A Sustainable Agricultural Future Relies on the Transition to Organic Agroecological Pest Management
Abstract
1. Achieving Needs for Agricultural Productivity and Pest Management Sustainably
2. Issues with Managing Agricultural Pests through Pesticides
2.1. Pesticides Impact Human Health
2.2. Pesticides Disrupt Ecosystems and Ecosystem Services for Agriculture
2.3. Pesticides Create Risk in Production Systems
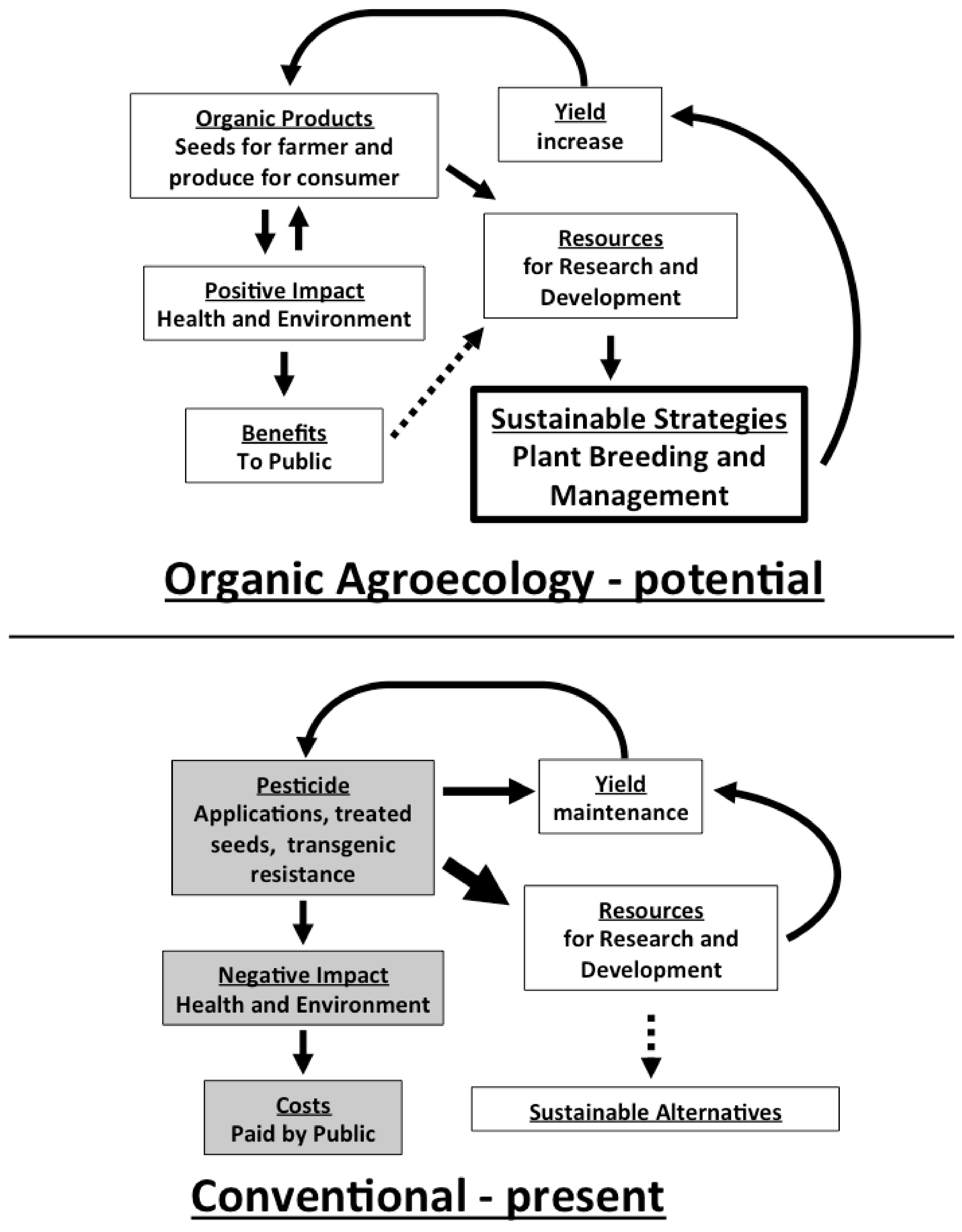
3. Investment in Organic Agroecological Research for Sustainable Pest Management Moves toward Eliminating the Conventional-Organic Yield Gap
3.1. Rhizosphere-Associated Microbiome
- Which soil microbes contribute to disease suppressive soils [167], and in what context are they effective in significant disease suppression on organic farms?
3.2. Trans-Generational Defense Priming
- What underlying conserved mechanisms are responsible for transgenerational defense priming?
- What are the biotic and abiotic triggers of plant defense priming, and how effective is the response to the broad spectrum of pests the progeny may encounter? Does this have ramifications for where and how we could produce organic seed?
- Are certain plant genotypes best suited for a response to transgenerational priming?
3.3. Plant Breeding for Indirect Resistance
- How can we identify unique volatiles that affect insect behavior (pests, and natural enemies) in a high-throughput manner? Of these volatiles, is there sufficient variation to select for enhanced phenotypes within cultivated plants?
- What procedures should be developed to ensure enhanced volatile phenotypes are effective at field scale for pest management while ensuring minimal disruption to other beneficial organisms of the plant (i.e., pollinators) [190]?
- How quickly will pest communities evolve to overcome disruptions in host finding via volatiles? How durable can we expect this pest management method to be?
3.4. Quantitative Resistance
- What is the best method for breeding for quantitative resistance in organic agroecological systems? How can we improve our ability to detect and select quantitative resistance traits in an agroecosystem with extensive biological diversity?
- Will there be tradeoffs between selecting for quantitative resistance, and other quantitative traits important to fruit and vegetable crops, including flavor and yield?
- Can we breed for any quantitative resistance traits that provide protection to multiple disease or insect pest pressure [196]?
3.5. Genetically Diverse Cultivars
- For cultivar mixtures, what is the most effective method to screen mixture combinations? Can we employ genomic tools to predict mixing ability to make the most rapid progress?
- For plant populations, how can we ensure that genetic diversity is maintained to respond to evolving pressures?
- How can participatory breeding methods be best employed to develop plant populations for organic growers?
- Can development of plant populations be incentivized in the private sector; what market changes would allow plant populations greater fit into the business model of seed companies? Are there resources for public plant breeders to meet this need?
3.6. Diverse Farmscapes
- How can we effectively identify functional groupings of botanical diversity for organic growers, given the contextual dependency of the field, farm, and landscape on the relative effect of adding botanical diversity to the farm?
- Are diverse organic agroecological farming operations scalable? How can we drive innovation in harvesting equipment and food distribution to allow growers to enhance the degree to which intercropping strategies, for example, are deployed on farm? While excellent local production models exist, can we develop a system to allow efficient coalescence into major markets, like cities?
- Can we develop strategies to augment botanical diversity on organic farms, without increasing the total area of land under cultivation?
3.7. Interactions between Modes of Defense
- There are innumerable combinations of modes of defense on organic farms. Can we leverage citizen science data or empirical grower knowledge to best identify the most promising areas of research for organic agroecological systems?
- How do other organic pest-related (i.e., adding biological control) and non-pest related (i.e., tillage) management practices impact these synergistic interactions?
