Liquid-Phase Respiration Activity Assays to Assess Organic Waste Stability: A Comparison of Two Tests
Abstract
1. Introduction
- (i)
- to develop a new, simple to use, liquid-phase respirometric method using manometric principles, and
- (ii)
- to compare the indices calculated by this new test with the indices calculated by an already published test (SOUR method) [2].
2. Materials and Methods
2.1. Substrates
2.1.1. Raw Substrates
2.1.2. Processed Substrates
2.2. Initial Substrate Characterization
2.3. Manometric Liquid Phase Test
2.4. SOUR Liquid Phase Test
3. Results and Discussion
3.1. Substrates’ Characterization
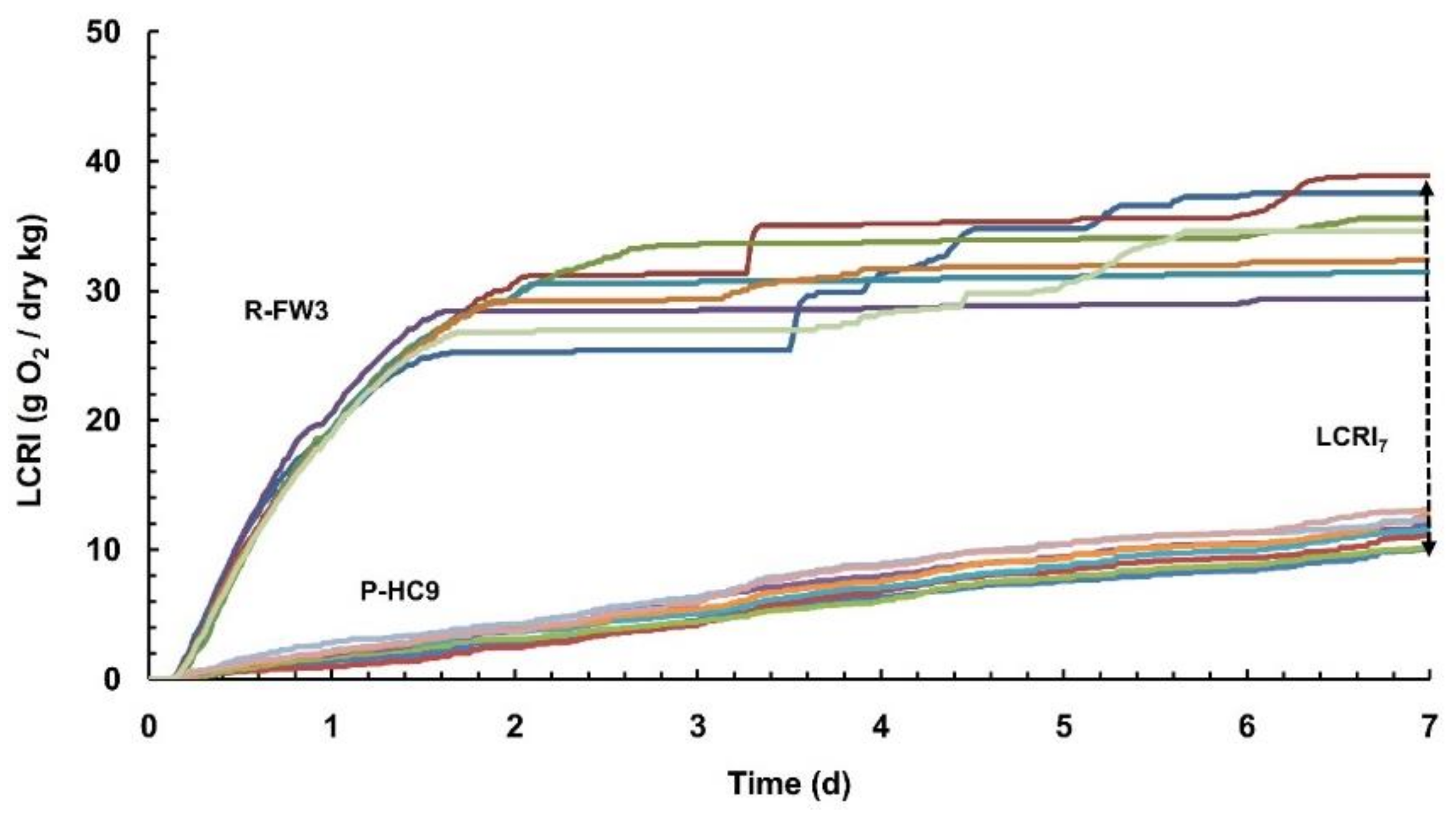
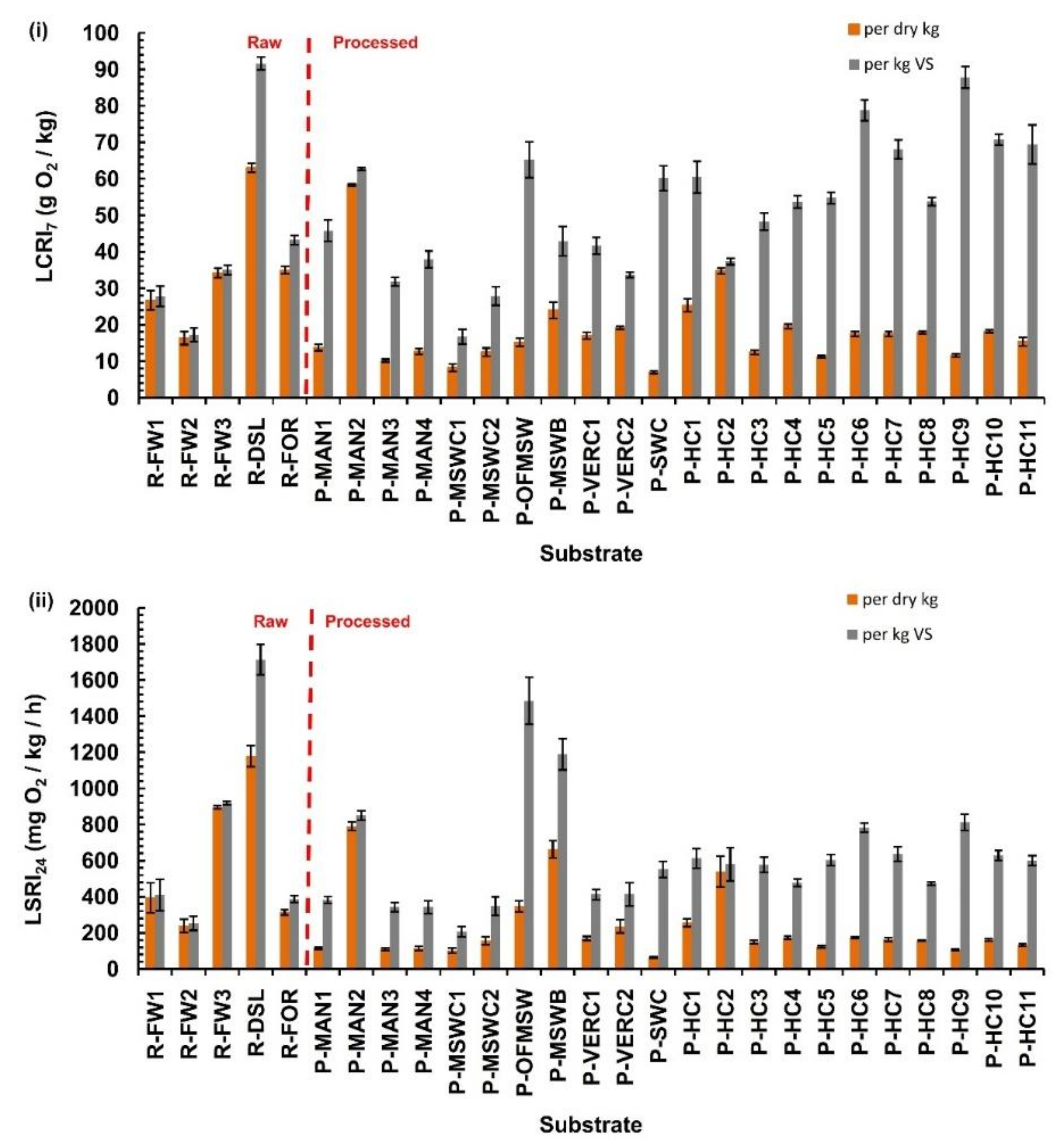
3.2. Manometric Liquid Phase Indices
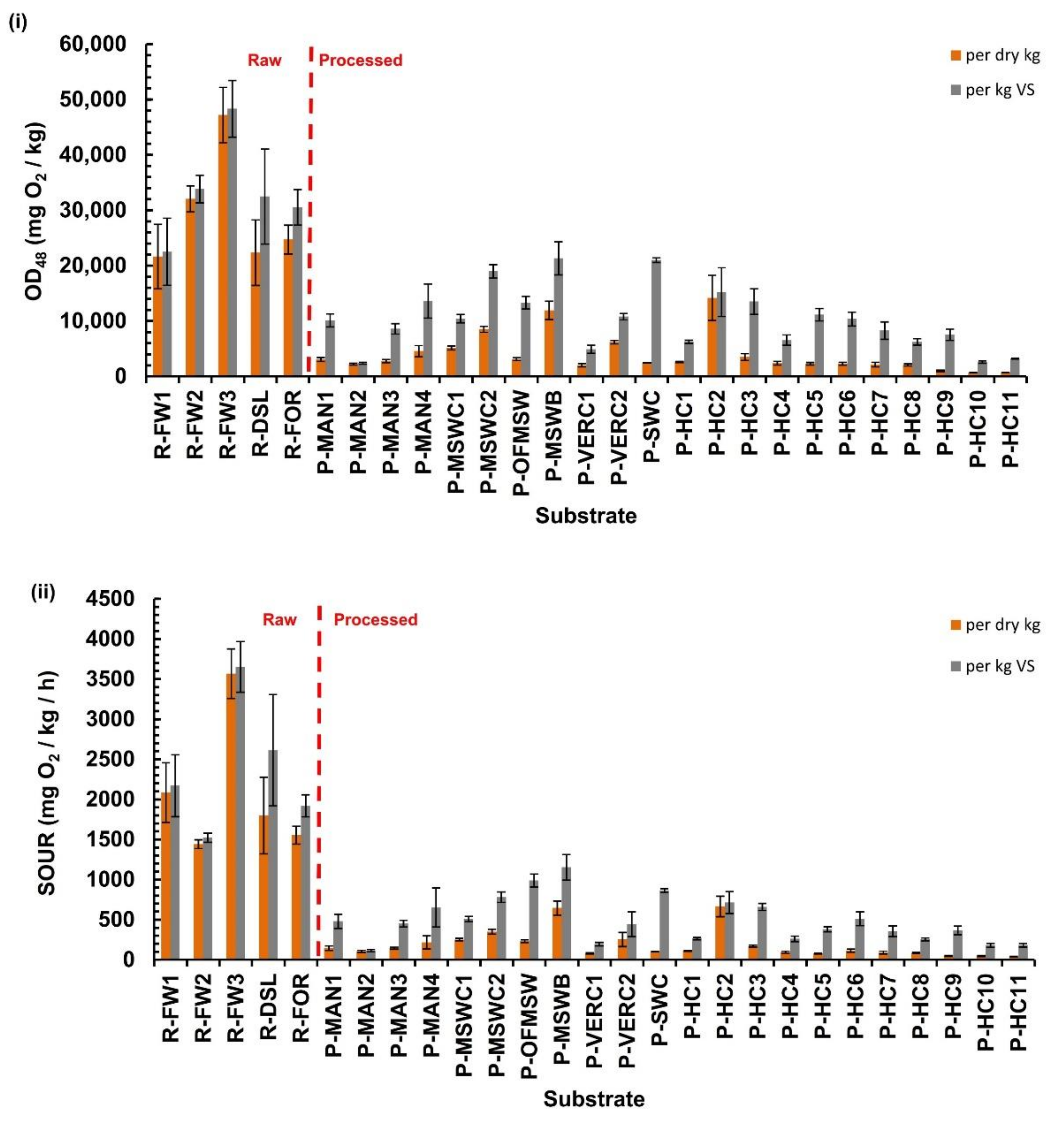
3.3. SOUR Test Indices
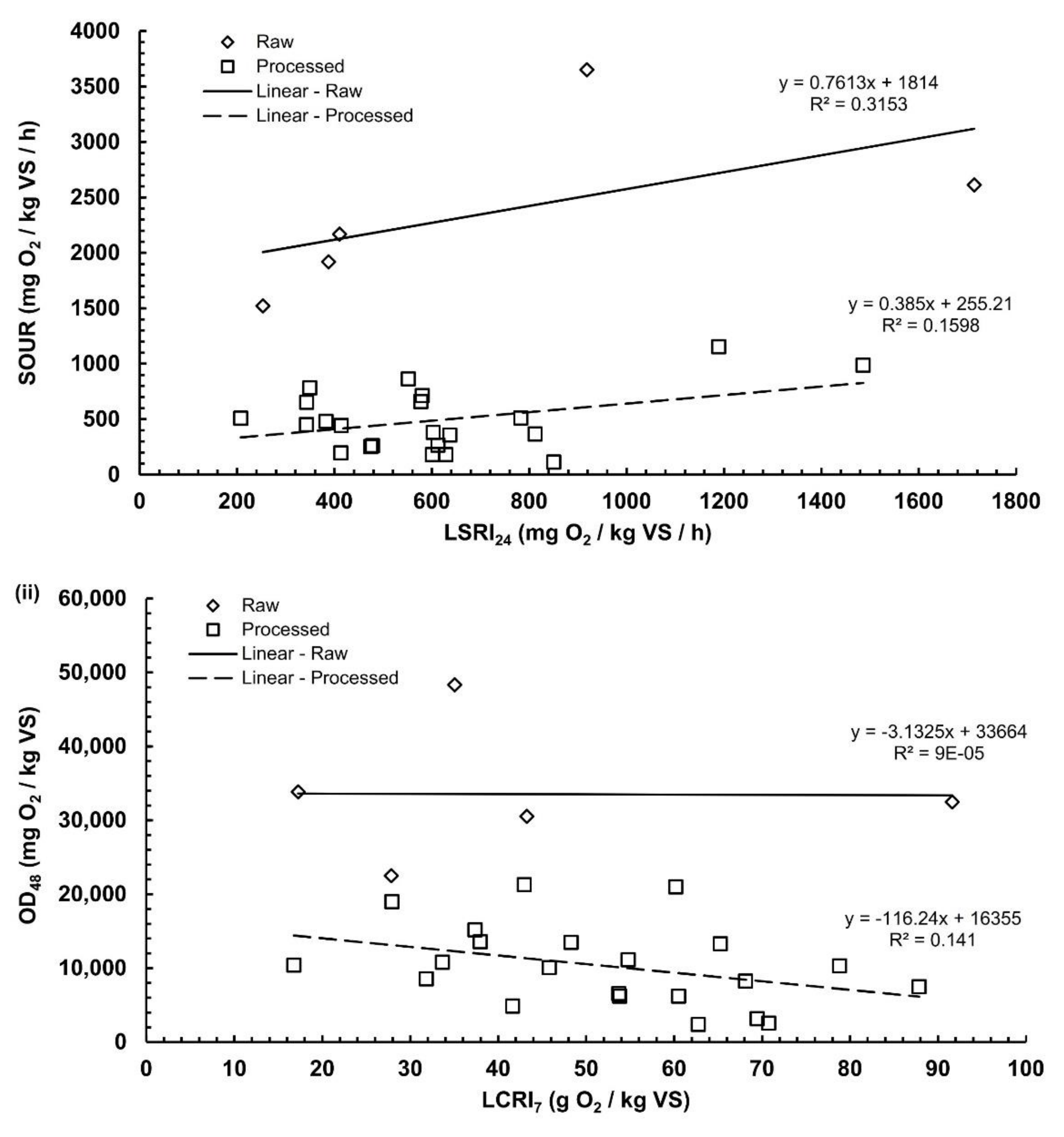
3.4. Correlation Analysis
3.5. Statistical Differences among Substrate Groups and Types
4. Conclusions
- A significant correlation among the respiration indices of both tests was found for the raw substrates only (n = 5). On the other hand, no significant correlation was found for the processed substrates (n = 22).
- The limits of the SOUR that had been proposed in the past (1000 mg O2/kg VS-h for matured composts) are verified here too, since almost all processed materials had SOUR values below that limit.
- The newly introduced liquid phase test can be used as an alternative test to assess organic waste stability. The 24 h oxygen consumption rates (LSRI24) for the raw and processed substrates ranged from 240 to 1180 mg O2/dry kg-h (250 to 1700 mg O2/kg VS-h) and from 64 to 792 mg O2/dry kg-h (210 to 1480 mg O2/kg VS-h), respectively. A clearer differentiation between fresh and more stabilized substrates is thus achieved when the above index is expressed on a per dry matter basis than on a per VS basis.
- The respiration activity indices calculated here were significantly lower in the processed substrates compared to the raw ones. On the other hand, no statistical differences were found between substrates when grouped by source material (food waste, manures, MSW, etc.).
