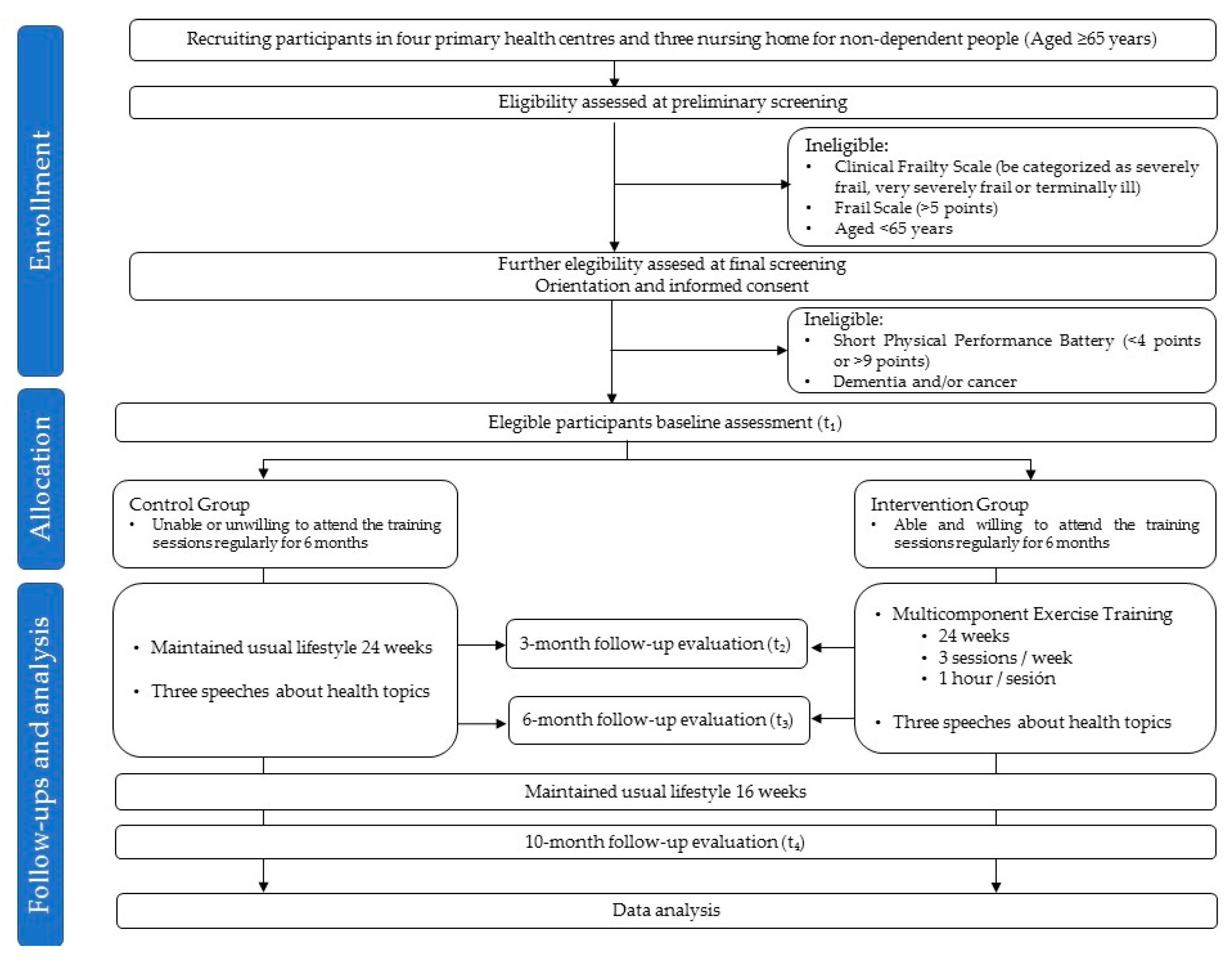
2.2. Study Design
2.3. Ethical Committee
2.4. Study Sample
2.4.1. Sample Size
2.4.2. Recruitment and Screening
2.5. Exercise Intervention
2.5.1. Elder-Fit Strength Training Protocol
Familiarization Period (Weeks 1–4)
Accumulation Period (Weeks 5–14)
Transformation Period (Weeks 15–21)
Functional Period (Weeks 22–24)
2.5.2. Elder-Fit Endurance Training Protocol
Familiarization Period (Weeks 1–4)
Development Period (Weeks 5–14)
Maintenance Period (Weeks 15–21)
2.6. Adherence and Motivation Strategies
2.7. Evaluation and Equipment
2.7.1. Fitness
Balance and Agility
Flexibility
Dynamic Strength
Maximum Isometric Strength
Aerobic Endurance
Walking Speed
2.7.2. Frailty Status & Functional Capacity
Frail Scale
Clinical Frail Scale (Adapted Version)
Frailty Phenotype of Fried
Short Physical Performance Battery (SPPB)
2.7.3. Body Composition and Anthropometric Measurements
Bone Mineral Density and Strength Indices
Height, Weight, Fat Mass and Muscle Mass
Anthropometrics
2.7.4. Physical Activity and Sedentary Behaviors
Accelerometry
Physical Activity Scale for the Elderly (PASE)
Elderly EXERNET Physical Activity Questionnaire (EEPAQ)
Adapted Version of Chasan–Tarber Physical Activity Questionnaire (CT-PAQ)
2.7.5. Nutrition
Mediterranean Diet Adherence Screener (MEDAS)
Mini Nutritional Assessment (MNA)
Food Frequency Questionnaire (FFQ)
2.7.6. Cognitive State
Mini-Mental State (MMS)
2.7.7. Health-Related Quality of Life
Euroqol-5D Questionnaire (EQ-5D)
Instrumental Activities of Daily Living Scale (IADL)
Barthel Index (BI)
2.7.8. Fall Risk and Fear of Falling
Falls Efficacy Scale I (FES-I)
Ad-hoc Risk of Fall and Fear Questionnaire
2.7.9. Change of Behavior Aspects
Modified Version of Transtheoretical Model of Physical Exercise Changes Questionnaire (TMPECQ)
2.7.10. Sociotype
Sociotype Questionnaire (SOCQ)
2.7.11. Clinical and Analytical Parameters
Blood Collection and Genomic, Proteomic and Biochemical Analysis
Blood Pressure (BP)
2.7.12. General Questionnaire
2.8. Data Collection Procedure
3. Discussion
Perspective
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Collard, R.M.; Boter, H.; Schoevers, R.A.; Oude Voshaar, R.C. Prevalence of frailty in community-dwelling older persons: A systematic review. J. Am. Geriatr. Soc. 2012, 60, 1487–1492. [Google Scholar] [CrossRef] [PubMed]
- Giné-Garriga, M.; Roqué-Fíguls, M.; Coll-Planas, L.; Sitjà-Rabert, M.; Salvà, A. Physical exercise interventions for improving performance-based measures of physical function in community-dwelling, frail older adults: A systematic review and meta-analysis. Arch. Phys. Med. Rehabil. 2014, 95, 753–769. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Cabello, A.; Vicente Rodríguez, G.; Vila-Maldonado, S.; Casajús, J.A.; Ara, I. Aging and body composition: The sarcopenic obesity in Spain. Nutr. Hosp. 2012, 27, 22–30. [Google Scholar] [CrossRef] [PubMed]
- Sternberg, S.A.; Schwartz, A.W.; Karunananthan, S.; Bergman, H.; Mark Clarfield, A. The identification of frailty: A systematic literature review. J. Am. Geriatr. Soc. 2011, 59, 2129–2138. [Google Scholar] [CrossRef] [PubMed]
- Bauer, J.M.; Kaiser, M.J.; Anthony, P.; Guigoz, Y.; Sieber, C.C. The mini nutritional assessment®-its history, today’s practice, and future perspectives. Nutr. Clin. Pract. 2008, 23, 388–396. [Google Scholar] [CrossRef]
- Goodpaster, B.H.; Krishnaswami, S.; Harris, T.B.; Katsiaras, A.; Kritchevsky, S.B.; Simonsick, E.M.; Nevitt, M.; Holvoet, P.; Newman, A.B. Obesity, regional body fat distribution, and the metabolic syndrome in older men and women. Arch. Intern. Med. 2005, 165, 777–783. [Google Scholar] [CrossRef] [PubMed]
- Alonso, A.; Martínez-González, M.Á. Olive oil consumption and reduced incidence of hypertension: The SUN study. Lipids 2004, 39, 1233–1238. [Google Scholar] [CrossRef] [PubMed]
