Biogas Potential of Coffee Processing Waste in Ethiopia
Abstract
1. Introduction
2. Materials and Methods
2.1. Raw Materials
2.2. Inoculum
2.3. Chemical Analysis
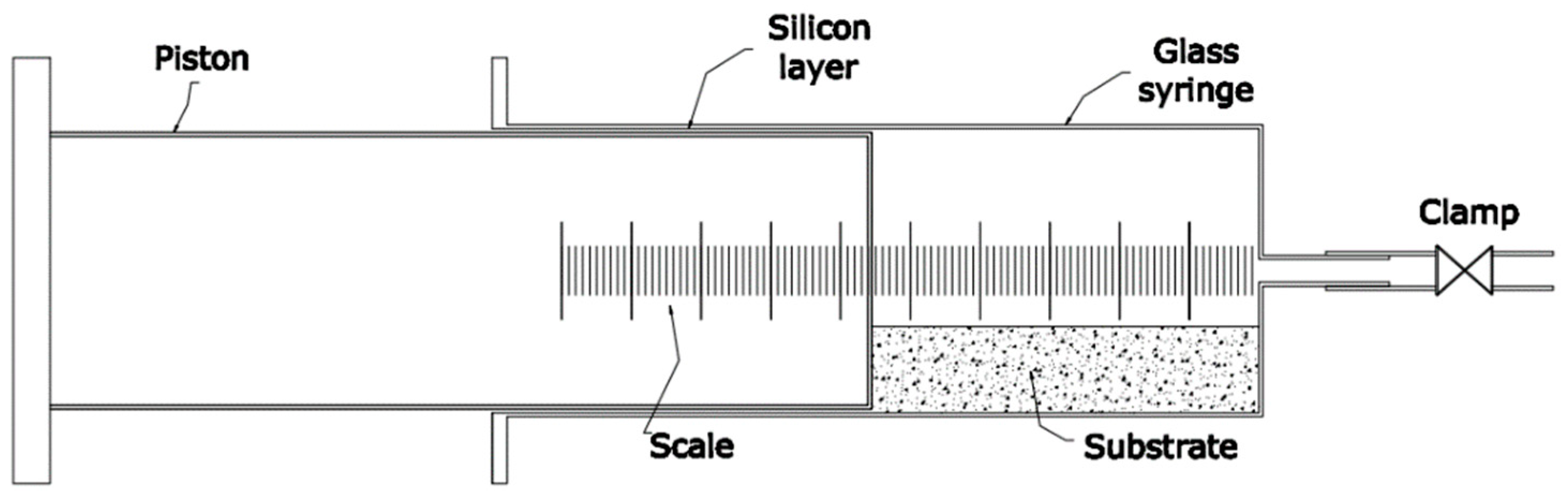
2.4. Anaerobic Batch Digestion Tests
3. Results and Discussion
3.1. Chemical Composition
3.2. Elemental Analysis
3.3. HBT Analysis
3.4. Energy Potential of Coffee by-Products in Ethiopia
4. Conclusions
Nomenclatures
| Symbol | Definition |
| Degree of volatile solids degradation | |
| Concentration of volatile solids in dry substrate | |
| Mass of biogas | |
| Molar mass of methane | |
| Molar mass of carbon dioxide | |
| Concentration of methane in the biogas | |
| Concentration of carbon dioxide in the biogas | |
| GE | Gross energy of substrate |
| SMY | Specific methane yield |
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chanakya, H.N.; De Alwis, A.A.P. Environmental issues and management in primary coffee processing. Process Saf. Environ. 2004, 82, 291–300. [Google Scholar] [CrossRef]
- Labouisse, J.-P.; Bellachew, B.; Kotecha, S.; Bertrand, B. Current status of coffee (coffea arabica l.) genetic resources in ethiopia: Implications for conservation. Genet. Resour. Crop Evol. 2008, 55, 1079–1093. [Google Scholar] [CrossRef]
- Petit, N. Ethiopia’s coffee sector: A bitter or better future? J. Agrar. Chang. 2007, 7, 225–263. [Google Scholar] [CrossRef]
- Minten, B.; Tamru, S.; Kuma, T.; Nyarko, Y. Structure and Performance of Ethiopia’s Coffee Export Sector; International Food Policy Research Institute: Washington, DC, USA, 2014; Volume 66. [Google Scholar]
- Food Agriculture Organization of the United Nation. Faostat Statistics Database; Food Agriculture Organization of the United Nation: Rome, Italy, 2018. [Google Scholar]
- Gadhamshetty, V.; Arudchelvam, Y.; Nirmalakhandan, N.; Johnson, D.C. Modeling dark fermentation for biohydrogen production: Adm1-based model vs. Gompertz model. Int. J. Hydrog. Energy 2010, 35, 479–490. [Google Scholar] [CrossRef]
- Franca, A.S.; Oliveira, L.S. Coffee processing solid wastes: Current uses and future perspectives. In Agricultural Wastes; Ashworth, G.S., Azevedo, P., Eds.; Nova Science Publishers, Inc.: Hauppauge, NY, USA, 2009; pp. 155–190. [Google Scholar]
