An Experimental Brackish Aquaponic System Using Juvenile Gilthead Sea Bream (Sparus aurata) and Rock Samphire (Crithmum maritimum)
Abstract
1. Introduction
2. Materials and Methods
2.1. Plants and Fish
2.2. Experimental Set-Up and Operation
2.3. Abiotic Factors
- HRT (min) = (surface area water x depth x porosity of gravel trough/flow rate)
- SSA (m2/m3) = Surface area of filter media/volume of the filter media
- Vmedia (m3) = surface area of the filter media/SSA
- r = volume of recycled water/volume of the system
2.4. Biological Analyses
- SGR (%/day) = ((ln Wfin − lnWin)/Δt) × 100
- WG (gr) = Wfin − Win
- FCR = Food offered (gr)/weight gain (gr)
- DFI (%/d) = 100 × ((food offered/weight gain)/feeding days)
- Condition factor (K) = (W × L−3) × 100 where W is the body weight of the fish (gr) and L is the total length of the fish (cm)
- S = ((final number of fish − initial number of fish)/initial number of fish) × 100
- Plant Growth (cm/d) = height of plant/day
2.5. Statistical Analysis
3. Results
3.1. Abiotic Factors
3.2. Biotic Factors
4. Discussion
4.1. Abiotic Environment
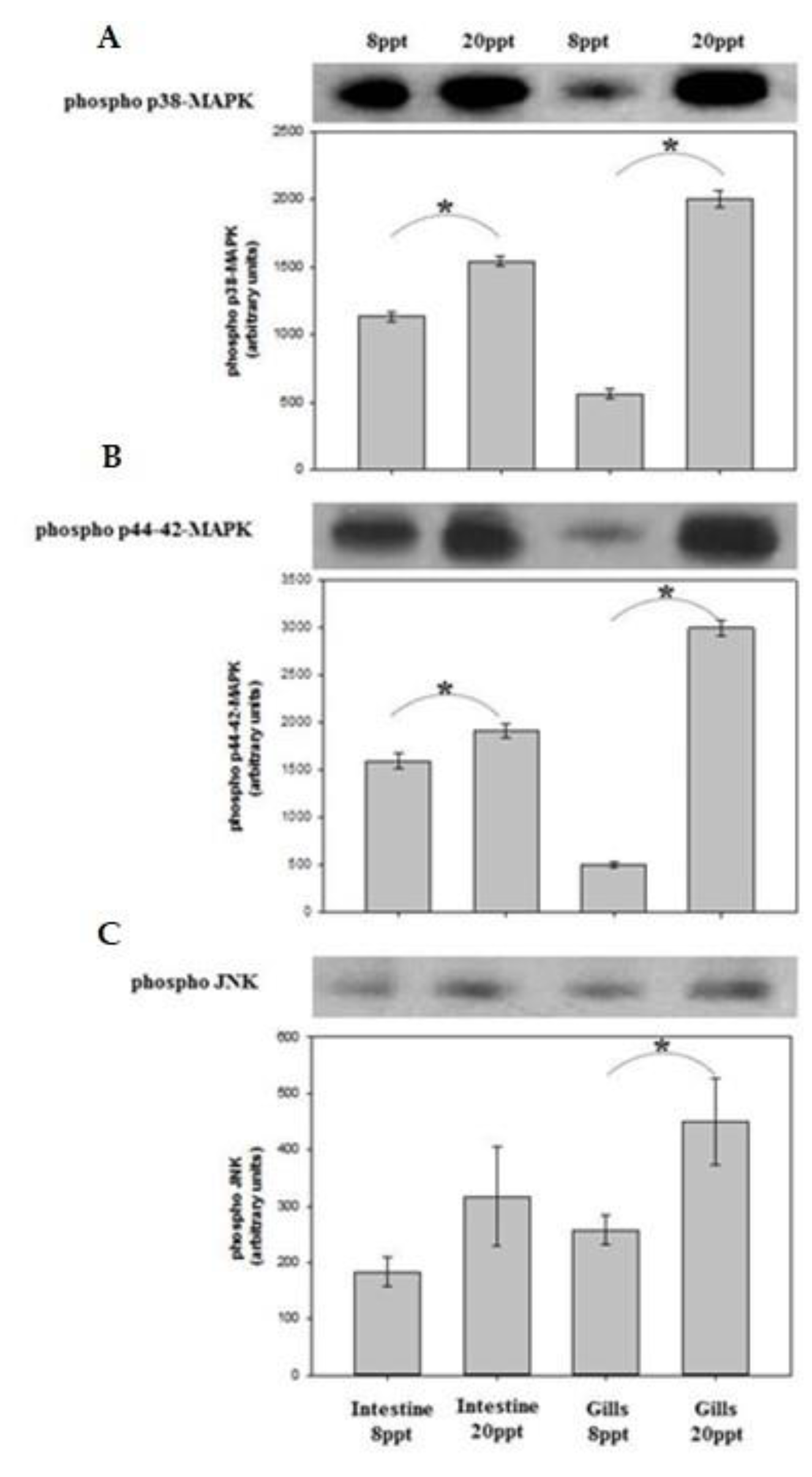
4.2. Biological Elements
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Love, D.C.; Fry, J.P.; Li, X.; Hill, E.S.; Genello, L.; Semmnes, K.; Thompson, R.E. Commercial aquaponics production and profitability: Findings from an international survey. Aquaculture 2015, 435, 67–74. [Google Scholar] [CrossRef]
- Thorarinsdottir, R. Introduction—What is aquaponics. In Aquapoincs Guidelines; Haskolaprent: Reykjavik, Iceland, 2015; Chapter 1; p. 58. ISBN 978-9935-9283-1-3. [Google Scholar]