- Bes-Rastrollo, M.; Sánchez-Villegas, A.; Gómez-Gracia, E.; Martínez, J.A.; Pajares, R.M.; Martínez-González, M.A. Predictors of weight gain in a Mediterranean cohort: The Seguimiento Universidad de Navarra Study. Am. J. Clin. Nutr. 2006, 83, 362–370. [Google Scholar] [CrossRef]
- Martínez-González, M.A.; Sánchez-Villegas, A.; De Irala, J.; Marti, A.; Martínez, J.A. Mediterranean diet and stroke: Objectives and design of the SUN project. Nutr. Neurosci. 2002, 5, 65–73. [Google Scholar] [CrossRef]
- Psaltopoulou, T.; Naska, A.; Orfanos, P.; Trichopoulos, D.; Mountokalakis, T.; Trichopoulou, A. Olive oil, the Mediterranean diet, and arterial blood pressure: The Greek European Prospective Investigation into Cancer and Nutrition (EPIC) study. Am. J. Clin. Nutr. 2004, 80, 1012–1018. [Google Scholar] [CrossRef]
- Rodríguez-Mañas, L.; Féart, C.; Mann, G.; Viña, J.; Chatterji, S.; Chodzko-Zajko, W.; Gonzalez-Colaço Harmand, M.; Bergman, H.; Carcaillon, L.; Nicholson, C.; et al. Searching for an operational definition of frailty: A delphi method based consensus statement. the frailty operative definition-consensus conference project. J. Gerontol 2013, 68, 62–67. [Google Scholar] [CrossRef] [PubMed]
- Gobbens, R.J.; Luijkx, K.G.; Wijnen-Sponselee, M.T.; Schols, J.M. Toward a conceptual definition of frail community dwelling older people. Nurs. Outlook 2010, 58, 76–86. [Google Scholar] [CrossRef] [PubMed]
- Robertson, D.A.; Savva, G.M.; Kenny, R.A. Frailty and cognitive impairment—A review of the evidence and causal mechanisms. Ageing Res. Rev. 2013, 12, 840–851. [Google Scholar] [CrossRef] [PubMed]
- Aniuliene, R.; Aniulis, P.; Steibliene, V. Risk factors and types of urinary incontinence among middle-aged and older male and female primary care patients in Kaunas region of Lithuania: Cross sectional study. Urol. J. 2016, 13, 2552–2561. [Google Scholar] [CrossRef] [PubMed]
- Felde, G.; Ebbesen, M.H.; Hunskaar, S. Anxiety and depression associated with urinary incontinence. A 10-year follow-up study from the Norwegian HUNT study (EPINCONT). Neurourol. Urodyn. 2017, 36, 322–328. [Google Scholar] [CrossRef]
- Crowley, K. Sleep and sleep disorders in older adults. Neuropsychol. Rev. 2011, 21, 41–53. [Google Scholar] [CrossRef]
- Wyskida, M.; Wieczorowska-Tobis, K.; Chudek, J. Prevalence and factors promoting the occurrence of vitamin D deficiency in the elderly. Postepy Hig. Med. Dosw. 2017, 71, 198–204. [Google Scholar] [CrossRef]
- Ferrucci, L.; Guralnik, J.M.; Studenski, S.; Fried, L.P.; Cutler, G.B.; Walston, J.D. Designing randomized, controlled trials aimed at preventing or delaying functional decline and disability in frail, older persons: A consensus report. J. Am. Geriatr. Soc. 2004, 52, 625–634. [Google Scholar] [CrossRef]
- Fried, L.P.; Tangen, C.M.; Walston, J.; Newman, A.B.; Hirsch, C.; Gottdiener, J.; Seeman, T.; Tracy, R.; Kop, W.J.; Burke, G.; et al. Frailty in older adults: Evidence for a phenotype. J. Gerontol. Ser. Biol. Sci. Med. Sci. 2001, 56, M146–M157. [Google Scholar] [CrossRef]
- Rodriguez-Mañas, L.; Fried, L.P. Frailty in the clinical scenario. Lancet 2015, 385, e7–e9. [Google Scholar] [CrossRef]
- Freiberger, E.; De vreede, P.; Schoene, D.; Rydwik, E.; Mueller, V.; Frändin, K.; Hopman-Rock, M. Performance-based physical function in older community-dwelling persons: A systematic review of instruments. Age Ageing 2012, 41, 712–721. [Google Scholar] [CrossRef] [PubMed]
- Scully, T. Demography: To the limit. Nature 2012, 492, S2–S3. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization United Nations Department of Economics and Social Affairs: Population Division. World Population Ageing: 1950–2050. Available online: http://www.un.org/%0Aesa/population/publications/worldageing19502050/. (accessed on 31 March 2020).
