Biostimulants as a Tool for Improving Environmental Sustainability of Greenhouse Vegetable Crops
Abstract
1. Introduction
2. Materials and Methods
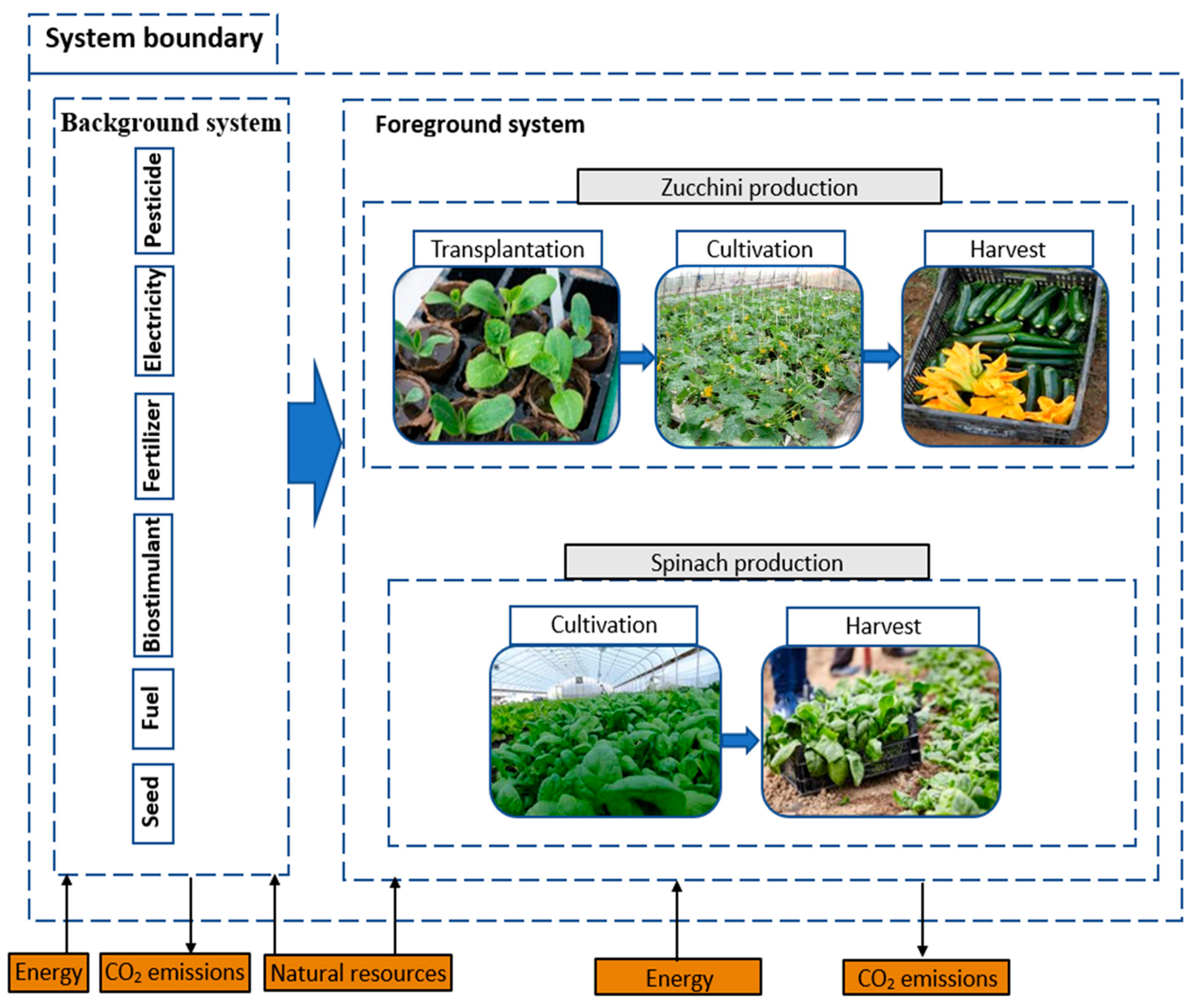
2.1. Life Cycle Assessment and Carbon Footprint
- an assessment of the overall environmental impact of the production chain for crops following a cradle to gate perspective (plant cultivation phase up to harvest) considering both the direct emissions of the different phases of the process and the indirect emissions associated with the production of raw materials as inputs in the production chain;
- an environmental comparison of different ways of managing the production chain that considers 1 ton of cultivated spinach and zucchini squash as a functional unit to identify the most sustainable way.
