Plant Biodiversity Knowledge Varies by Gender in Sustainable Amazonian Agricultural Systems Called Chacras
Abstract
1. Introduction
2. Materials and Methods
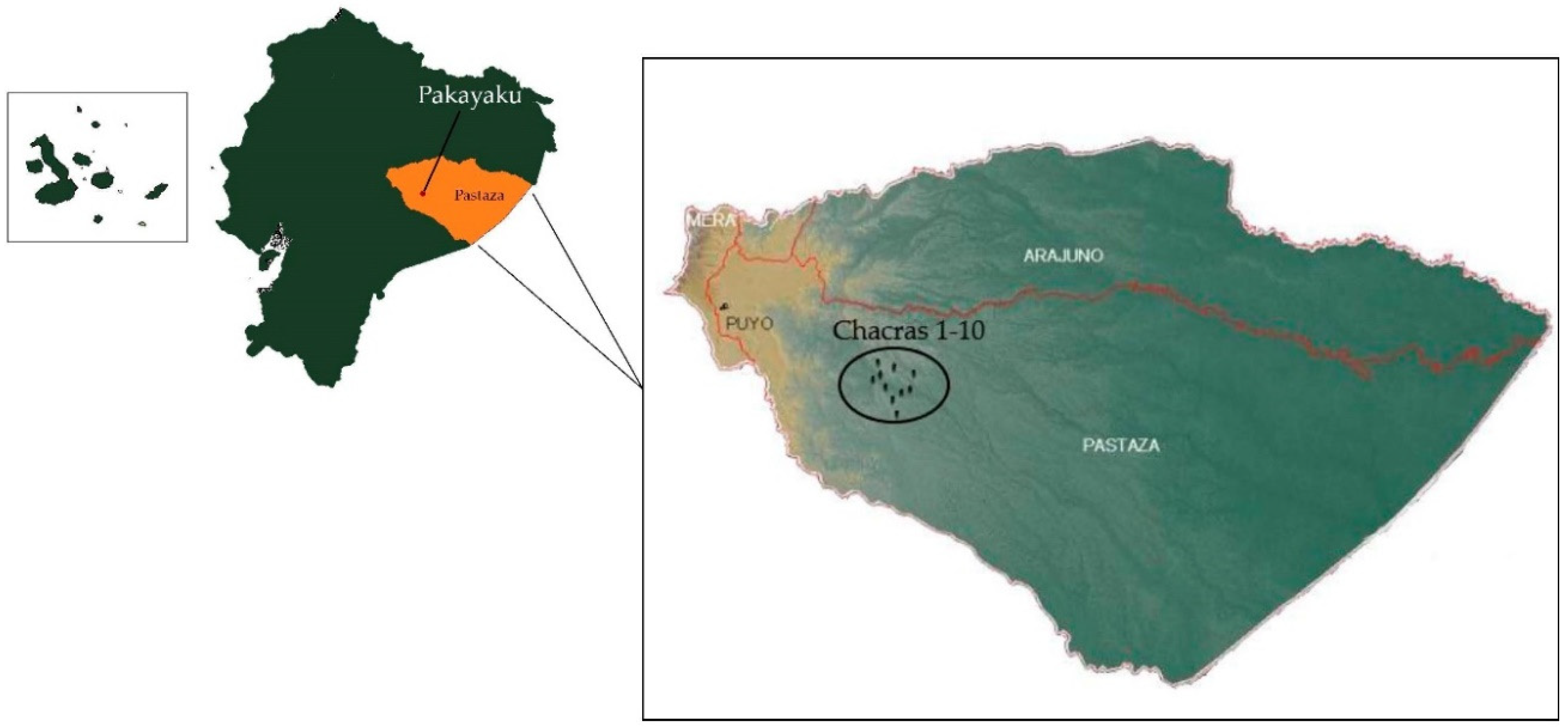
2.1. Study Area and Permissions
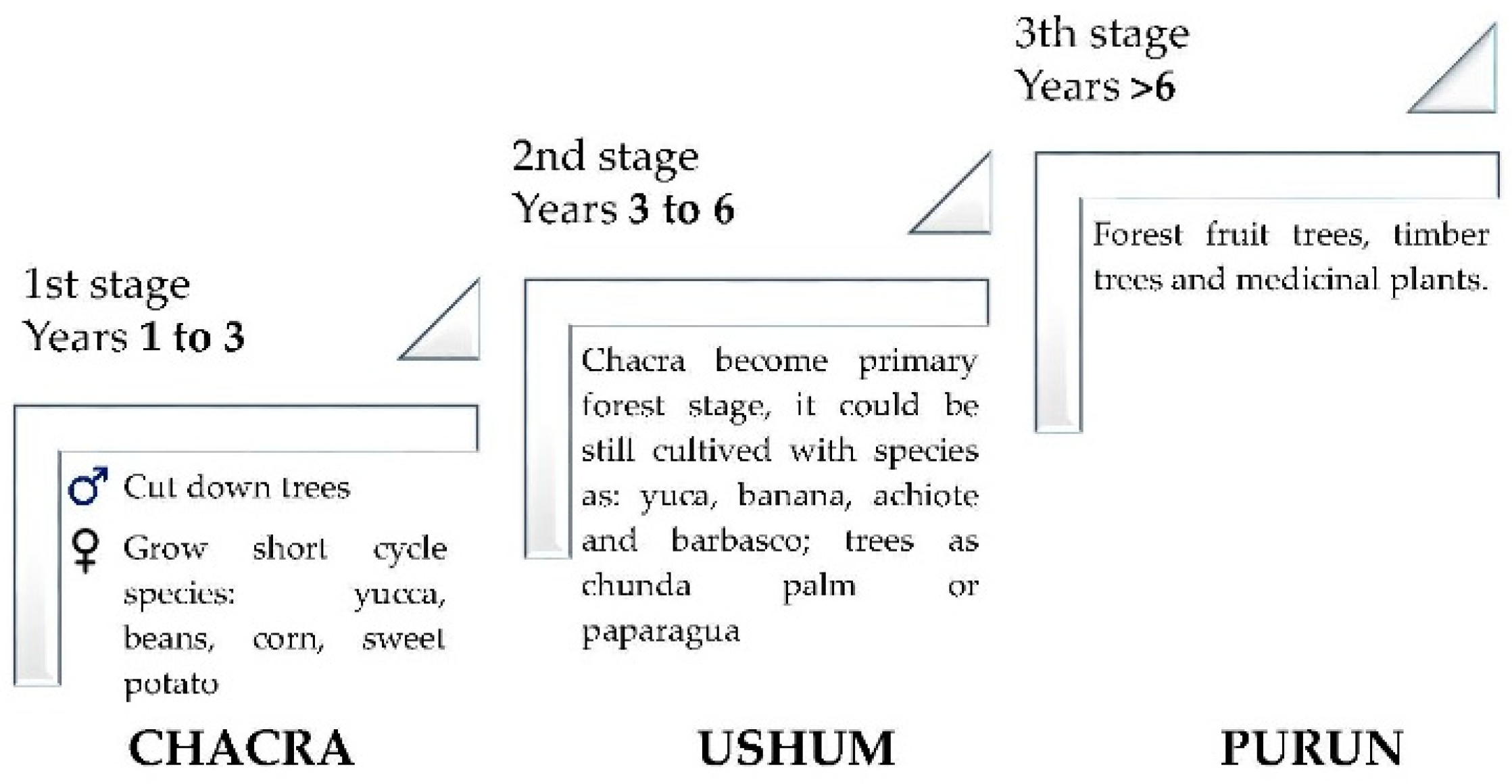
2.2. Plant Material
2.3. Workshop Design
