The Influence of Different Levels of Sodium Nitrite on the Safety, Oxidative Stability, and Color of Minced Roasted Beef
Abstract
:1. Introduction
2. Materials and Methods
2.1. Preparation and Storage of Product
2.2. Analysis of Product Safety
2.2.1. Water Activity and pH Value Measurement
2.2.2. Microbial Analysis
2.2.3. N-Nitrosamine Content
2.3. Analysis of Oxidation Stability
2.3.1. Measurement of Thiobarbituric Acid Reactive Substances (TBARS) Values
2.3.2. Measurement of Oxidation–Reduction Potential (ORP)
2.4. Analysis of Color
2.4.1. Total Pigments (OZB) and Heme Iron (Fe) Analyses
2.4.2. Product Color Evaluation
2.5. Statistical Procedure
3. Results
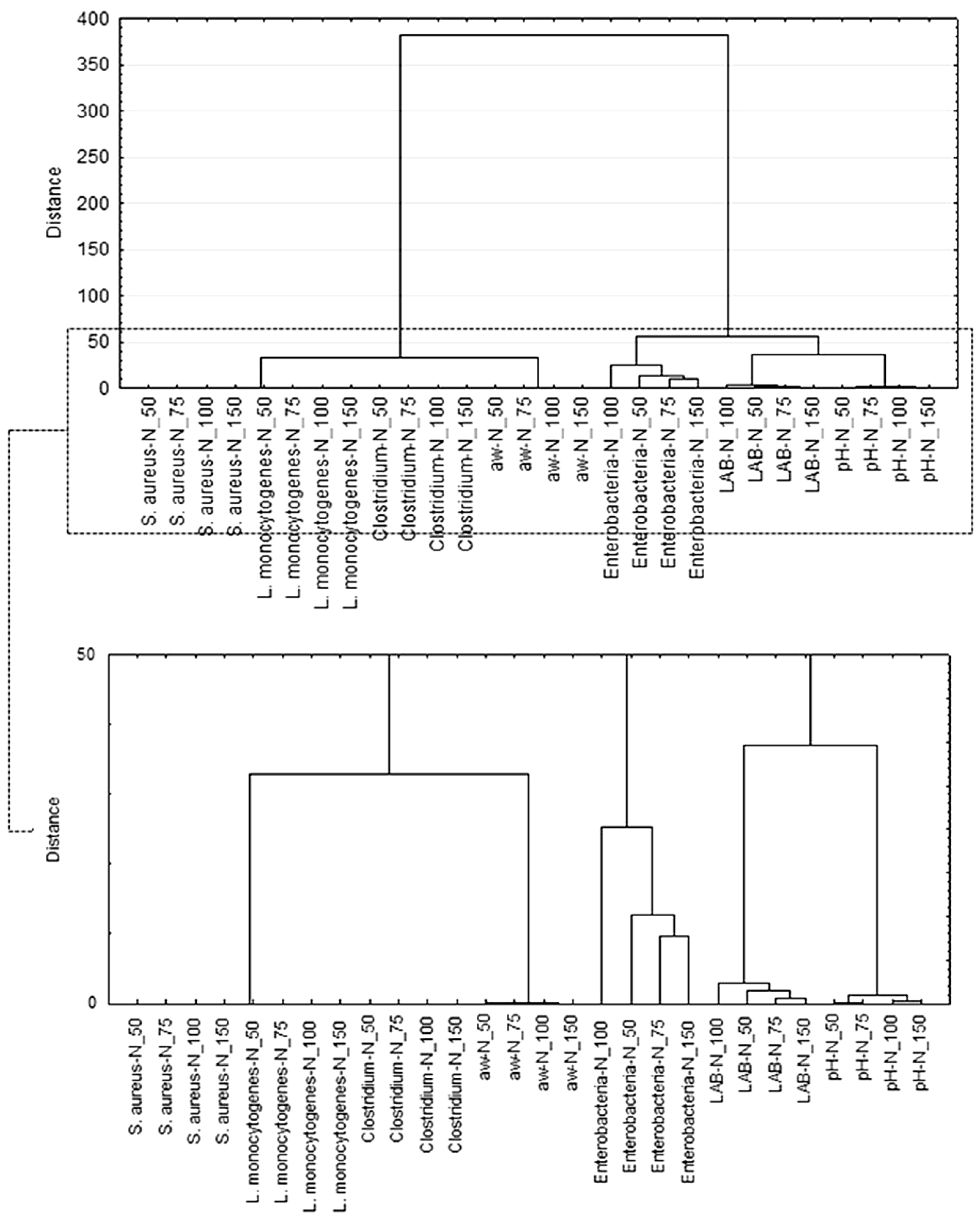
3.1. Determination of Product Safety
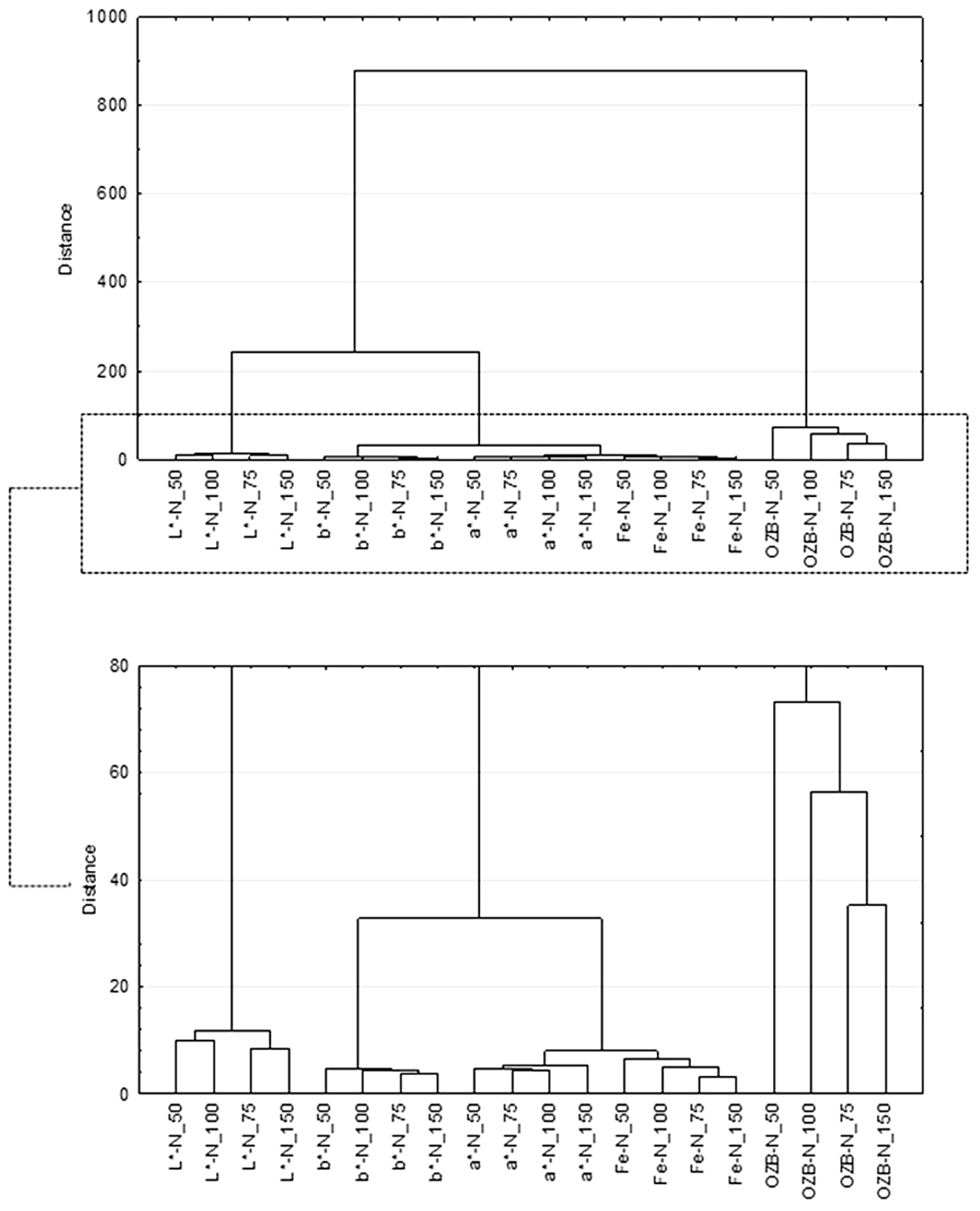
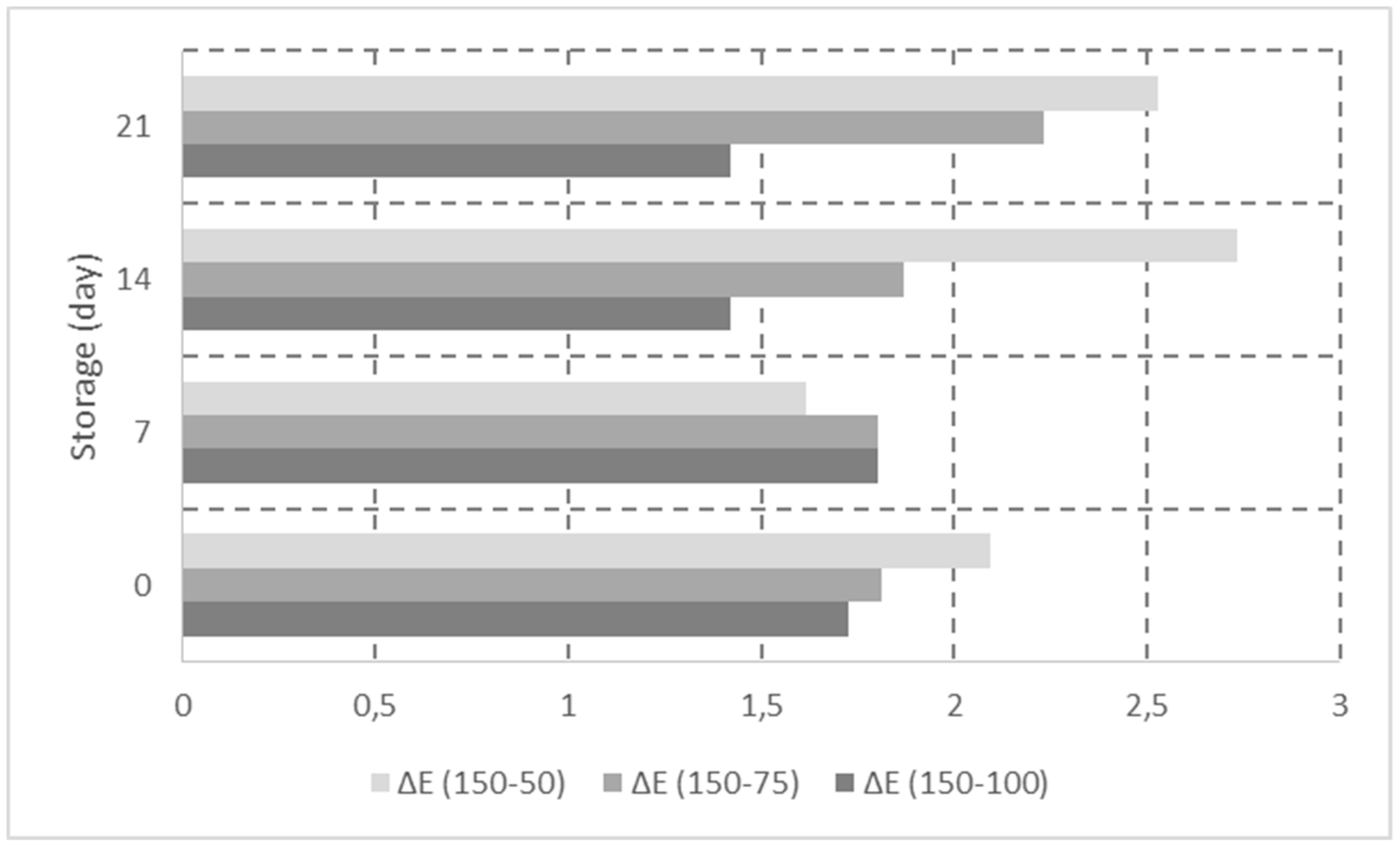
3.2. Determination of Color Parameters
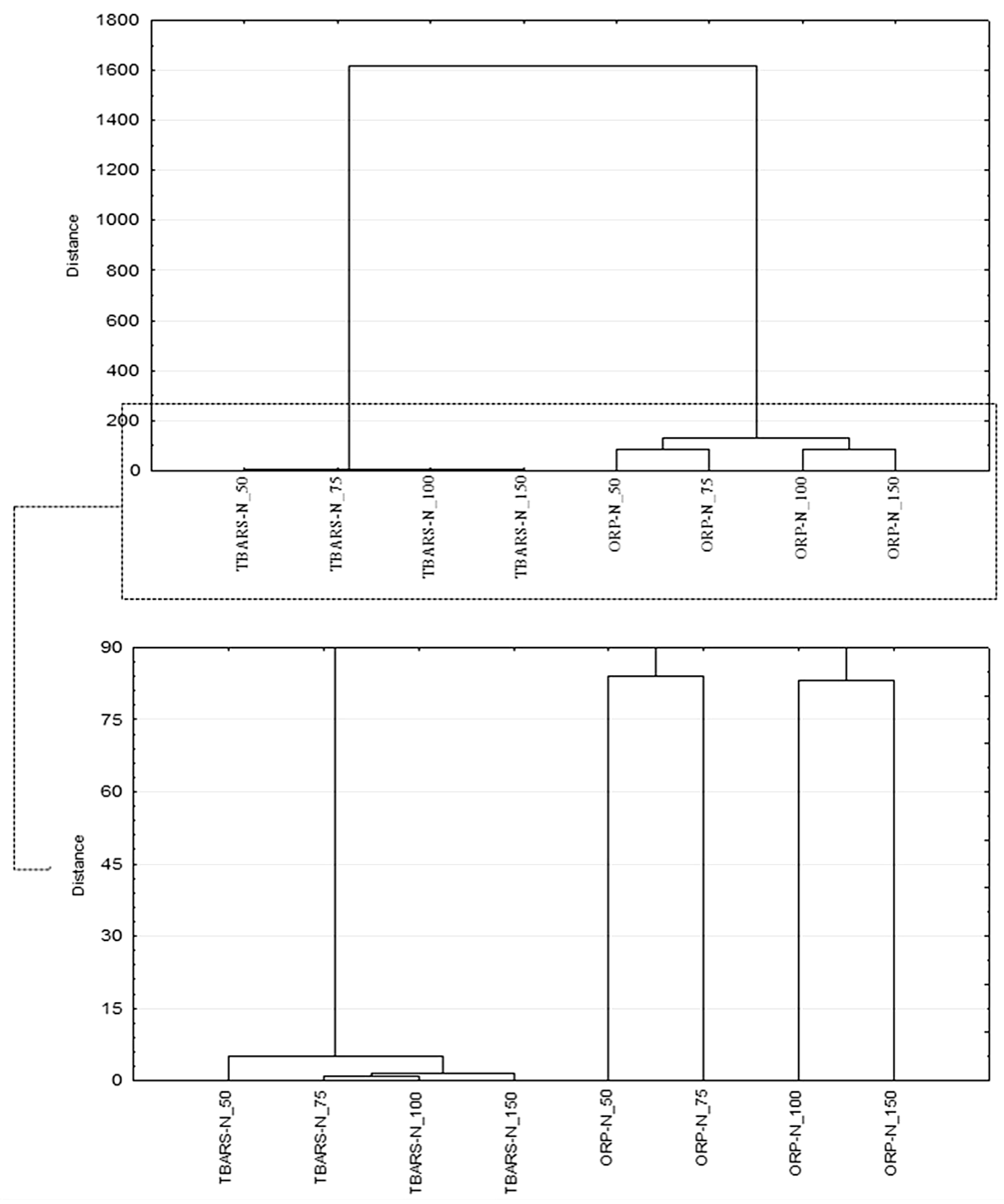
3.3. Determination of Oxidative Stability
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Püssa, T. Toxicological issues associated with production and processing of meat. Meat Sci. 2013, 95, 844–853. [Google Scholar] [CrossRef] [PubMed]
- Directive. Directive 2006/52/EC of the European Parliament and of the Council of 5 July 2006 amending Directive 95/2/EC on food additives other than colours and sweeteners and Directive 95/35/EC on sweeteners for use in foodstuffs. Off. J. Eur. Union 2006, 204, 10–22. [Google Scholar]
- EU Commission. Commission regulation (EU) n° 1129/2011 of 11 November 2011 amending Annex II to Regulation (EC) n° 1333/2008 of the European Parliament and of the Council by establishing a Union list of food additives. Off. J. Eur. Union 2011, L295, 1–177. [Google Scholar]
- MacDougall, D.B.; Hetherington, M.J. The minimum quantity of nitrite required to stain sliced and homogenised cooked pork. Meat Sci. 1992, 31, 201–209. [Google Scholar] [CrossRef]
