Pulp and Paper Mill Fly Ash: A Review
Abstract
1. Introduction
2. Solid Waste Generation and Management in Pulp and Paper Mills
3. Major Properties of Pulp and Paper Mill Fly Ash
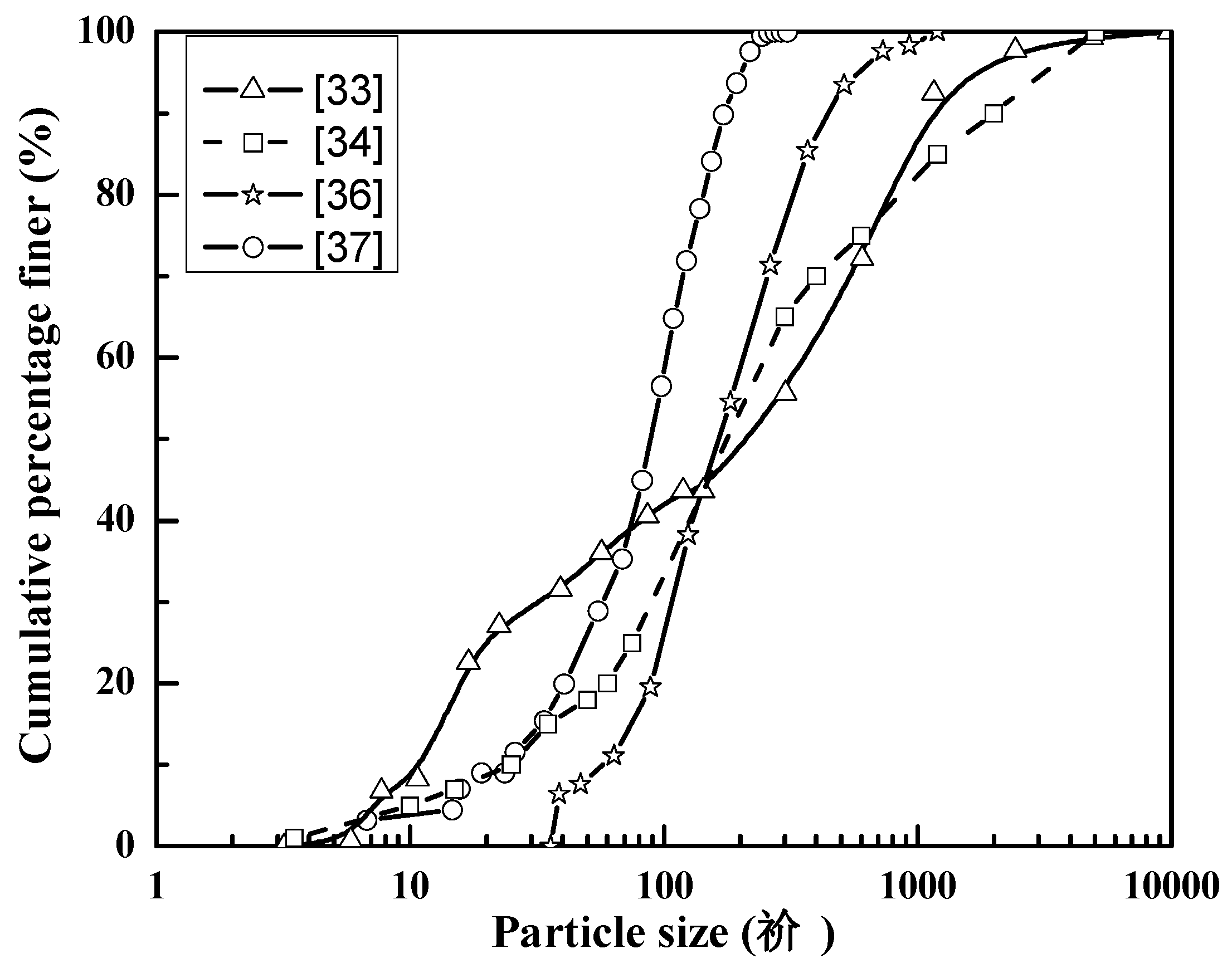
3.1. Physical Properties
3.2. Chemical Properties
3.3. Morphological and Mineralogical Properties
3.4. Leachate Characteristics
3.4.1. Trace Metal Concentrations
3.4.2. Inorganic and Organic Compounds
3.5. Radioactivity
4. Valorization of Pulp and Paper Mill Fly Ash
5. Existing and Prospective Applications of Pulp and Paper Mill Fly Ash and Value-Added Geomaterials
5.1. Soil Amendments in Agriculture and Forestry
5.2. Supplementary Cementitious Material in Construction Industry
5.3. Binder Components for Geotechnical Engineering Applications
5.4. Raw Materials for the Material Manufacturing Industry
5.5. Adsorbent for Remediation of Contaminated Environments
6. Summary
Author Contributions
Funding
Conflicts of Interest
References
- Elliot, A.; Mahmood, T. Beneficial uses of pulp and paper power boiler ash residues. TAPPI J. 2006, 5, 9–16. [Google Scholar]
- Lamers, F.; Cremers, M.; Matschegg, D.; Schmidl, C.; Hannam, K.; Hazlett, P.; Madrali, S.; Dam, B.P.; Roberto, R.; Mager, R.; et al. Options for Increased Use of Ash from Biomass Combustion and Co-Firing. IEA Bioenergy: Task 32. 2019. Available online: https://www.ieabioenergy.com/wp-content/uploads/2019/02/IEA-Bioenergy-Ash-management-report-revision-5-november.pdf (accessed on 12 January 2019).
