Environmental Sustainability: A Review of Termite Mound Soil Material and Its Bacteria
Abstract
1. Introduction
2. Termite Mound as Hotspot for Bacteria Concentration
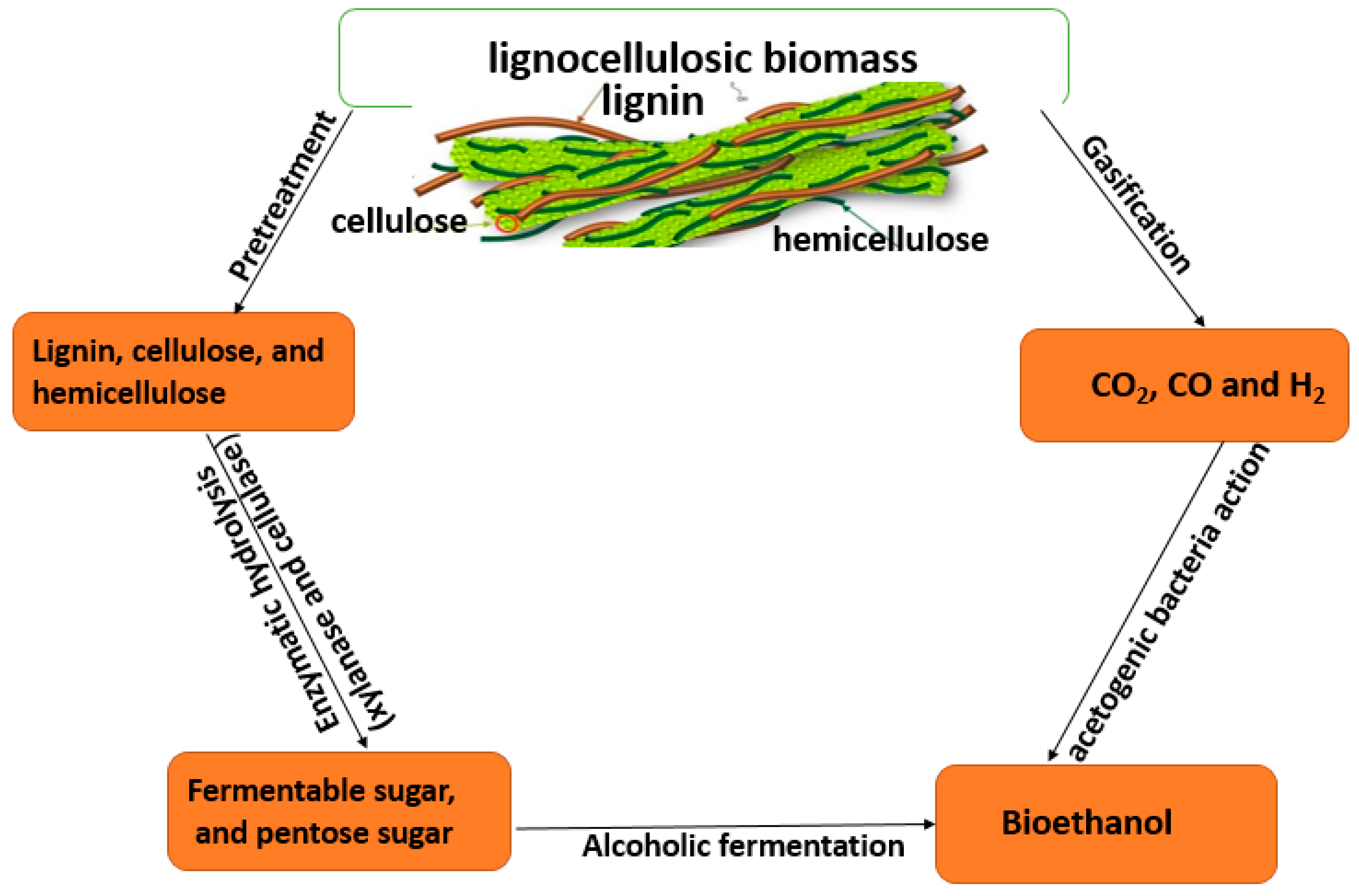
3. Degradation of Lignocellulose for Biofuel Production
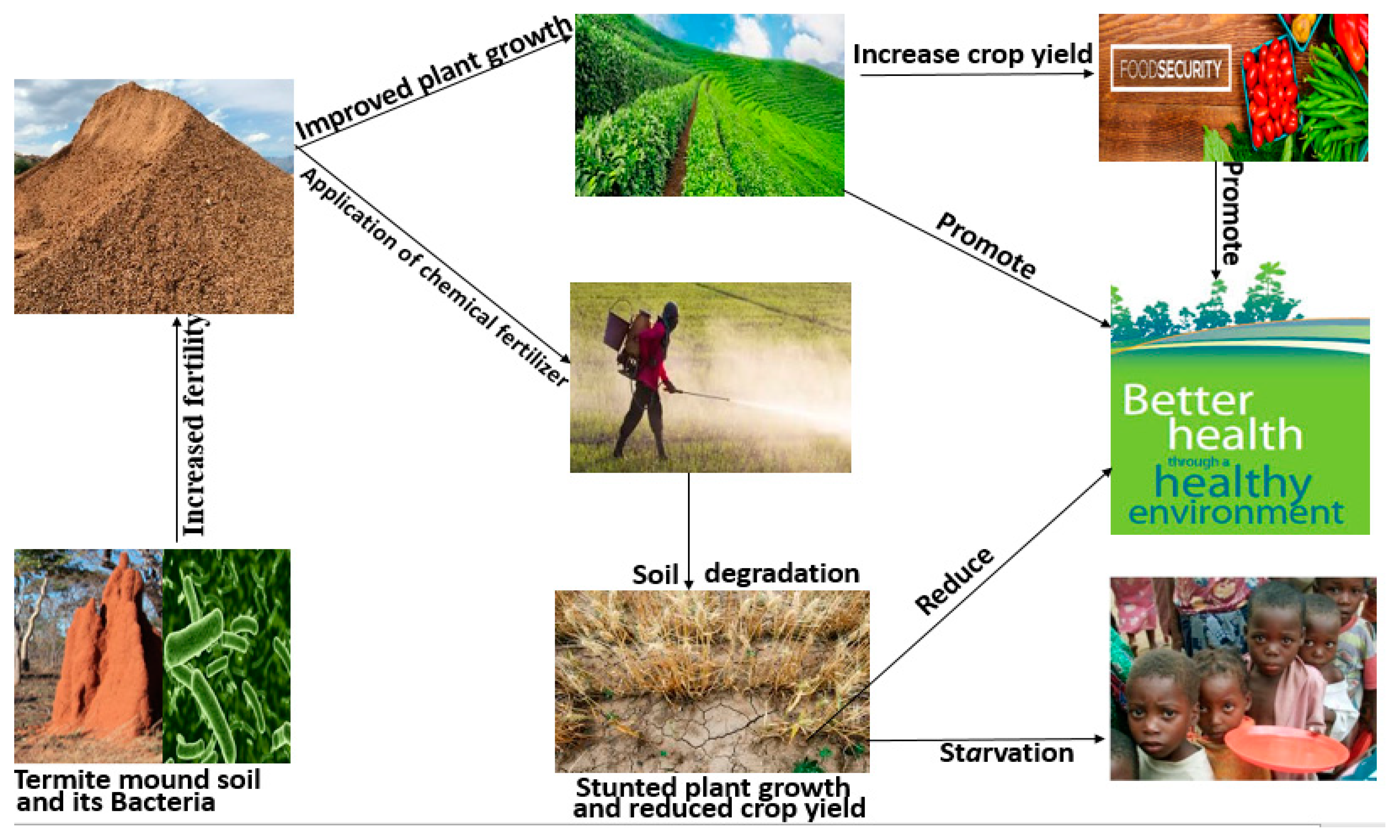
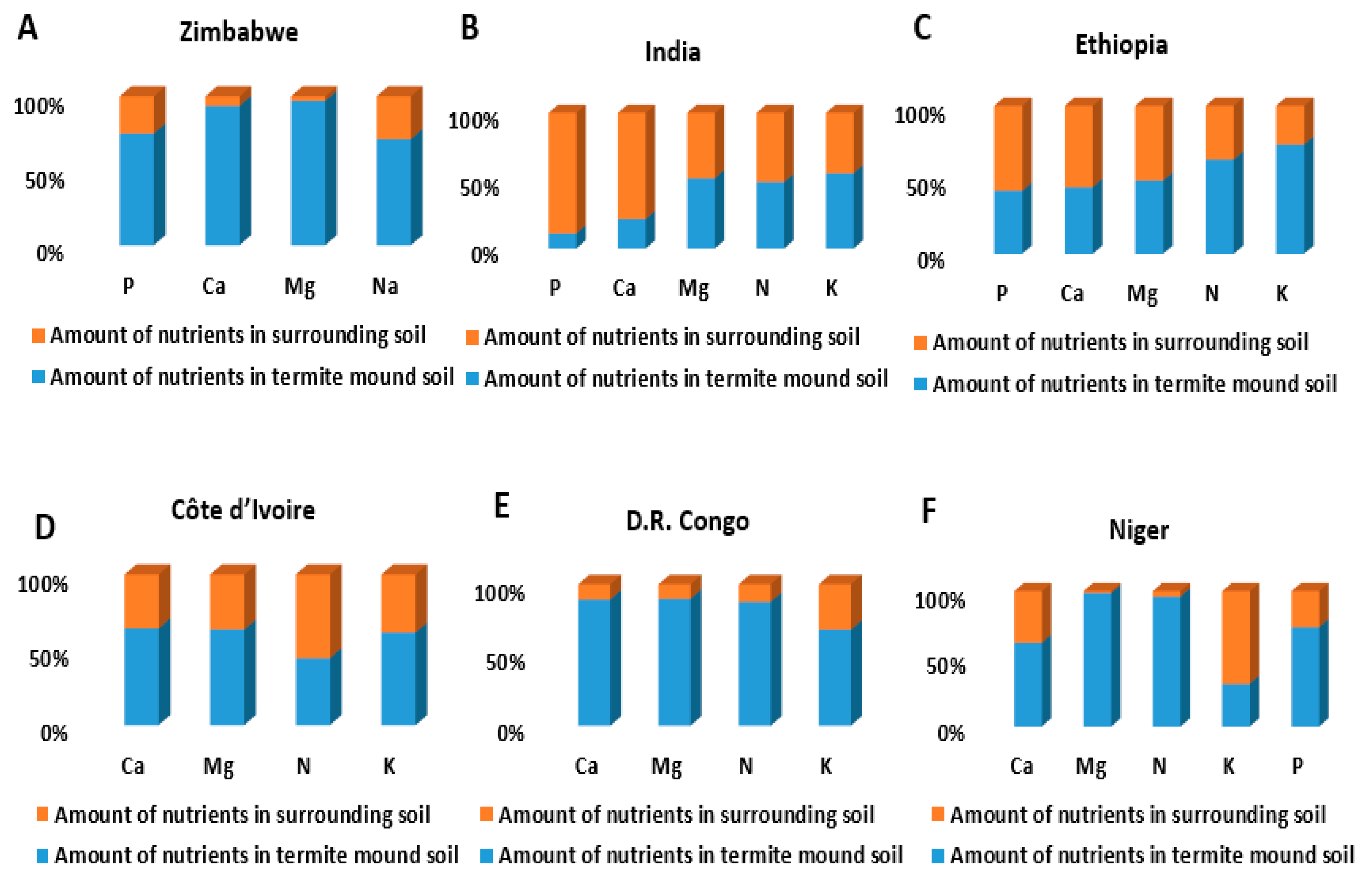
4. Termite Mound Soil as Soil Amendment
5. The Role of Termite Mound Soil and Its Bacteria in Bioremediation and Bio-Filtering
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Ethical Approval
References
- Bhardwaj, N.; Tomar, S. Indigenous knowledge of termite (Odontotermes obesus) control: A Review. Int. J. Adv. Sci. Res. 2018, 1, 124–127. [Google Scholar]
- Negassa, W.; Sileshi, G.W. Integrated soil fertility management reduces termite damage to crops on degraded soils in western Ethiopia. Agric. Ecosyst. Environ. 2018, 251, 124–131. [Google Scholar] [CrossRef]
- Calovi, D.S.; Bardunias, P.; Carey, N.; Scott Turner, J.; Nagpal, R.; Werfel, J. Surface curvature guides early construction activity in mound-building termites. Philos. Trans. R. Soc. B 2019, 374, 20180374. [Google Scholar] [CrossRef] [PubMed]
- Deke, A.L.; Adugna, W.T.; Fite, A.T. Soil physic-chemical properties in termite mounds and adjacent control soil in Miyo and Yabello Districts of Borana Zone, Southern Ethiopia. Amer. J. Agric. For. 2016, 4, 69–74. [Google Scholar]
- Dawes, T.Z. Reestablishment of ecological functioning by mulching and termite invasion in a degraded soil in an Australian savanna. Soil Biol. Biochem. 2010, 42, 1825–1834. [Google Scholar] [CrossRef]
- Khucharoenphaisan, K.; Sripairoj, N.; Sinma, K. Isolation and identification of actinomycetes from termite’s gut against human pathogen. Asian J. Anim. Vet. Adv. 2012, 7, 68–73. [Google Scholar] [CrossRef][Green Version]
- Nithyatharani, R.; Kavitha, U.S. Termite soil as bio-Indicator of soil fertility. Int. J. Res. Appl. Sci. Eng. Technol. 2018, 6, 659–661. [Google Scholar] [CrossRef]
- Jouquet, P.; Guilleux, N.; Caner, L.; Chintakunta, S.; Ameline, M.; Shanbhag, R. Influence of soil pedological properties on termite mound stability. Geoderma 2016, 262, 45–51. [Google Scholar] [CrossRef]
- Kumari, R.; Sachdev, M.; Prasad, R.; Garg, A.P.; Sharma, S.; Giang, P.H.; Varma, A. Microbiology of termite hill (mound) and soil. In Intestinal Microorganisms of Termites and Other Invertebrates; Springer: Berlin, Germany, 2006; pp. 351–372. [Google Scholar]
- Sujada, N.; Sungthong, R.; Lumyong, S. Termite nests as an abundant source of cultivable actinobacteria for biotechnological purposes. Microbes Environ. 2014, 29, 211–219. [Google Scholar] [CrossRef]
- Abe, S.S.; Watanabe, Y.; Onishi, T.; Kotegawa, T.; Wakatsuki, T. Nutrient storage in termite (Macrotermes bellicosus) mounds and the implications for nutrient dynamics in a tropical savanna Ultisol. Soil Sci. Plant Nutr. 2011, 57, 786–795. [Google Scholar] [CrossRef]