- Sindelar, J.J.; Milkowski, A.L. Human safety controversies surrounding nitrate and nitrite in the diet. Nitric Oxide 2012, 26, 259–266. [Google Scholar] [CrossRef]
- Villaverde, A.; Ventanas, J.; Estévez, M. Nitrite promotes protein carbonylation and Strecker aldehyde formation in experimental fermented sausages: Are both events connected? Meat Sci. 2014, 98, 665–672. [Google Scholar] [CrossRef] [PubMed]
- Berardo, A.; De Maere, H.; Stavropoulou, D.A.; Rysmna, T.; Leroy, F.; De Smet, S. Differential Effects of the Curring Agents Sodium Ascorbate and Sodium Nitrite on Protein Oxidation in Dry Fermented Sausages. In Proceedings of the 61st International Congress of Meat Science and Technology, Clermont-Ferrand, France, 23–28 August 2015. [Google Scholar]
- Majou, D.; Christieans, S. Mechanisms of the bactericidal effects of nitrate and nitrite in cured meats. Meat Sci. 2018, 145, 273–284. [Google Scholar] [CrossRef]
- FCEC. Study on the Monitoring of the Implementation of Directive 2006/52/EC as Regards the Use of Nitrites by Industry in Different Categories of Meat Products: Final Report. Available online: http://www.fecic.es/img/galeria/file/BUTLLETi%20INTERNACIONAL/ARXIUS%20BUTLLETI%20INTERNACIONAL/setmana%205/05.pdf (accessed on 12 February 2019).
- Alahakoon, A.; Jayasena, D.D.; Ramachandra, S.; Jo, C. Alternatives to nitrite in processed meat: Up to date. Trends Food Sci. Technol. 2015, 45, 37–49. [Google Scholar] [CrossRef]
- ISO. ISO 15214:1998: Microbiology of Food and Animal Feeding Stuffs—Horizontal Method for the Enumeration of Mesophilic Lactic Acid Bacteria—Colony-Count Technique at 30°C; ISO: Geneva, Switzerland, 1998. [Google Scholar]
