Higher Sustainability and Lower Opportunistic Behaviour in Healthcare: A New Framework for Performing Hospital-Based Health Technology Assessment
Abstract
:1. Introduction
- An exploration of the literature background concerning technology adoption decision-making and the practices of HTA, with a particular focus on the hospital level;
- The development of a conceptual framework to support hospital-based technology management, aligning strategy with operations.
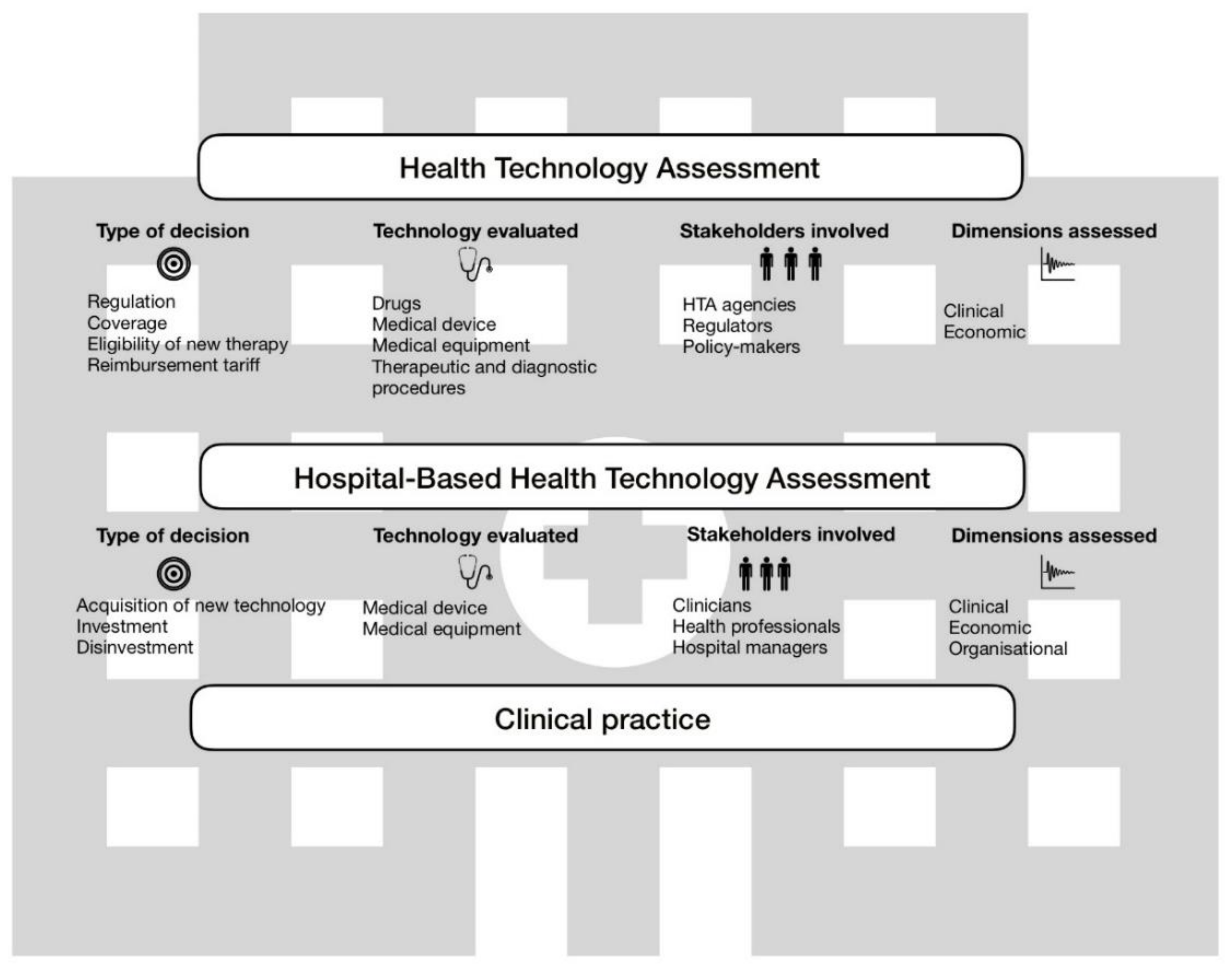
2. Health Technology Assessment across the National Health System
