Kū Hou Kuapā: Cultural Restoration Improves Water Budget and Water Quality Dynamics in Heʻeia Fishpond
Abstract
1. Introduction
1.1. Native Hawaiian Fishpond Mariculture and Food Security
1.2. The Legacy of Land Use Change and Invasive Species on loko iʻa
1.3. Revitilization of loko iʻa: Heʻeia Fishpond as a Model
1.4. Biocultural Restoration of Heʻeia Fishpond: 2012–2018
2. Methods and Materials
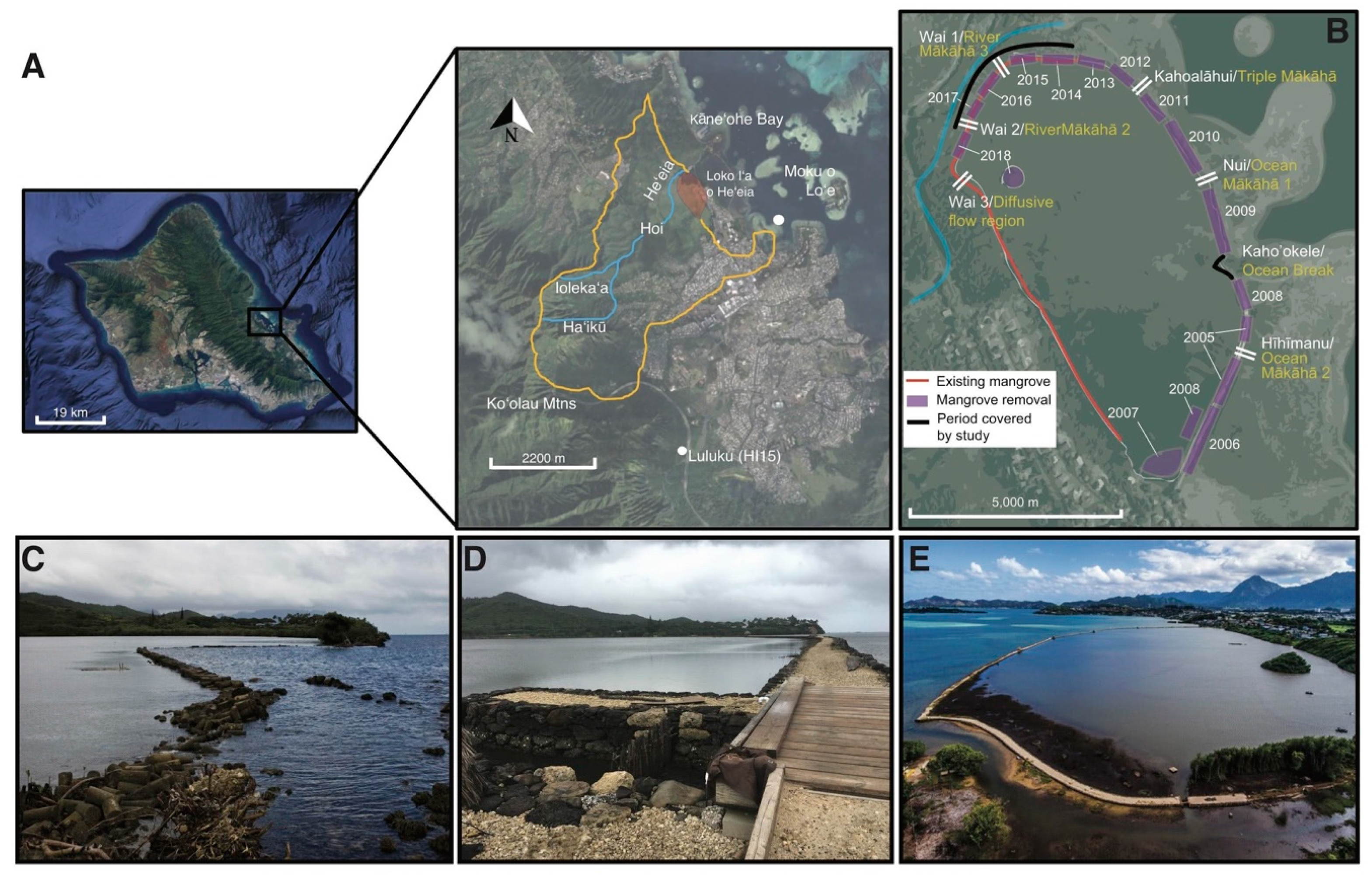
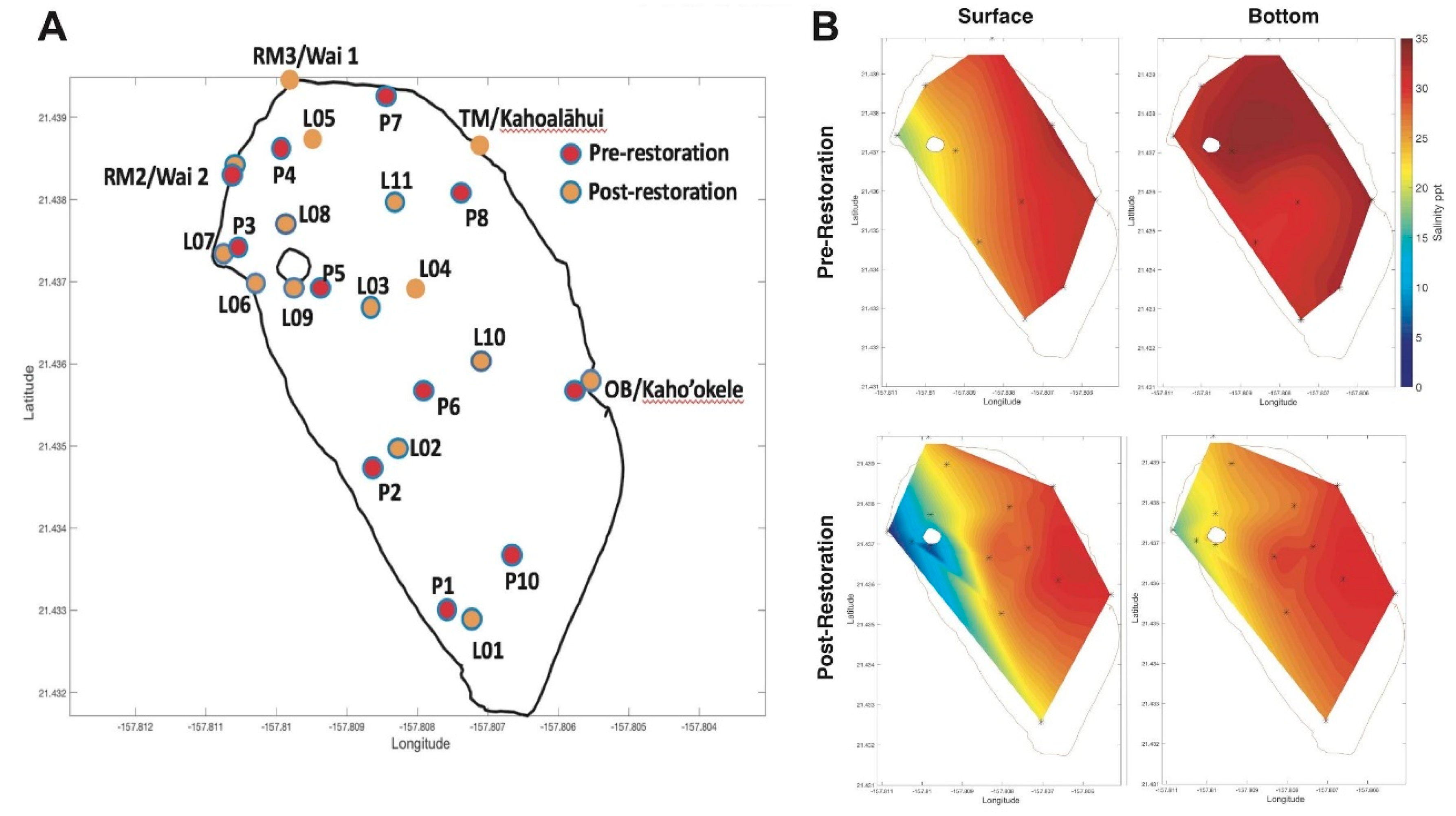
2.1. Study Site
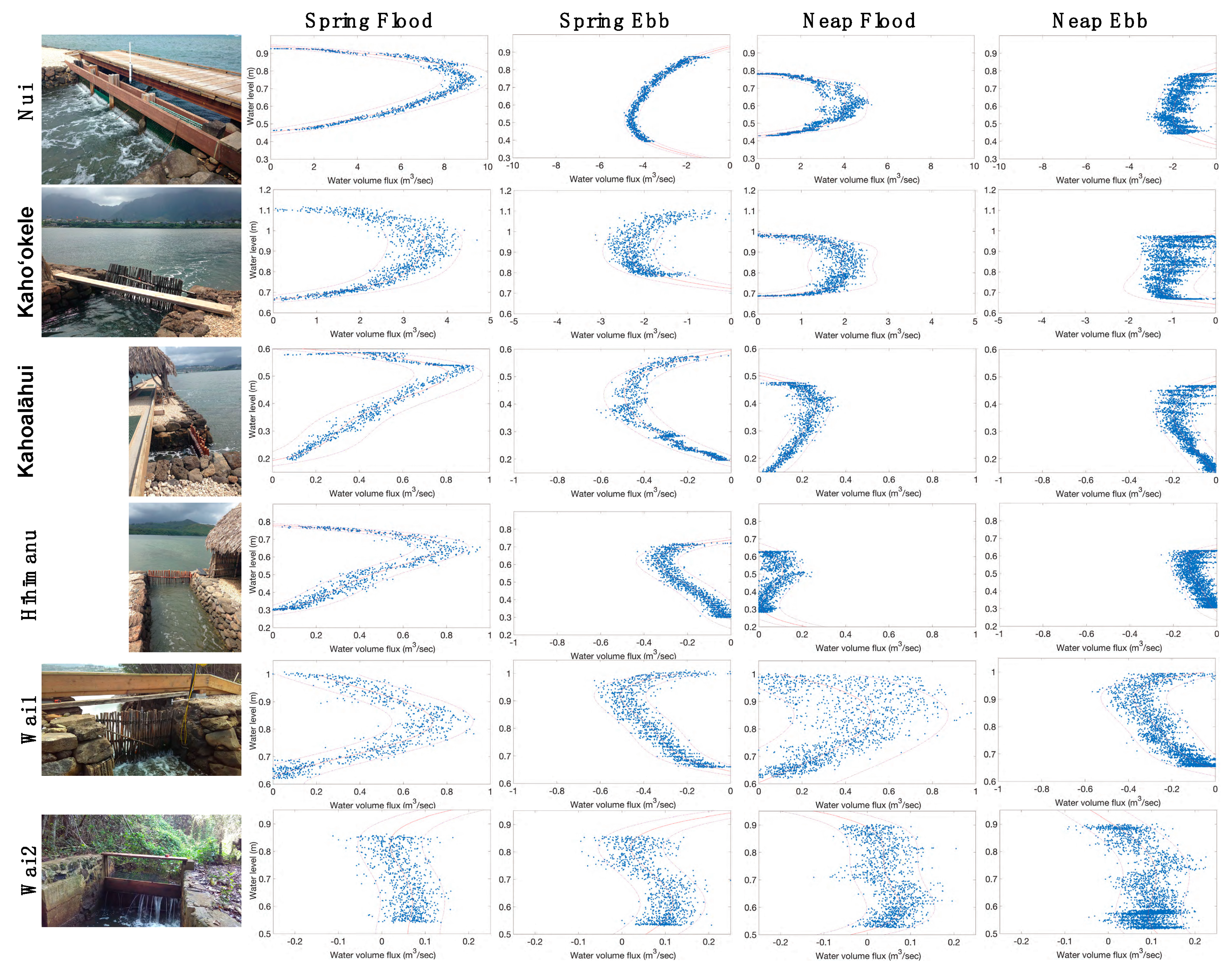
2.2. Water Volume Flux and Volume Change Calculations
2.3. Water Quality Sampling Regime