Selection of the People and Participation
2.4. Data Digitalization
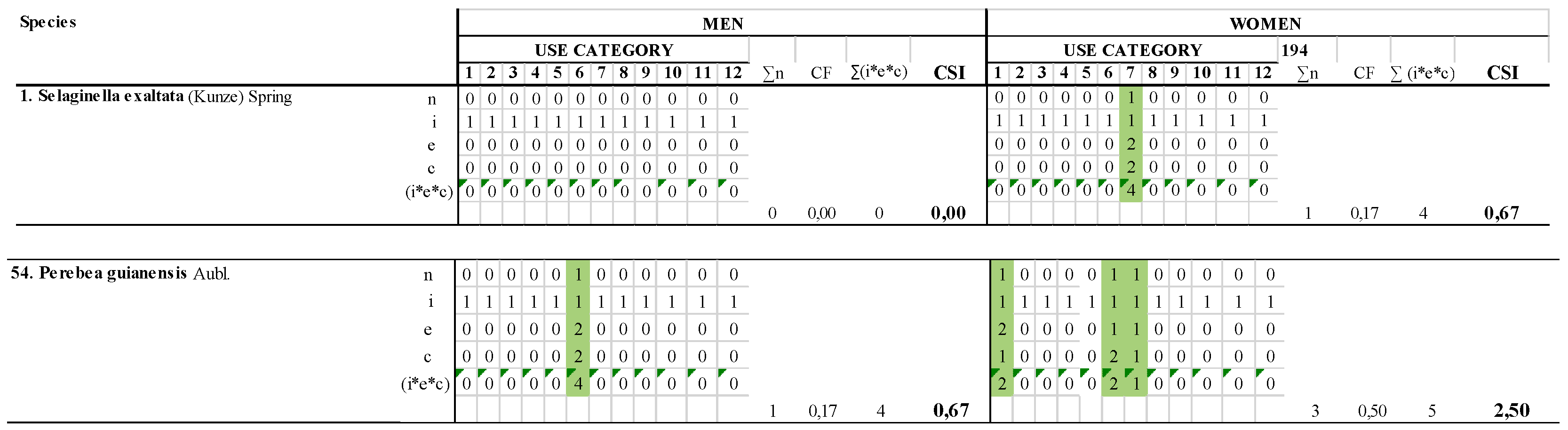
- Row i (management): Significance of the plant from an agronomical point of view. For wild plants, value, = 1. For plants referenced as cultivated in the Encyclopedia of Useful Plants of Ecuador [29] value, = 2.