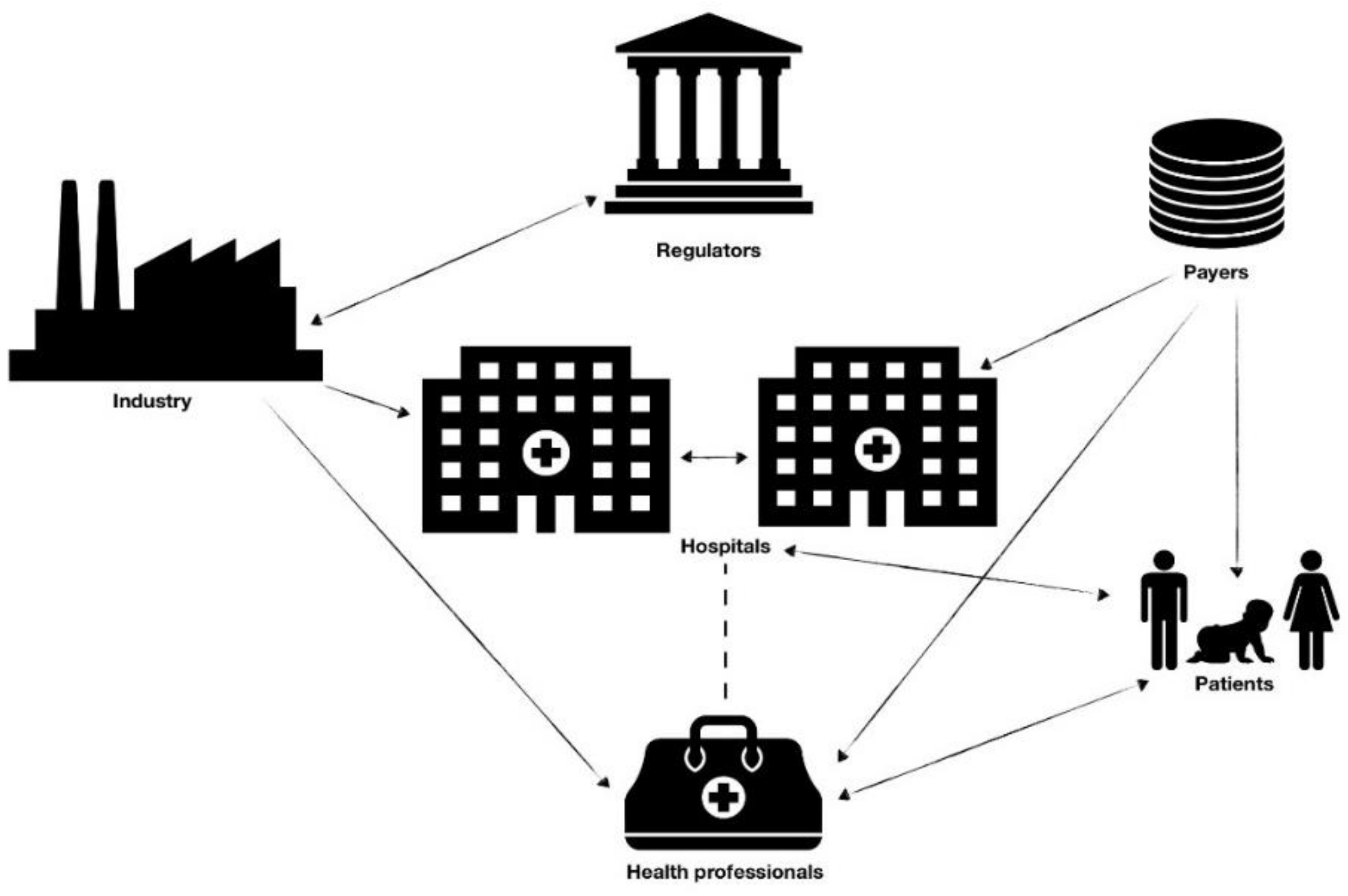
3. Opportunistic Behavior in Procurement of Health Technology
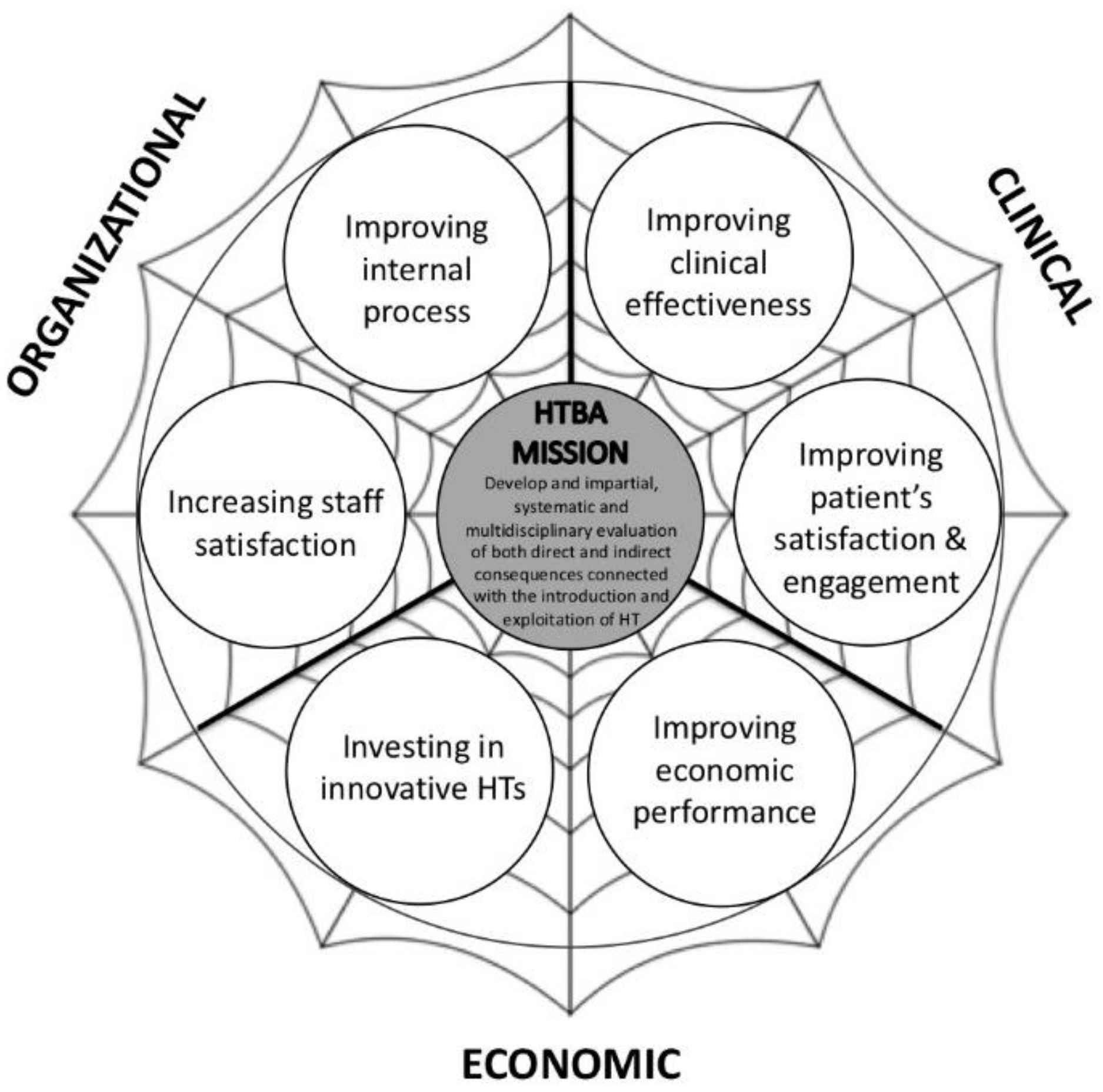
4. “Health Technology Balanced Assessment” Framework
- improving clinical effectiveness and increasing patients’ satisfaction and engagement from the clinical perspective;
- optimising employment of financial resources, maintaining (or improving) the same level of access and quality of health services from the economic perspective;
- improving the efficacy and effectiveness of internal processes and increasing staff satisfaction from the organisational perspective.
