Implementation of Floating Treatment Wetlands for Textile Wastewater Management: A Review
Abstract
1. Introduction
2. Potential Pollutants in Textile Wastewater
2.1. Dyes
2.2. Dissolved Solids
2.3. Suspended Solids
2.4. Heavy Metals
3. Available Technologies for Treatment of Textile Effluent
4. Floating Treatment Wetlands for Textile Effluent Treatment
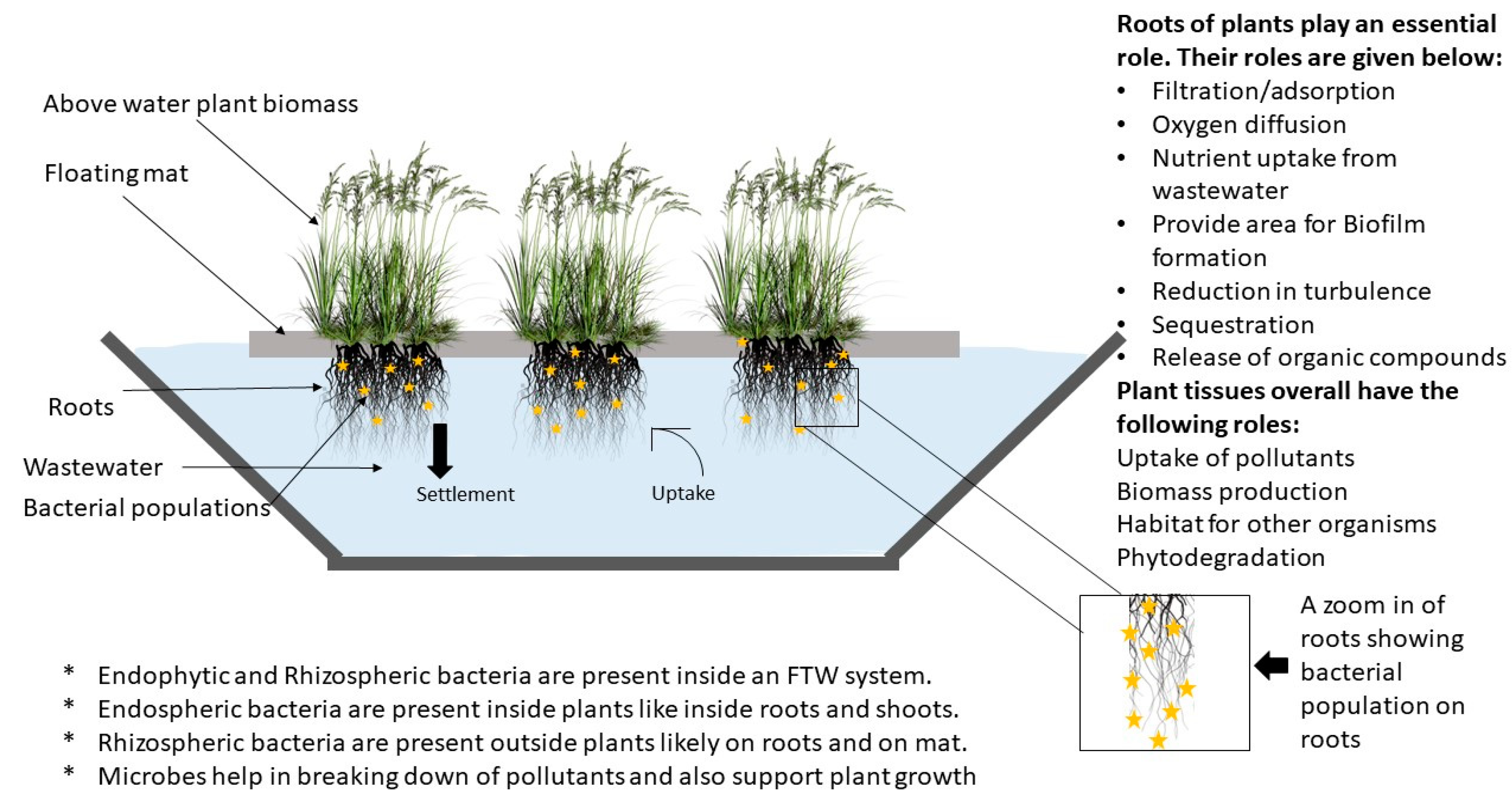
4.1. Role of Plants
- Direct uptake of pollutants by the roots [120].
- Extracellular enzyme production by roots [113].
- Provide a surface area for the growth of biofilm [117].
- Roots secrete root exudates that help in denitrification [121].
- Suspended particles are entrapped in the roots [111].
- Macrophytes also enhance flocculation of suspended matter [113].
4.2. Role of Microorganism
5. Removal of Pollutants
5.1. Removal of Dissolved and Suspended Solids
5.2. Removal of Organic Matter
5.3. Removal of Heavy Metals
6. Factors Affecting the Performance of FTWs
6.1. Plant Selection
6.2. Plant Coverage
6.3. Aeration and Dissolve Oxygen
6.4. Bacterial Inoculation
7. Care and Maintenance of FTWs
8. Conclusions and Recommendations
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Owa, F. Water pollution: Sources, effects, control and management. Mediterr. J. Soc. Sci. 2013, 4, 65. [Google Scholar] [CrossRef]
- Keiser, D.A. The missing benefits of clean water and the role of mismeasured pollution. J. Assoc. Environ. Resour. Econ. 2019, 6, 669–707. [Google Scholar] [CrossRef]
- Júnior, M.A.C. Advances in the Treatment of Textile Effluents: A Review. OALib. J. 2019, 6, 1–13. [Google Scholar] [CrossRef]
- Hussein, F.H. Chemical Properties of Treated Textile Dyeing Wastewater. Asian J. Chem. 2013, 25, 9393–9400. [Google Scholar] [CrossRef]
- Khandare, R.V.; Govindwar, S.P. Phytoremediation of textile dyes and effluents: Current scenario and future prospects. Biotechnol. Adv. 2015, 33, 1697–1714. [Google Scholar] [CrossRef]
- Ayadi, I.; Souissi, Y.; Jlassi, I.; Peixoto, F.; Mnif, W. Chemical synonyms, molecular structure and toxicological risk assessment of synthetic textile dyes: A critical review. J. Dev. Drugs 2016, 5, 2. [Google Scholar] [CrossRef]
- Rovira, J.; Domingo, J.L. Human health risks due to exposure to inorganic and organic chemicals from textiles: A review. Environ. Res. 2019, 168, 62–69. [Google Scholar] [CrossRef] [PubMed]
- Saratale, R.G.; Saratale, G.D.; Chang, J.-S.; Govindwar, S.P. Bacterial decolorization and degradation of azo dyes: A review. J. Taiwan Inst. Chem. Eng. 2011, 42, 138–157. [Google Scholar] [CrossRef]
- Khan, S.; Malik, A. Environmental and health effects of textile industry wastewater. In Environmental Deterioration and Human Health; Springer: Berlin/Heidelberg, Germany, 2014; pp. 55–71. [Google Scholar]
- Yaseen, D.A.; Scholz, M. Textile dye removal using experimental wetland ponds planted with common duckweed under semi-natural conditions. Environ. Prot. Eng. 2017, 43, 39–60. [Google Scholar] [CrossRef]
- Crini, G.; Lichtfouse, E. Advantages and disadvantages of techniques used for wastewater treatment. Environ. Chem. Lett. 2019, 17, 145–155. [Google Scholar] [CrossRef]
- Kalra, S.S.; Mohan, S.; Sinha, A.; Singh, G. Advanced Oxidation Processes for Treatment of Textile And Dye Wastewater: A Review; 2nd International Conference on Environmental Science and Development, 2011; IACSIT Press: Singapore, 2011; pp. 271–275. [Google Scholar]
- Ashraf, S.; Ali, Q.; Zahir, Z.A.; Ashraf, S.; Asghar, H.N. Phytoremediation: Environmentally sustainable way for reclamation of heavy metal polluted soils. Ecotoxicol. Environ. Saf. 2019, 174, 714–727. [Google Scholar] [CrossRef] [PubMed]
- Anning, A.K.; Akoto, R. Assisted phytoremediation of heavy metal contaminated soil from a mined site with Typha latifolia and Chrysopogon zizanioides. Ecotoxicol. Environ. Saf. 2018, 148, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Jeevanantham, S.; Saravanan, A.; Hemavathy, R.; Kumar, P.S.; Yaashikaa, P.; Yuvaraj, D. Removal of toxic pollutants from water environment by phytoremediation: A survey on application and future prospects. Environ. Technol. Inno. 2019, 13, 264–276. [Google Scholar] [CrossRef]
- El-Esawi, M.A.; Al-Ghamdi, A.A.; Ali, H.M.; Ahmad, M. Overexpression of AtWRKY30 Transcription Factor Enhances Heat and Drought Stress Tolerance in Wheat (Triticum aestivum L.). Genes 2019, 10, 163. [Google Scholar] [CrossRef] [PubMed]
- El-Esawi, M.A.; Alayafi, A.A. Overexpression of rice Rab7 gene improves drought and heat tolerance and increases grain yield in rice (Oryza sativa L.). Genes 2019, 10, 56. [Google Scholar] [CrossRef]
- Khamparia, S.; Jaspal, D.; Malviya, A. Optimization of adsorption process for removal of sulphonated di azo textile dye. OPTIMIZATION 2015, 1, 61–66. [Google Scholar] [CrossRef]
- Ali, N.; El-Mohamedy, R. Microbial decolourization of textile waste water. J. Saudi Chem. Soc. 2012, 16, 117–123. [Google Scholar] [CrossRef]
- Wesenberg, D.; Kyriakides, I.; Agathos, S.N. White-rot fungi and their enzymes for the treatment of industrial dye effluents. Biotechnol. Adv. 2003, 22, 161–187. [Google Scholar] [CrossRef]
- Feng, C.; Fang-yan, C.; Yu-bin, T. Isolation, identification of a halotolerant acid red B degrading strain and its decolorization performance. Apcbee Procedia 2014, 9, 131–139. [Google Scholar] [CrossRef][Green Version]
- Mahmoud, M. Decolorization of certain reactive dye from aqueous solution using Baker’s Yeast (Saccharomyces cerevisiae) strain. HBRC J. 2016, 12, 88–98. [Google Scholar] [CrossRef]
- El-Kassas, H.Y.; Mohamed, L.A. Bioremediation of the textile waste effluent by Chlorella vulgaris. Egypt. J. Aqua. Res. 2014, 40, 301–308. [Google Scholar] [CrossRef]
- Rather, L.J.; Akhter, S.; Hassan, Q.P. Bioremediation: Green and sustainable technology for textile effluent treatment. In Sustainable Innovations in Textile Chemistry and Dyes; Springer: Berlin/Heidelberg, Germany, 2018; pp. 75–91. [Google Scholar]
- Chung, K.T.; Stevens, S.E., Jr. Degradation azo dyes by environmental microorganisms and helminths. Environ. Toxicol. Chem. Int. J. 1993, 12, 2121–2132. [Google Scholar]
- Sandesh, K.; Kumar, G.; Chidananda, B.; Ujwal, P. Optimization of direct blue-14 dye degradation by Bacillus fermus (KX898362) an alkaliphilic plant endophyte and assessment of degraded metabolite toxicity. J. Hazard. Mater. 2019, 364, 742–751. [Google Scholar]
- Stolz, A. Basic and applied aspects in the microbial degradation of azo dyes. Appl. Microbiol. Biotechnol. 2001, 56, 69–80. [Google Scholar] [CrossRef]
- Kumar, P.S.; Saravanan, A. Sustainable wastewater treatments in textile sector. In Sustainable Fibres and Textiles; Elsevier: Amsterdam, The Netherlands, 2017; pp. 323–346. [Google Scholar]
- Nawaz, M.S.; Ahsan, M. Comparison of physico-chemical, advanced oxidation and biological techniques for the textile wastewater treatment. Alex. Eng. J. 2014, 53, 717–722. [Google Scholar] [CrossRef]
- Dey, S.; Islam, A. A review on textile wastewater characterization in Bangladesh. Resour. Environ. 2015, 5, 15–44. [Google Scholar]
- Kant, R. Textile dyeing industry an environmental hazard. Nat. Sci. 2011, 4, 22–26. [Google Scholar] [CrossRef]
- Ghaly, A.; Ananthashankar, R.; Alhattab, M.; Ramakrishnan, V. Production, characterization and treatment of textile effluents: A critical review. J. Chem. Eng. Process Technol. 2014, 5, 1–19. [Google Scholar]
- Shehzadi, M.; Afzal, M.; Khan, M.U.; Islam, E.; Mobin, A.; Anwar, S.; Khan, Q.M. Enhanced degradation of textile effluent in constructed wetland system using Typha domingensis and textile effluent-degrading endophytic bacteria. Water Res. 2014, 58, 152–159. [Google Scholar] [CrossRef]
- Watharkar, A.D.; Khandare, R.V.; Waghmare, P.R.; Jagadale, A.D.; Govindwar, S.P.; Jadhav, J.P. Treatment of textile effluent in a developed phytoreactor with immobilized bacterial augmentation and subsequent toxicity studies on Etheostoma olmstedi fish. J. Hazard. Mater. 2015, 283, 698–704. [Google Scholar] [CrossRef]
- Tara, N.; Iqbal, M.; Mahmood Khan, Q.; Afzal, M. Bioaugmentation of floating treatment wetlands for the remediation of textile effluent. Water Environ. J. 2019, 33, 124–134. [Google Scholar] [CrossRef]
- Hussain, Z.; Arslan, M.; Malik, M.H.; Mohsin, M.; Iqbal, S.; Afzal, M. Integrated perspectives on the use of bacterial endophytes in horizontal flow constructed wetlands for the treatment of liquid textile effluent: Phytoremediation advances in the field. J. Environ. Manag. 2018, 224, 387–395. [Google Scholar] [CrossRef] [PubMed]