2.4. Microbial Source Tracking
2.5. Statistics
3. Results
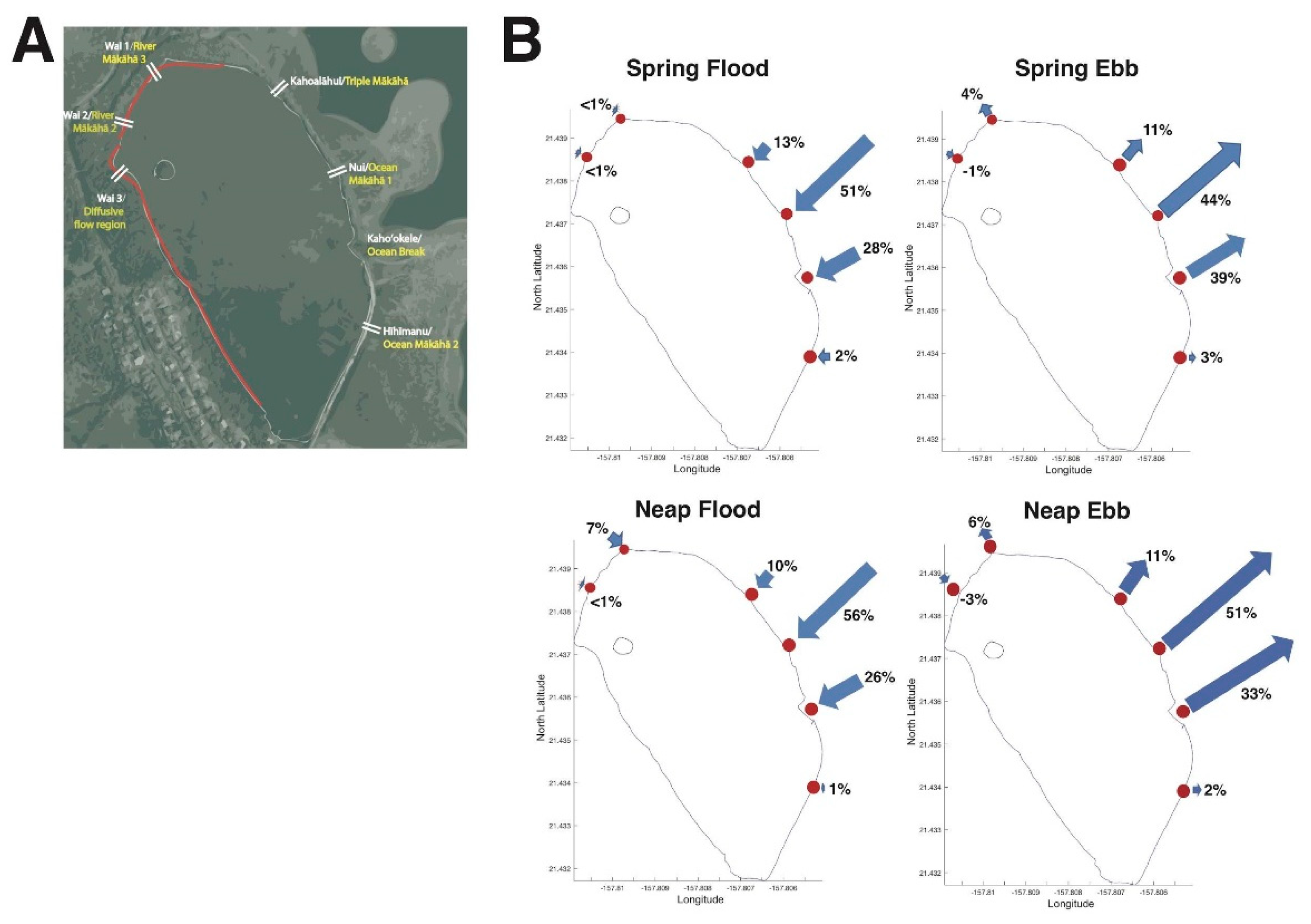
3.1. Restoration from 2014–2018 Shifted Relative Water Volume Flux Contributions of Each mākāhā
3.1.1. Characterizing mākāhā Water Volume Flux Post-Restoration (2018)
3.1.2. Changes in Relative Water Volume Flux Post-Restoration
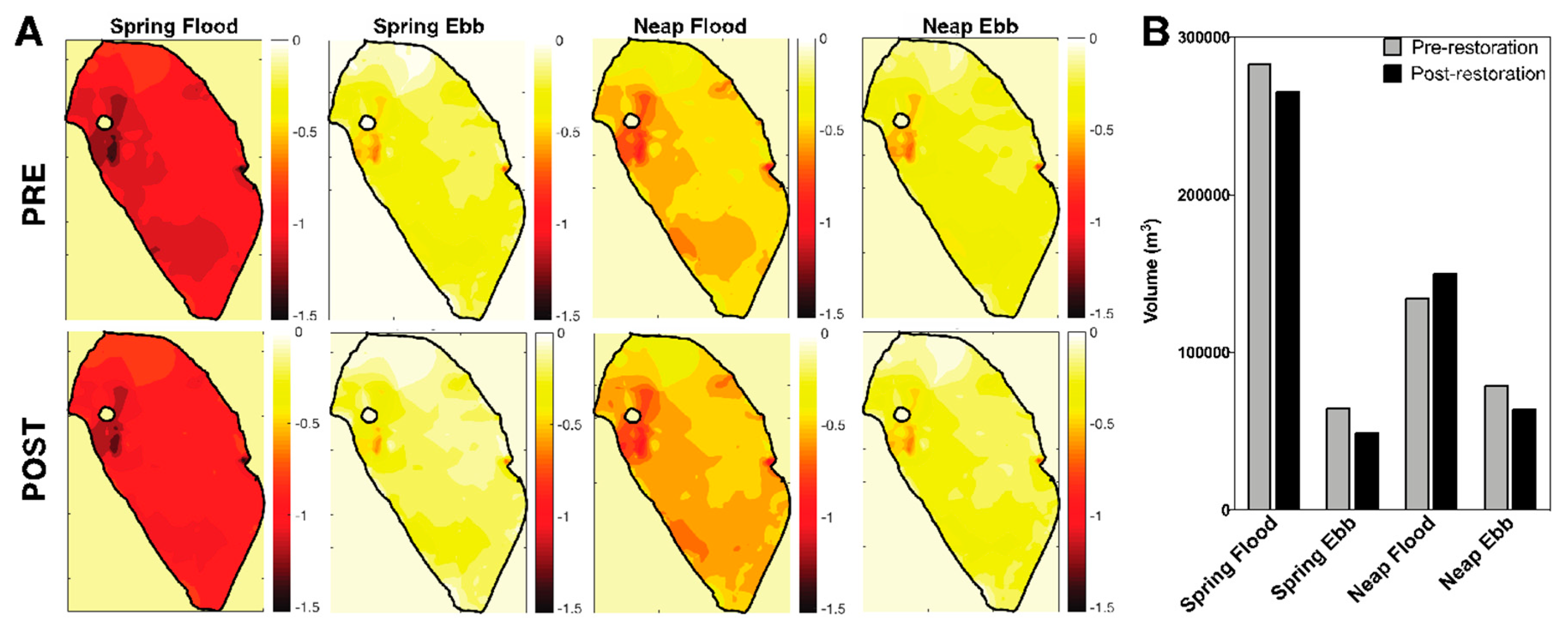
3.2. Decrease in loko iʻa Volume and Residence Time Post-Restoration
3.3. Spatial Salinity Distribution Significantly Altered due to Restoration
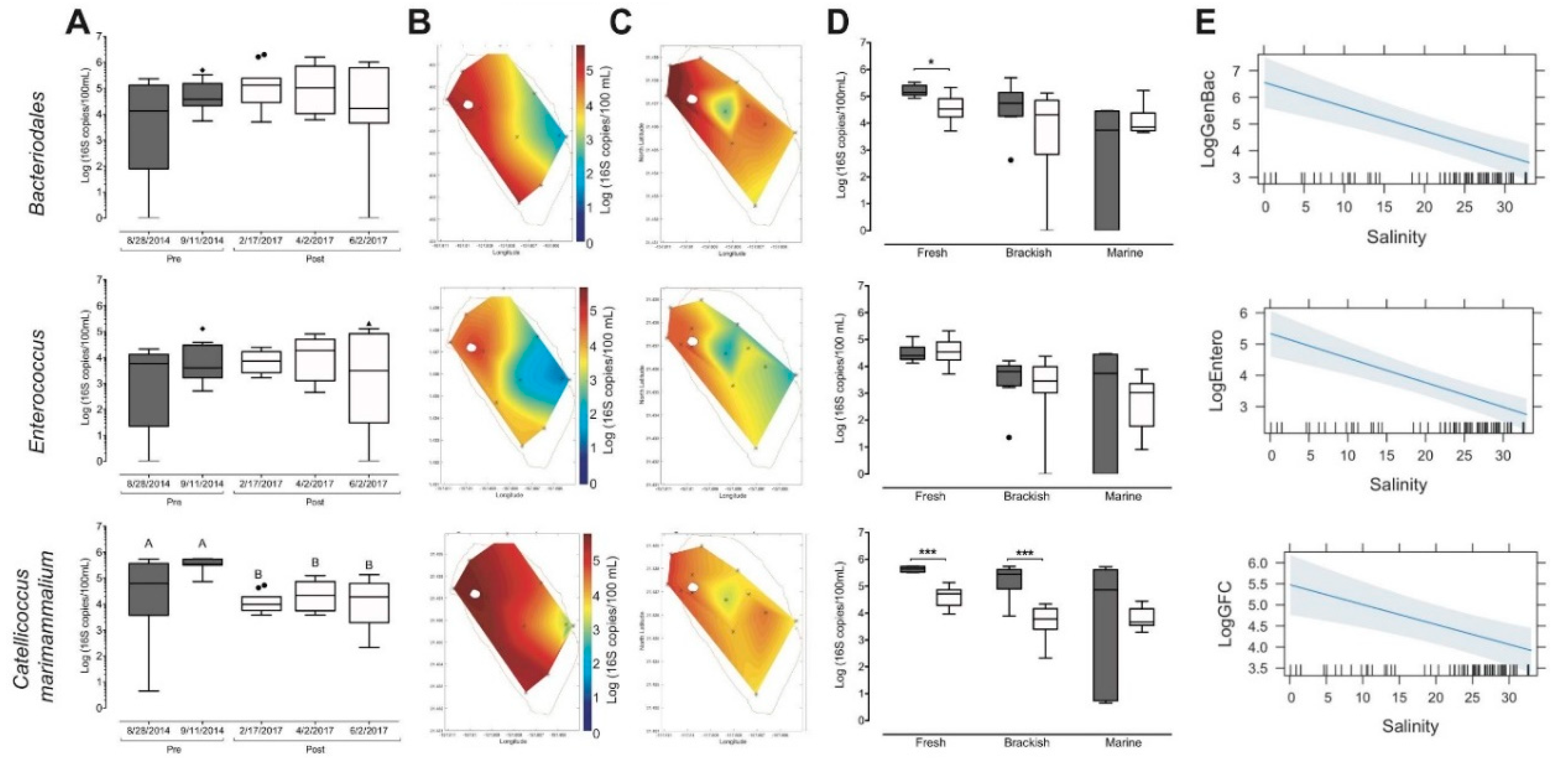
3.4. Restoration-Driven Changes to Circulation Altered Microbial Biomarker Spatial Distribution