- Somerville, C.; Cohen, M.; Pantanella, E.; Stankus, A.; Lovatelli, A. Small-Scale Aquaponic Food Production—Integrated Fish and Plant Farming; Fisheries and Aquaclture Technical Paper, No 589; FAO: Rome, Italy, 2014; 262p, ISSN 2070-7010. Available online: http://www.fao.org/3/a-i4021e.pdf (accessed on 2 September 2019).
- Tyson, R.C.; Treadwell, D.D.; Simonne, E.H. Opportunities and challenges to sustainability in aquaponic systems. HortTechnology 2011, 21, 6–13. [Google Scholar] [CrossRef]
- Fronte, B.; Galliano, G.; Bibbiani, C. From freshwater to marine aquaponic: New opportunities for marine fish species production. In Proceedings of the 4th Conference VIVUS with International Participation on Agriculture, Environmentalism, Horticulture, Floristics, Food Production and Processing, Naklo, Slovenia, 20–21 April 2016; pp. 514–521. [Google Scholar]
- Spotte, S. Biological process. In Captive Seawater Fishes; John Wiley & Sons: New York, NY, USA, 1992; Chapter 2; pp. 79–91. [Google Scholar]
- Huguenin, E.J.; Colt, J. Design and Operating Guide for Aquaculture Seawater Systems, 2nd ed.; Elsevier: Amsterdam, The Netherlands, 2002; pp. 20–24. [Google Scholar]
- Alessio, G.; Allegrucci, G.; Angle, G. Acquacoltura Responsabile—Verso le Produzioni Acquatiche del Terzo Millennio; FAO per L’autorizzazione, Unimar-Uniprom; FAO: Rome, Italy, 2001; p. 683. Available online: http://www.unimar.it/wp-content/uploads/2017/04/20.-acquacolturaresp_rid.pdf (accessed on 2 September 2019).