- The main advantage of the newly proposed manometric liquid phase respiration activity test is that it can provide stability indices more representative of the composting process, than the 48 h SOUR test, due to its longer experimental duration (7 days), which is closer to actual composting periods. In addition, it is cheaper to set up, than the SOUR test, since it uses equipment widely available in most environmental laboratories.
Author Contributions
Acknowledgments
Conflicts of Interest
Abbreviations
| SOUR | specific oxygen uptake rate |
| VS | volatile solids |
| DO | dissolved oxygen |
| OD12,20,48 | cumulative oxygen demand after 12, 20, 48 h, respectively |
| OUR | oxygen uptake rate |
| db | dry weight basis |
| wb | wet weight basis |
| LOI | loss on ignition |
| MRA | microbial respiration activity |
| LCRI | liquid cumulative respiration index |
| LSRI | liquid static respiration index |
| BF | biodegradable fraction |
| ThOD | theoretical oxygen demand |
| C/N | carbon to nitrogen ratio |
| L/S | liquid to solid ratio |
| MBT | mechanical biological treatment |
| WWTP | wastewater treatment plant |
| BOD | biochemical oxygen demand |
| MSW | municipal solid waste |
References
- European Council. Directive, 2008/98/EC of 19 November 2008 on Waste and Repealing Certain Directives; Official Journal of the European Communities, L 312, 22/11/2008; European Council: Brusseles, Belgium, 2008. [Google Scholar]
- Lasaridi, K.; Stentiford, E. A simple respirometric technique for assessing compost stability. Water Res. 1998, 32, 3717–3723. [Google Scholar] [CrossRef]
- Oviedo-Ocana, E.R.; Torres-Lozada, P.; Marmolejo-Rebellon, L.F.; Hoyos, L.V.; Gonzales, S.; Barrena, R.; Komilis, D.; Sánchez, A. Stability and maturity of biowaste composts derived by small municipalities: Correlation among physical, chemical and biological indices. Waste Manag. 2015, 44, 63–71. [Google Scholar] [CrossRef] [PubMed]
- Barrena, R.; d’Imporzano, G.; Ponsa, S.; Gea, T.; Artola, A.; Vazquez, F.; Sánchez, A.; Adani, F. In search of a reliable technique for the determination of the biological stability of the organic matter in the mechanical-biological treated waste. J. Hazard. Mater. 2009, 162, 1065–1072. [Google Scholar] [CrossRef] [PubMed]
- Ponsá, S.; Gea, T.; Sánchez, A. Different indices to express biodegradability in organic solid wastes. J. Environ. Qual. 2010, 39, 706–712. [Google Scholar] [CrossRef] [PubMed]