4. Discussion
4.1. Hoʻoniho ka niho (Interlock the Stones [44]): Water Volume Flux Changes due to Kuapā Repair
4.2. Paepae ke alo (Raise the Face of the Wall [44]): Volume, Residence Time, and Salinity
4.3. Pani hakahaka (Close Gaps/Vacancies [44]): Microbial Indicators as Markers of Watershed Connectivity
4.4. Pōhaku ka papale (Place the Capstone on the Top [44]): Future Implications of Revitalizing Customary Fishpond Infrastructure
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mathieson, A.M. The state of world fisheries and aquaculture. In World Review of Fisheries and Aquaculture; FAO: Rome, Italy, 2012. [Google Scholar]
- Loke, M.K.; Geslani, C.; Takenaka, B.; Leung, P. Seafood consumption and supply sources in Hawaii, 2000–2009. Mar. Fish. Rev. 2012, 74, 44–51. [Google Scholar]
- Keala, G.; Hollyer, J.R. LOKO I’A; College of Tropical Agriculture and Human Resources, University of Hawai’i Mānoa: Honolulu, HI, USA, 2007; pp. 1–76. [Google Scholar]
- Kikuchi, W.K. Prehistoric Hawaiian fishponds. Science 1976, 193, 295–299. [Google Scholar] [CrossRef] [PubMed]
- Cobb, J.N. The Commercial Fisheries of the Hawaiian Islands in 1903; U.S. Government Printing Office: Washington, DC, USA, 1905.
