1. Introduction
California is still the third-largest oil-producing state in the United States [
1]. During the oil production process, substantial volumes of oilfield produced water (OPW, also referred to as oilfield brine, connate water, or formation water) are typically generated, especially as the well and oilfield matures. The origins of the briny water include flow from above, within, and below the targeted hydrocarbon zone as well as flow from injected fluids and additives employed during the production process [
2]. OPW is the largest waste stream by volume in the exploration and extraction of oil, and over the lifespan of an oilfield the total volume of produced water can exceed tenfold the volume of hydrocarbons produced [
3]. In 2012, for example, California generated an estimated 21 billion barrels of OPW, i.e., approximately 15 barrels of OPW for every barrel of oil [
4].
In general, OPW is high in salts and dissolved solids and may also contain metals, alkylphenols, trace elements, hydrocarbons, polycyclic aromatic hydrocarbons (PAHs), volatile organic compounds (VOCs), benzene, toluene, ethylbenzene, and xylenes (collectively known as BTEX), naturally occurring radioactive materials (NORMs), biocides, and other compounds used during the production process [
3]. Concentrations of total dissolved solids (TDS) in OPW range from a few thousand mg/L to several hundred thousand mg/L [
2]. In the Western U.S., including several California oilfields, measured concentrations range from 1000 mg/L to 400,000 mg/L [
5]. A case study analyzing the composition of OPW from 630 producing oil and gas wells in California that were stimulated through hydraulic fracturing found that the average concentration of Cr(VI) in the OPW was 8.5 μg/L, while the average concentrations of Sb was 1100 μg/L, As 1100 μg/L, Cd 40 μg/L, Pb 420 μg/L, Se 1900 μg/L, and Zn 420 μg/L [
4]. Many of the elements and compounds used in oil well drilling and production can have varying adverse health effects on the gastrointestinal tract, the nervous system, and the reproductive system when ingested.
Although the most common method for managing OPW in California is deep-well injection and disposal in evaporation-percolation pits, accounting for 60% and 18% of total OPW disposal, respectively, a small fraction of California’s OPW is applied to land surfaces [
6]. The Clean Water Act stipulates effluent guidelines for on-shore oil and gas extraction facilities that prohibit the discharge of pollutants into surface waters, except for wastewater that is of good enough quality for use in agricultural and wildlife propagation for onshore facilities located in the continental United States and west of the 98th meridian. This creates the possibility for reusing OPW in agricultural crop irrigation, as is the case in California’s Central Valley.
Although OPW has been reused in surface applications for more than 20 years, it gained wider prominence and interest during the exceptional drought that gripped California between 2011–2016 as a means to supplement heavily constrained agricultural freshwater supplies [
7,
8]. However, the question of whether it can be done safely for consumers, agricultural workers, and communities living near OPW application sites has not been sufficiently answered to date [
9]. Among the concerns voiced by public health and environmental groups is whether toxic OPW compounds, such as heavy metals, may become bioavailable and accumulate in parts of the crops intended for human consumption [
9]. Concerns about long-term soil health and productivity have also been raised [
9]. There is a substantial body of literature, involving field and laboratory studies, regarding the origins, environmental and health effects, and remediation of soil contamination with heavy metals, including Ba, Cd, Pb, and As [
10,
11,
12,
13,
14,
15,
16]. Studies by Fliessbach et al. (1994) and Khan et al. (2000) show the negative effects on soil productivity resulting from heavy metal contamination [
17,
18,
19]. Furthermore, Pontoni et al. (2016a,b) studied the accumulation, mobility, and fate in soil of trace amounts of Ni, Cd, Cu, and Zn and found, inter alia, that the metals accumulate mostly in the top soil layer and that their accumulation pattern depends on the mobility of colloids in the case of Ni, Cd, and Cu, while for Zn the wastewater content of dissolved organic matter and its salinity played greater roles [
20,
21].
While soil accumulation has been widely documented, Toze (2009) found evidence that the seeds and leaves of flax, cotton, and rice store only a small percentage of the heavy metals compared to soils [
22]. However, even small concentrations may pose health risks if the crop is consumed frequently, such as rice, or in larger amounts over a short period of time, such as seasonal crops like mandarin oranges. A study in Tongzhou District, Beijing, China, in 2007 examined wastewater-irrigated soils used to grow radish, maize, spinach, green cabbage, cauliflower, turnip, and lettuce [
23]. It found significant correlations between heavy metal concentration in soils and the edible parts of plants for Cd, Cu, Pb, and Zn. In particular, the concentrations of Cd, Cr, and Ni exceeded regulatory limits set by the State Environmental Protection Administration [
21]. Similar issues are known to arise in the proximity of metal ore mines. For example, the soil and corn, jujube, perilla, red pepper, soybean, and spring onions grown near a copper-tungsten mine in South East Korea were found to have elevated concentrations of Cu, Zn, Cd, and Pb [
24]. Concentrations varied with crop species and higher concentrations were found in the leafy material of the crops compared with the fruit and seeds. The study also determined that the most dominant factor affecting metal uptake in plants was the metal content in the surface soil, which may be problematic in the long-term application of OPW wastewater [
24]. Owsianiak et al. (2013, 2015) cautioned against estimating toxicity impacts from measured total soluble metal concentrations alone. They find that the bioaccumulation patterns and resulting eco-toxicity depend on the metals’ accessibility and bioavailability, which are influenced by environmental chemistry factors such as soil pH, organic matter content, and the pore water concentration of other metals [
25,
26].
While the existing literature demonstrates the potential for plant uptake of metals through soil and irrigation water, it is of specific interest to look into the potential soil and crop impacts of OPW reuse in agriculture in California’s Central Valley. As of now, water quality monitoring involving OPW is limited and not federally regulated [
5]. In response to concerns raised by environmental and public health organizations, the Central Valley Regional Water Board initiated a Food Safety Expert Panel tasked with examining the available evidence and conducting studies for testing the risk involved in OPW reuse in the region’s agricultural operations, primarily citrus, almond, pistachio, and table grape farming and to a lesser extent garlic, carrot, and tomato plantings [
9]. The panel commissioned sampling campaigns in the spring and summer of 2017 for table grapes, citrus, almonds, garlic, and pistachios and tested the edible fruit for over 100 organic, inorganic, and metal analytes. A total of 16 compound or elements were detected, among them the metals Ba, Cu, Mo, Ni, Sr, and Zn, although at mostly low concentrations [
27].
