From Rare to Neglected Diseases: A Sustainable and Inclusive Healthcare Perspective for Reframing the Orphan Drugs Issue
Abstract
1. Introduction
2. Orphan Drugs: Definitions, Facts, and Scope of the Issue
- Products intended to treat rare diseases: developed to treat patients suffering from serious diseases for which no treatments are available or satisfactory. The number of such rare diseases is estimated to be between 4000 and 5000 worldwide.
- Products withdrawn from the market for economic or therapeutic reasons: diseases for which no satisfactory treatment is available.
- Products that have not been developed: this is the case of products derived from a research process that cannot be patented or of products that concern important but unprofitable markets (as in the case of third-world countries).
3. Materials and Methods
4. Results
4.1. An Overview of Key Regulatory Approaches to Orphan Drugs
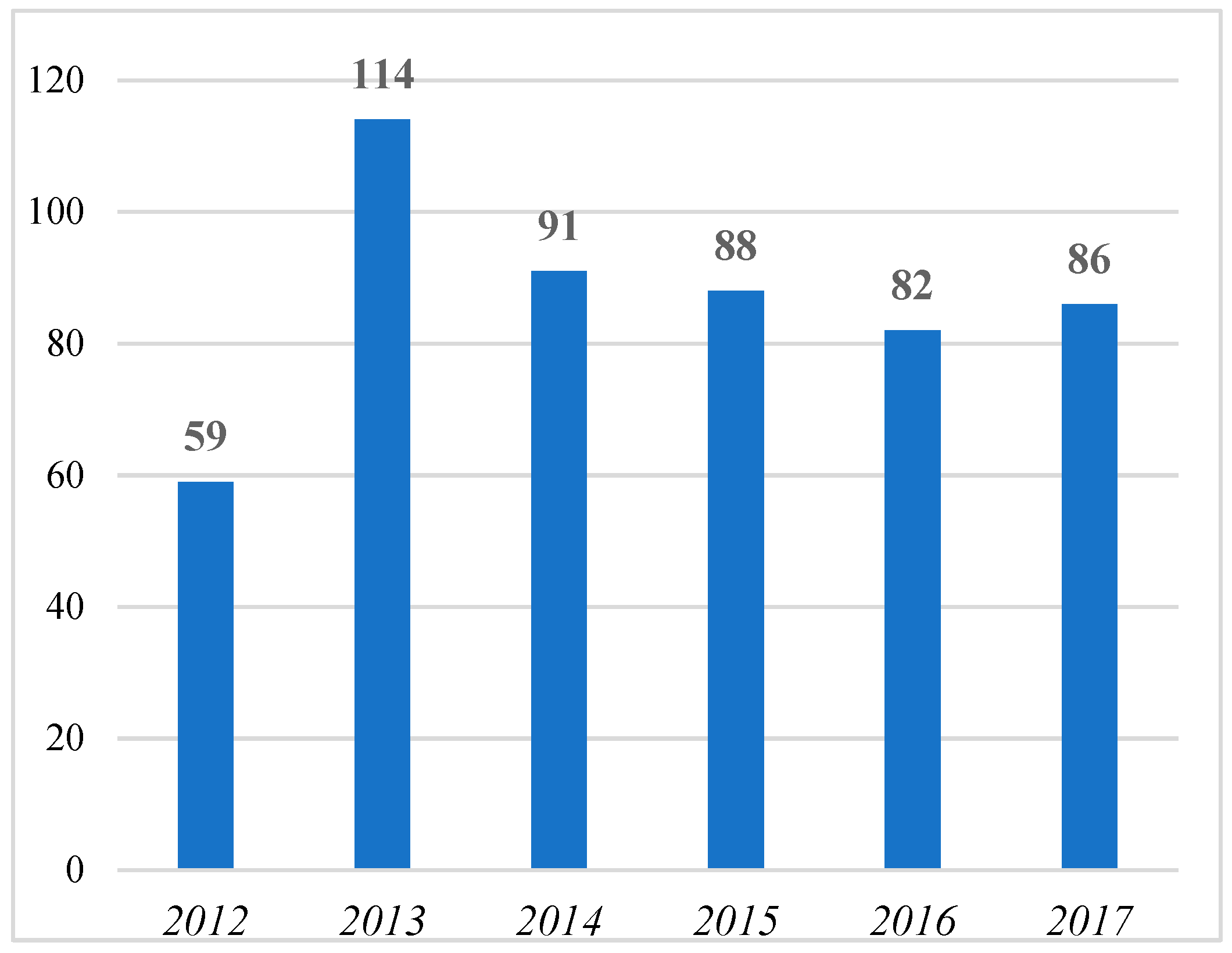
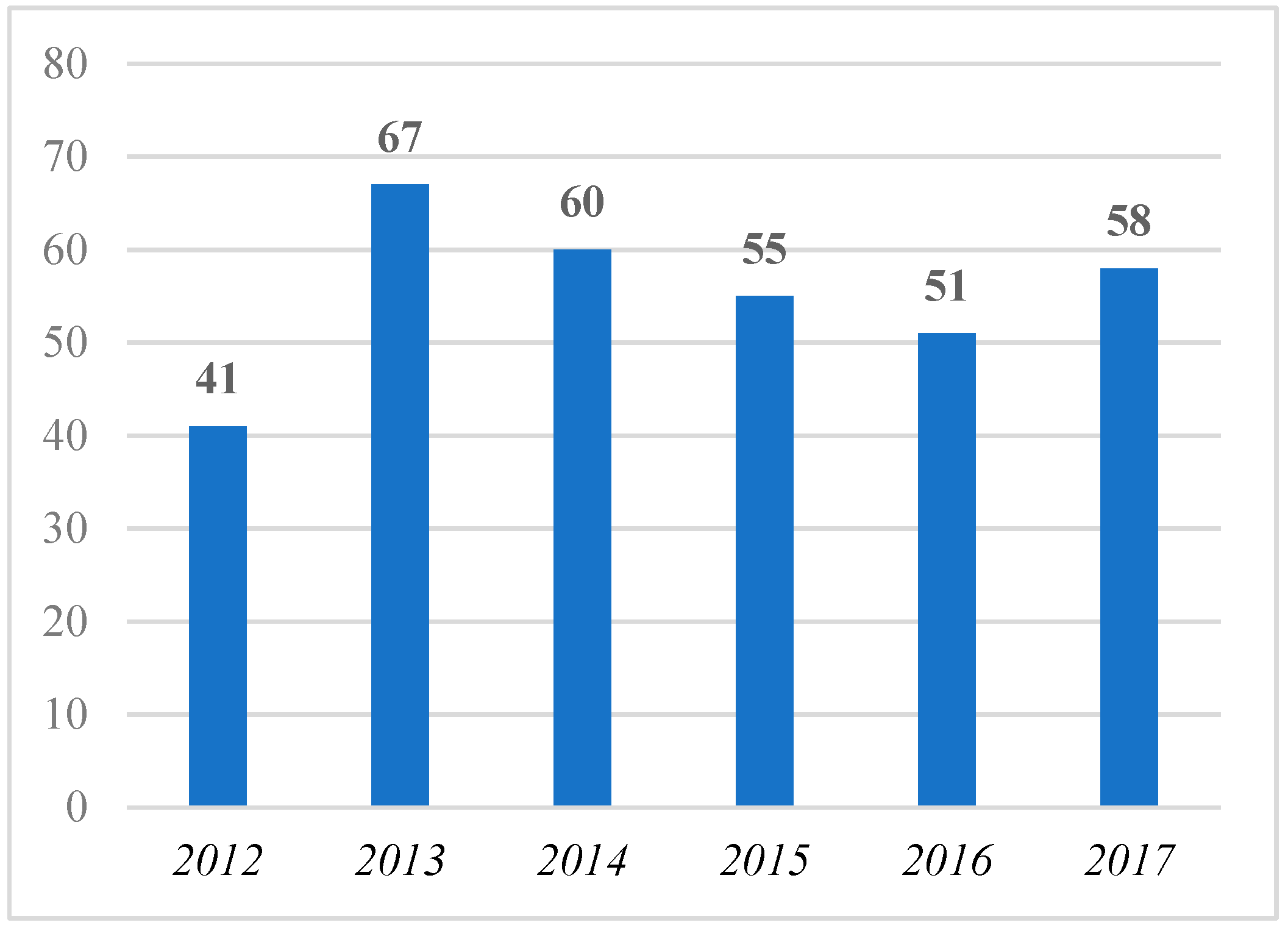
4.2. Results of the Brief Review of the Orphan Drugs Literature
- Three journals (“Chinese Pharmaceutical Journal”, “Expert Opinion on Orphan Drugs”, and “Nature Reviews Drug Discovery”) published at least one contribution with “orphan dru*” in its title for five of the six investigated years;
- Eight journals (“BioWorld Insight”, “Chain Drug Review”, “Deutsche Apotheker Zeitung”, “Drug Discovery Today”, “Manufacturing Chemist”, “Pharmazeutische Zeitung”, “PLoS ONE”, and “Worldwide Biotech”) published at least one contribution with “orphan dru*” in its title for four of the six investigated years;
- 13 journals published at least one contribution with “orphan dru*” in its title for three of the six investigated years;
- 33 journals published at least one contribution with “orphan dru*” in its title for two of the six investigated years;
- 166 journals published at least one contribution with “orphan dru*” in its title for only one of the six investigated years.
- 27 journals are interested in the Business, Law, and Economics fields;
- 25 journals are interested in the Health Policy field;
- Seven journals are interested in the Innovation and Technology fields;
- 165 journals are interested in the Medicine and Pharmacy fields;
- One journal is interested in the Science field.
5. Discussion
5.1. Key Traits of the Main Regulatory Approaches to Orphan Drugs
- Africa and Oceania, where there is no clear approach to orphan drugs and rare diseases issues. Few incentives are given to companies interested in contributing to the research on orphan diseases, and low attention is paid to the topic of orphan diseases from a social perspective [42].