- ISO. ISO 7937:2004: Microbiology of Food and Animal Feeding Stuffs—Horizontal Method for the Enumeration of Clostridium Perfringens—Colony-Count Technique; ISO: Geneva, Switzerland, 2004. [Google Scholar]
- ISO. ISO 6888-2:1999/A1:2003: Microbiology of Food and Animal Feeding Stuffs—Horizontal Method for the Enumeration of Coagulase-Positive Staphylococci (Staphylococcus Aureus and Other Species)—Part 2: Technique Using Rabbit Plasma Fibrinogen Agar Medium—Amendment 1: Inclusion of Precision Data; ISO: Geneva, Switzerland, 1999. [Google Scholar]
- ISO. ISO 11290-2:1998—Microbiology of Food and Animal Feeding Stuffs—Horizontal Method for the Detection and Enumeration of Listeria Monocytogenes—Part 2—Enumeration Method; ISO: Geneva, Switzerland, 1998. [Google Scholar]
- ISO. ISO 21528-2:2004—Microbiology of Food and Animal Feeding Stuffs—Horizontal Method for the Detection and Enumeration of Enterobacteriaceae.—Colony-Count Technique; ISO: Geneva, Switzerland, 2004. [Google Scholar]
- Pikul, J.; Leszczyński, D.E.; Kummerow, F.A. Evaluation of three modified TBA methods for measuring lipid oxidation in chicken meat. J. Agric. Food Chem. 1989, 37, 1309–1313. [Google Scholar] [CrossRef]
- Hornsey, H.C. The color of cooked cured pork, I. Estimation of the nitric oxide-heme pigments. J. Sci. Food Agric. 1956, 7, 534–540. [Google Scholar] [CrossRef]
- Clark, E.M.; Mahoney, A.W.; Carpenter, C.E. Heme and total iron in ready-to-eat chicken. J. Agric. Food Chem. 1997, 45, 124–126. [Google Scholar] [CrossRef]
- Hunt, R.G.W. Measuring Colour; Ellis Horwood Limited: Chichester, UK, 1987. [Google Scholar]
- Suman, S.P.; Poulson, J. Myoglobin chemistry and meat color. Annu. Rev. Food Sci. Technol. 2013, 4, 79–99. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, B.; Diez, V. The effect of nitrite and starter culture on microbiological quality of “chorizo”—A Spanish dry cured sausage. Meat Sci. 2002, 60, 295–298. [Google Scholar] [CrossRef]