- Keala, G.; Hollyer, J.R.; Castro, L. Loko Ia: A Manual on Hawaiian Fishpond Restoration and Management; University of Hawaii at Mānoa: Honolulu, HI, USA, 2007. [Google Scholar]
- Munro, G.C. Island of Moloka ʻi. In First Report of the Board of Commissioners of Agriculture and Forestry of the Territory of Hawaii for the Period from July; Board of Commissioners of Agriculture and Forestry: Honolulu, HI, USA, 1904; Volume 1, pp. 94–96. [Google Scholar]
- Gedan, K.B.; Kirwan, M.L.; Wolanski, E.; Barbier, E.B.; Silliman, B.R. The present and future role of coastal wetland vegetation in protecting shorelines: Answering recent challenges to the paradigm. Clim. Chang. 2011, 106, 7–29. [Google Scholar] [CrossRef]
- Twilley, R.W.; Lugo, A.E.; Patterson-Zucca, C. Litter production and turnover in basin mangrove forests in southwest Florida. Ecology 1986, 67, 670–683. [Google Scholar] [CrossRef]
- Chimner, R.A.; Fry, B.; Kaneshiro, M.Y.; Cormier, N. Current Extent and Historical Expansion of Introduced Mangroves on O’ahu, Hawai’i. Pac. Sci. 2006, 60, 377–383. [Google Scholar] [CrossRef]
- Allen, J.A. Mangroves as Alien Species: The Case of Hawaii. Glob. Ecol. Biogeogr. Lett. 1998, 7, 61–71. [Google Scholar] [CrossRef]
- Drigot, D.C. Mangrove Removal and Related Studies at Marine Corps Base Hawaii; Tech Note M-3N in Technical Notes: Case Studies from the Department of Defense Conservation Program; US Department of Defense Legacy Resource Management Program Publication: Kaneohe Bay, HI, USA, 1999; pp. 170–174. [Google Scholar]
- Walsh, G.E. An ecological study of a Hawaiian mangrove swamp. Estuaries 1967, 83, 420–431. [Google Scholar]
- Crooks, J.A. Characterizing ecosystem-level consequences of biological invasions: The role of ecosystem engineers. Oikos 2002, 97, 153–166. [Google Scholar] [CrossRef]
- Demopoulos, A.W.J.; Fry, B.; Smith, C.R. Food web structure in exotic and native mangroves: A Hawaii–Puerto Rico comparison. Oecologia 2007, 153, 675–686. [Google Scholar] [CrossRef]
- Sweetman, A.K.; Middelburg, J.J.; Berle, A.M.; Bernardino, A.F.; Schander, C.; Demopoulos, A.W.J.; Smith, C.R. Impacts of exotic mangrove forests and mangrove deforestation on carbon remineralization and ecosystem functioning in marine sediments. Biogeosciences 2010, 7, 2129–2145. [Google Scholar] [CrossRef]
- Wester, L. Introduction and Spread of Mangroves in the Hawaiian Islands. Yearb. Assoc. Pac. Coast Geogr. 1981, 43, 125–137. [Google Scholar] [CrossRef]
- Force, H.G.M.S.T.; Matsuoka, J.K. Hawaii. Governor’s Molokaʻi Subsistence Task Force Final Report; Task Force: Washington DC, USA, 1994. [Google Scholar]
- Matsuoka, J.K.; McGregor, D.P.; Minerbi, L. Molokai: A Study of Hawaiian Subsistence and Community Sustainability; Sustainable Community Development: Studies in Economic, Environmental, and Cultural Revitalizations; CRC Press: Boca Raton, FL, USA, 1998; pp. 25–44. [Google Scholar]
- Farber, J.M. Ancient Hawaiian Fishponds: Can Restoration Succeed on Moloka’i? Neptune House Publications: Encinitas, CA, USA, 1997. [Google Scholar]
- Declaration of Hui Malama Loko Iʻa. In. 2012. Available online: http://dlnr.hawaii.gov/occl/files/2015/07/Declaration-of-Hui-Malama.pdf (accessed on 19 November 2018).
- Kelly, M. Loko I’a O He’eia: Heeia Fishpond; Department of Anthropology, Bernice P. Bishop Museum: Honolulu, HI, USA, 1975. [Google Scholar]
- Banner, A.H. A Fresh-Water “Kill” on the Coral Reefs of Hawaii; Hawaii Institute of Marine Biology, University of Hawaii: Honolulu, HI, USA, 1968. [Google Scholar]
- Water Levels—NOAA Tides & Currents. Available online: https://tidesandcurrents.noaa.gov/waterlevels.html?id=1612480&units=standard&bdate=19650101&edate=19651201&timezone=GMT&datum=MLLW&interval=m&action=data (accessed on 15 September 2018).
- Brooks, M. Heʻeia Fishpond. In Proceedings of The Governor’s Molokaʻi Fishpond Restoration Workshop; Wyban, C.A., Ed.; Office of Hawaiian Affairs: Hilo, HI, USA, 1991; pp. 20–24. [Google Scholar]
- Vasconcellos, S.M.K. Distribution and Characteristics of a Photosynthetic Benthic Microbial Community in a Marine Coastal Pond; University of Hawaii Mānoa: Honolulu, HI, USA, 2007. [Google Scholar]
- Scott, T.M.; Rose, J.B.; Jenkins, T.M.; Farrah, S.R.; Lukasik, J. Microbial source tracking: Current methodology and future directions. Appl. Environ. Microbiol. 2002, 68, 5796–5803. [Google Scholar] [CrossRef] [PubMed]
- Kirs, M.; Kisand, V.; Wong, M.; Caffaro-Filho, R.A.; Moravcik, P.; Harwood, V.J.; Yoneyama, B.; Fujioka, R.S. Multiple lines of evidence to identify sewage as the cause of water quality impairment in an urbanized tropical watershed. Water Res. 2017, 116, 23–33. [Google Scholar] [CrossRef]
- McCoy, D.; McManus, M.A.; Kotubetey, K.; Kawelo, A.H.; Young, C.; D’Andrea, B.; Ruttenberg, K.C.; Alegado, R.A. Large-scale climatic effects on traditional Hawaiian fishpond aquaculture. PLoS ONE 2017, 12, e0187951. [Google Scholar] [CrossRef] [PubMed]
- Young, C.W. Perturbation of Nutrient Level Inventories and Phytoplankton Community Composition During Storm Events in a Tropical Coastal System: Heeia Fishpond, Oahu, Hawaii. Master’s Thesis, University of Hawaii Mānoa, Honolulu, HI, USA, 2011. [Google Scholar]
- National Weather Service Honolulu, HI. Available online: http://www.prh.noaa.gov/hnl/hydro/hydronet/hydronet-data.php (accessed on 19 November 2018).