- Arhin, E.; Boadi, S.; Esoah, M.C. Identifying pathfinder elements from termite mound samples for gold exploration in regolith complex terrain of the Lawra belt, NW Ghana. J. Afr. Earth. Sci. 2015, 109, 143–153. [Google Scholar] [CrossRef]
- Dhembare, A. Physico-chemical properties of termite mound soil. Arch. Appl. Sci. Res. 2013, 5, 123–126. [Google Scholar]
- Jouquet, P.; Guilleux, N.; Shanbhag, R.R.; Subramanian, S. Influence of soil type on the properties of termite mound nests in Southern India. Appl. Soil Ecol. 2015, 96, 282–287. [Google Scholar] [CrossRef]
- Menichetti, L.; Landi, L.; Nannipieri, P.; Katterer, T.; Kirchmann, H.; Renella, G. Chemical properties and biochemical activity of colonized and abandoned litter-feeding termite (Macrotermes spp.) mounds in chromic Cambisol Area on the Borana Plateau, Ethiopia. Pedosphere 2014, 24, 399–407. [Google Scholar]
- Kumar, P.; Tilak, M.; Sivakumar, K.; Saranya, K. Studies on the assessment of major nutrients and microbial population of termite mound soil. Int. J. For. Crop Improv. 2018, 9, 13–17. [Google Scholar] [CrossRef]
- Devi, R.; Thakur, R. Screening and identification of bacteria for plant growth promoting traits from termite mound soil. J. Pharm. Phytochem. 2018, 7, 1681–1686. [Google Scholar]
- Poulsen, M.; Hu, H.; Li, C.; Chen, Z.; Xu, L.; Otani, S.; Nygaard, S.; Nobre, T.; Klaubauf, S.; Schindler, P.M.; et al. Complementary symbiont contributions to plant decomposition in a fungus-farming termite. Proc. Natl. Acad. Sci. USA 2014, 111, 14500–14505. [Google Scholar] [CrossRef]
- Benndorf, R.; Guo, H.; Sommerwerk, E.; Weigel, C.; Garcia-Altares, M.; Martin, K.; Hu, H.; Küfner, M.; de Beer, Z.; Poulsen, M. Natural products from actinobacteria associated with fungus-growing termites. Antibiotics 2018, 7, 83. [Google Scholar] [CrossRef]
- Karak, T.; Sonar, I.; Paul, R.K.; Das, S.; Boruah, R.K.; Dutta, A.K.; Das, D.K. Composting of cow dung and crop residues using termite mounds as bulking agent. Bioresour. Technol. 2014, 169, 731–741. [Google Scholar] [CrossRef]
- Omobowale, M.O.; Armstrong, P.R.; Mijinyawa, Y.; Igbeka, J.C.; Maghirang, E.B. Maize storage in termite mound clay, concrete, and steel silos in the humid tropics: Comparison and effect on bacterial and fungal counts. Trans. Asabe 2016, 59, 1039–1048. [Google Scholar]
- Krishanti, N.P.R.A.; Zulfina, D.; Wikantyoso, B.; Zulfitri, A.; Yusuf, S. Antimicrobial production by an actinomycetes isolated from the termite nest. J. Trop. Life Sci. 2018, 8, 279–288. [Google Scholar]
- Enagbonma, B.J.; Babalola, O.O. Potentials of termite mound soil bacteria in ecosystem engineering for sustainable agriculture. Ann. Microbiol. 2019, 69, 211–219. [Google Scholar] [CrossRef]