- Hospital, X.F.; Hierro, E.; Fernàndez, M. Survival of Listeria innocua in dry fermented sausages and changes in the typical microbiota and volatile profile as affected by the concentration of nitrate and nitrite. Int. J. Food Microbiol. 2012, 153, 395–401. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Foegeding, P.M. Principles of control. In Clostridium Botulinum: Ecology and Control in Foods; Hauschild, A.H.W., Dodds, K.L., Eds.; Marcel Dekker, Inc.: New York, NY, USA, 1993; Volume 54, pp. 121–176. [Google Scholar]
- Leistner, L. Hurdle effect and energy saving. In Food Quality and Nutrition; Downey, W.K., Ed.; Applied Science Publishers: London, UK, 1978; pp. 553–557. [Google Scholar]
- Archer, D.L. Evidence that ingested nitrate and nitrite are beneficial to health. J. Food Protect 2002, 65, 872–8751. [Google Scholar] [CrossRef]
- Roberts, T.A.; Gibson, A.M. Chemical methods for controlling Clostridium botulinum in processed meats. Food Technol. 1986, 40, 163–171. [Google Scholar]
- Pierson, M.D.; Smoot, L.A. Nitrite, nitrite alternatives, and the control of Clostridium botulinum in cured meats. Crit. Rev. Food Sci. Nutr. 1982, 17, 141–187. [Google Scholar] [CrossRef]
- Lövenklev, M.; Artin, I.; Hagberg, O.; Borch, E.; Holst, E.; Rådström, P. Quantitative interaction effects of carbon dioxide, sodium chloride, and sodium nitrite on neurotoxin gene expression in nonproteolytic Clostridium botulinum type B. Appl. Environ. Microbiol. 2004, 70, 2928–2934. [Google Scholar] [CrossRef]
- Cui, H.; Gabriel, A.A.; Nakano, H. Antimicrobial efficacies of plant extracts and sodium nitrite against Clostridium botulinum. Food Control 2010, 21, 1030–1036. [Google Scholar] [CrossRef]