- Esquivel, P.; Jiménez, V.M. Functional properties of coffee and coffee by-products. Food Res. Int. 2012, 46, 488–495. [Google Scholar] [CrossRef]
- Beyene, A.; Kassahun, Y.; Addis, T.; Assefa, F.; Amsalu, A.; Legesse, W.; Kloos, H.; Triest, L. The impact of traditional coffee processing on river water quality in ethiopia and the urgency of adopting sound environmental practices. Environ. Monit. Assess. 2012, 184, 7053–7063. [Google Scholar] [CrossRef] [PubMed]
- Shemekite, F.; Gómez-Brandón, M.; Franke-Whittle, I.H.; Praehauser, B.; Insam, H.; Assefa, F. Coffee husk composting: An investigation of the process using molecular and non-molecular tools. Waste Manag. 2014, 34, 642–652. [Google Scholar] [CrossRef] [PubMed]
- Bakker, R. Availability of Lignocellulosic Feedstocks for Lactic Acid Production-Feedstock Availability, Lactic Acid Production Potential and Selection criteria; Wageningen UR-Food & Biobased Research: Wageningen, The Netherlands, 2013. [Google Scholar]
- Mengistu, M.G.; Simane, B.; Eshete, G.; Workneh, T.S. A review on biogas technology and its contributions to sustainable rural livelihood in ethiopia. Renew. Sustain. Energy Rev. 2015, 48, 306–316. [Google Scholar] [CrossRef]
- Gwavuya, S.G.; Abele, S.; Barfuss, I.; Zeller, M.; Müller, J. Household energy economics in rural ethiopia: A cost-benefit analysis of biogas energy. Renew. Energy 2012, 48, 202–209. [Google Scholar] [CrossRef]
- Tong, X.; Smith, L.H.; McCarty, P.L. Methane fermentation of selected lignocellulosic materials. Biomass 1990, 21, 239–255. [Google Scholar] [CrossRef]
- Triolo, J.M.; Sommer, S.G.; Møller, H.B.; Weisbjerg, M.R.; Jiang, X.Y. A new algorithm to characterize biodegradability of biomass during anaerobic digestion: Influence of lignin concentration on methane production potential. Bioresour. Technol. 2011, 102, 9395–9402. [Google Scholar] [CrossRef] [PubMed]
- Mussatto, S.I.; Fernandes, M.; Milagres, A.M.F.; Roberto, I.C. Effect of hemicellulose and lignin on enzymatic hydrolysis of cellulose from brewer’s spent grain. Enzyme Microb. Technol. 2008, 43, 124–129. [Google Scholar] [CrossRef]
- Fischer, E.; Schmidt, T.; Hora, S.; Geirsdorf, J.; Stinner, W.; Scholwin, F. Agro-Industrial Biogas in Kenya: Potentials, Estimates for Tariffs, Policy and Business Recommendations; German International Cooperation (GIZ): Berlin, Germany, 2010. [Google Scholar]