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Alexandratos, N.; Bruinsma, J. World Agriculture towards 2030/2050: the 2012 Revision; FAO: Italy, Rome, 2012. [Google Scholar]
- Altieri, M.A. Agroecology, Small Farms, and Food Sovereignty. Mon. Rev. 2009, 61, 102–113. [Google Scholar] [CrossRef]
- National Research Council. Toward Sustainable Agricultural Systems in the 21st Century; The National Academies Press: Washington, DC, USA, 2010. [Google Scholar]
- Badgley, C.; Moghtader, J.; Quintero, E.; Zakem, E.; Chappell, M.J.; Avilés-Vázquez, K.; Samulon, A.; Perfecto, I. Organic agriculture and the global food supply. Renew. Agric. Food Syst. 2007, 22, 86–108. [Google Scholar] [CrossRef]
- De Ponti, T.; Rijk, B.; van Ittersum, M.K. The crop yield gap between organic and conventional agriculture. Agric. Syst. 2012, 108, 1–9. [Google Scholar] [CrossRef]
- Seufert, V.; Ramankutty, N.; Foley, J.A. Comparing the yields of organic and conventional agriculture. Nature 2012, 485, 229–232. [Google Scholar] [CrossRef] [PubMed]
- Reganold, J.P.; Wachter, J.M. Organic agriculture in the twenty-first century. Nat. Plants 2016, 2, 15221. [Google Scholar] [CrossRef] [PubMed]
- Muller, A.; Schader, C.; El-Hage Scialabba, N.; Brüggemann, J.; Isensee, A.; Erb, K.H.; Smith, P.; Klocke, P.; Leiber, F.; Stolze, M.; et al. Strategies for feeding the world more sustainably with organic agriculture. Nat. Commun. 2017, 8, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Seufert, V.; Ramankutty, N. Many shades of gray—The context-dependent performance of organic agriculture. Sci. Adv. 2017, 3, e1602638. [Google Scholar] [CrossRef] [PubMed]
- Willer, H.; Lernoud, J. Organic Agriculture Worldwide 2017: Current Statistics; Research Institute of Organic Agriculture: Frick, Switzerland, 2017. [Google Scholar]
- Tomich, T.P.; Brodt, S.; Ferris, H.; Galt, R.; Horwath, W.R.; Kebreab, E.; Leveau, J.H.J.; Liptzin, D.; Lubell, M.; Merel, P.; et al. Agroecology: A Review from a Global-Change Perspective. Annu. Rev. Environ. Resour. 2011, 36, 193–222. [Google Scholar] [CrossRef]
- Altieri, M.A.; Nicholls, C.I. Agroecology: Rescuing organic agriculture from a specialized Industrial model of production and distribution. In Policy Matters; IUCN Commission on Environmental, Economic & Social Policy: Gland, Switzerland, 2003; pp. 34–41. [Google Scholar]
- Migliorini, P.; Wezel, A. Converging and diverging principles and practices of organic agriculture regulations and agroecology. A review. Agron. Sustain. Dev. 2017, 37, 1–18. [Google Scholar] [CrossRef]
- Oerke, E.-C. Crop losses to pests. J. Agric. Sci. 2006, 144, 31–43. [Google Scholar] [CrossRef]
- Popp, J.; Peto, K.; Nagy, J. Pesticide productivity and food security. A review. Agron. Sustain. Dev. 2013, 33, 243–255. [Google Scholar] [CrossRef]
- Savary, S.; Ficke, A.; Aubertot, J.N.; Hollier, C. Crop losses due to diseases and their implications for global food production losses and food security. Food Secur. 2012, 4, 519–537. [Google Scholar] [CrossRef]
- Juroszek, P.; von Tiedemann, A. Plant pathogens, insect pests and weeds in a changing global climate: A review of approaches, challenges, research gaps, key studies and concepts. J. Agric. Sci. 2013, 151, 163–188. [Google Scholar] [CrossRef]
- Bebber, D.P.; Ramotowski, M.A.T.; Gurr, S.J. Crop pests and pathogens move polewards in a warming world. Nat. Clim. Chang. 2013, 3, 985–988. [Google Scholar] [CrossRef]
- Peshin, R.; Bandral, R.S.; Zhang, W.; Wilson, L.; Dhawan, A.K. Integrated Pest Managment: A Global Overview of History, Programs and Adoption. In Integrated Pest Management: Innovation-Development Process; Peshin, R., Dhawan, A.K., Eds.; Springer: Berlin, Germany, 2009; pp. 317–329. [Google Scholar]
- Fernandez-Cornejo, J.; Vialou, A. Pesticide Use in U. S. Agriculture: 21 Selected Crops, 1960–2008. USDA Econ. Inf. Bull. 2014, 80. [Google Scholar] [CrossRef]
- NASS. USDA NASS Quick Stats Lite Browser. Available online: https://www.nass.usda.gov/Quick_Stats/Lite/ (accessed on 27 May 2018).
- Pimentel, D. Environmental and economic costs of the application of pesticides primarily in the United States. Environ. Dev. Sustain. 2005, 7, 229–252. [Google Scholar] [CrossRef]
- Bourguet, D.; Guillemaud, T. The Hidden and External Costs of Pesticide Use. In Sustainable Agriculture Reviews; Lichtfouse, E., Ed.; Springer: Cham, Switzerland, 2016. [Google Scholar]
- Howard, P.H. Visualizing consolidation in the global seed industry: 1996-2008. Sustainability 2009, 1, 1266–1287. [Google Scholar] [CrossRef]
- Lammerts van Bueren, E.T.; Myers, J.R. Organic Crop Breeding: Integrating Organic Agricultural Approaches and Traditional and Modern Plant Breeding Methods. Org. Crop Breed. 2011, 1–13. [Google Scholar] [CrossRef]
- Döring, T.F.; Pautasso, M.; Wolfe, M.S.; Finckh, M.R. Pest and Disease Management in Organic Farming: Implications and Inspirations for Plant Breeding. Org. Crop Breed. 2012, 39–59. [Google Scholar] [CrossRef]
- Coleman, E. The New Organic Grower, 2nd ed.; Chelsea Green Publishing Company: White River Junction, VT, USA, 1995. [Google Scholar]
- Stenberg, J.A. A Conceptual Framework for Integrated Pest Management. Trends Plant Sci. 2017, 22, 759–769. [Google Scholar] [CrossRef] [PubMed]
- Hanley, M.E.; Lamont, B.B.; Fairbanks, M.M.; Rafferty, C.M. Plant structural traits and their role in anti-herbivore defence. Perspect. Plant Ecol. Evol. Syst. 2007, 8, 157–178. [Google Scholar] [CrossRef]
- Aragón, W.; Reina-Pinto, J.J.; Serrano, M.; Dominguez, E. The intimate talk between plants and microorganisms at the leaf surface. J. Exp. Bot. 2017, 68, 5339–5350. [Google Scholar] [CrossRef] [PubMed]
- Mohler, C.L. Weed life history: identifying vulnerabilities. In Ecological Management of Agricultural Weeds; Liebman, M., Mohler, C.L., Staver, C.P., Eds.; Cambridge University Press: Cambridge, UK, 2004; pp. 40–68. [Google Scholar]
- Ando, K.; Grumet, R.; Terpstra, K.; Kelly, J.D. Manipulation of plant architecture to enhance crop disease control. CAB Rev. Perspect. Agric. Vet. Sci. Nutr. Nat. Resour. 2007, 2, 1–8. [Google Scholar] [CrossRef]
- Kessler, A.; Baldwin, I.T. Defensive Function of Herbivore-Induced Plant Volatile Emissions in Nature. Science 2001, 291, 2141–2144. [Google Scholar] [CrossRef] [PubMed]
- Jones, J.D.G.; Dangl, J.L. The plant immune system. Nature 2006, 444, 323–329. [Google Scholar] [CrossRef] [PubMed]
- Howe, G.A.; Jander, G. Plant Immunity to Insect Herbivores. Annu. Rev. Plant Biol. 2008, 59, 41–66. [Google Scholar] [CrossRef] [PubMed]
- Mithöfer, A.; Boland, W. Plant Defense Against Herbivores: Chemical Aspects. Annu. Rev. Plant Biol. 2012, 63, 431–450. [Google Scholar] [CrossRef] [PubMed]
- Farooq, M.; Jabran, K.; Cheema, Z.A.; Wahid, A.; Siddique, K.H. The role of allelopathy in agricultural pest management. Pest Manag. Sci. 2011, 67, 493–506. [Google Scholar] [CrossRef] [PubMed]
- Jabran, K.; Mahajan, G.; Sardana, V.; Chauhan, B.S. Allelopathy for weed control in agricultural systems. Crop Prot. 2015, 72, 57–65. [Google Scholar] [CrossRef]
- Kessler, A.; Heil, M. The multiple faces of indirect defences and their agents of natural selection. Funct. Ecol. 2011, 25, 348–357. [Google Scholar] [CrossRef]
- Hilker, M.; Fatouros, N.E. Plant Responses to Insect Egg Deposition. Annu. Rev. Entomol. 2015, 60, 493–515. [Google Scholar] [CrossRef] [PubMed]
- Tamiru, A.; Khan, Z.R.; Bruce, T.J.A. New directions for improving crop resistance to insects by breeding for egg induced defence. Curr. Opin. Insect Sci. 2015, 9, 51–55. [Google Scholar] [CrossRef]
- Smith, C.M.; Clement, S.L. Molecular bases of plant resistance to arthropods. Annu. Rev. Entomol. 2012, 57, 309–328. [Google Scholar] [CrossRef] [PubMed]
- Poland, J.A.; Balint-Kurti, P.J.; Wisser, R.J.; Pratt, R.C.; Nelson, R.J. Shades of gray: the world of quantitative disease resistance. Trends Plant Sci. 2009, 14, 21–29. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, C.; Brennan, R.M.; Graham, J.; Karley, A.J. Plant Defense against Herbivorous Pests: Exploiting Resistance and Tolerance Traits for Sustainable Crop Protection. Front. Plant Sci. 2016, 7, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Koch, K.G.; Chapman, K.; Louis, J.; Heng-Moss, T.; Sarath, G. Plant Tolerance: A Unique Approach to Control Hemipteran Pests. Front. Plant Sci. 2016, 7, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Van Bruggen, A.H.C.; Finckh, M.R. Plant Diseases and Management Approaches in Organic Farming Systems. Annu. Rev. Phytopathol. 2016, 54, 25–54. [Google Scholar] [CrossRef] [PubMed]
- Metcalf, R.L.; Luckmann, W.H. Introduction to Insect Pest Management, 3rd ed.; John Wiley & Sons: Hoboken, NY, USA, 1994. [Google Scholar]
- Haramoto, E.R.; Gallandt, E.R. Brassica cover cropping for weed management: A review. Renew. Agric. Food Syst. 2004, 19, 187–198. [Google Scholar] [CrossRef]
- Zehnder, G.; Gurr, G.M.; Kühne, S.; Wade, M.R.; Wratten, S.D.; Wyss, E. Arthropod Pest Management in Organic Crops. Annu. Rev. Entomol. 2007, 52, 57–80. [Google Scholar] [CrossRef] [PubMed]
- Pickett, J.A.; Woodcock, C.M.; Midega, C.A.O.; Khan, Z.R. Push-pull farming systems. Curr. Opin. Biotechnol. 2014, 26, 125–132. [Google Scholar] [CrossRef] [PubMed]
- Miller, J.R.; Cowles, R.S. Stimulo-deterrent diversion: A concept and its possible application to onion maggot control. J. Chem. Ecol. 1990, 16, 3197–3212. [Google Scholar] [CrossRef] [PubMed]
- Harman, G.E.; Howell, C.R.; Viterbo, A.; Chet, I.; Lorito, M. Trichoderma species - opportunistic avirulent plant symbionts. Nat. Rev. Microbiol. 2004, 2, 43–56. [Google Scholar] [CrossRef] [PubMed]
- Wallenstein, M.D. Managing and manipulating the rhizosphere microbiome for plant health: A systems approach. Rhizosphere 2017, 3, 230–232. [Google Scholar] [CrossRef]
- Melander, B.; Liebman, M.; Davis, A.S.; Gallandt, E.R.; Barberi, P.; Moonen, A.-C.; Rasmussen, J.; van der Weide, R.; Vidotto, F. Non-Chemical Weed Management. In Weed Research: Expanding Horizons; Hatcher, P.E., Froud-Williams, R.J., Eds.; John Wiley & Sons: New York, NY, USA, 2017; pp. 245–270. [Google Scholar]
- Vincent, C.; Hallman, G.; Panneton, B.; Fleurat-Lessard, F. Management of Agricultural Insects with Physical Control Methods. Annu. Rev. Entomol. 2003, 48, 261–281. [Google Scholar] [CrossRef] [PubMed]
- FAO. Global Food Losses and Food Waste—Extent, Causes and Prevention; FAO: Rome, Italy, 2011. [Google Scholar]
- Pimentel, D.; Kirby, C.; Shroff, A. The Relationship between “Cosmetic Standards” for Foods and Pesticide Use. In The Pesticide Question; Pimentel, D., Lehman, H., Eds.; Springer: Boston, MA, USA, 1993; pp. 85–105. [Google Scholar]
- Hubbard, K.; Zystro, J. State of Organic Seed 2016. 2016. Available online: https://stateoforganicseed.org/ (accessed on 15 May 2018).