- USGS Water Data for the Nation. Available online: https://waterdata.usgs.gov/nwis (accessed on 19 November 2018).
- Weather Observations: Moku o Loʻe, Oʻahu|PacIOOS. Available online: http://www.pacioos.hawaii.edu/weather/obs-mokuoloe/ (accessed on 19 November 2018).
- Timmerman, H.V.; Young, C.; Briggs, R.; D’Andrea, B.; McManus, M.A.; Vasconcellos, S.; Ruttenberg, K.S. Dynamics of Land-Ocean Linkages in a Semi-Enclosed Tropical Coastal System. Unpublished work. 2018. [Google Scholar]
- Ludwig, W.; Schleifer, K.H. How quantitative is quantitative PCR with respect to cell counts? Syst. Appl. Microbiol. 2000, 23, 556–562. [Google Scholar] [CrossRef]
- Haugland, R.A.; Siefring, S.C.; Wymer, L.J.; Brenner, K.P.; Dufour, A.P. Comparison of Enterococcus measurements in freshwater at two recreational beaches by quantitative polymerase chain reaction and membrane filter culture analysis. Water Res. 2005, 39, 559–568. [Google Scholar] [CrossRef] [PubMed]
- Method 1611: Enterococci in Water by TaqMan® Quantitative Polymerase Chain Reaction (qPCR) Assay; United States Environmental Protection Agency Office of Water (4303T): Washingdon, DC, USA, 2012.
- Dick, L.K.; Field, K.G. Rapid estimation of numbers of fecal Bacteroidetes by use of a quantitative PCR assay for 16S rRNA genes. Appl. Environ. Microbiol. 2004, 70, 5695–5697. [Google Scholar] [CrossRef]
- Siefring, S.; Varma, M.; Atikovic, E.; Wymer, L.; Haugland, R.A. Improved real-time PCR assays for the detection of fecal indicator bacteria in surface waters with different instrument and reagent systems. J. Water Health 2008, 6, 225–237. [Google Scholar] [CrossRef]
- Method B: Bacteroidales in Water by TaqMan® Quantitative Polymerase Chain Reaction (qPCR) Assay; United States Environmental Protection Agency Office of Water (4303T): Washingdon, DC, USA, 2010.
- Green, H.C.; Dick, L.K.; Gilpin, B.; Samadpour, M.; Field, K.G. Genetic markers for rapid PCR-based identification of gull, Canada goose, duck, and chicken fecal contamination in water. Appl. Environ. Microbiol. 2012, 78, 503–510. [Google Scholar] [CrossRef]
- Moehlenkamp, P. Kū Hou Kuapā: Increase of Water Exchange Rates and Changes in Microbial Source Tracking Markers Resulting from Restoration Regimes at Heʻeia Fishpond. Master’s Thesis, University of Hawaii Mānoa, Honolulu, HI, USA, 2018. [Google Scholar]
- Kleven, A. Coastal Groundwater Discharge as a Source of Nutrients to Heeia Fishpond, Oahu, HI. Bachelor’s Thesis, University of Hawaii at Mānoa, Honolulu, HI, USA, 2014. [Google Scholar]
- Paepae o Heʻeia. Hoʻoniho ka Niho. Unpublished work. 2015. [Google Scholar]
- Ertekin, R.C.; Yang, L.; Sundararaghavan, H. Hawaiian Fishpond Studies: Web Page Development and the Effect of Runoff from the Streams on Tidal Circulation; University of Hawaii at Mānoa: Honolulu, HI, USA, 1999; pp. 1–53. [Google Scholar]
- Ertekin, R.C. Molokai Fishpond Tidal Circulation Study; Final Report Submitted to the University of Hawaii Sea Grant College Program; University of Hawaii Sea Grant College Program: Honolulu, HI, USA, 1996. [Google Scholar]
- Yang, L. A Circulation Study of Hawaiian Fishponds; University of Hawaii, Department of Ocean and Resources Engineering: Honolulu, HI, USA, 2000. [Google Scholar]
- Dulai, H.; Kleven, A.; Ruttenberg, K.; Briggs, R.; Thomas, F. Evaluation of Submarine Groundwater Discharge as a Coastal Nutrient Source and Its Role in Coastal Groundwater Quality and Quantity. In Emerging Issues in Groundwater Resources; Advances in Water Security; Springer: Cham, Switzerland, 2016; pp. 187–221. ISBN 9783319320069. [Google Scholar]
- Noble, R.T.; Lee, I.M.; Schiff, K.C. Inactivation of indicator micro-organisms from various sources of faecal contamination in seawater and freshwater. J. Appl. Microbiol. 2004, 96, 464–472. [Google Scholar] [CrossRef]
- Shanks, O.C.; Kelty, C.A.; Sivaganesan, M.; Varma, M.; Haugland, R.A. Quantitative PCR for genetic markers of human fecal pollution. Appl. Environ. Microbiol. 2009, 75, 5507–5513. [Google Scholar] [CrossRef] [PubMed]
