The Performance of Early-Generation Perennial Winter Cereals at 21 Sites across Four Continents
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sites
2.2. Experiment Design and Germplasm
2.3. Management
2.4. Sampling
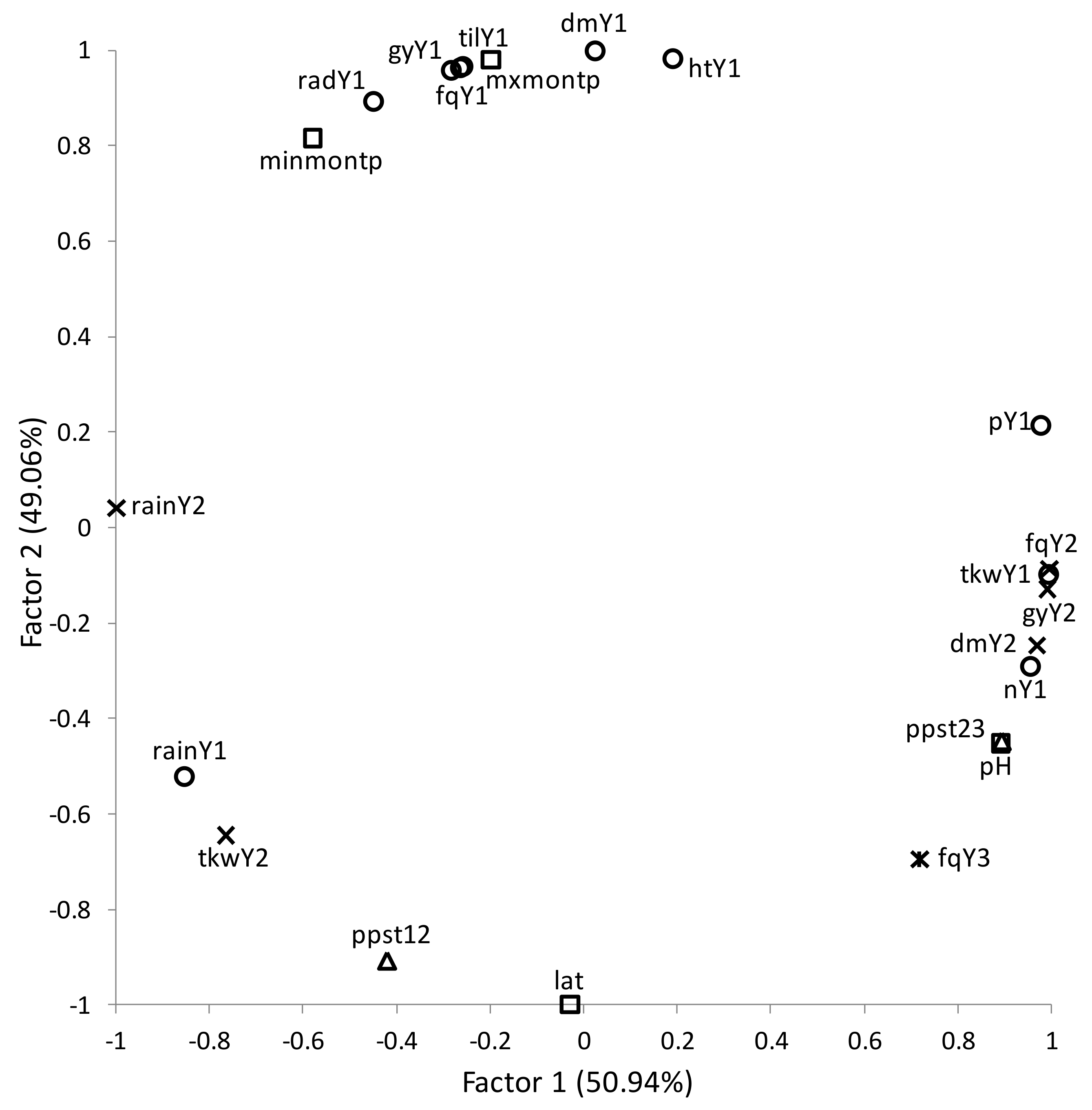
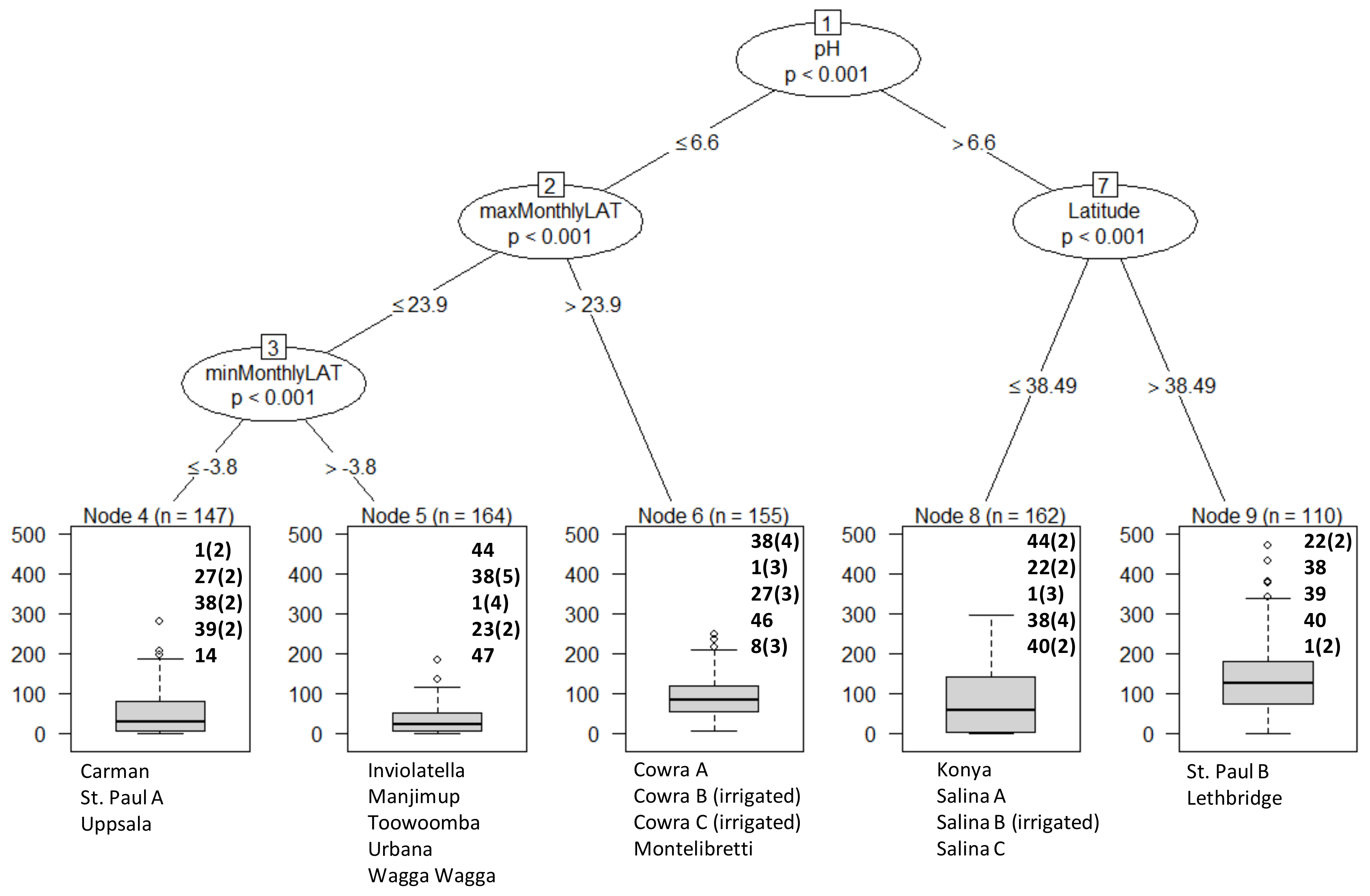
2.5. Statistical Analysis
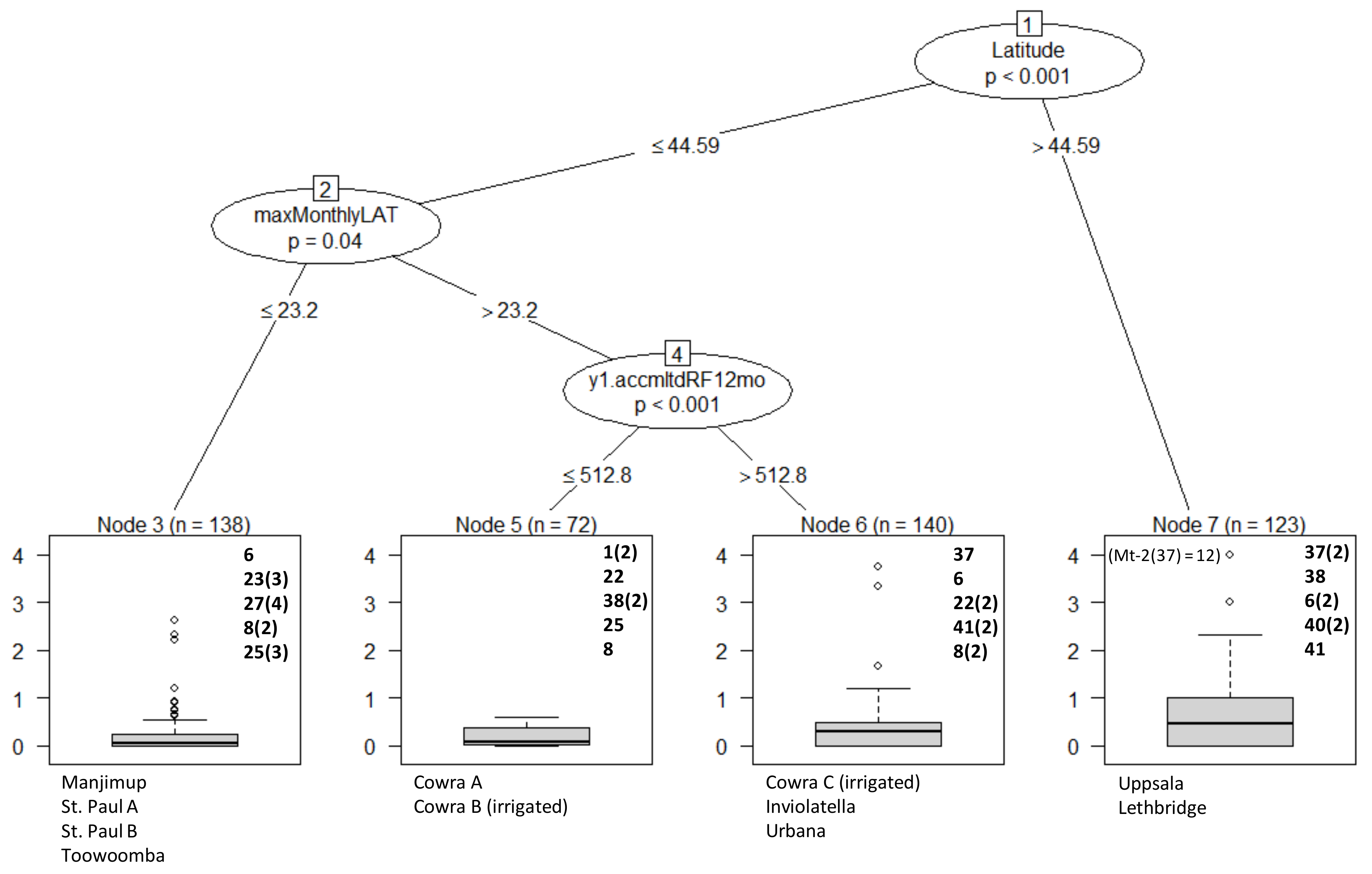
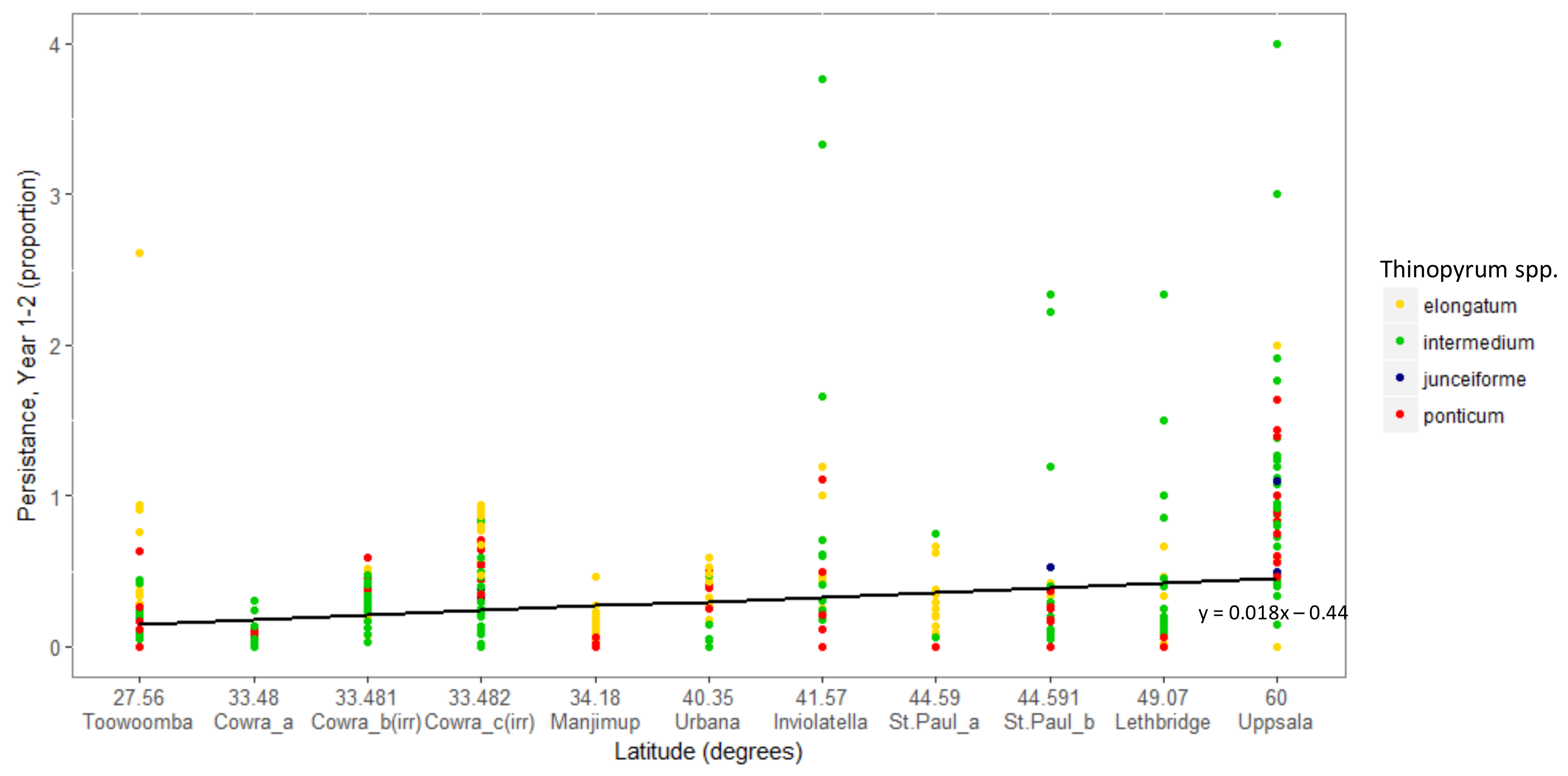
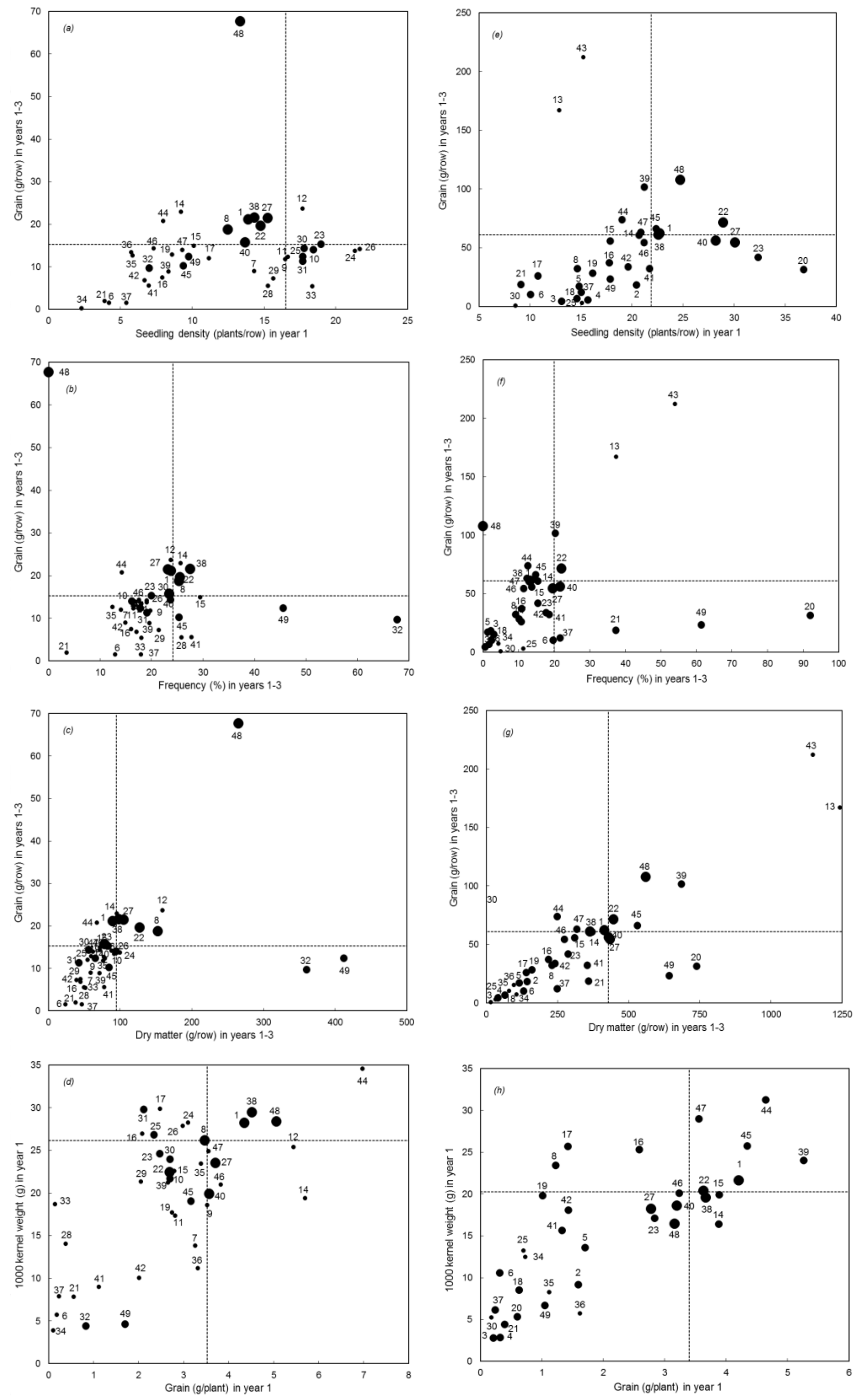
3. Results
Overall Performance
4. Discussion
4.1. Wheat Derivatives
4.2. Rye Derivatives
4.3. Barley Derivatives
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Wagoner, P. Perennial grain development: Past efforts and potential for the future. Crit. Rev. Plant Sci. 1990, 9, 381–408. [Google Scholar] [CrossRef]
- Curwen-McAdams, C.; Jones, S.S. Breeding Perennial Grain Crops Based on Wheat. Crop Sci. 2017, 57, 1172–1188. [Google Scholar] [CrossRef]
- Tsitsin, N.V. The significance of wide hybridization in the evolution and production of new species and forms of plants and animals. In Wide Hybridization in Plants; Tsitsin, N.V., Ed.; Isreal Program for Science Translations: Jerusalem, Isreal, 1960. [Google Scholar]
- Sharma, H.C.; Gill, B.S. Current status of wide hybridization in wheat. Euphytica 1983, 32, 17–31. [Google Scholar] [CrossRef]
- Cox, T.S.; Van Tassel, D.L.; Cox, C.M.; DeHaan, L.R. Progress in breeding perennial grains. Crop Pasture Sci. 2010, 61, 513–521. [Google Scholar] [CrossRef]
- DeHaan, L.R.; Wang, S.; Larsen, S.R.; Cattani, D.J.; Zhang, X.; Kantarski, T. Current efforts to develop perennial wheat and domesticate Thinopyrum intermedium as a perennial grain. In Proceedings of the Perennial Crops for Food Security FAO Expert Workshop, Rome, Italy, 28–30 August 2014. [Google Scholar]
- Zhang, X.; Larsen, S.R.; Gao, L.; Teh, S.L.; DeHaan, L.R.; Fraser, M.; Sallam, A.; Kantarski, T.; Frels, K.; Poland, J.; et al. Uncovering the genetic architecture of seed weight and size in intermediate wheatgrass through linkage and association mapping. Plant Genome 2017, 10. [Google Scholar] [CrossRef] [PubMed]
- Reimann-Philipp, R. Breeding perennial rye. Plant Breed. Rev. 1995, 13, 265–292. [Google Scholar]
- Larkin, P.J.; Newell, M.T.; Hayes, R.C.; Aktar, J.; Norton, M.R.; Moroni, S.J.; Wade, L.J. Progress in developing perennial wheats for grain and grazing. Crop Pasture Sci. 2014, 65, 1147–1164. [Google Scholar] [CrossRef]
- Jaikumar, N.S.; Snapp, S.S.; Murphy, K.; Jones, S.S. Agronomic Assessment of perennial wheat and perennial rye as cereal crops. Agron. J. 2012, 104, 1716–1726. [Google Scholar] [CrossRef]
- Hayes, R.C.; Newell, M.T.; Crews, T.E.; Peoples, M.B. Perennial cereal crops: An initial evaluation of wheat derivatives grown in mixtures with a regenerating legume. Renew. Agric. Food Syst. 2016, 32, 276–290. [Google Scholar] [CrossRef]
- Zhang, D.; Bowden, R.L.; Yu, J.; Carver, B.F.; Bai, G. Association Analysis of Stem Rust Resistance in U.S. Winter Wheat. PLoS ONE 2014, 9, e103747. [Google Scholar] [CrossRef]
- Banks, P.M.; Xu, S.J.; Wang, R.R.C.; Larkin, P.J. Varying chromosome composition of 56-chromosome wheat x Thinopyrum intermedium partial amphiploids. Genome 1993, 36, 207–215. [Google Scholar] [CrossRef] [PubMed]
- Jauhar, P.P.; Peterson, T.S.; Xu, S.S. Cytogenetic and molecular characterization of a durum alien disomic addition line with enhanced tolerance to Fusarium head blight. Genome 2009, 52, 467–483. [Google Scholar] [CrossRef] [PubMed]
- Larter, E.N.; Elliot, F.C. An evaluation of different ionizing radiations for possible use in the genetic transfer of bunt resistance from Agropyron to wheat. Can. J. Bot. 1956, 34, 817–823. [Google Scholar] [CrossRef]
- Murphy, K.M.; Lyon, S.R.; Balow, K.A.; Jones, S.S. Post-sexual cycle regrowth and grain yield in Thinopyrum elongatum × Triticum aestivum amphiploids. Plant Breed. 2010, 129, 480–483. [Google Scholar] [CrossRef]