- Row e (preference): Proportion of the workshop participants who selected that use category as “the most preferred”. It is expressed as per participant, not in percentage. Maximum value 2 was given to the preferred category. Non-preferred categories value = 1. Non-used categories = 0.
- Row c (frequency): Proportion of the workshop who selected that use category as “the most frequently used in the community”. It is expressed as per participant, not in percentage. Maximum value 2 was given to the selected category. Non-selected categories value = 1. Non-used categories = 0.
- Row (i*c*e): Value obtained for a category of use of the species in the workshop. Maximum value (2 × 2 × 2) = 8. Minimum value (1 × 1 × 1) = 1 Non-used categories = 0.
- Column ∑n: Minimum value 0. maximum value = 12. H = the highest value obtained in the compared workshops by one species.
- Column ∑(i*e*c): Global value of all the uses of the species. The maximum value that one species can obtain is ((2 × 2 × 2) + [11 × (2 × 1 × 1)) = 30.
2.5. Quantitative Analysis of Data: Cultural Significance Indexes (CSI) Calculations and Statistics
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Herren, H.R. The Sustainable Development Goals: Challenge or opportunity. In Farming, Food and Nature; Taylor and Francis: London, UK, 2018; pp. 171–173. [Google Scholar]
- Peña-Venegas, P.; Mazorra Valderrama, A.; Acosta Muñoz, L.E.; Pérez Rúa, M.N. Seguridad Alimentaria en Comunidades Indígenas del Amazonas: Ayer y Hoy; Instituto Sinchi: Bogotá, Colombia, 2009. [Google Scholar]
- Bioversity International. Sustainable Agriculture for Food and Nutrition Security; Bioversity International: Rome, Italy, 2011; ISBN 978-92-9043-898-4. [Google Scholar]
- Ortiz, R.; Nowak, A.; Lavado, A.; Parker, L. Food Security in the Amazon—A Report for the Amazonia Security Agenda Project; Global Canopy Foundation: Oxford, UK, 2013. [Google Scholar]
- Tapia-Armijos, M.F.; Homeier, J.; Espinosa, C.I.; Leuschner, C.; De La Cruz, M. Deforestation and forest fragmentation in south Ecuador since the 1970s—Losing a hotspot of biodiversity. PLoS ONE 2015, 10. [Google Scholar] [CrossRef] [PubMed]
- FAO-CEPAL. Seguridad Alimentraria, Nutrición y Erradicación del Hambre, Celac 2025: Elementos Para el Debate y la Cooperación Regionales; United Nations: New York, NY, USA, 2016. [Google Scholar]
- Calero, C.J. Seguridad Alimentaria en Ecuador Desde un Enfoque de Acceso a Alimentos. Master’s Thesis, FLACSO Ecuador, Quito, Ecuador, 2011. [Google Scholar]
- Ortiz, T.P. El laberinto de la autonomía indígena en el Ecuador. Las circunscripciones territoriales indígenas en la Amazonía Central, 2010–2012. Lat. Am. Caribb. Ethn. Stud. 2015, 10, 60–86. [Google Scholar]
- COP-6-CDB UNEP/CBD/COP/6. Decisions. Available online: https://www.cbd.int/kb/record/meetingDocument/2303?RecordType=meetingDocument&Event=COP-06 (accessed on 24 June 2019).