- Hackett, G.A.R.; Easton, C.A.; Duff, S.J.B. Composting of pulp and paper mill fly ash with wastewater treatment sludge. Bioresour. Technol. 1999, 70, 217–224. [Google Scholar] [CrossRef]
- Xie, A. Characteristics of Wastepaper Sludge Ash and its Potential Applications in Concrete. Master’s Thesis, University of Sherbrooke, Sherbrooke, QC, Canada, November 2009. [Google Scholar]
- Kamali, M.; Khodaparast, Z. Review on recent developments on pulp and paper mill wastewater treatment. Ecotoxicol. Environ. Saf. 2015, 114, 326–344. [Google Scholar] [CrossRef] [PubMed]
- Scheepers, G.P.; du Toil, B. Potential use of wood ash in South African forestry: A review. South. For. J. For. Sci. 2016, 78, 255–266. [Google Scholar] [CrossRef]
- Fauberta, P.; Barnabéb, S.; Boucharda, S.; Côtéa, R.; Villeneuvea, C. Pulp and paper mill sludge management practices: What are the challenges to assess the impacts on greenhouse gas emissions. Resour. Conserv. Recycl. 2016, 108, 107–133. [Google Scholar] [CrossRef]
- Wysong, M.L. CZ’s solid waste problems at Wuana are reduced by composting. Pulp Pap. 1976, 50, 112–113. [Google Scholar]
- Campbell, A.G. Recycling and disposing of wood ash. TAPPI J. 1990, 73, 141–146. [Google Scholar]
- Sikora, L.J. Benefits and drawbacks to composting organic by-products. In Beneficial Co-Utilization of Agricultural, Municipal and Industrial By-Products; Springer Nature: Dordrecht, Netherlands, 1998; pp. 69–77. [Google Scholar]
- Muse, J.K.; Mitchell, C.C. Paper mill boiler ash and lime by-products as soil liming materials. Agron. J. 1995, 87, 432–438. [Google Scholar] [CrossRef]
- Risse, L.M.; Gaskin, J.W. Best Management Practices for Wood Ash as Agricultural Soil Amendment; Bulletin 1142; University of Georgia: Athens, GA, USA, 2010; p. 4. Available online: https://extension.uga.edu/publications/detail.html?number=B1142&title=Best%20Management%20Practices%20for%20Wood%20Ash%20as%20Agricultural%20Soil%20Amendment (accessed on 24 May 2019).
- Väätäinen, K.; Sirparanta, E.; Räisänen, M.; Tahvanainen, T. The cost and profitability of using granulated wood ash as a forest fertilizer in drained peatland forest. Biomass Bioenergy 2011, 35, 3335–3341. [Google Scholar] [CrossRef]
- Bang-Andreasen, T.; Nielsen, J.T.; Voriskova, J.; Heeise, J.; Rønn, R.; Kjøller, R.; Hansen, H.C.B.; Jacobsen, C.S. Wood ash induced pH changes strongly affect soil bacterial numbers and community composition. Front. Microbiol. 2017, 8, 1400. [Google Scholar] [CrossRef] [PubMed]
- Etiégni, L.; Campbell, A.; Mahler, R. Evaluation of wood ash disposal on agricultural land. 1. potential as a soil additive and liming agent. Commun. Soil Sci. Plant Anal. 1991, 22, 243–256. [Google Scholar] [CrossRef]
- Pitman, R.M. Wood ash in forestry: A review of the environmental impacts. J. For. 2006, 79, 563–588. [Google Scholar] [CrossRef]
- Toller, S.; Kärrman, E.; Gustafsson, J.P.; Magnusson, Y. Environmental assessment of incinerator residue utilisation. J. Waste Manag. 2009, 29, 2071–2077. [Google Scholar] [CrossRef] [PubMed]
- Perumal, P.; Ganesh, G.M.; Santhi, A.S. A review on artificial aggregates. Int. J. Earth Sci. Eng. 2012, 5, 540–546. [Google Scholar]
- Qin, J.; Cui, C.; Cui, X.; Hussain, A.; Yang, C.; Yang, S. Recycling of lime mud and fly ash for fabrication of anorthite ceramic at low sintering temperature. Ceram. Int. 2015, 41, 5648–5655. [Google Scholar] [CrossRef]
- Zumrawi, M.M.E. Stabilization of pavement subgrade by using fly ash activated by cement. Am. J. Civ. Eng. Archit. 2015, 3, 218–224. [Google Scholar] [CrossRef]
- Škēls, P.S.; Bondars, K.; Plonis, R.; Haritonovs, V.; Paeglītis, A. Usage of wood fly ash in stabilization of unbound pavement layers and soils. In Proceedings of the 13th Baltic Sea Geotechnical Conference, Vilnius, Lithuania, 22–24 September 2016. [Google Scholar] [CrossRef]
- Murmu, A.L.; Dhole, N.; Patel, A. Stabilization of black cotton soil for subgrade application using fly ash geopolymer. Road Mater. Pavement Des. 2018, 1–19. [Google Scholar] [CrossRef]
- Simão, L.; Hotza, D.; Raupp-Pereira, F.; Labrincha, J.A.; Montedo, O.R.K. Characterization of pulp and paper mill waste for the production of waste-based cement. Rev. Int. Contam. Ambient. 2019, 35, 237–246. [Google Scholar] [CrossRef]
- Bajpai, P. Generation of waste in pulp and paper mills. In Management of Pulp and Paper Mill Waste; Springer: New York, NY, USA, 2015; pp. 9–17. ISBN 978-3-319-11787-4. [Google Scholar]
- Simão, L.; Hotza, D.; Raupp-Pereira, F.; Labrincha, J.A.; Montedo, O.R.K. Wastes from pulp and paper mills—A review of generation and recycling alternatives. Cerâmica 2018, 64, 443–453. [Google Scholar] [CrossRef]
- Gavrilescu, D. Solid waste generation in Kraft pulp mills. Environ. Eng. Manag. J. 2004, 3, 399–404. [Google Scholar] [CrossRef]
- Tran, H.; Vakkilainnen, E.K. The Kraft chemical recovery process. In Proceedings of the Tappi Kraft Pulping Short Course, St. Petersburg, FL, USA, 7–10 January 2008; pp. 1.1:1–1.1:8. Available online: https://www.tappi.org/content/events/08kros/manuscripts/1-1.pdf (accessed on 4 December 2018).