- Kadam, S.K.; Watharkar, A.D.; Chandanshive, V.V.; Khandare, R.V.; Jeon, B.-H.; Jadhav, J.P.; Govindwar, S.P. Co-planted floating phyto-bed along with microbial fuel cell for enhanced textile effluent treatment. J. Clean. Prod. 2018, 203, 788–798. [Google Scholar] [CrossRef]
- Tara, N.; Arslan, M.; Hussain, Z.; Iqbal, M.; Khan, Q.M.; Afzal, M. On-site performance of floating treatment wetland macrocosms augmented with dye-degrading bacteria for the remediation of textile industry wastewater. J. Clean. Prod. 2019, 217, 541–548. [Google Scholar] [CrossRef]
- Bafana, A.; Devi, S.S.; Chakrabarti, T. Azo dyes: Past, present and the future. Environ. Rev. 2011, 19, 350–371. [Google Scholar] [CrossRef]
- Essandoh, M.; Garcia, R.A. Efficient removal of dyes from aqueous solutions using a novel hemoglobin/iron oxide composite. Chemosphere 2018, 206, 502–512. [Google Scholar] [CrossRef]
- Uddin, M.G.; Islam, M.M.; Islam, M.R. Effects of reductive stripping of reactive dyes on the quality of cotton fabric. Fash. Text. 2015, 2, 8. [Google Scholar] [CrossRef]
- El Harfi, S.; El Harfi, A. Classifications, properties and applications of textile dyes: A review. Appl. J. Environ. Eng. Sci. 2017, 3, 311–320. [Google Scholar]
- Benkhaya, S.; El Harfi, A. A critical review of surface water contaminated with dyes from textile industry effluent: Possible approaches. Appl. J. Environ. Eng. Sci. 2018, 4, 1–12. [Google Scholar]
- Imran, M.; Shaharoona, B.; Crowley, D.E.; Khalid, A.; Hussain, S.; Arshad, M. The stability of textile azo dyes in soil and their impact on microbial phospholipid fatty acid profiles. Ecotoxicol. Environ. Saf. 2015, 120, 163–168. [Google Scholar] [CrossRef]
- Uday, U.S.P.; Mahata, N.; Sasmal, S.; Bandyopadhyay, T.K.; Mondal, A.; Bhunia, B. Dyes Contamination in the Environment: Ecotoxicological Effects, Health Hazards, and Biodegradation and Bioremediation Mechanisms for Environmental Cleanup. In Environmental Pollutants and Their Bioremediation Approaches; CRC Press: Boca Raton, FL, USA, 2017; pp. 127–176. [Google Scholar]
- Bokare, A.D.; Chikate, R.C.; Rode, C.V.; Paknikar, K.M. Iron-nickel bimetallic nanoparticles for reductive degradation of azo dye Orange G in aqueous solution. Appl. Catal. B-Environ. 2008, 79, 270–278. [Google Scholar] [CrossRef]
- Ismail, A.; Toriman, M.E.; Juahir, H.; Zain, S.M.; Habir, N.L.A.; Retnam, A.; Kamaruddin, M.K.A.; Umar, R.; Azid, A. Spatial assessment and source identification of heavy metals pollution in surface water using several chemometric techniques. Mar. Pollut. Bull. 2016, 106, 292–300. [Google Scholar] [CrossRef]
- Khatri, J.; Nidheesh, P.; Singh, T.A.; Kumar, M.S. Advanced oxidation processes based on zero-valent aluminium for treating textile wastewater. Chem. Eng. J. 2018, 348, 67–73. [Google Scholar] [CrossRef]
- Bhatia, D.; Sharma, N.R.; Kanwar, R.; Singh, J. Physicochemical assessment of industrial textile effluents of Punjab (India). Appl. Water Sci. 2018, 8, 83. [Google Scholar] [CrossRef]
- Maruthi, Y.; Rao, S.; Kiran, D. Evaluation of ground water pollution potential in Chandranagar, Visakhapatnam: A case study. J. Ecobiol. 2004, 16, 423–430. [Google Scholar]
- Mohabansi, N.P.; Tekade, P.; Bawankar, S. Physico-chemical and microbiological analysis of textile industry effluent of Wardha region. Water Res. Dev. 2011, 1, 40–44. [Google Scholar]
- Kolhe, A.; Pawar, V. Physico-chemical analysis of effluents from dairy industry. Rec. Res. Sci. Technol. 2011, 3, 29–32. [Google Scholar]
- Elango, G.; Govindasamy, R. Removal of Colour from Textile Dyeing Effluent Using Temple Waste Flowers as Ecofriendly Adsorbent. IOSR J. Appl. Chem. 2018, 11, 19–28. [Google Scholar]
- Bilotta, G.; Brazier, R. Understanding the influence of suspended solids on water quality and aquatic biota. Water Res. 2008, 42, 2849–2861. [Google Scholar] [CrossRef]
- Durotoye, T.O.; Adeyemi, A.A.; Omole, D.O.; Onakunle, O. Impact assessment of wastewater discharge from a textile industry in Lagos, Nigeria. Cogent Eng. 2018, 5, 1531687. [Google Scholar] [CrossRef]
- Ubale, M.A.; Salkar, V.D. Experimental study on electrocoagulation of textile wastewater by continuous horizontal flow through aluminum baffles. Korean J. Chem. Eng. 2017, 34, 1044–1050. [Google Scholar] [CrossRef]
- Ismail, M.; Akhtar, K.; Khan, M.; Kamal, T.; Khan, M.A.; M Asiri, A.; Seo, J.; Khan, S.B. Pollution, toxicity and carcinogenicity of organic dyes and their catalytic bio-remediation. Curr. Pharm. Des. 2019, 25, 3645–3663. [Google Scholar] [CrossRef] [PubMed]
- Sultana, T.; Arooj, F.; Nawaz, M.; Alam, S. Removal of Heavy Metals from Contaminated Soil using Plants: A Mini-Review. PSM Biol. Res. 2019, 4, 113–117. [Google Scholar]
- Yin, K.; Wang, Q.; Lv, M.; Chen, L. Microorganism remediation strategies towards heavy metals. Chem. Eng. J. 2019, 360, 1553–1563. [Google Scholar] [CrossRef]
- Rezaei, A.; Hassani, H.; Hassani, S.; Jabbari, N.; Mousavi, S.B.F.; Rezaei, S. Evaluation of groundwater quality and heavy metal pollution indices in Bazman basin, southeastern Iran. Groundw. Sustain. Dev. 2019, 9, 100245. [Google Scholar] [CrossRef]
- Roque, F.; Diaz, K.; Ancco, M.; Delgado, D.; Tejada, K. Biodepuration of domestic sewage, textile effluents and acid mine drainage using starch-based xerogel from recycled potato peels. Water Sci. Technol. 2018, 77, 1250–1261. [Google Scholar] [CrossRef]
- Järup, L. Hazards of heavy metal contamination. Br. Med. Bull. 2003, 68, 167–182. [Google Scholar] [CrossRef]
- Noreen, M.; Shahid, M.; Iqbal, M.; Nisar, J. Measurement of cytotoxicity and heavy metal load in drains water receiving textile effluents and drinking water in vicinity of drains. Measurement 2017, 109, 88–99. [Google Scholar] [CrossRef]
- Mulugeta, M.; Tibebe, D. Assessment of some selected metals from textile effluents in amhara region using AAS and ICPOES. Assessment 2019, 7, 27–31. [Google Scholar] [CrossRef]
- Wijeyaratne, W.D.N.; Wickramasinghe, P.M.U. Treated textile effluents: Cytotoxic and genotoxic effects in the natural aquatic environment. Bull. Environ. Contam. Toxicol. 2020, 104, 245–252. [Google Scholar] [CrossRef]
- Paździor, K.; Bilińska, L.; Ledakowicz, S. A review of the existing and emerging technologies in the combination of AOPs and biological processes in industrial textile wastewater treatment. Chem. Eng. J. 2019, 376, 120597. [Google Scholar] [CrossRef]
- Holkar, C.R.; Jadhav, A.J.; Pinjari, D.V.; Mahamuni, N.M.; Pandit, A.B. A critical review on textile wastewater treatments: Possible approaches. J. Environ. Manag. 2016, 182, 351–366. [Google Scholar] [CrossRef] [PubMed]
- Deng, D.; Aryal, N.; Ofori-Boadu, A.; Jha, M.K. Textiles wastewater treatment. Water Environ. Res 2018, 90, 1648–1662. [Google Scholar] [CrossRef] [PubMed]
- Vandevivere, P.C.; Bianchi, R.; Verstraete, W. Treatment and reuse of wastewater from the textile wet-processing industry: Review of emerging technologies. J. Chem. Technol. Biotechnol. Int. Res. Process Environ. Clean Technol. 1998, 72, 289–302. [Google Scholar] [CrossRef]
- Rodrigues, C.S.; Madeira, L.M.; Boaventura, R.A. Synthetic textile wastewaters treatment by coagulation/flocculation using ferric salt as coagulant. Environ. Eng. Manag. J. 2017, 16, 1881–1889. [Google Scholar]
- Queiroz, R.D.C.S.D.; Lôbo, I.P.; de Ribeiro, V.S.; Rodrigues, L.B.; de Almeida Neto, J.A. Assessment of autochthonous aquatic macrophytes with phytoremediation potential for dairy wastewater treatment in floating constructed wetlands. Int. J. Phytorem. 2020, 22, 518–528. [Google Scholar] [CrossRef]
- Chandran, D. A review of the textile industries waste water treatment methodologies. Int. J. Sci. Eng. Res. 2016, 7, 2229–5518. [Google Scholar]
- Dąbrowski, A. Adsorption—From theory to practice. Adv. Colloid Interface Sci. 2001, 93, 135–224. [Google Scholar] [CrossRef]
- Amrial, M.; Kusumandari, K.; Saraswati, T.; Suselo, Y. Textile Wastewater Treatment by Using Plasma Corona Discharge in a Continuous Flow System; IOP Conference Series: Materials Science and Engineering, 2019; IOP Publishing: Bristol, UK, 2019; p. 012016. [Google Scholar]
- Paprowicz, J.; Słodczyk, S. Application of biologically activated sorptive columns for textile waste water treatment. Environ. Technol. 1988, 9, 271–280. [Google Scholar] [CrossRef]
- Choo, K.-H.; Choi, S.-J.; Hwang, E.-D. Effect of coagulant types on textile wastewater reclamation in a combined coagulation/ultrafiltration system. Desalination 2007, 202, 262–270. [Google Scholar] [CrossRef]
- Kalaiarasi, K.; Lavanya, A.; Amsamani, S.; Bagyalakshmi, G. Decolourization of textile dye effluent by non-viable biomass of Aspergillus fumigatus. Braz. Arc. Biol. Technol. 2012, 55, 471–476. [Google Scholar] [CrossRef]
