Soil Reclamation of Abandoned Mine Lands by Revegetation in Northwestern Part of Transylvania: A 40-Year Retrospective Study
Abstract
1. Introduction
2. Materials and Methods
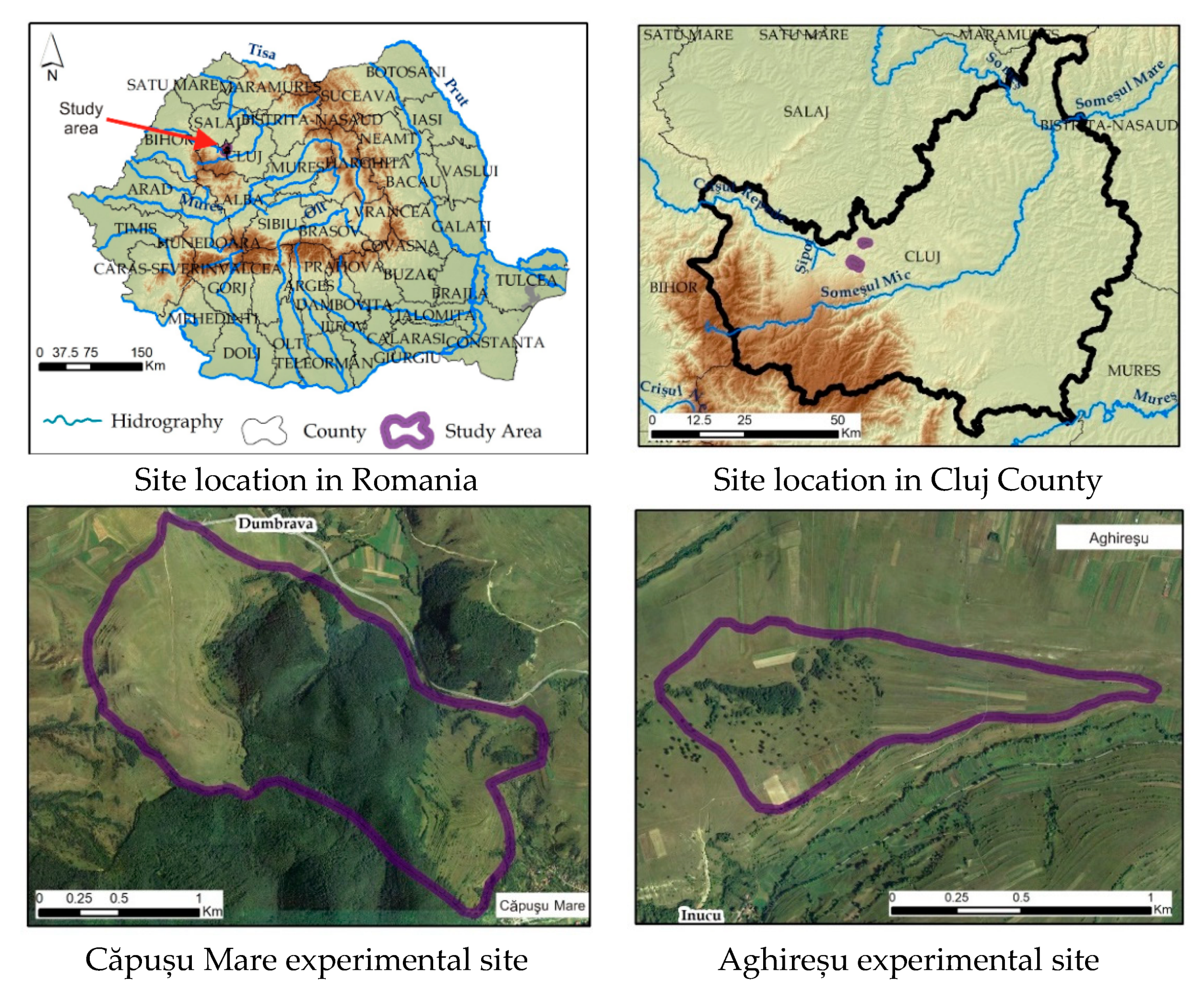
2.1. Site Location and Study Area
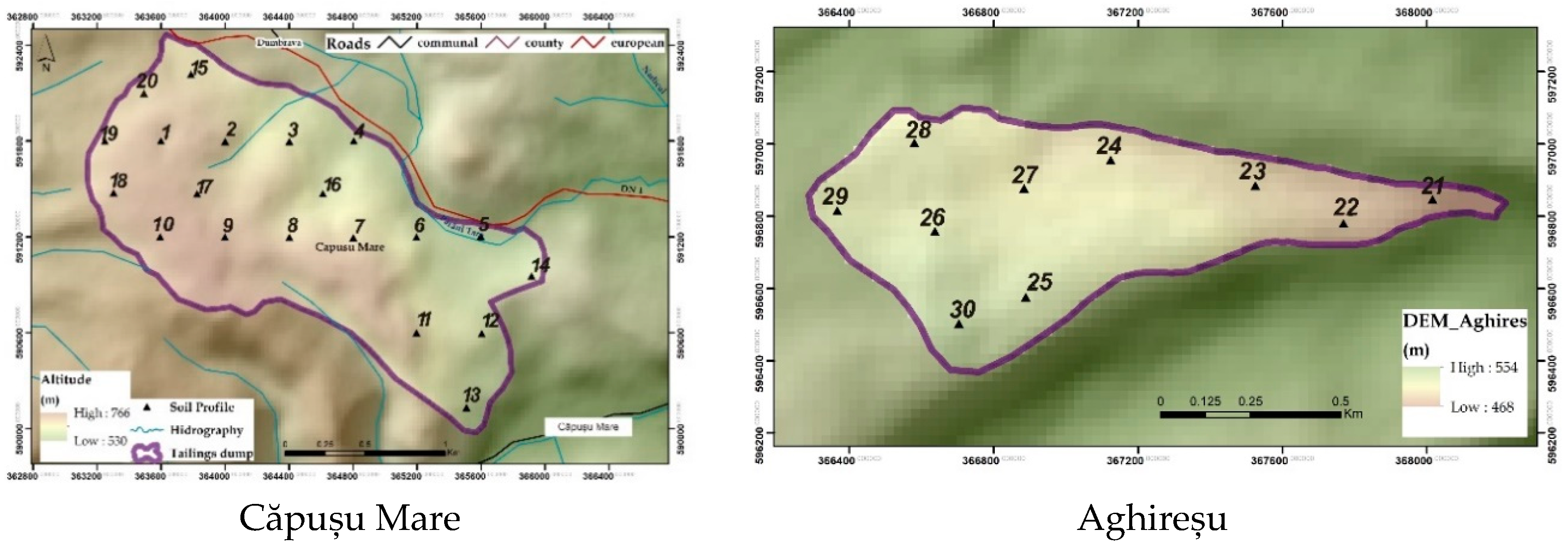
2.1.1. Căpușu Mare
2.1.2. Aghireșu
2.2. Soil Sampling
2.3. Soil Chemical, Physical, and Biological Analyses
2.4. Land Suitability Analysis
2.5. GIS Techniques
2.6. Statistical Analysis
3. Results and Discussion
3.1. Soil Chemical, Physical, and Biological Status