- Christieansa, S.; Picgirarda, L.; Parafitaa, E.; Lebertb, A.; Gregori, T. Impact of reducing nitrate/nitrite levels on the behavior of Salmonella Typhimurium and Listeria monocytogenes in French dry fermented sausages. Meat Sci. 2018, 137, 160–167. [Google Scholar] [CrossRef]
- Ahn, D.U.; Maurer, A.J. Concentration of nitrate and nitrite in raw turkey breast meat and the microbial conversion of added nitrate to nitrite in tumbled turkey breast meat. Poult. Sci. 1987, 66, 1957–1960. [Google Scholar] [CrossRef]
- Heaton, K.M.; Cornforth, D.P.; Moiseev, I.V.; Egbert, W.R.; Carpenter, C.E. Minimum sodium nitrite levels for pinking of various cooked meats as related to use of direct or indirect-dried soy isolates in poultry rolls. Meat Sci. 2000, 55, 321–329. [Google Scholar] [CrossRef]
- Deda, M.S.; Bloukas, J.G.; Fista, G.A. Effect of tomato paste and nitrite level on processing and quality characteristics of frankfurters. Meat Sci. 2007, 76, 501–508. [Google Scholar] [CrossRef]
- Hayes, J.E.; Canonico, I.; Allen, P. Effects of organic tomato pulp powder and nitrite level on the physicochemical, textural and sensory properties of pork luncheon roll. Meat Sci. 2013, 95, 755–762. [Google Scholar] [CrossRef] [PubMed]
- Sindelar, J.J.; Cordray, J.C.; Sebranek, J.G.; Love, J.A.; Ahn, D.U. Effects of varying levels of vegetable juice powder and incubation time on color, residual nitrate and nitrite, pigment, pH, and trained sensory attributes of ready-to-eat uncured ham. J. Food Sci. 2007, 72, 388–395. [Google Scholar] [CrossRef]
- Vasavada, M.N.; Cornforth, D.P. Evaluation of milk mineral antioxidant activity in beef meatballs and nitrite-cured sausage. J. Food Sci. 2005, 70, 250–253. [Google Scholar] [CrossRef]
- Dethmers, A.E.; Rock, H. Effect of added sodium nitrite and sodium nitrate on sensory quality and nitrosoamine formation in Thuringer Sausage. J. Food Sci. 1975, 40, 491–497. [Google Scholar] [CrossRef]