- Forss, J.; Lindh, M.V.; Pinhassi, J.; Welander, U. Microbial biotreatment of actual textile wastewater in a continuous sequential rice husk biofilter and the microbial community involved. PLoS ONE 2017, 12, e0170562. [Google Scholar] [CrossRef] [PubMed]
- Can, O.; Kobya, M.; Demirbas, E.; Bayramoglu, M. Treatment of the textile wastewater by combined electrocoagulation. Chemosphere 2006, 62, 181–187. [Google Scholar] [CrossRef]
- GilPavas, E.; Dobrosz-Gómez, I.; Gómez-García, M.-Á. Optimization and toxicity assessment of a combined electrocoagulation, H2O2/Fe2+/UV and activated carbon adsorption for textile wastewater treatment. Sci. Total Environ. 2019, 651, 551–560. [Google Scholar] [CrossRef] [PubMed]
- Aleem, M.; Cao, J.; Li, C.; Rashid, H.; Wu, Y.; Nawaz, M.I.; Abbas, M.; Akram, M.W. Coagulation-and Adsorption-Based Environmental Impact Assessment and Textile Effluent Treatment. Water Air Soil Pollut. 2020, 231, 45. [Google Scholar] [CrossRef]
- Bilińska, L.; Blus, K.; Gmurek, M.; Ledakowicz, S. Coupling of electrocoagulation and ozone treatment for textile wastewater reuse. Chem. Eng. J. 2019, 358, 992–1001. [Google Scholar] [CrossRef]
- Dotto, J.; Fagundes-Klen, M.R.; Veit, M.T.; Palácio, S.M.; Bergamasco, R. Performance of different coagulants in the coagulation/flocculation process of textile wastewater. J. Clean. Prod. 2019, 208, 656–665. [Google Scholar] [CrossRef]
- Khandegar, V.; Saroha, A.K. Electrocoagulation for the treatment of textile industry effluent–a review. J. Environ. Manag. 2013, 128, 949–963. [Google Scholar] [CrossRef]
- Beltrán-Heredia, J.; Sánchez-Martín, J.; Rodríguez-Sánchez, M. Textile wastewater purification through natural coagulants. Appl. Water Sci. 2011, 1, 25–33. [Google Scholar] [CrossRef]
- Abouri, M.; Souabi, S.; Jada, A. Optimization of Coagulation Flocculation Process for the Removal of Heavy Metals from Real Textile Wastewater. Adv. Intell. Syst. Sustain. Dev. (AI2SD’2018) Vol 3 Adv. Intell. Syst. Appl. Environ. 2019, 913, 257. [Google Scholar]
- Solano, A.M.S.; de Araújo, C.K.C.; de Melo, J.V.; Peralta-Hernandez, J.M.; da Silva, D.R.; Martínez-Huitle, C.A. Decontamination of real textile industrial effluent by strong oxidant species electrogenerated on diamond electrode: Viability and disadvantages of this electrochemical technology. Appl. Catal. B Environ. 2013, 130, 112–120. [Google Scholar] [CrossRef]
- Martínez-Huitle, C.A.; Panizza, M. Electrochemical oxidation of organic pollutants for wastewater treatment. Curr. Opin. Electrochem. 2018, 11, 62–71. [Google Scholar] [CrossRef]
- Marin, N.M.; Pascu, L.F.; Demba, A.; Nita-Lazar, M.; Badea, I.A.; Aboul-Enein, H. Removal of the Acid Orange 10 by ion exchange and microbiological methods. Int. J. Environ. Sci. Technol. 2019, 16, 6357–6366. [Google Scholar] [CrossRef]
- Yetim, T.; Tekin, T. A kinetic study on photocatalytic and sonophotocatalytic degradation of textile dyes. Period. Polytech. Chem. Eng. 2017, 61, 102–108. [Google Scholar] [CrossRef]
- Jorfi, S.; Barzegar, G.; Ahmadi, M.; Soltani, R.D.C.; Takdastan, A.; Saeedi, R.; Abtahi, M. Enhanced coagulation-photocatalytic treatment of Acid red 73 dye and real textile wastewater using UVA/synthesized MgO nanoparticles. J. Environ. Manag. 2016, 177, 111–118. [Google Scholar] [CrossRef]
- Naseem, Z.; Bhatti, H.N.; Iqbal, M.; Noreen, S.; Zahid, M. Fenton and photo-fenton oxidation for the remediation of textile effluents: An experimental study. Text. Cloth. 2019, 235–251. [Google Scholar] [CrossRef]
- Sharma, A.; Syed, Z.; Brighu, U.; Gupta, A.B.; Ram, C. Adsorption of textile wastewater on alkali-activated sand. J. Clean. Prod. 2019, 220, 23–32. [Google Scholar] [CrossRef]
- Dasgupta, J.; Sikder, J.; Chakraborty, S.; Curcio, S.; Drioli, E. Remediation of textile effluents by membrane based treatment techniques: A state of the art review. J. Environ. Manag. 2015, 147, 55–72. [Google Scholar] [CrossRef] [PubMed]
- Liang, Y.; Zhu, H.; Bañuelos, G.; Yan, B.; Zhou, Q.; Yu, X.; Cheng, X. Constructed wetlands for saline wastewater treatment: A review. Ecol. Eng. 2017, 98, 275–285. [Google Scholar] [CrossRef]
- Shi, L.; Huang, J.; Zeng, G.; Zhu, L.; Gu, Y.; Shi, Y.; Yi, K.; Li, X. Roles of surfactants in pressure-driven membrane separation processes: A review. Environ. Sci. Pollut. Res. 2019, 26, 30731–30754. [Google Scholar] [CrossRef]
- Van der Bruggen, B.; Canbolat, Ç.B.; Lin, J.; Luis, P. The potential of membrane technology for treatment of textile wastewater. In Sustainable Membrane Technology for Water and Wastewater Treatment; Springer: Berlin/Heidelberg, Germany, 2017; pp. 349–380. [Google Scholar]
- Saeed, T.; Khan, T. Constructed wetlands for industrial wastewater treatment: Alternative media, input biodegradation ratio and unstable loading. J. Environ. Chem. Eng. 2019, 7, 103042. [Google Scholar] [CrossRef]
- Periasamy, D.; Mani, S.; Ambikapathi, R. White Rot Fungi and Their Enzymes for the Treatment of Industrial Dye Effluents. In Recent Advancement in White Biotechnology Through Fungi; Springer: Berlin/Heidelberg, Germany, 2019; pp. 73–100. [Google Scholar]
- Anastasi, A.; Spina, F.; Prigione, V.; Tigini, V.; Giansanti, P.; Varese, G.C. Scale-up of a bioprocess for textile wastewater treatment using Bjerkandera adusta. Bioresour. Technol. 2010, 101, 3067–3075. [Google Scholar] [CrossRef] [PubMed]
- Fazal, T.; Mushtaq, A.; Rehman, F.; Khan, A.U.; Rashid, N.; Farooq, W.; Rehman, M.S.U.; Xu, J. Bioremediation of textile wastewater and successive biodiesel production using microalgae. Renew. Sust. Energ. Rev. 2018, 82, 3107–3126. [Google Scholar] [CrossRef]
- Sekomo, C.B.; Kagisha, V.; Rousseau, D.; Lens, P. Heavy metal removal by combining anaerobic upflow packed bed reactors with water hyacinth ponds. Environ. Technol. 2012, 33, 1455–1464. [Google Scholar] [CrossRef]
- Yaseen, D.A.; Scholz, M. Treatment of synthetic textile wastewater containing dye mixtures with microcosms. Environ. Sci. Pollut. Rese. 2018, 25, 1980–1997. [Google Scholar] [CrossRef] [PubMed]
- Siddique, K.; Rizwan, M.; Shahid, M.J.; Ali, S.; Ahmad, R.; Rizvi, H. Textile wastewater treatment options: A critical review. In Enhancing Cleanup of Environmental Pollutants; Springer: Berlin/Heidelberg, Germany, 2017; pp. 183–207. [Google Scholar]
- Simsek, H.; Kasi, M.; Ohm, J.-B.; Blonigen, M.; Khan, E. Bioavailable and biodegradable dissolved organic nitrogen in activated sludge and trickling filter wastewater treatment plants. Water Res. 2013, 47, 3201–3210. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.P.; Singh, P.K.; Gupta, R.; Singh, R.L. Treatment and recycling of wastewater from textile industry. In Advances in Biological Treatment of Industrial Waste Water and their Recycling for a Sustainable Future; Springer: Berlin/Heidelberg, Germany, 2019; pp. 225–266. [Google Scholar]
- Winanti, E.; Rahmadyanti, E.; Fajarwati, I. Ecological Approach of Campus Wastewater Treatment Using Constructed Wetland; IOP Conference Series: Materials Science and Engineering, 2018; IOP Publishing: Bristol, UK, 2018; p. 012062. [Google Scholar]
- Afzal, M.; Rehman, K.; Shabir, G.; Tahseen, R.; Ijaz, A.; Hashmat, A.J.; Brix, H. Large-scale remediation of oil-contaminated water using floating treatment wetlands. NPJ Clean Water 2019, 2, 1–10. [Google Scholar] [CrossRef]
- El-Esawi, M.A.; Alaraidh, I.A.; Alsahli, A.A.; Alzahrani, S.M.; Ali, H.M.; Alayafi, A.A.; Ahmad, M. Serratia liquefaciens KM4 Improves Salt Stress Tolerance in Maize by Regulating Redox Potential, Ion Homeostasis, Leaf Gas Exchange and Stress-Related Gene Expression. Int. J. Mol. Sci. 2018, 19, 3310. [Google Scholar] [CrossRef]
- El-Esawi, M.A.; Al-Ghamdi, A.A.; Ali, H.M.; Alayafi, A.A. Azospirillum lipoferum FK1 confers improved salt tolerance in chickpea (Cicer arietinum L.) by modulating osmolytes, antioxidant machinery and stress-related genes expression. Environ. Exp. Bot. 2019, 159, 55–65. [Google Scholar] [CrossRef]
- Borne, K.E.; Fassman, E.A.; Tanner, C.C. Floating treatment wetland retrofit to improve stormwater pond performance for suspended solids, copper and zinc. Ecol. Eng. 2013, 54, 173–182. [Google Scholar] [CrossRef]
- Ijaz, A.; Shabir, G.; Khan, Q.M.; Afzal, M. Enhanced remediation of sewage effluent by endophyte-assisted floating treatment wetlands. Ecol. Eng. 2015, 84, 58–66. [Google Scholar] [CrossRef]
- Yeh, N.; Yeh, P.; Chang, Y.-H. Artificial floating islands for environmental improvement. Renew Sustain Energy Rev. 2015, 47, 616–622. [Google Scholar] [CrossRef]
- El-Esawi, M.A. Genetic diversity and evolution of Brassica genetic resources: From morphology to novel genomic technologies—A review. Plant Genet. Resour. 2017, 15, 388–399. [Google Scholar] [CrossRef]