3.1.1. Land Suitability
3.1.2. Soil PH
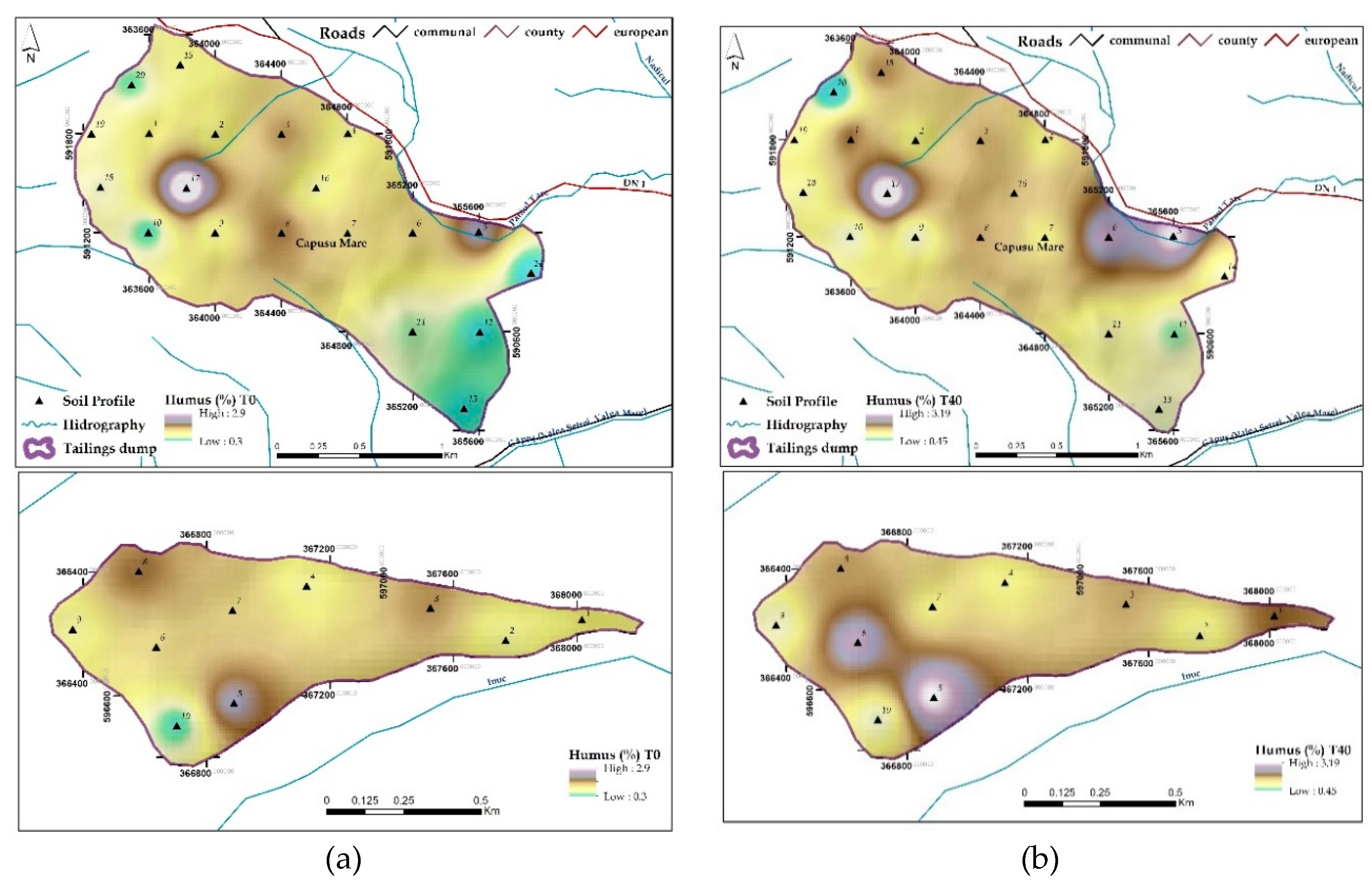
3.1.3. Organic Matter
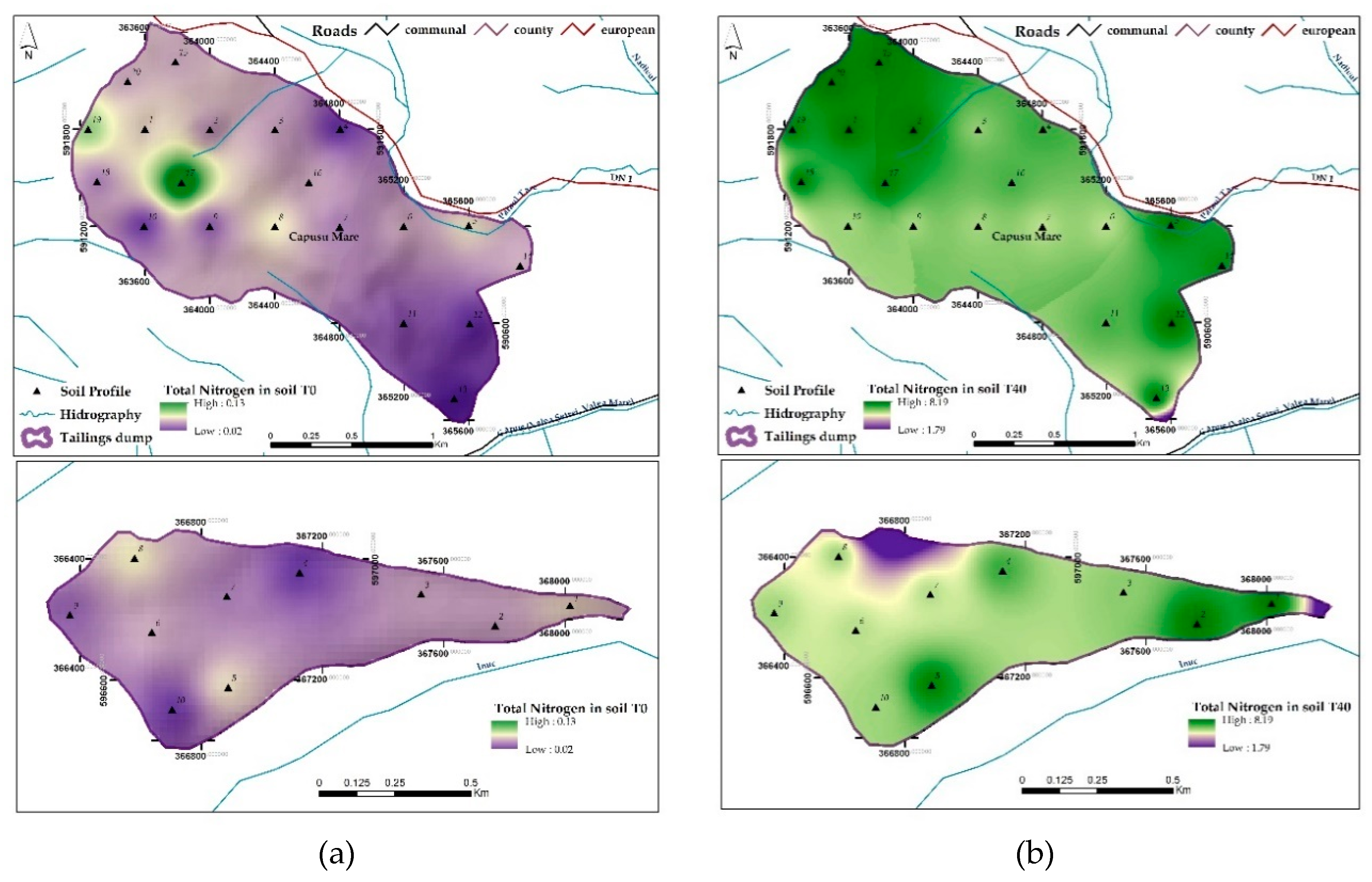
3.1.4. Total Nitrogen
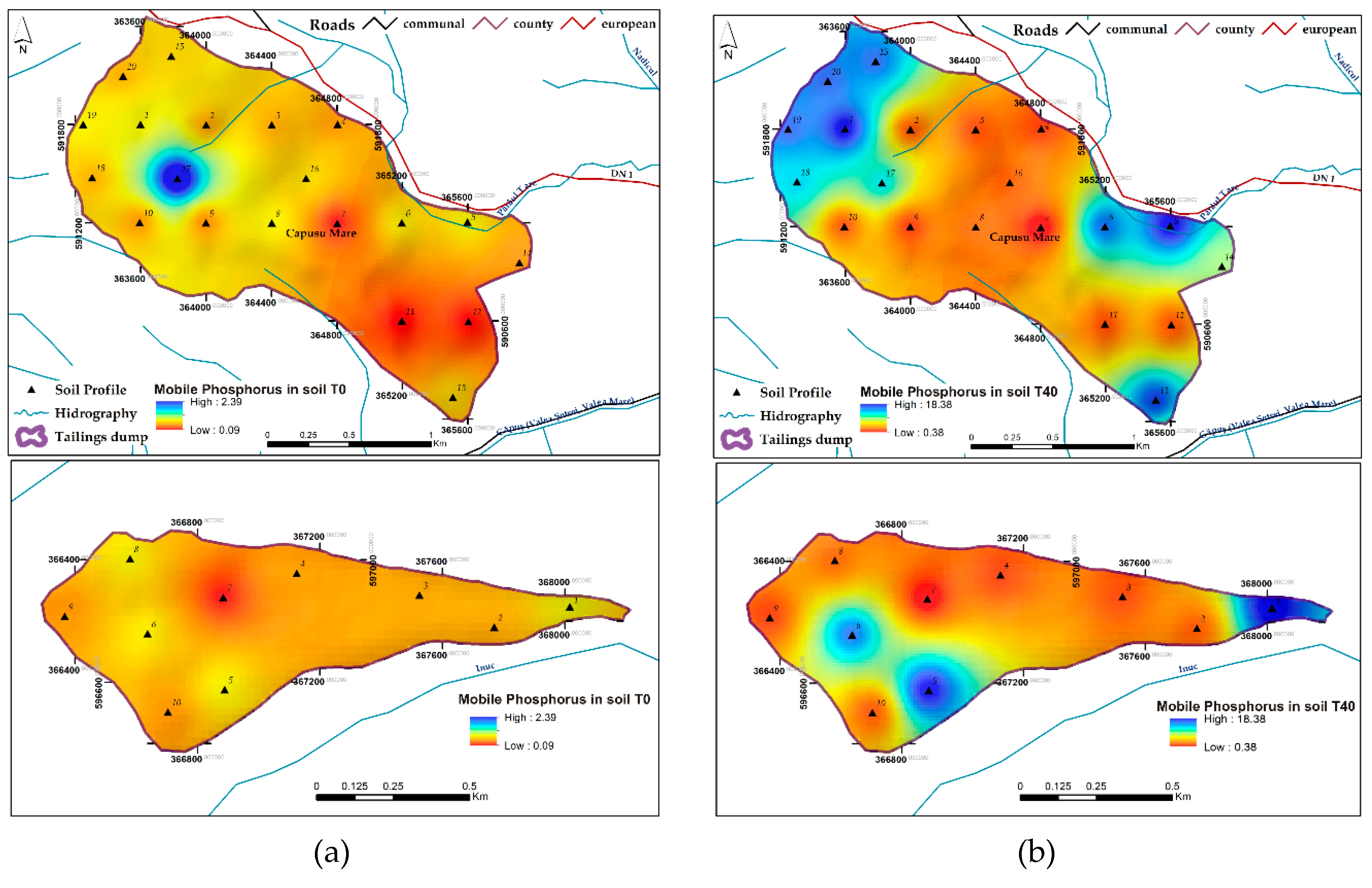
3.1.5. Available Phosphorus
3.1.6. Available Potassium
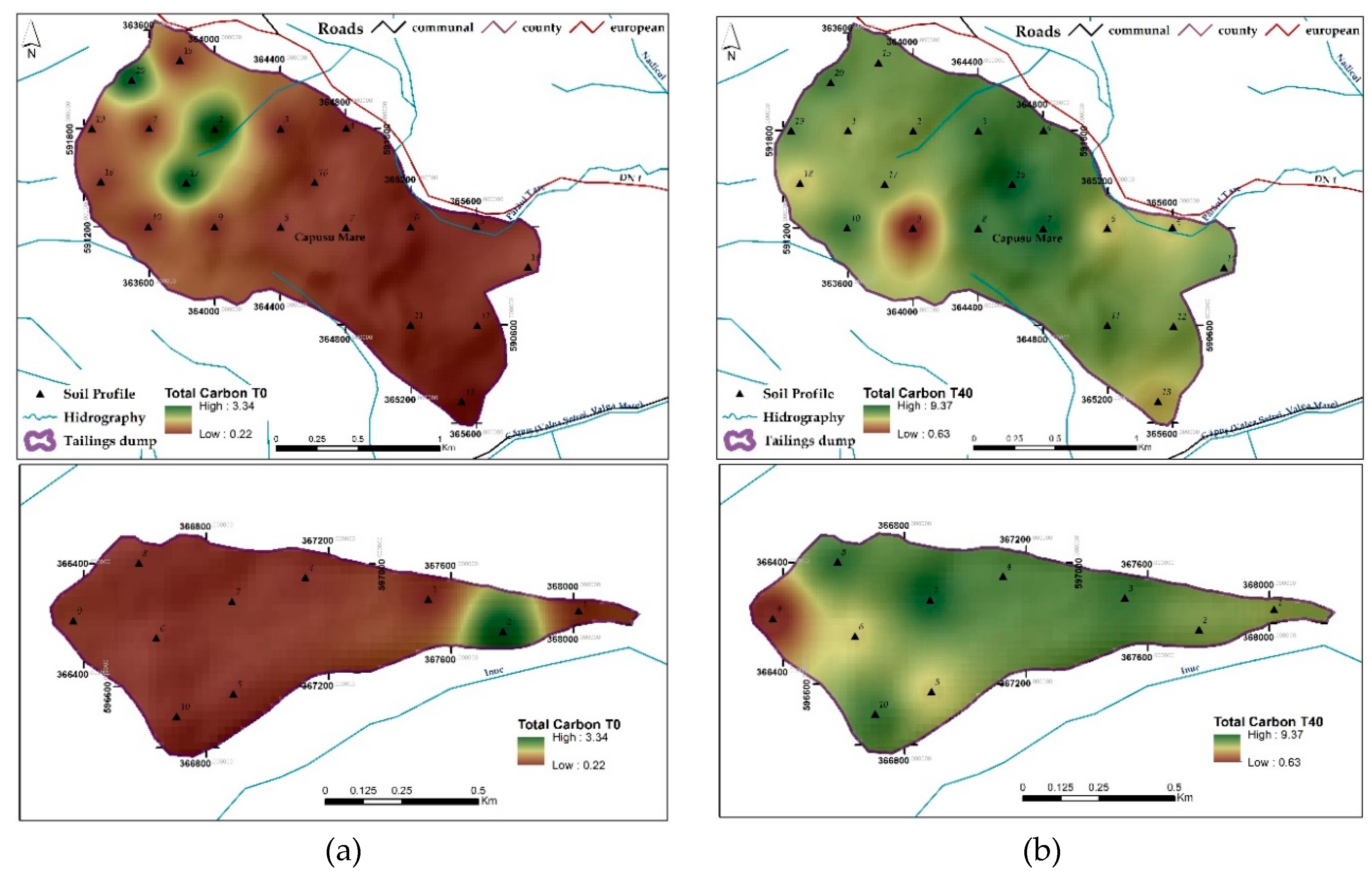
3.1.7. Total Organic Carbon
3.1.8. Soil Enzymatic Activity
- Catalase activity—referring to the descomposition of hydrogen peroxide with the production of molecular oxygen and water and is strongly correlated with dehydrogenases [48]. A general increase in soil catalase activity was observed during the reclamation process. The highest improvements in catalase activity after 40 years of soil reclamation was reached under Pinus sylvestris (1.47 mg H2O2/g soil), Picea abies (0.98 mg H2O2/g soil) tree plantations and under natural grassland cover (1.56 mg H2O2/g soil) followed by pasture sites (1.81 mg H2O2/g soil). In Robinia pseudacacia layers, the reclamation process seemed to be less effective, showing an improvement in catalase activity of only 0.29% at the end of the reclamation years examined.