- the clinical perspective deals with medical and technical issues in order to assess if a technology represents an effective treatment of a specific disease and how it works. Moreover, it is aimed at capturing patients’ needs and implications, referring not only to their health status but also to their satisfaction and physiological sphere;
- the economic perspective involves all financial issues, presented by the investment effort, cost savings or potential additional revenues;
- the organisational perspective assesses which implications on current arrangements and procedures are by the adoption of new technology. In particular, it is performed to understand if (and which) organisational changes, in terms of roles and competencies, and training activities are required for a successful implementation.
- Collect the information needed for the HT evaluation based on different qualitative and quantitative scales;
- Aggregate this information set based on specific assessment requirements;
- Analyse the results gathered by the assessment process;
- Summarise the results of the assessment in order to make them comparable with the targeted expectations.
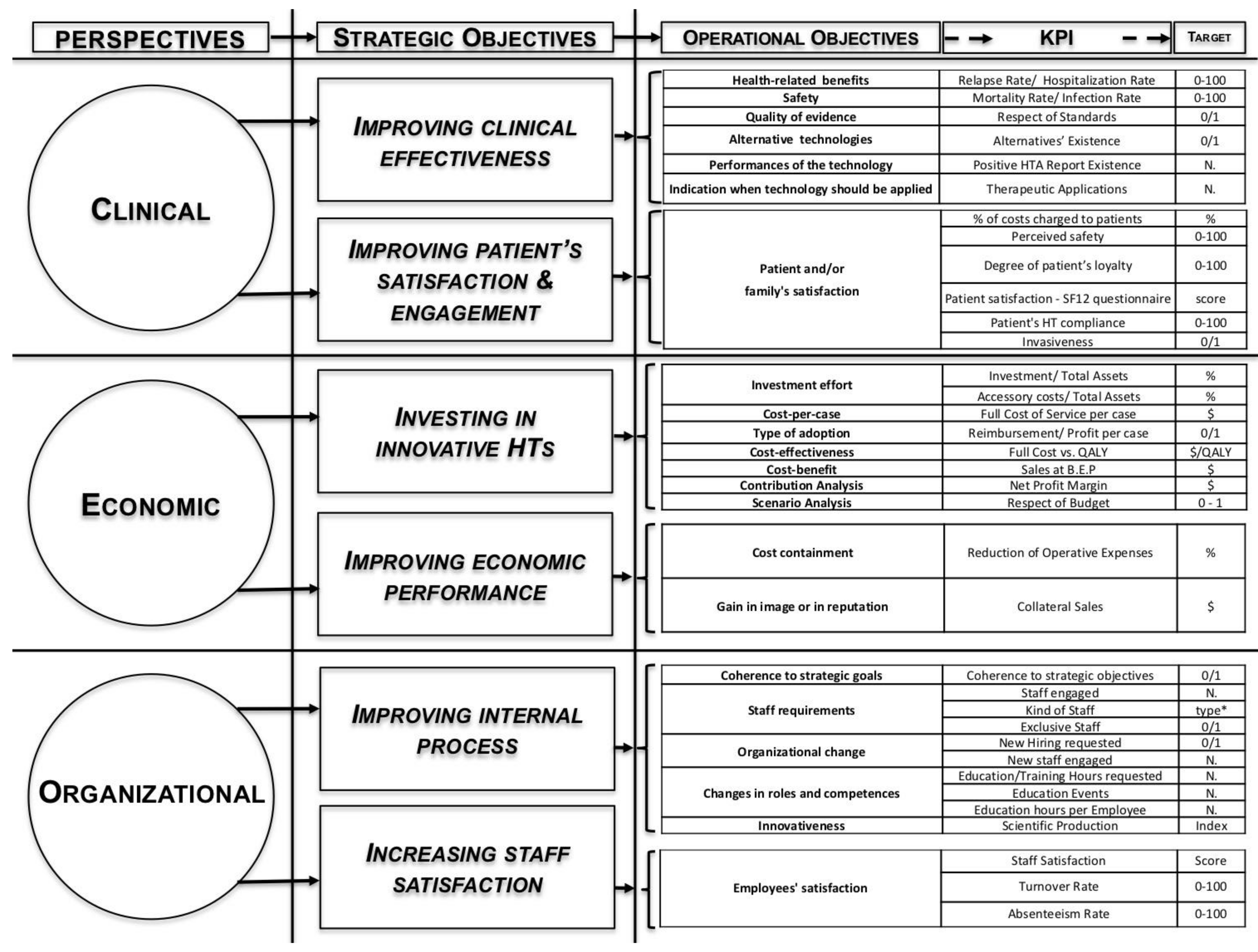
5. A Performance-Driven Hospital-Based HTA for Sustainable Development
- the consistency with overall strategy;
- the cause-effect relationship among different strategic and operating objectives;
- impacts on internal processes;
- the overall sustainability of hospitals taking into consideration the external environment, social needs and patients’ expectations.