- Miller, M.; Justiniano, M.; McQueen, S. Energy and Environmental Profile of the U.S. Pulp and Paper Industry; Industrial Technologies Program; U.S. Department of Energy: Washington, DC, USA, 2005; p. 89. Available online: https://www.energy.gov/sites/prod/files/2013/11/f4/pulppaper_profile.pdf (accessed on 12 October 2018).
- NRC Natural Resources Canada. The Model Kraft Market Pulp Mill. 2005. Available online: http://oee.nrcan.gc.ca/publications/infosource/pub/cipec/pulp-paperindustry/Section-01.cfm?text=N&printview=N (accessed on 10 April 2019).
- Jesús, A.G.; de Alda, O. Feasibility of recycling pulp and paper mill sludge in the paper and board industries. Resour. Conserv. Recycl. 2008, 52, 965–972. [Google Scholar] [CrossRef]
- Cabrera, M.N. Pulp mill wastewater: Characteristics and treatment. In Biological Wastewater Treatment and Resource Recovery; Farooq, R., Ahmad, Z., Eds.; InTech: Rijeka, Croatia, 2017; pp. 120–139. ISBN 978-953-51-3045-1. [Google Scholar]
- Demeyer, A.; Voundi, N.J.C.; Verloo, M.G. Characteristics of wood ash and influence on soil properties and nutrient uptake: An overview. Bioresour. Technol. 2001, 77, 287–295. [Google Scholar] [CrossRef]
- Naik, T.R.; Kraus, R.N. A new source of pozzolanic material. Concr. Int. 2003, 25, 55–62. [Google Scholar]
- Grau, F.; Choo, H.; Hu, J.W.; Jung, J. Engineering behaviour and characteristics of wood ash and sugarcane bagasse ash. Mater. J. 2015, 8, 6962–6977. [Google Scholar] [CrossRef] [PubMed]
- Etiégni, L.; Campbell, A.G. Physical and chemical characteristics of wood ash. Bioresour. Technol. 1991, 37, 173–178. [Google Scholar] [CrossRef]
- Sivasundaram, M. Glass Ceramics from Pulp and Paper Waste Ash. Master’s Thesis, McGill University, Montreal, QC, Canada, March 2000. [Google Scholar]
- Monosi, S.; Sani, D.; Ruello, M.L. Reuse of paper mill ash in plaster blends. Open Waste Manag. J. 2012, 5, 5–10. [Google Scholar] [CrossRef]
- Cheah, C.B.; Ramli, M. The implementation of wood waste ash: Review paper. Resour. Conserv. Recycl. 2011, 55, 669–685. [Google Scholar] [CrossRef]
- Pöykiö, R.; Nurmesniemi, H.; Perämäki, P.; Kuokkanen, T.; Välimäki, I. Leachability of metals in fly ash from a pulp and paper mill complex and environmental risk characterisation for eco-efficient utilization of the fly ash as a fertilizer. Chem. Speciat. Bioavailab. 2005, 17, 1–9. [Google Scholar] [CrossRef]
- Guerrini, I.A.; Moro, L.; Lopes, M.A.F.; Boas, R.L.V.; Benedetti, V. Application of wood ash and pulp and paper sludge to Eucalyptus grandis in three Brazilian soils. In The Forest Alternative: Principles and Practice of Residuals Use; Henry, C.L., Ed.; College of Forest Resources; University of Washington: Seattle, WA, USA, 2000; pp. 127–131. Available online: https://www.researchgate.net/publication/242412209_Application_of_Wood_Ash_and_Pulp_and_Paper_Sludge_to_Eucalyptus_grandis_in_Three_Brazilian_Soils (accessed on 24 November 2018).
- Park, B.P.; Yanai, R.D.; Sahm, J.M.; Lee, D.K.; Abrahamson, L.P. Wood ash effects on plant and soil in a willow bioenergy plantation. Biomass Bioenergy 2005, 28, 355–365. [Google Scholar] [CrossRef]
- Vassilev, S.; Baxter, D.; Andersen, L.K.; Vassileva, C.G. An overview of the composition and application of biomass ash. part 1. phase-mineral and chemical composition and classification. Fuel 2013, 105, 40–76. [Google Scholar] [CrossRef]
- Malhotra, V.M.; Ramezanianpour, A.A. Fly Ash in Concrete, 2nd ed.; Canada Centre for Mineral and Energy Technology (CANMET), Natural Resources Canada: Ottawa, ON, Canada, 1994; ISBN 13: 978-0660157641.