- Hayes, R.C.; Newell, M.T.; DeHaan, L.R.; Murphy, K.M.; Crane, S.; Norton, M.R.; Wade, L.J.; Newberry, M.; Fahim, M.; Jones, S.S.; et al. Perennial cereal crops: An initial evaluation of wheat derivatives. Field Crops Res. 2012, 133, 68–89. [Google Scholar] [CrossRef]
- Fasoula, V.A.; Fasoula, D.A. Honeycomb breeding: Principles and applications. In Plant Breeding Reviews; Janick, J., Ed.; John Wiley & Sons Inc.: Hoboken, NJ, USA, 2000. [Google Scholar]
- WRB. World Reference Base for Soil Resources 2014, Update 2015. International Soil Classification System for Naming Soils and Creating Legends for Soil Maps; World Soil Resources Reports No. 106; Food and Agriculture Organization of the United Nations: Rome, Italy, 2015. [Google Scholar]
- Newell, M.T.; Hayes, R.C. An initial investigation of forage production and feed quality of perennial wheat derivatives. Crop Pasture Sci. 2017, 68, 1141–1148. [Google Scholar] [CrossRef]
- Lodge, G.; Gleeson, A. A comparison of methods of estimating lucerne population for monitoring persistence. Aust. J. Exp. Agric. 1984, 24, 174–177. [Google Scholar] [CrossRef]
- Fox, J.; Weisburg, S. An {R} Companion to Applied Regression, 2nd ed.; Sage: Thousand Oaks, CA, USA, 2011. [Google Scholar]
- Hothorn, T.; Hornik, K.; Zeileis, A. Unbiased Recursive Partitioning: A Conditional Inference Framework. J. Comput. Graph. Stat. 2006, 15, 651–674. [Google Scholar] [CrossRef]
- VSN International. Genstat for Windows, 19th ed.; VSN International: Hemel Hempstead, UK, 2018. [Google Scholar]
- Smith, A.B.; Cullis, B.R.; Gilmour, A.R. The analysis of crop variety evaluation data in Australia. Aust. N. Z. J. Stat. 2001, 43, 129–145. [Google Scholar] [CrossRef]
- Gilmour, A.R.; Gogel, B.J.; Cullis, B.R.; Thompson, R. ASReml User Guide, Release 3; VSN International: Hemel Hempstead, UK, 2009. [Google Scholar]
- Tewolde, H.; Fernandez, C.J.; Erickson, C.A. Wheat Cultivars Adapted to Post-Heading High Temperature Stress. J. Agron. Crop Sci. 2006, 192, 111–120. [Google Scholar] [CrossRef]
- Li, G.D.; Lodge, G.M.; Moore, G.A.; Craig, A.D.; Dear, B.S.; Boschma, S.P.; Albertson, T.O.; Miller, S.M.; Harden, S.; Hayes, R.C.; et al. Evaluation of perennial pasture legumes and herbs to identify species with high herbage production and persistence in mixed farming zones in southern Australia. Aust. J. Exp. Agric. 2008, 48, 449–466. [Google Scholar] [CrossRef]
- Gazza, L.; Galassi, E.; Ciccoritti, R.; Cacciatori, P.; Pogna, N.E. Qualitative traits of perennial wheat lines derived from different Thinopyrum species. Genet. Res. Crop Evol. 2016, 63, 209–219. [Google Scholar] [CrossRef]
- Fedak, G.; Chen, Q.; Conner, R.L.; Laroche, A.; Petroski, R.; Armstrong, K.W. Characterization of wheat-Thinopyrum partial amphiploids by meiotic analysis and genomic in situ hybridization. Genome 2000, 43, 712–719. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q. Detection of alien chromatin introgression from Thinopyrum into wheat using S genomic DNA as a probe—A landmark approach for Thinopyrum genome research. Cytogenet. Genome Res. 2005, 109, 350–359. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Ahmad, F.; Collin, J.; Comeau, A.; Fedak, G.; St-Pierre, C.; Chen, Q. Genomic constitution of a partial amphiploid OK7211542 used as a source of immunity to barley yellow dwarf virus for bread wheat. Plant Breed. 1998, 117, 1–6. [Google Scholar] [CrossRef]
- Wang, R.R.C. Agropyron and Psathyrostachys. In Wild Crop Relatives: Genomic and Breeding Resources; Springer: Berlin/Heidelberg, Germany, 2011; pp. 77–108. [Google Scholar]
- USDA. Grass Varieties in the United States; Alderson, J.A.S.W.C., Ed.; CRC-Lewis Publishers: New York, NY, USA, 1994.
- Norton, M.R.; Lelièvre, F.; Volaire, F. Summer dormancy in Dactylis glomerata L.: The influence of season of sowing and a simulated mid-summer storm on two contrasting cultivars. Aust. J. Agric. Res. 2006, 57, 565–575. [Google Scholar] [CrossRef]