- Ulloa, A. The Ecological Native: Indigenous Peoples’ Movements and Eco-Governmentality in Colombia; Routledge: Abingdon, UK, 2013; ISBN 9780203958674. [Google Scholar]
- Indigenous-Women-of-America. Memoria: IV Encuentro Continental de las Mujeres Indígenas de las Américas: Lima, 4–7 abril del 2004: Un Solo Continente, un Solo Espíritu; Chirapaq, Centro de Culturas Indígenas de Perú: Lima, Perú, 2004; ISBN 9873-676-11-2. [Google Scholar]
- Vera, R.R.; Cota-Sánchez, J.H.; Grijalva Olmedo, J.E.; Coq-Huelva, D.; Higuchi, A.; Alfalla-Luque, R.; Burgos-Morán, R.; Arias-Gutiérrez, R.; Panduro, A.; Mass, W.; et al. La chagra: Patrimonio colectivo de las comunidades indígenas amazónicas. Sustainability 2017, 9, 21–37. [Google Scholar] [CrossRef]
- Garí, J. Biodiversity and Indigenous Agroecology in Amazonia: The Indigenous Peoples of Pastaza. Ecol. Res. 2001, 5, 21–37. [Google Scholar]
- Santi, E. Por la Tierra, Por la Vida, Despertemos. Confrontación Entre visiones Sobre el Territorio; PPGCSPA: Sao Louis, Brazil, 2016. [Google Scholar]
- Panduro, A.; Mass, W.; Reig, M.C.; Pinedo, J.M. Chacras Amazónicas; Programa de Cooperación Hispano Peruano, Agencia Española de cooperación Internacional para el Desarrollo y Universidad de Córdoba: Iquitos, Perú, 2010; ISBN 9786124565045. [Google Scholar]
- Vera, R.R.; Cota-Sánchez, J.H.; Grijalva Olmedo, J.E. Biodiversity, dynamics, and impact of chakras on the Ecuadorian Amazon. J. Plant. Ecol. 2017, 12, 34–44. [Google Scholar] [CrossRef]
- Robert, A.V. Are women reservoirs of traditional plant knowledge? Gender, ethnobotany and globalization in northeast Brazil. Singap. J. Trop. Geogr. 2007, 28, 7–20. [Google Scholar]
- Pfeiffer, J.; Butz, R. Assessing cultural and ecological variation in ethnobiological research: The importance of gender. J. Ethnobiol. 2005, 25, 240–279. [Google Scholar] [CrossRef]
- Müller, J.G.; Boubacar, R.; Guimbo, I.D. The “How” and “Why” of Including Gender and Age in Ethnobotanical Research and Community-Based Resource Management. Ambio 2014, 28, 7–20. [Google Scholar] [CrossRef] [PubMed]
- Howard, P.; Cuijpers, W. Gender and the Management and Conservation of Plant Biodiversity. In Encyclopaedia of Life Support Systems (EOLSS); Doelle, H.W., DaSilva, E., Eds.; EOLSS: Oxford, UK, 2002. [Google Scholar]
- Luce, D. Gender and Global Biodiversity From ’Women and Plants’ to International Law. Ph.D. Thesis, University of Ottawa, Ottawa, Canada, 2010. [Google Scholar]
- Zent, E.L. Women and Plants: Gender Relations in Biodiversity Management and Conservation. J. Ethnobiol. 2008, 25, 151–154. [Google Scholar] [CrossRef]
- Arias Gutiérrez, R.I.; González Sousa, R.; Herrera Sorzano, A.; Alemán Pérez, R.D. Diagnóstico integral de comunidades Kichwa amazónicas ecuatorianas para la elaboración de la estrategia de desarrollo sostenible. II. Indicadores socio-económicos. Centro Agrícola 2015, 42, 73–79. [Google Scholar]
- Santi Gualinga, F. Diagnóstico de la Realidad Socio-Económica de las Comunidades Kichwa de la Cuenca del Bobonaza con enfoque de Género. Bachelor’s Thesis, Universidad de Cuenca, Cuenca, Ecuador, 2015. [Google Scholar]
- GADR-Sarayaku. PODT Sarayaku. Plan. de Desarrollo y Ordenamiento Territorial de la Parroquia de Sarayakyu; GAD Pastaza: Puyo, Ecuador, 2009. [Google Scholar]
- Luzuriaga-Quichimbo, C.X. Estudio Etnobotánico en Comunidades Kichwas Amazónicas de Pastaza. Ph.D. Thesis, Universidad de Extremadura, Ecuador, Badajoz, Spain, 2017. [Google Scholar]
- Chávez, G.; Lara, R.; Moreno, M.-A. El Pueblo del Cenit.Identidad y Construcción Étnica: Informe antropológico —Jurídico Sobre los Impactos Sociales y Culturales de la Presencia de la Compañía CGC en Sarayaku; FLACSO Sede Académica de Ecuador: Quito, Ecuador, 2005. [Google Scholar]
- United-Nations Convention on Biological Diversity. Available online: https://www.cbd.int/convention/ (accessed on 24 June 2019).