- Europe, where the topics of orphan drugs and rare diseases are debated from both economic and social perspectives. Several guidelines are proposed to face problems; multiple strategies are planned to support pharmaceutical and health companies’ involvement in defining shared approaches, and several activities are organized to promote bottom-up involvements of citizens and local organizations [71].
- Asia, where the societal view seems to prevail. Several incentives are given to companies interested in contributing to the orphan drugs issue, but their application is always linked to the possibility for the population to obtain more effective health products and services [33].
- America, where the main guidelines and strategies regarding the orphan drugs and rare diseases issue are defined more by considering the view of health and pharmaceutical companies. There are few activities to increase citizen participation and to evaluate the sustainability of companies’ strategies [72].
- A dominant pure cost-effectiveness analysis in the evaluation of orphan drugs and diseases [74]. As Aronson argues [9], the main criterion currently used by the National Institute for Health and Clinical Excellence (NICE) for the approval of an orphan drug is its cost. In several countries, like the United Kingdom, recognition as an orphan drug and the related advantages for pharmaceutical companies are not implemented unless their costs are below £30,000 per Quality Adjusted Life Year (QALY).
- Insufficient success in implementing (economic) incentive mechanisms to support research, development, and marketing of orphan drugs. In the USA, for example, after the introduction of the regulation, from 1983 to 2002, about 1100 drugs and biological products were proposed as orphan products, but only 231 were approved [9].
- Millions of people all over the world are reported to be affected by rare diseases with no access to potentially available treatments.
5.2. Main Literature Perspectives on the Orphan Drugs Problem
- The economic one, which “requires that a business have a social responsibility to manufacture products and offer services that society demands and to sell them at a profit”.
- The legal one, which requires that a business “remain economically viable while complying with the laws and regulations of the sovereign nation”.
- The ethical one, which “involves a moral obligation to embrace those activities and practices that are expected, or prohibited, by society even though they may not be codified into law”.