6. Final Remarks
Author Contributions
Funding
Conflicts of Interest
References
- Organisation for Economic Co-operation and Development (OECD). Health at a Glance 2015: OECD Indicators; OECD Publishing: Paris, France, 2015. [Google Scholar] [CrossRef]
- Velasco Garrido, M.; Kristensen, F.B.; Nielsen, C.P.; Busse, R. Health Technology Assessment and Health Policy-Making in Europe: Current Status, Challenges and Potential; World Health Organization on Behalf of the European Observatory on Health Systems and Policies: London, UK, 2008; ISBN 9789289042932. [Google Scholar]
- Nielsen, C.P.; Funch, T.M.; Kristensen, F.B. Health Technology Assessment: Research Trends and Future Priorities in Europe. J. Health Ser. Res. Policy 2011, 16, 6–15. [Google Scholar] [CrossRef] [PubMed]
- Commission Services (Directorate-General for Economic and Financial Affairs); Economic Policy Committee (Ageing Working Group). Joint Report on Health Care and Long-Term Care Systems & Fiscal Sustainability; European Commission: Brussels, Belgium, 2016; Volume 1. Available online: https://publications.europa.eu/en/publication-detail/-/publication/d6042a45-b535-11e6-9e3c-01aa75ed71a1/language-en (accessed on 25 July 2018).
- Braithwaite, J. Flipping markets to virtue with qui tam and restorative justice. Account. Org. Soc. 2013, 38, 458–468. [Google Scholar] [CrossRef]
- Neu, D.; Everett, J.; Rahaman, A.S.; Martinez, D. Accounting and networks of corruption. Account. Org. Soc. 2013, 38, 505–524. [Google Scholar] [CrossRef]
- Petkov, M.; Cohen, D. Diagnosing Corruption in Healthcare; Transparency International: London, UK, October 2016; ISBN 9781910778609. [Google Scholar]
- European Union. Updated Study on Corruption in the Healthcare Sector; Final Report; Publications Office of the European Union: Luxembourg, 2017. [Google Scholar] [CrossRef]
- Cooper, D.J.; Dacin, T.; Palmer, D. Fraud in accounting, organizations and society. Account. Org. Soc. 2013, 38, 440–457. [Google Scholar] [CrossRef]
- Hood, C. A Public Management for All Seasons. Public Adm. 1991, 69, 3–19. [Google Scholar] [CrossRef]
- Dunleavy, P.; Hood, C. From Old Public Administration to New Public Management. Public Money Manag. 1994, 14, 9–16. [Google Scholar] [CrossRef]
- Barberis, P. The New Public Management and a New Accountability. Public Adm. 1998, 76, 451–470. [Google Scholar] [CrossRef]
- Osborne, S.P. The New Public Governance? Public Manag. Rev. 2006, 8, 377–387. [Google Scholar] [CrossRef]
- Bovaird, A.G.; Bovaird, T.; Löffler, E. Public Management and Governance, 2nd ed.; Routledge: London, UK, 2009; ISBN 0415430429. [Google Scholar]
- World Health Organization. Institutionalisation of Health Technology Assessment: Report of a WHO Meeting, Bonn, Germany, June 30–July 1 2001; WHO Regional Office for Europe: Copenhagen, Denmark, 2001. [Google Scholar]
- Office of Technology Assessment. Development of Medical Technology: Opportunities for Assessment; Government Printing Office: Washington, DC, USA, 1976. [Google Scholar]
- Jonsson, E.; Banta, D. Management of health technologies: An international view. BMJ 1999, 319, 1293. [Google Scholar] [CrossRef] [PubMed]
- Battista, R.N. Towards a paradigm for technology assessment. In The Scientific Basis of Health Services; Peckham, M., Smith, R., Eds.; BMJ Publishing Group: London, UK, 1996. [Google Scholar]
- Kidholm, K.; Ølholm, A.M.; Birk-Olsen, M.; Cicchetti, A.; Fure, B.; Halmesmäki, E.; Kahveci, R.; Kiivet, R.-A.; Wasserfallen, J.B.; Wild, C.; et al. Hospital Managers’ Need for Information in Decision-Making: An Interview Study in Nine European Countries. Health Policy 2015, 119, 1424–1432. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moreno, E.; Girón, F.J.; Vázquez-Polo, F.J.; Negrín, M.A. A Bayesian Net Benefit Approach to Cost-Effectiveness Analysis in Health Technology Assessment. Int. J. Econ. Bus. 2009, 16, 323–345. [Google Scholar] [CrossRef]
- Craven, M.P.; Morgan, S.P.; Crowe, J.A.; Lu, B. Deploying a spreadsheet tool for early economic value assessment of medical device innovations with healthcare decision makers. J. Manag. Mark. Healthc. 2009, 2, 278–292. [Google Scholar] [CrossRef] [Green Version]
- Hartz, S.; John, J. Public health policy decisions on medical innovations: What role can early economic evaluation play? Health Policy 2009, 89, 184–192. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Douw, K.; Vondeling, H.; Oortwijn, W. Priority setting for horizon scanning of new health technologies in Denmark: Views of health care stakeholders and health economists. Health Policy 2006, 76, 334–345. [Google Scholar] [CrossRef] [PubMed]
- Clark, S.; Weale, A. Social values in health priority setting: A conceptual framework. J. Health Organ. Manag. 2012, 26, 293–316. [Google Scholar] [CrossRef] [PubMed]
- Whitty, J.A.; Littlejohns, P. Social values and health priority setting in Australia: An analysis applied to the context of health technology assessment. Health Policy 2015, 119, 127–136. [Google Scholar] [CrossRef] [PubMed]
- Commission Services (Directorate-General for Economic and Financial Affairs); Economic Policy Committee (Ageing Working Group). Joint Report on Health Care and Long-Term Care Systems & Fiscal Sustainability; Country Documents; European Commission: Brussels, Belgium, 2016; Volume 2. Available online: https://publications.europa.eu/en/publication-detail/-/publication/8f7a1f6b-b538-11e6-9e3c-01aa75ed71a1/language-en (accessed on 25 July 2018).