- De la Torre, L.; Navarrete, H.; Muriel, P.; Marcia, M.; Balslev, H. Enciclopedia De Plantas Utiles Del Ecuador; Herbario QCA & Herbario AAU: Quito, Ecuador, 2008; Volume 1, ISBN 978-9978-77-135-8. [Google Scholar]
- Hoffman, B.; Gallaher, T. Importance indices in ethnobotany. Ethnobot. Res. Appl. 2007, 5, 201–218. [Google Scholar] [CrossRef]
- Da Silva, V.A.; Andrade, L.D.H.C.; de Albuquerque, U.P. Revising the Cultural Significance Index: The Case of the Fulni-ô in Northeastern Brazil. Field Methods 2006. [Google Scholar] [CrossRef]
- Paymal, N.; Sosa, C. Mundos Amazónicos: Pueblos y Culturas de la Amazonia Ecuatoriana; Fundación Sinchi Sach: Quito, Ecuador, 1993; ISBN 9978823654. [Google Scholar]
- Whitten, N.W. Arte, Cultura y Poder de los Canelos Quichua de la Amazonia Ecuatoriana; Departamento de Etnografía del Banco Central de Ecuador: Quito, Ecuador, 1987. [Google Scholar]
- Cerón, C.; Reyes, C.I.; Jiménez, E.D.; Simba, D.J. Plantas útiles de los Kichwa, Centro-Norte de la Amazonía Ecuatoriana. Cinchonia 2012, 12, 1–202. [Google Scholar]
- Arias-Gutierrez, R.I.; Herrera Sorzano, A.; Gonzalez Sousa, R. Amazonian indigenous settlement and local development in Pastaza, Ecuador. Noved. Población 2016, 16, 544. [Google Scholar]
- Pérez Quintana, M.; Arias Gutiérrez, R.; Sablón Cossio, N. Amazonia, Healthy Food and Rural Communities, Pastaza-Ecuador. 2017. Available online: https://www.researchgate.net/publication/312555679_Amazonia_healthy_food_and_rural_communities_Pastaza-Ecuador (accessed on 24 June 2019).
- Torres-Avilez, W.; Medeiros, P.M.D.; Albuquerque, U.P. Effect of Gender on the Knowledge of Medicinal Plants: Systematic Review and Meta-Analysis. Evidence Based Complement. Altern. Med. 2016, 2016, 1–13. [Google Scholar] [CrossRef] [PubMed]
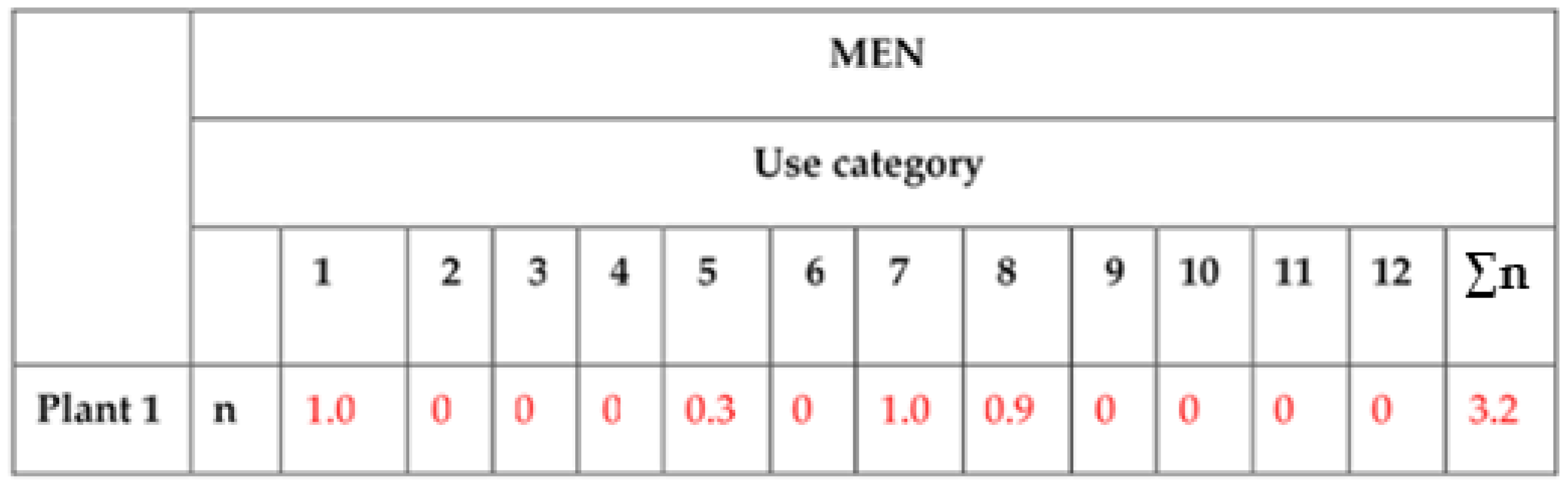
| Reference Number: Plant 1 (See List in Materials and Methods) | |||
|---|---|---|---|
| Nº People that Consider it Useful Plant: 10 Sex: Men | |||
| Category | Nº Persons Who Cite it in: | Select the Most Frequent Use (Only 1) & Sign as +F | Select the Preferred Use (Only 1) & Sign it as +P |
| 1 Food for human | 10 | +F | |
| 2 Food for animal | |||
| 3 Utensils and tools | |||
| 4 Handcrafts | |||
| 5 Construction | 3 | ||
| 6 Cultural uses | |||
| 7 Human medicine | 10 | +P | |
| 8 Veterinary uses | 9 | ||
| 9 Poisonous plants | |||
| 10 Ornamental plants | |||
| 11 Environmental uses | |||
| 12 Plants for fuel | |||
| Men | Women | |||
|---|---|---|---|---|
| ∑n | CSIm | ∑n | CSIw | |
| 1. Selaginella exaltata (Kunze) Spring | 0 | 0.00 | 1 | 0.67 |
| 2. Danaea ulei Christ | 1 | 0.67 | 0 | 0.00 |
| 3. Siparuna sp. | 2 | 1.33 | 3 | 3.00 |
| 4. Annona muricata L. | 1 | 1.33 | 2 | 3.33 |
| 5. Guatteria multinervis Wall. | 3 | 3.00 | 3 | 3.00 |