- Yêyinou Loko, L.E.; Orobiyi, A.; Agre, P.; Dansi, A.; Tamò, M.; Roisin, Y. Farmers’ perception of termites in agriculture production and their indigenous utilization in Northwest Benin. J. Ethnobiol. Ethnomed. 2017, 13, 64. [Google Scholar] [CrossRef]
- Bama, P.S.; Ravindran, A.D. Dynamics of P sorption and solubilising activity in termite nest material. Asian J. Res. Soc. Sci. Hum. 2012, 2, 231–237. [Google Scholar]
- Smith, F. Environmental Sustainability: Practical Global Applications; CRC Press: Boca Raton, FL, USA, 2019; p. 279. [Google Scholar]
- Miyagawa, S.; Koyama, Y.; Kokubo, M.; Matsushita, Y.; Adachi, Y.; Sivilay, S.; Kawakubo, N.; Oba, S. Indigenous utilization of termite mounds and their sustainability in a rice growing village of the Central Plain of Laos. J. Ethnobiol. Ethnomed. 2011, 7, 24. [Google Scholar] [CrossRef]
- Kaiser, D.; Lepage, M.; Konaté, S.; Linsenmair, K.E. Ecosystem services of termites (Blattoidea: Termitoidae) in the traditional soil restoration and cropping system Zaï in northern Burkina Faso (West Africa). Agric. Ecosyst. Environ. 2017, 236, 198–211. [Google Scholar] [CrossRef]
- Ahmad, T.; Nabi, S.; Humera, Q. Biotechnology: A tool in termite management. In Termites and Sustainable Management: Economic Losses and Management; Khan, M.A., Ahmad, W., Eds.; Springer International Publishing: Cham, Germany, 2018; Volume 2, pp. 289–315. [Google Scholar]
- Gougoulias, C.; Clark, J.M.; Shaw, L.J. The role of soil microbes in the global carbon cycle: Tracking the below-ground microbial processing of plant-derived carbon for manipulating carbon dynamics in agricultural systems. J. Sci. Food Agric. 2014, 94, 2362–2371. [Google Scholar] [CrossRef]
- Haynes, R.J. Nature of the belowground ecosystem and its development during pedogenesis. Adv. Agron. 2014, 127, 43–109. [Google Scholar]
- Fall, S.; Hamelin, J.; Ndiaye, F.; Assigbetse, K.; Aragno, M.; Chotte, J.L.; Brauman, A. Differences between bacterial communities in the gut of a soil-feeding termite (Cubitermes niokoloensis) and its mounds. Appl. Environ. Microbiol. 2007, 73, 5199–5208. [Google Scholar] [CrossRef]
- Chauhan, A.K.; Maheshwari, D.K.; Dheeman, S.; Bajpai, V.K. Termitarium-inhabiting Bacillus spp. enhanced plant growth and bioactive component in turmeric (Curcuma longa L.). Curr. Microbiol. 2017, 74, 174–192. [Google Scholar] [CrossRef]
- Manjula, A.; Pushpanathan, M.; Sathyavathi, S.; Gunasekaran, P.; Rajendhran, J. Comparative analysis of microbial diversity in termite gut and termite nest using ion sequencing. Curr. Microbiol. 2016, 72, 267–275. [Google Scholar] [CrossRef]
- Spain, A.V.; Gordon, V.; Reddell, P.; Correll, R. Ectomycorrhizal fungal spores in the mounds of tropical Australian termites (Isoptera). Eur. J. Soil Biol. 2004, 40, 9–14. [Google Scholar] [CrossRef]
- Makonde, H.M.; Mwirichia, R.; Osiemo, Z.; Boga, H.I.; Klenk, H.P. 454 Pyrosequencing-based assessment of bacterial diversity and community structure in termite guts, mounds and surrounding soils. SpringerPlus 2015, 4, 471. [Google Scholar] [CrossRef]