- Levy, A.R.; Mitton, C.; Johnston, K.M.; Harrigan, B.; Briggs, A.H. International Comparison of Comparative Effectiveness Research in Five Jurisdictions. PharmacoEconomics 2010, 28, 813–830. [Google Scholar] [CrossRef] [PubMed]
- Sampietro-Colom, L.; Lach, K.; Cicchetti, A.; Kidholm, K.; Pasternack, I.; Fure, B.; Rosenmöller, M.; Wild, C.; Kahveci, R.; Wasserfallen, J.B.; et al. The AdHopHTA handbook: A Handbook of Hospital-Based Health Technology Assessment (HB-HTA); Public Deliverable; The AdHopHTA Project (FP7/2007-13 Grant Agreement nr. 305018). 2015. Available online: http://www.adhophta.eu/handbook (accessed on 20 July 2018).
- Knies, S.; Lombardi, G.; Commers, M.; Douben, H.P.; Evers, S.; Michelsen, K.; Oortwijn, W.; Opara, C.; Brand, H. Supporting decision making in cross-border regions: A health technology assessment tool for hospitals. Int. J. Technol. Assess. Health Care 2013, 29, 71–78. [Google Scholar] [CrossRef] [PubMed]
- Martelli, N.; Lelong, A.S.; Prognon, P.; Pineau, J. Hospital-Based Health Technology Assessment for Innovative Medical Devices in University Hospitals and the Role of Hospital Pharmacists: Learning from International Experience. Int. J. Technol. Assess. Health Care 2013, 29, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Martelli, N.; Hansen, P.; van den Brink, H.; Boudard, A.; Cordonnier, A.L.; Devaux, C.; Pineau, J.; Prognon, P.; Borget, I. Combining Multi-Criteria Decision Analysis and Mini-Health Technology Assessment: A Funding Decision-Support Tool for Medical Devices in a University Hospital Setting. J. Biomed. Inform. 2016, 59, 201–208. [Google Scholar] [CrossRef] [PubMed]
- Banta, D. The development of health technology assessment. Health Policy 2003, 63, 121–132. [Google Scholar] [CrossRef]
- Velasco Garrido, M.; Busse, R. Health Technology Assessment—An Introduction on Objectives, Role of Evidence, and Structure in Europe; European Observatory on Health Systems and Policies: Brussels, Belgium, 2005. [Google Scholar]
- Cicchetti, A.; Marchetti, M.; Dibidino, R.; and Corio, M. Hospital Based Health Technology Assessment World-Wide Survey; Hospital Based Health Technology Assessment Sub-Interest Group: Roma, Italy, 2008; 41p. [Google Scholar]
- Gagnon, M.P.; Lepage-Savary, D.; Gagnon, J.; St-Pierre, M.; Simard, C.; Rhainds, M.; Lemieux, R.; Gauvin, F.P.; Desmartis, M.; Légaré, F. Introducing patient perspective in health technology assessment at the local level. BMC Health Serv. Res. 2009, 9, 54. [Google Scholar] [CrossRef] [PubMed]
- Gagnon, M.P.; Gagnon, J.; St-Pierre, M.; Gauvin, F.; Piron, F.; Rhainds, M.; Coulombe, M.; Lepage-Savary, D.; Desmartis, M.; Dipankui, M.T.; et al. Involving patients in HTA activities at local level: A study protocol based on the collaboration between researchers and knowledge users. BMC Health Serv. Res. 2012, 12, 14. [Google Scholar] [CrossRef] [PubMed]
- Billaux, M.; Borget, I.; Prognon, P.; Pineau, J.; Martelli, N. Innovative medical devices and hospital decision making: A study comparing the views of hospital pharmacists and physicians. Aust. Health Rev. 2016, 40, 257–261. [Google Scholar] [CrossRef] [PubMed]
- Demirdjian, G. A 10-Year Hospital-Based Health Technology Assessment Program in A Public Hospital in Argentina. Int. J. Technol. Assess. Health Care 2015, 31, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Martelli, N.; Billaux, M.; Borget, I.; Pineau, J.; Prognon, P.; van den Brink, H. Introduction of Innovative Medical Devices at French University Hospitals: An Overview of Hospital-Based Health Technology Assessment Initiatives. Int. J. Technol. Assess. Health Care 2015, 31, 12–18. [Google Scholar] [CrossRef] [PubMed]
- Halmesmäki, E.; Pasternack, I.; Roine, R. Hospital-based health technology assessment (HTA) in Finland: A case study on collaboration between hospitals and the national HTA unit. BMC Health Res. Policy Syst. 2016, 14, 25. [Google Scholar] [CrossRef] [PubMed]