- The discretionary one, which requires that “business be a good corporate citizen by contributing to the overall well-being of the community through philanthropic activities”.
- The high price of orphan drugs that “raises the question as to the extent to which high prices are indeed a fair reflection of the costs incurred by the industry rather than serving to generate profits for the industry”.
- The evidence that “some orphan drugs do not require a high level of investment to bring the drug to market”.
- The fact that “the lack of economic viability can be questioned for certain orphan drugs that have proved to be effective against multiple (sometimes non-orphan) diseases and, thus, target a larger number of patients”.
5.3. From Rare to Neglected Diseases: A Perspective Change towards a Sustainable and Inclusive Healthcare View of the Orphan Drugs Issue
- An economic approach where equality is considered with reference to the total members of a community without differences in the light of individual conditions. In this approach, inclusiveness is not considered a priority, while attention is focused on approaches and strategies for reducing costs and increasing effectiveness.
- A disease-based approach (I) in which the equality is evaluated with reference to each category of patient or disease, while inclusiveness is not considered as a relevant pillar. In this approach, healthcare management is differentiated on the basis of the patients’ category or disease under evaluation.
- A disease-based approach (II) in which equality is defined without considering the differences between the members of a community, and inclusiveness is considered a key priority of the healthcare system. In such a perspective, health services and processes are ensured to all the members of a community in the same way. This produces a high level of satisfaction among the patients that do not require personalized treatments and low level of satisfaction among the patients with needs that are “different from the average”, as in the case of orphan diseases.
- An equal and inclusive approach where equality is considered in the light of the specific conditions of patients, while inclusiveness represents a key pillar of healthcare. This perspective incorporates the basic principles of sustainability because it underlines the need to recognize the substantial differences between individuals, proposing personalized health treatments to ensure healthy lives and well-being for all.
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Robinson, S.; Williams, I.; Dickinson, H.; Freeman, T.; Rumbold, B. Priority-setting and rationing in healthcare: Evidence from the English experience. Soc. Sci. Med. 2012, 75, 2386–2393. [Google Scholar] [CrossRef] [PubMed]
- Divino, V.; DeKoven, M.; Kleinrock, M.; Wade, R.L.; Kaura, S. Orphan drug expenditures in the United States: A historical and prospective analysis, 2007–18. Health Aff. 2016, 35, 1588–1594. [Google Scholar] [CrossRef] [PubMed]
- Drummond, M.F.; Wilson, D.A.; Kanavos, P.; Ubel, P.; Rovira, J. Assessing the economic challenges posed by orphan drugs. Int. J. Technol. Assess. Health Care 2007, 23, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Genetic and Rare Diseases Information Center. Neglected Diseases. Available online: https://rarediseases.info.nih.gov/files/Neglected_Diseases_FAQs.pdf (accessed on 21 February 2019).
- Orphanet. Available online: https://www.orpha.net/consor/cgi-bin/Education_AboutOrphanDrugs.php?lng=EN (accessed on 17 December 2018).
- Eurodis. Available online: https://www.eurordis.org/about-rare-diseases (accessed on 12 January 2019).
- Schieppati, A.; Henter, J.I.; Daina, E.; Aperia, A. Why rare diseases are an important medical and social issue. Lancet 2008, 371, 2039–2041. [Google Scholar] [CrossRef]
- Lavandeira, A. Orphan drugs: Legal aspects, current situation. Haemophilia 2002, 8, 194–198. [Google Scholar] [CrossRef] [PubMed]
- Aronson, J.K. Rare diseases and orphan drugs. Br. J. Clin. Pharmacol. 2006, 61, 243–245. [Google Scholar] [CrossRef] [PubMed]
- Joppi, R.; Garattini, S. Orphan drugs, orphan diseases. The first decade of orphan drug legislation in the EU. Eur. J. Clin. Pharmacol. 2013, 69, 1009–1024. [Google Scholar] [CrossRef] [PubMed]
- Griggs, R.C.; Batshaw, M.; Dunkle, M.; Gopal-Srivastava, R.; Kaye, E.; Krischer, J.; Merkel, P.A. Clinical research for rare disease: Opportunities, challenges, and solutions. Mol. Genet. Metab. 2009, 96, 20–26. [Google Scholar] [CrossRef] [PubMed]
- Haffner, M.E.; Whitley, J.; Moses, M. Two decades of orphan product development. Nat. Rev. Drug Discov. 2002, 1, 821–825. [Google Scholar] [CrossRef] [PubMed]
- Franco, P. Orphan drugs: The regulatory environment. Drug Discov. Today 2013, 18, 163–172. [Google Scholar] [CrossRef] [PubMed]
- Khanna, I. Drug discovery in pharmaceutical industry: Productivity challenges and trends. Drug Discov. Today 2012, 17, 1088–1102. [Google Scholar] [CrossRef] [PubMed]