- Komilis, D.; Kontou, I.; Ntougias, S. A modified static respiration assay and its relationship with an enzymatic test to assess compost stability and maturity. Bioresour. Technol. 2011, 102, 5863–5872. [Google Scholar] [CrossRef] [PubMed]
- Evangelou, A.; Calabrò, P.S.; Greco, R.; Sánchez, A.; Komilis, D. Biodegradation activity of eight organic substrates: A correlation study of different test methods. Waste Biomass Valoriz. 2016, 7, 1067–1080. [Google Scholar] [CrossRef][Green Version]
- Tremier, A.; de Guardia, A.; Massiani, C.; Paul, E.; Martel, J.L. A respirometric method for characterizing the organic composition and biodegradation kinetics and the temperature influence on the biodegradation kinetics, for a mixture of sludge and bulking agent to be composted. Bioresour. Technol. 2005, 96, 169–180. [Google Scholar] [CrossRef] [PubMed]
- Adani, F.; Gigliotti, G.; Valentini, F.; Laraia, R. Respiration index determination: A comparative study of different methods. Compost Sci. Util. 2003, 11, 144–151. [Google Scholar] [CrossRef]
- Scaglia, B.; Tambone, F.; Genevini, P.L.; Adani, F. Respiration index determination: A dynamic and static approach. Compost Sci. Util. 2000, 8, 90–98. [Google Scholar] [CrossRef]
- Adani, F.; Ubbiali, C.; Generini, P. The determination of biological stability of composts using the dynamic respiration index: The results of experience after two years. Waste Manag. 2006, 26, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Komilis, D.; Kanellos, D. A modified dynamic respiration test to assess compost stability: Effect of sample size and air flowrate. Bioresour. Technol. 2012, 117, 300–309. [Google Scholar] [CrossRef] [PubMed]
- Binner, E.; Zach, A. Laboratory Tests Describing the Biological Reactivity of Pretreated Residual Wastes. In Proceedings of the ORBIT Symposium, Weimar, Germany, 2–4 September 1999. [Google Scholar]
- Iannotti, D.A.; Pang, T.; Toth, B.L.; Elwell, D.L.; Keener, H.M.; Hoitink, H.A.J. A quantitative respirometric method for monitoring compost stability. Compost Sci. Util. 1993, 1, 52–65. [Google Scholar] [CrossRef]