The present study complements the work of the Food Expert Panel with an observational study of the uptake of Ba, Cr, Pb, and Ag in the soil, leaves, and fruit of mandarin orange plants as a result of the controlled application of heavy metal tainted irrigation water. The goal of the study is to identify if and to what extent the tested heavy metals accumulate in the soil, leaves, and/or fruit in response to sampling time and the concentration of heavy metals used in the irrigation water.
2. Materials and Methods
A total of 23 mandarin orange trees, representing six varieties found in California’s Central Valley and residential gardens, were purchased in 5 gal. pots from the same local nursery in late spring 2017. The plants were semi-randomly assigned to receiving irrigation water with three different concentration levels of Ba, Cr, Pb, and Ag ions, ranging from 25 mg/L–3000 mg/L for Ba, 0.01 mg/L–0.10 mg/L for Cr, 5.0 mg/L–30 mg/L for Pb, and 1.0 mg/L–15 mg/L for Ag (see
Table 1 for details). These ranges reflect measurements reported in the OPW literature [
2,
28]. The tested metals are known for their risks to human health and soil productivity. Nitrate salts were used due to their high solubilities and the general lack of adverse effects on plant growth at the levels employed in this study. The high concentration level was selected to test for substantial plant effects such as growth decline and mortality. The procedure was semi-random, because fruit-bearing plants were allocated such that each concentration level had at least two plants with fruits.
Prior to commencing irrigation (or treatment) with the three levels of heavy metal concentrations, the baseline (or control) concentrations in the soil and leaves were determined. Plants were then watered twice (cold season) or three times (hot season) weekly. The watering frequency was based on recommendations received from the nursery regarding watering amounts and frequencies supporting healthy plant growth. During August and mid-December through mid-January the plants were watered with freshwater due to breaks in the academic calendar. Samples of soil and leaves were taken every two weeks during the active experimentation period. The fruit was harvested for analysis at the end of the experiment in March 2018.
Watering solutions were prepared on the days of irrigation. Each plant received 0.5 gal. (1.89 L) of heavy metal solution at every watering for a total of 22.5 gal. received at w = 43, i.e., the day that the final soil and leaf samples were collected. The soil and leaf samples were then processed by microwave-assisted digestion using U.S. Environmental Protection Agency (EPA) Method 3051A. Method blanks were run for each analysis. Specifically, in a typical procedure, 0.300 g of soil or leaf were dried at 60 °C for 3 days, digested with 12 mL of (3:1 HNO3:HCl, A.C.S. reagent grade, Spectrum Chemical Mfg. Corp., New Brunswick, NJ, USA), and filtered through a 2-μm glass fiber filter (Millipore Sigma AP2501300). The supernatant solution was diluted to 50.0 mL and stored in polypropylene test tubes. The samples were then analyzed using inductively coupled plasma-optical emission spectroscopy (ICP-OES). A PerkinElmer 8300 ICP-OES was used to construct calibration curves over the range of 0–400 mg/L Ba; 0–5.0 mg/L Cr; 0–50 mg/L Pb; and 0–0.25 mg/L Ag. The plasma operated at 1500 Watts of radio frequency (RF) power with argon gas flows of 10 L/min, 0.4 L/min, and 0.6 L/min in plasma, auxiliary, and nebulizer supplies, respectively. The oranges were analyzed in three parts: the juice, the pith, and the peel. The pith and peels were dried at 60 °C for 3 days prior to being processed as described above.
A total of seven samples of soils and leaves were collected and analyzed. In addition, 11 mandarin oranges were harvested and processed at the end of the experiment. The vector of watering days is
w = (0, 20, 26, 31, 35, 39, 43). Thus,
w = 0 represents the baseline (control) concentrations of Ba, Cr, Pb, and Ag in the soil and leaves at the start of the experiment, i.e., when no heavy metal irrigation water had been applied yet, while
w = 26 refers to the concentrations measured after 26 waterings. Analysis of covariance (ANCOVA) was carried out to model the change in measured metal concentrations in both the soil and leaf samples as a function of the heavy metal concentration in the irrigation water (the treatment) while controlling for waterings (
w). Analysis of variance (ANOVA) was used to estimate the effects of the irrigation water concentrations on the pith, peel, and juice of the fruit. Since the sampling design was not fully balanced (the low concentration level had only seven mandarin orange plants), type II and III sums of squares were used [
29]. Tukey’s honest significant difference test (Tukey HSD) was applied to identify differences in means between factor level combinations. All analyses were carried out in the R statistical language and computing environment, version 3.4.3 [
30]. Results are presented by metal.
4. Discussion
The study presents the result of a controlled experiment of the accumulation of four heavy metals (Ba, Cd, Pb, Ag) in the soil, leaves, and fruit of 23 mandarin orange trees that were irrigated with water containing concentrations of these elements at three levels that are representative of OPW conditions. The study’s goals were to detect the soil and plant (leaves, fruit) concentrations of these elements, which are frequently found in similar concentrations in oilfield produced water (OPW), and to examine if there were detectable trends over time and as a function of the metals’ concentration in the irrigation water. In the absence of federal (and state) regulations on the permissible concentrations of these and other compounds found in OPW, it is of interest to learn about their fate when OPW is reused for agricultural crop irrigation on a frequent basis, as is the case in several water districts in California’s agriculturally important Central Valley.