- Yavuzcan Yildiz, H.; Robaina, L.; Pirhonen, J.; Mente, E.; Domínguez, D.; Parisi, G. Fish welfare in aquaponic systems: Its relation to water quality with an emphasis on feed and faeces—A review. Water 2017, 9, 13. [Google Scholar] [CrossRef]
- Colt, J.; Lamoureux, J.; Patterson, R.; Rogers, G. Reporting standards for the biofilter performance studies. Aquac. Eng. 2006, 34, 377–388. [Google Scholar] [CrossRef]
- Rojas-Tirado, P.; per Bovbjerg, P.; Lars-Flemming, P. Bacterial activity dynamics in the water recirculating phase during start-up of aquaculture systems. Aquac. Eng. 2018, 78, 24–31. [Google Scholar] [CrossRef]
- Gerardi, M. Wastewater Bacteria; John Willey & Sons: Hoboken, NJ, USA, 2006; p. 251. [Google Scholar]
- Feidantsis, K.; Antonopoulou, E.; Lazou, A.; Pörtner, H.O.; Michaelidis, B. Seasonal variations of cellular stress response of the gilthead sea bream (Sparus aurata). J. Comp. Physiol. B 2013, 183, 625–639. [Google Scholar] [CrossRef]
- Stensløkken, K.O.; Ellefsen, S.; Larsen, H.K.; Vaage, J.; Nilsson, G.E. Expression of heat shock proteins in anoxic crucian carp (Carassius carassius): Support for cold as a preparatory cue for anoxia. Am. J. Physiol. Integr. Comp. Physiol. 2010, 298, R1499–R1508. [Google Scholar] [CrossRef]
- Dean, E.E.; Kelly, S.P.; Luk, J.C.; Woo, N.Y. Chronic salinity adaptation modulates hepatic heat shock protein and insulin-like growth factor Iexpresseion in black sea bream. Mar. Biotechnol. 2002, 4, 193–205. [Google Scholar]
- Seger, R.; Krebs, E.G. The MAPK signaling cascade. FASEB J. 1995, 9, 726–735. [Google Scholar] [CrossRef]
- Kaloyianni, M.; Feidantsis, K.; Ntelli, I.; Stergiou, P.; Tsoulia, T.; Dimitriadi, A.; Antonopoulou, E.; Bobori, D. Biochemical and molecular responses of cyprinids in two Mediterranean lacustrine ecosystems: Opputunities for ecological assesment and biomonitoring. Aquat. Toxicol. 2019, 211, 105–115. [Google Scholar] [CrossRef]
- Roufidou, C.; Feidantsis, K.; Mente, E.; Sarropoulou, E.; Antonopoulou, E. Heat shock protein (HSP) expression and mitogen—Activated protein kinase (MAPK) phosphorylation during early embryonic developmental stages of the gilthead sea bream (Sparus aurata). Mediterr. Mar. Sci. 2018, 19, 240–247. [Google Scholar] [CrossRef]
- Endut, A.; Jusoh, A.; Ali, N.; Wan Nik, W.B.; Hassan, A. A study on the optimal hydraulic loading rate and plant ratios in recirculation aquaponic system. Bioresour. Technol. 2010, 101, 1511–1517. [Google Scholar] [CrossRef] [PubMed]
- Palm, W.H.; Bissa, K.; Knaus, U. Significant factors affecting the economic sustainability of closed aquaponic systems. Part II: Fish and plant growth. AACL Bioflux 2014, 7, 162–175. [Google Scholar]
- Khater, E.G.; Bahnasawy, A.H.; Shams, A.E.S.; Hassaan, M.S.; Hassan, Y.A. Utilization of effluent fishfarms in tomato cultivation. Ecol. Eng. 2015, 83, 199–207. [Google Scholar] [CrossRef]
- Kotzen, B.; Appelbaum, S. An investigation of aquaponics using brackish water resources in the NegevDesert. J. Appl. Aquac. 2010, 22, 297–320. [Google Scholar] [CrossRef]
- Boxman, S. Resource Recovery through Halophyte Production in Marine Aquaponics: An Evaluation of the Nutrient Cycling and the Environmental Sustainability of Aquaponics. Ph.D. Thesis, University of South Florida, Tampa, FL, USA, 2015; pp. 6–86. [Google Scholar]
- Pantanella, E.; Colla, G. Saline aquaponics opportunities for integraed marine aquaculture. In Proceedings of the International Aquaponic Conference: Aquaponics and Global Food Security, Stevens Point, WI, USA, 19–21 June 2013; p. 52. [Google Scholar]