- Tieman, D.; Zhu, G.; Resende, M.F.R.; Lin, T.; Nguyen, C.; Bies, D.; Rambla, J.L.; Beltran, K.S.O.; Taylor, M.; Zhang, B.; et al. A chemical genetic roadmap to improved tomato flavor. Science 2017, 355, 391–394. [Google Scholar] [CrossRef] [PubMed]
- National Organic Program, 7 205 C.F.R §206; 2000. Available online: https://www.gpo.gov/fdsys/pkg/CFR-2011-title7-vol3/pdf/CFR-2011-title7-vol3-part205.pdf (accessed on 10 November 2017).
- IFOAM. The IFOAM Norms for Organic Production and Processing; IFOAM: Bonn, Germany, 2014. [Google Scholar]
- Bernardo, R. Genotype x Environment Interaction. In Breeding for Quantitative Traits in Plants; Stemma Press: Woodbury, MN, USA, 2010; pp. 177–202. [Google Scholar]
- Murphy, K.M.; Campbell, K.G.; Lyon, S.R.; Jones, S.S. Evidence of varietal adaptation to organic farming systems. Field Crops Res. 2007, 102, 172–177. [Google Scholar] [CrossRef]
- Kiers, E.T.; Hutton, M.G.; Denison, R.F. Human selection and the relaxation of legume defences against ineffective rhizobia. Proc. R. Soc. B Biol. Sci. 2007, 274, 3119–3126. [Google Scholar] [CrossRef] [PubMed]
- Legzdina, L.; Kokare, A.; Lammerts van Bueren, E. Genotype and environment interaction of various spring barley genotypes in organic and conventional growing conditions. In Plant Breeding for Organic and Sustainable, Low-input Agriculture: Dealing with Genotypeenvironment Interactions; Lammerts van Bueren, E., Goldringer, I., Scholten, O., Østergård, H., Eds.; Wageningen University: Wageningen, The Netherlands, 2007; p. 17. [Google Scholar]
- Lammerts Van Bueren, E.T.; Jones, S.S.; Tamm, L.; Murphy, K.M.; Myers, J.R.; Leifert, C.; Messmer, M.M. The need to breed crop varieties suitable for organic farming, using wheat, tomato and broccoli as examples: A review. NJAS—Wageningen J. Life Sci. 2011, 58, 193–205. [Google Scholar] [CrossRef]
- Roberts, J.R.; Reigart, J.R. Introduction. In Recognition and Management of Pesticide Poisonings; US EPA Office of Pesticide Programs: Washington, DC, USA, 2013; pp. 2–12. [Google Scholar]
- EPA. Regulatory Impact Analysis of Worker Protection Standard for Agricultural Pesticides; EPA: Washington, DC, USA, 1992. [Google Scholar]
- Alavanja, M.C.R.; Bonner, M.R. Occupational pesticide exposures and cancer risk: A review. J. Toxicol. Environ. Heal. Part B Crit. Rev. 2012, 15, 238–263. [Google Scholar] [CrossRef] [PubMed]
- Roberts, J.R.; Reigart, J.R. Chronic Effects. In Recognition and Management of Pesticide Poisonings; US EPA Office of Pesticide Programs: Washington, DC, USA, 2013; pp. 212–230. [Google Scholar]
- International Agency for Research on Cancer. IARC Monographs Volume 112: Evaluation of Five Organophosphate Insecticides and Herbicides; International Agency for Research on Cancer: Lyon, France, 2015; Volume 112. [Google Scholar]
- Kremen, C.; Iles, A.; Bacon, C. Diversified farming systems: An agroecological, systems-based alternative to modern industrial agriculture. Ecol. Soc. 2012, 17. [Google Scholar] [CrossRef]
- Butler-Dawson, J.; Galvin, K.; Thorne, P.S.; Rohlman, D.S. Organophosphorus pesticide exposure and neurobehavioral performance in Latino children living in an orchard community. Neurotoxicology 2016, 53, 165–172. [Google Scholar] [CrossRef] [PubMed]
- Roberts, J.R.; Karr, C.J. Pesticide Exposure in Children. Pediatrics 2012, 130, e1765–e1788. [Google Scholar] [CrossRef] [PubMed]
- EPA. About Pesticide Tolerances. Available online: https://www.epa.gov/pesticide-tolerances/about-pesticide-tolerances (accessed on 13 December 2017).
- Keikotlhaile, B.M.; Spanoghe, P.; Steurbaut, W. Effects of food processing on pesticide residues in fruits and vegetables: A meta-analysis approach. Food Chem. Toxicol. 2010, 48, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Liang, Y.; Liu, Y.; Ding, Y.; Liu, X.J. Meta-analysis of food processing on pesticide residues in fruits. Food Addit. Contam.—Part A Chem. Anal. Control. Expo. Risk Assess. 2014, 31, 1568–1573. [Google Scholar] [CrossRef] [PubMed]
- Barr, D.B.; Bravo, R.; Weerasekera, G.; Caltabiano, L.M.; Whitehead, R.D.; Olsson, A.; Caudill, S.P.; Schober, S.E.; Pirkle, J.L.; Sampson, E.J.; et al. Concentrations of dialkyl phosphate metabolites of organophosphorus pesticides in the U.S. population. Environ. Health Perspect. 2004, 112, 186–200. [Google Scholar] [CrossRef] [PubMed]
- Barr, D.B.; Allen, R.; Olsson, A.O.; Bravo, R.; Caltabiano, L.M.; Montesano, A.; Nguyen, J.; Udunka, S.; Walden, D.; Walker, R.D.; et al. Concentrations of selective metabolites of organophosphorus pesticides in the United States population. Environ. Res. 2005, 99, 314–326. [Google Scholar] [CrossRef] [PubMed]
- Barr, D.B.; Olsson, A.O.; Wong, L.Y.; Udunka, S.; Baker, S.E.; Whitehead, R.D.; Magsumbol, M.S.; Williams, B.L.; Needham, L.L. Urinary concentrations of metabolites of pyrethroid insecticides in the general u.s. population: National health and nutrition examination survey 1999–2002. Environ. Health Perspect. 2010, 118, 742–748. [Google Scholar] [CrossRef] [PubMed]
- Hughner, R.S.; McDonagh, P.; Prothero, A.; Shultz, C.J.; Stanton, J. Who are organic food consumers? A compilation and review of why people purchase organic food. J. Consum. Behav. 2007, 6, 94–110. [Google Scholar] [CrossRef]
- Baker, B.P.; Benbrook, C.M.; Groth, E.; Benbrook, K.L. Pesticide residues in conventional, integrated pest management (IPM)-grown and organic foods: insights from three US data sets. Food Addit. Contam. 2002, 19, 37–41. [Google Scholar] [CrossRef] [PubMed]
- Smith-Spangler, C.; Brandeau, M.L.; Hunter, G.E.; Bavinger, J.C.; Pearson, M.; Eschbach, P.J. Are Organic Foods Safer or Healthier Than Conventional Alternatives?: A Systematic Review. Ann. Intern. Med. 2012, 157, 348–366. [Google Scholar] [CrossRef] [PubMed]
- Curl, C.L.; Fenske, R.A.; Elgethun, K. Organophosphorus Pesticide Exposure of Urban and Suburban Preschool Children with Organic and Conventional Diets. Environ. Health Perspect. 2002, 111, 377–382. [Google Scholar] [CrossRef]
- Oates, L.; Cohen, M.; Braun, L.; Schembri, A.; Taskova, R. Reduction in urinary organophosphate pesticide metabolites in adults after a week-long organic diet. Environ. Res. 2014, 132, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Bradman, A.; Quirós-Alcalá, L.; Castorina, R.; Schall, R.A.; Camacho, J.; Holland, N.T.; Barr, D.B.; Eskenazi, B. Effect of organic diet intervention on pesticide exposures in young children living in low-income urban and agricultural communities. Environ. Health Perspect. 2015, 123, 1086–1093. [Google Scholar] [CrossRef] [PubMed]
- Costa, C.; Garcia-Leston, J.; Costa, S.; Coelho, P.; Silva, S.; Pingarilho, M.; Valdiglesias, V.; Mattei, F.; Dall’Armi, V.; Bonassi, S.; et al. Is organic farming safer to farmers’ health? A comparison between organic and traditional farming. Toxicol. Lett. 2014, 230, 166–176. [Google Scholar] [CrossRef] [PubMed]
- EPA. Biopesticides. Available online: https://www.epa.gov/pesticides/biopesticides (accessed on 18 December 2017).