- Manjula, A.; Sathyavathi, S.; Pushpanathan, M.; Gunasekaran, P.; Rajendhran, J. Microbial diversity in termite nest. Curr. Sci. 2014, 106, 1430–1434. [Google Scholar]
- Yilmaz, N.; Atmanli, A. Sustainable alternative fuels in aviation. Energy 2017, 140, 1378–1386. [Google Scholar] [CrossRef]
- Batista-García, R.A.; Del Rayo Sánchez-Carbente, M.; Talia, P.; Jackson, S.A.; O’Leary, N.D.; Dobson, A.D.; Folch-Mallol, J.L. From lignocellulosic metagenomes to lignocellulolytic genes: Trends, challenges and future prospects. Biofuels Bioprod. Bioref. 2016, 10, 864–882. [Google Scholar] [CrossRef]
- Williams, C.L.; Westover, T.L.; Emerson, R.M.; Tumuluru, J.S.; Li, C. Sources of biomass feedstock variability and the potential impact on biofuels production. Bioenergy Res. 2016, 9, 1–14. [Google Scholar] [CrossRef]
- Molino, A.; Larocca, V.; Chianese, S.; Musmarra, D. Biofuels production by biomass gasification: A review. Energies 2018, 11, 811. [Google Scholar] [CrossRef]
- Goldemberg, J.; Coelho, S.T.; Guardabassi, P.; Nastari, P.M. Bioethanol from Sugar: The Brazilian Experience; Springer Nature: New York, NY, USA, 2019; pp. 925–954. [Google Scholar]
- Nigam, P.S.; Singh, A. Production of liquid biofuels from renewable resources. Prog. Energy Com. Sci. 2011, 37, 52–68. [Google Scholar] [CrossRef]
- Sethi, A.; Scharf, M.E. Biofuels: Fungal, bacterial and insect degraders of lignocellulose. In e LS; John Wiley & Sons, Ltd.: Chichester, UK, 2013; p. 19. [Google Scholar]
- Sharma, H.K.; Xu, C.; Qin, W. Biological pretreatment of lignocellulosic biomass for biofuels and bioproducts: An overview. Waste Biomass Valorization 2019, 10, 235–251. [Google Scholar] [CrossRef]
- Putro, J.N.; Soetaredjo, F.E.; Lin, S.-Y.; Ju, Y.-H.; Ismadji, S. Pretreatment and conversion of lignocellulose biomass into valuable chemicals. RSC Adv. 2016, 6, 46834–46852. [Google Scholar] [CrossRef]
- Moreno, A.D.; Ibarra, D.; Alvira, P.; Tomás-Pejó, E.; Ballesteros, M. A review of biological delignification and detoxification methods for lignocellulosic bioethanol production. Crit. Rev. Biotechnol. 2015, 35, 342–354. [Google Scholar] [CrossRef]
- Elshahed, M.S. Microbiological aspects of biofuel production: Current status and future directions. J. Adv. Res. 2010, 1, 103–111. [Google Scholar] [CrossRef]
- Talia, P.; Arneodo, J. Lignocellulose degradation by termites. In Termites and Sustainable Management; Springer: Cham, Germany, 2018; pp. 101–117. [Google Scholar]
- Varghese, L.M.; Agrawal, S.; Sharma, D.; Mandhan, R.P.; Mahajan, R. Cost-effective screening and isolation of xylano-cellulolytic positive microbes from termite gut and termitarium. Biotechnology 2017, 7, 108. [Google Scholar] [CrossRef]
- Koeck, D.E.; Pechtl, A.; Zverlov, V.V.; Schwarz, W.H. Genomics of cellulolytic bacteria. Curr. Opin. Biotechnol. 2014, 29, 171–183. [Google Scholar] [CrossRef]