- Serafimova, E.; Mladenov, M.; Mihailova, I.; Pelovski, Y. Study on the characteristics of waste wood ash. J. Univ. Chem. Technol. Metall. 2011, 46, 31–34. [Google Scholar]
- Misra, M.K.; Ragland, K.W.; Baker, A.J. Wood ash composition as a function of furnace temperature. Biomass Bioenergy 1993, 4, 103–116. [Google Scholar] [CrossRef]
- Patterson, S. The Agronomic Benefit of Pulp Mill Boiler Wood Ash. Master’s Thesis, University of Lethbridge, Lethbridge, AB, Canada, May 2001. [Google Scholar]
- Camberato, J.J.; Gagnon, B.; Angers, D.A.; Chantigny, M.H.; Pan, W.L. Pulp and paper mill by-products as soil amendments and plant nutrient sources. Can. J. Soil Sci. 2011, 86, 641–653. [Google Scholar] [CrossRef]
- Sear, L.K.A.; Weatherley, A.J.; Dawson, A. The environmental impacts of using fly ash—The UK producers’ perspective. In Proceedings of the International Ash Utilization Symposium, Lexington, KY, USA, 20–22 October 2003; pp. 1–14. Available online: http://www.flyash.info/2003/20sear.pdf (accessed on 1 September 2018).
- Ohno, T.; Erich, M.S. Incubation-derived calcium carbonate equivalence of papermill boiler ashes derived from sludge and wood sources. Environ. Pollut. 1993, 79, 175–180. [Google Scholar] [CrossRef]
- Abdullahi, M. Characteristics of wood ash/OPC concrete. Leonardo Electron. J. Pract. Technol. 2006, 8, 9–16. [Google Scholar]
- Chowdhury, S.; Mishra, M.; Suganya, O. The incorporation of wood waste ash as a partial cement replacement material for making structural grade concrete: An overview. Ain Shams Eng. J. 2015, 6, 429–437. [Google Scholar] [CrossRef]
- Johakimu, J.K.; Roopchund, R.; Sithole, B.B. Beneficiation of fly ash from pulp and paper mills: Valorisation into heat-resistant geo-polymers. In Proceedings of the Technical Association of the Pulp and Paper Industry of South Africa National Conference and Exhibition, Durban, South Africa, 21–22 September 2016; Available online: https://www.researchgate.net/publication/308663771_Beneficiation_of_fly_ash_from_pulp_and_paper_mills_valorisation_into_heat-resistant_geo-polymers (accessed on 15 November 2018).
- Olanders, B.; Steenari, B. Characterization of ashes from wood and straw. Biomass Bioenergy 1995, 8, 105–115. [Google Scholar] [CrossRef]
- Gu, X.; Jin, X.; Zhou, Y. Basic Principles of Concrete Structures; Springer: New York, NY, USA, 1995; ISBN 978-3-662-48563-7. [Google Scholar]
- Ulery, A.L.; Graham, R.C.; Amrhein, C. Wood-ash composition and soil pH following intense burning. Soil Sci. 1993, 156, 358–364. [Google Scholar] [CrossRef]
- Zhou, H.; Smith, D.W.; Sego, D.C. Characterization and use of pulp mill fly ash and lime by-products as road construction amendments. Can. J. Civ. Eng. 2000, 27, 581–593. [Google Scholar] [CrossRef]
- Kuokkanen, T.; Nurmesniemi, H.; Pöykiö, R.; Kujala, K.; Kaakinen, J.; Kuokkanen, M. Chemical and leaching properties of paper mill sludge. Chem. Speciat. Bioavailab. 2008, 20, 111–122. [Google Scholar] [CrossRef]
- Knapp, B.A.; Insam, H. Recycling of biomass ashes-current technologies and research needs. In Recycling of Biomass Ashes; Knapp, B.A., Insam, H., Eds.; Springer: New York, NY, USA, 2011; ISBN 978-3-642-19353-8. [Google Scholar]
- Environmental Protection Agency. Standards for the use or disposal of sewage sludge. In Clean Water Act; Rules and Regulations, Federal Register, USEPA: Washington, DC, USA, 1993; Volume 58, pp. 9248–9404. Available online: https://www.epa.gov/sites/production/files/2015-10/documents/58_fr_9248_9404_standards_for_the_disposal_of_sewage_sludge_final_reduced.pdf (accessed on 9 April 2019).
- Pöykiö, R.; Mäkelä, M.; Watkins, G.; Nurmesniemi, H.; Dahl, O. Heavy metal leaching in bottom ash and fly ash fractions from industrial-scale BFB-boiler for environmental risks assessment. Trans. Nonferrous Met. Soc. China 2016, 26, 256–264. [Google Scholar] [CrossRef]
- Alakangas, E. Properties of Wood Fuels Used in Finland-BIOSOUTH-Project; Project Report PRO2/P2030/05; Technical Research Centre of Finland: Jyväskylä, Finland, 2005; p. 90. Available online: https://ec.europa.eu/energy/intelligent/projects/sites/iee-projects/files/projects/documents/bio-south_wood_fuel_properties.pdf (accessed on 11 November 2018).
- Matysik, M.A.; Gilmore, D.W.; Mozaffari, M.; Halbach, T.R. Application of Wood Ash, Biosolids, and Papermill Residuals to Forest Soils—A Review of the Literature; Staff Paper Series #153; University of Minnesota: St. Paul, MN, USA, 2001; Available online: https://www.forestry.umn.edu/sites/forestry.umn.edu/files/Staffpaper153.PDF (accessed on 6 December 2018).