- Nozzi, V.; Parisi, G.; Di Crescenzo, D.; Giordano, M.; Carnevali, O. Evaluation of Dicentrarchus labrax meats and the vegetable quality of Beta vulgaris var. cicla farmed in fresh water and salt water aquaponic systems. Water 2016, 8, 423. [Google Scholar] [CrossRef]
- Goddek, S.; Joyce, A.; Kotzen, B.; Burnell, G.M. Aquaponics Food Production Systems: Compined Aquaculture and Hydroponic Production Technology for the Future; Springer: Berlin/Heidelberg, Germany, 2019; p. 619. [Google Scholar]
- Flowers, T.J.; Colmer, T.D. Salinity tolerance in halophytes. New Phytol. 2008, 179, 945–963. [Google Scholar] [CrossRef]
- Hamdani, F.; Derridj, A.; Rogers, H.J. Diverse salinity responses in Critmum maritimum tissues at different salinities over time. J. Soil Sci. Plant Nutr. 2017, 17, 716–734. [Google Scholar] [CrossRef]
- Rakocy, J.; Shultz, R.C.; Bailey, D.S.; Thoman, E.S. Aquaponic production of tilapia and basil: Comparing a batch and staggered cropping system. In Proceedings of the South Pacific Soilless Culture Conference 2003, Palmerston North, New Zealand, 10–13 February 2003; pp. 63–69. [Google Scholar]
- Hotos, G.N.; Vlahos, N.G. Observations on the conditioning of filter bed in aquaria with water of very highsalinity (hypersalinity). Aquac. Eng. 1999, 19, 215–222. [Google Scholar] [CrossRef]
- Vlahos, N.; Kormas, K.; Pachiadaki, G.M.; Meziti, A.; Hotos, G. Changes of bacterioplankton apparent species richness in two ornamental fish aquaria. SpringerPlus 2013, 2, 66. [Google Scholar] [CrossRef]
- Bekiari, V.; Avramidis, P. Data quality in water analysis: Validation of combustion-infrared and combustion-chemiluminescence methods for the simultaneous determination of Total Organic Carbon (TOC) and Total Nitrogen (TN). Int. J. Environ. Anal. Chem. 2013, 94, 65–76. [Google Scholar] [CrossRef]
- Porter, K.G.; Feig, Y.S. The use of DAPI for identifying and counting aquatic microflora. Limnol. Oceanogr. 1980, 25, 943–948. [Google Scholar] [CrossRef]
- Mente, E.; Solovyev, M.M.; Vlahos, N.; Rotllant, G.; Gisbert, E. Digestive enzymes activity during initial ontogeny and after feeding diets with different protein sources in zebra cichlid Archocentrus nigrofasciatus. J. World Aquac. Soc. 2016, 48, 831–848. [Google Scholar] [CrossRef]
- Antonopoulou, E.; Chouri, E.; Feidantsis, K.; Lazou, A.; Chatzifotis, S. Effects of partial dietary supplementation of fish meal with soymeal on the stress and apoptosis response in the digestive system of common dentex (Dentex dentex). J. Boil. Res. 2017, 24, 14. [Google Scholar] [CrossRef] [PubMed]
- Zar, J.H. Biostatistical Analysis; Prentice-Hall: London, UK, 1999; p. 718. [Google Scholar]
- Endut, A.; Josoh, A.; Ali, N.A. Nitrogen budget and effluent nitrogen components in aquaponics recirculation system. Desalt. Water Treat. 2014, 52, 744–752. [Google Scholar] [CrossRef]
- Hu, Z.; Lee, J.W.; Chandran, K.; Kim, S.; Brotto, A.C.; Khanal, S.K. Effect of plants species on nitrogen recovery in aquaponics. Bioresour. Technol. 2015, 188, 92–98. [Google Scholar] [CrossRef] [PubMed]
- Nozzi, V.; Graber, A.; Schmautz, Z.; Mathis, A.; Junge, R. Nutrient Management in Aquaponics: Comparison of Three Approaches for Cultivating Lettuce, Mint and Mushroom Herb. Agronomy 2018, 8, 27. [Google Scholar] [CrossRef]
- Sadek, S.S. Seabream culture in Egypt, status, constraints and potential. Fish Physiol. Biochem. 2000, 22, 171–178. [Google Scholar] [CrossRef]