- Bandounas, L.; Wierckx, N.J.; De Winde, J.H.; Ruijssenaars, H.J. Isolation and characterization of novel bacterial strains exhibiting ligninolytic potential. BMC Biotechnol. 2011, 11, 94. [Google Scholar] [CrossRef]
- Kumar, A.; Singh, J.; Baskar, C. Lignocellulosic biomass for bioethanol production through microbes: Strategies to improve process efficiency. In Prospects of Renewable Bioprocessing in Future Energy Systems; Rastegari, A.A., Yadav, A.N., Gupta, A., Eds.; Springer International Publishing: Cham, Germany, 2019; pp. 357–386. [Google Scholar]
- You, Y.; Wang, J.; Huang, X.; Tang, Z.; Liu, S.; Sun, O.J. Relating microbial community structure to functioning in forest soil organic carbon transformation and turnover. Ecol. Evol. 2014, 4, 633–647. [Google Scholar] [CrossRef]
- Traoré, S.; Bottinelli, N.; Aroui, H.; Harit, A.; Jouquet, P. Termite mounds impact soil hydrostructural properties in southern Indian tropical forests. Pedobiologia 2019, 74, 1–6. [Google Scholar] [CrossRef]
- Fall, S.; Brauman, A.; Chotte, J.-L. Comparative distribution of organic matter in particle and aggregate size fractions in the mounds of termites with different feeding habits in Senegal: Cubitermes niokoloensis and Macrotermes bellicosus. Appl. Soil Ecol. 2001, 17, 131–140. [Google Scholar] [CrossRef]
- Fall, S.; Nazaret, S.; Chotte, J.L.; Brauman, A. Bacterial density and community structure associated with aggregate size fractions of soil-feeding termite mounds. Microb. Ecol. 2004, 48, 191–199. [Google Scholar] [CrossRef]
- Dhembare, A.; Pokale, A. Physico-Chemical Properties of Termite Mound Soil from Lohare, Ahmednagar Maharashtra; International E Publication: Ahmednagar Maharashtra, India, 2013; p. 243. [Google Scholar]
- Muvengwi, J.; Mbiba, M.; Nyenda, T. Termite mounds may not be foraging hotspots for mega-herbivores in a nutrient-rich matrix. J. Trop. Ecol. 2013, 29, 551–558. [Google Scholar] [CrossRef]
- Jouquet, P.; Tessier, D.; Lepage, M. The soil structural stability of termite nests: Role of clays in Macrotermes bellicosus (Isoptera, Macrotermitinae) mound soils. Eur. J. Soil Biol. 2004, 40, 23–29. [Google Scholar] [CrossRef]
- Adhikary, N.; Erens, H.; Weemaels, L.; Deweer, E.; Mees, F.; Mujinya, B.B.; Baert, G.; Boeckx, P.; Van Ranst, E. Effects of spreading out termite mound material on ferralsol fertility, Katanga, DR Congo. Comm. Soil Sci. Plant Anal. 2016, 47, 1089–1100. [Google Scholar] [CrossRef]
- Garba, M.; Cornelis, W.M.; Steppe, K. Effect of termite mound material on the physical properties of sandy soil and on the growth characteristics of tomato (Solanum lycopersicum L.) in semi-arid Niger. Plant Soil 2011, 338, 451–466. [Google Scholar] [CrossRef]
- Batalha, L.; Da Silva Filho, D.; Martius, C. Using termite nests as a source of organic matter in agrosilvicultural production systems in Amazonia. Sci. Agric. 1995, 52, 318–325. [Google Scholar] [CrossRef]