- Ulsido, M.D.; Zeleke, G.; Li, M. Biogas potential assessment from a coffee husk: An option for solid waste management in gidabo watershed of ethiopia. Eng. Rural Dev. 2016, 1348–1354. [Google Scholar]
- Kivaisi, A.K.; Rubindamayugi, M.S.T. The potential of agro-industrial residues for production of biogas and electricity in tanzania. Renew. Energy 1996, 9, 917–921. [Google Scholar] [CrossRef]
- Baier, U.; Schleiss, K. Greenhouse gas emission reduction through anaerobic digestion of coffee pulp. In Proceedings of the 4th International Symposium Anaerobic Digestion of Solid Waste, Copenhagen, Denmark, 31 August–2 September 2005. [Google Scholar]
- Adams, M.R.; Dougan, J. Waste products. In Coffee: Volume 2: Technology; Clarke, R.J., Macrae, R., Eds.; Springer Netherlands: Dordrecht, The Netherlands, 1987; pp. 257–291. [Google Scholar]
- Jayachandra, T.; Venugopal, C.; Anu Appaiah, K.A. Utilization of phytotoxic agro waste—Coffee cherry husk through pretreatment by the ascomycetes fungi mycotypha for biomethanation. Energy Sustain. Dev. 2011, 15, 104–108. [Google Scholar] [CrossRef]
- Porres, C.; Alvarez, D.; Calzada, J. Caffeine reduction in coffee pulp through silage. Biotechnol. Adv. 1993, 11, 519–523. [Google Scholar] [CrossRef]
- Jungbluth, T.; Büscher, W.; Krause, M. Technik Tierhaltung; Eugen Ulmer: Stuttgart, Germany, 2017; Volume 2641. [Google Scholar]
- Weiland, P. Biogas production: Current state and perspectives. Appl. Microbiol. Biotechnol. 2010, 85, 849–860. [Google Scholar] [CrossRef] [PubMed]
- Roati, C.; Fiore, S.; Ruffino, B.; Marchese, F.; Novarino, D.; Zanetti, M. Preliminary evaluation of the potential biogas production of food-processing industrial wastes. Am. J. Environ. Sci. 2012, 8, 291. [Google Scholar]
- The World Factbook: 2018; Central Intelligence Agency: Washington, DC, USA, 2018.
- WorldBank. Worlddatabank:Ethiopia; WorldBank: Washington, DC, USA, 2018. [Google Scholar]
- Mittweg, G.; Oechsner, H.; Hahn, V.; Lemmer, A.; Reinhardt-Hanisch, A. Repeatability of a laboratory batch method to determine the specific biogas and methane yields. Eng. Life Sci. 2012, 12, 270–278. [Google Scholar] [CrossRef]