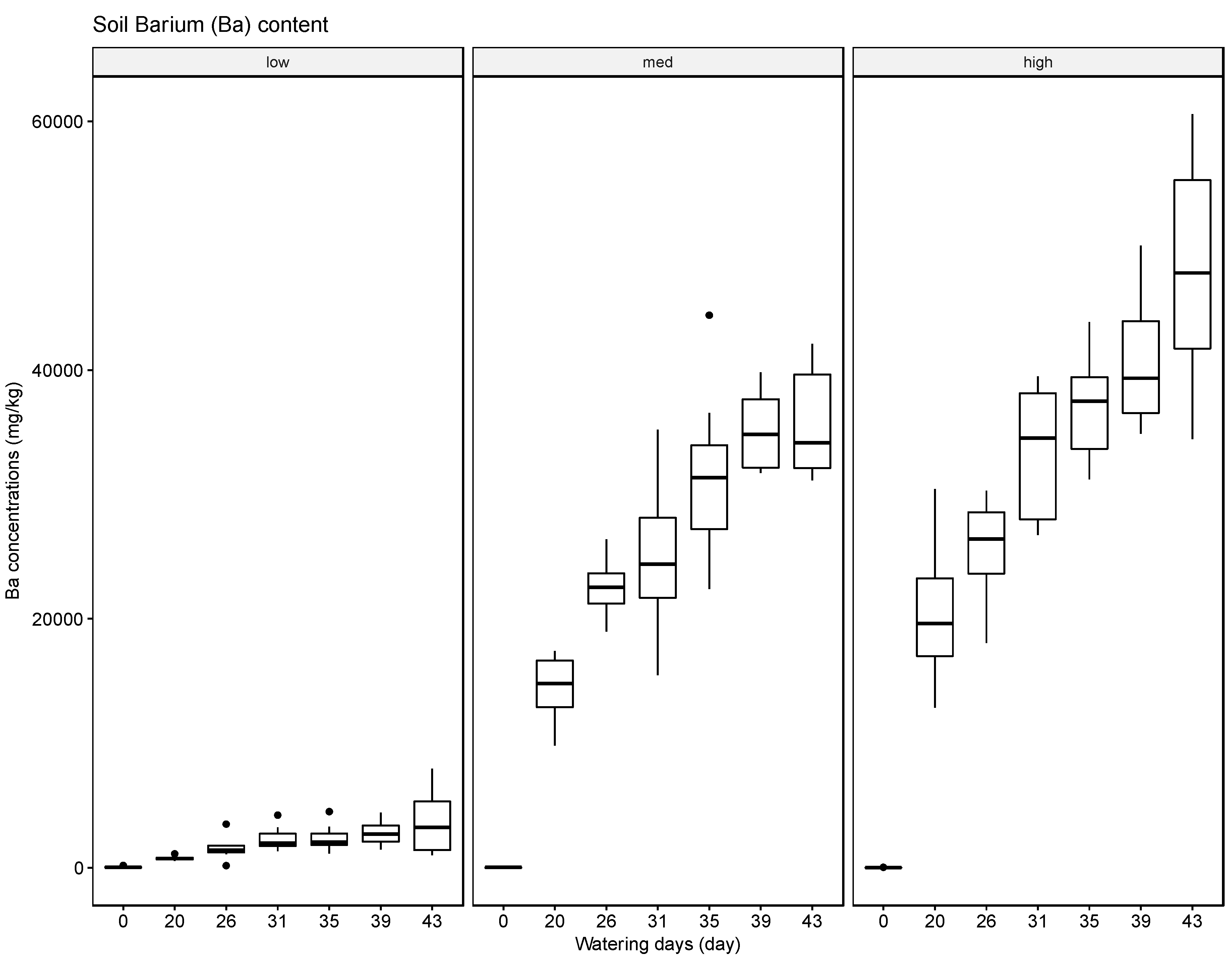
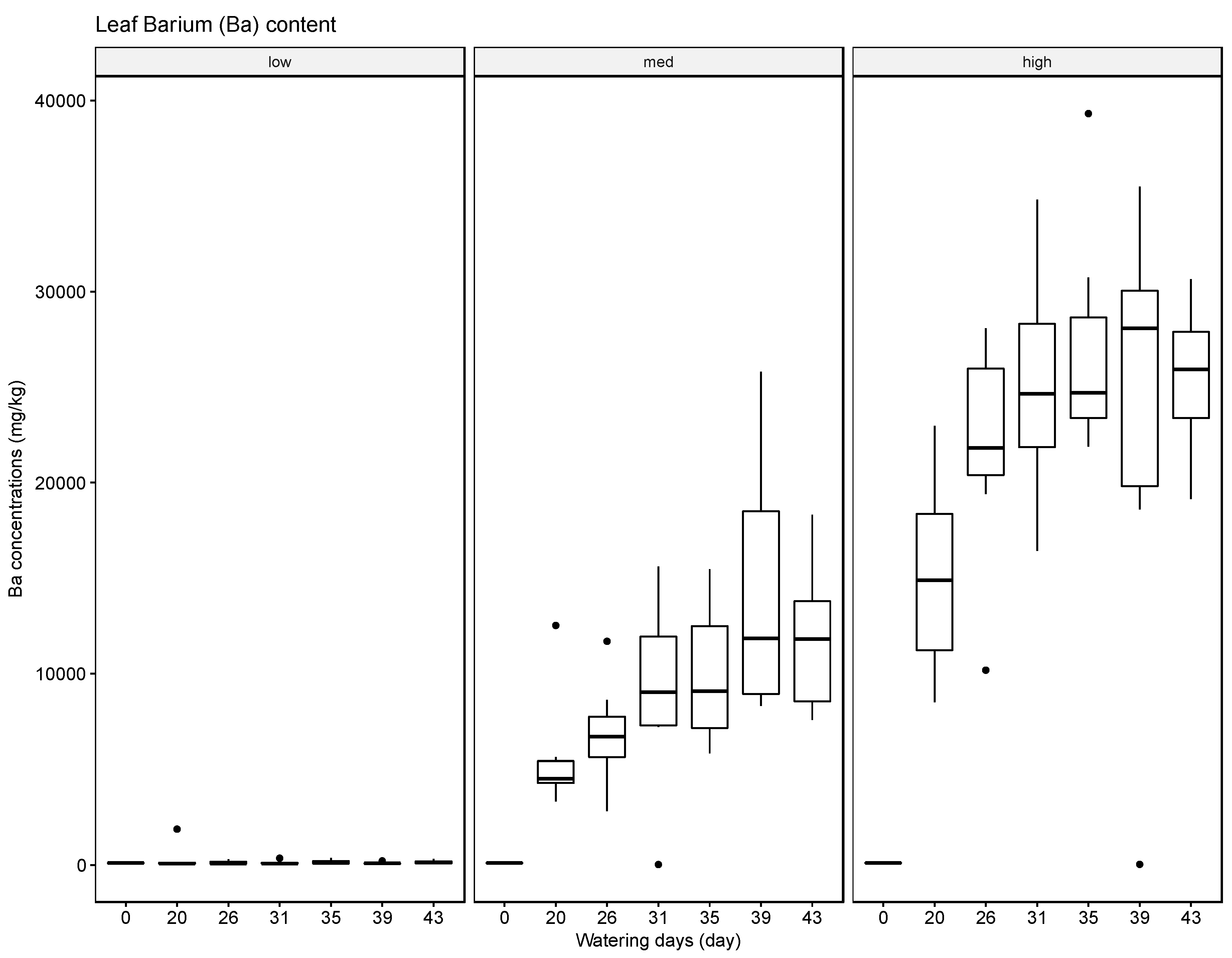
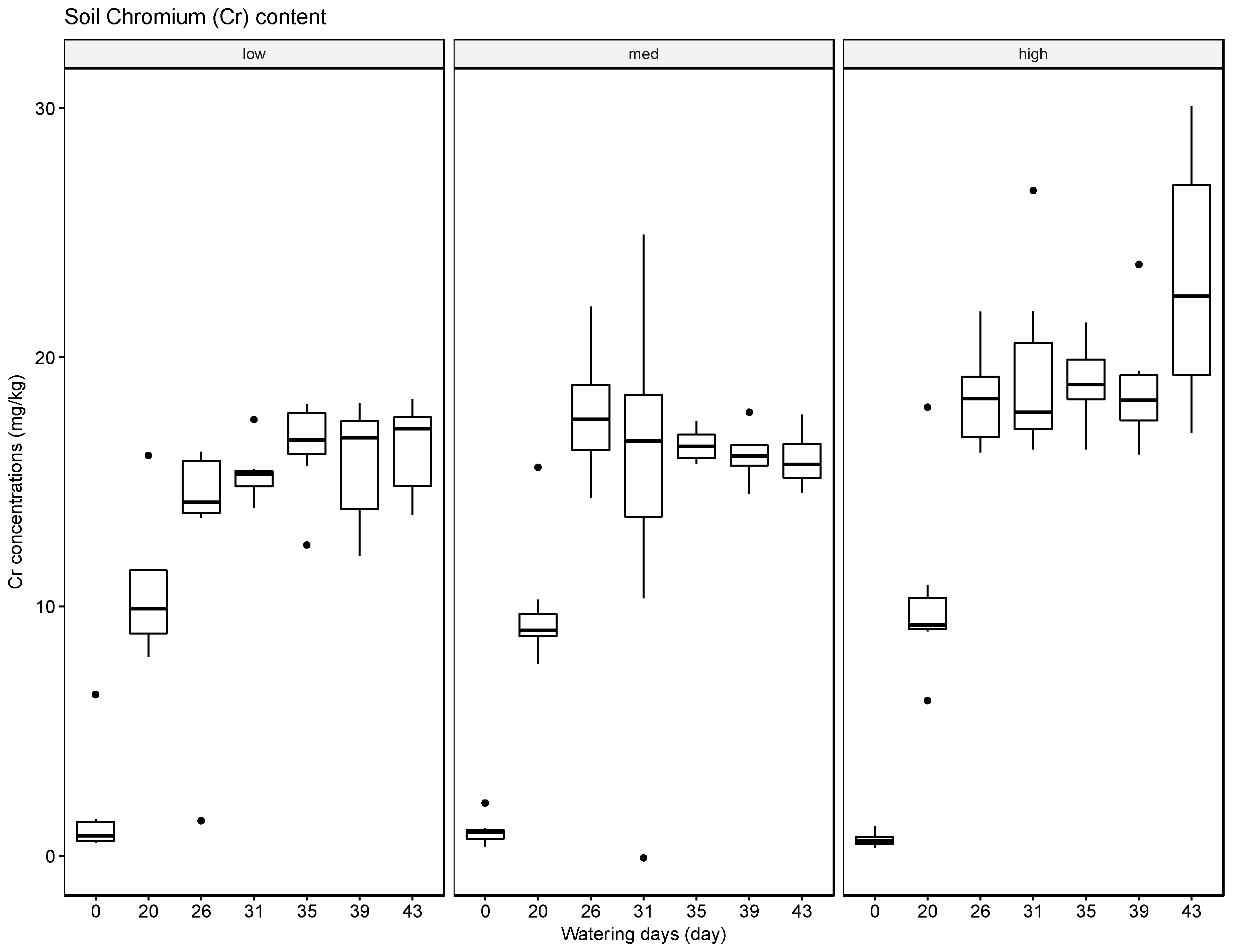
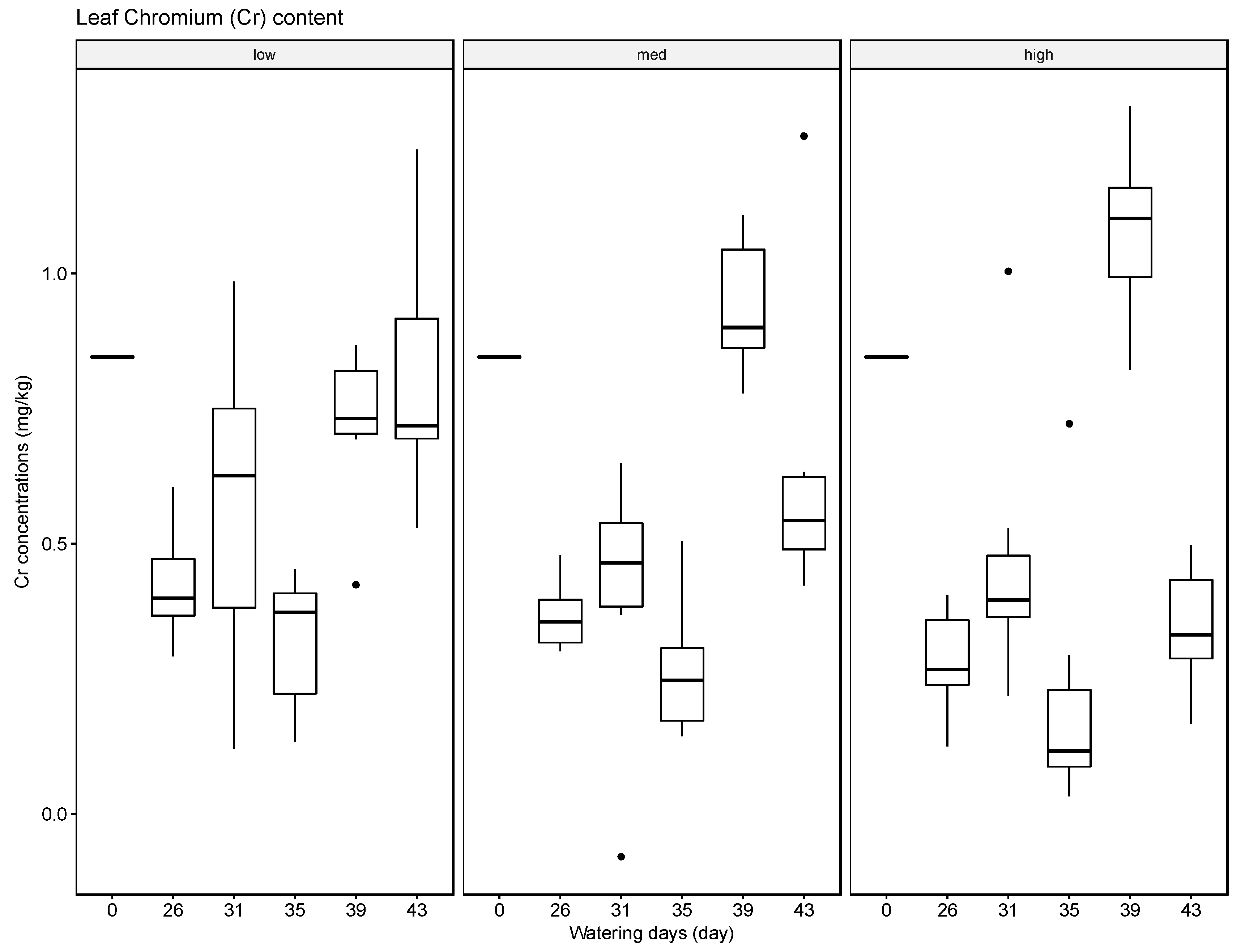
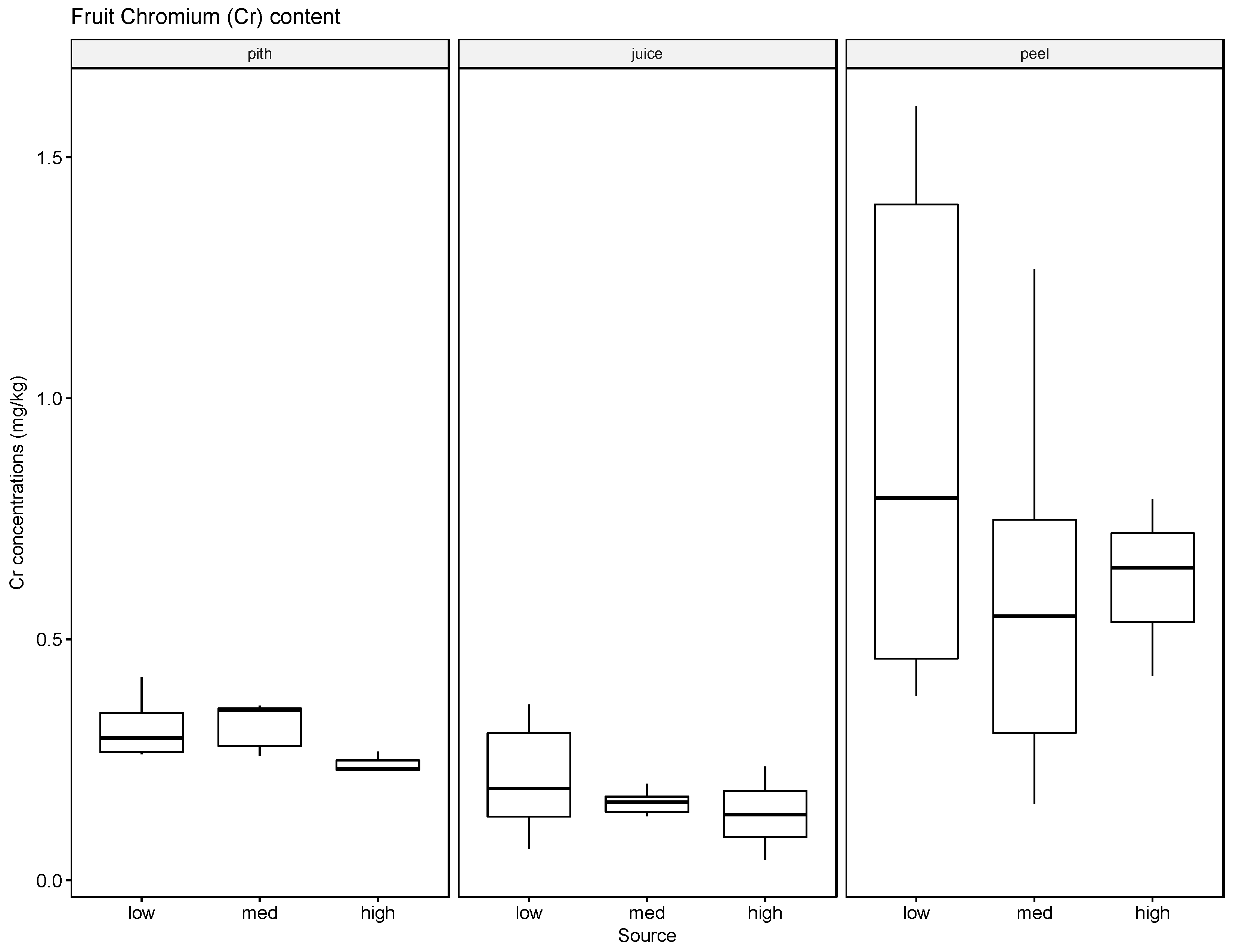
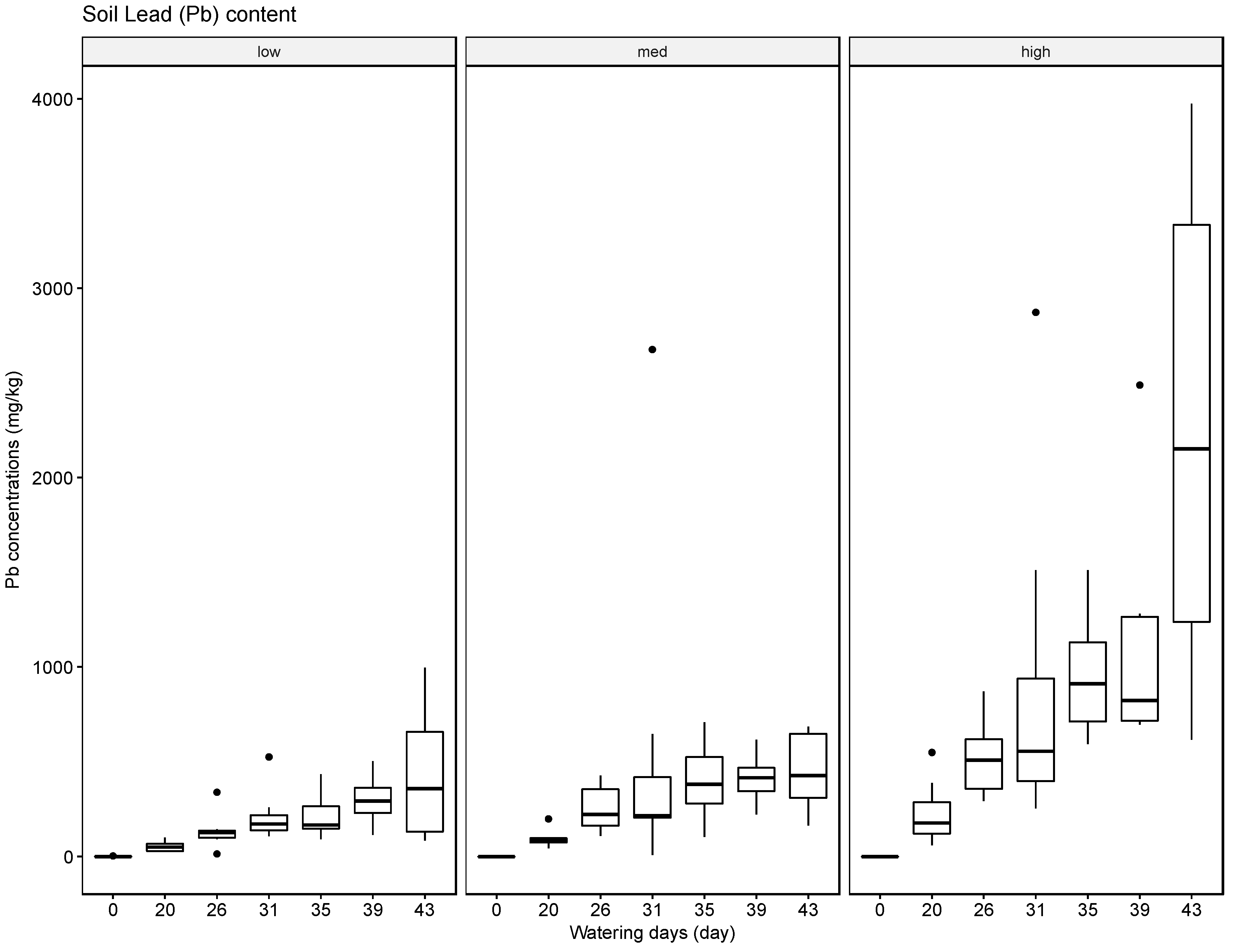
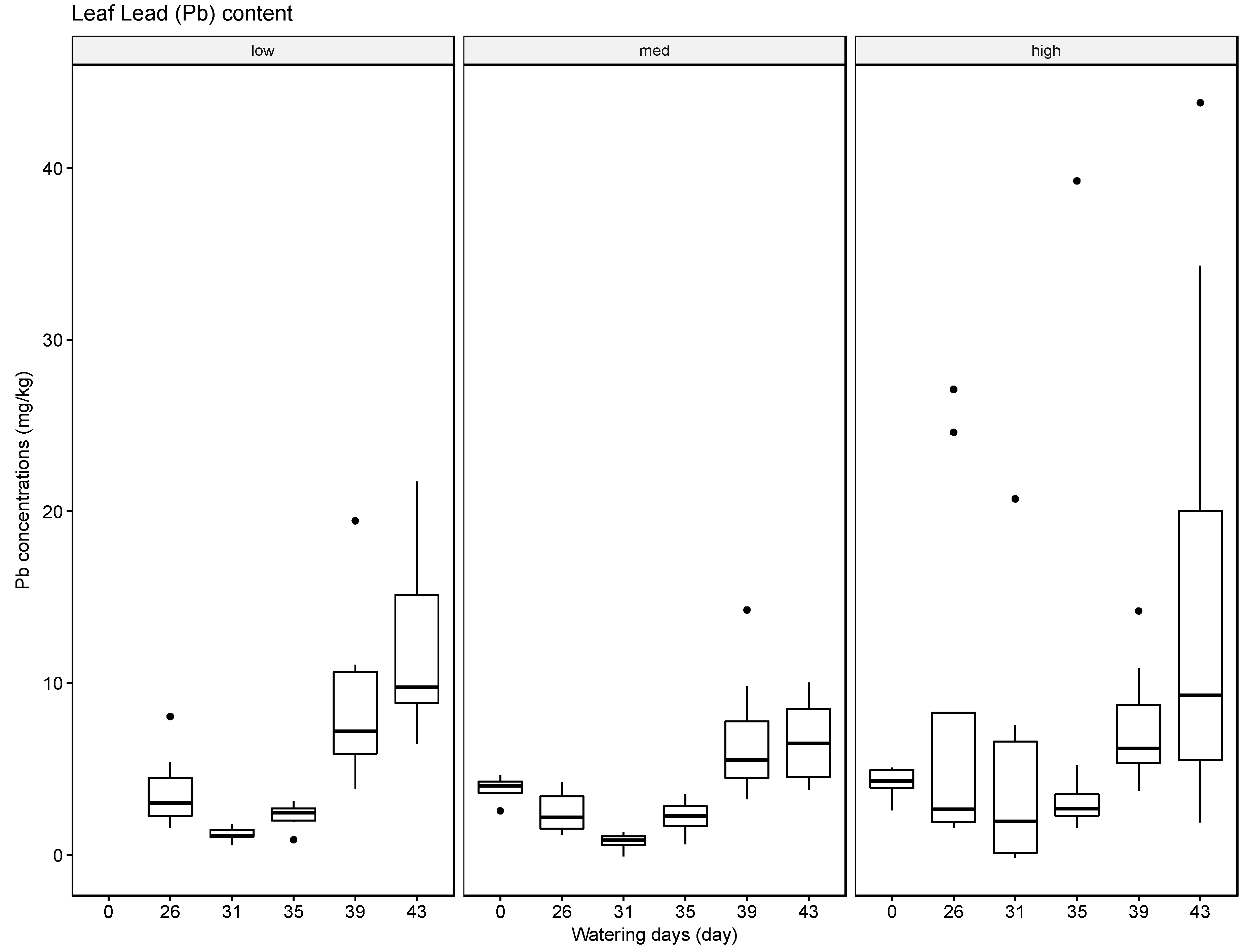
The results indicate that soils can accumulate several of the tested elements, in particular Ba, but to a lesser extent also Pb and Cr, and that generally the accumulated amounts are higher when these heavy metals are supplied in high concentrations in the irrigation water. The results for Ag were inconclusive. Compared with the soil samples, the leaf material showed consistently lower concentrations of the tested elements. The most significant uptake was observed for Ba at the high concentration level. This finding is supported by some reports in the literature that green leafy material, with the exception of leafy produce such as spinach and lettuce and senescing leaves in trees such as poplar, tends to store little heavy metals [
32,
33,
34].
There is very little peer-reviewed literature on the concentrations of the tested heavy metals in soils treated with OPW wastewater and hence little data available to compare our concentrations against. Odeigah et al. (1997) tested the general toxicity and genotoxicity of OPW on the bulbs of
Allium cepa L. and found significant phytotoxic and chromosomal effects [
35]. Pichtel (2016) gives an overview on the chemicals contained in OPW and their effects on soil health and productivity, while a grey literature report assesses how OPW use could impact the long-term health of agricultural production in California’s Central Valley [
6,
36]. Instead, most of the OPW literature focuses on treatment procedures and technologies prior to its application to farm fields.