- Dehydrogenase activity—reflects the total range of oxidative activity of soil microorganisms being considered a valuable indicator of oxidative metabolisms and microbiological activity [48]. Dehydrogenase activity exhibited an increasing tendency during the reclamation process underneath the herbaceous covers and Robinia pseudacacia plantations, but decreased under the coniferous ground covers as compared to initial values. It was also observed that dehydrogenase activity was higher in the topsoil layers than in lower depths. The results showed a low to moderate (0.41–1.03 μg formazan/g soil) dehydrogenase activity correlated with catalase activity in all sited under study. Similar results have also been reported by Brzezińska et al. (2001) [49] and Frîncu et al. (2015) [48].
- Saccharase activity—saccharase activity together with urease are strongly related to carbon and nitrogen cycles, carbohydrate supplies in soil and organic matter which make saccharase to be considered a reliable indicator of soil fertility or contamination [45,50]. The results show that soil saccharase activity was the highest under Robinia pseudacacia stands with a linear increase of the plantation age. Similar findings have been reported by Wang et al. (2012) [51] who investigated the effect of black locust on soil chemical and microbiological properties in an eroded hilly area from China. Their results are in agreement with our findings confirming the fact that Robinia pseudacacia greatly improves soil properties when grown in degraded soils. Likewise, natural grassland and pasture vegetation also improved saccharase activity of the soil. Saccharase activity decreased slightly under the coniferous covers being related to the lower soil pH and soil fertility.
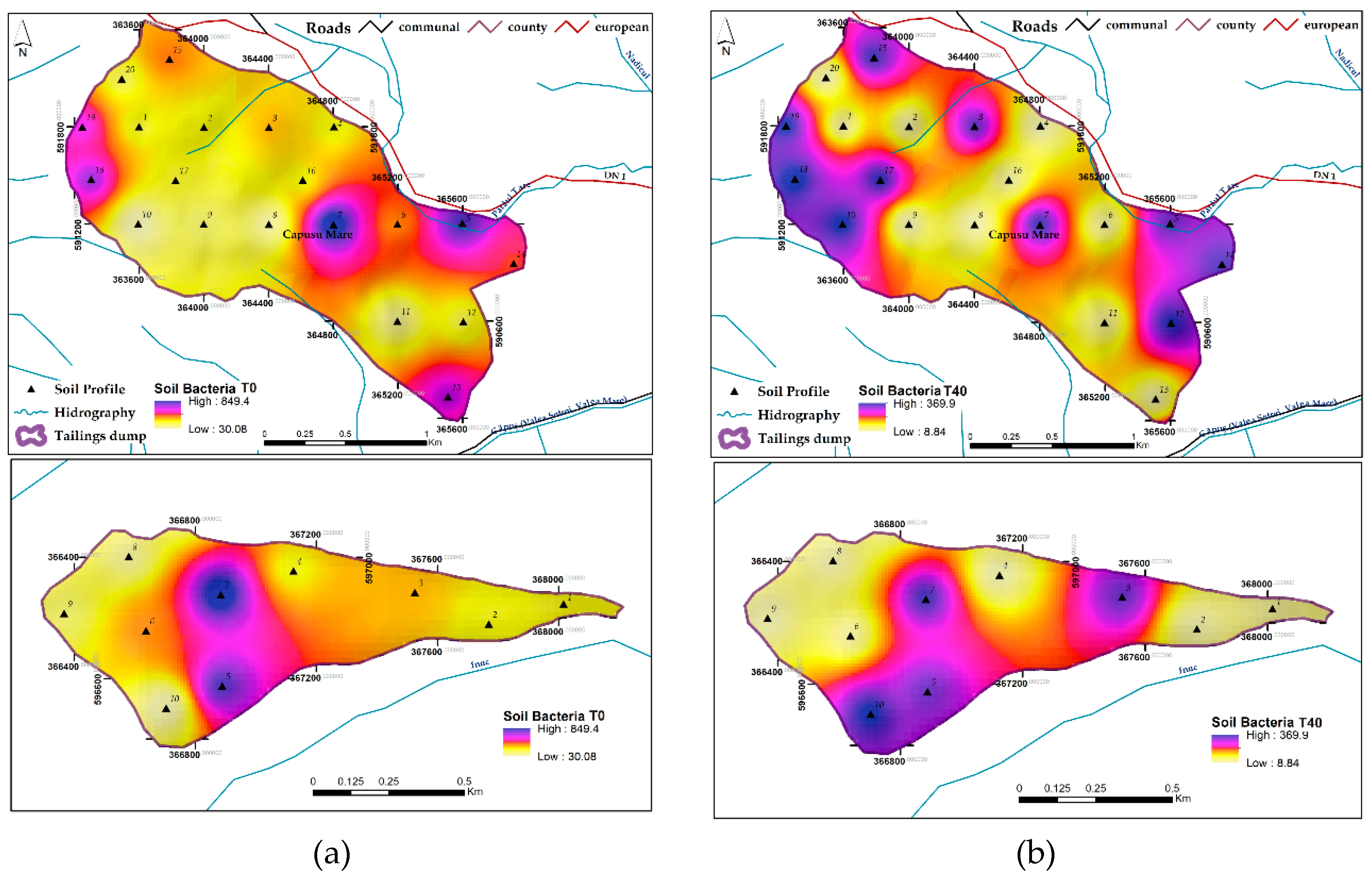
3.1.9. Soil Bacteria
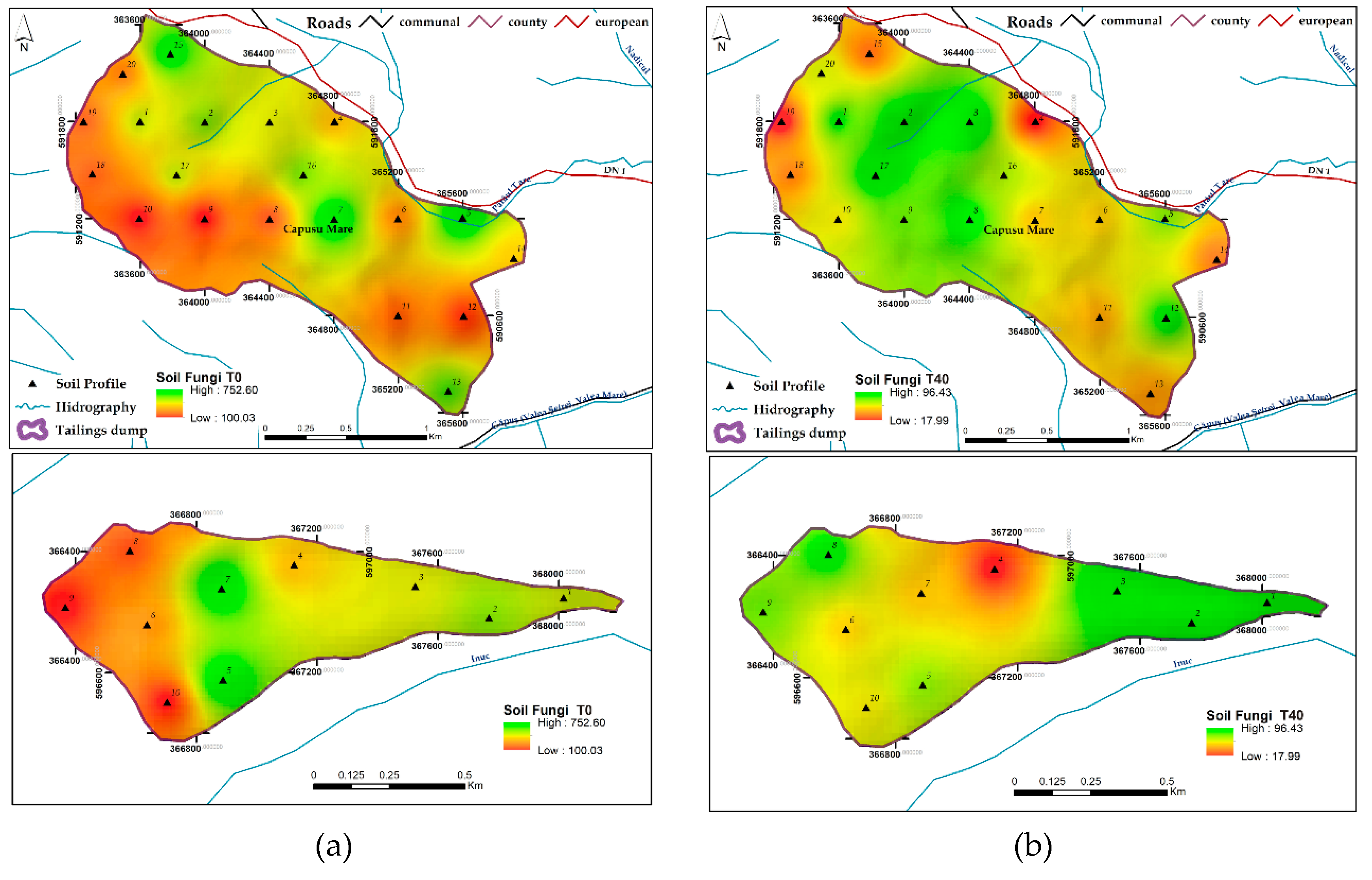
3.1.10. Soil Fungi
3.1.11. Soil Respiration
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Worldbank and International Finance Corporation. Available online: http://siteresources.worldbank.org/INTOGMC/Resources/treasureortrouble.pdf (accessed on 15 March 2019).