- Helffrich, D.; Oechsner, H. The hohenheim biogas yield test: Comparison of different laboratory techniques for the digestion of biomass. Landtechnik 2003, 58, 148–149. [Google Scholar]
- VDI. Vdi 4630—Fermentation of organic materials, characterisation of substrate, sampling, collection of material data, fermentation tests [1872]vdi gesellschaft energietechnik. In VDI Handbuch Energietechnik; Beuth Verlag GmbH: Berlin, Germany, 2006; pp. 44–59. [Google Scholar]
- Lindner, J.; Zielonka, S.; Oechsner, H.; Lemmer, A. Effect of different ph-values on process parameters in two-phase anaerobic digestion of high-solid substrates. Environ. Technol. 2015, 36, 198–207. [Google Scholar] [CrossRef] [PubMed]
- Mönch-Tegeder, M.; Lemmer, A.; Oechsner, H.; Jungbluth, T. Investigation of the methane potential of horse manure. Agric. Eng. Int. CIGR J. 2013, 15, 161–172. [Google Scholar]
- Mao, C.; Feng, Y.; Wang, X.; Ren, G. Review on research achievements of biogas from anaerobic digestion. Renew. Sustain. Energy Rev. 2015, 45, 540–555. [Google Scholar] [CrossRef]
- Oechsner, H.; Lemmer, A.; Ramhold, D.; Mathies, E.; Mayrhuber, E.; Preissler, D. Method for Producing Biogas in Controlled Concentrations of Trace Elements. U.S. Patent Application DE200850002359, 29 May 2008. [Google Scholar]
- Schattauer, A.; Abdoun, E.; Weiland, P.; Plöchl, M.; Heiermann, M. Abundance of trace elements in demonstration biogas plants. Biosyst. Eng. 2011, 108, 57–65. [Google Scholar] [CrossRef]
- Facchin, V.; Cavinato, C.; Fatone, F.; Pavan, P.; Cecchi, F.; Bolzonella, D. Effect of trace element supplementation on the mesophilic anaerobic digestion of foodwaste in batch trials: The influence of inoculum origin. Biochem. Eng. J. 2013, 70, 71–77. [Google Scholar] [CrossRef]
- Zhang, L.; Lee, Y.W.; Jahng, D. Anaerobic co-digestion of food waste and piggery wastewater: Focusing on the role of trace elements. Bioresour. Technol. 2011, 102, 5048–5059. [Google Scholar] [CrossRef] [PubMed]
- Chala, B.; Oechsner, H.; Fritz, T.; Latif, S.; Müller, J. Increasing the loading rate of continuous stirred tank reactor for coffee husk and pulp: Effect of trace elements supplement. Eng. Life Sci. 2017. [Google Scholar] [CrossRef]