- Hsia, C.Y.; Wu, C.W.; Lui, W.Y. Heterotopic pancreas: A difficult diagnosis. J. Clin. Gastroenterol. 1999, 28, 144–147. [Google Scholar] [CrossRef] [PubMed]
- Slattery, W.H.; Schwartz, M.S.; Fisher, L.M.; Oppenheimer, M. Acoustic neuroma surgical cost and outcome by hospital volume in California. Otolaryngol. Head Neck Surg. 2004, 130, 726–735. [Google Scholar] [CrossRef] [PubMed]
- Nitschke, Y.; Rutsch, F. Genetics in arterial calcification: Lessons learned from rare diseases. Trends Cardiovasc. Med. 2012, 22, 145–149. [Google Scholar] [CrossRef] [PubMed]
- Knight, A.W.; Senior, T.P. The common problem of rare disease in general practice. Med. J. Aust. 2006, 185, 82–83. [Google Scholar] [PubMed]
- Tambuyzer, E. Rare diseases, orphan drugs and their regulation: Questions and misconceptions. Nat. Rev. Drug Discov. 2010, 9, 921–929. [Google Scholar] [CrossRef] [PubMed]
- Kesselheim, A.S.; Myers, J.A.; Avorn, J. Characteristics of clinical trials to support approval of orphan vs nonorphan drugs for cancer. JAMA 2011, 305, 2320–2326. [Google Scholar] [CrossRef] [PubMed]
- Crompton, H. Mode 2 knowledge production: Evidence from orphan drug networks. Sci. Public Policy 2007, 34, 199–211. [Google Scholar] [CrossRef]
- Meekings, K.N.; Williams, C.S.; Arrowsmith, J.E. Orphan drug development: An economically viable strategy for biopharma R&D. Drug Discov. Today 2012, 17, 660–664. [Google Scholar] [PubMed]
- Mallett, R.; Hagen-Zanker, J.; Slater, R.; Duvendack, M. The benefits and challenges of using systematic reviews in international development research. J. Dev. Eff. 2012, 4, 445–455. [Google Scholar] [CrossRef]
- Khan, K.S.; Kunz, R.; Kleijnen, J.; Antes, G. Five steps to conducting a systematic review. J. R. Soc. Med. 2003, 96, 118–121. [Google Scholar] [CrossRef] [PubMed]
- Fleming, P.S.; Koletsi, D.; Pandis, N. Blinded by PRISMA: Are systematic reviewers focusing on PRISMA and ignoring other guidelines? PLoS ONE 2014, 9, e96407. [Google Scholar] [CrossRef] [PubMed]
- Tranfield, D.; Denyer, D.; Smart, P. Towards a methodology for developing evidence-informed management knowledge by means of systematic review. Br. J. Manag. 2003, 14, 207–222. [Google Scholar] [CrossRef]
- Meho, L.I.; Yang, K. Impact of data sources on citation counts and rankings of LIS faculty: Web of Science versus Scopus and Google Scholar. J. Am. Soc. Inf. Sci. Technol. 2007, 58, 2105–2125. [Google Scholar] [CrossRef]
- Fu, L.; Thomes, C. Implementing discipline-specific searches in EBSCO Discovery Service. New Libr. World 2014, 115, 102–115. [Google Scholar] [CrossRef]
- Wellman-Labadie, O.; Zhou, Y. The US Orphan Drug Act: Rare disease research stimulator or commercial opportunity? Health Policy 2010, 95, 216–228. [Google Scholar] [CrossRef] [PubMed]
- Berndt, E.R.; Cockburn, I.M. The hidden cost of low prices: Limited access to new drugs in India. Health Aff. 2014, 33, 1567–1575. [Google Scholar] [CrossRef] [PubMed]
- McCaughan, M. Pricing Orphan Drugs. Health Aff. 2017. [Google Scholar] [CrossRef]
- Sharma, A.; Jacob, A.; Tandon, M.; Kumar, D. Orphan drug: Development trends and strategies. J. Pharm. Bioallied Sci. 2010, 2, 290–299. [Google Scholar] [CrossRef] [PubMed]
- Song, P.; Gao, J.; Inagaki, Y.; Kokudo, N.; Tang, W. Rare diseases, orphan drugs, and their regulation in Asia: Current status and future perspectives. Intractable Rare Dis. Res. 2012, 1, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Office of Inspector General. United States Department of Health and Human Services. The Orphan Drug Act Implementation and Impact. 2001. Available online: http://oig.hhs.gov/oei/reports/oei-09-00-00380.pdf (accessed on 23 November 2018).
- European Parliament Regulation (EC) no 141/2000 of the European Parliament and of the council of 16 December 1999 on Orphan Medicinal Products. 2000. Available online: https://ec.europa.eu/health/sites/health/files/files/eudralex/vol-1/reg_2000_141_cons-2009-07/reg_2000_141_cons-2009-07_en.pdf (accessed on 13 December 2018).
- Ministry of Health, Labour, and Welfare. Designated Intractable Disease. Available online: https://www.mhlw.go.jp/stf/seisakunitsuite/bunya/0000084783.html (accessed on 13 December 2018).
- Australian Government Therapeutic Goods Regulations 1990. Statutory Rules No. 394, 1990 Made under the Therapeutic Goods Act 1989. 1990. Available online: https://www.legislation.gov.au/Details/F2018C00897 (accessed on 13 December 2018).
- India Drug Manufacturers’ Association. Available online: https://www.idma-assn.org/index.html (accessed on 1 February 2019).
- Law & Regulations database of the Republic of China. Takumi Disease Prevention and Medicine Law. 2014. Available online: https://law.moj.gov.tw/LawClass/LawAll.aspx?PCode=L0030003 (accessed on 13 December 2018).
- Ministry of Government Legislation. Pharmaceutical Affairs Act. 2011. Available online: www.moleg.go.kr/FileDownload.mo?flSeq=39483 (accessed on 13 December 2018).
- The Hong Kong Alliance for Rare Diseases, What Is Rare Disease? Available online: http://www.hkard.org/index/about-rare-disease (accessed on 13 December 2018).