- Schandl, H.; Poldy, F.; Turner, G.M.; Measham, T.G.; Walker, D.H.; Eisenmenger, N. Australia’s resource use trajectories. J. Ind. Ecol. 2008, 12, 669–685. [Google Scholar] [CrossRef]
- Keeling, A.; Sandlos, J. Environmental justice goes underground? Historical notes from Canada’s northern mining frontier. Environ. Justice 2009, 2, 117–125. [Google Scholar] [CrossRef]
- Sahu, H.B.; Dash, S. Land degradation due to mining in India and its mitigation measures. In Proceedings of the Second International Conference on Environmental Science and Technology, Singapore, 26–28 February 2011. [Google Scholar]
- Majer, M. The practice of mining companies in building relationships with local communities in the context of CSR formula. J. Sustain. Min. 2013, 12, 38–47. [Google Scholar] [CrossRef]
- Hilson, G. Small-scale mining and its socio-economic impact in developing countries. Nat. Resour. Forum 2002, 26, 3–13. [Google Scholar] [CrossRef]
- Martin, J.H. Surface mining and the environment in Europe. Int. J. Surf. Min. Reclam. Environ. 1993, 7, 91–104. [Google Scholar]
- Sheoran, V.; Sheoran, A.S.; Poonia, P. Soil reclamation of abandoned mine land by revegetation: A review. J. Soil Sediment. 2010, 3, 13. [Google Scholar]
- Šourková, M.; Frouz, J.; Šantrùčková, H. Accumulation of carbon, nitrogen and phosphorus during soil formation on alder spoil heaps after brown-coal mining, near Sokolov (Czech Republic). Geoderma 2005, 124, 203–214. [Google Scholar] [CrossRef]
- Blaga, G.; Dumitru, M.; Bunescu, V.; Lechinţan, T.; Păcurar, I. Bonitarea terenurilor degradate de exploatarea minieră de suprafaţă Mirşid (jud. Sălaj), şi a haldelor nivelate (protosoluri antropice). Agronomie 1992, 35, 54–58. [Google Scholar]
- Florea, N.; Munteanu, I. Solonchaks. (Soolonceacuri). In Romanian Soil Taxonomy System (SRTS); Sitech: Craiova, Romania, 2012; pp. 132–140. [Google Scholar]
- IUSS Working Group WRB. (2015). World Reference Base for Soil Resources 2014. Update2015, International Soil Classification System for Naming Soils and Creating Legends for Soilmaps; World Soil Resources Reports No. 106; FAO: Rome, Italy, 2015; Available online: http://www.fao.org/3/i3794en/I3794en.pdf (accessed on 19 April 2019).
- Simon, A.; Gáll, E.; Tonk, S.; Tamás, L.; Maxim, A.; Janicsik, P.; Coroiu, T. Atlas; Suncart: Cluj-Napoca, Romania, 2003; pp. 80–95. [Google Scholar]
- Schollenberger, C.J. A rapid approximate method for determining soil organic matter. Soil Sci. 1927, 24, 65–68. [Google Scholar] [CrossRef]
- Schuman, G.E.; Stanley, M.A.; Knudsen, D. Automated total nitrogen analysis of soil and plant samples 1. Soil Sci. Soc. Am. J. 1973, 37, 480–481. [Google Scholar] [CrossRef]
- Oreshkin, N.G. Extraction of available phosphorus by the Egner-Riehm-Domingo method. Agrokhimiya 1980, 8, 135–138. [Google Scholar]
- Walkley, A.; Black, I.A. An examination of the Degtjareff method for determining soil organic matter, and a proposed modification of the chromic acid titration method. Soil Sci. 1934, 37, 29–38. [Google Scholar] [CrossRef]
- Carter, M.R.; Gregorich, E.G. Soil Sampling and Methods of Analysis; Lewis: London, UK, 1993. [Google Scholar]
- Casida, L.E.; Klein, D.A.; Santoro, T. Soil dehydrogenase activity. Soil Sci. 1964, 98, 371–376. [Google Scholar] [CrossRef]
- Zibilske, L.M. Part 2-Microbiological and biochemical properties. In Methods of Soil Analysis, 2nd ed.; Weaver, R.W., Angle, J.S., Bottomley, P.S., Eds.; SSSA Book Series 5.2; ACSESS Digital Library: Madison, WI, USA, 1994; pp. 835–863. [Google Scholar]
- Florea, N.; Bălăceanu, V.; Rauţă, C.; Canarache, A. Methodology of Pedological Studies, 3rd ed.; Pedological and Agrochemical Institute Bucharest, Redacţia de propagandă tehnico-agricolă: Bucharest, Romania, 1978. [Google Scholar]
- Martinez-Toledo, M.V.; Salmeron, V.; Gonzalez-Lopez, J. Effect of an organophosphorus insecticide, profenofos, on agricultural soil microflora. Chemosphere 1992, 24, 71–80. [Google Scholar] [CrossRef]
- Roșca, S. Application of Soil Loss Scenarios Using the ROMSEM Model Depending on Maximum Land Use Pretability Classes. A Case Study. Studia UBB Geographia 2014, 59, 101–116. [Google Scholar]
- Bilașco, S.; Roșca, S.; Fodorean, I.; Vescan, I.; Filip, S.; Petrea, D. Quantitative evaluation of the risk induced by dominant geomorphological processes on different land uses, based on GIS spatial analysis models. Front. Earth. Sci. 2017, 12, 1–14. [Google Scholar] [CrossRef]