- Castell, M.V.; Gutiérrez-Misis, A.; Sánchez-Martínez, M.; Prieto, M.A.; Moreno, B.; Nuñez, S.; Triano, R.; De Antonio, M.P.; Mateo, C.; Cano, M.D.; et al. Effectiveness of an intervention in multicomponent exercise in primary care to improve frailty parameters in patients over 70 years of age (MEFAP-project), a randomised clinical trial: Rationale and study design. BMC Geriatr. 2019, 19, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Gates, S.; Fisher, J.D.; Cooke, M.W.; Carter, Y.H.; Lamb, S.E. Multifactorial assessment and targeted intervention for preventing falls and injuries among older people in community and emergency care settings: Systematic review and meta-analysis. BMJ 2008, 336, 130–133. [Google Scholar] [CrossRef] [PubMed]
- Izquierdo, M.; Casas-Herrero, A.; Martínez-Velilla, N.; Alonso-Bouzón, C.; Rodríguez-Mañas, L. An example of cooperation for implementing programs associated with the promotion of exercise in the frail elderly. European Erasmus + «Vivifrail» program. Rev. Esp. Geriatr. Gerontol. 2017, 52, 110–111. [Google Scholar] [CrossRef] [PubMed]
- Lutz, W.; Sanderson, W.; Scherbov, S. Doubling of world population unlikely. Nature 1997, 387, 803–805. [Google Scholar] [CrossRef]
- Bouaziz, W.; Lang, P.O.; Schmitt, E.; Kaltenbach, G.; Geny, B.; Vogel, T. Health benefits of multicomponent training programmes in seniors: A systematic review. Int. J. Clin. Pract. 2016, 70, 520–536. [Google Scholar] [CrossRef]
- Hildebrand, M.; Van Hees, V.T.; Hansen, B.H.; Ekelund, U. Age group comparability of raw accelerometer output from wrist-and hip-worn monitors. Med. Sci. Sports Exerc. 2014, 46, 1816–1824. [Google Scholar] [CrossRef]
- Physical Activity Guidelines Advisory Committee. Physical activity guidelines advisory committee report, 2008: To the Secretary of Health and Human Services. Nutr. Rev. 2009, 67, 114–120. [Google Scholar]
- Fiuza-Luces, C.; Garatachea, N.; Berger, N.A.; Lucia, A. Exercise is the real polypill. Physiology 2013, 28, 330–358. [Google Scholar] [CrossRef]
- Fragala, M.S.; Cadore, E.L.; Dorgo, S.; Izquierdo, M.; Kraemer, W.J.; Peterson, M.D.; Ryan, E.D. Resistance training for older adults: Position statement from the national strength and conditioning association. J. Strength Cond. Res. 2019, 33, 2019–2052. [Google Scholar] [CrossRef] [PubMed]
- Colley, R.C.; Garriguet, D.; Janssen, I.; Craig, C.L.; Clarke, J.; Tremblay, M.S. Physical activity of canadian children and youth: Accelerometer results from the 2007 to 2009 canadian health measures survey. Health Rep. 2011, 22. [Google Scholar]
- Sardinha, L.B.; Santos, D.A.; Silva, A.M.; Baptista, F.; Owen, N. Breaking-up sedentary time is associated with physical function in older adults. J. Gerontol. 2015, 70, 119–124. [Google Scholar] [CrossRef] [PubMed]
- Cadore, E.L.; Casas-Herrero, A.; Zambom-Ferraresi, F.; Idoate, F.; Millor, N.; Gómez, M.; Rodriguez-Mañas, L.; Izquierdo, M. Multicomponent exercises including muscle power training enhance muscle mass, power output, and functional outcomes in institutionalized frail nonagenarians. Age 2014, 36, 773–785. [Google Scholar] [CrossRef] [PubMed]
- Cadore, E.L.; Rodríguez-Mañas, L.; Sinclair, A.; Izquierdo, M. Effects of different exercise interventions on risk of falls, gait ability, and balance in physically frail older adults: A systematic review. Rejuvenation Res. 2013, 16, 105–114. [Google Scholar] [CrossRef]
- Esain, I.; Rodriguez-Larrad, A.; Bidaurrazaga-Letona, I.; Gil, S.M. Health-related quality of life, handgrip strength and falls during detraining in elderly habitual exercisers. Health Qual. Life Outcomes 2017, 15. [Google Scholar] [CrossRef]
- Rockwood, K.; Song, X.; MacKnight, C.; Bergman, H.; Hogan, D.B.; McDowell, I.; Mitnitski, A. A global clinical measure of fitness and frailty in elderly people. Can. Med. Assoc. J. 2005, 173, 489–495. [Google Scholar] [CrossRef]
- Slade, S.C.; Dionne, C.E.; Underwood, M.; Buchbinder, R. Consensus on Exercise Reporting Template (CERT): Explanation and elaboration statement. Br. J. Sports Med. 2016, 50, 1428–1437. [Google Scholar] [CrossRef]
- De Labra, C.; Guimaraes-Pinheiro, C.; Maseda, A.; Lorenzo, T.; Millán-Calenti, J.C. Effects of physical exercise interventions in frail older adults: A systematic review of randomized controlled trials Physical functioning, physical health and activity. BMC Geriatr. 2015, 15. [Google Scholar] [CrossRef]
- Abellan Van Kan, G.; Vellas, B. Is the mini nutritional assessment an appropriate tool to assess frailty in older adults? J. Nutr. Heal. Aging 2011, 15, 159–161. [Google Scholar] [CrossRef]
- Visvanathan, R. Under-nutrition in older people: A serious and growing global problem! J. Postgrad. Med. 2003, 49, 352–360. [Google Scholar] [PubMed]
- Cederholm, T.; Barazzoni, R.; Austin, P.; Ballmer, P.; Nutrition, G.B.-C. Undefined ESPEN Guidelines on Definitions and Terminology of Clinical Nutrition; Elsevier: Amsterdam, The Netherlands, 2017. [Google Scholar]
- De La Fuente-Arrillaga, C.; Vzquez Ruiz, Z.; Bes-Rastrollo, M.; Sampson, L.; Martinez-González, M.A. Reproducibility of an FFQ validated in Spain. Public Health Nutr. 2010, 13, 1364–1372. [Google Scholar] [CrossRef] [PubMed]
- Martin-moreno, J.M.; Boyle, P.; Gorgojo, L.; Maisonneuve, P.; Fernandez-rodriguez, J.C.; Salvini, S.; Willett, W.C. Development and validation of a food frequency questionnaire in Spain. Int. J. Epidemiol. 1993, 22, 512–519. [Google Scholar] [CrossRef] [PubMed]