- Palomo, A.; Grrutzeck, M.W.; Blanco, M.T. Alkali-activated fly ashes: A cement for future. Cem. Concr. Res. 1999, 29, 1323–1329. [Google Scholar] [CrossRef]
- Rios, S.; Cristelo, N.; Miranda, T.; Araújo, N.; Oliviera, J.; Lucas, E. Increasing the reaction kinetics of alkali activated fly ash for stabilisation of a silty sand pavement sub-base. Road Mater. Pavement Des. 2018, 19, 201–222. [Google Scholar] [CrossRef]
- Cristelo, N.; Glendinning, S.; Miranda, T.; Oliveira, D.; Silva, R. Soil stabilisation using alkaline activation of fly ash for self-compacting rammed earth construction. Constr. Build. Mater. 2012, 36, 727–735. [Google Scholar] [CrossRef]
- Thokchom, S.; Dutta, D.; Ghosh, S. Effect of incorporating silica fume in fly ash geopolymers. World Acad. Sci. Eng. Technol. 2011, 60, 243–247. [Google Scholar]
- Miccio, F.; Medri, V.; Papa, E.; Natali, M.A.; Landi, E. Geopolymerization as effective measure for reducing risks during coal ashes handling, storage and disposal. Chem. Eng. Trans. 2014, 36, 133–138. [Google Scholar] [CrossRef]
- Leong, H.Y.; Ong, D.E.L.; Sanjayan, J.G.; Nazari, A. Strength development of soil–fly ash geopolymer: Assessment of soil, fly ash, alkali activators, and water. J. Mater. Civ. Eng. 2018, 30, 04018171. [Google Scholar] [CrossRef]
- Querol, X.; Moreno, N.; Umaña, J.T.; Alastuey, A.; Hernández, E.; Lopez-Soler, A.; Plana, F. Synthesis of zeolites from coal fly ash: An overview. Int. J. Coal Geol. 2002, 50, 413–423. [Google Scholar] [CrossRef]
- Sathia, R.; Babu, K.G.; Santhanam, M. Durability study of low calcium fly ash geopolymer concrete. In Proceedings of the 3rd Asian Concrete Federation International Conference, Ho Chi Minh City, Vietnam, 11–13 November 2008; pp. 1153–1159. Available online: https://www.scribd.com/document/117851741/durability-study-of-low-calcium-fly-ash-geopolymer-concrete (accessed on 10 June 2018).
- Cristelo, N.; Glendinning, S.; Fernandes, L.; Pinto, A.T. Effects of alkaline-activated fly ash and Portland cement on soft soil stabilisation. Acta Geotech. 2013, 8, 395–405. [Google Scholar] [CrossRef]
- Pandya, R.R.; Shah, A.J. Effect of alkali activated fly ash on the strength of clayeysoil. In Proceedings of the Indian Geotechnical Conference, Assam, India, 14–16 December 2017; pp. 1–4. Available online: http://www.igs.org.in/portal/igc-proceedings/Theme09/Th09_287.pdf (accessed on 16 September 2018).
- Steenbruggen, G.; Hollman, G.G. The synthesis of zeolites from fly ash and the properties of the zeolite products. J. Geochem. Explor. 1998, 62, 305–309. [Google Scholar] [CrossRef]
- Li, Z.; Ohunki, T.; Ikeda, K. Development of paper sludge ash-based geopolymer and application to treatment of hazardous water contaminated with radioisotopes. Materials 2016, 9, 633. [Google Scholar] [CrossRef]
- Hardjito, D.; Wallah, S.E.; Sumajouw, D.M.J.; Rangan, B.V. Fly ash-based geopolymer concrete. Aust. J. Struct. Eng. 2010, 6, 77–86. [Google Scholar] [CrossRef]
- Pachamuthu, S.; Thangaraju, P. Effect of incinerated paper sludge ash on fly ash-based geopolymer concrete. Gradevinar 2016, 69, 851–859. [Google Scholar] [CrossRef]
- Parhi, P.S.; Garanayak, L.; Mahamaya, M.; Das, S.K. Stabilization of an expansive soil using alkali activated fly ash based geopolymer. In Advances in Characterization and Analysis of Expansive Soils and Rocks; Hoyos, L., McCartney, J., Eds.; Springer: New York, NY, USA, 2017; pp. 36–50. [Google Scholar] [CrossRef]
- Udawattha, C.; Halwatura, R. Geopolymerized self-compacting mud concrete masonry units. Case Stud. Constr. Mater. 2018, 9, e00177. [Google Scholar] [CrossRef]
- de Sousa, S.M.T.; de Carvalho, C.M.; Torres, S.M.; Barbosa, N.M.; Gomes, K.M.; Ghavami, K. Mechanical Properties and Durability of Geopolymer Stabilised Earth Blocks. 2011, pp. 1–11. Available online: https://www.researchgate.net/publication/245024883_Mechanical_Properties_and_Durability_of_Geopolymer_Stabilizated_Earth_Blocks (accessed on 14 April 2019).