- Boț, A.I.; Păcurar, I.; Szajdak, L.; Roșca, S.; Păcurar, H.M.; Pleșa, V.C. Determining the Amount of Total Nitrogen, Total Organic Carbon and C:N Ratio for Eight Blueberry Plantations from Northwest Region of Development. Proenvironment/Promediu 2017, 10, 86–94. [Google Scholar]
- Mîndru, M.; Păcurar, I.; Roșca, S.; Bilașco, S.; Păcurar, H.M. Research Regarding the Soil Characteristics and their Influence on Forest Productivity in the Mociar Forest, Mureş County. Bull. Uasvm Hortic. 2017, 74, 155–163. [Google Scholar] [CrossRef]
- Sandor, M. Soil respiration: Concept and measurement methods. ProEnvironment 2010, 3, 54–57. [Google Scholar]
- Rhoades, C.; Binkley, D. Factors influencing decline in soil pH in Hawaiian Eucalyptus and Albizia plantations. For. Ecol. Manag. 1996, 80, 47–56. [Google Scholar] [CrossRef]
- Hendershot, W.H.; Lalande, H. Soil Reaction and Exchangeable Acidity. In Soil Sampling and Methods of Analysis, 2nd ed.; Carter, M.R., Gregorich, E.G., Eds.; CRC Press: Boca Raton, FL, USA, 2007; pp. 173–179. [Google Scholar]
- Cheng, Y.; Wang, J.; Mary, B.; Zhang, J.B.; Cai, Z.C.; Chang, S.X. Soil pH has contrasting effects on gross and net nitrogen mineralizations in adjacent forest and grassland soils in central Alberta, Canada. Soil. Biol. Biochem. 2013, 57, 848–857. [Google Scholar] [CrossRef]
- Gitt, M.J.; Dollhopf, D.J. Coal waste reclamation using automated weathering to predict lime requirement. J. Environ. Qual. 1991, 20, 285–288. [Google Scholar] [CrossRef]
- Gould, A.B.; Hendrix, J.W.; Ferriss, R.S. Relationship of mycorrhizal activity to time following reclamation of surface mine land in western Kentucky. I. Propagule and spore population densities. Can. J. Bot. 1996, 74, 247–261. [Google Scholar] [CrossRef]
- Reeves, D.W. The role of soil organic matter in maintaining soil quality in continuous cropping systems. Soil Till. Res. 1997, 43, 131–167. [Google Scholar] [CrossRef]
- Hu, Z.; Duo, L.; Shao, F. Optimal Thickness of Soil Cover for Reclaiming Subsided Land with Yellow River Sediments. Sustainability 2018, 10, 3853. [Google Scholar] [CrossRef]
- Drinkwater, L.E.; Wagoner, P.; Sarrantonio, M. Legume-based cropping systems have reduced carbon and nitrogen losses. Nature 1998, 396, 262–265. [Google Scholar] [CrossRef]
- McVay, K.A.; Radcliffe, D.E.; Hargrove, W.L. Winter legume effects on soil properties and nitrogen fertilizer requirements. Soil Sci. Soc. Am. J. 1989, 53, 1856–1862. [Google Scholar] [CrossRef]
- Unger, P.W.; Kaspar, T.C. Soil compaction and root growth. A review. Agron. J. 1994, 86, 759–766. [Google Scholar] [CrossRef]
- Williams, S.M.; Weil, R.R. Crop cover root channels may alleviate soil compaction effects on soybean crop. Soil Sci Soc. Am. J. 2004, 68, 1403–1409. [Google Scholar] [CrossRef]
- Elkins, N.Z.; Lawrence, W.P.; Aldon, E.; Whitford, W.G. Responses of Soil Biota to Organic Amendments in Stripmine Spoils in Northwestern New Mexico. J. Environ. Qual. 1984, 13, 215–219. [Google Scholar] [CrossRef]
- Burke, I.C.; Lauenroth, W.K.; Coffin, D.P. Soil organic matter recovery in semiarid grasslands: Implications for the conservation reserve program. Ecol. Appl. 1995, 5, 793–801. [Google Scholar] [CrossRef]
- Gyaneshwar, P.; Kumar, G.N.; Parekh, L.J.; Poole, P.S. Role of soil microorganisms in improving P nutrition of plants. Plant. Soil. 2002, 245, 83–93. [Google Scholar]
- Coppin, N.J.; Bradshaw, A.D. The Establishment of Vegetation in Quarries and Open-Pit Non-Metal Mines; Mining Journal Books: London, England, 1982; p. 112. [Google Scholar]
- Heiberg, S.O.; White, D.P. Potassium deficiency of reforested Pine and spruce stands in northern New York. In Proceedings of the 7th International Botanical Congress, Stockholm, Sweden, 128 July 1950. [Google Scholar]
- Brennan, R.F.; Bolland, M.D.A.; Bowden, J.W. Potassium deficiency, and molybdenum deficiency and aluminium toxicity due to soil acidification, have become problems for cropping sandy soils in south-western Australia. Aust. J. Exp. Agric. 2004, 44, 1031–1039. [Google Scholar] [CrossRef]
- Zhang, L.; Chen, W.; Burger, M.; Yang, L.; Gong, P.; Wu, Z. Changes in soil carbon and enzyme activity as a result of different long-term fertilization regimes in a greenhouse field. PLoS ONE 2015, 10, e0118371. [Google Scholar] [CrossRef] [PubMed]
- Akala, V.A.; Lal, R. Potential of mine land reclamation for soil organic carbon sequestration in Ohio. Land Degrad. Dev. 2000, 11, 289–297. [Google Scholar] [CrossRef]
- Indorante, S.J.; Jansen, I.J.; Boast, C.W. Surface mining and reclamation: Initial changes in soil character. J. Soil Water Conserv. 1981, 36, 347–350. [Google Scholar]