- Benítez, T.; Rincón, A.M.; Limón, M.C.; Codón, A.C. Biocontrol mechanisms of Trichoderma strains. Int. Microbiol. 2004, 7, 249–260. [Google Scholar]
- EPA. Trichoderma Species Summary Document: Registration Review; EPA: Washington, DC, USA, 2007. [Google Scholar]
- Bengtsson, J.; Ahnström, J.; Weibull, A.C. The effects of organic agriculture on biodiversity and abundance: A meta-analysis. J. Appl. Ecol. 2005, 42, 261–269. [Google Scholar] [CrossRef]
- Crowder, D.W.; Northfield, T.D.; Strand, M.R.; Snyder, W.E. Organic agriculture promotes evenness and natural pest control. Nature 2010, 466, 109–112. [Google Scholar] [CrossRef] [PubMed]
- Goulson, D.; Nicholls, E.; Botias, C.; Rotheray, E.L. Bee declines driven by combined stress from parasites, pesticides, and lack of flowers. Science 2015, 347, 1255957. [Google Scholar] [CrossRef] [PubMed]
- Potts, S.G.; Biesmeijer, J.C.; Kremen, C.; Neumann, P.; Schweiger, O.; Kunin, W.E. Global pollinator declines: Trends, impacts and drivers. Trends Ecol. Evol. 2010, 25, 345–353. [Google Scholar] [CrossRef] [PubMed]
- Geiger, F.; Bengtsson, J.; Berendse, F.; Weisser, W.W.; Emmerson, M.; Morales, M.B.; Ceryngier, P.; Liira, J.; Tscharntke, T.; Winqvist, C.; et al. Persistent negative effects of pesticides on biodiversity and biological control potential on European farmland. Basic Appl. Ecol. 2010, 11, 97–105. [Google Scholar] [CrossRef]
- Birkhofer, K.; Arvidsson, F.; Ehlers, D.; Mader, V.L.; Bengtsson, J.; Smith, H.G. Organic farming affects the biological control of hemipteran pests and yields in spring barley independent of landscape complexity. Landsc. Ecol. 2016, 31, 567–579. [Google Scholar] [CrossRef]
- Krauss, J.; Gallenberger, I.; Steffan-Dewenter, I. Decreased functional diversity and biological pest control in conventional compared to organic crop fields. PLoS ONE 2011, 6, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Desneux, N.; Decourtye, A.; Delpuech, J.-M. The Sublethal Effects of Pesticides on Beneficial Arthropods. Annu. Rev. Entomol. 2007, 52, 81–106. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Tu, C.; Hu, S.; Gumpertz, M.; Ristaino, J.B. Effect of organic, sustainable, and conventional management strategies in grower fields on soil physical, chemical, and biological factors and the incidence of Southern blight. Appl. Soil Ecol. 2007, 37, 202–214. [Google Scholar] [CrossRef]
- Lupatini, M.; Korthals, G.W.; de Hollander, M.; Janssens, T.K.S.; Kuramae, E.E. Soil microbiome is more heterogeneous in organic than in conventional farming system. Front. Microbiol. 2017, 7, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Losey, J.E.; Vaughan, M. The economic value of ecological services provided by insects. Bioscience 2006, 56, 311–323. [Google Scholar] [CrossRef]
- Hallmann, C.A.; Sorg, M.; Jongejans, E.; Siepel, H.; Hofland, N.; Schwan, H.; Stenmans, W.; Müller, A.; Sumser, H.; Hörren, T.; et al. More than 75 percent decline over 27 years in total flying insect biomass in protected areas. PLoS ONE 2017, 12, e0185809. [Google Scholar] [CrossRef] [PubMed]
- Beketov, M.A.; Kefford, B.J.; Schafer, R.B.; Liess, M. Pesticides reduce regional biodiversity of stream invertebrates. Proc. Natl. Acad. Sci. USA 2013, 110, 11039–11043. [Google Scholar] [CrossRef] [PubMed]
- Mac Intyre Allen, J.K.; Scott-Dupree, C.D.; Tolman, J.H.; Harris, C.R. Evaluation of application methods for the chemical control of striped cucumber beetle (Coleoptera: Chrysomelidae) attacking seedling cucurbits. J. Veg. Crop Prod. 2001, 7, 83–95. [Google Scholar] [CrossRef]
- Stoner, K.A.; Eitzer, B.D. Movement of soil-applied imidacloprid and thiamethoxam into nectar and pollen of squash (Cucurbita pepo). PLoS ONE 2012, 7, e39114. [Google Scholar] [CrossRef] [PubMed]
- Dively, G.P.; Kamel, A. Insecticide residues in pollen and nectar of a cucurbit crop and their potential exposure to pollinators. J. Agric. Food Chem. 2012, 60, 4449–4456. [Google Scholar] [CrossRef] [PubMed]
- Fairbrother, A.; Purdy, J.; Anderson, T.; Fell, R. Risks of neonicotinoid insecticides to honeybees. Environ. Toxicol. Chem. 2014, 33, 719–731. [Google Scholar] [CrossRef] [PubMed]
- Henry, M.; Béguin, M.; Requier, F.; Rollin, O.; Odoux, J.; Aupinel, P.; Aptel, J.; Tchamitchian, S.; Decourtye, A. A Common Pesticide Decreases Foraging Success and Survival in Honey Bees. Science 2012, 336, 348–350. [Google Scholar] [CrossRef] [PubMed]
- Krupke, C.H.; Hunt, G.J.; Eitzer, B.D.; Andino, G.; Given, K. Multiple routes of pesticide exposure for honey bees living near agricultural fields. PLoS ONE 2012, 7, e29268. [Google Scholar] [CrossRef] [PubMed]
- Gill, R.J.; Ramos-Rodriguez, O.; Raine, N.E. Combined pesticide exposure severely affects individual- and colony-level traits in bees. Nature 2012, 491, 105–108. [Google Scholar] [CrossRef] [PubMed]
- Whitehorn, P.R.; O’Connor, S.; Wackers, F.L.; Goulson, D. Neonicotinoid pesticide reduces bumble bee colony growth and queen production. Science 2012, 336, 351–352. [Google Scholar] [CrossRef] [PubMed]
- Laycock, I.; Lenthall, K.M.; Barratt, A.T.; Cresswell, J.E. Effects of imidacloprid, a neonicotinoid pesticide, on reproduction in worker bumble bees (Bombus terrestris). Ecotoxicology 2012, 21, 1937–1945. [Google Scholar] [CrossRef] [PubMed]
- Synder, W.E. Managing Cucumber Beetles in Organic Farming Systems. Available online: http://www.extension.org/pages/64274/managing-cucumber-beetles-in-organic-farming-systems (accessed on 3 March 2015).