- Gullian Klanian, M.; Arámburu Adame, C. Performance of Nile tilapia O. niloticus fingerlings in a hyper-intensive recirculating aquaculture system with low water-exchange. Lat. Am. J. Aquat. Res. 2013, 41, 150–162. [Google Scholar] [CrossRef]
- Sadek, S.S.; Fathy Osman, M.; Adel Mansour, M. Growth, survival and feed conversion rates of sea bream (Sparus aurata) cultured in earthen brackish water ponds fed different feed types. Aquac. Int. 2004, 12, 409–421. [Google Scholar] [CrossRef]
- Ebeling, J.M.; Timmons, M.B.; Bisogni, J.J. Engineering analysis of the stoichiometry of photoautotrophic, autotrophic, and heterotrophic removal of ammonia-nitrogen in aquaculture systems. Aquaculture 2006, 257, 346–358. [Google Scholar] [CrossRef]
- Timmons, M.B.; Ebeling, J.M. Recirculating Aquaculture; Cayuga Aqua Ventures: Ithaca, NY, USA, 2007; p. 959. [Google Scholar]
- Torno, J.; Naas, C.; Schroeder, J.P.; Schulz, C. Impact of hydraulic retendion time, backflushing interval, and C/N ratio on the SID-reactor denitrification performance in marine RAS. Aquaculture 2018, 496, 112–122. [Google Scholar] [CrossRef]
- Summerfelt, S.T. Design and management of conventional fluidized-sand filters. Aquac. Eng. 2006, 34, 275–302. [Google Scholar] [CrossRef]
- Summerfelt, S.T.; Zühlke, A.; Kolarevic, J.; Britt, K.; Reiten, M.; Selset, R.; Gutierrez, X.; Terjesen, B.F. Effects of alkalinity on ammonia removal, carbon dioxide stripping, and system pH in semi-commercial scale water recirculating aquaculture systems operated with moving bed bioreactors. Aquac. Eng. 2015, 65, 46–54. [Google Scholar] [CrossRef]
- Christianson, L.; Lepine, C.; Tsukuda, S.; Summerfelt, S.T. Nitrate removal effectiveness of fluidized sulfur-based autotrophic denitrification biofilters for recirculating aquaculture systems. Aquac. Eng. 2015, 68, 10–18. [Google Scholar] [CrossRef]
- Suhr, K.I.; Pedersen, P.B.; Arvin, E. End-of-pipe denitrification using RAS effluent waste streams: Effect of C/N-ratio and hydraulic retention time. Aquac. Eng. 2013, 53, 57–64. [Google Scholar] [CrossRef]
- Chen, S.; Ling, J.; Blancheton, J.-P. Nitrification kinetics of biofilm as affected by water quality factors. Aquac. Eng. 2006, 34, 179–197. [Google Scholar] [CrossRef]
- Zhu, S.; Chen, S. The impact of temperature on nitrification rate in fixed film biofilters. Aquac. Eng. 2002, 26, 221–237. [Google Scholar] [CrossRef]
- Ohashi, A.; VirajdeSilva, D.G.; Mobarry, B.; Manem, J.A.; Stahl, D.A.; Rittmann, B.E. Influence of substrate C/N ratio on the structure of multi-species biofilms consisting ofnitrifiers and heterotrophs. Water Sci. Technol. 1995, 32, 75–84. [Google Scholar] [CrossRef]
- Díaz, V.; Ibanez, R.; Gómez, P.; Utriaga, A.M.; Ortiz, I. Kinetics of nitrogen compounds in a commercial marine Recirculating Aquaculture System. Aquac. Eng. 2012, 50, 20–27. [Google Scholar] [CrossRef]
- Nogueira, R.; Melo, L.F.; Purkhold, U.; Wuertz, S.; Wagner, M. Nitrifying and heterotrophic population dynamics in biofilm reactors: Effects of hydraulic retention time and the presence of organic carbon. Water Res. 2002, 36, 469–481. [Google Scholar] [CrossRef]
- Avnimelech, Y. C/N ratio as a control element in aquaculture systems. Aquaculture 1999, 176, 3227–3235. [Google Scholar] [CrossRef]
- Sherr, E.B.; Sherr, B.F. Significance of predation by protists in aquatic microbial food webs. Antonie Leewenhoek 2002, 81, 293–308. [Google Scholar] [CrossRef]