- Gea, T.; Barrena, R.; Artola, A.; Sánchez, A. Monitoring the biological activity of the composting process: Oxygen uptake rate (OUR), respirometric index (RI), and respiratory quotient (RQ). Biotechnol. Bioeng. 2004, 88, 520–527. [Google Scholar] [CrossRef] [PubMed]
- Villaseñor, J.; Pérez, M.A.; Fernández, F.J.; Puchalski, C.M. Monitoring respiration and biological stability during sludge composting with a modified dynamic respirometer. Bioresour. Technol. 2011, 102, 6562–6568. [Google Scholar] [CrossRef] [PubMed]
- Scaglia, B.; Erriquens, F.G.; Gigliotti, G.; Taccari, M.; Ciani, M.; Genevini, P.L.; Adani, F. Precision determination for the specific oxygen uptake rate (SOUR) method used for biological stability evaluation of compost and biostabilized products. Bioresour. Technol. 2007, 98, 706–713. [Google Scholar] [CrossRef] [PubMed]
- Stentiford, E. The SOUR Method; Biowaste Workshop: Brussels, Belgium, 2002. [Google Scholar]
- Barrena, R.; Vazquez, F.; Sanchez, A. The use of respiration indices in the composting process: A review. Waste Manag. 2006, 24, 37–41. [Google Scholar] [CrossRef]
- U.S. Department of Agriculture (USDA); U.S. Composting Council (USCC). Test Methods for the Examination of Composting and Compost; Thomson, W., Ed.; The Composting Council Research and Education Foundation, Holbrook: New York, NY, USA, 2002; pp. 03.09.1–03.09.4. [Google Scholar]
- American Public Health Association (APHA); American Water Works Association (AWWA). Standard Methods for the Examination of Water and Wastewater, 20th ed.; American Public Health Association: Washington, DC, USA, 1998; ISBN 0875532357. [Google Scholar]
- Komilis, D.; Evangelou, A.; Giannakis, G.; Lymperis, C. Revisiting the elemental composition and the calorific value of the organic fraction of municipal solid wastes. Waste Manag. 2012, 32, 372–381. [Google Scholar] [CrossRef] [PubMed]
- Komilis, D.P.; Tziouvaras, I.S. A statistical analysis to assess the maturity and stability of six composts. Waste Manag. 2009, 29, 1504–1513. [Google Scholar] [CrossRef] [PubMed]
- Bernal, M.P.; Alburquerque, J.A.; Moral, R. Composting of animal manures and chemical criteria for compost maturity assessment. A review. Bioresour. Technol. 2009, 100, 5444–5453. [Google Scholar] [CrossRef] [PubMed]