- Bach, C.E. Effects of Plant Density and Diversity on the Population Dynamics of a Specialist Herbivore, the Striped Cucumber Beetle, Acalymma Vittata (Fab). Ecology 1980, 61, 1515–1530. [Google Scholar] [CrossRef]
- Adler, L.S.; Hazzard, R.V. Comparison of perimeter trap crop varieties: Effects on herbivory, pollination, and yield in butternut squash. Environ. Entomol. 2009, 38, 207–215. [Google Scholar] [CrossRef] [PubMed]
- Cavanagh, A.; Hazzard, R.; Adler, L.S.; Boucher, J. Using trap crops for control of Acalymma vittatum (Coleoptera: Chrysomelidae) reduces insecticide use in butternut squash. J. Econ. Entomol. 2009, 102, 1101–1107. [Google Scholar] [CrossRef] [PubMed]
- Cavanagh, A.F.; Adler, L.S.; Hazzard, R.V. Buttercup squash provides a marketable alternative to blue hubbard as a trap crop for control of striped cucumber beetles (Coleoptera: Chrysomelidae). Environ. Entomol. 2010, 39, 1953–1960. [Google Scholar] [CrossRef] [PubMed]
- Gardner, J.; Hoffmann, M.P.; Mazourek, M. Striped cucumber beetle (Coleoptera: Chrysomelidae) aggregation in response to cultivar and flowering. Environ. Entomol. 2015, 44, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Brzozowski, L.; Leckie, B.M.; Gardner, J.; Hoffmann, M.P.; Mazourek, M. Cucurbita pepo subspecies delineates striped cucumber beetle (Acalymma vittatum) preference. Hortic. Res. 2016, 3, 16028. [Google Scholar] [CrossRef] [PubMed]
- Toepfer, S.; Cabrera Walsh, G.; Eben, A.; Alvarez-Zagoya, R.; Haye, T.; Zhang, F.; Kuhlmann, U. A critical evaluation of host ranges of parasitoids of the subtribe Diabroticina (Coleoptera: Chrysomelidae: Galerucinae: Luperini) using field and laboratory host records. Biocontrol Sci. Technol. 2008, 18, 485–508. [Google Scholar] [CrossRef]
- Toepfer, S.; Haye, T.; Erlandson, M.; Goettel, M.; Lundgren, J.G.; Kleespies, R.G.; Weber, D.C.; Walsh, G.C.; Peters, A.; Ehlers, R.-U.; et al. A review of the natural enemies of beetles in the subtribe Diabroticina (Coleoptera: Chrysomelidae): implications for sustainable pest management. Biocontrol Sci. Technol. 2009, 19, 1–65. [Google Scholar] [CrossRef]
- Smyth, R.R.; Hoffmann, M.P. Seasonal incidence of two co-occurring adult parasitoids of Acalymma vittatum in New York State: Centistes (Syrrhizus) diabroticae and Celatoria setosa. BioControl 2010, 55, 219–228. [Google Scholar] [CrossRef]
- Pimentel, D.; Acquay, H.; Biltonen, M.; Rice, P.; Silva, M.; Nelson, J.; Lipner, V.; Giordano, S.; Horowitz, A.; Amore, M.D. Environmental and Economic Costs of Pesticide Use. Bioscience 1992, 42, 750–760. [Google Scholar] [CrossRef]
- Ponisio, L.C.; Kremen, C. System-level approach needed to evaluate the transition to more sustainable agriculture. Proc. R. Soc. B 2016, 283. [Google Scholar] [CrossRef] [PubMed]
- Van den Bosch, R. The Pesticide Conspiracy; University of California Press: Berkeley, CA, USA, 1978. [Google Scholar]
- Sparks, T.C.; Nauen, R. IRAC: Mode of action classification and insecticide resistance management. Pestic. Biochem. Physiol. 2015, 121, 122–128. [Google Scholar] [CrossRef] [PubMed]
- Torriani, S.F.F.; Brunner, P.C.; McDonald, B.A.; Sierotzki, H. QoI resistance emerged independently at least 4 times in European populations of Mycosphaerella graminicola. Pest Manag. Sci. 2009, 65, 155–162. [Google Scholar] [CrossRef] [PubMed]
- Hahn, M. The rising threat of fungicide resistance in plant pathogenic fungi: Botrytis as a case study. J. Chem. Biol. 2014, 7, 133–141. [Google Scholar] [CrossRef] [PubMed]
- Gassmann, A.J.; Petzold-Maxwell, J.L.; Keweshan, R.S.; Dunbar, M.W. Field-evolved resistance to Bt maize by Western corn rootworm. PLoS ONE 2011, 6. [Google Scholar] [CrossRef] [PubMed]
- Gassmann, A.J.; Petzold-Maxwell, J.L.; Clifton, E.H.; Dunbar, M.W.; Hoffmann, A.M.; Ingber, D.A.; Keweshan, R.S. Field-evolved resistance by western corn rootworm to multiple Bacillus thuringiensis toxins in transgenic maize. Proc. Natl. Acad. Sci. USA 2014, 111, 5141–5146. [Google Scholar] [CrossRef] [PubMed]
- Tabashnik, B.E.; Carrière, Y. Surge in insect resistance to transgenic crops and prospects for sustainability. Nat. Biotechnol. 2017, 35, 926–935. [Google Scholar] [CrossRef] [PubMed]
- ISAAA. Global Status of Commercialized Biotech/GM Crops: 2016. In ISAA Briefs; ISAAA: Ithaca, NY, USA, 2016; ISBN 978-1-892456-66-4. [Google Scholar]
- Savory, E.A.; Granke, L.L.; Quesada-ocampo, L.M.; Varbanova, M.; Hausbeck, M.K.; Day, B. Pathogen profile: The cucurbit downy mildew pathogen Pseudoperonospora cubensis. Mol. Plant Pathol. 2011, 12, 217–226. [Google Scholar] [CrossRef] [PubMed]
- Holmes, G.J.; Ojiambo, P.S.; Hausbeck, M.K.; Quesada-Ocampo, L.; Keinath, A.P. Resurgence of Cucurbit Downy Mildew in the United States: A Watershed Event for Research and Extension. Plant Dis. 2015, 99, 428–441. [Google Scholar] [CrossRef]
- Cohen, Y.; Van den Langenberg, K.M.; Wehner, T.C.; Ojiambo, P.S.; Hausbeck, M.; Quesada-Ocampo, L.M.; Lebeda, A.; Sierotzki, H.; Gisi, U. Resurgence of Pseudoperonospora cubensis: The Causal Agent of Cucurbit Downy Mildew. Phytopathology 2015, 105, 998–1012. [Google Scholar] [CrossRef] [PubMed]
- Urban, J.; Lebeda, A. Fungicide resistance in cucurbit downy mildew - Methodological, biological and population aspects. Ann. Appl. Biol. 2006, 149, 63–75. [Google Scholar] [CrossRef]
- Hausbeck, M.K. Downy Mildew Watch: Fungicides Recommended for Cucumber Disease Control. Available online: http://msue.anr.msu.edu/news/downy_mildew_watch_fungicides_recommended_for_cucumber_disease_control (accessed on 28 December 2017).
- Holdsworth, W.L.; Summers, C.F.; Glos, M.; Smart, C.D.; Mazourek, M. Development of downy mildew-resistant cucumbers for late-season production in the northeastern United States. HortScience 2014, 49, 10–17. [Google Scholar]
- Brzozowski, L.; Holdsworth, W.L.; Mazourek, M. “DMR-NY401”: A New Downy Mildew-resistant Slicing Cucumber. HortScience 2016, 51, 1294–1296. [Google Scholar] [CrossRef]
- Coleman, E. Biological Diplomacy. In The New Organic Grower; Chelsea Green Publishing Company: White River Junction, VT, USA, 1995; pp. 181–183. [Google Scholar]
- Heffner, E.L.; Sorrells, M.E.; Jannink, J.L. Genomic selection for crop improvement. Crop Sci. 2009, 49, 1–12. [Google Scholar] [CrossRef]
- Fahlgren, N.; Gehan, M.A.; Baxter, I. Lights, camera, action: High-throughput plant phenotyping is ready for a close-up. Curr. Opin. Plant Biol. 2015, 24, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Riesenfeld, C.S.; Schloss, P.D.; Handelsman, J. Metagenomics: Genomic Analysis of Microbial Communities. Annu. Rev. Genet. 2004, 38, 525–552. [Google Scholar] [CrossRef] [PubMed]
- Raguso, R.A.; Agrawal, A.A.; Douglas, A.E.; Jander, G.; Kessler, A.; Poveda, K.; Thaler, J.S. The raison d’etre of chemical ecology. Ecology 2015, 96, 617–630. [Google Scholar] [CrossRef] [PubMed]
- Doran, J.W.; Parkin, T.B. Defining and assessing soil quality. In Defining Soil Quality for a Sustainable Environment; SSSA Special Publication; Soil Science Society of America and American Society of Agronomy: Madison, WI, USA, 1994; Volume 35, pp. 1–21. [Google Scholar]