- Chen, Y.; Cheng, J.J.; Creamer, K.S. Inhibition of anaerobic digestion process: A review. Bioresour. Technol. 2008, 99, 4044–4064. [Google Scholar] [CrossRef] [PubMed]
- Triolo, J.M.; Pedersen, L.; Qu, H.; Sommer, S.G. Biochemical methane potential and anaerobic biodegradability of non-herbaceous and herbaceous phytomass in biogas production. Bioresour. Technol. 2012, 125, 226–232. [Google Scholar] [CrossRef] [PubMed]
- Rojas-Sossa, J.P.; Murillo-Roos, M.; Uribe, L.; Uribe-Lorio, L.; Marsh, T.; Larsen, N.; Chen, R.; Miranda, A.; Solís, K.; Rodriguez, W.; et al. Effects of coffee processing residues on anaerobic microorganisms and corresponding digestion performance. Bioresour. Technol. 2017, 245, 714–723. [Google Scholar] [CrossRef] [PubMed]
- Thomsen, S.T.; Spliid, H.; Østergård, H. Statistical prediction of biomethane potentials based on the composition of lignocellulosic biomass. Bioresour. Technol. 2014, 154, 80–86. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.T.; Brulé, M.; Maurer, C.; Argyropoulos, D.; Müller, J.; Oechsner, H. Batch anaerobic digestion of banana waste-energy potential and modelling of methane production kinetics. Agric. Eng. Int. CIGR J. 2016, 18, 110–128. [Google Scholar]
- Haag, N.L.; Nägele, H.-J.; Reiss, K.; Biertümpfel, A.; Oechsner, H. Methane formation potential of cup plant (silphium perfoliatum). Biomass Bioenergy 2015, 75, 126–133. [Google Scholar] [CrossRef]
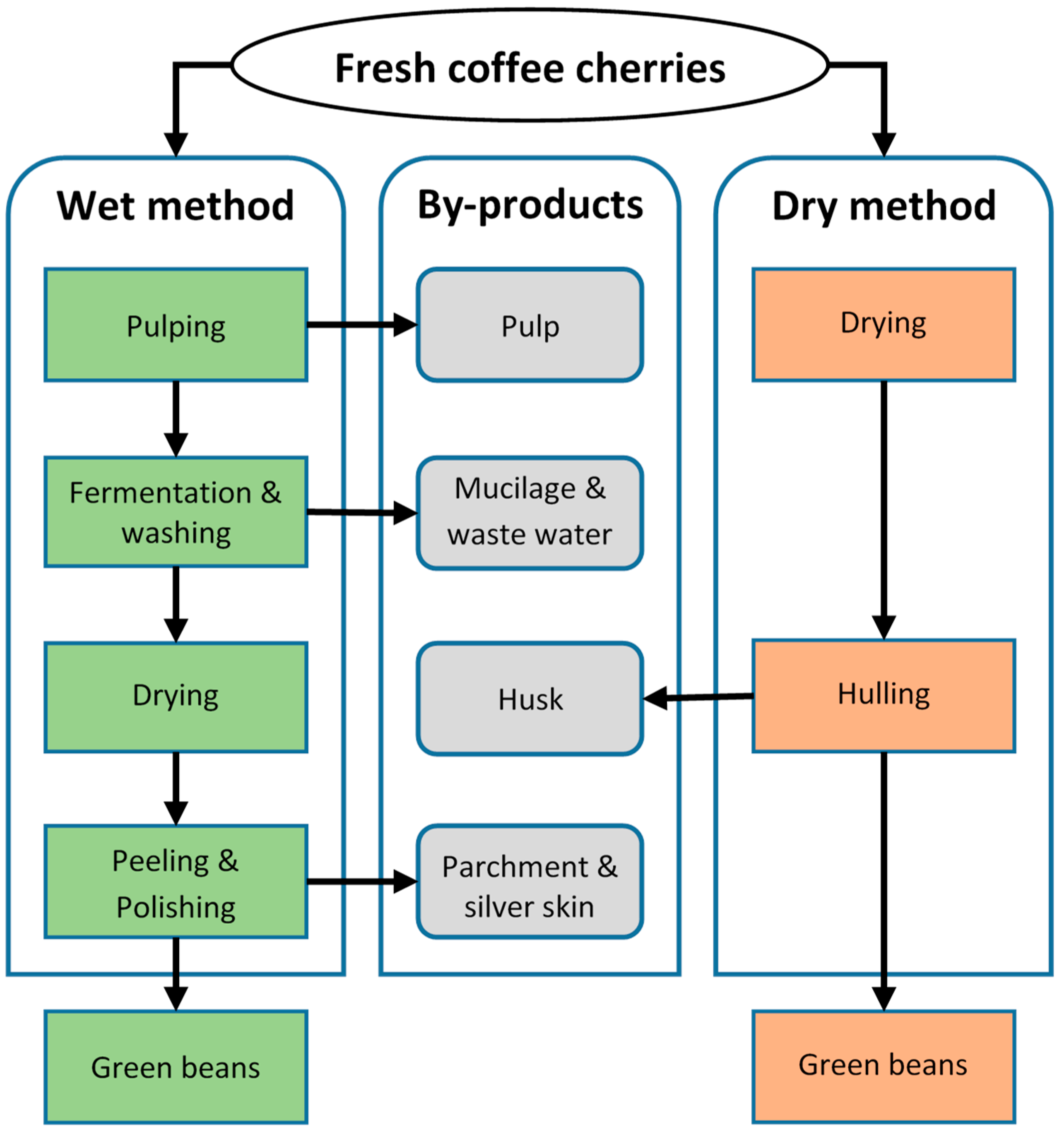
| Year | Area (ha) | Yield (kg/ha) | Green Beans Production (Mg) | Green Beans Export (Mg) | Export Value (×1000 USD) |
|---|---|---|---|---|---|
| 2007 | 407,147 | 671.5 | 273,400 | 158,467 | 417,323 |
| 2008 | 391,296 | 665.1 | 260,239 | 179,283 | 561,511 |
| 2009 | 395,003 | 672.1 | 265,469 | 129,833 | 365,689 |
| 2010 | 498,618 | 743.2 | 370,569 | 211,840 | 676,517 |
| 2011 | 515,882 | 730.4 | 376,823 | 159,135 | 844,555 |
| 2012 | 528,571 | 521.3 | 275,530 | 203,652 | 887,549 |
| 2013 | 538,466 | 728.0 | 392,006 | 218,937 | 803,965 |
| 2014 | 561,762 | 747.6 | 419,980 | 238,631 | 1,023,691 |
| 2015 | 653,910 | 698.9 | 457,014 | 234,218 | 1,018,149 |
| 2016 | 700,475 | 669.7 | 469,091 | 159,712 | 714,885 |