- Oram, R.N. Register of Australian Herbage Plant Cultivars, 3rd ed.; CSIRO Division of Plant Industries: Canberra, Australia, 1990. [Google Scholar]
- Hoen, K. The effect of plant size and developmental stage on summer survival of some perennial grasses. Aust. J. Exp. Agric. Anim. Husb. 1968, 8, 190–196. [Google Scholar] [CrossRef]
- Sandve, S.R.; Kosmal, A.; Rudi, H.; Fjellheim, S.; Rapacz, M.; Yamada, T.; Rognli, O.A. Molecular mechanisms underlying frost tolerance in perennial grasses adapted to cold climates. Plant Sci. 2011, 180, 69–77. [Google Scholar] [CrossRef] [PubMed]
- Egron, A. Optimal Regulation of the Balance between Productivity and Overwintering of Perennial Grasses in a Warmer Climate. Agronomy 2017, 7, 19. [Google Scholar] [CrossRef]
- Dalmannsdottir, S.; Jørgensen, M.; Rapacz, M.; Østrem, L.; Larsen, A.; Rødven, R.; Rognli, O.A. Cold acclimation in warmer extended autumns impairs freezing tolerance of perennial ryegrass (Lolium perenne) and timothy (Phleum pratense). Physiol. Plant. 2017, 160, 266–281. [Google Scholar] [CrossRef] [PubMed]
- Newell, M.T.; Hayes, R.C.; Virgona, J.M.; Larkin, P.J. Summer dormancy in Elymus scaber and its hybridity with wheat. Euphytica 2015, 204, 535–556. [Google Scholar] [CrossRef]
- Oram, R.N. Secale montanum—A wider role in Australasia? N. Z. J. Agric. Res. 1996, 39, 629–633. [Google Scholar] [CrossRef]
- Lawrence, T. Orbit tall wheatgrass. Can. J. Plant Sci. 1967, 47, 611–612. [Google Scholar] [CrossRef]
- Schulz-Schaeffer, J.; Haller, S.E. Registration of Montana-2 Perennial × Agrotriticum Intermediodurum Khizhnyak. Crop Sci. 1987, 22, 822. [Google Scholar] [CrossRef]
- Schulz-Schaeffer, J.; Friebe, B. Karyological characterization of a partial amphiploid, Triticum turgidum L. var. durum Agropyron intermedium (Host) P.B. Euphytica 1992, 62, 83–88. [Google Scholar] [CrossRef]
- Jones, T.; Zhang, X.; Wang, R. Genome characterization of MT-2 perennial and OK-906 annual wheat × intermediate wheatgrass hybrids. Crop Sci. 1999, 39, 1041–1043. [Google Scholar] [CrossRef]
- Monneveux, P.; Jing, R.; Misra, S.C. Phenotyping for drought tolerance in wheat using physiological traits. Front. Physiol. 2012, 3, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Angus, J.F.; Kirkegaard, J.A.; Hunt, J.R.; Ryan, M.H.; Ohlander, L.; Peoples, M.B. Break crops and rotations for wheat. Crop Pasture Sci. 2015, 66, 523–552. [Google Scholar] [CrossRef]
- Acharya, S.N.; Mir, Z.; Moyer, J.R. ACE-1 perennial cereal rye. Can. J. Plant Sci. 2004, 84, 819–821. [Google Scholar] [CrossRef]
- Reimann-Philipp, R. Perennial spring rye as a crop alternative. J. Agron. Crop Sci. 1986, 157, 281–285. [Google Scholar] [CrossRef]
- Miedaner, T.; Longin, F. St. John’s Rye-Definitely too good for Goats and Sheep. In Neglected Cereals: From Ancient Grains to Superfood; Erling Verlag GmbH & Co. KG. EU: Clenze, Germany, 2017. [Google Scholar]
- Westerbergh, A.; Lerceteau-Köhler, E.; Sameri, M.; Bedada, G.; Lundquist, P. Towards development of perennial barley for cold temperate climates—Implications from wild barley relatives and barley-Hordeum bulbosum introgression lines. Sustainability 2018. under review. [Google Scholar]
- Pickering, R. The production of fertile triploid hybrids between Hordeum vulgare L. (2n = 2x = 14) and H. bulbosum L. (2n = 4x = 28). Barley Genet. Newsl. 1988, 18, 25–29. [Google Scholar] [CrossRef]
- Johnston, P.A.; Timmerman-Vaughan, G.M.; Farnden, K.J.F.; Pickering, R. Marker development and characterisation of Hordeum bulbosum introgression lines: A resource for barley improvement. Theor. Appl. Genet. 2009, 118, 1429–1437. [Google Scholar] [CrossRef] [PubMed]
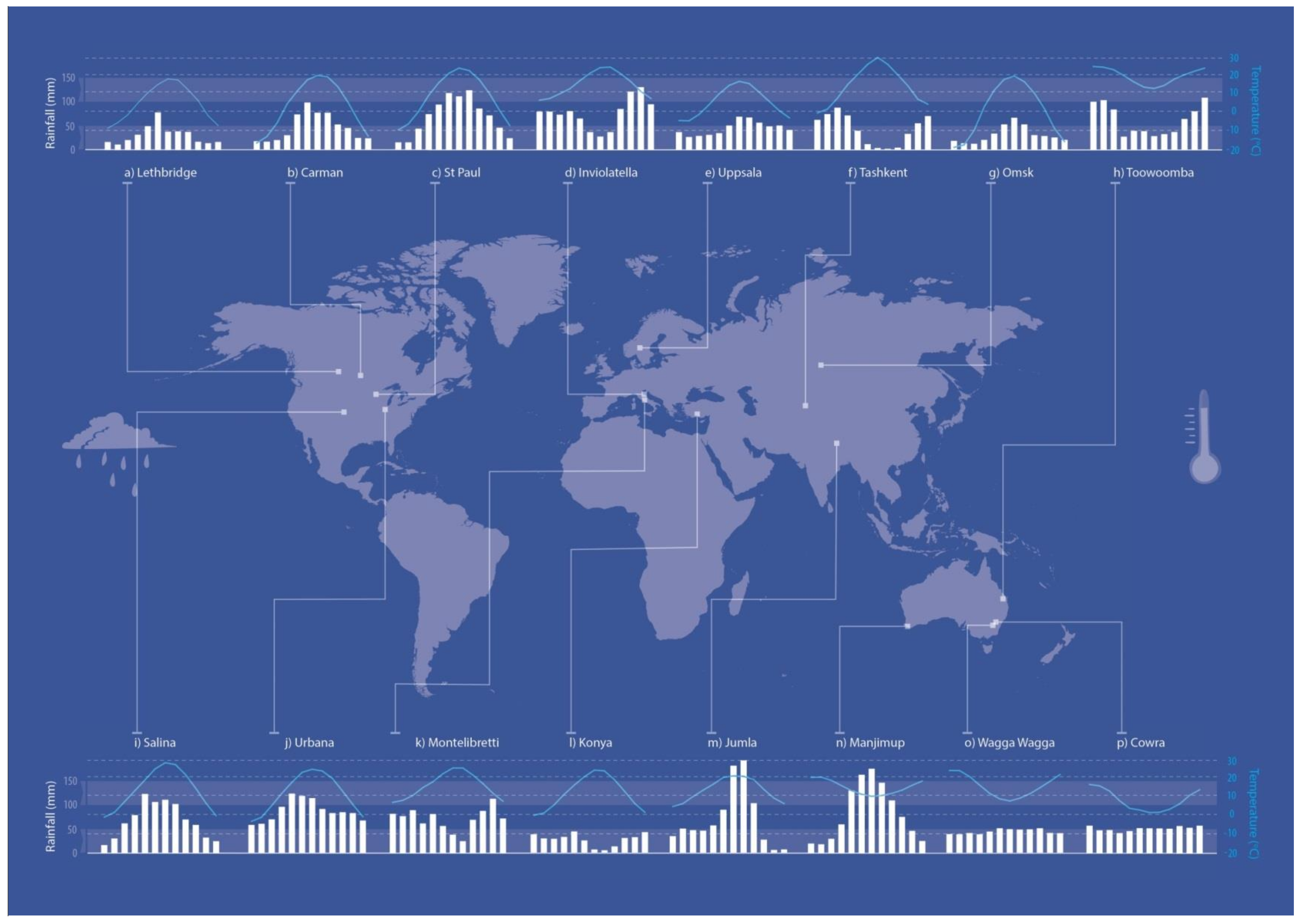
| Country | Site ID | Site | Latitude | Longitude | Koppen Classification | Elevation (m) | Planting Date | Soil Type § | Accumulated RF 12mths from Sowing (mm) | LAR (mm) | LAT (°C) | Historical Record | Soil pH Water | ||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mild winter environment | Year 1 | Year 2 | Year 3 | ||||||||||||