All tested metals have known adverse human health effects that range from mild discomforts to serious organ damage and even death. For example, Ba (in soluble form), Cr, and Pb ingested in small to moderate concentrations can impact the digestive and respiratory tracts, the musculoskeletal and neurological systems, and the reproductive organs. The effects depend on the exposure route, frequency, and magnitude of exposure, as well as the chemical formulation of the metal (e.g., soluble versus insoluble Ba). Although the OPW in this study is used for crop irrigation and does not directly enter drinking water sources, it is illustrative to compare the measured concentrations with applicable drinking water standards, because of the risk of exposure to consumers of the edible crop. For Ba, the federal Maximum Contaminant Level (MCL) is 2.0 mg/L, for Pb the EPA specified an Action Level of 0.015 mg/L. The MCL for total Cr is 0.1 mg/L, and for Ag the federal Secondary Drinking Water Standard is 2.0 mg/L [
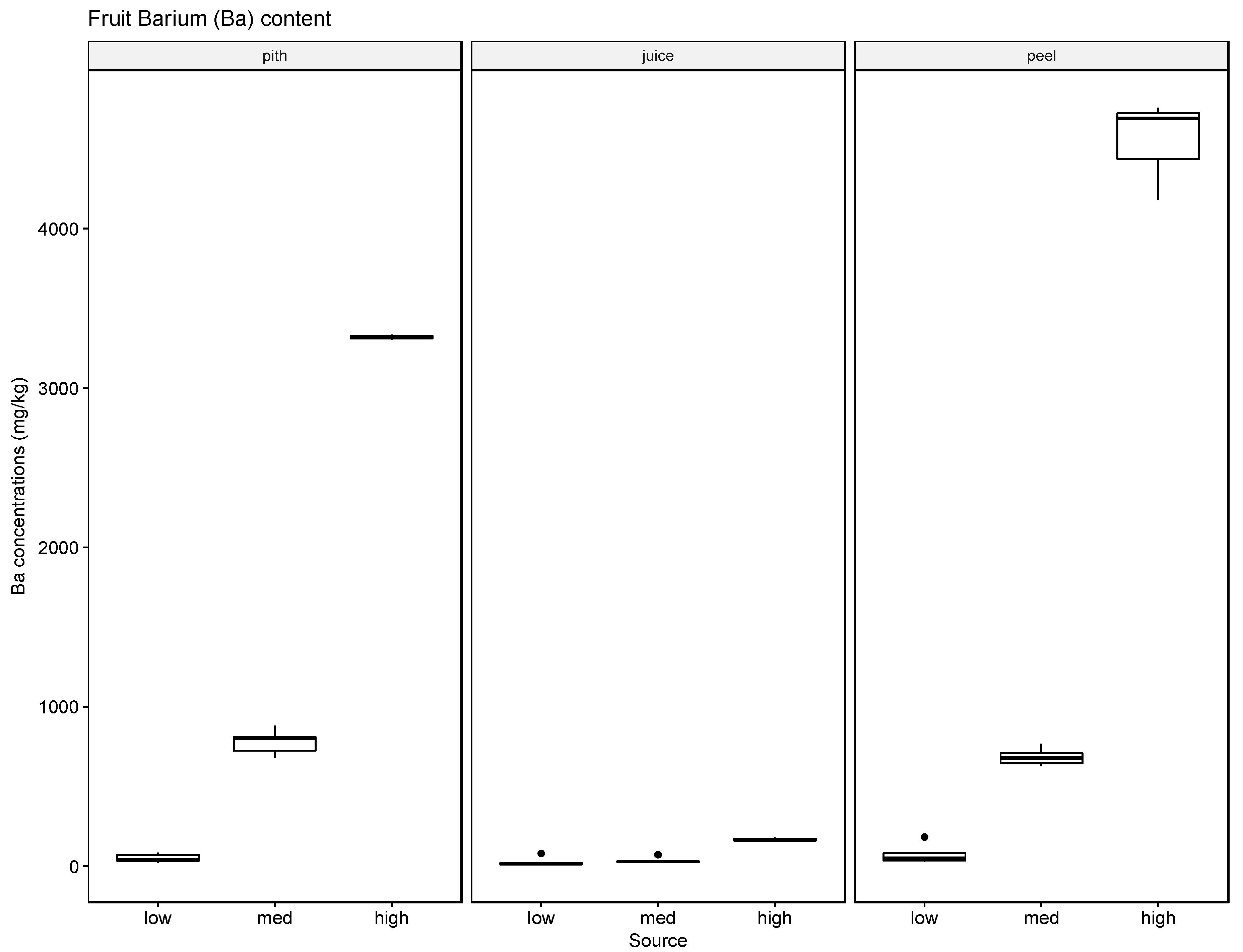
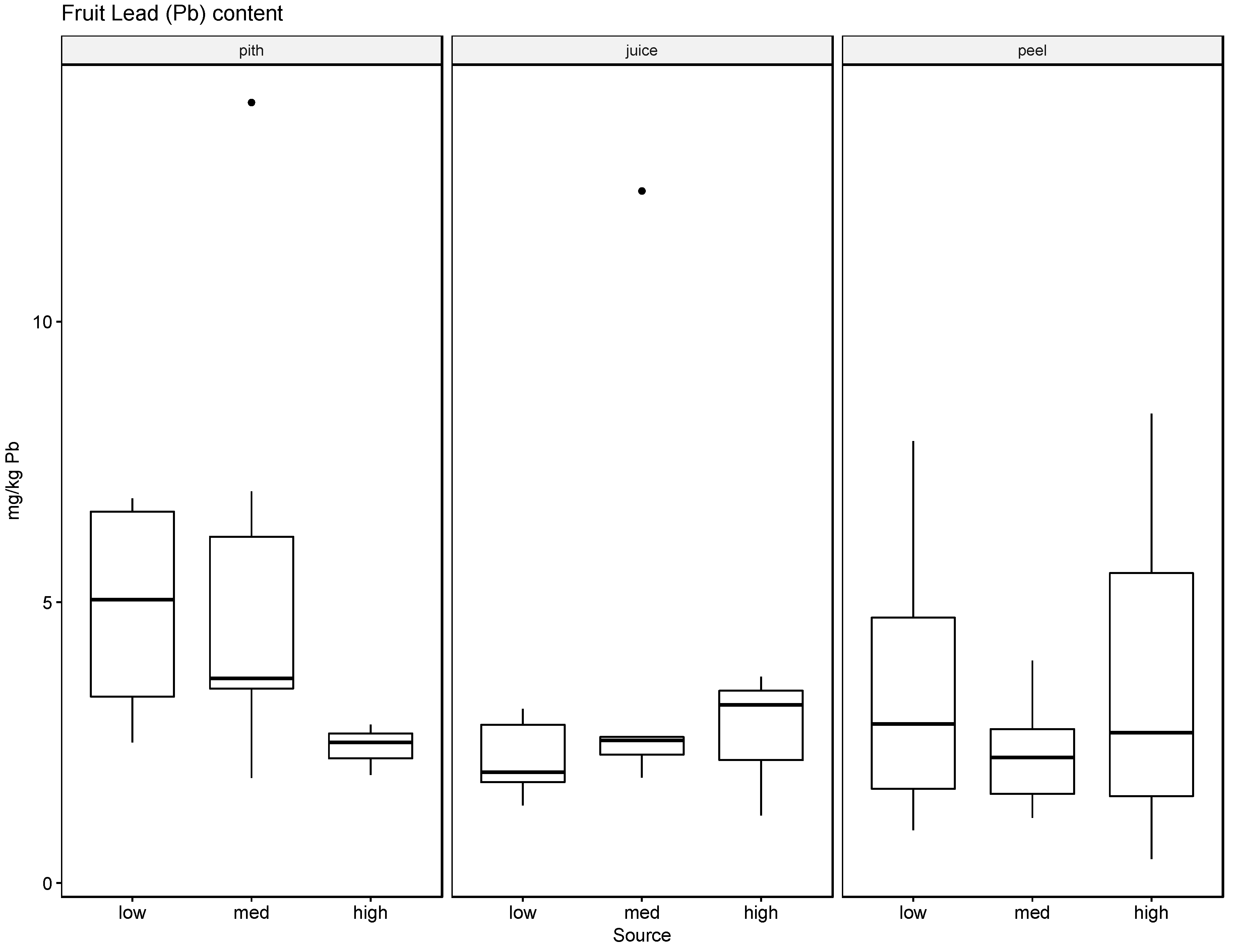
37]. The soil, leaf, and fruit concentrations measured for the four metals frequently exceeded these thresholds, noticeably at the medium and high watering concentrations. The potential exceedance of public health standards is especially concerning for the fruit measurements. The pith appears to be the most likely to accumulate the tested metals at concentrations potentially endangering the health of the consumer with concentrations for Ba ranging from 21.57 mg/kg to 4758.25 mg/kg, for Cr from 0.13 mg/kg to 1.61 mg/kg, for Pb from 0.43 mg/kg to 8.36 mg/kg, and for Ag from 0.10 mg/kg to 0.91 mg/kg.
It is noteworthy in this study that the concentrations reported are for total Ba, Cr, Pb, and Ag. While the heavy metals were applied in the watering solutions as the soluble species, the acid digestion and ICP-OES analysis provides no insights on soluble fraction, bioavailability, or oxidation state, i.e., Cr(III) versus Cr(VI), in soil, leaf, or fruit. Available research highlights the need for more differentiated study of the composition of the OPW and heavy metal speciation in soil and plant matter [
25,
26,
38,
39,
40,
41]. In particular, health risk assessments benefit from estimating the bioavailable fractions of the metals, which are generally smaller than the accessible fraction and the total concentration of the soluble salts. For example, in assessing the comparative toxicity potentials (CTPs) of Cu and Ni, the main influencing factors were the metals’ pH dependency with respect to dissolution and complexation; differences in metal availability for partitioning to the solution phase due to differences in solid-phase reactivity; and susceptibility of metal sorption, speciation, and toxicity to variations in soil chemistry parameters [
25]. A separate study of the bioavailability of Cd, Co, Cu, Ni, Pb, and Zn found that the emission source and aging mechanisms in the soil can also contribute to the metals’ reactivity [
26]. We intend to examine these issues in additional studies.