- Frîncu, M.; Dumitrache, C.; Dumitru, A.C.; Cimpeanu, P.C.; Mihai, L.P. Soil Fertility Assessment through Enzyme Activity. J. Young Sci. 2015, 3, 1283–1291. [Google Scholar]
- Brzezińska, M.; Stępniewska, Z.; Stępniewski, W. Dehydrogenase and catalase activity of soil irrigated with municipal wastewater. Pol. J. Environ. Stud. 2001, 10, 307–311. [Google Scholar]
- Levytska, O.; Davydova, I. Analysis of the Soil Enzymatic Activity for Lands Reclaimed From Brown-Coal Strip Mines in Stryzhivka. Logistyka 2013, 4, 302–309. [Google Scholar]
- Wang, B.; Liu, G.; Xue, S. Effect of black locust (Robinia pseudoacacia) on soil chemical and microbiological properties in the eroded hilly area of China’s Loess Plateau. Environ. Earth Sci. 2012, 65, 597–607. [Google Scholar] [CrossRef]
- Johnson, D.B.; Williamson, J.C.; Bailey, A.J. Microbiology of soils at opencast coal sites. I. Short-and long-term transformations in stockpiled soils. J. Soil Sci. 1991, 42, 1–8. [Google Scholar] [CrossRef]
- Hardoim, P.R.; Van Overbeek, L.S.; Berg, G.; Pirttila, A.M.; Compant, S.; Campisano, A.; Doring, M.; Sessitsch, A. The hidden world within plants: Ecological and evolutionary considerations for defining functioning of microbial endophytes. Microbiol. Mol. Biol Rev. 2015, 79, 293–320. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, A. Soil fungi. In Soil Biology Basics, Information Series; NSW Department of Primary Industries: Forests, NSW, Australia, 2005. [Google Scholar]
- Rousk, J.; Brookes, P.C.; Baath, E. Contrasting soil pH effects on fungal and bacterial growth suggest functional redundancy in carbon mineralization. Appl. Environ. Microbiol. 2009, 75, 1589–1596. [Google Scholar] [CrossRef] [PubMed]
- Ritz, K.; Young, I.M. Interactions between soil structure and fungi. Mycologis 2004, 18, 52–59. [Google Scholar] [CrossRef]
- Rives, C.S.; Bajwa, M.I.; Liberta, A.E.; Miller, R.M. Effects of topsoil storage during surface mining on the viability of VA mycorrhiza. Soil Sci. 1980, 129, 253–257. [Google Scholar] [CrossRef]
- O’Halloran, I.P.; Miller, M.H.; Arnold, G. Absorption of P by corn (Zea mays L.) as influenced by soil disturbance. Can. J. Soil Sci 1986, 66, 287–302. [Google Scholar]
- Jasper, D.A.; Abbott, L.K.; Robson, A.D. Soil disturbance reduces the infectivity of external hyphae of VA mycorrhizal fungi. New Phytol. 1989, 112, 93–99. [Google Scholar] [CrossRef]
- Jasper, D.A.; Abbott, L.K.; Robson, A.D. Hyphae of a vesicular arbuscular mycorrhizal fungus maintain infectivity in dry soil, except when the soil is disturbed. New Phytol. 1989, 112, 101–107. [Google Scholar] [CrossRef]
- Miller, R.M.; Jastrow, J.D.; Reinhardt, D.R. External hyphal production of vesicular-arbuscular mycorrhizal fungi in pasture and tallgrass prairie communities. Oecologia 1995, 103, 17–23. [Google Scholar] [CrossRef]
- Lladó, S.; López-Mondéjar, R.; Baldrian, P. Forest soil bacteria: Diversity, involvement in ecosystem processes, and response to global change. Microbiol. Mol. Biol. Rev. 2017, 81, e00063-16. [Google Scholar] [CrossRef]
- Larionova, A.A.; Yermolayev, A.M.; Blagodatsky, S.A.; Rozanova, L.N.; Yevdokimov, I.V.; Orlinsky, D.B. Soil respiration and carbon balance of gray forest soils as affected by land use. Biol. Fert. Soil. 1998, 27, 251–257. [Google Scholar] [CrossRef]
- Yiqi, L.; Zhou, X. Soil Respiration and the Environment; Elsevier: Burlington, MA, USA, 2006. [Google Scholar]
- Cho, S.J.; Kim, M.H.; Lee, Y.O. Effect of pH on soil bacterial diversity. J. Ecol. Nat. Environ. 2016, 40, 10. [Google Scholar] [CrossRef]
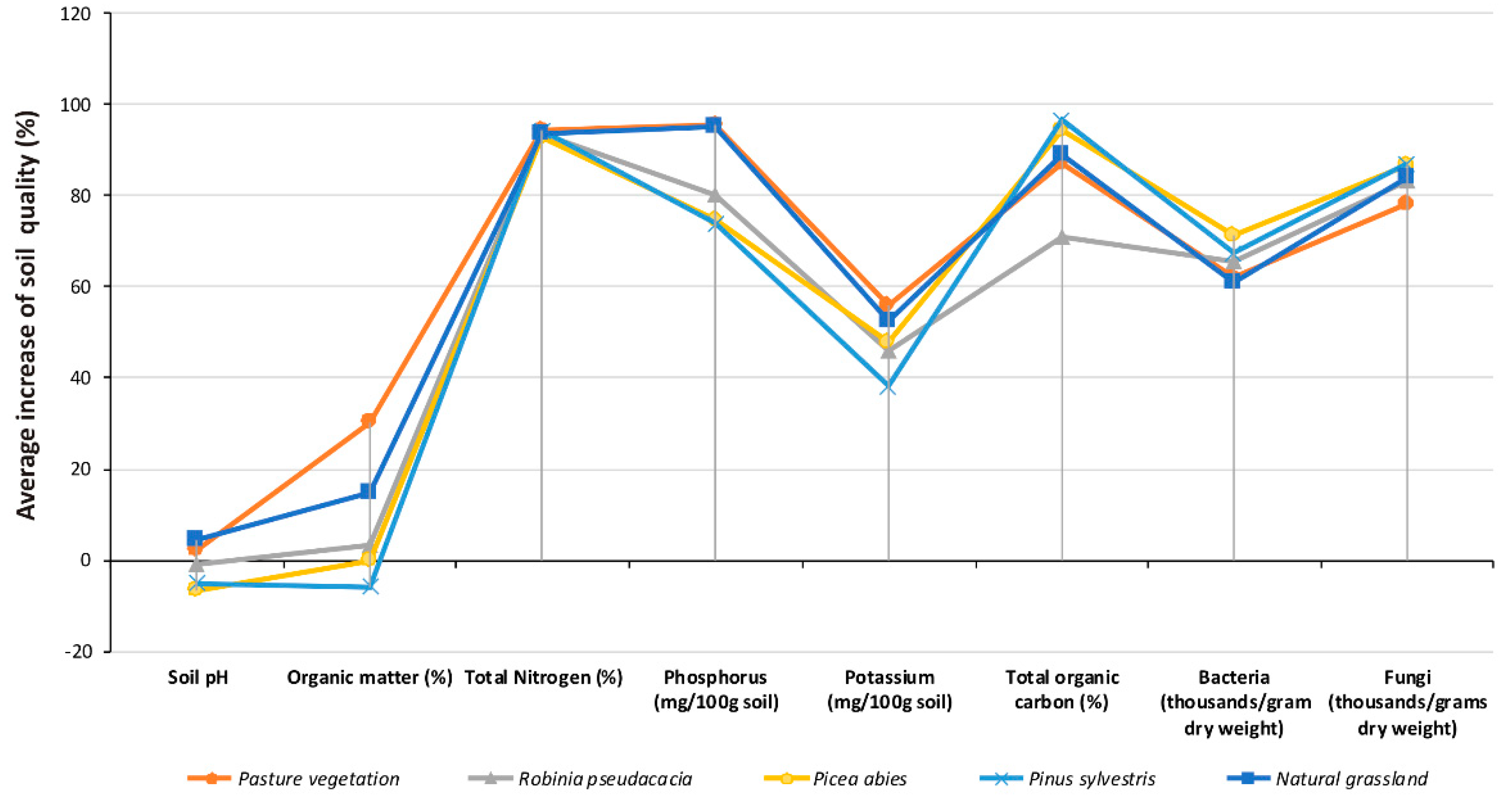
| Soil pH | Organic Matter (%) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Plant Cover | Natural grassland | Pasture vegetation | Robinia pseudacacia | Picea abies | Pinus sylvestris | Natural grassland | Pasture vegetation | Robinia pseudacacia | Picea abies | Pinus sylvestris |
| Time | ||||||||||
| T 0 | 6.65 ± 1.46 a | 6.10 ± 1.95 a | 7.85 ± 0.30 a | 7.72 ± 0.24 c | 7.03 ± 1.63 b | 0.69 ± 0.50 a | 0.50 ± 0.29 a | 0.89 ± 0.62 a | 0.86 ± 0.39 a | 1.06 ± 0.58 a |
| T 10 | 6.75 ± 1.35 a | 6.14 ± 1.93 a | 7.84 ± 0.35 a | 7.56 ± 0.11 b | 6.92 ± 1.57 ab | 0.72 ± 0.48 b | 0.56 ± 0.26 b | 0.90 ± 0.60 a | 0.86 ±0.39 a | 1.05 ± 0.57 a |
| T 20 | 6.84 ± 1.27 ab | 6.17 ± 1.91 a | 7.81 ± 0.38 a | 7.43 ± 0.01 ab | 6.84 ± 1.53 ab | 0.75 ± 0.47 c | 0.64 ± 0.21 c | 0.91 ± 0.58 ab | 0.86 ± 0.40 a | 1.03 ± 0.55 a |