- Hemphill, T.A. Extraordinary pricing of orphan drugs: Is it a socially responsible strategy for the US pharmaceutical industry? J. Bus. Ethics 2010, 94, 225–242. [Google Scholar] [CrossRef]
- Scherer, F.M. Pricing, profits, and technological progress in the pharmaceutical industry. J. Econ. Perspect. 1993, 7, 97–115. [Google Scholar] [CrossRef]
- McGuire, J.; Jabon, E.N.; Faseruk, A. Financial and economic implications of orphan drugs the Canadian economy in perspective. J. Financ. Manag. Anal. 2014, 27, 1–13. [Google Scholar]
- Paulden, M.; Stafinski, T.; Menon, D.; McCabe, C. Value-based reimbursement decisions for orphan drugs: A scoping review and decision framework. Pharmacoeconomics 2015, 33, 255–269. [Google Scholar] [CrossRef] [PubMed]
- Maresova, P.; Klimova, B.; Kuca, K. Financial and legislative aspects of drug development of orphan diseases on the European market–a systematic review. Appl. Econ. 2016, 48, 2562–2570. [Google Scholar] [CrossRef]
- Issa, A.M. Ethical perspectives on pharmacogenomic profiling in the drug development process. Nat. Rev. Drug Discov. 2002, 1, 300–308. [Google Scholar] [CrossRef] [PubMed]
- Cheng, A.; Xie, Z. Challenges in orphan drug development and regulatory policy in China. Orphanet J. Rare Dis. 2017, 12, 13–27. [Google Scholar] [CrossRef] [PubMed]
- Minn, M. Development of Orphan Drugs under European Regulatory Incentives and Patent Protection. Eur. J. Health Law 2017, 24, 239–263. [Google Scholar] [CrossRef]
- Vassal, G.; Kearns, P.; Blanc, P.; Scobie, N.; Heenen, D.; Pearson, A. Orphan Drug Regulation: A missed opportunity for children and adolescents with cancer. Eur. J. Cancer 2017, 84, 149–158. [Google Scholar] [CrossRef] [PubMed]
- Hughes, D.A.; Tunnage, B.; Yeo, S.T. Drugs for exceptionally rare diseases: Do they deserve special status for funding? QJM 2005, 98, 829–836. [Google Scholar] [CrossRef] [PubMed]
- Herder, M. Orphan drug incentives in the pharmacogenomic context: Policy responses in the USA and Canada. J. Law Biosci. 2016, 3, 158–172. [Google Scholar] [CrossRef] [PubMed]
- Le, T.T. Incentivizing Orphan Product Development: United States Food and Drug Administration Orphan Incentive Programs. In Rare Diseases Epidemiology: Update and Overview; Springer: Cham, Switzerland, 2017; pp. 183–196. [Google Scholar]
- Hughes-Wilson, W.; Palma, A.; Schuurman, A.; Simoens, S. Paying for the Orphan Drug System: Break or bend? Is it time for a new evaluation system for payers in Europe to take account of new rare disease treatments? Orphanet J. Rare Dis. 2012, 7, 74–82. [Google Scholar] [CrossRef] [PubMed]
- Gammie, T.; Lu, C.Y.; Babar, Z.U.D. Access to orphan drugs: A comprehensive review of legislations, regulations and policies in 35 countries. PLoS ONE 2015, 10, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Stockklausner, C.; Lampert, A.; Hoffmann, G.F.; Ries, M. Novel treatments for rare cancers: The US orphan drug act is delivering—A cross-sectional analysis. Oncologist 2016, 21, 487–493. [Google Scholar] [CrossRef] [PubMed]
- Boon, W.; Moors, E. Exploring emerging technologies using metaphors–a study of orphan drugs and pharmacogenomics. Soc. Sci. Med. 2008, 66, 1915–1927. [Google Scholar] [CrossRef] [PubMed]
- Scarpa, M.; Bellettato, C.; Lampe, C. Orphan Drugs. Drug Discovery and Evaluation: Pharmacological Assays; Springer-Verlag: Berlin/Heidelberg, Germany, 2014; pp. 1–20. [Google Scholar]
- Douglas, C.M.; Wilcox, E.; Burgess, M.; Lynd, L.D. Why orphan drug coverage reimbursement decision-making needs patient and public involvement. Health Policy 2015, 119, 588–596. [Google Scholar] [CrossRef] [PubMed]
- Mavris, M.; Le Cam, Y. Involvement of patient organisations in research and development of orphan drugs for rare diseases in Europe. Mol. Syndromol. 2012, 3, 237–243. [Google Scholar] [CrossRef] [PubMed]
- Cui, Y.; Han, J. A proposed definition of rare diseases for China: From the perspective of return on investment in new orphan drugs. Orphanet J. Rare Dis. 2015, 10, 28–40. [Google Scholar] [CrossRef] [PubMed]
- Henrard, S.; Hermans, C. Rare diseases and orphan drugs in Belgium and in the European Union: What is the current situation? Louvain Med 2015, 134, 527–534. [Google Scholar]
- Villa, S.; Compagni, A.; Reich, M.R. Orphan drug legislation: Lessons for neglected tropical diseases. Int. J. Health Plan. Manag. 2009, 24, 27–42. [Google Scholar] [CrossRef] [PubMed]
- Aymé, S.; Kole, A.; Groft, S. Empowerment of patients: Lessons from the rare diseases community. Lancet 2008, 371, 2048–2051. [Google Scholar] [CrossRef]
- López-Bastida, J.; Oliva-Moreno, J.; Linertová, R.; Serrano-Aguilar, P. Social/economic costs and health-related quality of life in patients with rare diseases in Europe. Eur. J. Health Econ. 2016, 17, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Linley, W.G.; Hughes, D.A. Societal views on NICE, cancer drugs fund and value-based pricing criteria for prioritising medicines: A cross-sectional survey of 4118 adults in Great Britain. Health Econ. 2013, 22, 948–964. [Google Scholar] [CrossRef] [PubMed]
- McCabe, C.; Stafinski, T.; Menon, D. Is it time to revisit orphan drug policies? BMJ 2010, 341. [Google Scholar] [CrossRef] [PubMed]
- Grabowski, H.G. Increasing R&D incentives for neglected diseases: Lessons from the Orphan Drug Act. International public goods and transfer of technology under a globalized intellectual property regime. In International Public Goods and Transfer of Technology Under a Globalized Intellectual Property Regime; Maskus, K., Reichman, J., Eds.; Cambridge University: Cambridge, UK, 2005; pp. 457–480. [Google Scholar]
- Gericke, C.A.; Riesberg, A.; Busse, R. Ethical issues in funding orphan drug research and development. J. Med. Ethics 2005, 31, 164–168. [Google Scholar] [CrossRef] [PubMed]