- Doolaege, E.H.A.; Vossen, E.; Raes, K.; Meulenaer, B.D.; Verhe, R.; Paelinck, H. Effect of rosemary extract inclusion level on lipid oxidation, color stability and antioxidant concentrations, in reduced nitrite liver pates. Meat Sci. 2012, 90, 925–931. [Google Scholar] [CrossRef]
- Antonini, E.; Brunoni, M. Hemoglobin and Myoglobin in Their Reaction with Ligands; North-Holland Publishing Co.: Amsterdam, The Netherlands, 1971. [Google Scholar]
- Ahn, D.U.; Nam, K.C. Effect of ascorbic acid and antioxidants on color, lipid oxidation and volatiles of irradiated ground beef. Radiat. Phys. Chem. 2003, 71, 149–154. [Google Scholar]
- Sayd, T.; Morzel, M.; Chambon, C.; Franck, M.; Figwer, P.; Larzul, C.; Laville, E. Proteome analysis of the sarcoplasmic fraction of pig semimembranosus muscle: Implications on meat color development. J. Agric. Food Chem. 2006, 54, 2732–2737. [Google Scholar] [CrossRef]
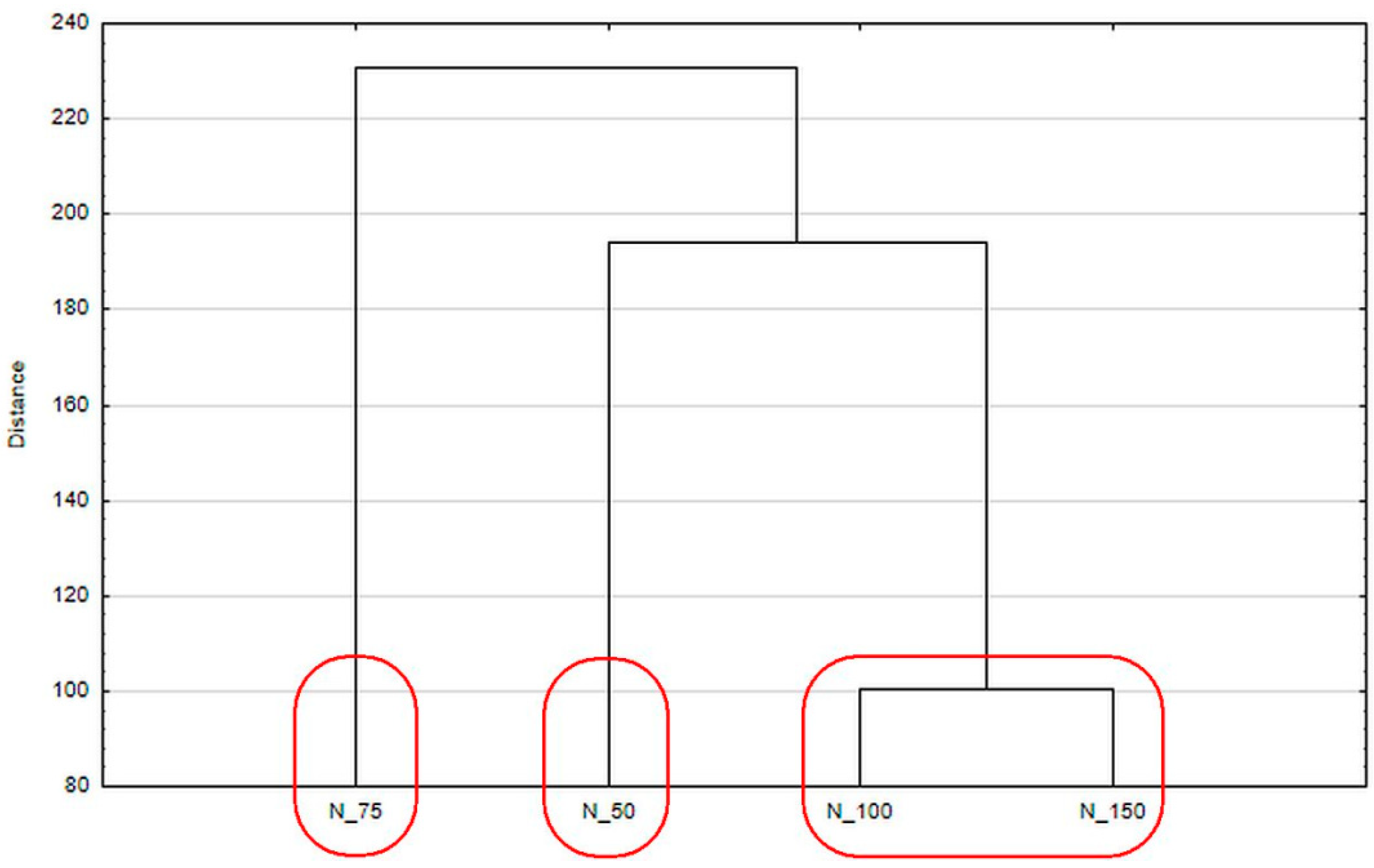
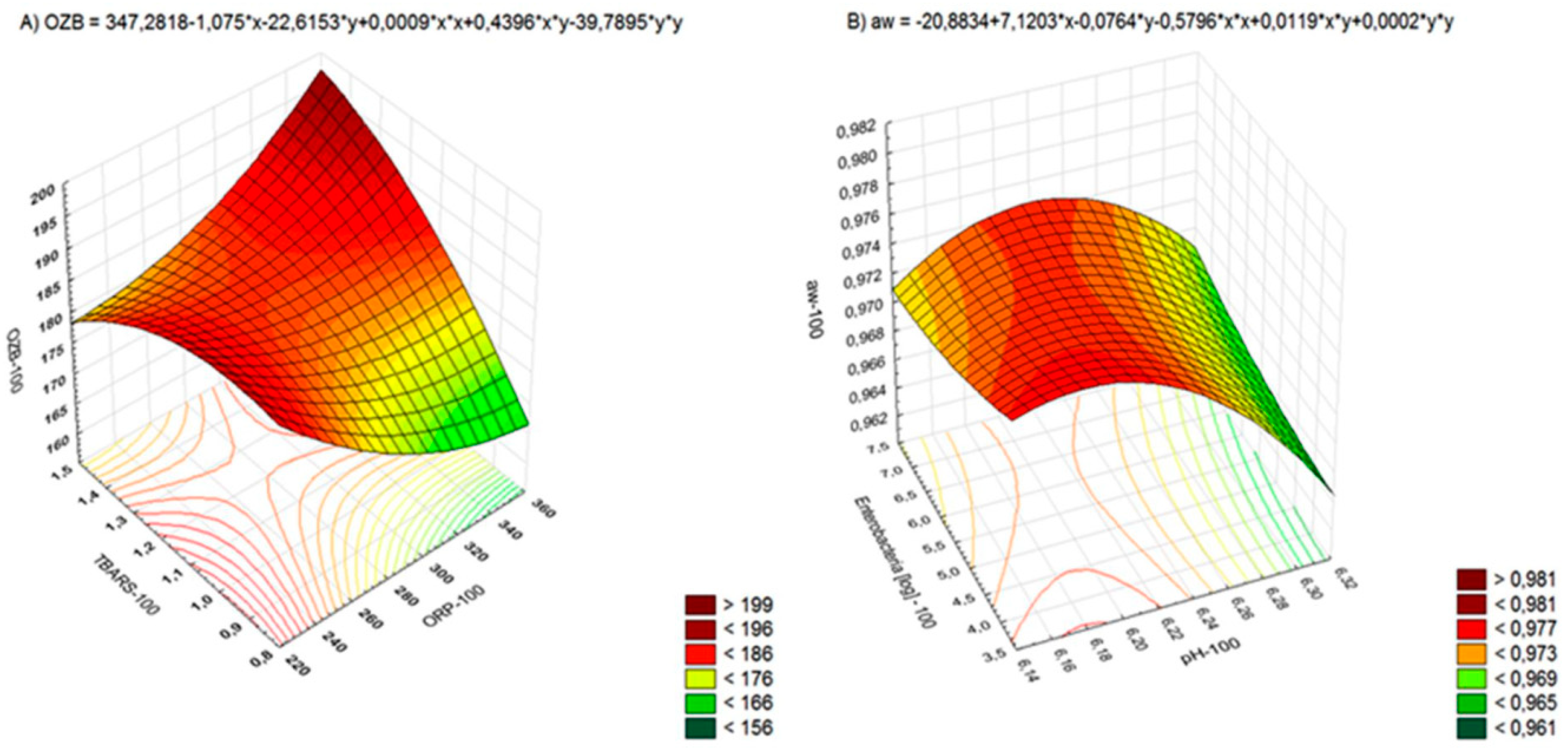
| Parameter | Sample | Day 0 | Day 7 | Day 14 | Day 21 |
|---|---|---|---|---|---|
| pH value | N_150 | 6.29 ± 0.01 bA | 6.25 ± 0.03 cA | 6.23 ± 0.01 dA | 6.31 ± 0.01 aA |
| N_100 | 6.27 ± 0.01 aA | 6.26 ± 0.01 bA | 6.17 ± 0.01 cB | 6.28 ± 0.01 abB | |
| N_75 | 6.17 ± 0.02 aC | 6.16 ± 0.02 aC | 6.07 ± 0.01 cD | 6.11 ± 0.04 bD | |
| N_50 | 6.21 ± 0.01 aB | 6.18 ± 0.01 bB | 6.08 ± 0.02 dC | 6.16 ± 0.01 cC | |
| Water activity | N_150 | 0.969 ± 0.00 bA | 0.97 ± 0.00 bB | 0.975 ± 0.00 aB | 0.975 ± 0.00 aB |
| N_100 | 0.970 ± 0.00 bA | 0.971 ± 0.00 bAB | 0.975 ± 0.00 aB | 0.975 ± 0.00 aB | |
| N_75 | 0.970 ± 0.00 bA | 0.970 ± 0.00 bB | 0.979 ± 0 aA | 0.979 ± 0 aA | |
| N_50 | 0.970 ± 0.00 bA | 0.972 ± 0.00 bB | 0.977 ± 0.00 aB | 0.977 ± 0.00 aB |