- Abizanda, P.; López, M.D.; García, V.P.; de Dios Estrella, J.; da Silva González, Á.; Vilardell, N.B.; Torres, K.A. Effects of an oral nutritional supplementation plus physical exercise intervention on the physical function, nutritional status, and quality of life in frail institutionalized older adults: The ACTIVNES study. J. Am. Med. Dir. Assoc. 2015, 16, 439.e9–439.e16. [Google Scholar] [CrossRef] [PubMed]
- Sanchis, J.; Núñez, E.; Ruiz, V.; Bonanad, C.; Fernández, J.; Cauli, O.; García-Blas, S.; Mainar, L.; Valero, E.; Rodríguez-Borja, E.; et al. Usefulness of clinical data and biomarkers for the identification of frailty after acute coronary syndromes. Can. J. Cardiol. 2015, 31, 1462–1468. [Google Scholar] [CrossRef]
- Halfon, M.; Phan, O.; Theta, D. Vitamin D: A review on its effects on muscle strength, the risk of fall, and frailty. Biomed. Res. Int. 2015, 2015, 1–11. [Google Scholar] [CrossRef]
- Granic, A.; Hil, T.R.; Davies, K.; Jagger, C.; Adamson, A.; Siervo, M.; Kirkwood, T.B.L.; Mathers, J.C.; Sayer, A.A. Vitamin d status, muscle strength and physical performance decline in very old adults: A prospective study. Nutrients 2017, 9, 379. [Google Scholar] [CrossRef]
- Zamboni, M.; Zoico, E.; Tosoni, P.; Zivelonghi, A.; Bortolani, A.; Maggi, S.; Di Francesco, V.; Bosello, O. Relation between vitamin D, physical performance, and disability in elderly persons. J. Gerontol. 2002, 57, M7–M11. [Google Scholar] [CrossRef]
- Hirani, V.; Cumming, R.G.; Naganathan, V.; Blyth, F.; Le Couteur, D.G.; Handelsman, D.J.; Waite, L.M.; Seibel, M.J. Associations between serum 25-hydroxyvitamin D concentrations and multiple health conditions, physical performance measures, disability, and all-cause mortality: The concord health and ageing in men project. J. Am. Geriatr. Soc. 2014, 62, 417–425. [Google Scholar] [CrossRef]
- Esain, I.; Gil, S.M.; Bidaurrazaga-Letona, I.; Rodriguez-Larrad, A. Effects of 3 months of detraining on functional fitness and quality of life in older adults who regularly exercise. Aging Clin. Exp. Res. 2019, 31, 503–510. [Google Scholar] [CrossRef]
- Henwood, T.R.; Taaffe, D.R. Detraining and retraining in older adults following long-term muscle power or muscle strength specific training. J. Gerontol. 2008, 63, 751–758. [Google Scholar] [CrossRef] [PubMed]
- Lobo, A.; Carvalho, J.; Santos, P. Effects of training and detraining on physical fitness, physical activity patterns, cardiovascular variables, and HRQoL after 3 health-promotion interventions in institutionalized elders. Int. J. Family Med. 2010, 2010, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Toraman, N.F.; Ayceman, N. Effects of six weeks of detraining on retention of functional fitness of old people after nine weeks of multicomponent training. Br. J. Sports Med. 2005, 39, 565–568. [Google Scholar] [CrossRef] [PubMed]
- Zwarenstein, M.; Treweek, S.; Gagnier, J.J.; Altman, D.G.; Tunis, S.; Haynes, B. Pragmatic trials in healthcare (Practihc) group. Improving the reporting of pragmatic trials: An extension of the CONSORT statement. BMJ 2008, 337, a2390. [Google Scholar] [CrossRef] [PubMed]
- World Medical Association. World Medical Association Declaration of Helsinki: Ethical principles for medical research involving human subjects. JAMA 2013, 310, 2191–2194. [Google Scholar] [CrossRef]
- Kelley, G.A.; Kelley, K.S. Dropouts and compliance in exercise interventions targeting bone mineral density in adults: A meta-analysis of randomized controlled trials. J. Osteoporos. 2013, 2013. [Google Scholar] [CrossRef]
- Morley, J.E.; Malmstrom, T.K.; Miller, D.K. A simple frailty questionnaire (FRAIL) predicts outcomes in middle aged african americans. J. Nutr. Health Aging 2012, 16, 601–608. [Google Scholar] [CrossRef]
- Guralnik, J.M.; Simonsick, E.M.; Ferrucci, L.; Glynn, R.J.; Berkman, L.F.; Blazer, D.G.; Scherr, P.A.; Wallace, R.B. A short physical performance battery assessing lower extremity function: Association with self-reported disability and prediction of mortality and nursing home admission. J. Gerontol. 1994, 49, M85–M94. [Google Scholar] [CrossRef]
- Treacy, D.; Hassett, L. The short physical performance battery. J. Physiother. 2018, 64, 61. [Google Scholar] [CrossRef]
- Rodriguez-Larrad, A.; Arrieta, H.; Rezola, C.; Kortajarena, M.; Yanguas, J.J.; Iturburu, M.; Susana, M.G.; Irazusta, J. Effectiveness of a multicomponent exercise program in the attenuation of frailty in long-term nursing home residents: Study protocol for a randomized clinical controlled trial. BMC Geriatr. 2017, 17, 60. [Google Scholar] [CrossRef]
- Casas-Herrero, A.; Anton-Rodrigo, I.; Zambom-Ferraresi, F.; Sáez De Asteasu, M.L.; Martinez-Velilla, N.; Elexpuru-Estomba, J.; Marin-Epelde, I.; Ramon-Espinoza, F.; Petidier-Torregrosa, R.; Sanchez-Sanchez, J.L.; et al. Effect of a multicomponent exercise programme (VIVIFRAIL) on functional capacity in frail community elders with cognitive decline: Study protocol for a randomized multicentre control trial. Trials 2019, 20, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Rikli, R.; Jones, C. Senior Fitness Test Manual, 2nd ed.; Tocco, A., Maurer, K., Cox, K., Shea, B., Feeney, J., Huls, S., Eds.; Human Kinetics: Champaign, IL, USA, 2002; ISBN 978-1-4504-1118-9. [Google Scholar]
- Oja, P.; Tuxworth, B. Eurofit Para Adultos: Evaluación de la Aptitud Física en Relación con la Salud; Ministry of Education and Culture: Montevideo, Uruguay, 1998.