- Gagnon, M.P. Hospital-Based Health Technology Assessment: Developments to Date. PharmacoEconomics 2014, 32, 819–824. [Google Scholar] [CrossRef] [PubMed]
- Sampietro-Colom, L.; Morilla-Bachs, I.; Gutierrez-Moreno, S.; Gallo, P. Development and Test of a Decision Support Tool for Hospital Health Technology Assessment. Int. J. Technol. Assess. Health Care 2012, 28, 460–465. [Google Scholar] [CrossRef] [PubMed]
- Tedesco, G.; Faggiano, F.C.; Leo, E.; Derrico, P.; Ritrovato, M. A Comparative Cost Analysis of Robotic-Assisted Surgery versus Laparoscopic Surgery and Open Surgery: The Necessity of Investing Knowledgeably. Surg. Endosc. 2016, 30, 5044–5051. [Google Scholar] [CrossRef] [PubMed]
- Lafortune, L.; Farand, L.; Mondou, I.; Sicotte, C.; Battista, R. Assessing the Performance of Health Technology Assessment Organizations: A Framework. Int. J. Technol. Assess. Health Care 2008, 24, 76–86. [Google Scholar] [CrossRef] [PubMed]
- Kosherbayeva, L.; Hailey, D.; Kurakbaev, K.; Tsoy, A.; Zhuzzhanov, O.; Donbay, A.; Kumar, A.; Nadyrov, K. Implementation of Health Technology Assessment Work in a Hospital in Kazakhstan. Int. J. Technol. Assess. Health Care 2016, 32, 78–80. [Google Scholar] [CrossRef] [PubMed]
- Miniati, R.; Dori, F.; Cecconi, G.; Gusinu, R.; Niccolini, F.; Biffi Gentili, G. HTA decision support system for sustainable business continuity management in hospitals. The case of surgical activity at the University Hospital in Florence. Technol. Health Care 2013, 21, 49–61. [Google Scholar] [CrossRef] [PubMed]
- Vuong, Q.H. The (ir)rational consideration of the cost of science in transition economies. Nat. Hum. Behav. 2018, 2, 5. [Google Scholar] [CrossRef]
- Hardyman, W.; Daunt, K.L.; Kitchener, M. Value Co-Creation through Patient Engagement in Health Care: A micro-level approach and research agenda. Public Manag. Rev. 2015, 17, 90–107. [Google Scholar] [CrossRef]
- Van Eijk, C.J.A.; Steen, T.P.S. Why People Co-Produce: Analysing citizens’ perceptions on co-planning engagement in health care services. Public Manag. Rev. 2014, 16, 358–382. [Google Scholar] [CrossRef]
- Vuong, Q.H.; Ho, M.T.; Nguyen, H.K.; Vuong, T.T. Healthcare consumers’ sensitivity to costs: A reflection on behavioural economics from an emerging market. Palgrave Commun. 2018, 4, 70. [Google Scholar] [CrossRef]
- Palumbo, R. Contextualizing co-production of health care: A systematic literature review. Int. J. Public Sect. Manag. 2016, 29, 72–90. [Google Scholar] [CrossRef]
- Renedo, A.; Marston, C.A.; Spyridonidis, D.; Barlow, J. Patient and Public Involvement in Healthcare Quality Improvement: How organizations can help patients and professionals to collaborate. Public Manag. Rev. 2015, 17, 17–34. [Google Scholar] [CrossRef]
- Gurtner, S. Making the Right Decisions about New Technologies: A Perspective on Criteria and Preferences in Hospitals. Health Care Manag. Rev. 2014, 39, 245–254. [Google Scholar] [CrossRef] [PubMed]
- Bardhan, P. Corruption and Development: A Review of Issues. J. Econ. Lit. 1997, 35, 1320–1346. [Google Scholar]
- Mauro, P. Corruption and the Composition of Public Expenditure. J. Public Econ. 1998, 69, 263–279. [Google Scholar] [CrossRef]
- Liu, E. A historical review of the control of corruption on economic crime in China. J. Financ. Crime 2016, 23, 4–21. [Google Scholar] [CrossRef]
- Pope, J. Confronting Corruption: The Elements of a National Integrity System; Transparency International: London, UK, 2000; ISBN 3980565785. [Google Scholar]
- Pope, J. National Integrity Systems: The key to building sustainable, just and honest government. In Promoting Integrity. Evaluating and Improving Public Institutions; Head, B.W., Brown, A.J., Connors, C., Eds.; Ashgate Publishing: Aldershot, UK, 2008; pp. 28–29. [Google Scholar]
- Organisation for Economic Co-operation and Development (OECD). Corruption: Glossary of International Criminal Standards; OECD Publishing: Paris, France, 2008; Available online: http://www.oecd.org/corruption/corruptionglossaryofinternationalcriminalstandards.htm (accessed on 24 July 2018).