| 6. Compsoneura sprucei (A. DC.) Warb. | 1 | 2.00 | 1 | 0.67 |
| 7. Colocasia sculenta (L.) Schott | 3 | 6.00 | 3 | 6.00 |
| 8. Homalomena crinipes Engl. | 2 | 1.67 | 3 | 3.00 |
| 9. Homalomena picturata (Linden & André) Regel | 2 | 1.67 | 3 | 3.00 |
| 10. Philodendron schmidtiae Croat & Cerón | 1 | 0.67 | 2 | 1.67 |
| 11. Xanthosoma saggitifolium (L.) Schott | 2 | 1.67 | 2 | 1.67 |
| 12. Dioscorea trifida L.f. | 2 | 3.33 | 2 | 3.33 |
| 13. Carludovica palmata Ruiz & Pav. | 2 | 3.33 | 6 | 18.00 |
| 14. Cyclanthus bipartitus Poit. ex A. Rich. | 2 | 1.33 | 4 | 4.00 |
| 15. Aphandra natalia (Balslev & A.J. Hend.) Barfod | 2 | 2.67 | 4 | 8.00 |
| 16. Bactris gasipaes Kunth | 4 | 8.00 | 3 | 6.00 |
| 17. Geonoma macrostachys Mart. | 1 | 1.33 | 3 | 6.00 |
| 18. Iriartea deltoidea Ruiz & Pav. | 2 | 3.33 | 6 | 18.00 |
| 19. Mauritia flexuosa L. f. | 2 | 3.33 | 3 | 6.00 |
| 20. Oenocarpus batatua Mart. | 4 | 9.33 | 6 | 18.00 |
| 21. Ananas comosus (L.) Merr. | 0 | 0.00 | 2 | 3.33 |
| 22. Ananas lucidus Mill. | 3 | 6.00 | 2 | 1.33 |
| 23. Paspalum pilosum Lam. | 0 | 0.00 | 0 | 0.00 |
| 24. Saccharum officinarum L. | 2 | 3.33 | 2 | 3.33 |
| 25. Rhynchospora radicans (Schltdl. & Cham.) H. Pfeiff. | 0 | 0.00 | 0 | 0.00 |
| 26. Scleria melaleuca Rchb. ex Schltdl. & Cham. | 0 | 0.00 | 1 | 0.67 |
| 27. Tripogandra serrulata (Vahl) Handlos | 1 | 0.67 | 1 | 0.67 |
| 28. Costus scaber Ruiz & Pav. | 2 | 1.67 | 3 | 2.50 |
| 29. Heliconia chartacea Lane ex Barreiros | 1 | 0.67 | 1 | 0.67 |
| 30. Heliconia episcopalis Vell. | 2 | 1.67 | 2 | 1.33 |
| 31. Heliconia hirsuta L. f. | 0 | 0.00 | 1 | 0.67 |
| 32. Heliconia rostrata Ruiz & Pav. | 2 | 1.67 | 2 | 1.33 |
| 33. Heliconia shumanniana Loes. | 0 | 0.00 | 2 | 1.67 |
| 34. Heliconia velutina L. Andersson | 1 | 0.67 | 1 | 0.67 |
| 35. Calathea lutea (Aubl.) Schult. | 1 | 0.67 | 1 | 0.67 |
| 36. Musa acuminata Colla | 1 | 1.33 | 3 | 6.00 |
| 37. Zingiber officinale Roscoe | 1 | 1.33 | 2 | 3.33 |
| 38. Tetracera volubilis L. | 0 | 0.00 | 1 | 0.67 |
| 39. Garcinia macrophylla Mart. | 2 | 1.67 | 3 | 3.00 |
| 40. Alchornea triplinervia (Spreng.) Müll. Arg. | 1 | 0.67 | 1 | 0.67 |
| 41. Croton lecheri Müll. Arg. | 2 | 1.67 | 2 | 1.67 |
| 42. Manihot esculenta Crantz | 2 | 3.33 | 2 | 3.33 |
| 43. Vismia baccifera (L.) Triana & Planch. | 0 | 2.00 | 2 | 1.67 |
| 44. Lunania parviflora Spruce ex Benth. | 1 | 0.67 | 4 | 4.00 |
| 45. Bauhinia tarapotensis Benth. | 4 | 4.67 | 2 | 1.67 |
| 46. Erythrina poeppigiana (Walp.) O.F. Cook | 1 | 0.67 | 3 | 3.00 |
| 47. Inga alba (Sw.) Willd. | 3 | 2.50 | 2 | 1.33 |
| 48. Inga auristellae Harms | 3 | 3.00 | 2 | 1.33 |
| 49. Inga edulis Mart. | 2 | 3.33 | 3 | 6.00 |
| 50. Inga sapindoides Willd. | 3 | 2.50 | 2 | 1.67 |