| Australia | 1 | Cowra A | 33°48.21’ S | 148°42.23’ E | Csc | 385 | 27/04/2012 | Lixisol | 414 | 513 | 442 | 623 | 16.2 | 1961–1990 | 6.1 |
| Australia | 2 | Cowra B | 33°48.21’ S | 148°42.23’ E | Csc | 385 | 25/04/2013 | Lixisol | 513 | 442 | - | 623 | 16.2 | 1961–1990 | 6.1 |
| Australia | 3 | Cowra C | 33°48.21’ S | 148°42.23’ E | Csc | 385 | 11/04/2016 | Lixisol | 858 | 613 | - | 623 | 16.2 | 1961–1990 | 6.1 |
| Australia | 4 | Manjimup | 34°18.38’ S | 116°07.60’ E | Csb | 262 | 14/06/2012 | Luvisol | 871 | 946 | - | 994 | 15.1 | 1915–2017 | 5.1 |
| Australia | 5 | Toowoomba | 27°56’ S | 151°95’ E | Csc | 640 | 15/05/2012 | Nitisol | 1145 | 606 | - | 722 | 17.9 | 1996–2017 | 6.1 |
| Australia | 6 | Wagga Wagga | 35°05’ S | 147°35’ E | Csc | 220 | 13/06/2012 | Ferralsol | 293 | 417 | - | 530 | 15.8 | 1898–2016 | 5.2 |
| Italy | 7 | Montelibretti | 42°08’ N | 12°44’ E | Csa | 20 | 30/12/2011 | Arenosol | 694 | 1069 | 1102 | 848 | 15.9 | 2005–2016 | 6.3 |
| Italy | 8 | Inviolatella | 41°57’ N | 12°47’ E | Csa | 20 | 20/11/2015 | Arenosol | 766 | 277 | - | 902 | 14.7 | 1973–2016 | 6.6 |
| Turkey | 9 | Konya | 37°51’ N | 32°33’ E | BSk | 1011 | 10/11/2015 | Xerosol | 243 | 264 | - | 322 | 11.6 | 1929–2016 | 8.0 |
| Cold winter environment | |||||||||||||||
| Nepal | 10 | Jumla | 29°17’ N | 82°10’ E | Cwb | 2300 | 12/10/2011 | Inceptisol | 763 | - | - | 794 | 13.2 | 2000–2015 | 8.1 |
| United States | 11 | Urbana | 40°3’50” N | 88°12’18” W | Cfa | 230 | 17/10/2012 | Mollic Gleysol | 879 | 962 | - | 1032 | 11.4 | 1989–2016 | 6.3 |
| United States | 12 | Salina A | 38°49’ N | 97°36’ W | Cfa | 373 | 5/10/2012 | Kastanozem | 750 | 716 | - | 800 | 13.4 | 1981–2010 | 6.7 |
| United States | 13 | Salina B | 38°49’ N | 97°36’ W | Cfa | 373 | 5/10/2012 | Kastanozem | 750 | 716 | - | - | - | - | 7.8 |
| United States | 14 | Salina C | 38°49’ N | 97°36’ W | Cfa | 373 | 10/12/2015 | Kastanozem | 767 | - | - | - | - | - | 7.8 |
| United States | 15 | St. Paul A | 44°52’ N | 93°13’ W | Dfa | 266 | 18/09/2012 | Phaeozem | 842 | 959 | - | 817 | 8.3 | 1981–2010 | 6.6 |
| United States | 16 | St Paul B | 44°52’ N | 93°13’ W | Dfa | 266 | 9/22/2015 | Phaeozem | 1051 | 963 | - | 820 | 8.3 | 1981–2010 | 6.9 |
| Canada | 17 | Lethbridge | 49°07’ N | 112°08’ W | BSk | 911 | 2/10/2015 | Chernozem | 302 | 245 | - | 379 | 5.6 | 1961–2016 | 7.6 |
| Canada | 18 | Carman | 49°30’ N | 98°13’ W | Dfb | 267 | 23/09/2015 | Chernozem | 568 | 364 | 548 | 3.4 | 1981–2010 | 6.3 | |
| Sweden | 19 | Uppsala | 60°00’ N | 17°42’ E | Cfb | 24 | 01/10/2015 | Cambisol | 609 | 542 | - | 527 | 5.7 | 1961–2016 | 6.6 |
| Uzbekistan | 20 | Tashkent † | 41°15′52″ N | 69°12′58″ E | Csa | 470 | 20/10/2016 | Serozem | 533 | - | - | 519 | 14.4 | 1923–2015 | 7.0 |
| Russia | 21 | Omsk † | 54°59′32″ N | 73°22’06″ E | Dfb | 87 | 19/05/2017 | Chernozem | 171 ‡ | - | - | 381 | 1.4 | 1948–2010 | 7.0 |
| ID | Genotype | Pedigree | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | 19 | 20 | 21 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 11955 | Triticum aestivum × Thinopyron ponticum | • | • | • | • | • | • | • | • | • | • | • | • | • | • | • | • | • | • | • | • | • |
| 2 | 12F3205 | T. aestivum × Th. intermedium | • | • | • | • | |||||||||||||||||
| 3 | 12F3256 | T. aestivum × Th. ponticum | • | • | • | • | |||||||||||||||||
| 4 | 12F3258 | T. aestivum × Th. ponticum | • | • | • | • | |||||||||||||||||
| 5 | 12F4090 | T. aestivum × Th. intermedium | • | • | • | • | |||||||||||||||||
| 6 | 12G401 F2 | T. turgidum × Th. intermedium | • | • | • | • | • | • | • | ||||||||||||||
| 7 | 14894 | T. aestivum × Secale cereale × Agropyron elongatum | • | • | • | ||||||||||||||||||
| 8 | 20238 | T. turgidum × Ag. elongatum | • | • | • | • | • | • | • | • | • | • | • | • | • | • | • | ||||||
| 9 | 4014 | Ag. derivative | • | • | • | ||||||||||||||||||
| 10 | 6754 | T. aestivum × Ag. elongatum | • | • | • | • | • | ||||||||||||||||