- Pognani, M.; Barrena, R.; Font, X.; Scaglia, B.; Adani, F.; Sánchez, A. Monitoring the organic matter properties in a combined anaerobic/aerobic full-scale municipal source separated waste treatment plant. Bioresour. Technol. 2010, 101, 6873–6877. [Google Scholar] [CrossRef] [PubMed]
- Sciubba, L.; Marzadori, C.; Cavani, L.; Grigatti, M.; Ciavatta, C. Relationships between stability, maturity, water-extractable organic matter of municipal sewage sludge composts and soil functionality. Environ. Sci. Pollut. Res. 2015, 22, 13393–13403. [Google Scholar] [CrossRef] [PubMed]
- Puyuelo, B.; Ponsá, S.; Gea, T.; Sánchez, A. Determining C/N ratios for typical organic wastes using biodegradable fractions. Chemosphere 2011, 85, 653–659. [Google Scholar] [CrossRef] [PubMed]
- Evangelou, A.; Gerassimidou, S.; Mavrakis, N.; Komilis, D. Monitoring the performances of a real scale municipal solid waste composting and a biodrying facility using respiration activity indices. Environ. Monit. Assess. 2016, 188, 302. [Google Scholar] [CrossRef] [PubMed]
- Aspray, T.J.; Dimambro, M.E.; Wallace, P.; Howell, G.; Frederickson, J. Static, dynamic and inoculum augmented respiration based test assessment for determining in-vessel compost stability. Waste Manag. 2015, 42, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Bayard, R.; Benbelkacem, H.; Gourdon, R.; Buffière, P. Characterization of selected municipal solid waste components to estimate their biodegradability. J. Environ. Manag. 2017. [Google Scholar] [CrossRef] [PubMed]

| Category | Substrate | Type | Description and Process Applied |
|---|---|---|---|
| Raw | R-FW1 | Food waste | Simulated mixture of apples, cooked meat, boiled pasta, bread (25% each, in wb). |
| R-FW2 | Food waste | Simulated mixture of cooked meat, bread (50% each, in wb). | |
| R-FW3 | Food waste | Simulated mixture of uncooked pasta, uncooked fries, beef-based dog food (50%, 30%, 20%, in wb). | |
| R-DSL | Dewatered sludge | Dewatered sludge derived from a WWTP | |
| R-FOR | Forest derived material | Leaves and small branches collected from a forest floor | |
| Processed | P-MAN1 | Cow manure | Fresh cow manure. Prepared in static piles for 1 month. |
| P-MAN2 | Cow manure and straw | Mixture of fresh cow manure and straw. Stored for 1 week in an open pile | |