- Murphy, H. Persistence of Pathogens in Sewage and Other Water Types. In Global Water Pathogens Project Part; Rose, J.B., Jiménez-Cisneros, B., Eds.; Michigan State University: Lansing, MI, USA, 2017; Volume 4. [Google Scholar]
- Ortega, C.; Solo-Gabriele, H.M.; Abdelzaher, A.; Wright, M.; Deng, Y.; Stark, L.M. Correlations between microbial indicators, pathogens, and environmental factors in a subtropical Estuary. Mar. Pollut. Bull. 2009, 58, 1374–1381. [Google Scholar] [CrossRef] [PubMed]
- Shehane, S.D.; Harwood, V.J.; Whitlock, J.E.; Rose, J.B. The influence of rainfall on the incidence of microbial faecal indicators and the dominant sources of faecal pollution in a Florida river. J. Appl. Microbiol. 2005, 98, 1127–1136. [Google Scholar] [CrossRef] [PubMed]
- Sinigalliano, C.D.; Ervin, J.S.; Van De Werfhorst, L.C.; Badgley, B.D.; Ballesté, E.; Bartkowiak, J.; Boehm, A.B.; Byappanahalli, M.; Goodwin, K.D.; Gourmelon, M.; et al. Multi-laboratory evaluations of the performance of Catellicoccus marimammalium PCR assays developed to target gull fecal sources. Water Res. 2013, 47, 6883–6896. [Google Scholar] [CrossRef]
- Ryu, H.; Griffith, J.F.; Khan, I.U.H.; Hill, S.; Edge, T.A.; Toledo-Hernandez, C.; Gonzalez-Nieves, J.; Santo Domingo, J. Comparison of gull feces-specific assays targeting the 16S rRNA genes of Catellicoccus marimammalium and Streptococcus spp. Appl. Environ. Microbiol. 2012, 78, 1909–1916. [Google Scholar] [CrossRef] [PubMed]
- Cloutier, D.D.; McLellan, S.L. Distribution and Differential Survival of Traditional and Alternative Indicators of Fecal Pollution at Freshwater Beaches. Appl. Environ. Microbiol. 2017, 83, e02881-16. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.; Marion, J.W.; Lee, J. Development and application of a quantitative PCR assay targeting Catellicoccus marimammalium for assessing gull-associated fecal contamination at Lake Erie beaches. Sci. Total Environ. 2013, 454–455, 1–8. [Google Scholar] [CrossRef]
| Mākāhā | Latitude | Longitude | Heading | Width (m) |
|---|---|---|---|---|
| Hīhīmanu/Ocean Mākāhā 2 | 21.4357389 | −157.80531 | 111°/291° | 2.00 |
| Kahoʻokele/Ocean Break | 21.4372333 | −157.80583 | 80°/260° | 3.05 |
| Nui/Ocean Mākāā 1 | 21.4384222 | −157.80675 | 63°/243° | 6.48 |
| Kahoalāhui Kealohi/Triple Mākāhā 1 | 21.4396667 | −157.80993 | 48°/228° | 1.88 |
| Kahoalāhui Koʻa Mano/Triple Mākāhā 2 | 21.4396667 | −157.80993 | 48°/228° | 1.78 |
| Kahoalāhui Kekepa/Triple Mākāhā 3 | 21.4396667 | −157.80993 | 48°/228° | 1.55 |
| Wai 1/River Mākāhā 3 | 21.4386034 | −157.81072 | 310°/130° | 2.18 |
| Wai 2/River Mākāhā 2 | 21.4379231 | −157.80782 | 290°/110° | 1.85 |
| Diffuse flow region/River Makāhā 1 | 21.4386583 | −157.81077 | n/a | n/a |
| Target | Primer | Sequence | References |
|---|---|---|---|
| Enteroccocus | Entero1af | AGAAATTCCAAACGAACTTG | [35,36,37] |
| Entero1ar | CAGTGCTCTACCTCCATCATT | [35,36,37] | |
| Entero1ap | 6-FAM™/TGGTTCTCT/ZEN™/CCGAAATAGCTTTAGGGCTA/IB®FQ/ | [35,36,37] | |
| Bacteroidales | GenBac3f | GGGGTTCTGAGAGGAAGGT | [38,39,40] |
| GenBac3r | CCGTCATCCTTCACGCTACT | [38,39,40] | |
| GenBac3p | 6-FAM™/CAATATTCC/ZEN™/TCACTGCTGCCTCCCGTA/IB®FQ/ | [38,39,40] | |
| Catellicoccus marimammalium | GFCf | CCC TTG TCG TTA GTT GCC ATC ATT C | [41] |
| GFCr | GCC CTC GCG AGT TCG CTG C | [41] |
| Mean WVF (m3 s−1) | Peak WVF (m3 s−1) | Tidal Cycle Length (h) | Cum. Flux per Tidal Cycle (m3) | WVF Rate (m3 h−1) | Volume Exchanged per Tidal Cycle (m3) | Relative WVF | |
|---|---|---|---|---|---|---|---|
| Spring Flood | 191660 | 31778 | 191660 | 100.00% | |||
| Wai 2 | 0.05 | 0.16 | 4.43 | 840 | 190 | 840 | 0.44% |
| Wai1 | 0.40 | 0.93 | 4.55 | 7140 | 1569 | 7140 | 3.37% |
| Kahoalāhui | 1.47 | 2.76 | 4.36 | 24420 | 5601 | 24420 | 12.74% |
| Nui | 4.18 | 9.70 | 6.29 | 97800 | 15548 | 97800 | 51.03% |
| Kahoʻokele | 2.02 | 4.69 | 7.29 | 54380 | 7460 | 54380 | 28.37% |
| Hīhīmanu | 0.39 | 0.95 | 5.02 | 7080 | 1410 | 7080 | 3.69% |
| Spring Ebb | −174880 | −30851 | −174880 | 100.00% | |||
| Wai 2 | 0.07 | −0.09 | 5.50 | 1560 | 284 | 1560 | −0.89% |
| Wai1 | −0.32 | −0.63 | 6.32 | −7600 | −1203 | −7600 | 4.35% |
| Kahoalāhui | −0.87 | −1.86 | 6.31 | −20220 | −3204 | −20220 | 11.56% |
| Nui | −3.60 | −4.86 | 5.53 | −76320 | −13801 | −76320 | 43.64% |