- Bui, Q.B.; Pfrudhomme, E.; Grillet, A.C.; Prime, N. An experimental study on earthen materials stabilized by geopolymer. In Lecture Notes in Civil Engineering, Proceedings of the 4th Congrès International de Géotechnique-Ouvrages-Structures, CIGOS 2017, Ho Chi Minh City, Vietnam, 26–27 October 2017; Tran-Nguyen, H.H., Wong, H., Ragueneau, F., Ha-Minh, C., Eds.; Springer: Singapore, 2018; Volume 8, pp. 319–328. ISBN 978-981-10-6712-9. [Google Scholar]
- Okoronkwo, C.D. Developing Sustainable and Environmentally Friendly Building Materials in Rammed Earth Construction. Ph.D. Thesis, University of Wolverhampton, Wolverhampton, UK, April 2015. [Google Scholar]
- Ondova, M.; Sicakova, A. Review of current trends in ways of fly ash application. In Proceedings of the 14th International Multidisciplinary Scientific Geoconference, Albena, Bulgaria, 17–26 June 2014. [Google Scholar] [CrossRef]
- Hannam, K.D.; Deschamps, C.D.; Kwiaton, M.; Venier, L.; Hazlett, P.W. Regulations and Guidelines for the Use of Wood Ash as a Soil Amendment in Canadian Forests; Natural Resources Canada Report GLC-X-17; Canadian Forest Service: Toronto, ON, Canada, 2016; p. 53. Available online: https://cfs.nrcan.gc.ca/publications?id=37781 (accessed on 10 October 2018).
- Hazlett, P.; Emilson, C. Applying Wood Ash Waste to Soil: Contributing to Sustainable Forest Management in Canada. Canadian Institute of Forestry (CIF) e-Lecture. 27 February 2019. Available online: http://www.cif-ifc.org/wp-content/uploads/2018/12/Hazlett_Emilson-eLecture-CIF-2019.pdf (accessed on 2 February 2019).
- Robichaud, P.R.; Ashmun, L.E.; Sims, B.D. Post-Fire Treatment Effectiveness for Hillslope Stabilization; General Technical Reports RMRS-GTR-240; U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: Fort Collins, CO, USA, 2010; p. 62. Available online: https://www.fs.fed.us/rm/pubs/rmrs_gtr240.pdf (accessed on 18 December 2018).
- Wohlgemuth, P.M.; Beyers, J.L.; Hubbert, K.R. Rehabilitation strategies after fire: The California, USA experience. In Fire Effects on Soils and Restoration Strategies. Land Reconstruction and Management Series; Cerdá, A., Robichaud, P.R., Eds.; CRC Press: Boca Raton, FL, USA, 2009; Volume 5, pp. 511–536. ISBN 1439843333. [Google Scholar]
- McLaughlin, R.A.; Brown, T.T. Evaluation of erosion control products with and without added and polyacrylamide. J. Am. Water Resour. Assoc. 2006, 42, 675–684. [Google Scholar] [CrossRef]
- Zhang, X.C.; Miller, W.P.; Nearing, M.A.; Norton, L. Effects of surface treatments on surface sealing, runoff, and interrill erosion. Trans. ASAE 1998, 41, 989–994. [Google Scholar] [CrossRef]
- Simão, L.; Jiusti, J.; Lóh, N.J.; Hotza, D.; Raupp-Pereira, F.; Labrincha, J.A.; Montedo, O.R.K. Waste-containing clinkers: Valorization of alternative mineral sources from pulp and paper mill. Process Saf. Environ. 2017, 109, 106–116. [Google Scholar] [CrossRef]
- Martínez-Lage, M.; Velay-Lizancos, P.; Vázquez-Burgo, M.; Rivas-Fernández, C.; Vázquez-Herrero, A.; Ramírez-Rodríguez, M.; Martín-Cano, M. Concretes and mortars with waste paper industry: Biomass ash and dregs. J. Environ. Manag. 2016, 181, 863–873. [Google Scholar] [CrossRef]
- Oliveira, K.A.; Nazáriob, B.I.; de Oliveira, A.P.N.; Hotza, D.; Raupp-Pereira, F. Industrial wastes as alternative mineral addition in Portland cement and as aggregate in coating mortars. Mater. Res. 2017, 20, 358–364. [Google Scholar] [CrossRef][Green Version]
- Mehta, P.K. Pozzolanic and cementitious by-products as mineral admixtures for concrete—A critical review. In Proceedings of the First CANMET/ACI International Conference on the Use of Fly Ash, Silica Fume and Other Mineral By-Products in Concrete, Montebello, QC, Canada, 31 July–5 August 1983; Abstract #51. pp. 1–46. [Google Scholar] [CrossRef]
- Siddique, R. Waste Materials and By-products in Concrete; Springer: Berlin, Germany, 2008; ISBN 978-3-540-74293-7. [Google Scholar]
- Bremseth, S.J. Fly Ash in Concrete a Literature Study of the Advantages and Disadvantages. COIN Pr. Rep. 18, SINTEF Building and Infrastructure. 2008, p. 37. Available online: https://www.sintefbok.no/book/download/1000 (accessed on 28 January 2019).
- Dockter, B.A.; Eylands, K.E.; Hamre, L.L. Use of bottom ash and fly ash in rammed-earth construction. In Proceedings of the International Ash Utilization Symposium: Materials for the Next Millennium, Lexington, KY, USA, 18–20 October 1999; Paper #56. Available online: http://www.flyash.info/1999/construc/pflug22.pdf (accessed on 5 May 2018).