- Gugino, B.K.; Abawi, G.S.; Idowu, O.J.; Schindelbeck, R.R.; Smith, L.L.; Thies, J.E.; Wolfe, D.W.; van Es, H.M. Cornell Soil Health Assessment Training Manual; Cornell University: Ithaca, NY, USA, 2009. [Google Scholar]
- Ryan, M.R.; Peigné, J. Applying Agroecological Principles for Regenerating Soils. In Agroecological Practices For Sustainable Agriculture: Principles, Applications, And Making The Transition; Alexander, W., Ed.; World Scientific Publishing Company: Singapore, 2017; pp. 53–84. [Google Scholar]
- Sugiyama, A.; Vivanco, J.M.; Jayanty, S.S.; Manter, D.K. Pyrosequencing Assessment of Soil Microbial Communities in Organic and Conventional Potato Farms. Plant Dis. 2010, 94, 1329–1335. [Google Scholar] [CrossRef]
- Berendsen, R.L.; Pieterse, C.M.J.; Bakker, P.A.H. M. The rhizosphere microbiome and plant health. Trends Plant Sci. 2012, 1, 478–486. [Google Scholar] [CrossRef] [PubMed]
- Chaparro, J.M.; Sheflin, A.M.; Manter, D.K.; Vivanco, J.M. Manipulating the soil microbiome to increase soil health and plant fertility. Biol. Fertil. Soils 2012, 48, 489–499. [Google Scholar] [CrossRef]
- Berg, G.; Grube, M.; Schloter, M.; Smalla, K. Unraveling the plant microbiome: Looking back and future perspectives. Front. Microbiol. 2014, 5, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Lakshmanan, V.; Selvaraj, G.; Bais, H.P. Functional Soil Microbiome: Belowground Solutions to an Aboveground Problem. Plant Physiol. 2014, 166, 689–700. [Google Scholar] [CrossRef] [PubMed]
- Müller, D.B.; Vogel, C.; Bai, Y.; Vorholt, J.A. The Plant Microbiota: Systems-Level Insights and Perspectives. Annu. Rev. Genet. 2016, 50, 211–234. [Google Scholar] [CrossRef] [PubMed]
- Bezemer, T.M.; Van Dam, N.M. Linking aboveground and belowground interactions via induced plant defenses. Trends Ecol. Evol. 2005, 20, 617–624. [Google Scholar] [CrossRef] [PubMed]
- Zamioudis, C.; Pieterse, C.M.J. Modulation of Host Immunity by Beneficial Microbes. Mol. Plant-Microbe Interact. 2012, 25, 139–150. [Google Scholar] [CrossRef] [PubMed]
- Pieterse, C.M.J.; Zamioudis, C.; Berendsen, R.L.; Weller, D.M.; Van Wees, S.C.M.; Bakker, P.A.H.M. Induced Systemic Resistance by Beneficial Microbes. Annu. Rev. Phytopathol. 2014, 52, 347–375. [Google Scholar] [CrossRef] [PubMed]
- Hacquard, S.; Spaepen, S.; Garrido-Oter, R.; Schulze-Lefert, P. Interplay Between Innate Immunity and the Plant Microbiota. Annu. Rev. Phytopathol. 2017, 55, 565–589. [Google Scholar] [CrossRef] [PubMed]
- Mendes, R.; Kruijt, M.; de Bruijn, I.; Dekkers, E.; van der Voort, M.; Schneider, J.H.M.; Piceno, Y.M.; DeSantis, T.Z.; Andersen, G.L.; Bakker, P.A.H. M.; et al. Deciphering the Rhizosphere Microbiome for Disease-Suppressive Bacteria. Science 2011, 332, 1097–1100. [Google Scholar] [CrossRef] [PubMed]
- Cha, J.Y.; Han, S.; Hong, H.J.; Cho, H.; Kim, D.; Kwon, Y.; Kwon, S.K.; Crusemann, M.; Bok Lee, Y.; Kim, J.F.; et al. Microbial and biochemical basis of a Fusarium wilt-suppressive soil. ISME J. 2016, 10, 119–129. [Google Scholar] [CrossRef] [PubMed]
- Bakker, M.G.; Manter, D.K.; Sheflin, A.M.; Weir, T.L.; Vivanco, J.M. Harnessing the rhizosphere microbiome through plant breeding and agricultural management. Plant Soil 2012, 360, 1–13. [Google Scholar] [CrossRef]
- Philippot, L.; Raaijmakers, J.M.; Lemanceau, P.; Van Der Putten, W.H. Going back to the roots: The microbial ecology of the rhizosphere. Nat. Rev. Microbiol. 2013, 11, 789–799. [Google Scholar] [CrossRef] [PubMed]
- Oburger, E.; Schmidt, H. New Methods To Unravel Rhizosphere Processes. Trends Plant Sci. 2016, 21, 243–255. [Google Scholar] [CrossRef] [PubMed]
- Poole, P. Shining a light on the dark world of plant root–microbe interactions. Proc. Natl. Acad. Sci. USA 2017, 114, 4281–4283. [Google Scholar] [CrossRef] [PubMed]
- Van Dam, N.M.; Bouwmeester, H.J. Metabolomics in the Rhizosphere: Tapping into Belowground Chemical Communication. Trends Plant Sci. 2016, 21, 256–265. [Google Scholar] [CrossRef] [PubMed]
- Schenk, P.M.; Carvalhais, L.C.; Kazan, K. Unraveling plant-microbe interactions: Can multi-species transcriptomics help? Trends Biotechnol. 2012, 30, 177–184. [Google Scholar] [CrossRef] [PubMed]
- Guttman, D.S.; McHardy, A.C.; Schulze-Lefert, P. Microbial genome-enabled insights into plant-microorganism interactions. Nat. Rev. Genet. 2014, 15, 797–813. [Google Scholar] [CrossRef] [PubMed]
- Finkel, O.M.; Castrillo, G.; Herrera Paredes, S.; Salas González, I.; Dangl, J.L. Understanding and exploiting plant beneficial microbes. Curr. Opin. Plant Biol. 2017, 38, 155–163. [Google Scholar] [CrossRef] [PubMed]
- Haney, C.H.; Samuel, B.S.; Bush, J.; Ausubel, F.M. Associations with rhizosphere bacteria can confer an adaptive advantage to plants. Nat. Plants 2015, 1, 15051. [Google Scholar] [CrossRef] [PubMed]
- Pieterse, C.M.J.; de Jonge, R.; Berendsen, R.L. The Soil-Borne Supremacy. Trends Plant Sci. 2016, 21, 171–173. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.Y.; Zeng, R. Sen; Xu, J.F.; Li, J.; Shen, X.; Yihdego, W.G. Interplant communication of tomato plants through underground common mycorrhizal networks. PLoS ONE 2010, 5. [Google Scholar] [CrossRef] [PubMed]
- Babikova, Z.; Gilbert, L.; Bruce, T.J.A.; Birkett, M.; Caulfield, J.C.; Woodcock, C.; Pickett, J.A.; Johnson, D. Underground signals carried through common mycelial networks warn neighbouring plants of aphid attack. Ecol. Lett. 2013, 16, 835–843. [Google Scholar] [CrossRef] [PubMed]
- Conrath, U.; Beckers, G.J.M.; Langenbach, C.J.G.; Jaskiewicz, M.R. Priming for Enhanced Defense. Annu. Rev. Phytopathol. 2015, 53, 97–119. [Google Scholar] [CrossRef] [PubMed]
- Holeski, L.M.; Jander, G.; Agrawal, A.A. Transgenerational defense induction and epigenetic inheritance in plants. Trends Ecol. Evol. 2012, 27, 618–626. [Google Scholar] [CrossRef] [PubMed]
- Latzel, V.; Zhang, Y.; Karlsson Moritz, K.; Fischer, M.; Bossdorf, O. Epigenetic variation in plant responses to defence hormones. Ann. Bot. 2012, 110, 1423–1428. [Google Scholar] [CrossRef] [PubMed]
- Ito, H. Plant Models of Transgenerational Epigenetic Inheritance. In Transgenerational Epigenetics; Elsevier: New York, NY, USA, 2014; pp. 147–161. [Google Scholar]
- Bilichak, A.; Ilnytskyy, Y.; Woycicki, R.; Kepeshchuk, N.; Fogen, D.; Kovalchuk, I. The elucidation of stress memory inheritance in Brassica rapa plants. Front. Plant Sci. 2015, 6, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, A.A.; Laforsch, C.; Tollrian, R. Transgenerational induction of defences in animals and plants. Nature 1999, 401, 60–63. [Google Scholar] [CrossRef]
- Holeski, L.M. Within and between generation phenotypic plasticity in trichome density of Mimulus guttatus. J. Evol. Biol. 2007, 20, 2092–2100. [Google Scholar] [CrossRef] [PubMed]
- Holeski, L.M.; Zinkgraf, M.S.; Couture, J.J.; Whitham, T.G.; Lindroth, R.L. Transgenerational effects of herbivory in a group of long-lived tree species: Maternal damage reduces offspring allocation to resistance traits, but not growth. J. Ecol. 2013, 101, 1062–1073. [Google Scholar] [CrossRef]