| 11 | 6755 | T. aestivum × Ag. elongatum | • | • | • | ||||||||||||||||||
| 12 | 6770 | T. aestivum × Ag. elongatum | • | • | • | ||||||||||||||||||
| 13 | ACE-1 | S. cereale | • | ||||||||||||||||||||
| 14 | Agrotana | T. aestivum × Th. ponticum | • | • | • | • | • | • | • | • | • | • | |||||||||||
| 15 | B1126 | T. aestivum × Th. intermedium | • | • | • | • | • | • | • | • | |||||||||||||
| 16 | B1321 | T. aestivum × Th. intermedium | • | • | • | • | • | • | • | • | • | • | |||||||||||
| 17 | B373 | T. aestivum × Th. intermedium | • | • | • | • | • | • | • | • | • | • | |||||||||||
| 18 | B374-6-7s | T. aestivum × Th. intermedium | • | • | • | • | |||||||||||||||||
| 19 | B913 | T. aestivum × Th. junceiforme | • | • | • | • | • | • | • | • | • | • | |||||||||||
| 20 | C3-2627 | Th. intermedium | • | • | • | • | |||||||||||||||||
| 21 | C3-3471 | Th. intermedium | • | • | • | • | • | • | • | • | • | ||||||||||||
| 22 | CPI147235a | T. aestivum × Th. ponticum × T. aestivum | • | • | • | • | • | • | • | • | • | • | • | • | • | • | • | • | • | • | • | ||
| 23 | CPI147236a | T. aestivum × Th. ponticum × T. aestivum | • | • | • | • | • | • | • | • | • | • | • | ||||||||||
| 24 | CPI147242b | T. aestivum × Th. ponticum × T. aestivum | • | • | • | ||||||||||||||||||
| 25 | CPI147244b | T. aestivum × Th. ponticum × T. aestivum | • | • | • | • | • | • | • | • | • | • | • | ||||||||||
| 26 | CPI147247a | T. aestivum × Th. ponticum × T. aestivum | • | • | • | ||||||||||||||||||
| 27 | CPI147251b | T. aestivum × Th. ponticum × T. aestivum | • | • | • | • | • | • | • | • | • | • | • | • | • | • | • | • | • | • | • | • | • |
| 28 | CPI147257b | T. carthlicum × Th. intermedium × T. aestivum | • | • | |||||||||||||||||||
| 29 | CPI147279a | T. carthlicum × Th. intermedium × T. aestivum | • | • | |||||||||||||||||||
| 30 | CPI147280b | T. aestivum × Th. intermedium | • | • | • | • | • | • | • | • | • | • | |||||||||||
| 31 | CPI147281b | T. aestivum × Th. intermedium | • | • | • | • | • | • | |||||||||||||||
| 32 | CPI148055 | Th. intermedium | • | • | • | • | • | • | • | ||||||||||||||
| 33 | IL107 | Hordeum vulgare × H. bulbosum | • | ||||||||||||||||||||
| 34 | IL118 | H. vulgare × H. bulbosum | • | • | • | ||||||||||||||||||
| 35 | IL24 | H. vulgare × H. bulbosum | • | • | • | • | • | ||||||||||||||||
| 36 | IL46 | H. vulgare × H. bulbosum | • | • | • | • | |||||||||||||||||
| 37 | Mt-2 | T. turgidum × Th. intermedium | • | • | • | • | • | • | • | ||||||||||||||
| 38 | OK7211542 | T. aestivum × Th. ponticum | • | • | • | • | • | • | • | • | • | • | • | • | • | • | • | • | • | • | • | • | |
| 39 | Ostankinskaya | T. aestivum × Th. intermedium | • | • | • | • | • | • | • | • | |||||||||||||
| 40 | Otrastajuscaja 38 | T. aestivum × Th. intermedium | • | • | • | • | • | • | • | • | • | • | • | • | • | • | • | • | • | • | |||
| 41 | P15 | T. aestivum × Th. ponticum × T. aestivum | • | • | • | • | • | • | • | • | |||||||||||||
| 42 | PWM706/PWM3 | T. aestivum × Th. ponticum | • | • | • | • | • | • | • | • | |||||||||||||
| 43 | Reimann-Phillip | S. cereale | • | ||||||||||||||||||||
| 44 | Summer 1 | T. aestivum × Th. intermedium | • | • | • | • | • | • | • | • | • | ||||||||||||
| 45 | TAF 46 | T. aestivum × Th. intermedium | • | • | • | • | • | • | • | • | • | • | |||||||||||
| 46 | Zhong 3 | T. aestivum × Th. intermedium | • | • | • | • | • | • | • | • | |||||||||||||
| 47 | Zhong 7 | T. aestivum × Th. intermedium | • | • | • | • | • | • | • | • | • | ||||||||||||
| 48 | Crop control | ||||||||||||||||||||||
| Arapahoe | T. aestivum | • | • | ||||||||||||||||||||
| Bezostaya1 | T. aestivum | • | |||||||||||||||||||||
| Ensco | T. aestivum | • | • | ||||||||||||||||||||
| Everest | T. aestivum | • | |||||||||||||||||||||
| Falcon | T. aestivum | • | |||||||||||||||||||||
| Fuller | T. aestivum | • | • | ||||||||||||||||||||
| Gregory | T. aestivum | • | |||||||||||||||||||||