- Burroughs, S. Recommendations for the selection, stabilization, and compaction of soil for rammed earth wall construction. J. Green Build. 2010, 5, 101–114. [Google Scholar] [CrossRef]
- Suresh, A.; Anand, K.B. Strength and durability of rammed earth for walling. J. Archit. Eng. 2017, 23, 06017004. [Google Scholar] [CrossRef]
- Consoli, N.C.; da Rocha, C.G.; Saldanha, R.B. Coal fly ash-carbide lime bricks: An environment friendly building product. Constr. Build. Mater. 2014, 69, 301–309. [Google Scholar] [CrossRef]
- Siddiqua, S.; Barreto, P.N.M. Chemical stabilization of rammed earth using calcium carbide residue and fly ash. Constr. Build. Mater. 2018, 169, 364–367. [Google Scholar] [CrossRef]
- Magnusson, Y. Environmental Systems Analysis for Utilization of Bottom Ash in Ground Constructions. Master’s Thesis, KTH Royal Institute of Technology, Stockholm, Sweden, March 2005. [Google Scholar]
- Rani, M.U.; Jenifer, J.M. Analysis of strength characteristics of black cotton soil using wood ash as stabilizer. Int. J. Res. Sci. Technol. 2016, 6, 171–179. [Google Scholar]
- Jhariya, S.; Parte, S.S. Stabilization of black cotton soil by the waste sludge (hypo-sludge). Int. J. Sci. Dev. Res. 2018, 3, 445–449. [Google Scholar]
- Ondova, M.; Stevulova, N. Slovak Fly Ash as Cement Substitution in the Concrete Road Pavements; Lambert Academic Publishing: Verlag, Germany, 2013; p. 108, ISBN-13 978-3-659-51273-5. [Google Scholar]
- Arm, M.; Vestin, J.; Lind, B.; Lagerkvist, A.; Nordmark, D.; Hallgren, P. Pulp mill fly ash for stabilization of low-volume unpaved forest roads—Field performance. Can. J. Civ. Eng. 2014, 41, 955–963. [Google Scholar] [CrossRef]
- James, J. Strength benefits of saw dust/wood ash amendment in stabilization of an expansive soil. Rev. Fac. Ing. 2018, 28, 44–61. [Google Scholar] [CrossRef]
- Moo-Young, H.K., Jr.; Zimmie, T.F. Waste minimization and re-use of paper sludges in landfill covers: A case study. Waste Manag. Res. 1997, 15, 593–605. [Google Scholar] [CrossRef]
- Slim, G.I.; Morales, M.; Alrumaidhin, L.; Bridgman, P.; Gloor, J.; Hoff, S.T.; Odem, W.I. Optimization of polymer-amended fly ash and paper pulp millings mixture for alternative landfill liner. Procedia Eng. 2016, 145, 312–318. [Google Scholar] [CrossRef][Green Version]
- Ribeiro, A.S.M.; Monteiro, R.C.C.; Davim, E.J.R.; Fernandes, M.H.V. Ash from a pulp mill boiler-characterisation and vitrification. J. Hazard. Mater. 2010, 179, 303–308. [Google Scholar] [CrossRef]
- Ghouleh, Z. Production of Glass-Ceramics from Municipal Solid Waste (MSW) Fly Ash. Master’s Thesis, McGill University, Montreal, QC, Canada, December 2008. [Google Scholar]
- Vu, D.H.; Wang, K.S.; Chen, J.H.; Nam, B.X.; Bac, B.H. Glass-ceramics from mixtures of bottom ash and fly ash. Waste Manag. J. 2012, 32, 2306–2314. [Google Scholar] [CrossRef]
- Goel, G.; Kalamdhad, A.S. Paper mill sludge (PMS) and degraded municipal solid waste (DMSW) blended fired bricks—A review. MOJ Civ. Eng. 2018, 4, 81–85. [Google Scholar] [CrossRef]
- Rodella, N.; Pasquali, M.; Zacco, A.; Bilo, F.; Borgese, L.; Bontempi, N.; Tomasoni, G.; Depero, L.E.; Bontempi, E. Beyond waste: New sustainable fillers from fly ashes stabilization, obtained by low cost raw materials. Heliyon 2016, 2, e00163. [Google Scholar] [CrossRef]
- Huang, X.; Hwang, J.Y.; Gillis, J.M. Processed low NOx fly ash as a filler in plastics. J. Min. Mater. Charact. Eng. 2013, 2, 11–31. [Google Scholar] [CrossRef]
- Sansui, O.M.; Komolafe, O.D.; Ogundana, T.O.; Olaleke, M.O.; Sanni, Y.Y. Development of wood-ash/resin polymer matrix composite for body armour application. FUOYE J. Eng. Technol. 2016, 1, 10–14. [Google Scholar]
- Sansui, O.M.; Oyinlola, A.K.; Akindapo, J.O. Influence of wood ash on the mechanical properties of polymer matrix composite developed from fibre glass and epoxy resin. Int. J. Eng. Res. Technol. 2013, 2, 344–352. [Google Scholar]
- Malakootian, M.; Almasi, A.; Hossaini, H. Pb and Co removal from paint industries effluent using wood ash. Int. J. Environ. Sci. Technol. 2008, 5, 217–222. [Google Scholar] [CrossRef]
- Laohaprapanon, S.; Marques, M.; Hogland, W. Removal of organic pollutants from wastewater using wood fly ash as a low-cost sorbent. Clean-Soil Air Water 2010, 38, 1055–1061. [Google Scholar] [CrossRef]
- Sahoo, P.K.; Tripathy, S.; Panigrahi, M.K.; Equeenuddin, S.M. Evaluation of the use of an alkali modified fly ash as a potential adsorbent for the removal of metals from acid mine drainage. Appl. Water Sci. 2013, 3, 567–576. [Google Scholar] [CrossRef]
- Tewari, N.; Verma, V.K.; Rai, J.P.N. Comparative evaluation of natural adsorbent for pollutants removal from distillery spent wash. J. Sci. Ind. Res. 2006, 65, 935–938. [Google Scholar]
- Ahmed, Z.T. The Quantification of the Fly Ash Adsorption Capacity for the Purpose of Characterization and Use in Concrete. Ph.D. Thesis, Michigan Technological University, Houghton, MI, USA, December 2012. Available online: https://digitalcommons.mtu.edu/etds/789 (accessed on 6 March 2019).