- Keuter, S.; Kruse, M.; Lipski, A.; Spieck, E. Relevance of Nitrospira for nitrite oxidation in a marine recirculation aquaculture system and physiological features of a Nitrospira marina-like isolate. Environ. Microbiol. 2011, 13, 2536–2547. [Google Scholar] [CrossRef] [PubMed]
- Keuter, S.; Quantz, G.; Spieck, S.E. Long-term monitoring of nitrification and nitrifying communities during bio filter activation of two marine recirculation aquaculture systems (RAS). Int. J. Aquac. Fish. Sci. 2017, 3, 51–61. [Google Scholar]
- Attramadal, K.J.; Truong, T.M.H.; Bakke, I.; Skjermo, J.; Olsen, Y.; Vadstein, O. RAS and microbial maturation as tools for K-selection of microbial communities improve survival in cod larvae. Aquaculture 2014, 432, 483–490. [Google Scholar] [CrossRef]
- Alleman, J.E.; Preston, K. Behavior and physiology of nitrifying bacteria. In Proceedings of the second annual conference on commercial aquaculture. CES 1991, 240, 1–13. [Google Scholar]
- Marquis, R.E. Salt-induced contraction of bacterial cell walls. J. Bacteriol. 1968, 95, 775–781. [Google Scholar]
- Waller, U.; Buhmann, A.K.; Ernst, A.; Hanke, V.; Kulakowski, A.; Wecker, B.; Orellana, J.; Papenbrock, J. Integrated multi-trophic aquaculture in a zero-exchange recirculation aquaculture system for marine fishand hydroponic halophyte production. Aquac. Int. 2015, 23, 1473–1489. [Google Scholar] [CrossRef]
- Ben Fattoum, R.; Zaghdoud, C.; Atia, A.; Ben Khedher, A.; Gouia, H.; Chaffei Haouari, C. Recovery capacity of the edible halophyte Crithmum maritimum from temporary salinity in relation to nutrient accumulation and nitrogen metabolism. Biol. Sect. Bot. 2016, 71, 1345–1352. [Google Scholar] [CrossRef]
- Nebel, C.; Romestand, B.; Nègre-Sadargues, G.; Grousset, E.; Aujoulat, F.; Bacal, J.; Bonhomme, F.; Charmantier, G. Differential freshwater adaptation in juvenile sea-bass Dicentrarchus labrax: Involvement of gills and urinary system. J. Exp. Boil. 2005, 208, 3859–3871. [Google Scholar] [CrossRef] [PubMed]
- Herman, R.L. Systemic Noninfectious Granulomatoses of Fishes; National Biological Service, Leetown Science Center, National Fish Health Research Laboratory: Kearneysville, WV, USA, 1996; p. 9.
- Baudin-Laurencin, F.; Messager, J.L. Granulomatous hypertyrosonaemia. In ICES (International Council for the Exploration of the Sea) Identification Leaflets for Diseases and Parasites of Fish and Shellfish; DK-1261; ICES: Copenhagen, Denmark, 1991; Volume 41, p. 4. ISSN 0109-2510. [Google Scholar] [CrossRef]
- Giffard-Mena, I.; Lorin-Nebel, C.; Charmantier, G.; Castille, R.; Boulo, V. Adaptation of the sea-bass (Dicentrarchus labrax) to fresh water: Role of aquaporins and Na+/K+-ATPases. Comp. Biochem. Physiol. Part A Mol. Integr. Physiol. 2008, 150, 332–338. [Google Scholar] [CrossRef] [PubMed]
- Laiz-Carrión, R.; Guerreiro, P.M.; Fuentes, J.; Canario, A.V.; Martín Del Río, M.P.; Mancera, J.M. Branchial osmoregulatory response to salinity in the gilthead sea bream, Sparus auratus. J. Exp. Zool. Part A Comp. Exp. Boil. 2005, 303, 563–576. [Google Scholar] [CrossRef] [PubMed]
- Masroor, W.; Farcy, E.; Gros, R.; Lorin-Nebel, C. Effect of combined stress (salinity and temperature) in European sea bass Dicentrarchus labrax osmoregulatory processes. Comp. Biochem. Physiol. Part A Mol. Integr. Physiol. 2018, 215, 45–54. [Google Scholar] [CrossRef] [PubMed]








| 8 ppt | 20 ppt | |
|---|---|---|
| TAN (mg/L) | 0.39 ± 0.1 a | 0.33 ± 0.07 a |
| NO2− (mg/L) | 0.89 ± 0.3 a | 0.82 ± 0.3 a |