- Watson, J. The use of mounds of the termite Macrotermes falciger (Gerstäcker) as a soil amendment. Eur. J. Soil Sci. 1977, 28, 664–672. [Google Scholar] [CrossRef]
- Bama, P.S.; Ravindran, A.D. Influence of Combined Termite Mound Materials and Inorganic Fertilizers on Growth Parameters of Maize under Non Sterilized Pot Culture Study. Elixir Appl. Zool. 2018, 125, 52303–52305. [Google Scholar]
- Kisa, M.; Duponnois, R.; Assikbetse, K.; Ramanankierana, H.; Thioulouse, J.; Lepage, M. Litter-forager termite mounds enhance the ectomycorrhizal symbiosis between Acacia holosericea A. Cunn. Ex G. Don and Scleroderma dictyosporum isolates. FEMS Microbiol. Ecol. 2006, 56, 292–303. [Google Scholar]
- Suzuki, S.; Noble, A.D.; Ruaysoongnern, S.; Chinabut, N. Improvement in water-holding capacity and structural stability of a sandy soil in Northeast Thailand. Arid Land Res. Manag. 2007, 21, 37–49. [Google Scholar] [CrossRef]
- Das, S.; Dash, H.R.; Chakraborty, J. Genetic basis and importance of metal resistant genes in bacteria for bioremediation of contaminated environments with toxic metal pollutants. Appl. Microbiol. Biotechnol. 2016, 100, 2967–2984. [Google Scholar] [CrossRef]
- Dangi, A.K.; Sharma, B.; Hill, R.T.; Shukla, P. Bioremediation through microbes: Systems biology and metabolic engineering approach. Crit. Rev. Biotechnol. 2019, 39, 79–98. [Google Scholar] [CrossRef]
- Burd, G.I.; Dixon, D.G.; Glick, B.R. Plant growth-promoting bacteria that decrease heavy metal toxicity in plants. Can. J. Microbiol. 2000, 46, 237–245. [Google Scholar] [CrossRef]
- Wasi, S.; Tabrez, S.; Ahmad, M. Use of Pseudomonas spp. for the bioremediation of environmental pollutants: A review. Environ. Monit. Assess. 2013, 185, 8147–8155. [Google Scholar] [CrossRef]
- Duponnois, R.; Kisa, M.; Assigbetse, K.; Prin, Y.; Thioulouse, J.; Issartel, M.; Moulin, P.; Lepage, M. Fluorescent pseudomonads occuring in Macrotermes subhyalinus mound structures decrease Cd toxicity and improve its accumulation in sorghum plants. Sci. Total Environ. 2006, 370, 391–400. [Google Scholar] [CrossRef]
- Abdus-Salam, N.; Itiola, A.D. Potential application of termite mound for adsorption and removal of Pb(II) from aqueous solutions. J. Iran. Chem. Soc. 2012, 9, 373–382. [Google Scholar] [CrossRef]
- Fufa, F.; Alemayehu, E.; Lennartz, B. Defluoridation of groundwater using termite mound. Water Air Soil Pollut. 2013, 224, 1552. [Google Scholar] [CrossRef]
- Fufa, F.; Alemayehu, E.; Lennartz, B. Sorptive removal of arsenate using termite mound. J. Environ. Manag. 2014, 132, 188–196. [Google Scholar] [CrossRef]
- Abdus-Salam, N.; Bello, M.O. Kinetics, thermodynamics and competitive adsorption of lead and zinc ions onto termite mound. Int. J. Environ. Sci. Technol. 2015, 12, 3417–3426. [Google Scholar] [CrossRef]