- Podsiadlo, D.; Richardson, S. The timed “up & go”: A test of basic functional mobility for frail elderly persons. J. Am. Geriatr. Soc. 1991, 39, 142–148. [Google Scholar] [PubMed]
- Hofheinz, M.; Mibs, M. The prognostic validity of the timed up and go test with a dual task for predicting the risk of falls in the elderly. Gerontol. Geriatr. Med. 2016, 2, 233372141663779. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, C.; Sunnerhagen, K.S.; Willén, C. Walking speed and distance in different environments of subjects in the later stage post-stroke. Physiother. Theory Pract. 2010, 26, 519–527. [Google Scholar] [CrossRef]
- Kim, H.; Park, I.; On, L. Reliability and validity of gait speed test. J Exerc. Nutr. Biochem. 2016, 20, 46–50. [Google Scholar] [CrossRef]
- Izquierdo, M. Multicomponent physical exercise program: Vivifrail. Nutr. Hosp. 2019, 36, 50–56. [Google Scholar] [CrossRef]
- Marfell-Jones, M.J.; Stewart, A.D.; De Ridder, J.H. International Standards for Anthropometric Assessment; International Society for the Advancement of Kinanthropometry: Wellington, New Zealand, 2012; ISBN 0620362073. [Google Scholar]
- Plasqui, G.; Bonomi, A.G.; Westerterp, K.R. Daily physical activity assessment with accelerometers: New insights and validation studies. Obes. Rev. 2013, 14, 451–462. [Google Scholar] [CrossRef]
- Washburn, R.A.; Smith, K.W.; Jette, A.M.; Janney, C.A. The physical activity scale for the elderly (PASE): Development and evaluation. J. Clin. Epidemiol. 1993, 46, 153–162. [Google Scholar] [CrossRef]
- López-Rodríguez, C.; Laguna, M.; Gómez-Cabello, A.; Gusi, N.; Espino, L.; Villa, G.; Pedrero-Chamizo, R.; Casajus, J.A.; Ara, I.; Aznar, S. Validation of the self-report EXERNET questionnaire for measuring physical activity and sedentary behavior in elderly. Arch. Gerontol. Geriatr. 2017, 69, 156–161. [Google Scholar] [CrossRef]
- Chasan-taber, L.; Erickson, J.B.; Nasca, P.C.; Chasan-taber, S.; Freedson, P.S. Validity and reproducibility of a physical activity questionnaire in women. Med. Sci. Sports Exerc. 2002, 34, 987–992. [Google Scholar] [CrossRef]
- Chasan-Taber, L.; Erickson, J.B.; McBride, J.W.; Nasca, P.C.; Chasan-Taber, S.; Freedson, P.S. Reproducibility of a self-administered lifetime physical activity questionnaire among female college alumnae. Am. J. Epidemiol. 2002, 155, 282–289. [Google Scholar] [CrossRef] [PubMed]
- Guirao-Goris, J.A.; Cabrero-García, J.; Moreno Pina, J.P.; Muñoz-Mendoza, C.L. Revisión estructurada de los cuestionarios y escalas que miden la actividad física en los adultos mayores y ancianos. Gac. Sanit. 2009, 23, 334-e51. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Sattler, M.C.; Jaunig, J.; Watson, E.D.; van Poppel, M.N.M.; Mokkink, L.B.; Terwee, C.B.; Dietz, P. Physical activity questionnaires for pregnancy: A systematic review of measurement properties. Sport. Med. 2018, 48, 2317–2346. [Google Scholar] [CrossRef] [PubMed]
- Martínez-González, M.Á.; Corella, D.; Salas-salvadó, J.; Ros, E.; Covas, M.I.; Fiol, M.; Wärnberg, J.; Arós, F.; Ruíz-Gutiérrez, V.; Lamuela-Raventós, R.M.; et al. Cohort profile: Design and methods of the PREDIMED study. Int. J. Epidemiol. 2012, 41, 377–385. [Google Scholar] [CrossRef] [PubMed]
- Papadaki, A.; Johnson, L.; Toumpakari, Z.; England, C.; Rai, M.; Toms, S.; Penfold, C.; Zazpe, I.; Martínez-González, M.A.; Feder, G. Validation of the English version of the 14-item mediterranean diet adherence screener of the PREDIMED study, in people at high cardiovascular risk in the UK. Nutrients 2018, 10, 138. [Google Scholar] [CrossRef]
- Schröder, H.; Fitó, M.; Estruch, R.; Martínez-González, M.A.; Corella, D.; Salas-Salvadó, J.; Lamuela-Raventós, R.; Ros, E.; Salaverría, I.; Fiol, M.; et al. A short screener is valid for assessing mediterranean diet adherence among older spanish men and women. J. Nutr. 2011, 141, 1140–1145. [Google Scholar] [CrossRef]
- Mataix, J. Tabla de Composición de Alimentos, 5th ed.; Universidad de Granada, Ed.; Faculty of Pharmacy: Granada, Spain, 2009. [Google Scholar]
- Moreiras, O.; Carbajal, Á.; Cabrera, L.; Cuadrado, C. Tablas de Composición de Alimentos; Organizacion Panamericana de la Salud: Washington, DC, USA, 2003. [Google Scholar]
- Folstein, M.F.; Folstein, S.E.; McHugh, P.R. “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J. Psychiatr. Res. 1975, 12, 189–198. [Google Scholar] [CrossRef]
- Group, T.E. EuroQol—A new facility for the measurement of health-related quality of life. Health Policy 1990, 16, 199–208. [Google Scholar] [CrossRef]
- Lawton, M.P.; Brody, E.M. Assessment of older people: Self-maintaining and instrumental activities of daily living. Gerontologist 1965, 9, 179–186. [Google Scholar] [CrossRef]
- Cabañero-Martínez, M.J.; Cabrero-García, J.; Richart-Martínez, M.; Muñoz-Mendoza, C.L. The Spanish versions of the Barthel index (BI) and the Katz index (KI) of activities of daily living (ADL): A structured review. Arch. Gerontol. Geriatr. 2009, 49, e77–e84. [Google Scholar] [CrossRef]
- Mahoney, F.I.; Barthel, D.W. Functional evaluation: The Barthel Index: A simple index of independence useful in scoring improvement in the rehabilitation of the chronically ill. Md. State Med. J. 1965, 14, 61–65. [Google Scholar] [PubMed]
- Sainsbury, A.; Seebass, G.; Bansal, A.; Young, J.B. Reliability of the Barthel Index when used with older people. Age Ageing 2005, 34, 228–232. [Google Scholar] [CrossRef] [PubMed]
- Yardley, L.; Beyer, N.; Hauer, K.; Kempen, G.; Piot-Ziegler, C.; Todd, C. Development and initial validation of the Falls Efficacy Scale-International (FES-I). Age Ageing 2005, 34, 614–619. [Google Scholar] [CrossRef] [PubMed]