- Terhorst, C.P.; Lau, J.A. Direct and indirect transgenerational effects alter plant-herbivore interactions. Evol. Ecol. 2012, 26, 1469–1480. [Google Scholar] [CrossRef]
- Colicchio, J. Transgenerational effects alter plant defence and resistance in nature. J. Evol. Biol. 2017, 30, 664–680. [Google Scholar] [CrossRef] [PubMed]
- Rasmann, S.; De Vos, M.; Casteel, C.L.; Tian, D.; Halitschke, R.; Sun, J.Y.; Agrawal, A.A.; Felton, G.W.; Jander, G. Herbivory in the Previous Generation Primes Plants for Enhanced Insect Resistance. Plant Physiol. 2012, 158, 854–863. [Google Scholar] [CrossRef] [PubMed]
- Vivas, M.; Zas, R.; Sampedro, L.; Solla, A. Environmental Maternal Effects Mediate the Resistance of Maritime Pine to Biotic Stress. PLoS One 2013, 8. [Google Scholar] [CrossRef] [PubMed]
- Poveda, K.; Gomez Jiminez, M.I.; Kessler, A. The enemy as ally: herbivore-induced increase in crop yield. Ecol. Appl. 2010, 20, 1787–1793. [Google Scholar] [CrossRef] [PubMed]
- Tamiru, A.; Bruce, T.J.A.; Woodcock, C.M.; Caulfield, J.C.; Midega, C.A.O.; Ogol, C.K.P. O.; Mayon, P.; Birkett, M.A.; Pickett, J.A.; Khan, Z.R. Maize landraces recruit egg and larval parasitoids in response to egg deposition by a herbivore. Ecol. Lett. 2011, 14, 1075–1083. [Google Scholar] [CrossRef] [PubMed]
- Mutyambai, D.M.; Bruce, T.J.A.; Midega, C.A.O.; Woodcock, C.M.; Caulfield, J.C.; Van Den Berg, J.; Pickett, J.A.; Khan, Z.R. Responses of Parasitoids to Volatiles Induced by Chilo partellus Oviposition on Teosinte, a Wild Ancestor of Maize. J. Chem. Ecol. 2015, 41, 323–329. [Google Scholar] [CrossRef] [PubMed]
- De Lange, E.S.; Balmer, D.; Mauch-Mani, B.; Turlings, T.C.J. Insect and pathogen attack and resistance in maize and its wild ancestors, the teosintes. New Phytol. 2014, 204, 329–341. [Google Scholar] [CrossRef]
- Chen, Y.H.; Gols, R.; Benrey, B. Crop Domestication and Its Impact on Naturally Selected Trophic Interactions. Annu. Rev. Entomol. 2015, 60, 35–58. [Google Scholar] [CrossRef] [PubMed]
- Reddy, G.V.P.; Guerrero, A. Interactions of insect pheromones and plant semiochemicals. Trends Plant Sci. 2004, 9, 253–261. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, I.; Lewis, D. What happens when crops are turned on? Simulating constitutive volatiles for tritrophic pest suppression across an agricultural landscape. Pest Manag. Sci. 2015, 71, 139–150. [Google Scholar] [CrossRef] [PubMed]
- Niks, R.E.; Qi, X.; Marcel, T.C. Quantitative Resistance to Biotrophic Filamentous Plant Pathogens: Concepts, Misconceptions, and Mechanisms. Annu. Rev. Phytopathol. 2015, 53, 445–470. [Google Scholar] [CrossRef] [PubMed]
- Palloix, A.; Ayme, V.; Moury, B. Durability of plant major resistance genes to pathogens depends on the genetic background, experimental evidence and consequences for breeding strategies. New Phytol. 2009, 183, 190–199. [Google Scholar] [CrossRef] [PubMed]
- Nelson, R.; Wiesner-Hanks, T.; Wisser, R.; Balint-Kurti, P. Navigating complexity to breed disease-resistant crops. Nat. Rev. Genet. 2017, 19, 21–33. [Google Scholar] [CrossRef] [PubMed]
- Kloth, K.J.; Thoen, M.P.M.; Bouwmeester, H.J.; Jongsma, M.A.; Dicke, M. Association mapping of plant resistance to insects. Trends Plant Sci. 2012, 17, 311–319. [Google Scholar] [CrossRef] [PubMed]
- McDonald, B.A.; Linde, C. Pathogen population genetics, evolutionary potential and durable resistance. Annu. Rev. Phytopathol. 2002, 40, 349–379. [Google Scholar] [CrossRef] [PubMed]
- Wiesner-Hanks, T.; Nelson, R. Multiple Disease Resistance in Plants. Annu. Rev. Phytopathol. 2016, 54, 229–252. [Google Scholar] [CrossRef] [PubMed]
- Tooker, J.F.; Frank, S.D. Genotypically diverse cultivar mixtures for insect pest management and increased crop yields. J. Appl. Ecol. 2012, 49, 974–985. [Google Scholar] [CrossRef]
- Zhu, Y.; Chen, H.; Fan, J.; Wang, Y.; Li, Y.; Chen, J.; Fan, J.; Yang, S.; Hu, L.; Leung, H.; et al. Genetic diversity and disease control in rice. Nature 2000, 406, 718–722. [Google Scholar] [CrossRef] [PubMed]
- Chateil, C.; Goldringer, I.; Tarallo, L.; Kerbiriou, C.; Le Viol, I.; Ponge, J.F.; Salmon, S.; Gachet, S.; Porcher, E. Crop genetic diversity benefits farmland biodiversity in cultivated fields. Agric. Ecosyst. Environ. 2013, 171, 25–32. [Google Scholar] [CrossRef]
- Pan, P.; Qin, Y. Genotypic diversity of soybean in mixed cropping can affect the populations of insect pests and their natural enemies. Int. J. Pest Manag. 2014, 60, 287–292. [Google Scholar] [CrossRef]
- Ceccarelli, S. GM crops, organic agriculture and breeding for sustainability. Sustainability 2014, 6, 4273–4286. [Google Scholar] [CrossRef]
- DeLucia, E.H.; Nabity, P.D.; Zavala, J.A.; Berenbaum, M.R. Climate Change: Resetting Plant-Insect Interactions. Plant Physiol. 2012, 160, 1677–1685. [Google Scholar] [CrossRef] [PubMed]
- Brooker, R.W.; Jones, H.G.; Paterson, E.; Watson, C.; Brooker, R.W.; Bennett, A.E.; Cong, W.; Daniell, T.J.; George, T.S.; Hallett, P.D.; et al. Improving intercropping: a synthesis of research in agronomy, plant physiology and ecology. New Phytol. 2015, 206, 107–117. [Google Scholar] [CrossRef] [PubMed]
- Barot, S.; Allard, V.; Cantarel, A.; Enjalbert, J.; Gauffreteau, A.; Goldringer, I.; Lata, J.C.; Le Roux, X.; Niboyet, A.; Porcher, E. Designing mixtures of varieties for multifunctional agriculture with the help of ecology. A review. Agron. Sustain. Dev. 2017, 37. [Google Scholar] [CrossRef]
- Dawson, J.C.; Goldringer, I. Breeding for Genetically Diverse Populations: Variety Mixtures and Evolutionary Populations. Org. Crop Breed. 2011, 77–98. [Google Scholar] [CrossRef]
- Landis, D.A.; Wratten, S.D.; Gurr, G.M. Habitat Management to Conserve Natural Enemies of Arthropod Pests in Agriculture. Annu. Rev. Entomol. 2000, 45, 175–201. [Google Scholar] [CrossRef] [PubMed]
- Ren, W.; Hu, L.; Zhang, J.; Sun, C.; Tang, J.; Yuan, Y.; Chen, X. Can positive interactions between cultivated species help to sustain modern agriculture? Front. Ecol. Environ. 2014, 12, 507–514. [Google Scholar] [CrossRef]
- Brooker, R.W.; Karley, A.J.; Newton, A.C.; Pakeman, R.J.; Schöb, C. Facilitation and sustainable agriculture: A mechanistic approach to reconciling crop production and conservation. Funct. Ecol. 2016, 30, 98–107. [Google Scholar] [CrossRef]
- Ratnadass, A.; Fernandes, P.; Avelino, J.; Habib, R. Plant Species Diversity for Sustainable Management of Crop Pests and Diseases in Agroecosystems: A review. Agron. Sustain. Dev. 2012, 32, 273–303. [Google Scholar] [CrossRef]
- Letourneau, D.K.; Armbrecht, I.; Rivera, B.S.; Lerma, J.; Carmona, E.J.; Daza, M.C.; Escobar, S.; Galindo, V.; GutiéRrez, C.; LóPez, S.D.; et al. Does plant diversity benefit agroecosystems? A synthetic review. Ecol. Appl. 2011, 21, 9–21. [Google Scholar] [CrossRef] [PubMed]
- Suso, M.J.; Bebeli, P.J.; Christmann, S.; Mateus, C.; Negri, V.; Pinheiro de Carvalho, M.A.A.; Torricelli, R.; Veloso, M.M. Enhancing Legume Ecosystem Services through an Understanding of Plant–Pollinator Interplay. Front. Plant Sci. 2016, 7, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Balmer, O.; Pfiffner, L.; Schied, J.; Willareth, M.; Leimgruber, A.; Luka, H.; Traugott, M. Noncrop flowering plants restore top-down herbivore control in agricultural fields. Ecol. Evol. 2013, 3, 2634–2646. [Google Scholar] [CrossRef] [PubMed]