- El-Esawi, M.A.; Germaine, K.; Bourke, P.; Malone, R. AFLP analysis of genetic diversity and phylogenetic relationships of Brassica oleracea in Ireland. Comptes Rendus Biol. 2016, 339, 163–170. [Google Scholar] [CrossRef]
- Tanner, C.C.; Headley, T.R. Components of floating emergent macrophyte treatment wetlands influencing removal of stormwater pollutants. Ecol. Eng. 2011, 37, 474–486. [Google Scholar] [CrossRef]
- Zhang, L.; Zhao, J.; Cui, N.; Dai, Y.; Kong, L.; Wu, J.; Cheng, S. Enhancing the water purification efficiency of a floating treatment wetland using a biofilm carrier. Environ. Sci. Pollut. Rese. 2016, 23, 7437–7443. [Google Scholar] [CrossRef] [PubMed]
- Borne, K.E.; Fassman-Beck, E.A.; Winston, R.J.; Hunt, W.F.; Tanner, C.C. Implementation and maintenance of floating treatment wetlands for urban stormwater management. J. Environ. Eng. 2015, 141, 04015030. [Google Scholar] [CrossRef]
- Ali, S.; Abbas, Z.; Rizwan, M.; Zaheer, I.E.; Yavaş, İ.; Ünay, A.; Abdel-Daim, M.M.; Bin-Jumah, M.; Hasanuzzaman, M.; Kalderis, D. Application of Floating Aquatic Plants in Phytoremediation of Heavy Metals Polluted Water: A Review. Sustainability 2020, 12, 1927. [Google Scholar] [CrossRef]
- Chang, N.-B.; Xuan, Z.; Marimon, Z.; Islam, K.; Wanielista, M.P. Exploring hydrobiogeochemical processes of floating treatment wetlands in a subtropical stormwater wet detention pond. Ecol. Eng. 2013, 54, 66–76. [Google Scholar] [CrossRef]
- Wand, H.; Vacca, G.; Kuschk, P.; Krüger, M.; Kästner, M. Removal of bacteria by filtration in planted and non-planted sand columns. Water Res. 2007, 41, 159–167. [Google Scholar] [CrossRef] [PubMed]
- Vymazal, J. Constructed wetlands for treatment of industrial wastewaters: A review. Ecol. Eng. 2014, 73, 724–751. [Google Scholar] [CrossRef]
- Shahid, M.J.; Ali, S.; Shabir, G.; Siddique, M.; Rizwan, M.; Seleiman, M.F.; Afzal, M. Comparing the performance of four macrophytes in bacterial assisted floating treatment wetlands for the removal of trace metals (Fe, Mn, Ni, Pb, and Cr) from polluted river water. Chemosphere 2020, 243, 125353. [Google Scholar] [CrossRef] [PubMed]
- Nawaz, N.; Ali, S.; Shabir, G.; Rizwan, M.; Shakoor, M.B.; Shahid, M.J.; Afzal, M.; Arslan, M.; Hashem, A.; Abd_Allah, E.F. Bacterial Augmented Floating Treatment Wetlands for Efficient Treatment of Synthetic Textile Dye Wastewater. Sustainability 2020, 12, 3731. [Google Scholar] [CrossRef]
- Borne, K.E. Floating treatment wetland influences on the fate and removal performance of phosphorus in stormwater retention ponds. Ecol. Eng. 2014, 69, 76–82. [Google Scholar] [CrossRef]
- Ashraf, S.; Afzal, M.; Naveed, M.; Shahid, M.; Ahmad Zahir, Z. Endophytic bacteria enhance remediation of tannery effluent in constructed wetlands vegetated with Leptochloa fusca. Int. J. Phytorem. 2018, 20, 121–128. [Google Scholar] [CrossRef]
- Shahid, M.; Arslan, M.; Ali, S.; Siddique, M.; Afzal, M. Floating wetlands: A sustainable tool for wastewater treatment. CLEAN Soil Air Water 2018, 46, 1–13. [Google Scholar] [CrossRef]
- Zhu, L.; Li, Z.; Ketola, T. Biomass accumulations and nutrient uptake of plants cultivated on artificial floating beds in China’s rural area. Ecol. Eng. 2011, 37, 1460–1466. [Google Scholar] [CrossRef]
- Khin, T.; Annachhatre, A.P. Novel microbial nitrogen removal processes. Biotechnol. Adv. 2004, 22, 519–532. [Google Scholar] [CrossRef]
- Borne, K.E.; Fassman-Beck, E.A.; Tanner, C.C. Floating treatment wetland influences on the fate of metals in road runoff retention ponds. Water Res. 2014, 48, 430–442. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Qiao, S.; Shao, D.; Jin, R.; Zhou, J. Simultaneous nitrogen and phosphorus removal by combined anammox and denitrifying phosphorus removal process. J. Chem. Technol. Biotechnol. 2018, 93, 94–104. [Google Scholar] [CrossRef]
- Shahid, M.J.; Tahseen, R.; Siddique, M.; Ali, S.; Iqbal, S.; Afzal, M. Remediation of polluted river water by floating treatment wetlands. Water Supply 2019, 19, 967–977. [Google Scholar] [CrossRef]
- Chua, L.H.; Tan, S.B.; Sim, C.; Goyal, M.K. Treatment of baseflow from an urban catchment by a floating wetland system. Ecol. Eng. 2012, 49, 170–180. [Google Scholar] [CrossRef]
- Nakphet, S.; Ritchie, R.J.; Kiriratnikom, S. Aquatic plants for bioremediation in red hybrid tilapia (Oreochromis niloticus× Oreochromis mossambicus) recirculating aquaculture. Aquacult. Int. 2017, 25, 619–633. [Google Scholar] [CrossRef]
- Chaturvedi, H.; Singh, V.; Gupta, G. Potential of bacterial endophytes as plant growth promoting factors. J. Plant Pathol. Microbiol. 2016, 7, 1–6. [Google Scholar] [CrossRef]
- Arslan, M.; Imran, A.; Khan, Q.M.; Afzal, M. Plant–bacteria partnerships for the remediation of persistent organic pollutants. Environ. Sci. Pollut. Res. 2017, 24, 4322–4336. [Google Scholar] [CrossRef] [PubMed]
- Fatima, K.; Afzal, M.; Imran, A.; Khan, Q.M. Bacterial rhizosphere and endosphere populations associated with grasses and trees to be used for phytoremediation of crude oil contaminated soil. Bull. Environ. Contam. Toxicol. 2015, 94, 314–320. [Google Scholar] [CrossRef]
- Shehzadi, M.; Fatima, K.; Imran, A.; Mirza, M.; Khan, Q.; Afzal, M. Ecology of bacterial endophytes associated with wetland plants growing in textile effluent for pollutant-degradation and plant growth-promotion potentials. Plant Biosyst. 2016, 150, 1261–1270. [Google Scholar] [CrossRef]
- Rehman, K.; Imran, A.; Amin, I.; Afzal, M. Inoculation with bacteria in floating treatment wetlands positively modulates the phytoremediation of oil field wastewater. J. Hazard. Mater. 2018, 349, 242–251. [Google Scholar] [CrossRef]
- Branda, S.S.; Vik, Å.; Friedman, L.; Kolter, R. Biofilms: The matrix revisited. Trends Microbiol. 2005, 13, 20–26. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Singh, S.K.; Chowdhury, I.; Singh, R. Understanding the mechanism of bacterial biofilms resistance to antimicrobial agents. Open Microbiol. J. 2017, 11, 53–62. [Google Scholar] [CrossRef] [PubMed]
- Urakawa, H.; Dettmar, D.L.; Thomas, S. The uniqueness and biogeochemical cycling of plant root microbial communities in a floating treatment wetland. Ecol. Eng. 2017, 108, 573–580. [Google Scholar] [CrossRef]
- Sun, Z.; Xie, D.; Jiang, X.; Fu, G.; Xiao, D.; Zheng, L. Effect of eco-remediation and microbial community using multilayer solar planted floating island (MS-PFI) in the drainage channel. BioRxiv 2018, 327965. [Google Scholar] [CrossRef]
- Achá, D.; Iniguez, V.; Roulet, M.; Guimaraes, J.R.D.; Luna, R.; Alanoca, L.; Sanchez, S. Sulfate-reducing bacteria in floating macrophyte rhizospheres from an Amazonian floodplain lake in Bolivia and their association with Hg methylation. Appl. Environ. Microbiol. 2005, 71, 7531–7535. [Google Scholar] [CrossRef]
- Lamers, L.P.; Van Diggelen, J.M.; Op Den Camp, H.J.; Visser, E.J.; Lucassen, E.C.; Vile, M.A.; Jetten, M.S.; Smolders, A.J.; Roelofs, J.G. Microbial transformations of nitrogen, sulfur, and iron dictate vegetation composition in wetlands: A review. Front. Microbiol. 2012, 3, 156. [Google Scholar] [CrossRef]
- Shahid, M.J.; Arslan, M.; Siddique, M.; Ali, S.; Tahseen, R.; Afzal, M. Potentialities of floating wetlands for the treatment of polluted water of river Ravi, Pakistan. Ecol. Eng. 2019, 133, 167–176. [Google Scholar] [CrossRef]
- Zhao, T.; Fan, P.; Yao, L.; Yan, G.; Li, D.; Zhang, W. Ammonifying bacteria in plant floating island of constructed wetland for strengthening decomposition of organic nitrogen. Trans. Chin. Soc. Agric. Eng. 2011, 27, 223–226. [Google Scholar]
- Govarthanan, M.; Mythili, R.; Selvankumar, T.; Kamala-Kannan, S.; Rajasekar, A.; Chang, Y.-C. Bioremediation of heavy metals using an endophytic bacterium Paenibacillus sp. RM isolated from the roots of Tridax procumbens. 3 Biotech 2016, 6, 242. [Google Scholar] [CrossRef] [PubMed]
- Barathi, S.; Karthik, C.; Nadanasabapathi, S.; Padikasan, I.A. Biodegradation of textile dye Reactive Blue 160 by Bacillus firmus (Bacillaceae: Bacillales) and non-target toxicity screening of their degraded products. Toxicol. Rep. 2020, 7, 16–22. [Google Scholar] [CrossRef]
- Franca, R.D.G.; Vieira, A.; Carvalho, G.; Oehmen, A.; Pinheiro, H.M.; Crespo, M.T.B.; Lourenço, N.D. Oerskovia paurometabola can efficiently decolorize azo dye Acid Red 14 and remove its recalcitrant metabolite. Ecotoxicol. Environ. Saf. 2020, 191, 110007. [Google Scholar] [CrossRef] [PubMed]
- Garg, N.; Garg, A.; Mukherji, S. Eco-friendly decolorization and degradation of reactive yellow 145 textile dye by Pseudomonas aeruginosa and Thiosphaera pantotropha. J. Environ. Manag. 2020, 263, 110383. [Google Scholar] [CrossRef] [PubMed]