| Parameter | Sample | Day 0 | Day 7 | Day 14 | Day 21 |
|---|---|---|---|---|---|
| Lightness | N_150 | 56.22 ± 0.5 aB | 56.04 ± 0.61 aB | 56.00 ± 1.09 aA | 57.71 ± 1.30 aA |
| L* | N_100 | 55.61 ± 0.98 aB | 55.66 ± 0.51 aB | 55.70 ± 0.58 aA | 57.16 ± 0.56 aAB |
| N_75 | 56.82 ± 0.72 aAB | 57.56 ± 0.71 aA | 56.74 ± 1.05 aA | 57.78 ± 0.77 Aa | |
| N_50 | 55.21 ± 1.12 aBC | 55.97 ± 1.22 aB | 55.96 ± 1.16 aA | 55.76 ± 1.07 Ba | |
| Redness | N_150 | 15.65 ± 0.71 aA | 15.16 ± 0.32 aA | 14.61 ± 0.66 aA | 14.77 ± 0.69 aA |
| a* | N_100 | 14.80 ± 0.42 aA | 14.89 ± 0.50 aA | 14.61 ± 0.40 aA | 14.5 ± 0.49 aA |
| N_75 | 14.66 ± 0.33 aA | 14.11 ± 0.18 aA | 14.7 ± 0.36 aA | 14.86 ± 0.52 aA | |
| N_50 | 14.97 ± 0.69 aA | 15.04 ± 0.34 aA | 15.14 ± 0.44 aA | 14.79 ± 0.27 aA | |
| Yellowness | N_150 | 10.30 ± 0.58 aA | 9.69 ± 0.39 aA | 9.79 ± 0.38 aB | 9.88 ± 0.65 aA |
| b* | N_100 | 9.78 ± 0.71 aA | 9.30 ± 0.28 aAB | 9.38 ± 0.26B aC | 9.19 ± 0.37 aA |
| N_75 | 9.85 ± 0.44 aA | 9.05 ± 0.21 aB | 9.56 ± 0.28 aB | 9.83 ± 0.53 aA | |
| N_50 | 9.64 ± 0.59 aA | 9.62 ± 0.17 aA | 10.00 ± 0.29 aAB | 9.47 ± 0.30 aA | |
| OZB | N_150 | 177.91 ± 3.83 aB | 176.63 ± 3.08 abB | 171.45 ± 7.14 bB | 174.59 ± 3.18 abC |
| (mg/kg) | N_100 | 183.01 ± 6.13 bAB | 187.09 ± 2.63 abA | 177.4 ± 3.55B cbA | 183.18 ± 3.43 bB |
| N_75 | 174.68 ± 3.18 aBC | 175.7 ± 5.06 aB | 173.57 ± 3.63 aB | 175.61 ± 5.89 aC | |
| N_50 | 181.56 ± 8.7 bB | 180.2 ± 4.22 bB | 181.48 ± 3.36 bA | 196.945 ± 5.87 aA | |
| Fe | N_150 | 15.69 ± 0.34 aA | 15.58 ± 0.27 abB | 15.12 ± 0.63 bB | 15.4 ± 0.28 abC |
| (mg/kg) | N_100 | 16.14 ± 0.54 cA | 16.5 ± 0.23 bcA | 15.64 ± 0.31 acAB | 16.15 ± 0.3 cB |
| N_75 | 15.4 ± 0.28 aAB | 15.49 ± 0.45 aA | 15.31 ± 0.32 aB | 15.49 ± 0.52 aC | |
| N_50 | 16.01 ± 0.77 bA | 15.89 ± 0.37 bB | 16 ± 0.56 bA | 17.38 ± 0.52 aA |