- Ayanda, O.S.; Amodu, O.S.; Adubiaro, H.; Olutona, G.O.; Ebenezer, O.T.; Nelana, S.M.; Naidoo, E.B. Effectiveness of termite hill as an economic adsorbent for the adsorption of alizarin red dye. J. Water Reuse Desalin. 2019, 9, 83–93. [Google Scholar] [CrossRef]
- Araújo, B.; Reis, J.; Rezende, E.; Mangrich, A.; Wisniewski, A., Jr.; Dick, D.; Romão, L. Application of termite nest for adsorption of Cr (VI). J. Environ. Manag. 2013, 129, 216–223. [Google Scholar] [CrossRef]
- Da Costa, R.R.; Hu, H.; Pilgaard, B.; Vreeburg, S.M.; Schückel, J.; Pedersen, K.S.; Kračun, S.K.; Busk, P.K.; Harholt, J.; Sapountzis, P. Enzyme activities at different stages of plant biomass decomposition in three species of fungus-growing termites. Appl. Environ. Microbiol. 2018, 84, e01815–e01817. [Google Scholar] [CrossRef]
- Gomati, V.; Ramasamy, K.; Kumar, K.; Sivaramaiah, N.; Mula, R.V. Green house gas emissions from termite ecosystem. Afr. J. Environ. Sci. Technol. 2011, 5, 56–64. [Google Scholar]
- Ho, A.; Erens, H.; Mujinya, B.B.; Boeckx, P.; Baert, G.; Schneider, B.; Frenzel, P.; Boon, N.; Van Ranst, E. Termites facilitate methane oxidation and shape the methanotrophic community. Appl. Environ. Microbiol. 2013, 79, 7234–7240. [Google Scholar] [CrossRef]
- Jamali, H.; Livesley, S.; Hutley, L.B.; Fest, B.; Arndt, S. The relationships between termite mound CH4/CO2 emissions and internal concentration ratios are species specific. Biogeosciences 2013, 10, 2229–2240. [Google Scholar] [CrossRef]
- Jamali, H.; Livesley, S.J.; Dawes, T.Z.; Hutley, L.B.; Arndt, S.K. Termite mound emissions of CH4 and CO2 are primarily determined by seasonal changes in termite biomass and behaviour. Oecologia 2011, 167, 525–534. [Google Scholar] [CrossRef]
- Sugimoto, A.; Inoue, T.; Kirtibutr, N.; Abe, T. Methane oxidation by termite mounds estimated by the carbon isotopic composition of methane. Global Biogeochem. Cycles 1998, 12, 595–605. [Google Scholar] [CrossRef]
- Nauer, P.A.; Hutley, L.B.; Arndt, S.K. Termite mounds mitigate half of termite methane emissions. Proc. Nat. Acad. Sci. USA 2018, 115, 13306–13311. [Google Scholar] [CrossRef]
- Knief, C.; Dunfield, P.F. Response and adaptation of different methanotrophic bacteria to low methane mixing ratios. Environ. Microbiol. 2005, 7, 1307–1317. [Google Scholar] [CrossRef]
| Molecular Method Used | Bacteria Reported | Reference |
|---|---|---|
| 454-pyrosequencing based analysis of 16S ribosomal ribonucleic acid gene sequences | Nitrospirae, Cyanobacteria, Bacteroidetes, Spirochaetes, Actinobacteria, Fibrobacteres, Candidate division TM7, Chloroflexi, Proteobacteria, WCHB1-60, Elusimicrobia, Planctomycetes, Spirochaetes, Chlorobi, Gemmatimonadetes, Armatimonadetes, Acidobacteria, SM2F11, Firmicutes, Candidate division WS3 | [36] |
| PCR-denaturing gradient gel electrophoresis analysis, cloning and sequencing of PCR-amplified 16S rRNA gene fragments | Firmicutes, Actinobacteria, Chloroflexi | [32] |