| Mace | T. aestivum | • | |||||||||||||||||||||
| Pioneer_25R47 | T. aestivum | • | |||||||||||||||||||||
| Stava | T. aestivum | • | |||||||||||||||||||||
| TAM_SOFT700 | T. aestivum | • | |||||||||||||||||||||
| UKR-OD 952.92/AE.1 Squarrosa (1031) | T. aestivum | • | |||||||||||||||||||||
| W512 | T. aestivum | • | |||||||||||||||||||||
| Wedgetail | T. aestivum | • | • | • | • | • | • | • | • | • | • | • | • | ||||||||||
| WK1204 | T. aestivum | • | |||||||||||||||||||||
| Hindmarsh | H. vulgare | • | |||||||||||||||||||||
| P-845 | H. vulgare | • | |||||||||||||||||||||
| 49 | Grass control | ||||||||||||||||||||||
| Accession (Turkey) | H. bulbosum | • | |||||||||||||||||||||
| Barcel | Festuca arundinacea | • | |||||||||||||||||||||
| C3-3864 | Th. intermedium | • | • | ||||||||||||||||||||
| Dundas | Th. ponticum | • | • | • | • | • | • | • | |||||||||||||||
| Family10 | S. montanum | • | • | • | • | • | • | • | • | • | • | ||||||||||||
| Local variety (Minnesota) | Th. intermedium | • | |||||||||||||||||||||
| Local variety (Turkey) | Th. intermedium | • | |||||||||||||||||||||
| TLI_C3-2925 | Th. intermedium | • | |||||||||||||||||||||
| Total | 24 | 24 | 24 | 16 | 16 | 16 | 15 | 22 | 27 | 15 | 18 | 18 | 18 | 27 | 18 | 22 | 24 | 22 | 25 | 12 | 12 |
| Site | Year 1 | Year 2 | ||||
|---|---|---|---|---|---|---|
| Pesticides | Fertilizers | Irrigation | Pesticides | Fertilizers | Irrigation | |
| Cowra A | 250 mL/ha Propiconazole (fungicide) | 18 kg/ha N, 20 kg/ha P | Nil | Nil | 18 kg/ha N, 20 kg/ha P | Nil |
| Cowra B | 250 mL/ha Propiconazole (fungicide) | 18 kg/ha N, 20 kg/ha P | Summer irrigation | Nil | 18 kg/ha N, 20 kg/ha P | Summer irrigation |
| Cowra C | Nil | 18 kg/ha N, 20 kg/ha P | at establishment & summer | Nil | 18 kg/ha N, 20 kg/ha P | Summer irrigation |
| Manjimup | 12.5 g/ha bifenthrin (insecticide); 129 g/ha tebuconazole (fungicide) | 80 kg/ha N, 22 kg/ha P, 60 kg/ha K | Nil | Nil | 37 kg/ha N, 6 k g/ha P, 22 kg/ha K | Nil |
| Toowoomba | Nil | 18 kg/ha N, 20 kg/ha P | at establishment | Nil | 18 kg/ha N, 20 kg/ha P | Nil |
| Wagga Wagga | Nil | 26 kg/ha N, 28 kg/ha P | Summer & autumn irrigation | Nil | 26 kg/ha N, 28 kg/ha P | Summer & autumn irrigation |
| Montelibretti | Nil | 31 kg/ha N, 20 kg/ha P | Summer irrigation | Nil | 31 kg/ha N, 20 kg/ha P | Nil |
| Inviolatella | Nil | 31 kg/ha N, 20 kg/ha P | Nil | Nil | 31 kg/ha N, 20 kg/ha P | Nil |
| Konya | Nil | 30 kg/ha N | Nil | Nil | Nil | Nil |
| Jumla | Nil | 60 kg/ha N, 30 kg/ha P, 30 kg/ha K | at establishment | - | - | - |
| Urbana | Nil | 30 kg/ha N; 20 kg/ha P | Nil | 175 kg/ha Pendamethalin (herbicide) | Nil | Nil |
| Salina A | Nil | 34 kg/ha N | at establishment & summer | Nil | Nil | Nil |
| Salina B | Nil | 34 kg/ha N | at establishment | - | - | - |
| Salina C | Nil | 34 kg/ha N | at establishment | Nil | Nil | Nil |
| St. Paul A | Nil | Nil | at establishment | Nil | Nil | Nil |
| St Paul B | establishment: 1.1 L/ha 2-4-D (herbicide); 145 mL/ha Tebuconazole (fungicide); 109 mL/ha lambda-cyhalothrin (insecticide); 2 L/ha Fenoxaprop-p-Ethyl, Pyrasulfotole, Bromoxynil Octanoate, Bromoxynil Heptanoate (herbicide) | Nil | Nil | Nil | 56 kg/ha N | Nil |
| Lethbridge | 500 mL/ha pyroxsulam | 87 kg/ha N, 58 kg/ha P, 24 kg/ha K | Spring & Summer (152.4 mm) | 0.007 kg/ha metsulfuron-methyl | 87 kg/ha N, 58 kg/ha P, 24 kg/ha K | Autumn, Spring & summer (292.1 mm) |
| Carman | Nil | Nil | Nil | Nil | Nil | Nil |
| Uppsala | Nil | 30 kg/ha N | Nil | Nil | Nil | Nil |
| Tashkent | Nil | 184 kg/ha N total applied 3 times | 1 after planting + 3 during the season (total 235 mm); 1 irrigation after harvest (70 mm) | - | - | - |
| Omsk | Nil | Nil | Nil | - | - | - |
| Planting Year | 2008 | 2009 | 2010 | 2011 | 2012 | 2013 | 2016 | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sampling Year | Yr. 1 | Yr. 2 | Yr. 3 | Yr. 4 | Yr. 1 | Yr. 2 | Yr. 1 | Yr. 2 | Yr. 1 | Yr. 2 | Yr. 1 | Yr. 2 | Yr. 3 | Yr. 1 | Yr. 2 | Yr. 1 | Yr. 2 | |