| Regional State | Wet Milling Stations | Dry Milling Stations | Grand Total | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Private | Cooperative | State | Total | Private | Cooperative | State | Total | ||
| Oromia | 297 | 95 | 15 | 407 | 524 | 20 | 6 | 550 | 957 |
| SNNP | 470 | 146 | 616 | 136 | 4 | -- | 140 | 756 | |
| Gambella | 3 | -- | -- | 3 | 6 | -- | -- | 6 | 9 |
| Total | 770 | 241 | 15 | 1026 | 666 | 24 | 6 | 696 | 1722 |
| Substrates | Methane Yield (L kg−1 VS) | Reference |
|---|---|---|
| Basic substrates: farm manures | ||
| Cattle | 130–300 | [24] |
| Pig | 210–320 | [24] |
| Poultry | 250–400 | [24] |
| Agricultural products | ||
| Straw | 71–240 | [24] |
| Maize silage | 320–400 | [24] |
| Grass | 286–324 | [25] |
| Sunflower | 235–347 | [25] |
| Agro industrial wastes | ||
| Potato pulp | 250–400 | [24] |
| Vegetable waste | 400 | [24] |
| Brewer grains | 370–390 | [24] |
| Municipal wastes | ||
| Bio-wastes | 200–600 | [24] |
| Rumen content (slaughterhouse waste) | 160–400 | [24] |
| Kitchen waste | 350–600 | [24] |
| Sewage sludge | 250–350 | [26] |
| Variant | Inoculum (mg) | Substrate (mg) | TS (% FM) | VS (% TS) |
|---|---|---|---|---|
| Blank | 50,000 | --- | 4.99 | 61.55 |
| Hay standard | 30,000 | 400 | 90.56 | 89.99 |
| Pulp | 30,000 | 400 | 91.62 | 88.26 |
| Husk | 30,000 | 400 | 92.65 | 93.09 |
| Parchment | 30,000 | 400 | 95.44 | 99.59 |
| Mucilage | 30,000 | 400 | 94.38 | 85.99 |
| Substrate | DM (% FM) | VS | XA | XP | XL | XF | NfE | NDF | ADF | ADL | NFC | GE (MJ kg−1) | C:NRatio | Sugars * | Organic Acids ** |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Husk | 93.5 ± 0.0 | 92.9 ± 0.1 | 7.2 ± 0.05 | 11.1 ± 0.0 | 1.5 ± 0.0 | 39.9 ± 0.1 | 40.4 ± 0.0 | 64.0 ± 1.1 | 49.5 ± 0.0 | 17.5 ± 1.6 | 16.2 ± 1.1 | 18.8 ± 0.0 | 24.9 | 4.45 | 1.22 |
| Pulp | 92.3 ± 0.1 | 88.3 ± 0.2 | 11.7 ± 0.2 | 14.1 ± 0.0 | 1.0 ± 0.1 | 24.8 ± 1.2 | 48.5 ± 0.0 | 55.6 ± 1.4 | 47.1 ± 0.1 | 15.5 ± 1.6 | 17.6 ± 1.4 | 17.4 ± 0.0 | 18.4 | 1.73 | 0.13 |
| Parchment | 96.0 ± 0.1 | 99.6 ± 0.1 | 0.45 ± 0.0 | 1.6 ± 0.0 | 0.9 ± 0.1 | 76.9 ± 0.3 | 20.1 ± 0.0 | 96.8 ± 0.3 | 76.9 ± 0.2 | 32.2 ± 0.0 | 0.3 ± 0.3 | 19.7 ± 0.1 | 190 | 0 | 0 |
| Mucilage | 94.8 ± 0.0 | 85.1 ± 0.1 | 14.9 ± 0.1 | 18.5 ± 0.0 | 0.1 ± 0.1 | 19.4 ± 0.4 | 47.1 ± 0.0 | 37.7 ± 1.2 | 36.9 ± 0.6 | 5.0 ± 0.3 | 28.8 ± 1.2 | 17.7 ± 0.1 | 14 | 0.46 | 5.19 |