- Morel, C.M.; Serruya, S.J.; Penna, G.O.; Guimarães, R. Co-authorship network analysis: A powerful tool for strategic planning of research, development and capacity building programs on neglected diseases. PLoS Negl. Trop. Dis. 2009, 3, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Michel, M.; Toumi, M. Access to orphan drugs in Europe: Current and future issues. Expert Rev. Pharm. Outcomes Res. 2012, 12, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Braun, M.M.; Farag-El-Massah, S.; Xu, K.; Coté, T.R. Emergence of orphan drugs in the United States: A quantitative assessment of the first 25 years. Nat. Rev. Drug Discov. 2010, 9, 519–522. [Google Scholar] [CrossRef] [PubMed]
- Ehrenberg, J.P.; Ault, S.K. Neglected diseases of neglected populations: Thinking to reshape the determinants of health in Latin America and the Caribbean. BMC Public Health 2005, 5, 119–132. [Google Scholar] [CrossRef] [PubMed]
- Denis, A.; Mergaert, L.; Fostier, C.; Cleemput, I.; Simoens, S. Issues surrounding orphan disease and orphan drug policies in Europe. Appl. Health Econ. Health Policy 2010, 8, 343–350. [Google Scholar] [CrossRef] [PubMed]
- Simoens, S.; Dooms, M. How much is the life of a cancer patient worth? A pharmaco-economic perspective. J. Clin. Pharm. Ther. 2011, 36, 249–256. [Google Scholar] [CrossRef] [PubMed]
- Callahan, D.; Jennings, B. Ethics and public health: Forging a strong relationship. Am. J. Public Health 2002, 92, 169–176. [Google Scholar] [CrossRef] [PubMed]
- Leisinger, K.M. The corporate social responsibility of the pharmaceutical industry: Idealism without illusion and realism without resignation. Bus. Ethics Q. 2005, 15, 577–594. [Google Scholar] [CrossRef] [PubMed]
- Joppi, R.; Bertele, V.; Garattini, S. Orphan drug development is progressing too slowly. Br. J. Clin. Pharmacol. 2006, 61, 355–360. [Google Scholar] [CrossRef] [PubMed]
- Wästfelt, M.; Fadeel, B.; Henter, J.I. A journey of hope: Lessons learned from studies on rare diseases and orphan drugs. J. Intern. Med. 2006, 260, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Seoane-Vazquez, E.; Rodriguez-Monguio, R.; Szeinbach, S.L.; Visaria, J. Incentives for orphan drug research and development in the United States. Orphanet J. Rare Dis. 2008, 3, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Simoens, S. Pricing and reimbursement of orphan drugs: The need for more transparency. Orphanet J. Rare Dis. 2011, 6, 42–50. [Google Scholar] [CrossRef] [PubMed]
- O’sullivan, B.P.; Orenstein, D.M.; Milla, C.E. Pricing for orphan drugs: Will the market bear what society cannot? JAMA 2013, 310, 1343–1344. [Google Scholar] [CrossRef] [PubMed]
- Trouiller, P.; Olliaro, P.; Torreele, E.; Orbinski, J.; Laing, R.; Ford, N. Drug development for neglected diseases: A deficient market and a public-health policy failure. Lancet 2002, 359, 2188–2194. [Google Scholar] [CrossRef]
- Iskrov, G.; Stefanov, R. Post-marketing access to orphan drugs: A critical analysis of health technology assessment and reimbursement decision-making considerations. Orphan Drugs Res. Rev. 2014, 4, 1–9. [Google Scholar] [CrossRef]
- Cohen, J.P.; Felix, A. Are payers treating orphan drugs differently? J. Mark. Access Health Policy 2014, 2, 23513. [Google Scholar] [CrossRef] [PubMed]
- Gulliford, M.; Figueroa-Munoz, J.; Morgan, M.; Hughes, D.; Gibson, B.; Beech, R.; Hudson, M. What does ‘access to health care’ mean? J. Health Serv. Res. Policy 2002, 7, 186–188. [Google Scholar] [CrossRef] [PubMed]
- Diener, E.; Seligman, M.E. Beyond money: Toward an economy of well-being. Psychol. Sci. Public Interest 2004, 5, 1–31. [Google Scholar] [CrossRef] [PubMed]
- Saviano, M.; Caputo, F.; Napoli, B. Addressing the social and economic challenges of Orphan Drugs: A managerial perspective. Int. J. Pharm. Sci. Bus. Manag. 2015, 3, 1–26. [Google Scholar]
- Nwaka, S.; Ridley, R.G. Science & society: Virtual drug discovery and development for neglected diseases through public–private partnerships. Nat. Rev. Drug Discov. 2003, 2, 919–928. [Google Scholar] [PubMed]
- Morel, C.M.; Acharya, T.; Broun, D.; Dangi, A.; Elias, C.; Ganguly, N.K.; Hotez, P.J. Health innovation networks to help developing countries address neglected diseases. Science 2005, 309, 401–404. [Google Scholar] [CrossRef] [PubMed]
- World Health Organizations. World Health Statistics 2014. 2014. Available online: https://www.who.int/features/qa/58/en/ (accessed on 21 November 2018).
- NIH Office of Rare Diseases Research. Neglected Diseases; 2012. Available online: https://rarediseases.info.nih.gov/files/Neglected_Diseases_FAQs.pdf (accessed on 25 November 2018).
- Sverdrup, H.; Svensson, M.G. Defining the concept of sustainability—A matter of systems thinking and applied systems analysis. In Systems Approaches and their Application; Olsson, M.O., Sjöstedt, G., Eds.; Springer: Dordrecht, The Netherlands, 2004; pp. 143–164. [Google Scholar]
- Kuhlman, T.; Farrington, J. What is sustainability? Sustainability 2010, 2, 3436–3448. [Google Scholar] [CrossRef]
- Charron, D.F. Ecosystem approaches to health for a global sustainability agenda. EcoHealth 2012, 9, 256–266. [Google Scholar] [CrossRef] [PubMed]
- Barile, S.; Saviano, M.; Iandolo, F.; Calabrese, M. The viable systems approach and its contribution to the analysis of sustainable business behaviors. Syst. Res. Behav. Sci. 2014, 31, 683–695. [Google Scholar] [CrossRef]
- Carayannis, E.G.; Grigoroudis, E.; Del Giudice, M.; Della Peruta, M.R.; Sindakis, S. An exploration of contemporary organizational artifacts and routines in a sustainable excellence context. J. Knowl. Manag. 2017, 21, 35–56. [Google Scholar] [CrossRef]
- Sustainable Development Goals. Knowledge Platform. Available online: https://sustainabledevelopment.un.org/ (accessed on 15 January 2019).