| ID | Genotype | |||||||||||||||||
| 1 | 11955 | - | - | - | - | - | - | 117.1 | 15.1 | 178.5 | 66 | 197.9 | 0 | 0 | 104.9 | 0 | 84.6 | 3.7 |
| 7 | 14894 | - | - | - | - | - | - | 68.1 | 0.3 | - | - | 97.6 | 0 | 0 | 46.6 | 0 | - | - |
| 8 | 20238 | - | - | - | - | - | - | 136.3 | 23.9 | 152.8 | 16.2 | 153.7 | 0 | 0 | 62.9 | 0 | 123.2 | 30.7 |
| 9 | 4014 | - | - | - | - | - | - | 108.8 | 4.6 | - | - | 193.7 | 0 | 0 | 10.2 | 0 | - | - |
| 10 | 6754 | - | - | - | - | - | - | 151.7 | 3.2 | - | - | 170.1 | 0 | 0 | 8.3 | 5 | - | - |
| 11 | 6755 | - | - | - | - | - | - | 175.6 | 3.5 | 253.6 | 53.2 | 240 | 0 | 0 | 6.5 | 0 | - | - |
| 12 | 6770 | - | - | - | - | - | - | 158.3 | 0.3 | - | - | 169.1 | 0 | 0 | 46 | 0 | - | - |
| 22 | CPI147235a | 73.5 | 25.9 | 0 | 0 | 33.7 | 120.1 | 82.6 | 4.4 | 102.1 | 58.3 | 107.4 | 0 | 0 | 44.4 | 1.9 | 60.2 | 43.5 |
| 23 | CPI147236a | 81.7 | 45.9 | 11.2 | 0 | 32.2 | 36.2 | 74.5 | 0.1 | 152.5 | 20.1 | 117 | 0 | 0 | 52.5 | 0 | - | - |
| 24 | CPI147242b | - | - | - | - | 24.4 | 9.8 | 47.3 | 3.2 | 107.3 | 13.1 | 128.2 | 0 | 0 | 69.7 | 1.5 | - | - |
| 25 | CPI147244b | - | - | - | - | 9.5 | 37.7 | 31.3 | 0 | 36.9 | 5.1 | 73.9 | 0 | 0 | 52.2 | 0 | - | - |
| 26 | CPI147247a | - | - | - | - | 40.5 | 0 | 62 | 6.3 | 145 | 0 | 137.4 | 0 | 0 | 53.7 | 0 | - | - |
| 27 | CPI147251b | 70.9 | 9.4 | 0.5 | 0.4 | 58.5 | 0 | 56.3 | 2.8 | 150.4 | 16.5 | 142.1 | 0 | 0 | 89.8 | 0 | 160.6 | 26.7 |
| 28 | CPI147257b | 7.8 | 12.6 | 4.7 | 0.7 | 2.6 | 4.3 | 0.2 | 0 | 13.9 | 4.1 | 11.4 | 3.8 | 0 | 2.9 | 2.4 | - | - |
| 29 | CPI147279a | - | - | - | - | 34.3 | 30.5 | - | - | - | - | 72.1 | 0 | 0 | 9.5 | 0 | - | - |
| 30 | CPI147280b | - | - | - | - | 21 | 31 | - | - | - | - | 99 | 3.3 | 0 | 28.9 | 0 | - | - |
| 31 | CPI147281b | - | - | - | - | 22.8 | 12.1 | - | - | - | - | 70.8 | 0 | 0 | 9.4 | 0 | - | - |
| 32 | CPI148055 | 0.4 | 3.6 | 57.7 | 0 | - | - | 81.7 | 0 | 13.5 | 43.5 | 4.9 | 55.2 | 2.8 | 16 | 2.1 | 7.7 | 26.7 |
| 38 | OK7211542 | 93.6 | 13.3 | 0 | 0 | 107 | 17.6 | 83.3 | 4.1 | 196.7 | 78.9 | 203.1 | 0 | 0 | 80.5 | 0 | 138.9 | 0 |
| 39 | Ostankinskaya | - | - | - | - | - | - | - | - | 101.8 | 54.8 | - | - | - | - | - | 54.3 | 1.2 |
| 40 | Otrastajuscaja 38 | 29.7 | 11.2 | 0 | 0 | 33.8 | 63.6 | 83.9 | 0 | - | - | 81 | 0 | 0 | 50.1 | 1.4 | 36 | 2.2 |
| 44 | Summer 1 | 128 | 0 | 0 | 0 | 217.1 | 0 | - | - | - | - | - | - | - | - | - | 105.1 | 0 |
| 45 | TAF46 | - | - | - | - | 1.5 | 43.8 | 20.6 | 0.4 | 43.5 | 57.9 | 49.8 | 7.6 | 0 | 42.5 | 0 | 47.1 | 0 |
| 48 | Wedgetail | 204.1 | 0 | 0 | 0 | 264.9 | 0 | 319.5 | 0 | 352.9 | 0 | 392.5 | 0 | 0 | 145.6 | 137.5 † | 263 | 137.4 † |
| 49 | Dundas | 0 | 9.4 | 207.7 | 268.9 | 6.4 | 128.5 | 20.7 | 0.6 | 18.4 | 72 | 9.6 | 101 | 8.4 | 17.9 | 6.2 | - | - |
| 49 | Family10 | 12.5 | 0.9 | 82 | 0 | 0.2 | 66.9 | 46.5 | 0.2 | - | - | 79.6 | 42.7 | 1 | 26.5 | 12.1 | - | - |
| ID | Genotype | Index | Rank | Crop Control | Grass Control | ID | Genotype | Index | Rank | Crop Control | Grass Control |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Mild winter | Cold winter | ||||||||||
| 38 | OK7211542 | 46.3 | 1 | 0.035 | <0.001 | 39 | Ostankinskaya | 53.1 | 1 | <0.001 | <0.001 |
| 1 | 11955 | 45.2 | 2 | 0.082 | <0.001 | 44 | Summer 1 | 52.2 | 2 | <0.001 | <0.001 |
| 12 | 6770 | 45.0 | 3 | 0.092 | <0.001 | 1 | 11955 | 46.9 | 3 | 0.008 | 0.003 |
| 24 | CPI147242b | 43.1 | 4 | 0.294 | 0.001 | 22 | CPI147235a | 46.5 | 4 | 0.010 | 0.005 |
| 44 | Summer 1 | 42.9 | 5 | 0.334 | 0.002 | 47 | Zhong 7 | 45.5 | 5 | 0.021 | 0.010 |
| 8 | 20238 | 42.3 | 6 | 0.439 | 0.003 | 45 | TAF 46 | 45.0 | 6 | 0.029 | 0.014 |
| 26 | CPI147247a | 42.2 | 7 | 0.467 | 0.004 | 38 | OK7211542 | 44.6 | 7 | 0.036 | 0.019 |
| 27 | CPI147251b | 40.4 | 8 | 0.894 | 0.020 | 20 | C3-2627 | 44.0 | 8 | 0.054 | 0.028 |
| 31 | CPI147281b | 40.1 | 9 | 0.987 | 0.026 | 40 | Otrastajuscaja 38 | 43.2 | 9 | 0.085 | 0.047 |
| 48 | Crop control | 40.0 | 10 | NA | 0.027 | 27 | CPI147251b | 41.0 | 10 | 0.247 | 0.153 |