- Leyton, M.; Batista, M.; Lobato, S.; JimÉnez, R. Validation of the questionnaire of the transtheoretical model of change of physical exercise Validación del cuestionario del modelo transteőrico del cambio de ejercicio físico. Rev. Int. Med. Ciencias Act. Fis. Deport. 2019, 19, 329–350. [Google Scholar] [CrossRef]
- Marcus, B.H.; Simkin, L.R. The transtheoretical model: Applications to exercise behavior. Med. Sci. Sports Exerc. 1994, 26, 1400–1404. [Google Scholar] [CrossRef] [PubMed]
- Marijuán, P.C.; Montero-Marín, J.; Navarro, J.; García-Campayo, J.; Del Moral, R. The “sociotype” construct: Gauging the structure and dynamics of human sociality. PLoS ONE 2017, 12, e0189568. [Google Scholar] [CrossRef]
- Simundic, A.; Bölenius, K.; Cadamuro, J.; Church, S.; Cornes, M.P.; van Dongen-Lases, E.C.; Hoke, R. Joint EFLM-COLABIOCLI Recommendation for venous blood sampling. Clin. Chem. Lab. Med. 2018, 56, 2015–2038. [Google Scholar] [CrossRef]
- Binder, E.F.; Schechtman, K.B.; Ehsani, A.A.; Steger-May, K.; Brown, M.; Sinacore, D.R.; Yarasheski, K.E.; Holloszy, J.O. Effects of exercise training on frailty in community-dwelling older adults: Results of a randomized, controlled trial. J. Am. Geriatr. Soc. 2002, 50, 1921–1928. [Google Scholar] [CrossRef]
- Means, K.M.; Rodell, D.E.; O’Sullivan, P.S. Balance, mobility, and falls among community-dwelling elderly persons: Effects of a rehabilitation exercise program. Am. J. Phys. Med. Rehabil. 2005, 84, 238–250. [Google Scholar] [CrossRef]
- Vaughan, S.; Wallis, M.; Polit, D.; Steele, M.; Shum, D.; Morris, N. The effects of multimodal exercise on cognitive and physical functioning and brain-derived neurotrophic factor in older women: A randomised controlled trial. Age Ageing 2014, 43, 623–629. [Google Scholar] [CrossRef]
- Nakamura, Y.; Tanaka, K.; Yabushita, N.; Sakai, T.; Shigematsu, R. Effects of exercise frequency on functional fitness in older adult women. Arch. Gerontol. Geriatr. 2007, 44, 163–173. [Google Scholar] [CrossRef] [PubMed]
- Coubard, O.A.; Duretz, S.; Lefebvre, V.; Lapalus, P.; Ferrufino, L. Practice of contemporary dance improves cognitive flexibility in aging. Front. Aging Neurosci. 2011, 3, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Ward, D.S.; Evenson, K.R.; Vaughn, A.; Rodgers, A.B.; Troiano, R.P. Accelerometer use in physical activity: Best practices and research recommendations. Med. Sci. Sports Exerc. 2005, 37 (Suppl. 11), S582–S588. [Google Scholar] [CrossRef] [PubMed]
- Cadore, E.L.; Izquierdo, M. Muscle power training: A hallmark for muscle function retaining in frail clinical setting. J. Am. Med. Dir. Assoc. 2018, 19, 190–192. [Google Scholar] [CrossRef] [PubMed]
- Cadore, E.L.; Pinto, R.S.; Reischak-Oliveira, Á.; Izquierdo, M. Explosive type of contractions should not be avoided during resistance training in elderly. Exp. Gerontol. 2018, 102, 81–83. [Google Scholar] [CrossRef]
- Reid, K.F.; Fielding, R.A. Skeletal muscle power: A critical determinant of physical functioning in older adults. Exerc. Sport Sci. Rev. 2012, 40, 4–12. [Google Scholar] [CrossRef]
- Tarazona-Santabalbina, F.J.; Gómez-Cabrera, M.C.; Pérez-Ros, P.; Martínez-Arnau, F.M.; Cabo, H.; Tsaparas, K.; Salvador-Pascual, A.; Rodriguez-Mañas, L.; Viña, J. A multicomponent exercise intervention that reverses frailty and improves cognition, emotion, and social networking in the community-dwelling frail elderly: A randomized clinical trial. J. Am. Med. Dir. Assoc. 2016, 17, 426–433. [Google Scholar] [CrossRef]
- Bréchat, P.-H.; Lonsdorfer, J.; Vogel, T. Health promotion by safe, individualized, and accessible physical activities and sports. Presse Med. 2007, 36, 379–380. [Google Scholar] [CrossRef]
- Farooqui, M.A.; Tan, Y.T.; Bilger, M.; Finkelstein, E.A. Effects of financial incentives on motivating physical activity among older adults: Results from a discrete choice experiment. BMC Public Health 2014, 14, 141. [Google Scholar] [CrossRef]
| Phase | Familiarization (Weeks 1–4) | Accumulation (Weeks 5–14) | Transformation (Weeks 15–21) | Functional (Weeks 22–24) | |||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Strength Goals | Cause Training Adaptations | Increase Strength Levels | Enhance Intermuscular Coordination | Increase Power | Improve Performance DLA | ||||||||||||||||||||
| Learn Technical Executions | Increase Muscle Endurance | Increase Muscle Endurance and Strength Level | Increase Strength Levels | Increase Power and Coordination | |||||||||||||||||||||
| Method summary | S Ex | 6–7 Ex, 1–2 sets, 8–10↓ rep, 90 s rest | 7–8 Ex, 2 sets, 10–15→ rep, 60 s rest | 7 Ex (LL+UL), 2 sets, 12–15→ rep, 60–75 s rest | 6–7 Ex, 2 sets, 12–15↑ rep, 90 s rest | FS: 6 Ex (DLA), 2 sets, 60–90 s, 60–90 s rest (a) | |||||||||||||||||||
| PS: 6–7 Ex (LL+UL), 2–3 sets, 12–15↑rep, 90 s rest (a) | |||||||||||||||||||||||||
| B Ex | 2 Ex, 2 sets, 15–20 s | 2 Ex, 2 sets, 30 s | Include in S Ex | 6–7 Ex, 2 sets, 20 s | FS: 6 Ex, 2–3 sets, 30–60 s | ||||||||||||||||||||
| PS: 6–7 Ex, 2–3 sets, 30 s | |||||||||||||||||||||||||
| Weeks | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | 19 | 20 | 21 | 22 | 23 | 24 | |
| Exercises | 6 | 6 | 7 | 7 | 7 ● | 7 | 7 | 8 ‡ | 8 ● | 8 | 8 ‡ | 8 ● | 8 | 8 ‡ | 7 ● | 7 | 7 ● | 7 | 6 ● | 6 ‡ | 7 | 6 ● | 6 ‡ | 6 | |
| Sets | 1 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 FS-3 PS | 2 FS-3 PS | |
| Repetitions & execution velocity | 8↓ | 8↓ | 10↓ | 10↓ | 10→ | 12→ | 15→ | 12→ | 12→ | 15→ | 12→ | 12→ | 15→ | 12→ | 12→ | 15→ | 12→ | 15→ | 12↑ | 12↑ | 15↑ | FS: 60 s | FS: 75 s | FS: 90 s | |
| PS: 12↑ | PS: 12↑ | PS: 15↑ | |||||||||||||||||||||||
| Rest time (s) | 90 | 90 | 90 | 90 | 60 | 60 | 60 | 60 | 60 | 60 | 60 | 60 | 60 | 60 | 75 | 75 | 75 | 75 | 90 (20 a) | 90 (20 a) | 90 (20 a) | FS: 60 (30 a) | FS: 75 (45 a) | FS: 90 (60 a) | |