2.1.1. Inventory Data Collection
2.1.2. Calculation of the Carbon Footprint
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lu, C.; Tia, H. Global nitrogen and phosphorus fertilizer use for agriculture production in the past half century: Shifted hot spots and nutrient imbalance. Earth Syst. Sci. Data 2017, 9, 181–192. [Google Scholar] [CrossRef]
- Tylecote, A. Biotechnology as a new techno-economic paradigm that will help drive the world economy and mitigate climate change. Res. Policy 2018, 48, 858–868. [Google Scholar] [CrossRef]
- James, M.; Oosten, V.; Pepe, O.; De Pascale, S.; Silletti, S.; Maggio, A. The role of biostimulants and bioeffectors as alleviators of abiotic stress in crop plants. Chem. Biol. Technol. Agric. 2017, 4, 5. [Google Scholar]
- Colla, G.; Nardi, S.; Cardarelli, M.; Ertani, A.; Lucini, L.; Canaguier, R.; Rouphael, Y. Protein hydrolysates as biostimulants in horticulture. Sci. Hortic. 2015, 196, 28–38. [Google Scholar] [CrossRef]
- European Parliament. EU Fertilising Products; European Parliament: Luxembourg, 2019. [Google Scholar]
- European Biostimulants Industry Council (EBIC). Towards an Optimal Regulatory Framework for Biostimulants; EBIC: Antwerp, Belgium, 2015. [Google Scholar]
- European Biostimulants Industry Council (EBIC). Provide Fertile Ground for EU’ s Continued Leadership in the Biostimulants Industry: EBIC Urges Policy Makers to Adopt the Draft Fertilising Products Regulation; EBIC: Antwerp, Belgium, 2018. [Google Scholar]
- Rouphael, Y.; Franken, P.; Schneider, C.; Schwarz, D.; Giovannetti, M.; Agnolucci, M.; De Pascale, S.; Bonini, P.; Colla, G. Arbuscular mycorrhizal fungi act as biostimulants in horticultural crops. Sci. Hortic. 2015, 196, 91–108. [Google Scholar] [CrossRef]
- Layek, J.; Das, A.; Ramkrushna, G.I.; Trivedi, K.; Yesuraj, D.; Kubavat, D.; Chandramohan, M.; Agarwal, P.K.; Ghosh, A. Seaweed sap: A sustainable way to improve productivity of maize in North-East India. Int. J. Environ. Stud. 2015, 72, 305–315. [Google Scholar] [CrossRef]
- Layek, J.; Das, A.; Idapuganti, R.G.; Sarkar, D.; Ghosh, A.; Zodape, S.T.; Lal, R.; Yadav, G.S.; Panwar, A.S.; Ngachan, S.; et al. Seaweed extract as organic bio-stimulant improves productivity and quality of rice in eastern Himalayas. J. Appl. Phycol. 2017, 30, 547–558. [Google Scholar] [CrossRef]
- Ronga, D.; Biazzi, E.; Parati, K.; Carminati, D.; Carminati, E.; Tava, A. Microalgal biostimulants and biofertilisers in crop productions. Agronomy 2019, 9, 192. [Google Scholar] [CrossRef]
- Marg, G.B. Foliar nutrient management through Kappaphycus and Gracilaria saps in rice-potato-green gram crop sequence. J. Sci. Ind. Res. 2014, 73, 613–617. [Google Scholar]
- Pramanick, B.; Brahmachari, K.; Ghosh, A.; Zodape, S. Effect of seaweed saps on growth and yield improvement of transplanted rice in old alluvial soil of west Bengal. Bangladesh J. Bot. 2014, 43, 53–58. [Google Scholar] [CrossRef]
- Carillo, P.; Colla, G.; Fusco, G.M.; Aversana, E.D.; El-Nakhel, C.; Giordano, M.; Pannico, A.; Cozzolino, E.; Mori, M.; Reynaud, H.; et al. Morphological and physiological responses induced by protein hydrolysate-based biostimulant and nitrogen rates in greenhouse spinach. Agronomy 2019, 9, 450. [Google Scholar] [CrossRef]
- ISO 14040. Environmental Management—Life Cycle Assessment—Principles and Framework; ISO: Geneva, Switzerland, 2006. [Google Scholar]
- ISO 14044. Environmental Management—Life Cycle Assessment—Requirements and Guidelines; ISO: Geneva, Switzerland, 2006. [Google Scholar]
- Carbon Trust, Department for Environment, Food and Rural Affairs. Guide to PAS 2050: How to Assess the Carbon Footprint of Goods and Services; BSI: London, UK, 2008. [Google Scholar]
- Cardarelli, M.; Rouphael, Y.; Rea, E.; Colla, G. Mitigation of alkaline stress by arbuscular mycorrhiza in zucchini plants grown under mineral and organic fertilization. J. Plant. Nutr. Soil Sci. 2010, 173, 778–787. [Google Scholar] [CrossRef]
- Ribaudo, F. Prontuario di Agricoltura; Hoepli: Milano, Italy, 2017. [Google Scholar]
- Lestrange, M.; Koike, S. Spinach Production in California; Vegetable Research and Information Center, University of California: Oakland, CA, USA, 2011. [Google Scholar]
- Giunta Regionale Della Campania. Available online: http://www.agroqualita.it/_files/sqnpi_disciplinare_campania_it.pdf (accessed on 22 June 2020).