| NO3− (mg/L) | 76.4 ± 11.2 a | 77.2 ± 11.8 a |
| pH | 7.54 ± 0.05 a | 7.73 ± 0.042 a |
| 20 ppt | 8 ppt | |
|---|---|---|
| TN (mg/L) | 23.89 ± 6.30 a | 14.42 ± 7.77 b |
| TOC (mg/L) | 16.57 ± 2.0 a | 8.1 ± 0.78 b |
| C/N | 1.14 ± 0.32 a | 0.66 ± 0.1 a |
| 8 ppt | 20 ppt | |
|---|---|---|
| HLR (m/day) | 1.85 ± 0.005 a | 1.85 ± 0.005 a |
| R (min) | 0.014 ± 0.003 a | 0.014 ± 0.003 a |
| HRT (min) | 9.7 ± 0.01 a | 9.7 ± 0.02 a |
| SSA (m2/m3) | 162 ± 0.30 a | 224 ± 0.31 b |
| Vmedia (m3) | 6.73 ± 0.02a | 6.94 ± 0.08 b |
| V (L) | 32.1 ± 0.001 a | 32.1 ± 0.001 a |
| 8 ppt | 20 ppt | |
|---|---|---|
| Gilthead seabream growth performance | ||
| Initial weight (Win, g) | 2.54 ± 0.05 a | 2.55 ± 0.06 a |
| Final weight (Wfin, g) | 27.91 ± 0.84 a | 28.07 ± 0.85 a |
| Weight gain (WG, g) | 25.36 ± 0.83 a | 25.51 ± 0.87 a |
| Specific growth rate (SGR, %/day) | 3.17 ± 0.04 a | 3.17 ± 0.06 a |
| Survival (%) | 99% | 97% |
| Initial condition factor (Kin) | 1.35 ± 0.02 a | 1.46 ± 0.02 b |
| Final condition factor (Kfin) | 1.58 ± 0.03 a | 1.46 ± 0.02 b |
| Food Conversion rate (FCR) | 1.80 ± 0.06 a | 1.84 ± 0.07 b |
| Daily Feed Intake (DFI, %/day) | 2.39 ± 0.08 a | 2.45 ± 0.1 a |
| Initial length (Lin, cm) | 5.73 ± 0.03 a | 5.57 ± 0.04 a |
| Final length (Lfin, cm) | 12.04 ± 0.11 a | 12.40 ± 0.16 a |
| Rock samphire growth performance | ||
| Initial height (cm) | 8.45 ± 0.34 a | 7.94 ± 0.46 a |
| Final height (cm) | 10.32 ± 0.58 a | 8.03 ± 0.47 b |
| Height gain of plant | 1.87 ± 0.56 a | 0.99 ± 0.28 b |
| Final number of lateral branches | 4.83 ± 0.47 a | 2.94 ± 0.42 b |
| Final number of shoots | 4.22 ± 0.59 a | 1.94 ± 0.21 b |
| 8 ppt | 20 ppt | |
|---|---|---|
| Midgut | 0 | 0 |
| Liver | 2 | 1 |
| Kidney | 3 | 2 |
| Gills | 2 | 2 |
| 8 ppt | 20 ppt | |
|---|---|---|
| Initial bacterial abundance (cells × 105/mL) | 19.9 × 105 ± 0.72 a | 20.2 × 105 ± 0.92 a |
| Bacterial abundance (cells × 105/mL) at the 1st day of salinity changes | 1.08 × 105 ± 1.82 a | 20.2 × 105 ± 0.92 b |
| Final bacterial abundance (cells × 105/mL) | 6.8 × 105 ± 1.9 a | 18.6 × 105 ± 0.91 b |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vlahos, N.; Levizou, E.; Stathopoulou, P.; Berillis, P.; Antonopoulou, E.; Bekiari, V.; Krigas, N.; Kormas, K.; Mente, E. An Experimental Brackish Aquaponic System Using Juvenile Gilthead Sea Bream (Sparus aurata) and Rock Samphire (Crithmum maritimum). Sustainability 2019, 11, 4820. https://doi.org/10.3390/su11184820
Vlahos N, Levizou E, Stathopoulou P, Berillis P, Antonopoulou E, Bekiari V, Krigas N, Kormas K, Mente E. An Experimental Brackish Aquaponic System Using Juvenile Gilthead Sea Bream (Sparus aurata) and Rock Samphire (Crithmum maritimum). Sustainability. 2019; 11(18):4820. https://doi.org/10.3390/su11184820
Chicago/Turabian StyleVlahos, Nikolaos, Efi Levizou, Paraskevi Stathopoulou, Panagiotis Berillis, Efthimia Antonopoulou, Vlasoula Bekiari, Nikos Krigas, Konstantinos Kormas, and Eleni Mente. 2019. "An Experimental Brackish Aquaponic System Using Juvenile Gilthead Sea Bream (Sparus aurata) and Rock Samphire (Crithmum maritimum)" Sustainability 11, no. 18: 4820. https://doi.org/10.3390/su11184820
APA StyleVlahos, N., Levizou, E., Stathopoulou, P., Berillis, P., Antonopoulou, E., Bekiari, V., Krigas, N., Kormas, K., & Mente, E. (2019). An Experimental Brackish Aquaponic System Using Juvenile Gilthead Sea Bream (Sparus aurata) and Rock Samphire (Crithmum maritimum). Sustainability, 11(18), 4820. https://doi.org/10.3390/su11184820