- Sorenson, C.; Kanavos, P. Medical technology procurement in Europe: A cross-country comparison of current practice and policy. Health Policy 2011, 100, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Palmer, D. Extending the process Model of Collective Corruption. Res. Organ. Behav. 2008, 28, 107–135. [Google Scholar] [CrossRef]
- Beeri, I.; Navot, D. Local Political Corruption: Potential structural malfunctions at the central-local, local-local and intra-local levels. Public Manag. Rev. 2012, 15, 712–739. [Google Scholar] [CrossRef]
- Drummond, M.; Tarricone, R.; Torbica, A. Assessing the Added Value of Health Technologies: Reconciling Different Perspectives. Value Health 2013, 16, 7–13. [Google Scholar] [CrossRef] [PubMed]
- Cicchetti, A.; Iacopino, V.; Coretti, S.; Fiore, A.; Marchetti, M.; Sampietro-Colom, L.; Kidholm, K.; Wasserfallen, J.-B.; Kahveci, R.; Halmesmäki, E.; et al. Toward a Contingency Model for Hospital-Based Health Technology Assessment: Evidence from AdHopHTA Project. Int. J. Technol. Assess. Health Care 2018, 34, 205–211. [Google Scholar] [CrossRef] [PubMed]
- Miniati, R.; Frosini, F.; Cecconi, G.; Dori, F.; Biffi Gentili, G. Development of Sustainable Models for Technology Evaluation in Hospital. Technol. Health Care 2014, 22, 729–739. [Google Scholar] [CrossRef] [PubMed]
- Poulin, P.; Austen, L.; Scott, C.M.; Waddell, C.D.; Dixon, E.; Poulin, M.; Lafrenière, R. Multi-Criteria Development and Incorporation into Decision Tools for Health Technology Adoption. J. Health Organ. Manag. 2013, 27, 246–265. [Google Scholar] [CrossRef] [PubMed]
- Ritrovato, M.; Faggiano, F.C.; Tedesco, G.; Derrico, P. Decision-Oriented Health Technology Assessment: One Step Forward in Supporting the Decision-Making Process in Hospitals. Value Health 2015, 18, 505–511. [Google Scholar] [CrossRef] [PubMed]
- Barile, S. Introduzione. In Condizioni di Efficacia Relazionale e di Performance Dell’azienda Sanitaria; Saviano, M., Ed.; Giappichelli: Torino, Italy, 2012; pp. 11–13. ISBN 9788834838938. [Google Scholar]
- EUnetHTA. The HTA Core Model®. Guiding Principles on Use. 2015. Available online: https://www.eunethta.eu/wp-content/uploads/2018/01/The-HTA-Core-Model_Guiding-principles-on-use_20151218.pdf (accessed on 25 September 2018).
- Ølholm, A.M.; Kidholm, K.; Birk-Olsen, M.; Christensen, J.B. Hospital Managers’ Need for Information on Health Technology Investments. Int. J. Technol. Assess. Health Care 2015, 31, 414–425. [Google Scholar] [CrossRef] [PubMed]
- Gastaldi, L.; Corso, M. Smart healthcare digitalization: Using ICT to effectively balance exploration and exploitation within hospitals. Int. J. Eng. Bus. Manag. 2012, 4, 4–9. [Google Scholar] [CrossRef]
- Berman, S.; Marshall, A. The next digital transformation: From an individual-centered to an everyone-to-everyone economy. Strateg. Leadersh. 2014, 42, 9–17. [Google Scholar] [CrossRef]
- Barile, S.; Polese, F.; Antonucci, E.; Carrubbo, L. Al confine dell’innovazione tecnologica in sanità: La medicina traslazionale. In Le Responsabilità in Ambito Sanitario; Aleo, S., De Matteis, R., Vecchio, G., Eds.; CEDAM: Milano, Italy, 2014; Volume 1, pp. 1233–1267. ISBN 9788813347079. [Google Scholar]
- Cappellaro, G.; Fattore, G.; Torbica, A. Funding health technologies in decentralized systems: A comparison between Italy and Spain. Health Policy 2009, 92, 313–321. [Google Scholar] [CrossRef] [PubMed]
- Galani, C.; Rutten, F.F. Self-reported healthcare decision-makers’ attitudes towards economic evaluations of medical technologies. Curr. Med. Res. Opin. 2008, 24, 3049–3058. [Google Scholar] [CrossRef] [PubMed]
- Otley, D. Performance management: A framework for management control systems research. Manag. Account. Res. 1999, 10, 363–382. [Google Scholar] [CrossRef]
- Lebas, M.; Euske, K. A conceptual and operational delineation of performance. In Business Performance Measurement: Theory and Practice; Neely, A., Ed.; Cambridge University Press: Cambridge, UK, 2002; pp. 65–79. ISBN 0511041578. [Google Scholar]
- Ferreira, A.; Otley, D. The Design and Use of Performance Management Systems: An Extended Framework for Analysis. Manag. Account. Res. 2009, 20, 263–282. [Google Scholar] [CrossRef]
- Kaplan, R.S.; Norton, D.P. The Balanced Scorecard: Translating Strategy into Action; Harvard Business School Press: Boston, MA, USA, 1996; ISBN 9780875846514. [Google Scholar]
- Neely, A.; Adams, C.; Crowe, P. The performance prism in practice. Meas. Bus. Excell. 2001, 5, 6–13. [Google Scholar] [CrossRef]
- Fernandez, A. Les Nouveaux Tableaux de Bord des Managers: Le Projet Business Intelligence Clés en Main, 6th ed.; Eyrolles: Paris, France, 2013; ISBN -13. [Google Scholar]
- Figueira, J.; Greco, S.; Ehrgott, M. Multiple Criteria Decision Analysis: State of the Art Surveys; Springer: New York, NY, USA, 2005; pp. 4–7. ISBN 9780387230818. [Google Scholar]
- Vuong, Q.H. Sociodemographic Factors Influencing Vietnamese Patient Satisfaction with Healthcare Services and Some Meaningful Empirical Thresholds. Iran. J. Public Health 2018, 47, 119–126. [Google Scholar] [PubMed]
- Sloane, E.B.; Liberatore, M.J.; Nydick, R.L.; Luo, W.; Chung, Q.B. Using the Analytic Hierarchy Process as a Clinical Engineering Tool to Facilitate an Iterative, Multidisciplinary, Microeconomic Health Technology Assessment. Comput. Oper. Res. 2003, 30, 1447–1465. [Google Scholar] [CrossRef]
- European Union. Directive 2014/24/EU of the European Parliament and of the Council of 26 February 2014 on public procurement and repealing Directive 2004/18/EC. Off. J. Eur. Union 2014, 65–242. Available online: http://data.europa.eu/eli/dir/2014/24/oj (accessed on 20 July 2018).