- Rouphael, Y.; Cardarelli, M.; Rea, E.; Battistelli, A.; Colla, G. Comparison of the subirrigation and drip-irrigation systems for greenhouse zucchini squash production using saline and non-saline nutrient solutions. Agric. Water Manag. 2006, 82, 99–117. [Google Scholar] [CrossRef]
- Weidema, B.P.; Bauer, C.; Hischier, R.; Mutel, C.; Nemecek, T.; Reinhard, J.; Vadenbo, C.O.; Wernet, G. Overview and methodology. In Data Quality Guideline for the Ecoinvent Database; The Ecoinvent Centre: St. Gallen, Switzerland, 2013; version 3. [Google Scholar]
- Frischknecht, R.; Jungbluth, N.; Althaus, H.J.; Doka, G.; Dones, R.; Heck, T.; Hellweg, S.; Hischier, R.; Nemecek, T.; Rebitzer, R.; et al. The ecoinvent database: Overview and methodological framework. Int. J. Life Cycle Assess 2005, 10, 3–9. [Google Scholar] [CrossRef]
- Colantoni, A.; Hamedani, S.R. Biostimolanti e sostenibilità ambientale delle colture. In Biostimolanti per Un’agricoltura Sostenibile, 1st ed.; L’informatore Agrario: Verona, Italy, 2019; pp. 143–153. [Google Scholar]
- Colantoni, A.; Recchia, L.; Bernabei, G.; Cardarelli, M.; Rouphael, Y.; Colla, G. Analyzing the environmental impact of chemically-produced protein hydrolysate from leather waste vs. enzymatically-produced protein hydrolysate from legume grains. Agriculture 2017, 7, 62. [Google Scholar] [CrossRef]
- Hara, M. Fertilizer Pellets Made from Composted Livestock Manure; Food and Fertilizer Technology Center: Taiwan, China, 2001. [Google Scholar]
- Fantozzi, F.; Buratti, C. Life cycle assessment of biomass chains: Wood pellet from short rotation coppice using data measured on a real plant. Biomass Bioenergy 2010, 34, 1796–1804. [Google Scholar] [CrossRef]
- Hamedani, S.R.; Colantoni, A.; Gallucci, F.; Salerno, M.; Silvestri, C.; Villarini, M. Comparative energy and environmental analysis of agro-pellet production from orchard woody biomass. Biomass Bioenergy 2019, 129, 105334. [Google Scholar] [CrossRef]
- Hamedani, S.R.; Del Zotto, L.; Bocci, E.; Colantoni, A.; Villarini, M. Eco-efficiency assessment of bioelectricity production from Iranian vineyard biomass gasification. Biomass Bioenergy 2019, 127, 105271. [Google Scholar] [CrossRef]