| 51. Lonchocarpus utilis A.C. Sm. | 1 | 1.33 | 0 | 0.00 |
| 52. Piptadenia sp. | 1 | 0.67 | 2 | 1.67 |
| 53. Clarisia racemosa Ruiz & Pav. | 2 | 1.67 | 2 | 1.67 |
| 54. Perebea guianensis Aubl. | 1 | 0.67 | 3 | 2.50 |
| 55. Perebea xanthochyma H. Karst. | 2 | 1.67 | 2 | 1.67 |
| 56. Cecropia engleriana Snethl. | 0 | 0.00 | 1 | 0.67 |
| 57. Cecropia ficifolia Warb. ex Snethl. | 1 | 0.67 | 2 | 2.00 |
| 58. Aciotis purpurascens (Aubl.) Triana | 0 | 0.00 | 1 | 0.67 |
| 59. Bellucia pentamera Naudin | 1 | 0.67 | 1 | 0.67 |
| 60. Clidemia dentata Pav. ex D. Don | 1 | 0.67 | 1 | 0.67 |
| 61. Clidemia octona (Bonpl.) L.O. Williams | 1 | 0.00 | 1 | 0.67 |
| 62. Graffenrieda gracilis (Triana) L.O. Williams | 1 | 0.00 | 1 | 0.67 |
| 63. Leandra catequensis Gleason | 0 | 0.00 | 1 | 0.67 |
| 64. Miconia aureoides Cogn. | 1 | 0.67 | 2 | 1.33 |
| 65. Miconia paleacea Cogn. | 1 | 0.67 | 1 | 0.67 |
| 66. Miconia punctata (Desr.) D. Don ex DC. | 1 | 0.67 | 3 | 2.50 |
| 67. Bixa orellana L. | 4 | 9.33 | 4 | 9.33 |
| 68. Apeiba aspera Aubl. | 1 | 0.67 | 1 | 0.67 |
| 69. Ochroma pyramidale (Cav. ex Lam.) Urb. | 2 | 1.67 | 5 | 7.50 |
| 70. Theobroma cacao L. | 2 | 2.67 | 2 | 3.33 |
| 71. Theobroma subincanum Mart. | 3 | 6.00 | 3 | 6.00 |
| 72. Carica papaya L. | 2 | 3.33 | 3 | 5.00 |
| 73. Minquartia guianensis Aubl. | 1 | 0.67 | 2 | 1.33 |
| 74. Agonandra sp. | 1 | 0.67 | 3 | 3.00 |
| 75. Cyathula prostrata (L.) Blume | 1 | 0.67 | 0 | 0.00 |
| 76. Phytolacca sp. | 0 | 0.00 | 2 | 1.33 |
| 77. Gustavia longifolia Poepp. ex O. Berg | 1 | 1.33 | 1 | 1.33 |
| 78. Pouteria caimito (Ruiz & Pav.) Radlk. | 3 | 6.00 | 2 | 3.33 |
| 79. Capsicum sp. | 3 | 6.00 | 4 | 9.33 |
| 80. Nicotiana tabacum L. | 1 | 1.33 | 1 | 1.33 |
| 81. Solanum quitoense Lam. | 2 | 3.33 | 3 | 6.00 |
| 82. Witheringa solanacea L’Hér. | 0 | 0.00 | 1 | 0.67 |
| 83. Aspidosperma excelsum Benth. | 1 | 0.67 | 1 | 0.67 |
| 84. Chelonanthus alatus (Aubl.) Pulle | 0 | 0.00 | 1 | 0.67 |
| 85. Spermacoce exilis (L.O. Williams) C.D. Adams | 0 | 0.00 | 1 | 0.67 |
| 86. Spermacoce remota Lam. | 1 | 0.67 | 1 | 0.67 |
| 87. Uncaria guianensis (Aubl.) J.F. Gmel. | 1 | 0.67 | 3 | 4.00 |
| 88. Warszewiczia coccinea (Vahl) Klotzsch | 1 | 1.33 | 2 | 2.67 |
| 89. Justicia comata (L.) Lam. | 0 | 0.00 | 1 | 0.67 |
| 90. Jacaranda copaia (Aubl.) D. Don | 2 | 1.33 | 2 | 1.33 |
| 91. Besleria sp. | 0 | 0.00 | 1 | 0.67 |
| 92. Hyptis obtusiflora C. Presl ex Benth. | 0 | 0.00 | 1 | 0.67 |
| 93. Cordia alliodora (Ruiz & Pav.) Oken | 1 | 0.67 | 1 | 0.67 |
| 94. Adenostemma fosbergii R.M. King & H. Rob. | 1 | 0.67 | 1 | 0.67 |
| 95. Conyza sumatrensis (Retz.) E. Walker | 0 | 0.00 | 1 | 0.67 |
| 96. Erechtites hieraciifolius (L.) Raf. ex DC. | 1 | 0.67 | 1 | 0.67 |
| 97. Piptocoma discolor (Kunth) Pruski | 4 | 4.00 | 3 | 2.50 |