- Roy, D.C.; Biswas, S.K.; Saha, A.K.; Sikdar, B.; Rahman, M.; Roy, A.K.; Prodhan, Z.H.; Tang, S.-S. Biodegradation of Crystal Violet dye by bacteria isolated from textile industry effluents. PeerJ 2018, 6, e5015. [Google Scholar] [CrossRef]
- Najme, R.; Hussain, S.; Maqbool, Z.; Imran, M.; Mahmood, F.; Manzoor, H.; Yasmeen, T.; Shehzad, T. Biodecolorization of Reactive Yellow-2 by Serratia sp. RN34 Isolated from textile wastewater. Water Environ. Res. 2015, 87, 2065–2075. [Google Scholar] [CrossRef]
- Bheemaraddi, M.C.; Patil, S.; Shivannavar, C.T.; Gaddad, S.M. Isolation and characterization of Paracoccus sp. GSM2 capable of degrading textile azo dye reactive violet 5. Sci. World J. 2014, 2014, 410704. [Google Scholar] [CrossRef]
- Singh, R.P.; Singh, P.K.; Singh, R.L. Bacterial decolorization of textile azo dye acid orange by Staphylococcus hominis RMLRT03. Toxicol. Int. 2014, 21, 160. [Google Scholar] [CrossRef] [PubMed]
- Garg, S.K.; Tripathi, M. Process parameters for decolorization and biodegradation of orange II (Acid Orange 7) in dye-simulated minimal salt medium and subsequent textile effluent treatment by Bacillus cereus (MTCC 9777) RMLAU1. Environ. Monit. Assess. 2013, 185, 8909–8923. [Google Scholar] [CrossRef]
- Lim, C.K.; Bay, H.H.; Aris, A.; Majid, Z.A.; Ibrahim, Z. Biosorption and biodegradation of Acid Orange 7 by Enterococcus faecalis strain ZL: Optimization by response surface methodological approach. Environ. Sci. Pollut. Res. 2013, 20, 5056–5066. [Google Scholar] [CrossRef]
- Surwase, S.V.; Deshpande, K.K.; Phugare, S.S.; Jadhav, J.P. Biotransformation studies of textile dye Remazol Orange 3R. 3 Biotech 2013, 3, 267–275. [Google Scholar] [CrossRef][Green Version]
- Deive, F.; Domínguez, A.; Barrio, T.; Moscoso, F.; Morán, P.; Longo, M.; Sanromán, M. Decolorization of dye Reactive Black 5 by newly isolated thermophilic microorganisms from geothermal sites in Galicia (Spain). J. Hazard. Mater. 2010, 182, 735–742. [Google Scholar] [CrossRef]
- Wang, H.; Su, J.Q.; Zheng, X.W.; Tian, Y.; Xiong, X.J.; Zheng, T.L. Bacterial decolorization and degradation of the reactive dye Reactive Red 180 by Citrobacter sp. CK3. Int. Biodeterior. Biodegrad. 2009, 63, 395–399. [Google Scholar] [CrossRef]
- Kolekar, Y.M.; Pawar, S.P.; Gawai, K.R.; Lokhande, P.D.; Shouche, Y.S.; Kodam, K.M. Decolorization and degradation of Disperse Blue 79 and Acid Orange 10, by Bacillus fusiformis KMK5 isolated from the textile dye contaminated soil. Bioresour. Technol. 2008, 99, 8999–9003. [Google Scholar] [CrossRef] [PubMed]
- Kalyani, D.C.; Patil, P.S.; Jadhav, J.P.; Govindwar, S.P. Biodegradation of reactive textile dye Red BLI by an isolated bacterium Pseudomonas sp. SUK1. Bioresour. Technol. 2008, 99, 4635–4641. [Google Scholar] [CrossRef] [PubMed]
- Alhassani, H.A.; Rauf, M.A.; Ashraf, S.S. Efficient microbial degradation of Toluidine Blue dye by Brevibacillus sp. Dyes Pigments 2007, 75, 395–400. [Google Scholar] [CrossRef]
- Coughlin, M.F.; Kinkle, B.K.; Bishop, P.L. High performance degradation of azo dye Acid Orange 7 and sulfanilic acid in a laboratory scale reactor after seeding with cultured bacterial strains. Water Res. 2003, 37, 2757–2763. [Google Scholar] [CrossRef]
- Hu, T. Degradation of azo dye RP2B by Pseudomonas luteola. Water Sci. Technol. 1998, 38, 299–306. [Google Scholar] [CrossRef]
- Elango, G.; Rathika, G.; Elango, S. Physico-chemical parameters of textile dyeing effluent and its impacts with case study. Int. J. Res Chem. Environ. 2017, 7, 17–24. [Google Scholar]
- Mirbolooki, H.; Amirnezhad, R.; Pendashteh, A.R. Treatment of high saline textile wastewater by activated sludge microorganisms. J. Appl Res. Technol. 2017, 15, 167–172. [Google Scholar] [CrossRef]
- Hawes, M.C.; McLain, J.; Ramirez-Andreotta, M.; Curlango-Rivera, G.; Flores-Lara, Y.; Brigham, L.A. Extracellular trapping of soil contaminants by root border cells: New insights into plant defense. Agronomy 2016, 6, 5. [Google Scholar] [CrossRef]
- Cao, W.; Zhang, H.; Wang, Y.; Pan, J. Bioremediation of polluted surface water by using biofilms on filamentous bamboo. Ecol. Eng. 2012, 42, 146–149. [Google Scholar] [CrossRef]
- Prajapati, M.; van Bruggen, J.J.; Dalu, T.; Malla, R. Assessing the effectiveness of pollutant removal by macrophytes in a floating wetland for wastewater treatment. Appl Water Sci. 2017, 7, 4801–4809. [Google Scholar] [CrossRef]
- Chen, Z.; Cuervo, D.P.; Müller, J.A.; Wiessner, A.; Köser, H.; Vymazal, J.; Kästner, M.; Kuschk, P. Hydroponic root mats for wastewater treatment—A review. Environ. Sci. Pollut. Res. 2016, 23, 15911–15928. [Google Scholar] [CrossRef]
- Ijaz, A.; Iqbal, Z.; Afzal, M. Remediation of sewage and industrial effluent using bacterially assisted floating treatment wetlands vegetated with Typha domingensis. Water Sci. Technol. 2016, 74, 2192–2201. [Google Scholar] [CrossRef]
- Qamar, M.T.; Mumtaz, H.M.; Mohsin, M.; Asghar, H.N.; Iqbal, M.; Nasir, M. Development of floating treatment wetlands with plant-bacteria partnership to clean textile bleaching effluent. Ind. Text. 2019, 70, 502–511. [Google Scholar] [CrossRef]
- Kumar, S.; Pratap, B.; Dubey, D.; Dutta, V. Microbial Communities in Constructed Wetland Microcosms and Their Role in Treatment of Domestic Wastewater. In Emerging Eco-Friendly Green Technologies for Wastewater Treatment; Springer: Berlin/Heidelberg, Germany, 2020; pp. 311–327. [Google Scholar]
- Rehman, K.; Imran, A.; Amin, I.; Afzal, M. Enhancement of oil field-produced wastewater remediation by bacterially-augmented floating treatment wetlands. Chemosphere 2019, 217, 576–583. [Google Scholar] [CrossRef]
- Suryawan, I.; Helmy, Q.; Notodarmojo, S. Textile Wastewater Treatment: Colour and COD Removal of Reactive Black-5 by Ozonation; IOP Conference Series: Earth and Environmental Science, 2018; IOP Publishing: Bristol, UK, 2018; p. 012102. [Google Scholar]
- Mohan, S.; Vidhya, K.; Sivakumar, C.; Sugnathi, M.; Shanmugavadivu, V.; Devi, M. Textile Waste Water Treatment by Using Natural Coagulant (Neem-Azadirachta India). Int. Res. J. Multidis. Technovation 2019, 1, 636–642. [Google Scholar]
- Sathya, U.; Nithya, M.; Balasubramanian, N. Evaluation of advanced oxidation processes (AOPs) integrated membrane bioreactor (MBR) for the real textile wastewater treatment. J. Environ. Manag. 2019, 246, 768–775. [Google Scholar] [CrossRef]
- Tusief, M.Q.; Malik, M.H.; Asghar, H.N.; Mohsin, M.; Mahmood, N. Bioremediation of textile wastewater through floating treatment wetland system. Int. J. Agric. Biol. 2019, 22, 821–826. [Google Scholar]
- Darajeh, N.; Idris, A.; Masoumi, H.R.F.; Nourani, A.; Truong, P.; Sairi, N.A. Modeling BOD and COD removal from Palm Oil Mill Secondary Effluent in floating wetland by Chrysopogon zizanioides (L.) using response surface methodology. J. Environ. Manag. 2016, 181, 343–352. [Google Scholar] [CrossRef] [PubMed]
- Klomjek, P.; Nitisoravut, S. Constructed treatment wetland: A study of eight plant species under saline conditions. Chemosphere 2005, 58, 585–593. [Google Scholar] [CrossRef] [PubMed]
- Yulistyorini, A.; Puspasari, A.K.; Sari, A.A. Removal of BOD and TSS of Student Dormitory Greywater Using Vertical Sub-Surface Flow Constructed Wetland of Ipomoea Aquatica; IOP Conference Series: Materials Science and Engineering, 2019; IOP Publishing: Bristol, UK, 2019; p. 012056. [Google Scholar]
- Saeed, T.; Haque, I.; Khan, T. Organic matter and nutrients removal in hybrid constructed wetlands: Influence of saturation. Chem. Eng. J. 2019, 371, 154–165. [Google Scholar] [CrossRef]
- Vymazal, J. Removal of nutrients in various types of constructed wetlands. Sci. Total Environ. 2007, 380, 48–65. [Google Scholar] [CrossRef] [PubMed]
- Arivoli, A.; Sathiamoorthi, T.; Satheeshkumar, M. Treatment of Textile Effluent by Phytoremediation with the Aquatic Plants: Alternanthera sessilis. In Bioremediation and Sustainable Technologies for Cleaner Environment; Springer: Berlin/Heidelberg, Germany, 2017; pp. 185–197. [Google Scholar]
- Ladislas, S.; Gerente, C.; Chazarenc, F.; Brisson, J.; Andres, Y. Floating treatment wetlands for heavy metal removal in highway stormwater ponds. Ecol. Eng. 2015, 80, 85–91. [Google Scholar] [CrossRef]
- Ladislas, S.; El-Mufleh, A.; Gérente, C.; Chazarenc, F.; Andrès, Y.; Béchet, B. Potential of aquatic macrophytes as bioindicators of heavy metal pollution in urban stormwater runoff. Water Air Soil Pollut. 2012, 223, 877–888. [Google Scholar] [CrossRef]
- Gadd, G.M. Metals, minerals and microbes: Geomicrobiology and bioremediation. Microbiol. 2010, 156, 609–643. [Google Scholar] [CrossRef] [PubMed]
- Haq, S.; Bhatti, A.A.; Dar, Z.A.; Bhat, S.A. Phytoremediation of Heavy Metals: An Eco-Friendly and Sustainable Approach. In Bioremediation and Biotechnology; Springer: Berlin/Heidelberg, Germany, 2020; pp. 215–231. [Google Scholar]