- Yoder, J.; Galinato, S.; Granatstein, D.; Garcia-Pérez, M. Economic tradeoff between biochar and bio-oil production via pyrolysis. Biomass Bioenergy 2011, 35, 1851–1862. [Google Scholar] [CrossRef]
- Taki, M.; Rohani, A.; Rahmati-Joneidabad, M. Solar thermal simulation and applications in greenhouse. Inf. Process. Agric. 2018, 5, 83–113. [Google Scholar] [CrossRef]
- Arpa, S.D.; Colangelo, G.; Starace, G.; Petrosillo, I.; Bruno, D.E.; Uricchio, V.; Zurlini, G. Heating requirements in greenhouse farming in southern Italy: Evaluation of ground-source heat pump utilization compared to traditional heating systems. Energy Effic. 2016, 9, 1065–1085. [Google Scholar]
- Cuce, E.; Harjunowibowo, D.; Mert, P. Renewable and sustainable energy saving strategies for greenhouse systems: A comprehensive review. Renew Sustain. Energy Rev. 2016, 64, 34–59. [Google Scholar] [CrossRef]
- Yilmaz, G. Seasonal variation of cumulative CO2 emission from a vertisol under apricot orchard in semi-arid southeast Turkey. Pedosphere 2012, 22, 322–332. [Google Scholar] [CrossRef]
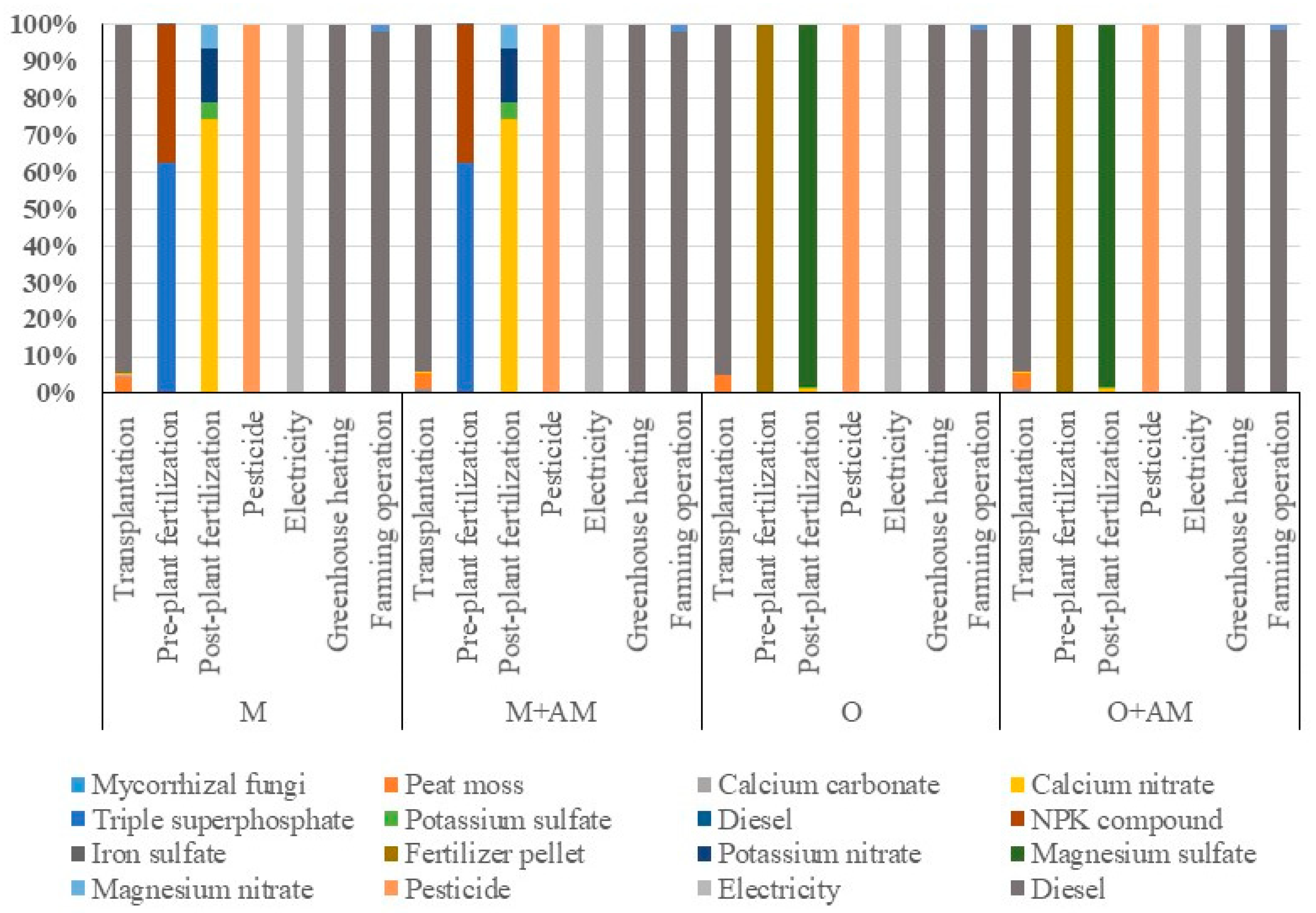
| Items | Unit | Quantity (Unit ha−1) | |||
|---|---|---|---|---|---|
| M | M + AM | O | O + M | ||
| Output to technosphere | |||||
| Fruit yield | kg | 88,634 | 96,024 | 56,696 | 64,838 |
| Input from the environment | |||||
| Water | m3 | 2440 | 2440 | 2440 | 2440 |
| Inputs from technosphere | |||||
| Seedling production | |||||
| Seeds | n | 20,000 | 20,000 | 20,000 | 20,000 |
| Mycorrhizal inoculum | kg | 0 | 7.94 | 0 | 7.94 |
| Peat based substrate | m3 | 0.53 | 0.53 | 0.53 | 0.53 |
| Calcium carbonate | kg | 1 | 1 | 1 | 1 |
| Calcium nitrate | kg | 0.50 | 0.50 | 0.50 | 0.50 |
| Triple superphosphate | kg | 0.15 | 0.15 | 0.15 | 0.15 |
| Potassium sulphate | kg | 0.25 | 0.25 | 0.25 | 0.25 |
| Gasoline | kg | 264.60 | 264.60 | 264.60 | 264.60 |
| Fruit production | |||||
| Pre-plant fertilization | |||||
| Slow-release mineral fertilizer | kg | 360 | 360 | 0 | 0 |
| Triple superphosphate | kg | 353 | 353 | 0 | 0 |
| Iron sulfate | kg | 0.55 | 0.55 | 0 | 0 |
| Pelletized organic-based organic fertilizer | kg | 0 | 0 | 1206 | 1206 |
| Fertigation | |||||
| Calcium nitrate | kg | 991.70 | 991.70 | 0 | 0 |
| Monopotassium phosphate | kg | 66.60 | 66.60 | 0 | 0 |