- McMurray, A. Community Health and Wellness: A Socio-ecological Approach; Elsevier: Chatswood, Australia, 2006. [Google Scholar]
- Travis, P.; Bennett, S.; Haines, A.; Pang, T.; Bhutta, Z.; Hyder, A.A.; Evans, T. Overcoming health-systems constraints to achieve the Millennium Development Goals. Lancet 2004, 364, 900–906. [Google Scholar] [CrossRef]
- Guenther, R.; Vittori, G. Sustainable Healthcare Architecture; John Wiley & Sons: New York, NY, USA, 2008. [Google Scholar]
- Buchanan, D.A.; Fitzgerald, L.; Ketley, D. (Eds.) The Sustainability and Spread of Organizational Change: Modernizing Healthcare; Routledge: London, UK, 2006. [Google Scholar]
- Buffoli, M.; Capolongo, S.; Bottero, M.; Cavagliato, E.; Speranza, S.; Volpatti, L. Sustainable Healthcare: How to assess and improve healthcare structures’ sustainability. Ann Ig 2013, 25, 411–418. [Google Scholar] [PubMed]
- Calabrese, M.; Iandolo, F.; Caputo, F.; Sarno, D. From mechanical to cognitive view: The changes of decision making in business environment. In Social Dynamics in a Systems Perspective; Barile, S., Pellicano, M., Polese, F., Eds.; Springer: Cham, Switzerland, 2018; pp. 223–240. [Google Scholar]
- Tronvoll, B.; Barile, S.; Caputo, F. A systems approach to understanding the philosophical foundation of marketing studies. In Social Dynamics in a Systems Perspective; Barile, S., Pellicano, M., Polese, F., Eds.; Springer: Cham, Switzerland, 2018; pp. 1–18. [Google Scholar]
- Saviano, M.; Bassano, C.; Piciocchi, P.; Di Nauta, P.; Lettieri, M. Monitoring Viability and Sustainability in Healthcare Organizations. Sustainability 2018, 10, 3548. [Google Scholar] [CrossRef]
- Saviano, M.; Caputo, F.; Mueller, J.; Belyaeva, Z. Competing through consonance: A stakeholder engagement view of corporate relational environment. Sinergie 2018, 36, 61–80. [Google Scholar]
- Swanson, R.C.; Cattaneo, A.; Bradley, E.; Chunharas, S.; Atun, R.; Abbas, K.M.; Best, A. Rethinking health systems strengthening: Key systems thinking tools and strategies for transformational change. Health Policy Plan. 2012, 27 (Suppl. 4), iv54–iv61. [Google Scholar] [CrossRef] [PubMed]
- Carlson, R.J. Holism and reductionism as perspectives in medicine and patient care. West. J. Med. 1979, 131, 466–480. [Google Scholar] [PubMed]
- Capolongo, S.; Bottero, M.C.; Lettieri, E.; Buffoli, M.; Bellagarda, A.; Birocchi, M.; Gola, M. Healthcare sustainability challenge. In Improving Sustainability During Hospital Design and Operation; Springer: Cham, Switzerland, 2015; pp. 1–9. [Google Scholar]
- Damschroder, L.J.; Aron, D.C.; Keith, R.E.; Kirsh, S.R.; Alexander, J.A.; Lowery, J.C. Fostering implementation of health services research findings into practice: A consolidated framework for advancing implementation science. Implement. Sci. 2009, 4, 50–65. [Google Scholar] [CrossRef] [PubMed]
- Draborg, E.; Gyrd-Hansen, D.; Poulsen, P.B.; Horder, M. International comparison of the definition and the practical application of health technology assessment. Int. J. Technol. Assess. Health Care 2005, 21, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Tantivess, S. Social and ethical analysis in health technology assessment. J. Med. Assoc. Thail. 2014, 97, S81–S86. [Google Scholar]
- Lehoux, P.; Williams-Jones, B. Mapping the integration of social and ethical issues in health technology assessment. Int. J. Technol. Assess. Health Care 2007, 23, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Papa, A.; Mital, M.; Pisano, P.; Del Giudice, M. E-health and wellbeing monitoring using smart healthcare devices: An empirical investigation. Technol. Forecast. Soc. Chang. 2018. [Google Scholar] [CrossRef]
- Dominici, G.; Yolles, M.; Caputo, F. Decoding the dynamics of value cocreation in consumer tribes: An agency theory approach. Cybern. Syst. 2017, 48, 84–101. [Google Scholar] [CrossRef]
- Porter, M.E. What is value in health care? N. Engl. J. Med. 2010, 363, 2477–2481. [Google Scholar] [CrossRef] [PubMed]
- McKeeargue, M. Budget crises, health, and social welfare programmes. BMJ 2010, 340. [Google Scholar] [CrossRef]
- Del Giudice, M.; Khan, Z.; De Silva, M.; Scuotto, V.; Caputo, F.; Carayannis, E. The microlevel actions undertaken by owner-managers in improving the sustainability practices of cultural and creative small and medium enterprises: A United Kingdom–Italy comparison. J. Organ. Behav. 2017, 38, 1396–1414. [Google Scholar] [CrossRef]
- Caputo, F.; Carrubbo, L.; Sarno, D. The Influence of Cognitive Dimensions on the Consumer-SME Relationship: A Sustainability-Oriented View. Sustainability 2018, 10, 3238. [Google Scholar] [CrossRef]
- Saviano, M.; Barile, S.; Spohrer, J.C.; Caputo, F. A service research contribution to the global challenge of sustainability. J. Serv. Theory Pract. 2017, 27, 951–976. [Google Scholar] [CrossRef]
- Yamey, G.; Shretta, R.; Binka, F.N. The 2030 sustainable development goal for health. BMJ 2014, 349. [Google Scholar] [CrossRef] [PubMed]
- Saviano, M.; Parida, R.; Caputo, F.; Kumar Datta, S. Health care as a worldwide concern. Insights on the Italian and Indian health care systems and PPPs from a VSA perspective. EuroMed J. Bus. 2014, 9, 198–220. [Google Scholar] [CrossRef]
- Caputo, F.; Masucci, A.; Napoli, L. Managing value co-creation in pharmacy. Int. J. Pharm. Healthc. Mark. 2018, 12, 374–390. [Google Scholar] [CrossRef]