- Bebbington, J.; Brown, J.; Frame, B. Accounting technologies and sustainability assessment models. Ecol. Econ. 2007, 61, 224–236. [Google Scholar] [CrossRef]
- Drummond, M.F.; Schwartz, J.; Luce, B.R.; Neumann, P.J.; Siebert, U.; Sullivan, S.D. Key principles for the improved conduct of health technology assessments for resource allocation decisions. Int. J. Technol. Assess. Health Care 2008, 24, 244–258. [Google Scholar] [CrossRef] [PubMed]
- Ball, A.; Bebbington, J. Editorial: Accounting and Reporting for Sustainable Development in Public Service Organizations. Public Money Manag. 2008, 28, 323–326. [Google Scholar] [CrossRef]
- Silva, P.; Ferreira, A. Performance management in primary healthcare service: Evidence from a field study. Qual. Res. Account. Manag. 2010, 7, 424–449. [Google Scholar] [CrossRef]
- Fama, E.F.; Jensen, M.C. Separation of ownership and control. J. Law Econ. 1983, 26, 301–325. [Google Scholar] [CrossRef]
- Mulgan, R. ‘Accountability’: An Ever Expanding Concept. Public Adm. 2000, 78, 555–573. [Google Scholar] [CrossRef]
- Fowler, C.J.; Cordery, C. From Community to Public Ownership: A Tale of Changing Accountabilities. Account. Audit. Account. J. 2015, 28, 128–153. [Google Scholar] [CrossRef]
- Klitgaard, R. Controlling Corruption, 1st ed.; University of California Press: Berkeley, CA, USA, 1988; ISBN 9780520074088. [Google Scholar]
- Carnegie, G.D.; West, B.P. Making Accounting Accountable in the Public Sector. Crit. Perspect. Account. 2005, 16, 905–928. [Google Scholar] [CrossRef]
- Osborne, S.P.; Ball, A. Social Accounting and Public Management. Accountability for the Public Good, 1st ed.; Routledge: London, UK, 2010; ISBN 9780415806497. [Google Scholar]
- Bracci, E.; Humphrey, C.; Moll, J.; Steccolini, I. Public Sector Accounting, Accountability and Austerity: More Than Balancing the Books? Account. Audit. Account. J. 2015, 28, 878–908. [Google Scholar] [CrossRef]
- Newberry, S. Public Sector Accounting: Shifting Concepts of Accountability. Public Money Manag. 2015, 35, 371–376. [Google Scholar] [CrossRef]
- Christensen, M.; Skærbæk, P. Framing and Overflowing of Public Sector Accountability Innovations. Account. Audit. Account. J. 2007, 20, 101–132. [Google Scholar] [CrossRef]
- D’Onza, G.; Brotini, F.; Zarone, V. Disclosure on Measures to Prevent Corruption Risks: A Study of Italian Local Governments. Int. J. Public Adm. 2017, 40, 612–624. [Google Scholar] [CrossRef]
- Kassel, D.S. Performance, Accountability, and the Debate over Rules. Public Adm. Rev. 2008, 68, 241–252. [Google Scholar] [CrossRef]
- Barrett, P. New Development: Financial Reform and Good Governance. Public Money Manag. 2014, 34, 59–66. [Google Scholar] [CrossRef]

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Palozzi, G.; Brunelli, S.; Falivena, C. Higher Sustainability and Lower Opportunistic Behaviour in Healthcare: A New Framework for Performing Hospital-Based Health Technology Assessment. Sustainability 2018, 10, 3550. https://doi.org/10.3390/su10103550
Palozzi G, Brunelli S, Falivena C. Higher Sustainability and Lower Opportunistic Behaviour in Healthcare: A New Framework for Performing Hospital-Based Health Technology Assessment. Sustainability. 2018; 10(10):3550. https://doi.org/10.3390/su10103550
Chicago/Turabian StylePalozzi, Gabriele, Sandro Brunelli, and Camilla Falivena. 2018. "Higher Sustainability and Lower Opportunistic Behaviour in Healthcare: A New Framework for Performing Hospital-Based Health Technology Assessment" Sustainability 10, no. 10: 3550. https://doi.org/10.3390/su10103550