| T 30 | 6.92 ± 1.18 b | 6.21 ± 1.90 a | 7.80 ± 0.42 a | 7.31 ± 0.09 a | 6.76 ± 1.49 a | 0.78 ± 0.47 d | 0.67 ± 0.19 d | 0.92 ± 0.56 b | 0.86 ± 0.40 a | 1.02 ± 0.54 a |
| T 40 | 6.97 ± 1.13 b | 6.23 ± 1.89 a | 7.78 ± 0.44 a | 7.23 ± 0.15 a | 6.69 ± 1.45 a | 0.81 ± 0.46 e | 0.72 ± 0.18 e | 0.92±0.54b | 0.86 ± 0.40 a | 1.00 ± 0.52 a |
| Total Nitrogen (%) | Phosphorus (mg/100 g soil) | |||||||||
| T 0 | 0.04 ± 0.02 a | 0.04 ± 0.03 a | 0.06 ± 0.02 a | 0.04 ± 0.01 a | 0.04 ± 0.02 a | 0.91 ± 0.42 a | 1.29 ± 0.61 a | 0.70 ± 0.28 a | 0.47 ± 0.10 a | 0.60 ± 0.25 a |
| T 10 | 0.18 ± 0.09 b | 0.21 ± 0.13 b | 0.27 ± 0.09 a | 0.17 ± 0.04 a | 0.20 ± 0.10 b | 5.35 ± 2.39 b | 8.04 ± 2.16 b | 1.41 ± 0.56 a | 0.81 ± 0.17 a | 1.03 ± 0.43 a |
| T 20 | 0.32 ± 0.16 c | 0.38 ± 0.23 c | 0.48 ± 0.17 b | 0.30 ± 0.07 ab | 0.36 ± 0.19 c | 9.78 ± 4.38 c | 14.80 ± 3.74 c | 2.11 ± 0.84 b | 1.16 ± 0.25 ab | 1.45 ± 0.60 b |
| T 30 | 0.46 ± 0.23 d | 0.54 ± 0.33 d | 0.69 ± 0.24 b | 0.43 ± 0.10 b | 0.52 ± 0.27 d | 14.22 ± 6.37 d | 21.55 ± 5.32 d | 2.82 ± 1.12 b | 1.50 ± 0.32 b | 1.87 ± 0.78 b |
| T 40 | 0.61 ± 0.29 e | 0.71 ± 0.43 e | 0.90 ± 0.32 c | 0.56 ± 0.13 c | 0.68 ± 0.35 e | 18.66 ± 8.36 e | 28.30 ± 6.90 e | 3.53 ± 1.40 c | 1.85 ± 0.39 b | 2.30 ± 0.95 c |
| Potassium (mg/100 g soil) | Total organic carbon (%) | |||||||||
| T 0 | 13.41 ± 4.09 a | 10.49 ± 3.98 a | 14.91 ± 1.74 a | 17.99 ± 3.77 a | 13.23 ± 2.54 a | 0.69 ± 0.49 a | 1.00 ± 0.58 a | 1.33 ± 0.01 a | 0.32 ± 0.04 a | 0.31 ± 0.03 a |
| T 10 | 17.11 ± 5.23 a | 13.82 ± 5.24 b | 18.04 ± 2.10 b | 22.11 ± 4.63 b | 15.27 ± 2.93 a | 2.09 ± 0.46 b | 2.65 ± 0.45 b | 2.13 ± 0.83 b | 1.67 ± 1.86 a | 2.53 ± 0.27 b |
| T 20 | 20.81 ± 6.36 ab | 17.15 ± 6.50 c | 21.18 ± 2.47 c | 26.24 ± 5.49 c | 17.31 ± 3.32 ab | 3.50 ± 0.61 c | 4.29 ± 0.32 c | 2.94 ± 1.67 b | 3.03 ± 3.68 b | 4.74 ± 0.51 c |
| T 30 | 24.52 ± 7.49 b | 20.48 ± 7.76 d | 24.31 ± 2.83 d | 30.37 ± 6.36 d | 19.35 ± 3.71 b | 4.91 ± 0.83 d | 5.94 ± 0.20 d | 3.75 ± 2.50 c | 4.38 ± 5.50 b | 6.96 ± 0.76 d |
| T 40 | 28.22 ± 8.62 c | 23.81 ± 9.02 e | 27.44 ± 3.20 e | 34.49 ± 7.22 e | 21.39 ± 4.11 c | 6.32 ± 1.09 e | 7.59 ± 0.12 e | 4.55 ± 3.34 d | 5.74 ± 7.32 c | 9.18 ± 1.00 e |
| Bacteria (thousands per gram dry weight) | Fungi (thousands per grams dry weight) | |||||||||
| T 0 | 122.80 ± 5.89 a | 124.93 ± 23.28 a | 87.81 ± 8.04 a | 120.03 ± 7.30 a | 113.42 ± 9.81 a | 56.87 ± 9.04 a | 52.66 ± 7.94 a | 60.82 ± 6.01 a | 55.85 ± 4.51 a | 52.55 ± 13.47 a |
| T 10 | 170.14 ± 9.30 b | 175.42 ± 27.91 b | 129.25 ± 7.72 b | 194.82 ± 8.80 b | 172.33 ± 11.84 b | 131.76 ± 5.53 b | 100.11 ± 5.43 b | 135.00 ± 7.81 b | 145.97 ± 0.79 b | 141.31 ± 3.22 b |
| T 20 | 217.48 ± 12.30 c | 225.91 ± 38.38 c | 170.68 ± 34.36 c | 269.60 ± 27.07 c | 231.24 ± 8.78 c | 206.76 ± 13.11 c | 146.57 ± 8.59 c | 210.94 ± 5.22 c | 239.98 ± 21.85 c | 229.08 ± 55.57 c |
| T 30 | 264.82 ± 1.23 d | 276.40 ± 7.62 d | 212.12 ± 16.48 d | 344.38 ± 25.33 d | 290.16 ± 34.18 d | 281.75 ± 3.08 d | 193.04 ± 3.41 d | 286.89 ± 0.44 d | 333.99 ± 7.47 d | 316.85 ± 4.38 d |
| T 40 | 312.16 ± 15.05 e | 326.89 ± 33.44 e | 253.56 ± 21.43 e | 419.17 ± 40.52 e | 349.07 ± 19.90 e | 356.43 ± 11.44 e | 242.44 ± 4.61 e | 357.56 ± 8.02 e | 416.33 ± 31.78 e | 407.60 ± 78.71 e |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Buta, M.; Blaga, G.; Paulette, L.; Păcurar, I.; Roșca, S.; Borsai, O.; Grecu, F.; Sînziana, P.E.; Negrușier, C. Soil Reclamation of Abandoned Mine Lands by Revegetation in Northwestern Part of Transylvania: A 40-Year Retrospective Study. Sustainability 2019, 11, 3393. https://doi.org/10.3390/su11123393
Buta M, Blaga G, Paulette L, Păcurar I, Roșca S, Borsai O, Grecu F, Sînziana PE, Negrușier C. Soil Reclamation of Abandoned Mine Lands by Revegetation in Northwestern Part of Transylvania: A 40-Year Retrospective Study. Sustainability. 2019; 11(12):3393. https://doi.org/10.3390/su11123393
Chicago/Turabian StyleButa, Mihai, Gheorghe Blaga, Laura Paulette, Ioan Păcurar, Sanda Roșca, Orsolya Borsai, Florina Grecu, Pauliuc Ecaterina Sînziana, and Cornel Negrușier. 2019. "Soil Reclamation of Abandoned Mine Lands by Revegetation in Northwestern Part of Transylvania: A 40-Year Retrospective Study" Sustainability 11, no. 12: 3393. https://doi.org/10.3390/su11123393
APA StyleButa, M., Blaga, G., Paulette, L., Păcurar, I., Roșca, S., Borsai, O., Grecu, F., Sînziana, P. E., & Negrușier, C. (2019). Soil Reclamation of Abandoned Mine Lands by Revegetation in Northwestern Part of Transylvania: A 40-Year Retrospective Study. Sustainability, 11(12), 3393. https://doi.org/10.3390/su11123393