| PS: 90 (30 a) | PS: 90 (30 a) | PS: 90 (30 a) | |||||||||||||||||||||||
| Aerobic and Skills Goals | Increase Aerobic Capacity (VO2 Max) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Improve Coordination and Functional Performance | |||||||||||||||||||||
| Enhance Motor Skills and Dynamic Balance | |||||||||||||||||||||
| Phase | Familiarization Period (Weeks 1–4) | Development Period (Weeks 5–14) | Maintenance Period (Weeks 15–21) | ||||||||||||||||||
| Method summary | 7 Ex, 2 sets of 35–40 s, 60–90 s rest | 7 Ex, 2 sets of 60–90 s, 90–60 s rest | 7 Ex, 2 sets of 90 s, 90 s rest (30–60 s a) | ||||||||||||||||||
| Weeks | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | 19 | 20 | 21 |
| Exercises | 7 | 7 | 7 | 7 | 7 ● | 7 | 7 | 7‡ | 7 ● | 7 | 7 | 7 ‡ | 7 ● | 7 | 7 | 7‡ | 7 ● | 7 | 7 | 7 ‡ | 7 |
| Sets | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 |
| Time (s) | 30 | 30 | 45 | 45 | 60 | 60 | 60 | 60 | 75 | 75 | 75 | 75 | 90 | 90 | 90 | 90 | 90 | 90 | 90 | 90 | 90 |
| Rest time (s) | 60 | 60 | 90 | 90 | 90 | 90 | 75 | 75 | 75 | 75 | 60 | 60 | 60 | 60 | 90 (30 a) | 90 (30 a) | 90 (45 a) | 90 (45 a) | 90 (60 a) | 90 (60 a) | 90 (60 a) |
| Total session WT (min) | 7 | 7 | 10.5 | 10.5 | 14 | 14 | 14 | 14 | 17.5 | 17.5 | 17.5 | 17.5 | 21 | 21 | 21 + 7 a | 21 + 7 a | 21 + 10.5 a | 21 + 10.5 a | 21 + 14 a | 21 + 14 a | 21 + 14 a |
| Ratio (WT:RT) (s) | 1:2 | 1:2 | 1:2 | 1:2 | 1:1.5 | 1:1.5 | 1:1.25 | 1:1.25 | 1:1 | 1:1 | 1.25:1 | 1.25:1 | 1.5:1 | 1.5:1 | 2:1 | 2:1 | 2.25:1 | 2.25:1 | 2.5:1 | 2.5:1 | 2.5:1 |
| Outcome/Variable | Test/Method | Data Collection | Equipment | Timeline Evaluation Period |
|---|---|---|---|---|
| FITNESS | ||||
| Balance and agility | ||||
| Static balance | Flamingo test | Time holding balance on one foot | Digital stopwatch (Fisher Scientific, Bartlesville, OK, USA) | t1, t2, t3, t4 |
| Agility and dynamic balance | 2.45-m up-and-go test | Time spent to rise from a chair, walk 2.45 m, turn, come back and sit | Digital stopwatch (Fisher Scientific, Bartlesville, OK, USA), chair and a cone | t1, t2, t3, t4 |
| TUG | Time spent to rise from a chair, walk 3 m, turn, come back and sit | |||
| TUGcog | The same as TUG but adding a cognitive task while performing the test | |||
| Flexibility | ||||
| Lower limb flexibility | Chair sit and reach test | Distance from the tips of the middle fingers to the toe end of the shoe (cm) performed from a sitting position with one leg extended | Rosscraft Anthrotape (Rosscraft Innovations Inc., Vancouver, BC, Canada) | t1, t2, t3, t4 |
| Upper limb flexibility | Back scratch test | Distance of overlap or the distance between the tips of the middle fingers of both arms | Rosscraft Anthrotape (Rosscraft Innovations Inc., Vancouver, BC, Canada) | t1, t2, t3, t4 |
| Dynamic strength | ||||
| Lower limb strength | Chair stand test | Number of stands from a chair | Digital stopwatch (Fisher Scientific, Bartlesville, OK, USA) and a chair | t1, t2, t3, t4 |
| Upper limb strength | Arm curl test | Number of curl lifts | Digital stopwatch (Fisher Scientific, Bartlesville, OK, USA), and dumbbells of 2.5 and 4 kg. | t1, t2, t3, t4 |
| Maximum isometric strength | ||||
| Grip strength | Handgrip test | PF (kg) | Dynamometer (Takei TKK 5401, Takei Scientific Instruments, Tokyo, Japan) | t1, t2 t3, t4 |
| Shoulder abduction Elbow flexion Knee extension | Hand-held Dynamometer | PF (kg), TPF (s.) and AVG-F (Kg) | Dynamometer (Lafayette Manual Muscle Testing Systems, Lafayette, IN, USA) | t1, t3, t4 |
| Walking speed | ||||
| Maximum gait speed | 30-m walk test | Time spent in covered 30-m as fast as possible | Digital stopwatch (Fisher Scientific, Bartlesville, OK, USA), and cones | t1, t2, t3, t4 |
| Usual gait speed | 6-m usual walking test | Time spent in covered 6-m as usual gait speed | Digital stopwatch (Fisher Scientific, Bartlesville, OK, USA), and cones | t1, t2, t3, t4 |
| Aerobic endurance | ||||
| Cardiorespiratory fitness | 6-min walk test | Maximum distance covered in 6-min time limit | Digital stopwatch (Fisher Scientific, Bartlesville, OK, USA), and cones | t1, t2, t3, t4 |
| Outcome/Variable | Test/Method | Data Collection | Equipment | Timeline Evaluation Period |
|---|---|---|---|---|
| FRAILTY STATUS & FUNCTIONAL CAPACITY | ||||
| SPPB | Static balance, gait speed and lower strength | Digital stopwatch (Fisher Scientific, Bartlesville, OK, USA), cones and a chair of 90 cm | t1, t2, t3, t4 | |
| Frailty phenotype of Fried | Shrinking or unintentional weight loss, weakness (grip strength), poor endurance and energy, slowness and low physical activity level. | Dynamometer (Takei TKK 5401, Takei Scientific Instruments, Tokyo, Japan) | t1, t2, t3, t4 | |
| Questionnaire | Degree of disease and dependence for DLA | Adapted version of the clinical frailty scale | t0 | |
| Fatigue, resistance, ambulation, illnesses and loss of weight | Frail scale questionnaire | t0 | ||
| BODY COMPOSITION | ||||
| BMD and strength indices | ||||
| pQCT | Bone size, mass, apparent tissue density and geometry of trabecular and cortical bone in radius and tibia of non-dominant limbs (leg at 4%, 38% and 66% sites and forearm at 4% and 66%) | XCT 2000 pQCT Scanner, Ortometrix, INC, Naples, FL, USA | t1, t3 | |
| Body height, weight, fat mass and muscle mass | ||||
| Portable stadiometer | Height | Stadiometer (SECA, Hamburg, Germany) | t1 | |
| BIA | Weight, FFM, FMT and FM% | TANITA BC 418-MA (Tanita Corp., Tokyo, Japan) | t1, t2, t3, t4 | |
| pQCT | Muscle area, muscle size index, muscle density, and index of intramuscular fat (forearm and leg 66% site scans) | XCT 2000 pQCT Scanner, Ortometrix, INC, Naples, FL, USA | t1, t3 | |