| P-MAN3 | Cow manure and straw | Mixture of fresh cow manure and straw. 1 week old, after passing through a solid-liquid separator. | |
| P-MAN4 | Cow manure compost | Mixture of fresh com manure and straw. 10 months active composting period. | |
| P-MSWC1 | MSW derived compost | Compost derived from commingled MSW, in a mechanical and biological aerobic pretreatment facility. | |
| P-MSWC2 | MSW derived compost | Compost derived from screened and undersized fraction of commingled MSW, in a MBT plant. | |
| P-OFMSW | Organic fraction of MSW | Organic fraction of municipal solid wastes. Aeration for 28 days in a MBT plant. | |
| P-MSWB | MSW wrapped in bales | Commingled MSW wrapped in bales. Sampling after 1 year of storage within the bales. | |
| P-VERC1 | Vermicompost | Mixture of dewatered wastewater sludge with straw. Vermicomposting process. | |
| P-VERC2 | Vermicompost | Mixture of dewatered wastewater sludge with straw. Vermicomposting process. | |
| P-SWC | Sea weeds derived compost | Prepared from 80% sea weeds and 20% cow manure and other agricultural products | |
| P-HC1 | Home composts | Mix of kitchen and garden waste and ash. 6 months composting and 6 months curing. | |
| P-HC2 | Mix of kitchen waste and wood chips. 3 weeks forced aeration and 2 weeks curing. | ||
| P-HC3 | Mix of kitchen waste, garden waste, wood chips, ash, sawdust and various household organic amendments. Composting for a period between 6 to 12 months, in home composters. | ||
| P-HC4 | |||
| P-HC5 | |||
| P-HC6 | |||
| P-HC7 | |||
| P-HC8 | |||
| P-HC9 | |||
| P-HC10 | |||
| P-HC11 |
| Substrate 1 | Moisture (% wb) + | Volatile Solids (% db) − | pH ^ | C/N * | Empirical Formula * |
|---|---|---|---|---|---|
| R-FW1 | 61% ± 1.7% | 96% ± 0.6% | 5.4 ± 0.0 | 13.8 ± 1.6 | C16H34.8NO10.6 |
| R-FW2 | 49% ± 0.2% | 95% ± 2.8% | 6.0 ± 0.0 | 7.7 ± 1.6 | C8.7H20.6NO4.9 |
| R-FW3 | 40% ± 3.6% | 98% ± 0.1% | 5.1 ± 0.0 | 19.6 ± 1.1 | C22.9H49.4NO24.9 |
| R-DSL | 82% ± 0.2% | 69% ± 0.1% | n/a | 8.2 ± 0.2 | C9.6H18.4NO4.2 |
| R-FOR | 73% ± 0.4% | 81% ± 0.9% | 6.2 ± 0.2 | 33.9 ± 3.1 | C39.5H81NO35.1 |
| P-MAN1 | 41% ± 1.3% | 30% ± 1.6% | n/a | 15.0 ± 0.7 | C17.5H14.2NO11.5 |
| P-MAN2 | 79% ± 0.2% | 93% ± 0.6% | 8.3 ± 0.1 | 8.2 ± 0.2 | C9.6H18.4NO8.9 |
| P-MAN3 | 40% ± 0.8% | 32% ± 0.3% | 8.1 ± 0.0 | 11.3 ± 0.9 | C13H24.6NO5.5 |