| Potassium sulfate | kg | 423 | 423 | 0 | 0 |
| Potassium nitrate | kg | 247.70 | 247.70 | 0 | 0 |
| Magnesium nitrate | kg | 468.50 | 468.50 | 0 | 0 |
| Micronutrient mix | kg | 38.80 | 38.80 | 0 | 0 |
| Organic liquid fertilizer | kg | 0 | 0 | 8840.09 | 8840.09 |
| Magnesium sulphate enriched with micronutrients | kg | 0 | 0 | 454.80 | 454.80 |
| Pesticides | |||||
| Flonicamid | kg | 0.40 | 0.40 | 0.40 | 0.40 |
| Deltametrin | L | 0.50 | 0.50 | 0.50 | 0.50 |
| Abamectin | L | 1.20 | 1.20 | 1.20 | 1.20 |
| Penconazole | L | 1.50 | 1.50 | 1.50 | 1.50 |
| Sulfur | kg | 10 | 10 | 10 | 10 |
| Electricity | kWh | 11.10 | 11.10 | 11.10 | 11.10 |
| Diesel | kg | 46,512 | 46,513 | 46,524 | 46,527 |
| Lubricant | kg | 3.50 | 3.50 | 3.50 | 3.50 |
| Output to environment | |||||
| Emissions to air 2 | |||||
| CO2 | kg | 193,491.70 | 193,495.82 | 193,541.58 | 193,554.06 |
| CH4 | kg | 23.25 | 23.25 | 23.26 | 23.26 |
| N2O | kg | 1.39 | 1.39 | 1.39 | 1.39 |
| Items | Unit | Quantity (Unit ha−1) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| N0 | N0 + B | N15 | N15 + B | N30 | N30 + B | N45 | N45 + B | ||
| Output to technosphere | |||||||||
| Leaf yield | kg | 4780 | 6370 | 8420 | 10,529 | 10,290 | 11,970 | 11,950 | 12,900 |
| Input from the environment | |||||||||
| Water | m3 | 620 | 620 | 620 | 620 | 620 | 620 | 620 | 620 |
| Inputs from technosphere | |||||||||
| Seeds | kg | 167 | 167 | 167 | 167 | 167 | 167 | 167 | 167 |
| Mineral fertilizer | |||||||||
| Ammonium nitrate | kg | 0 | 0 | 44 | 44 | 88 | 88 | 132 | 132 |
| Biostimulant | |||||||||
| Protein hydrolysate | kg | 0 | 8.5 | 0 | 8.5 | 0 | 8.5 | 0 | 8.5 |
| Pesticides | |||||||||
| Pyrethrin | kg | 0.96 | 0.96 | 0.96 | 0.96 | 0.96 | 0.96 | 0.96 | 0.96 |
| Copper oxychloride | kg | 7 | 7 | 7 | 7 | 7 | 7 | 7 | 7 |
| Electricity | kWh | 210 | 210 | 210 | 210 | 210 | 210 | 210 | 210 |
| Diesel | kg | 164 | 166 | 169 | 171 | 171 | 173 | 173 | 174 |
| Lubricant | kg | 5.2 | 5.2 | 5.2 | 5.2 | 5.2 | 5.2 | 5.2 | 5.2 |
| Output to environment | |||||||||
| Emissions to air 2 | |||||||||
| CO2 | kg | 682.24 | 690.56 | 703.04 | 711.36 | 711.36 | 719.68 | 719.68 | 719.68 |
| CH4 | kg | 0.08 | 0.08 | 0.08 | 0.08 | 0.08 | 0.08 | 0.08 | 0.08 |