| Parameter | Sample | Day 0 | Day 7 | Day 14 | Day 21 |
|---|---|---|---|---|---|
| TBARS | N_150 | 0.716 ± 0.05 bB | 0.711 ± 0.03 bD | 0.91 ± 0.13 aB | 0.945 ± 0.07 aB |
| (mg/kg) | N_100 | 0.999 ± 0.09 bB | 0.995 ± 0.04 bC | 0.98 ± 0.05 bAB | 1.158 ± 0.13 aA |
| N_75 | 0.955 ± 0.06 bB | 1.125 ± 0.07 aB | 1.164 ± 0.21 aAB | 1.12 ± 0.07 aA | |
| N_50 | 2.094 ± 0.71 aA | 1.823 ± 0.05 aA | 1.204 ± 0.34 bA | 1.121 ± 0.04 bA | |
| ORP | N_150 | 275.98 ± 6.32 bB | 263.09 ± 10.77 cA | 330.81 ± 1.19 aC | 251.24 ± 1.38 dD |
| (mV) | N_100 | 272.41 ± 6.51 bB | 239.69 ± 5.96 dC | 330.39 ± 3.44 aC | 262.74 ± 5.26 cC |
| N_75 | 284.7 ± 6.49 BbA | 243.36 ± 4.19 cCB | 342.73 ± 4.06 aA | 284.09 ± 9.02 bB | |
| N_50 | 293.98 ± 18.71 bA | 249.48 ± 3.14 cB | 336.81 ± 4.62 aB | 300.46 ± 10.73 bA |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wójciak, K.M.; Stasiak, D.M.; Kęska, P. The Influence of Different Levels of Sodium Nitrite on the Safety, Oxidative Stability, and Color of Minced Roasted Beef. Sustainability 2019, 11, 3795. https://doi.org/10.3390/su11143795
Wójciak KM, Stasiak DM, Kęska P. The Influence of Different Levels of Sodium Nitrite on the Safety, Oxidative Stability, and Color of Minced Roasted Beef. Sustainability. 2019; 11(14):3795. https://doi.org/10.3390/su11143795
Chicago/Turabian StyleWójciak, Karolina M., Dariusz M. Stasiak, and Paulina Kęska. 2019. "The Influence of Different Levels of Sodium Nitrite on the Safety, Oxidative Stability, and Color of Minced Roasted Beef" Sustainability 11, no. 14: 3795. https://doi.org/10.3390/su11143795
APA StyleWójciak, K. M., Stasiak, D. M., & Kęska, P. (2019). The Influence of Different Levels of Sodium Nitrite on the Safety, Oxidative Stability, and Color of Minced Roasted Beef. Sustainability, 11(14), 3795. https://doi.org/10.3390/su11143795