| P-MAN4 | 41% ± 0.2% | 34% ± 1.0% | n/a | 7.6 ± 0.8 | C8.8H11.8NO10.8 |
| P-MSWC1 | 8% ± 0.2% | 49% ± 1.1% | n/a | 14.6 ± 2.0 | C17H23.5NO8.7 |
| P-MSWC2 | 6% ± 0.0% | 45% ± 1.5% | n/a | 10.7 ± 1.0 | C12.4H21.6NO10.8 |
| P-OFMSW | 22% ± 2.4% | 23% ± 4.3% | 7.7 ± 0.1 | 21.9 ± 5.2 | C24.4H22.5NO2.3 |
| P-MSWB | 44% ± 1.9% | 56% ± 1.1% | n/a | 45.2 ± 4.9 | C51.9H88NO35.9 |
| P-VERC1 | 46% ± 0.5% | 41% ± 1.1% | n/a | 14.8 ± 1.0 | C17.2H33.1NO13.8 |
| P-VERC2 | 42% ± 0.5% | 57% ± 0.2% | n/a | 8.6 ± 0.2 | C10H16.9NO6.3 |
| P-SWC | 25% ± 0.2% | 12% ± 0.0% | n/a | 50.9 ± 10.1 | C57.5H17.9NO11.9 |
| P-HC1 | 65% ± 0.4% | 42% ± 0.6% | 8.8 ± 0.1 | 24.6 ± 3.9 | C28H23.9NO12 |
| P-HC2 | 62% ± 1.4% | 93% ± 0.3% | 7.2 ± 0.1 | 39.5 ± 5.3 | C45.3H68.8NO26.6 |
| P-HC3 | 51% ± 0.2% | 26% ± 1.7% | 9.4 ± 0.0 | 14.3 ± 1.7 | C17.7H24.1N1.1O |
| P-HC4 | 54% ± 1.0% | 37% ± 0.3% | 9.2 ± 0.0 | 15.0 ± 2.0 | C17.4H22.4NO9.4 |
| P-HC5 | 45% ± 0.1% | 20% ± 0.8% | 9.7 ± 0.0 | 18.4 ± 4.5 | C21H17.5NO11.1 |
| P-HC6 | 44% ± 0.7% | 22% ± 1.3% | 9.5 ± 0.0 | 14.4 ± 3.1 | C21.8H26.4N1.4O |
| P-HC7 | 39% ± 1.7% | 26% ± 1.1% | 8.4 ± 0.1 | 12.2 ± 1.9 | C14.1H19.7NO5.8 |
| P-HC8 | 35% ± 3.4% | 33% ± 0.9% | 9.4 ± 0.0 | 14.7 ± 4.3 | C16.5H25.4NO7 |
| P-HC9 | 24% ± 0.2% | 13% ± 0.5% | 8.4 ± 0.0 | 11.8 ± 1.5 | C13.7H14.7NO3.7 |
| P-HC10 | 51% ± 0.1% | 26% ± 0.4% | 7.3 ± 0.0 | 10.1 ± 1.1 | C11.7H16.1NO6.1 |
| P-HC11 | 26% ± 0.9% | 22% ± 0.9% | 7.9 ± 0.1 | 15.1 ± 2.5 | C17.1H27.8NO3.7 |
| Raw Substrates | |||||||
| LCRI7 | LSRI24 | OD48 | |||||
| LSRI24 | 0.830 | ||||||
| OD48 | n/s | n/s | |||||
| SOUR | n/s | 0.459 | 0.603 | ||||
| Processed substrates | |||||||
| LCRI7 | LSRI24 | OD48 | |||||
| LSRI24 | 0.617 | ||||||
| OD48 | −0.360 | n/s | |||||
| SOUR | −0.326 | n/s | 0.857 | ||||
| Liquid Manometric Test | Liquid SOUR Test | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| LCRI7 (g/dry kg) | LSRI24 (mg/dry kg-h) | LCRI7 (g/kg VS) | LSRI24 (mg/kg VS-h) | OD48 (g/dry kg) | SOUR (mg/dry kg-h) | OD48 (g/kg VS) | SOUR (mg/kg VS-h) | ||
| per dry kg | per kg VS | per dry kg | per kg VS | ||||||
| Groups | Raw | 32.9A ± 13.7 | 529A ± 350 | 39.3B ± 21.8 | 622A ± 473 | 31.1A ± 12.6 | 2026A ± 975 | 35.0A ± 12.5 | 2294A ± 1013 |
| (n = 35) | (n = 28) | ||||||||