| Ion Sequencing of V3 hypervariable region of 16S ribosomal deoxyribonucleic acid | Cyanobacteria, Proteobacteria, Chlorobi, Actinobacteria, Deinococcus-Thermus, Firmicutes, Synergistetes, Spirochaetes, and Bacteroidetes, | [34] |
| Bacterial tag encoded amplicon pyrosequencing | Gemmatimonadetes, Fibrobacteres, Nitrospirae, Verrucomicrobia, Bacteroidetes, Actinobacteria, Deinococcus, Planctomycetes, Chlamydiae, Firmicutes, Chloroflexi, Tenericutes, Acidobacteria, and Proteobacteria | [37] |
| Isolation and cultivation | Pseudonocardia, Nocardia, Micromonospora, Streptomyces, and Amycolatopsis | [10] |
| Isolation and cultivation | Bacillus, Pseudomonas, Nocardia, and Streptomyces | [16] |
| Test Plant | Effect on the Plant | Reference |
|---|---|---|
| Solanum lycopersicum | The soil amended with termite mound soils resulted in better plant height, as well as an increase in leave number, fruits, and dry matter than those plant grown on unamended soil. | [62] |
| Oryza sativa L. and Phaseolus vulgaris L. | Improve the growth of Oryza sativa L. and Phaseolus vulgaris L. | [27] |
| Solatium melongena | Combined use of 200 g of termite mound material with NPK led to a substantial increase in Solatium melongena production. | [63] |
| Lolium perenne | Lolium perenne gave higher dry-matter yields with substrates derived from termite mounds than the comparable soil. | [64] |
| Zea mays | Combined use of termite mound materials and inorganic fertilization significantly increased the Zea mays growth and yield. | [65] |
| Acacia holosericea | Termite mound materials consisting of Pseudomonas monteilii species enhanced the ectomycorrhizal development between Acacia holosericea and Scleroderma dictyosporum. | [66] |
| Sorghum sudanensis | Combining sandy soil with termite mound materials at a proportion of 120 Mg/ha improved porosity and transformed the pore size distribution, thus causing a stepping up in the obtainable water content for the crop growth. | [67] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Enagbonma, B.J.; Babalola, O.O. Environmental Sustainability: A Review of Termite Mound Soil Material and Its Bacteria. Sustainability 2019, 11, 3847. https://doi.org/10.3390/su11143847
Enagbonma BJ, Babalola OO. Environmental Sustainability: A Review of Termite Mound Soil Material and Its Bacteria. Sustainability. 2019; 11(14):3847. https://doi.org/10.3390/su11143847
Chicago/Turabian StyleEnagbonma, Ben Jesuorsemwen, and Olubukola Oluranti Babalola. 2019. "Environmental Sustainability: A Review of Termite Mound Soil Material and Its Bacteria" Sustainability 11, no. 14: 3847. https://doi.org/10.3390/su11143847
APA StyleEnagbonma, B. J., & Babalola, O. O. (2019). Environmental Sustainability: A Review of Termite Mound Soil Material and Its Bacteria. Sustainability, 11(14), 3847. https://doi.org/10.3390/su11143847