| BMI | BMI = weight/height2; kg/m2 | Height and weight | Stadiometer (SECA, Hamburg, Germany) and TANITA BC 418-MA (Tanita Corp., Tokyo, Japan) | t1, t2, t3, t4 |
| Anthropometry | ISAK protocol | Circumferences of arm, waist, hip and calf were registered | Rosscraft Anthrotape (Rosscraft Innovations Inc., Vancouver, BC, Canada) |
| Outcome/Variable | Test/Method | Data Collection | Equipment | Timeline Evaluation Period |
|---|---|---|---|---|
| PHYSICAL ACTIVITY AND SEDENTARY BEHAVIOR | ||||
| Accelerometery | Active and sedentary periods during a week | Accelerometers GENEActiv (ActivInsights Ltd., Kimbolton, Cambridgeshire, UK) | t1, t3, t4 | |
| Questionnaires | Leisure, household and occupational activities | PASE | t1, t2, t3, t4 | |
| Time sitting, walking and doing household activities | EEPAQ | t1, t2, t3, t4 | ||
| Recreational, household and occupational PA | CT-PAQ | t1 | ||
| NUTRITION | ||||
| Questionnaires | Frequency of amount consumption of 12 main components of the MedDiet and food habits related to it | MEDAS | t1, t3, t4 | |
| Dietary intakes of total calories, protein, fats, carbohydrate, alcohol, cholesterol, fibre and vitamins A and C | FFQ | t1 | ||
| Questionnaire and anthropometry | Nutrition, subjective health, BMI, calf circumference and midarm circumference | MNA and Rosscraft Anthrotape (Rosscraft Innovations Inc., Vancouver, BC, Canada) | t1, t3, t4 | |
| COGNITIVE STATE | Questionnaire | Mental functions (vocal responses, orientation, memory, attention and ability to name and follow verbal and written commands) | MMS | t1, t3, t4 |
| HEALTH-RELATED QUALITY OF LIFE | ||||
| QoL | Questionnaires | Mobility, self-care, main activity, social relationships, pain, mood and health subjective perception | EQ-5D | t1, t2, t3, t4 |
| DLA performance | Questionnaires | Telephoning, shopping, food preparation, housekeeping, laundering, use of transportation, use of medicine and financial behavior | IADL | t1, t3, t4 |
| Feeding, bathing, grooming, dressing, bowel control, bladder control, toileting, chair transfer, ambulation and stair climbing | BI | t1, t3, t4 |
| Outcome/Variable | Test/Method | Data Collection | Equipment | Timeline Evaluation Period |
|---|---|---|---|---|
| FALL RISK AND FEAR OF FALLING | ||||
| Questionnaires | Fear of falling when carrying out different ADL | FES-I | t1, t3, t4 | |
| Fear of falling, falls suffered and their consequences | EXERNET 3.0 general questionnaire | t1, t2, t3, t4 | ||
| CHANGE OF BEHAVIOR | Questionnaire | Participation in moderate PA in the present and past, and intention to participate in the future | TMPECQ | t1, t3, t4 |
| SOCIOTYPE | Questionnaire | Quality of the relationships with family, friends and acquaintances | SOCQ | t1, t3, t4 |
| CLINICAL AND ANALYTICAL PARAMETERS | ||||
| Blood test | Direct venous puncture fasting in the antecubital vein (10 mL) | Basic blood count, serology and vitD | Syringe, blood tubes, centrifuge, freezer, VITROS 25-OH VitD Total Reagent Pack and VITROS 25-PH vitD Total Calibrators | t1, t3, t4 |
| Blood pressure | Ankle-arm index | AAI = maximum systolic blood pressure in the ankle or foot/maximum systolic blood pressure in the arm | Sphygmomanometer (Omron, HEM-7112), manual continuous Doppler probe (Hadeco Bidop ES-100V3, Düsseldorf, Germany) and conductive gel | t1, t3, t4 |
| Outcome/Variable | Test/Method | Data Collection | Equipment/Method | Timeline Evaluation Period |
|---|---|---|---|---|
| COMPLEMENTARY INFORMATION | ||||
| Personal, socioeconomic and educational | Questionnaire | Age, sex and socioeconomic and educational level | EXERNET 3.0 general questionnaire | t1 |
| Health-related | Questionnaire | Urinary incontinence, insomnia, sun exposure, drug and tobacco consumption and chronic pains | EXERNET 3.0 general questionnaire | t1, t2, t3, t4 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fernández-García, Á.I.; Gómez-Cabello, A.; Moradell, A.; Navarrete-Villanueva, D.; Pérez-Gómez, J.; Ara, I.; Pedrero-Chamizo, R.; Subías-Perié, J.; Muniz-Pardos, B.; Casajús, J.A.; et al. How to Improve the Functional Capacity of Frail and Pre-Frail Elderly People? Health, Nutritional Status and Exercise Intervention. The EXERNET-Elder 3.0 Project. Sustainability 2020, 12, 6246. https://doi.org/10.3390/su12156246
Fernández-García ÁI, Gómez-Cabello A, Moradell A, Navarrete-Villanueva D, Pérez-Gómez J, Ara I, Pedrero-Chamizo R, Subías-Perié J, Muniz-Pardos B, Casajús JA, et al. How to Improve the Functional Capacity of Frail and Pre-Frail Elderly People? Health, Nutritional Status and Exercise Intervention. The EXERNET-Elder 3.0 Project. Sustainability. 2020; 12(15):6246. https://doi.org/10.3390/su12156246
Chicago/Turabian StyleFernández-García, Ángel Iván, Alba Gómez-Cabello, Ana Moradell, David Navarrete-Villanueva, Jorge Pérez-Gómez, Ignacio Ara, Raquel Pedrero-Chamizo, Jorge Subías-Perié, Borja Muniz-Pardos, José A. Casajús, and et al. 2020. "How to Improve the Functional Capacity of Frail and Pre-Frail Elderly People? Health, Nutritional Status and Exercise Intervention. The EXERNET-Elder 3.0 Project" Sustainability 12, no. 15: 6246. https://doi.org/10.3390/su12156246
APA StyleFernández-García, Á. I., Gómez-Cabello, A., Moradell, A., Navarrete-Villanueva, D., Pérez-Gómez, J., Ara, I., Pedrero-Chamizo, R., Subías-Perié, J., Muniz-Pardos, B., Casajús, J. A., & Vicente-Rodríguez, G. (2020). How to Improve the Functional Capacity of Frail and Pre-Frail Elderly People? Health, Nutritional Status and Exercise Intervention. The EXERNET-Elder 3.0 Project. Sustainability, 12(15), 6246. https://doi.org/10.3390/su12156246