| Mn | Zn | Co. | Mo | Fe | Ni | Se | W | ||
|---|---|---|---|---|---|---|---|---|---|
| Husk | 83 | 4.4 | 0.5 | 0.1 | 440 | 1.2 | 0.09 | <0.05 | |
| Pulp | 159 | 21.7 | 0.5 | 0.2 | 969 | 1.2 | 0.09 | <0.05 | |
| Mucilage | 149 | 125.5 | 1.0 | 0.3 | 1719 | 3.7 | 0.12 | <0.05 | |
| Parchment | 12 | 6.8 | 0.1 | <0.05 | 47 | 0.1 | <0.05 | <0.05 | |
| Optimum values | Min. | 100 | 30 | 0.4 | 1.0 | 1500 | 3.0 | 0.20 | 0.1 |
| Max. | 1500 | 300 | 5.0 | 6.0 | 3000 | 16.0 | 2.00 | 30.0 | |
| Substrate | SMY (L kg−1 VS) | Methane Content (%) | Degradability (%) | Methane Energy (MJ kg−1 VS) | Energy Recovery |
|---|---|---|---|---|---|
| Husk | 159.4 ± 1.8 | 51.5 | 35.3 | 6.33 | 33.7% |
| Pulp | 244.7 ± 6.4 | 56.8 | 63.0 | 9.75 | 56.1% |
| Parchment | 31.1 ± 2.0 | 84.2 | 3.4 | 1.12 | 5.7% |
| Mucilage | 294.5 ± 9.6 | 55.5 | 68.0 | 11.7 | 66.1% |
| Substrate | SMY (m3 Mg−1 VS) | Residues Production Ratio (RPR) kg VS kg−1 GB a | Biomass Yield (Mg VS year−1) | Methane Yield (m3 year−1) | CHP Production (MWh/year) | Diesel b Equivalent (m3) | Saved Fuel Cost c (USD) | |
|---|---|---|---|---|---|---|---|---|
| Electricity(MWhel year−1) | Heat(MWhTh year−1) | |||||||
| Husk | 159 | 0.923 | 289,748 | 46,069,906 | 160,729 | 183,690 | 46,070 | 27,181,244 |
| Pulp | 245 | 0.564 | 75,925 | 18,601,671 | 64,898 | 74,169 | 18,602 | 10,974,986 |
| Mucilage | 294 | 0.093 | 12,564 | 3,693,742 | 12,887 | 14,728 | 3,694 | 2,179,308 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chala, B.; Oechsner, H.; Latif, S.; Müller, J. Biogas Potential of Coffee Processing Waste in Ethiopia. Sustainability 2018, 10, 2678. https://doi.org/10.3390/su10082678
Chala B, Oechsner H, Latif S, Müller J. Biogas Potential of Coffee Processing Waste in Ethiopia. Sustainability. 2018; 10(8):2678. https://doi.org/10.3390/su10082678
Chicago/Turabian StyleChala, Bilhate, Hans Oechsner, Sajid Latif, and Joachim Müller. 2018. "Biogas Potential of Coffee Processing Waste in Ethiopia" Sustainability 10, no. 8: 2678. https://doi.org/10.3390/su10082678
APA StyleChala, B., Oechsner, H., Latif, S., & Müller, J. (2018). Biogas Potential of Coffee Processing Waste in Ethiopia. Sustainability, 10(8), 2678. https://doi.org/10.3390/su10082678