| TOTAL | 135 | 164.67 | 294 | 267.50 |
| CSIm | CSIw | CSIm + CSIw | |
|---|---|---|---|
| +20. Oenocarpus batatua Mart. | 9.33 | 18.00 | 27.33 |
| +13. Carludovica palmata Ruiz & Pav. | 3.33 | 18.00 | 21.33 |
| +18. Iriartea deltoidea Ruiz & Pav. | 3.33 | 18.00 | 21.33 |
| +67. Bixa orellana L. | 9.33 | 9.33 | 18.67 |
| +79. Capsicum sp. | 6.00 | 9.33 | 15.33 |
| +16. Bactris gasipaes Kunth | 8.00 | 6.00 | 14.00 |
| +7. Colocasia sculenta (L.) Schott | 6.00 | 6.00 | 12.00 |
| +71. Theobroma subincanum Mart. | 6.00 | 6.00 | 12.00 |
| +15. Aphandra natalia (Balslev & A.J. Hend.) Barfod | 2.67 | 8.00 | 10.67 |
| +78. Pouteria caimito (Ruiz & Pav.) Radlk. | 6.00 | 3.33 | 9.33 |
| +19. Mauritia flexuosa L. f. | 3.33 | 6.00 | 9.33 |
| +49. Inga edulis Mart. | 3.33 | 6.00 | 9.33 |
| +81. Solanum quitoense Lam. | 3.33 | 6.00 | 9.33 |
| +69. Ochroma pyramidale (Cav. ex Lam.) Urb. | 1.67 | 7.50 | 9.17 |
| +72. Carica papaya L. | 3.33 | 5.00 | 8.33 |
| 22. Ananas lucidus Mill. | 6.00 | 1.33 | 7.33 |
| +17. Geonoma macrostachys Mart. | 1.33 | 6.00 | 7.33 |
| +36. Musa acuminata Colla | 1.33 | 6.00 | 7.33 |
| 12. Dioscorea trifida L.f. | 3.33 | 3.33 | 6.67 |
| +24. Saccharum officinarum L. | 3.33 | 3.33 | 6.67 |
| +42. Manihot esculenta Crantz | 3.33 | 3.33 | 6.67 |
| 97. Piptocoma discolor (Kunth) Pruski | 4.00 | 2.50 | 6.50 |
| 45. Bauhinia tarapotensis Benth. | 4.67 | 1.67 | 6.33 |
| 5. Guatteria multinervis Wall | 3.00 | 3.00 | 6.00 |
| +70. Theobroma cacao L. | 2.67 | 3.33 | 6.00 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Luzuriaga-Quichimbo, C.X.; Hernández del Barco, M.; Blanco-Salas, J.; Cerón-Martínez, C.E.; Ruiz-Téllez, T. Plant Biodiversity Knowledge Varies by Gender in Sustainable Amazonian Agricultural Systems Called Chacras. Sustainability 2019, 11, 4211. https://doi.org/10.3390/su11154211
Luzuriaga-Quichimbo CX, Hernández del Barco M, Blanco-Salas J, Cerón-Martínez CE, Ruiz-Téllez T. Plant Biodiversity Knowledge Varies by Gender in Sustainable Amazonian Agricultural Systems Called Chacras. Sustainability. 2019; 11(15):4211. https://doi.org/10.3390/su11154211
Chicago/Turabian StyleLuzuriaga-Quichimbo, Carmen X., Míriam Hernández del Barco, José Blanco-Salas, Carlos E. Cerón-Martínez, and Trinidad Ruiz-Téllez. 2019. "Plant Biodiversity Knowledge Varies by Gender in Sustainable Amazonian Agricultural Systems Called Chacras" Sustainability 11, no. 15: 4211. https://doi.org/10.3390/su11154211
APA StyleLuzuriaga-Quichimbo, C. X., Hernández del Barco, M., Blanco-Salas, J., Cerón-Martínez, C. E., & Ruiz-Téllez, T. (2019). Plant Biodiversity Knowledge Varies by Gender in Sustainable Amazonian Agricultural Systems Called Chacras. Sustainability, 11(15), 4211. https://doi.org/10.3390/su11154211