- Ullah, A.; Heng, S.; Munis, M.F.H.; Fahad, S.; Yang, X. Phytoremediation of heavy metals assisted by plant growth promoting (PGP) bacteria: A review. Environ. Exp. Bot. 2015, 117, 28–40. [Google Scholar] [CrossRef]
- Hussain, I.; Aleti, G.; Naidu, R.; Puschenreiter, M.; Mahmood, Q.; Rahman, M.M.; Wang, F.; Shaheen, S.; Syed, J.H.; Reichenauer, T.G. Microbe and plant assisted-remediation of organic xenobiotics and its enhancement by genetically modified organisms and recombinant technology: A review. Sci. Total Environ. 2018, 628, 1582–1599. [Google Scholar] [CrossRef]
- Doty, S.L. Enhancing phytoremediation through the use of transgenics and endophytes. New Phytol. 2008, 179, 318–333. [Google Scholar] [CrossRef]
- Wang, C.-Y.; Sample, D.J.; Bell, C. Vegetation effects on floating treatment wetland nutrient removal and harvesting strategies in urban stormwater ponds. Sci. Total Environ. 2014, 499, 384–393. [Google Scholar] [CrossRef]
- Headley, T.; Tanner, C. Constructed wetlands with floating emergent macrophytes: An innovative stormwater treatment technology. Crit. Rev. Environ. Sci. Technol. 2012, 42, 2261–2310. [Google Scholar] [CrossRef]
- Correa-García, S.; Pande, P.; Séguin, A.; St-Arnaud, M.; Yergeau, E. Rhizoremediation of petroleum hydrocarbons: A model system for plant microbiome manipulation. Microb. Biotechnol. 2018, 11, 819–832. [Google Scholar] [CrossRef] [PubMed]
- Yergeau, E.; Tremblay, J.; Joly, S.; Labrecque, M.; Maynard, C.; Pitre, F.E.; St-Arnaud, M.; Greer, C.W. Soil contamination alters the willow root and rhizosphere metatranscriptome and the root–rhizosphere interactome. ISME J. 2018, 12, 869–884. [Google Scholar] [CrossRef]
- Sheoran, A. A laboratory treatment study of acid mine water of wetlands with emergent macrophyte (Typha angustata). Int. J. Min. Reclam. Environ. 2006, 20, 209–222. [Google Scholar] [CrossRef]
- Kumar, K.; Kumar, D.; Teja, V.; Venkateswarlu, V.; Kumar, M.; Nadendla, R. A review on Typha angustata. Int. J. Phytopharm. 2013, 4, 277–281. [Google Scholar]
- Vymazal, J. Plants used in constructed wetlands with horizontal subsurface flow: A review. Hydrobiologia 2011, 674, 133–156. [Google Scholar] [CrossRef]
- Vymazal, J. The use of hybrid constructed wetlands for wastewater treatment with special attention to nitrogen removal: A review of a recent development. Water Res. 2013, 47, 4795–4811. [Google Scholar] [CrossRef] [PubMed]
- Rezania, S.; Taib, S.M.; Din, M.F.M.; Dahalan, F.A.; Kamyab, H. Comprehensive review on phytotechnology: Heavy metals removal by diverse aquatic plants species from wastewater. J. Hazard. Mater. 2016, 318, 587–599. [Google Scholar] [CrossRef] [PubMed]
- Valipour, A.; Ahn, Y.-H. Constructed wetlands as sustainable ecotechnologies in decentralization practices: A review. Environ. Sci. Pollut. Res. 2016, 23, 180–197. [Google Scholar] [CrossRef]
- Fritioff, Å.; Greger, M. Aquatic and terrestrial plant species with potential to remove heavy metals from stormwater. Int. J. Phytorem. 2003, 5, 211–224. [Google Scholar] [CrossRef]
- Vymazal, J. Emergent plants used in free water surface constructed wetlands: A review. Ecol. Eng. 2013, 61, 582–592. [Google Scholar] [CrossRef]
- Di Luca, G.A.; Mufarrege, M.; Hadad, H.R.; Maine, M.A. Nitrogen and phosphorus removal and Typha domingensis tolerance in a floating treatment wetland. Sci. Total Environ. 2019, 650, 233–240. [Google Scholar] [CrossRef] [PubMed]
- Nichols, P.; Lucke, T.; Drapper, D.; Walker, C. Performance evaluation of a floating treatment wetland in an urban catchment. Water 2016, 8, 244. [Google Scholar] [CrossRef]
- Gao, L.; Zhou, W.; Huang, J.; He, S.; Yan, Y.; Zhu, W.; Wu, S.; Zhang, X. Nitrogen removal by the enhanced floating treatment wetlands from the secondary effluent. Bioresour. Technol. 2017, 234, 243–252. [Google Scholar] [CrossRef]
- Song, H.-L.; Li, X.-N.; Lu, X.-W.; Inamori, Y. Investigation of microcystin removal from eutrophic surface water by aquatic vegetable bed. Ecol. Eng. 2009, 35, 1589–1598. [Google Scholar] [CrossRef]
- Tambunan, J.A.M.; Effendi, H.; Krisanti, M. Phytoremediating Batik Wastewater Using Vetiver Chrysopogon zizanioides (L). Pol. J. Environ. Stud. 2018, 27, 1281–1288. [Google Scholar] [CrossRef]
- De Stefani, G.; Tocchetto, D.; Salvato, M.; Borin, M. Performance of a floating treatment wetland for in-stream water amelioration in NE Italy. Hydrobiologia 2011, 674, 157–167. [Google Scholar] [CrossRef]
- Park, J.B.; Sukias, J.P.; Tanner, C.C. Floating treatment wetlands supplemented with aeration and biofilm attachment surfaces for efficient domestic wastewater treatment. Ecol. Eng. 2019, 139, 105582. [Google Scholar] [CrossRef]
- Fahid, M.; Arslan, M.; Shabir, G.; Younus, S.; Yasmeen, T.; Rizwan, M.; Siddique, K.; Ahmad, S.R.; Tahseen, R.; Iqbal, S. Phragmites australis in combination with hydrocarbons degrading bacteria is a suitable option for remediation of diesel-contaminated water in floating wetlands. Chemosphere 2020, 240, 124890. [Google Scholar] [CrossRef]
- Afzal, M.; Arslan, M.; Müller, J.A.; Shabir, G.; Islam, E.; Tahseen, R.; Anwar-ul-Haq, M.; Hashmat, A.J.; Iqbal, S.; Khan, Q.M. Floating treatment wetlands as a suitable option for large-scale wastewater treatment. Nat. Sustain. 2019, 2, 863–871. [Google Scholar] [CrossRef]
- Nanayakkara, C. Floating wetlands for management of algal washout from waste stabilization pond effluent: Case study at hikkaduwa waste stabilization ponds. Engineer 2013, 46, 63–74. [Google Scholar]
- Weragoda, S.; Jinadasa, K.; Zhang, D.Q.; Gersberg, R.M.; Tan, S.K.; Tanaka, N.; Jern, N.W. Tropical application of floating treatment wetlands. Wetlands 2012, 32, 955–961. [Google Scholar] [CrossRef]
- Lopardo, C.R.; Zhang, L.; Mitsch, W.J.; Urakawa, H. Comparison of nutrient retention efficiency between vertical-flow and floating treatment wetland mesocosms with and without biodegradable plastic. Ecol. Eng. 2019, 131, 120–130. [Google Scholar] [CrossRef]
- Wang, C.-Y.; Sample, D.J. Assessment of the nutrient removal effectiveness of floating treatment wetlands applied to urban retention ponds. J. Environ. Manag. 2014, 137, 23–35. [Google Scholar] [CrossRef] [PubMed]
- Abhilash, P.; Jamil, S.; Singh, N. Transgenic plants for enhanced biodegradation and phytoremediation of organic xenobiotics. Biotechnol. Adv. 2009, 27, 474–488. [Google Scholar] [CrossRef]
- Xie, W.-Y.; Huang, Q.; Li, G.; Rensing, C.; Zhu, Y.-G. Cadmium accumulation in the rootless macrophyte Wolffia globosa and its potential for phytoremediation. Int. J. Phytorem. 2013, 15, 385–397. [Google Scholar] [CrossRef] [PubMed]
- Rezania, S.; Din, M.F.M.; Taib, S.M.; Dahalan, F.A.; Songip, A.R.; Singh, L.; Kamyab, H. The efficient role of aquatic plant (water hyacinth) in treating domestic wastewater in continuous system. Int. J. Phytorem. 2016, 18, 679–685. [Google Scholar] [CrossRef]
- Colares, G.S.; Dell’Osbel, N.; Wiesel, P.G.; Oliveira, G.A.; Lemos, P.H.Z.; da Silva, F.P.; Lutterbeck, C.A.; Kist, L.T.; Machado, Ê.L. Floating treatment wetlands: A review and bibliometric analysis. Sci. Total Environ. 2020, 714, 136776. [Google Scholar] [CrossRef]
- Ibekwe, A.M.; Lyon, S.; Leddy, M.; Jacobson-Meyers, M. Impact of plant density and microbial composition on water quality from a free water surface constructed wetland. J. Appl. Microbiol. 2007, 102, 921–936. [Google Scholar] [CrossRef]
- Chance, L.M.G.; White, S.A. Aeration and plant coverage influence floating treatment wetland remediation efficacy. Ecol. Eng. 2018, 122, 62–68. [Google Scholar] [CrossRef]
- Wu, H.; Zhang, J.; Ngo, H.H.; Guo, W.; Hu, Z.; Liang, S.; Fan, J.; Liu, H. A review on the sustainability of constructed wetlands for wastewater treatment: Design and operation. Bioresour. Technol. 2015, 175, 594–601. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Hu, Z.; Zhang, J.; Ngo, H.H.; Guo, W.; Liang, S.; Fan, J.; Lu, S.; Wu, H. Optimizations on supply and distribution of dissolved oxygen in constructed wetlands: A review. Bioresour. Technol. 2016, 214, 797–805. [Google Scholar] [CrossRef] [PubMed]
- Austin, D.; Nivala, J. Energy requirements for nitrification and biological nitrogen removal in engineered wetlands. Ecol. Eng. 2009, 35, 184–192. [Google Scholar] [CrossRef]
- Chen, M.; Wu, X.; Chen, Y.; Dong, M. Mechanism of nitrogen removal by adsorption and bio-transformation in constructed wetland systems. Chin. J. Environ. Eng. 2009, 3, 223–228. [Google Scholar]
- Pan, J.; Fei, H.; Song, S.; Yuan, F.; Yu, L. Effects of intermittent aeration on pollutants removal in subsurface wastewater infiltration system. Bioresour. Technol. 2015, 191, 327–331. [Google Scholar] [CrossRef]