| N2O | kg | 0.005 | 0.005 | 0.005 | 0.005 | 0.005 | 0.005 | 0.005 | 0.005 |
| Input | kg CO2 eq t−1 | |||
|---|---|---|---|---|
| M | M + AM | O | O + AM | |
| Nursery transplant production | 16.30 | 15.11 | 25.39 | 22.38 |
| Pre-plant fertilization | 9.91 | 9.14 | 2.63 | 2.30 |
| Post-plant fertilization | 45.56 | 42.05 | 1.96 | 1.71 |
| Pesticides | 1.38 | 1.28 | 2.16 | 1.89 |
| Electricity | 0.05 | 0.04 | 0.08 | 0.07 |
| Diesel | 2465.80 | 2277.18 | 3848.1 | 3366.78 |
| Total | 2539.00 | 2344.80 | 3880.32 | 3395.13 |
| Input | kg CO2 eq t−1 | |||||||
|---|---|---|---|---|---|---|---|---|
| N0 | N0 + B | N15 | N15 + B | N30 | N30 + B | N45 | N45 + B | |
| Seeds | 6.30 | 4.72 | 3.57 | 2.86 | 2.97 | 2.51 | 2.52 | 2.33 |
| Biostimulant | 0 | 0.18 | 0 | 0.11 | 0 | 0.09 | 0 | 0.09 |
| Ammonium nitrate | 0 | 0 | 41.32 | 33.04 | 67.63 | 58.14 | 87.33 | 80.92 |
| Pesticides | 9.64 | 7.14 | 5.40 | 4.32 | 4.42 | 3.80 | 3.80 | 3.52 |
| Electricity | 18.78 | 14.09 | 10.66 | 8.53 | 8.72 | 7.50 | 7.51 | 6.96 |
| Diesel | 160.30 | 121.94 | 93.77 | 75.99 | 77.64 | 67.62 | 67.63 | 63.11 |
| Total | 195.02 | 148.07 | 154.72 | 124.85 | 161.38 | 139.66 | 168.79 | 156.93 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rajabi Hamedani, S.; Rouphael, Y.; Colla, G.; Colantoni, A.; Cardarelli, M. Biostimulants as a Tool for Improving Environmental Sustainability of Greenhouse Vegetable Crops. Sustainability 2020, 12, 5101. https://doi.org/10.3390/su12125101
Rajabi Hamedani S, Rouphael Y, Colla G, Colantoni A, Cardarelli M. Biostimulants as a Tool for Improving Environmental Sustainability of Greenhouse Vegetable Crops. Sustainability. 2020; 12(12):5101. https://doi.org/10.3390/su12125101
Chicago/Turabian StyleRajabi Hamedani, Sara, Youssef Rouphael, Giuseppe Colla, Andrea Colantoni, and Mariateresa Cardarelli. 2020. "Biostimulants as a Tool for Improving Environmental Sustainability of Greenhouse Vegetable Crops" Sustainability 12, no. 12: 5101. https://doi.org/10.3390/su12125101
APA StyleRajabi Hamedani, S., Rouphael, Y., Colla, G., Colantoni, A., & Cardarelli, M. (2020). Biostimulants as a Tool for Improving Environmental Sustainability of Greenhouse Vegetable Crops. Sustainability, 12(12), 5101. https://doi.org/10.3390/su12125101