- Singh, P.; Sharda, S.; Cauhan, S.S. Domestic waste water treatment by fly and wood ash along with additive materials. Int. J. Civ. Eng. Technol. 2016, 7, 67–75. [Google Scholar]
- Kimura, K.; Wajima, T. Phosphate removal ability of calcined paper sludge from aqueous solution-effect of calcination temperature. Int. J. Environ. Sci. Dev. 2017, 8, 247–250. [Google Scholar] [CrossRef][Green Version]
- Jacobson, S. Addition of stabilised wood ashes to Swedish coniferous stands on mineral soils- effects on stem growth and needle nutrient concentrations. Silva Fenn. 2003, 37, 437–450. [Google Scholar] [CrossRef]
- Kellner, O.; Weibull, H. Effects of wood ash on bryophytes and lichens in a Swedish pine forest. Scand. J. For. Res. 1998, 13, 76–85. [Google Scholar]

| Reference | Composition of Major and Minor Oxides (%) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| SiO2 | Al2O3 | Fe2O3 | CaO | MgO | Na2O | K2O | TiO2 | SO3 | MnO | P2O5 | |
| Muse and Mitchell [11] | N/P | 1.25 | 0.63 | 12.0 | 0.77 | 0.14 | 1.33 | N/P | N/P | N/P | N/P |
| Pitman [16] | N/P | 9.1 | 5.1 | 1.66 | 0.11 | 0.01 | 0.26 | N/P | 20 | N/P | N/P |
| Naik et al. [33] | 26.5 | 9.0 | 5.4 | 16.0 | 3.0 | 1.7 | 5.0 | 0.51 | 4.8 | N/P | N/P |
| Vassilev et al. [42] | 31.6 | 13.2 | 5.12 | 28.9 | 5.4 | 1.42 | 13.2 | N/P | 2.67 | 2.77 | 2.64 |
| Serafimova et al. [44] | N/P | N/P | N/P | 52 | 1.32 | N/P | 2.39 | N/P | N/P | N/P | 0.72 |
| Ohno and Eric [49] | N/P | 8.21 | 1.43 | 9.49 | 0.65 | 0.67 | 1.03 | N/P | N/P | N/P | N/P |
| Abdullahi [50] | 31.8 | 28 | 2.34 | 10.53 | 9.32 | 6.5 | 10.38 | N/P | N/P | N/P | N/P |
| Chowdhury et al. [51] | 50.7 | 8.2 | 2.1 | 19.6 | 6.5 | 2.1 | 2.8 | N/P | N/P | N/P | N/P |
| Johakimu et al. [52] | 0.68–52.78 | 0.12–28.3 | 0.37–3.69 | 1.59–4.88 | 0.35–83.58 | N/P | 0.43–1.97 | 0.01–1.68 | 4.23–62.62 | N/P | N/P |
| Reference | Concentration of Trace Elements (mg/kg) | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| As | Ba | Bo | Cd | Co | Cr | Cu | Mo | Ni | Pb | Se | V | Zn | |
| USEPA [59] | 75 | N/P | N/P | 85 | N/P | 3000 | 4300 | 57 | 75 | 420 | 100 | N/P | 7500 |
| Muse and Mitchell [11] | N/P | 588 | 95 | <2 | 14 | 75 | 67 | 15 | 16 | 72 | N/P | N/P | 183 |
| Grau et al. [34] | N/D | N/D | 0.47 | N/D | N/D | 0.03 | N/D | N/D | N/D | N/D | N/D | N/D | N/D |
| Serafimova et al. [44] | 11.3 | N/P | N/P | 1.11 | N/P | 23 | N/P | N/P | 16.1 | 99.7 | N/P | N/P | 133 |
| Ohno and Eric [49] | N/D | 549 | N/D | N/D | N/D | 1036 | 151 | 61 | 65 | 32 | N/D | N/D | 423 |
| Zhou et al. [56] | N/D | 0.13 | 1.41 | N/D | N/P | 0.05 | N/P | N/D | N/D | N/D | 0.03 | N/P | 0.67 |
| Poykio et al. [60] | 13 | 745 | N/P | 2.9 | 6.6 | 66.9 | N/P | 3.8 | 32.4 | 28.7 | 3.3 | 92.7 | 295.3 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cherian, C.; Siddiqua, S. Pulp and Paper Mill Fly Ash: A Review. Sustainability 2019, 11, 4394. https://doi.org/10.3390/su11164394
Cherian C, Siddiqua S. Pulp and Paper Mill Fly Ash: A Review. Sustainability. 2019; 11(16):4394. https://doi.org/10.3390/su11164394
Chicago/Turabian StyleCherian, Chinchu, and Sumi Siddiqua. 2019. "Pulp and Paper Mill Fly Ash: A Review" Sustainability 11, no. 16: 4394. https://doi.org/10.3390/su11164394
APA StyleCherian, C., & Siddiqua, S. (2019). Pulp and Paper Mill Fly Ash: A Review. Sustainability, 11(16), 4394. https://doi.org/10.3390/su11164394