- Polese, F.; Carrubbo, L.; Caputo, F.; Sarno, D. Managing Healthcare Service Ecosystems: Abstracting a Sustainability-Based View from Hospitalization at Home (HaH) Practices. Sustainability 2018, 10, 3951. [Google Scholar] [CrossRef]
- Aquino, R.P.; Barile, S.; Caputo, F.; Corrente, M.I.; Grasso, A.; Saviano, M. Salute, farmaci e integratori in una visione sistemica: Vigilanza su prodotti a base di isoflavoni di soia. Rapporto di ricercar; Giappichelli: Torino, Italy, 2018. [Google Scholar]
- Garcés, J.; Ródenas, F.; Sanjosé, V. Towards a new welfare state: The social sustainability principle and health care strategies. Health Policy 2003, 65, 201–215. [Google Scholar]
| Inclusion Criteria |
|---|
|
| Area | Summary |
|---|---|
| United States | Orphan drugs production is motivated by the Orphan Drug Act’s incentives and by the Office of Orphan Products Development’s clinical superiority criteria [34]. |
| Europe | Orphan drugs are considered to be all medical products the pharmaceutical industry would be unwilling to develop under normal market conditions. A condition of not more than 5 affected persons per 10,000 is generally regarded as the appropriate threshold for defining orphan diseases, while the medicinal products considered “orphan” are those useful for treating diseases that affect not more than 5 per 10,000 citizens [35]. |
| Japan | Three conditions must be satisfied to be considered an orphan disease: (1) impact on less than 50,000 citizens in Japan; (2) absence of available health treatment; and (3) availability of a clear product development process that shows the need for a new drug. If all these conditions occur, the Ministry of Health, Labor, and Welfare evaluates case by case the approval for a new orphan drug [36]. |
| Australia | All the drugs intended to treat diseases that affect fewer than 5 in 10,000 individuals in Australia can be considered “orphan” [37]. |
| India | After the request formulated in 2001 by a group of pharmacologists during a conference held by the Indian Drugs Manufacturers Association, the possibility to define a specific regulatory framework for orphan diseases is under evaluation [38]. |
| Taiwan | Taiwan’s Legislative Yuan approved 20 regulated orphan drugs on 9 February. In April 2006, the government of Taiwan identified 159 kinds of rare diseases with 2117 cases confirmed. At present, the Taiwan Foundation for Rare Disorders (TFRD) serves 2252 citizens affected by 191 rare diseases, and in Taiwan, 77 orphan drugs and 40 special nutrients are recognized by the government for the treatment of rare disease [39]. |
| Korea | The Korean Orphan Drug Center manages the processes for approval, incentives, and management of orphan drugs [40]. |
| Hong Kong | There is no clear and shared definition of rare diseases and there are no specific public policies [41]. |
| Area | Key Elements |
|---|---|
| Africa & Oceania |
|
| Europe |
|
| Asia |
|
| America |
|
| Relevance of the Societal Perspective | |||
|---|---|---|---|
| High | Low | ||
| Relevance of the economic perspective | High | e.g., European approach | e.g., American approach |
| Low | e.g., Asian approach | e.g., African and Oceanian approach | |
| Perspectives | Brief Description | Sources Examples |
|---|---|---|
| Technical- scientific | Contributions in this area focus on the way in which orphan drugs can be developed from the chemical, biological, and pharmaceutical points of view. Most of these studies concern the development of new kinds of drugs or the definition of innovative production processes for existing orphan drugs. | [78,79] |
| Economic | Studies provided with reference to the economic area investigate the economic advantages and impacts on companies’ economic performances related to the “market of orphan diseases”. Most contributions that can be classified in this area attempt to define economic risks and opportunities for pharmaceutical companies interested in working in the orphan drugs sector. | [3,80,81,82]. |
| Managerial | The few contributions provided within the managerial and marketing area focus on the levers to improve companies’ efficiency in terms of human resources management, communication, and brand image. | [22,83,84] |
| Inclusiveness | |||
|---|---|---|---|
| Low | High | ||
| Equality | Horizontal | Economic approach | Disease-based approach (I) |
| Vertical | Disease-based approach (II) | Equal and Inclusive approach | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Saviano, M.; Barile, S.; Caputo, F.; Lettieri, M.; Zanda, S. From Rare to Neglected Diseases: A Sustainable and Inclusive Healthcare Perspective for Reframing the Orphan Drugs Issue. Sustainability 2019, 11, 1289. https://doi.org/10.3390/su11051289
Saviano M, Barile S, Caputo F, Lettieri M, Zanda S. From Rare to Neglected Diseases: A Sustainable and Inclusive Healthcare Perspective for Reframing the Orphan Drugs Issue. Sustainability. 2019; 11(5):1289. https://doi.org/10.3390/su11051289
Chicago/Turabian StyleSaviano, Marialuisa, Sergio Barile, Francesco Caputo, Mattia Lettieri, and Stefania Zanda. 2019. "From Rare to Neglected Diseases: A Sustainable and Inclusive Healthcare Perspective for Reframing the Orphan Drugs Issue" Sustainability 11, no. 5: 1289. https://doi.org/10.3390/su11051289
APA StyleSaviano, M., Barile, S., Caputo, F., Lettieri, M., & Zanda, S. (2019). From Rare to Neglected Diseases: A Sustainable and Inclusive Healthcare Perspective for Reframing the Orphan Drugs Issue. Sustainability, 11(5), 1289. https://doi.org/10.3390/su11051289