- Hu, Y.; Zhao, Y.; Zhao, X.; Kumar, J.L. High rate nitrogen removal in an alum sludge-based intermittent aeration constructed wetland. Environ. Sci. Technol. 2012, 46, 4583–4590. [Google Scholar] [CrossRef]
- Ahn, K.-H.; Song, K.-G.; Choa, E.; Cho, J.; Yun, H.; Lee, S.; Me, J. Enhanced biological phosphorus and nitrogen removal using a sequencing anoxic/anaerobic membrane bioreactor (SAM) process. Desalination 2003, 157, 345–352. [Google Scholar] [CrossRef]
- Wang, C.-Y.; Sample, D.J.; Day, S.D.; Grizzard, T.J. Floating treatment wetland nutrient removal through vegetation harvest and observations from a field study. Ecol. Eng. 2015, 78, 15–26. [Google Scholar] [CrossRef]
- Ong, S.-A.; Uchiyama, K.; Inadama, D.; Ishida, Y.; Yamagiwa, K. Performance evaluation of laboratory scale up-flow constructed wetlands with different designs and emergent plants. Bioresour. Technol. 2010, 101, 7239–7244. [Google Scholar] [CrossRef]
- Dunqiu, W.; Shaoyuan, B.; Mingyu, W.; Qinglin, X.; Yinian, Z.; Hua, Z. Effect of artificial aeration, temperature, and structure on nutrient removal in constructed floating islands. Water Environ. Res. 2012, 84, 405–410. [Google Scholar] [CrossRef]
- Fahid, M.; Ali, S.; Shabir, G.; Rashid Ahmad, S.; Yasmeen, T.; Afzal, M.; Arslan, M.; Hussain, A.; Hashem, A.; Abd Allah, E.F. Cyperus laevigatus L. Enhances Diesel Oil Remediation in Synergism with Bacterial Inoculation in Floating Treatment Wetlands. Sustainability 2020, 12, 2353. [Google Scholar] [CrossRef]
- Saleem, H.; Rehman, K.; Arslan, M.; Afzal, M. Enhanced degradation of phenol in floating treatment wetlands by plant-bacterial synergism. Int. J. Phytorem. 2018, 20, 692–698. [Google Scholar] [CrossRef] [PubMed]
- El-Esawi, M.A.; Alaraidh, I.A.; Alsahli, A.A.; Ali, H.M.; Alayafi, A.A.; Witczak, J.; Ahmad, M. Genetic variation and alleviation of salinity stress in barley. Molecules 2018, 23, 2488. [Google Scholar] [CrossRef] [PubMed]
- Vwioko, E.; Adinkwu, O.; El-Esawi, M.A. Comparative Physiological, Biochemical and Genetic Responses to Prolonged Waterlogging Stress in Okra and Maize Given Exogenous Ethylene Priming. Front. Physiol. 2017, 8, 632. [Google Scholar] [CrossRef]
- El-Esawi, M.A.; Al-Ghamdi, A.A.; Ali, H.M.; Alayafi, A.A.; Witczak, J.; Ahmad, M. Analysis of Genetic Variation and Enhancement of Salt Tolerance in French Pea (Pisum Sativum L.). Int. J. Mol. Sci. 2018, 19, 2433. [Google Scholar] [CrossRef]
- Hossen, M.Z.; Hussain, M.E.; Hakim, A.; Islam, K.; Uddin, M.N.; Azad, A.K. Biodegradation of reactive textile dye Novacron Super Black G by free cells of newly isolated Alcaligenes faecalis AZ26 and Bacillus spp obtained from textile effluents. Heliyon 2019, 5, e02068. [Google Scholar] [CrossRef]
- Pavlineri, N.; Skoulikidis, N.T.; Tsihrintzis, V.A. Constructed floating wetlands: A review of research, design, operation and management aspects, and data meta-analysis. Chem. Eng. J. 2017, 308, 1120–1132. [Google Scholar] [CrossRef]
- Thullen, J.S.; Sartoris, J.J.; Walton, W.E. Effects of vegetation management in constructed wetland treatment cells on water quality and mosquito production. Ecol. Eng. 2002, 18, 441–457. [Google Scholar] [CrossRef]
- Walton, W.E.; Jiannino, J.A. Vegetation management to stimulate denitrification increases mosquito abundance in multipurpose constructed treatment wetlands. J. Am. Mosq. Control Assoc. 2005, 21, 22–27. [Google Scholar] [CrossRef]
- Francesco, D. Biomass recovery from invasive species management in wetlands. Biomass Bioenergy 2017, 105, 259–265. [Google Scholar]
- Lishawa, S.C.; Lawrence, B.A.; Albert, D.A.; Larkin, D.J.; Tuchman, N.C. Invasive species removal increases species and phylogenetic diversity of wetland plant communities. Ecol. Evol. 2019, 9, 6231–6244. [Google Scholar] [CrossRef] [PubMed]
| Reference | [12] | [31] | [4] | [32] | [29] | [33] | [34] | [35] | [36] | [37] | [38] |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Country | India | India | Iraq | Canada | Pakistan | Pakistan | India | Pakistan | Pakistan | India | Pakistan |
| Temp (°C) | 35–45 | 33–45 | 35–45 | 25.4 | 42 | 38 | 38 | ||||
| pH | 6.0–10.0 | 9.2–11 | 5.5–10.5 | 6–10 | 8.5 | 12.93 | 7.8 | 10.7 | 8.8 | ||
| EC (μS/cm) | 8.07 | 7.1 | 8.4 | 8.2 | |||||||
| DO (mg/L) | 0.84 | ||||||||||
| Color (Pt–Co) | 50–2500 | 50–2500 | 456 | 53 (m−1) | 35.5 (m−1) | 68 (m−1) | 66 (m−1) | ||||
| COD (mg/L) | 150–10,000 | 465–1400 | 150–10,000 | 150–12,000 | 433.7 | 813 | 1090 | 471 | 493 | 1734 | 513 |
| BOD (mg/L) | 100–4000 | 130–820 | 100–4000 | 80–6000 | 224.6 | 422 | 141 | 249 | 190 | 1478 | 201 |
| Total solids (mg/L) | 3600–6540 | 5125 | 4961 | 5420 | 5420 | ||||||
| TSS (mg/L) | 100–5000 | 360–370 | 100–5000 | 15–8000 | 244 | 391 | 1004 | 391 | 324 | 6438 | 324 |
| TDS (mg/L) | 1800–6000 | 3230–6180 | 1500–6000 | 2900–3100 | 2570 | 4834 | 4569 | 5164 | 9060 | 5251 | |
| Total Alkalinity (mg/L) | 500–800 | 1250–3160 | |||||||||
| Hardness (mg/L) | 410 | 380 | |||||||||
| Total settleable solids (mg/L) | 24 | 38 | |||||||||
| Total Organic Carbon (mg/L) | 301 | 166 | 230 | 201 | |||||||
| TN (mg/L) | 55.8 | ||||||||||
| TP (mg/L) | 13 | ||||||||||
| Phenol (mg/L) | 0.86 | 0.85 | |||||||||
| Chlorine (mg/L) | 1000–6000 | 600 | |||||||||
| Chlorides (mg/L) | 1000–6000 | 200–6000 | 846 | 1382 | 1383 | ||||||
| Free chlorine (mg/L) | <10 | ||||||||||
| TA (mg/L) as CaCo3 | 500–800 | ||||||||||
| TH (mg/L) as CaCo3 | |||||||||||
| TKN (mg/L) | 70–80 | 70–80 | |||||||||
| Phosphate (mg/L) | <10 | 16.4 | |||||||||
| Sulphates (mg/L) | 500–700 | 600–1000 | 412 | 310 | 311 | ||||||
| Sulphides (mg/L) | 5–20 | ||||||||||
| Oil and grease (mg/L) | 10–50 | 10–30 | 28 | ||||||||
| Nitrogen (mg/L) | 28.6 | 28.7 | |||||||||
| Zink (mg/L) | 3–6 | <10 | |||||||||
| Nickel (mg/L) | <10 | 2.0 | 0.125 | 7.6 | 7.57 | ||||||
| Manganese (mg/L) | <10 | ||||||||||
| Iron (mg/L) | <10 | 3.3 | 1.171 | 14.3 | 14.4 | ||||||
| Copper (mg/L) | 2–6 | <10 | 0.503 | ||||||||
| Boron (mg/L) | <10 | ||||||||||
| Arsenic (mg/L) | <10 | 0.025 | 0.90 | ||||||||
| Silica (mg/L) | <15 | ||||||||||
| Mercury (mg/L) | <10 | ||||||||||
| Fluorine (mg/L) | <10 | ||||||||||
| Chromium (mg/L) | 2–5 | 0.21 | 0.812 | 9.7 | 3.7 | 9.67 | |||||
| Potassium (mg/L) | 30–50 | 858 | 242 | ||||||||
| Sodium (mg/L) | 610–2175 | 400–2175 | 7000 | 1656 | 1560 | ||||||
| Cadmium (mg/L) | 0.27 | 0.88 | 0.80 | 0.88 | |||||||
| Calcium (mg/L) | 80.16 | 110 | |||||||||
| Magnesium (mg/L) | 48.6 | 65 | |||||||||
| Sulfate (mg/L) | 412.54 | ||||||||||
| Phosphate (mg/L) | 10.08 | ||||||||||
| Nitrate (mg/L) | 24 | ||||||||||
| Lead (mg/L) | 0.880 | 0.40 | |||||||||
| Phosphorous (mg/L) | 16.4 | ||||||||||
| Aluminum (mg/L) | 2.5 |
| Type | Technique | Drawbacks | References |
|---|---|---|---|
| Chemical | Combined Electrocoagulation | The pH should be maintained below 6 during the process | [79,80] |
| Coagulation and Adsorption by Alum | Increase the concentration of sulfate and sulfide | [81] | |
| Ozonation | It has low COD reduction capacity | [82] | |
| Chemical coagulation | It is a slow technique and large amount of sludge is produced | [83,84] | |
| Electrochemical oxidation | Secondary salt contamination | [66] | |
| Coagulation | Coagulants can be associated with diseases like cancer or Alzheimer’s | [85,86] | |
| Electrochemical technology | Produce undesirable by-products that can be harmful for environment | [87,88] | |
| Ion exchange method | Not effective for all dyes | [89] | |
| Photochemical Sonolysis | Requires a lot of dissolved oxygen, high cost, and produces undesirable by-products | [90] | |
| Coagulation-photocatalytic treatment by nanoparticles | Sludge production, difficulty of light penetration in dark and colored wastewaters, high costs of nanoparticles preparation, and limited cycles of nanoparticles usage | [91] | |
| Fenton and Photo-Fenton process | Sludge production, accumulation of unused ferrous ions, and difficult maintenance of pH | [92] | |
| Physical | Adsorption/filtration (commercially activated carbon) | High cost of materials, costly operation, may not work with certain dyes and metals, performance depends upon the material types | [11] |
| Adsorption | It is a costly process | [93] | |
| Membrane based treatment | Membrane failing may happen, and costly method | [67,94] | |
| Pilot-scale bio-filter | Bio-filter has low efficiency to metabolize hydrophobic volatile organic compounds because of the massive transfer limitations | [95] | |
| Pressure-driven membranes | Sensitivity to fouling and scaling | [96,97] | |
| Biological | Constructed wetlands | High retention time and large area required for establishment | [66,98] |