| Kahoʻokele | −1.10 | −3.12 | 5.50 | −67520 | −12276 | −67520 | 38.61% |
| Hīhīmanu | −0.17 | −0.43 | 7.35 | −4780 | −650 | −4780 | 2.73% |
| Neap Flood | 141384 | 16717 | 141384 | 100.00% | |||
| Wai 2 | 0.05 | 0.20 | 7.41 | 1300 | 175 | 1300 | 0.92% |
| Wai1 | 0.32 | 0.98 | 8.29 | 9720 | 1172 | 9720 | 6.87% |
| Kahoalāhui | 0.51 | 1.08 | 7.31 | 13620 | 1863 | 13620 | 9.63% |
| Nui | 2.26 | 5.41 | 9.46 | 78744 | 8324 | 78744 | 55.70% |
| Kahoʻokele | 1.35 | 2.52 | 7.30 | 36440 | 4992 | 36440 | 25.77% |
| Hīhīmanu | 0.05 | 0.24 | 8.20 | 1560 | 190 | 1560 | 1.10% |
| Neap Ebb | −159938 | −10584 | −159938 | 100.00% | |||
| Wai 2 | 0.88 | −0.09 | 17.46 | 5640 | 323 | 5640 | −3.53% |
| Wai1 | −0.17 | −0.57 | 15.50 | −9880 | −637 | −9880 | 6.18% |
| Kahoalāhui | −0.30 | −0.9 | 15.50 | −17100 | −1103 | −17100 | 10.69% |
| Nui | −1.60 | −3.19 | 14.09 | −81298 | −5770 | −81298 | 50.83% |
| Kahoʻokele | −0.86 | −1.80 | 17.10 | −53280 | −3116 | −53280 | 33.31% |
| Hīhīmanu | −0.08 | −0.25 | 14.34 | −4020 | −280 | −4020 | 2.51% |
| Mākāhā | Flood Tide | Ebb Tide | ||||||
|---|---|---|---|---|---|---|---|---|
| Pre-Restoration | Post-Restoration | Pre-Restoration | Post-Restoration | |||||
| Volume Exchange per Tidal Cycle (m3) | Relative WVF | Volume Exchange per Tidal Cycle (m3) | Relative WVF | Volume Exchange per Tidal Cycle (m3) | Relative WVF | Volume Exchanged per Tidal Cycle (m3) | Relative WVF | |
| Wai 2 | 2057 | 0.85% | 1300 | 0.67% | −5515 | 2.28% | 5640 | −3.25% |
| Wai 1 | 2249 | 0.93% | 9720 | 5.10% | −5791 | 2.40% | −9880 | 5.70% |
| Kahoalāhui | 4106 | 1.71% | 24420 | 12.54% | −5802 | 2.41% | −20220 | 11.68% |
| Nui | 31101 | 12.88% | 97800 | 50.24% | −26886 | 11.12% | −76320 | 44.10% |
| Kahoʻokele/OB | 197820 | 81.94% | 54380 | 27.93% | −192780 | 79.76% | −67520 | 39.01% |
| Hīhīmanu | 4081 | 1.69% | 7080 | 3.61% | −4912 | 2.03% | −4780 | 2.76% |
| Mākāhā Total | 241,413 | 100.00% | 194,700 | 100.00% | −241,685 | 100.00% | −173,080 | 100.00% |
| Bacterial Indicator | Estimate | Std. Error | df | t Value | Pr (>|t|) | Signif. Codes 1 | ||
|---|---|---|---|---|---|---|---|---|
| C. marimammalium | Intercept | 4.99266 | 0.37574 | 9.16611 | 13.287 | 2.69 × 10−7 | *** | |
| Pre-vs. post repair | 0.96856 | 0.4402 | 4.42676 | 2.2 | 0.086085 | |||
| Salinity | −0.04652 | 0.01226 | 57.66017 | −3.794 | 0.000357 | *** | ||
| Bacteroidales | Intercept | 6.36231 | 0.48919 | 10.03302 | 13.006 | 1.32 × 10−7 | *** | |
| Pre-vs. post repair | 0.45987 | 0.56039 | 4.46269 | 0.821 | 0.453 | |||
| Salinity | −0.09205 | 0.01671 | 57.73214 | −5.509 | 8.75 × 10−7 | *** | ||
| Enterococcus | Intercept | 5.14077 | 0.36391 | 12.62958 | 14.127 | 4.12 × 10−9 | *** | |
| Pre-vs. post repair | 0.45003 | 0.39462 | 4.63367 | 1.14 | 0.31 | |||
| Salinity | −0.0794 | 0.01361 | 57.97529 | −5.823 | 2.66 × 10−7 | *** | ||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Möhlenkamp, P.; Beebe, C.K.; McManus, M.A.; Kawelo, A.H.; Kotubetey, K.; Lopez-Guzman, M.; Nelson, C.E.; Alegado, R.ʻ. Kū Hou Kuapā: Cultural Restoration Improves Water Budget and Water Quality Dynamics in Heʻeia Fishpond. Sustainability 2019, 11, 161. https://doi.org/10.3390/su11010161
Möhlenkamp P, Beebe CK, McManus MA, Kawelo AH, Kotubetey K, Lopez-Guzman M, Nelson CE, Alegado Rʻ. Kū Hou Kuapā: Cultural Restoration Improves Water Budget and Water Quality Dynamics in Heʻeia Fishpond. Sustainability. 2019; 11(1):161. https://doi.org/10.3390/su11010161
Chicago/Turabian StyleMöhlenkamp, Paula, Charles Kaiaka Beebe, Margaret A. McManus, Angela Hiʻilei Kawelo, Keliʻiahonui Kotubetey, Mirielle Lopez-Guzman, Craig E. Nelson, and Rosanna ʻAnolani Alegado. 2019. "Kū Hou Kuapā: Cultural Restoration Improves Water Budget and Water Quality Dynamics in Heʻeia Fishpond" Sustainability 11, no. 1: 161. https://doi.org/10.3390/su11010161
APA StyleMöhlenkamp, P., Beebe, C. K., McManus, M. A., Kawelo, A. H., Kotubetey, K., Lopez-Guzman, M., Nelson, C. E., & Alegado, R. ʻ. (2019). Kū Hou Kuapā: Cultural Restoration Improves Water Budget and Water Quality Dynamics in Heʻeia Fishpond. Sustainability, 11(1), 161. https://doi.org/10.3390/su11010161