| Processed | 17.6B ± 10.5 | 208B ± 82.3 | 52.6A ± 19.2 | 571A ± 256 | 3.35B ± 0.57 | 156B ± 59.9 | 9.27B ± 5.77 | 433B ± 276 | |
| (n = 149) | (n = 123) | ||||||||
| Types | Home composts | 18.0CD ± 6.55 | 189B ± 129 | 62.6A ± 15.7 | 618AB ± 147 | 2.75D ± 1.85 | 125D ± 63.7 | 8.15E ± 5.18 | 369D ± 215 |
| (n = 83) | (n = 77) | ||||||||
| Manures | 21.6BCD ± 9.25 | 253B ± 81.5 | 43.1BC ± 12.7 | 459B ± 214 | 2.96D ± 1.24 | 145D ± 79.1 | 8.01DE ± 4.90 | 395D ± 284 | |
| (n = 29) | (n = 25) | ||||||||
| MSW | 13.6D ± 6.30 | 258B ± 59.0 | 32.2CD ± 17.5 | 612AB ± 220 | 6.99CD ± 3.13 | 339D ± 146 | 15. 9C ± 4.87 | 799CD ± 257 | |
| (n = 22) | (n = 15) | ||||||||
| Food wastes | 25.7ABC ± 9.01 | 516A ± 317 | 26.7D ± 9.09 | 533AB ± 322 | 36.4A ± 12.7 | 2319A ± 1122 | 37.9A ± 12.8 | 2401A ± 1130 | |
| (n = 20) | (n = 16) | ||||||||
| Sludges | 32.2AB ± 21.5 | 511A ± 172 | 53.3AB ± 26.9 | 814A ± 139 | 13.2C ± 2.55 | 982BC ± 176 | 20.2BC ± 7.45 | 1466BC ± 531 | |
| (n = 13) | (n = 8) | ||||||||
| Forest waste | 35.0A ± 3.32 | 314AB ± 48.3 | 43.2BCD ± 4.10 | 388B ± 59.6 | 24.7B ± 7.35 | 1553B ± 315 | 30.5AB ± 9.08 | 1919AB ± 389 | |
| (n = 11) | (n = 8) | ||||||||
| Seaweed | 7.02D ± 0.97 | 64.4B ± 12.4 | 60.2AB ± 8.35 | 552AB ± 106 | 2.45CD ± 0.07 | 101CD ± 3.83 | 21.0BCDE ± 0.58 | 863BCD ± 32.8 | |
| (n = 6) | (n = 2) | ||||||||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Evangelou, A.; Komilis, D. Liquid-Phase Respiration Activity Assays to Assess Organic Waste Stability: A Comparison of Two Tests. Sustainability 2018, 10, 1441. https://doi.org/10.3390/su10051441
Evangelou A, Komilis D. Liquid-Phase Respiration Activity Assays to Assess Organic Waste Stability: A Comparison of Two Tests. Sustainability. 2018; 10(5):1441. https://doi.org/10.3390/su10051441
Chicago/Turabian StyleEvangelou, Alexandros, and Dimitrios Komilis. 2018. "Liquid-Phase Respiration Activity Assays to Assess Organic Waste Stability: A Comparison of Two Tests" Sustainability 10, no. 5: 1441. https://doi.org/10.3390/su10051441
APA StyleEvangelou, A., & Komilis, D. (2018). Liquid-Phase Respiration Activity Assays to Assess Organic Waste Stability: A Comparison of Two Tests. Sustainability, 10(5), 1441. https://doi.org/10.3390/su10051441