- Jones, G.A.; Sieving, K.E. Intercropping sunflower in organic vegetables to augment bird predators of arthropods. Agric. Ecosyst. Environ. 2006, 117, 171–177. [Google Scholar] [CrossRef]
- Ninkovic, V.; Dahlin, I.; Vucetic, A.; Petrovic-Obradovic, O.; Glinwood, R.; Webster, B. Volatile Exchange between Undamaged Plants - a New Mechanism Affecting Insect Orientation in Intercropping. PLoS ONE 2013, 8. [Google Scholar] [CrossRef] [PubMed]
- Stenberg, J.A.; Heil, M.; Ahman, I.; Bjorkman, C. Optimizing Crops for Biocontrol of Pests and Disease. Trends Plant Sci. 2015, 20, 698–712. [Google Scholar] [CrossRef] [PubMed]
- Pineda, A.; Kaplan, I.; Bezemer, T.M. Steering Soil Microbiomes to Suppress Aboveground Insect Pests. Trends Plant Sci. 2017, 22, 770–778. [Google Scholar] [CrossRef] [PubMed]
- Rasmann, S.; Bennett, A.; Biere, A.; Karley, A.; Guerrieri, E. Root symbionts: Powerful drivers of plant above- and belowground indirect defenses. Insect Sci. 2017, 947–960. [Google Scholar] [CrossRef] [PubMed]
- Birkhofer, K.; Bezemer, T.M.; Bloem, J.; Bonkowski, M.; Christensen, S.; Dubois, D.; Ekelund, F.; Fließbach, A.; Gunst, L.; Hedlund, K.; et al. Long-term organic farming fosters below and aboveground biota: Implications for soil quality, biological control and productivity. Soil Biol. Biochem. 2008, 40, 2297–2308. [Google Scholar] [CrossRef]
| Crop 1 | Fungicide | Herbicide | Insecticide | |||
|---|---|---|---|---|---|---|
| AI (MT) | Acreage (%) | AI (MT) | Acreage (%) | AI (MT) | Acreage (%) | |
| Vegetable crop2 | ||||||
| Asparagus | 16 | 53 | 41 | 88 | 20 | 90 |
| Beans, snap, processing | 36 | 49 | 138 | 97 | 11 | 58 |
| Broccoli | 17 | 38 | 61 | 46 | 31 | 68 |
| Carrots, fresh market | 188 | 75 | 73 | 77 | 3 | 35 |
| Carrots, processing | 28 | 100 | 5 | 100 | 0 | 100 |
| Cauliflower | 2 | 11 | 7 | 47 | 8 | 76 |
| Celery | 20 | 65 | 9 | 42 | 20 | 71 |
| Eggplant | 1 | 71 | 0 | 21 | 0 | 81 |
| Garlic | 16 | 88 | 29 | 89 | 4 | 58 |
| Lettuce, (excluding head) | 206 | 71 | 111 | 42 | 56 | 85 |
| Lettuce, head | 207 | 76 | 52 | 52 | 66 | 90 |
| Melons, cantaloupe | 99 | 79 | 42 | 54 | 31 | 85 |
| Melons, honeydew | 22 | 87 | 3 | 24 | 31 | 92 |
| Melons, watermelon | 282 | 84 | 31 | 52 | 114 | 80 |
| Onions, dry | 287 | 88 | 176 | 92 | 125 | 91 |
| Peas, green, processing | 2 | 14 | 60 | 98 | 2 | 29 |
| Peppers, bell | 199 | 84 | 17 | 44 | 46 | 81 |
| Pumpkins | 75 | 69 | 22 | 76 | 7 | 32 |
| Spinach, fresh market | 29 | 75 | 11 | 41 | 6 | 79 |
| Squash | 79 | 76 | 7 | 51 | 9 | 53 |
| Sweet corn, processing | 6 | 31 | 163 | 97 | 10 | 71 |
| Tomatoes, field, processing | 5073 | 87 | 268 | 69 | 216 | 79 |
| Total, vegetable crops | 6890 | 1326 | 816 | |||
| Fruit crop (bearing)3 | ||||||
| Apples | 2545 | 81 | 122 | 37 | 704 | 91 |
| Apricots | 18 | 70 | 7 | 55 | 5 | 77 |
| Avocados | ND | ND | 28 | 35 | 4 | 40 |
| Blackberries | 36 | 76 | 10 | 95 | 4 | 83 |
| Blueberries | 223 | 86 | 82 | 66 | 90 | 85 |
| Cherries, sweet | 627 | 83 | 48 | 36 | 150 | 83 |
| Cherries, tart | 252 | 95 | 14 | 54 | 30 | 90 |
| Dates | 0 | - | 3 | 23 | 0 | 8 |
| Figs | 0 | - | 19 | 60 | 0 | - |
| Grapefruit | 193 | 84 | 198 | 70 | 452 | 94 |
| Grapes 4 | 13,590 | 83 | 854 | 55 | 269 | 53 |
| Kiwifruit | 0 | - | 11 | 50 | 0 | 12 |
| Lemons | 25 | 34 | 51 | 60 | 53 | 74 |
| Nectarines | 73 | 55 | 16 | 49 | 7 | 55 |
| Olives | 50 | 43 | 23 | 39 | 2 | 26 |
| Oranges | 744 | 72 | 1890 | 72 | 2246 | 90 |
| Peaches | 824 | 82 | 46 | 43 | 68 | 78 |
| Pears | 566 | 90 | 24 | 45 | 564 | 92 |
| Plums | 27 | 51 | 14 | 47 | 3 | 70 |
| Prunes | 132 | 63 | 48 | 57 | 5 | 63 |
| Raspberries | 65 | 92 | 14 | 91 | 12 | 94 |
| Strawberries | 620 | 96 | 11 | 27 | 159 | 93 |
| Tangelos | 25 | 96 | 11 | 75 | 29 | 97 |
| Tangerines | 140 | 72 | 78 | 76 | 185 | 88 |
| Total, fruit crops | 20,775 | 3622 | 5041 | |||
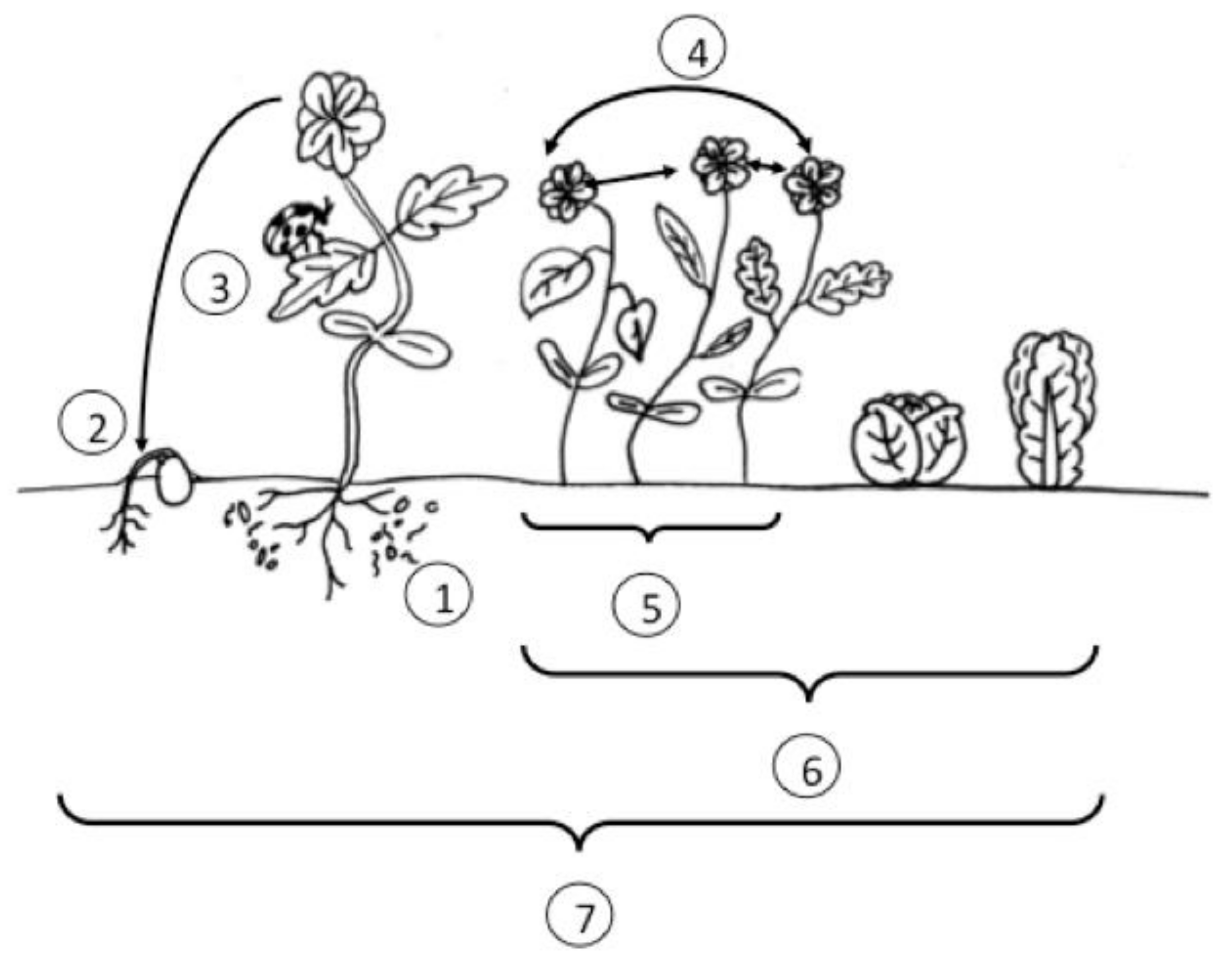
| Practice or Trait | Results | |
|---|---|---|
| Plant based resistance | Physical traits | • Deter or impede mobility of insect pests [29] or colonization of plant pathogens (i.e., cuticle composition) [30] • Canopy architecture can shade weeds [31], or alter environmental conditions (i.e., humidity) to slow pathogen growth [32] |
| Chemical traits | • Volatile deterrents for insect pests [33] • Harmful or deterrent secondary metabolites for pathogen and insect pests [34,35,36], and allelopathic compounds inhibit weed growth [37,38] • Volatile cues for insect predators or parasitoids about location of prey [39,40,41] • Qualitative gene-for-gene interactions [34,42] or quantitative resistance traits [42,43] | |
| Tolerance | • Plants exhibit no apparent yield or fitness cost to pest damage [44,45] | |
| Farm scale cultural practices | Sanitation | • Clean planting material and equipment stop inoculum from entering farm (pathogens, weeds and insects) [46,47] |
| Crop rotation | • Disrupt pest lifecycles (pathogens, weeds and insects) [46,48,49] | |
| Applying botanical diversity | • Trap crops or push-pull systems rely on differential plant attractiveness to lure and, or repel insect pests from main marketable crop [50,51] • Provide habitat and alternate food sources for plant beneficial insects [49] • Modify epidemiological factors to slow the spread of pathogens through crop rotations, intercropping, companion planting or growing a crop mixture [46] | |
| Crop targeted interventions | Beneficial organisms | • Beneficial insects that are predatory on pests, and nematodes and effective microbes can further suppress insect pest and pathogen populations [49,52,53] |
| Mechanical interventions | • Cultivation, thermal and mechanical measures to manage weeds or pathogens [46,54] • Specific passive traps (like trenches) or active control like vacuuming to manage particular insect pests [55] | |
| Naturally-derived products | • Non-synthetically derived products like oils, soaps, or extracts, can be used to supplement pest management efforts [46,49] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Brzozowski, L.; Mazourek, M. A Sustainable Agricultural Future Relies on the Transition to Organic Agroecological Pest Management. Sustainability 2018, 10, 2023. https://doi.org/10.3390/su10062023
Brzozowski L, Mazourek M. A Sustainable Agricultural Future Relies on the Transition to Organic Agroecological Pest Management. Sustainability. 2018; 10(6):2023. https://doi.org/10.3390/su10062023
Chicago/Turabian StyleBrzozowski, Lauren, and Michael Mazourek. 2018. "A Sustainable Agricultural Future Relies on the Transition to Organic Agroecological Pest Management" Sustainability 10, no. 6: 2023. https://doi.org/10.3390/su10062023
APA StyleBrzozowski, L., & Mazourek, M. (2018). A Sustainable Agricultural Future Relies on the Transition to Organic Agroecological Pest Management. Sustainability, 10(6), 2023. https://doi.org/10.3390/su10062023