| 22 | CPI147235a | 39.8 | 11 | 0.940 | 0.033 | 23 | CPI147236a | 38.2 | 11 | 0.686 | 0.496 |
| 23 | CPI147236a | 38.3 | 12 | 0.574 | 0.096 | 46 | Zhong 3 | 37.8 | 12 | 0.776 | 0.575 |
| 25 | CPI147244b | 38.3 | 13 | 0.570 | 0.097 | 15 | B1126 | 37.3 | 13 | 0.864 | 0.654 |
| 30 | CPI147280b | 37.6 | 14 | 0.416 | 0.156 | 16 | B1321 | 37.2 | 14 | 0.896 | 0.683 |
| 17 | B373 | 35.9 | 15 | 0.172 | 0.386 | 17 | B373 | 37.2 | 15 | 0.898 | 0.686 |
| 15 | B1126 | 35.9 | 16 | 0.163 | 0.402 | 48 | Crop control | 36.7 | 16 | NA | 0.782 |
| 45 | TAF 46 | 35.8 | 17 | 0.161 | 0.406 | 14 | Agrotana | 36.0 | 17 | 0.846 | 0.934 |
| 10 | 6754 | 35.6 | 18 | 0.141 | 0.446 | 49 | Grass control | 35.7 | 18 | 0.782 | NA |
| 47 | Zhong 7 | 35.4 | 19 | 0.125 | 0.485 | 42 | PWM706/PWM3 | 34.9 | 19 | 0.638 | 0.847 |
| 14 | Agrotana | 35.2 | 20 | 0.107 | 0.537 | 8 | 20238 | 34.4 | 20 | 0.543 | 0.740 |
| 32 | CPI148055 | 34.4 | 21 | 0.061 | 0.725 | 41 | P15 | 33.7 | 21 | 0.418 | 0.593 |
| 40 | Otrastajuscaja 38 | 34.3 | 22 | 0.055 | 0.762 | 19 | B913 | 31.3 | 22 | 0.147 | 0.239 |
| 35 | IL24 | 33.8 | 23 | 0.039 | 0.880 | 21 | C3-3471 | 30.0 | 23 | 0.074 | 0.129 |
| 49 | Grass control | 33.4 | 24 | 0.027 | NA | 25 | CPI147244b | 27.5 | 24 | 0.016 | 0.032 |
| 29 | CPI147279a | 33.3 | 25 | 0.025 | 0.978 | 5 | 12F4090 | 24.8 | 25 | 0.002 | 0.005 |
| 9 | 4014 | 32.5 | 26 | 0.013 | 0.762 | 34 | IL118 | 24.0 | 26 | 0.001 | 0.003 |
| 16 | B1321 | 32.2 | 27 | 0.010 | 0.694 | 6 | 12G401 F2 | 23.5 | 27 | <0.001 | 0.002 |
| 46 | Zhong 3 | 31.8 | 28 | 0.007 | 0.594 | 37 | Mt-2 | 22.1 | 28 | <0.001 | <0.001 |
| 11 | 6755 | 30.3 | 29 | 0.001 | 0.294 | 35 | IL24 | 20.1 | 29 | <0.001 | <0.001 |
| 39 | Ostankinskaya | 30.1 | 30 | 0.001 | 0.271 | 2 | 12F3205 | 20.1 | 30 | <0.001 | <0.001 |
| 19 | B913 | 28.7 | 31 | <0.001 | 0.117 | 36 | IL46 | 18.1 | 31 | <0.001 | <0.001 |
| 36 | IL46 | 28.4 | 32 | <0.001 | 0.098 | 18 | B374-6-7s | 14.9 | 32 | <0.001 | <0.001 |
| 28 | CPI147257b | 26.4 | 33 | <0.001 | 0.020 | 30 | CPI147280b | 12.0 | 33 | <0.001 | <0.001 |
| 7 | 14894 | 24.9 | 34 | <0.001 | 0.005 | 4 | 12F3258 | 7.4 | 34 | <0.001 | <0.001 |
| 41 | P15 | 24.6 | 35 | <0.001 | 0.004 | 3 | 12F3256 | 6.9 | 35 | <0.001 | <0.001 |
| 42 | PWM706/PWM3 | 22.3 | 36 | <0.001 | <0.001 | ||||||
| 37 | Mt-2 | 20.5 | 37 | <0.001 | <0.001 | ||||||
| 6 | 12G401 F2 | 15.4 | 38 | <0.001 | <0.001 | ||||||
| 21 | C3-3471 | 9.7 | 39 | <0.001 | <0.001 | ||||||
| l.s.d. | 5.87 | l.s.d. | 7.41 | ||||||||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hayes, R.C.; Wang, S.; Newell, M.T.; Turner, K.; Larsen, J.; Gazza, L.; Anderson, J.A.; Bell, L.W.; Cattani, D.J.; Frels, K.; et al. The Performance of Early-Generation Perennial Winter Cereals at 21 Sites across Four Continents. Sustainability 2018, 10, 1124. https://doi.org/10.3390/su10041124
Hayes RC, Wang S, Newell MT, Turner K, Larsen J, Gazza L, Anderson JA, Bell LW, Cattani DJ, Frels K, et al. The Performance of Early-Generation Perennial Winter Cereals at 21 Sites across Four Continents. Sustainability. 2018; 10(4):1124. https://doi.org/10.3390/su10041124
Chicago/Turabian StyleHayes, Richard C., Shuwen Wang, Matthew T. Newell, Kathryn Turner, Jamie Larsen, Laura Gazza, James A. Anderson, Lindsay W. Bell, Douglas J. Cattani, Katherine Frels, and et al. 2018. "The Performance of Early-Generation Perennial Winter Cereals at 21 Sites across Four Continents" Sustainability 10, no. 4: 1124. https://doi.org/10.3390/su10041124
APA StyleHayes, R. C., Wang, S., Newell, M. T., Turner, K., Larsen, J., Gazza, L., Anderson, J. A., Bell, L. W., Cattani, D. J., Frels, K., Galassi, E., Morgounov, A. I., Revell, C. K., Thapa, D. B., Sacks, E. J., Sameri, M., Wade, L. J., Westerbergh, A., Shamanin, V., ... Li, G. D. (2018). The Performance of Early-Generation Perennial Winter Cereals at 21 Sites across Four Continents. Sustainability, 10(4), 1124. https://doi.org/10.3390/su10041124