The study has some additional limitations. The sample size of 23 citrus plants is relatively small and the experiment was conducted on potted plants and not in an orchard in the Central Valley with actual OPW-supplemented irrigation water. Thus, the typical conditions, including soil cycling, soil types, and hydrological movements of irrigation water could not be fully replicated and no soil characterization and analysis of the transport mechanisms of the heavy metals in the soils and plants were done. These should be part of follow-up studies. The study also had a limited duration (June 2017 through March 2018) and was interrupted by two periods of watering with freshwater only. However, this watering, and a small number of precipitation events in the winter months, are assumed to have flushed out some of the metals that had accumulated in the soil as opposed to further enrich it. Finally, due to the time limits on the study, mandarin oranges could only be harvested and tested once.
5. Conclusions
The present study of the uptake of the heavy metals Ba, Cr, Pb, and Ag contained in soluble form in irrigation water by the soil and the leaves and fruit of mandarin orange plants allows the following conclusions. First, the elevated concentrations of Ba, Cr, and Pb indicate that a full characterization of their toxicity potentials is needed within the OPW context. The field sampling conducted by the Food Safety Expert Panel for citrus, almonds, pistachios, table grapes, and garlic tested for 108 elements and chemical compounds and detected 16 in the fruit and seed samples, among them Ba, Cu, Mo, Ni, Sr, and Zn. In several cases, concentrations of these metals in the treated crop were statistically higher than in the controls, but were still judged to be “non-alarming.” The present study indicates that field sampling, such as the studies conducted for the Food Safety Expert Panel, should be continued at least for citrus fruit to reaffirm that there is no risk for consumers. The results, furthermore, signal the need to conduct a systematic soil study to assess the risk of heavy metal accumulation for soil and crop health. The Regional Water Quality Control Board is in the process of planning additional studies for the new growing season to supplement the existing data. These studies aim to include root vegetables such as garlic, potatoes, and carrots, which may have a higher propensity to store heavy metals and other contaminants than tree crops. The present study suggests that soil analysis should be included as well.
The question of whether OPW use in agriculture poses risks to consumers of the impacted crops also requires testing the long-term effects of OPW application, because crops with very low to low concentrations of compounds with known adverse health effects may pose risks if the crop is consumed in large-enough quantities by consumers (e.g., the increase in consumption of mandarin oranges between October–February).
Since OPW chemical composition can change over time, often within short time periods, it remains advisable in the short term that OPW water quality testing continues for at least key contaminants that carry substantial health risks at even small concentrations, such as heavy metals, BTEX, and radioactive isotopes.
Adding more advanced treatment of OPW to regular OPW, crop, and soil quality monitoring can further ensure that harmful and toxic elements and chemicals do not reach agricultural products, especially those aimed for human consumption. With sufficient safeguards in place OPW could be considered a supplemental water resource for California’s Central Valley farms and hence make for a mutually beneficial partnership between the oil and agricultural industries in the region.