| Use of White-rot fungi along with bioreactor | It has long hydraulic retention time and requires large reactors | [99,100] | |
| Microalgae | Conditions hard to maintain, selection of suitable algae is critical | [101] | |
| Duckweed and algae ponds | Inefficient removal of heavy metals | [102,103] |
| Bacteria | Dye | Reference |
|---|---|---|
| Bacillus firmus | Reactive Blue 160 | [149] |
| Oerskovia paurometabola | Acid Red 14 | [150] |
| Pseudomonas aeruginosa and Thiosphaera pantotropha | Reactive Yellow 14 | [151] |
| Enterobacter sp. CV–S1 | Crystal Violet | [152] |
| Serratia sp. RN34 | Reactive Yellow 2 | [153] |
| Paracoccus sp. GSM2 | Reactive Violet 5 | [154] |
| Staphylococcus hominis RMLRT03 | Acid Orange | [155] |
| Bacillus cereus RMLAU1 | Orange II (Acid Orange 7) | [156] |
| Enterococcus faecalis strain ZL | Acid Orange 7 | [157] |
| Pseudomonas aeruginosa strain BCH | Orange 3R (RO3R) | [158] |
| Anoxybacillus pushchinoensis, Anoxybacillus kamchatkensis and Anoxybacillus flavithermus | Reactive Black 5 | [159] |
| Citrobacter sp. CK3 | Reactive Red 180 | [160] |
| Bacillus Fusiformis kmk 5 | Disperse Blue 79 (DB79) and Acid Orange 10 (AO10) | [161] |
| Pseudomonas sp. SUK1 | Red BLI | [162] |
| Brevibacillus sp. | Toluidine Blue dye (TB) | [163] |
| Bacterial strains 1CX and SAD4i | Acid Orange 7 | [164] |
| Pseudomonas luteola | Azo Dye RP2B | [165] |
| Country | Plant Name | Wastewater | Removal Efficiency | Reference |
|---|---|---|---|---|
| Argentina | Typha domingensis | Synthetic runoff effluent | Achieved 95% removal of total phosphorus, soluble reactive phosphorus, NH4+ and NO3− | [205] |
| Australia | Carex appressa | Runoff from low density residential area | The pollutants removal performance was 80% for TSS, 53% for total phosphorus, 17% for total nitrogen | [206] |
| China | Iris pseudacorus | Synthetic secondary effluent | Achieved 89.4% removal of TN in one day retention time | [207] |
| China | Cyperus ustulatus | Domestic wastewater | The average removal efficiency for total microcystin-RR and microcystin-LR were 63.0% and 66.7%, respectively | [208] |
| Indonesia | Chrysopogon zizanioides | Textile wastewater | The average removal rate for chromium was 40%, BOD was 98.47%, and COD was 89.05% | [209] |
| Italy | Phragmites australis, Carex elata, Juncus effusus, Typha latifolia, Chrysopogon zizanioides, Sparganium erectum, and Dactylis glomerata | Resurgent water | The COD, BOD, and TP were reduced by 66%, 52%, and 65%, respectively | [210] |
| New Zealand | Carex virgate | Storm water | The pond with FTWs achieved 41% TSS, 40% particulate zinc, 39% copper, and 16% dissolved copper removal more than pond without FTWs | [111] |
| New Zealand | Carex virgate | Domestic wastewater | The removal rate for both TSS and BOD was more than 93%, TP and dissolve reactive phosphorus removal rate were 44.9% and 29.7% | [211] |
| Pakistan | Phragmites australis | Synthetic diesel oil contaminated water | The hydrocarbons concentration was reduced to 95.8%, COD to 98.6%, BOD to 97.7%, and phenol to 98.9% | [212] |
| Pakistan | Phragmites australis, T. domingensis, Leptochloa fusca and Brachia mutica | Oil contaminated stabilization pit | Reduced COD 97.4%, BOD 98.9%, TDS 82.4%, hydrocarbons 99.1%, and heavy metals 80%. | [108] |
| Pakistan | Brachia mutica and Phragmites australis | Oil field-produced wastewater | The COD, BOD, and oil contents reduced by 93%, 97%, and 97%, respectively | [139] |
| Pakistan | Phragmites australis and Typha domingensis | Textile wastewater | The color, COD, and BOD were reduced by 97%, 87%, and 92%, respectively | [35] |
| Pakistan | Brachiaria mutica | Sewage effluent | The COD, BOD, and oil contents were approximately reduced by 80%, 95%, and 50% | [112] |
| Pakistan | Typha domingensis, Pistia stratiotes and Eichhornia crassipes | Textile effluent | The average reduction rate for color, COD, and BOD was 57%, 72%, and 78%, respectively | [138] |
| Pakistan | Phragmites australis, T. domingensis, Leptochloa fusca and Brachia mutica | Oil contaminated stabilization pit | The COD, BOD, and TDS contents were reduced by 79%, 88%, and 65% | [213] |
| Sri Lanka | Eichhornia crassipes | Sewage water | The removal rate was 74.8% for TP and 55.8% for TN | [214] |
| Sri Lanka | Typha angustifolia and Canna iridiflora | Sewage wastewater | Achieved 80% reduction in BOD and NH4+-N, and 40% reduction in NO3−-N | [215] |
| USA | Spartina patens | Synthetic marine aquaculture effluent | The TP concentration was dropped to ranging from 17–40% | [216] |
| USA | Pontederia cordata and Schoenoplectus tabernaemontani | Urban runoff | The TP and TN concentration were dropped to 60% and 40% in treated wastewater | [217] |
| Wastewater | Plant Specie | Inoculated Bacteria | Pollutant Removal | Retention Period | Reference |
|---|---|---|---|---|---|
| River water | Phragmites australis, Typha domingensis, Brachia mutica, Leptochloa fusca | Aeromonas salmonicida, Pseudomonas indoloxydans, Bacillus cerus, Pseudomonas gessardii, and Rhodococcus sp. | Significant reduction in trace metals contents (Fe, Mn, Ni, Pb, and Cr) | 5 weeks | [123] |
| Diesel contaminated water (1%, w/v) | Phragmites australis | Acinetobacter sp. BRRH61, Bacillus megaterium RGR14 32, and Acinetobacter iwoffii AKR1 | 95.8% hydrocarbon, 98.6% chemical oxygen demand (COD), 97.7% biological oxygen demand (BOD), 95.2%, total organic carbon (TOC), 98.9% Phenol removal | 3 months | [212] |
| Textile effluent | Phragmites australis | Acinetobacter junii, Pseudomonas indoloxydans, and Rhodococcus sp. | 97% color, 87% COD, and 92% BOD removal | 8 days | [38] |
| Oil contaminated water | Phragmites australis T. domingensis Leptochloa fusca Brachiaria mutica inoculated with bacteria | Ochrobactrum intermedium R2, Microbacterium oryzae R4, Pseudomonas aeruginosa R25, P. aeruginosa R21, Acinetobacter sp. LCRH81, Klebsiella sp. LCRI-87, Acinetobacter sp. BRSI56, P. aeruginosa BRRI54, Bacillus subtilus LORI66, and Acinetobacter junii TYRH47. | 97.43% COD, 98.83% BOD, 82.4% TDS, 99.1% hydrocarbon content, and 80% heavy metal removal | 18 months | [108] |
| Phenol contaminated water | Typha domingensis | Acinetobacter lwofii ACRH76, Bacillus cereus LORH97, and Pseudomonas sp. LCRH90 | COD was reduced from 1057 to 97 mg/L; BOD5 from 423 to 64 mg/L, and TOC from 359 to 37 mg/L Phenol removal of 0.166 g/m2/day | 15 days | [235] |
| River water | Phragmites australis, Brachia mutica | Aeromonas Salmonicida, Bacillus cerus Pseudomonas indoloxydans, Pseudomonas gessardii, and Rhodococcus sp. | 85.9% COD, 83.3% BOD, and 86.6% TOC reduction, respectively | 96 h | [146] |
| Oil field wastewater | Brachiara mutica and Phragmites australis | Bacillus subtilis LORI66, Klebsiella sp. LCRI87, Acinetobacter Junii TYRH47, Acinetobacter sp. LCRH81 | 97% COD 93%, and 97% BOD reduction, respectively | 42 days | [139] |
| Oil field produced wastewater | Typha domingensis | Bacillus subtilis LORI66, Klebsiella sp. LCRI87, Acinetobacter Junii TYRH47, and Acinetobacter sp. BRSI56 | 95% Hydrocarbon, 90% COD, and 93% BOD content removal | 42 days | [175] |
| Sewage effluent | Brachiaria mutica | Acinetobacter sp. strain BRSI56, Bacillus cereus strain BRSI57, and Bacillus licheniformis strain BRSI58 | Reduction in COD, BOD, Total nitrogen (TN), and phosphate (PO4) | 8 days | [112] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wei, F.; Shahid, M.J.; Alnusairi, G.S.H.; Afzal, M.; Khan, A.; El-Esawi, M.A.; Abbas, Z.; Wei, K.; Zaheer, I.E.; Rizwan, M.; et al. Implementation of Floating Treatment Wetlands for Textile Wastewater Management: A Review. Sustainability 2020, 12, 5801. https://doi.org/10.3390/su12145801
Wei F, Shahid MJ, Alnusairi GSH, Afzal M, Khan A, El-Esawi MA, Abbas Z, Wei K, Zaheer IE, Rizwan M, et al. Implementation of Floating Treatment Wetlands for Textile Wastewater Management: A Review. Sustainability. 2020; 12(14):5801. https://doi.org/10.3390/su12145801
Chicago/Turabian StyleWei, Fan, Munazzam Jawad Shahid, Ghalia S. H. Alnusairi, Muhammad Afzal, Aziz Khan, Mohamed A. El-Esawi, Zohaib Abbas, Kunhua Wei, Ihsan Elahi Zaheer, Muhammad Rizwan, and et al. 2020. "Implementation of Floating Treatment Wetlands for Textile Wastewater Management: A Review" Sustainability 12, no. 14: 5801. https://doi.org/10.3390/su12145801
APA StyleWei, F., Shahid, M. J., Alnusairi, G. S. H., Afzal, M., Khan, A., El-Esawi, M. A., Abbas, Z., Wei, K., Zaheer, I. E., Rizwan, M., & Ali, S. (2020). Implementation of Floating Treatment Wetlands for Textile Wastewater Management: A Review. Sustainability, 12(14), 5801. https://doi.org/10.3390/su12145801








