Assessment of Dynamic Knee Valgus between Lateral Step-Down Test and Running in Female Runners with and without Patellofemoral Pain Using Two-Dimensional Video Analysis
Abstract
1. Introduction
2. Materials and Methods
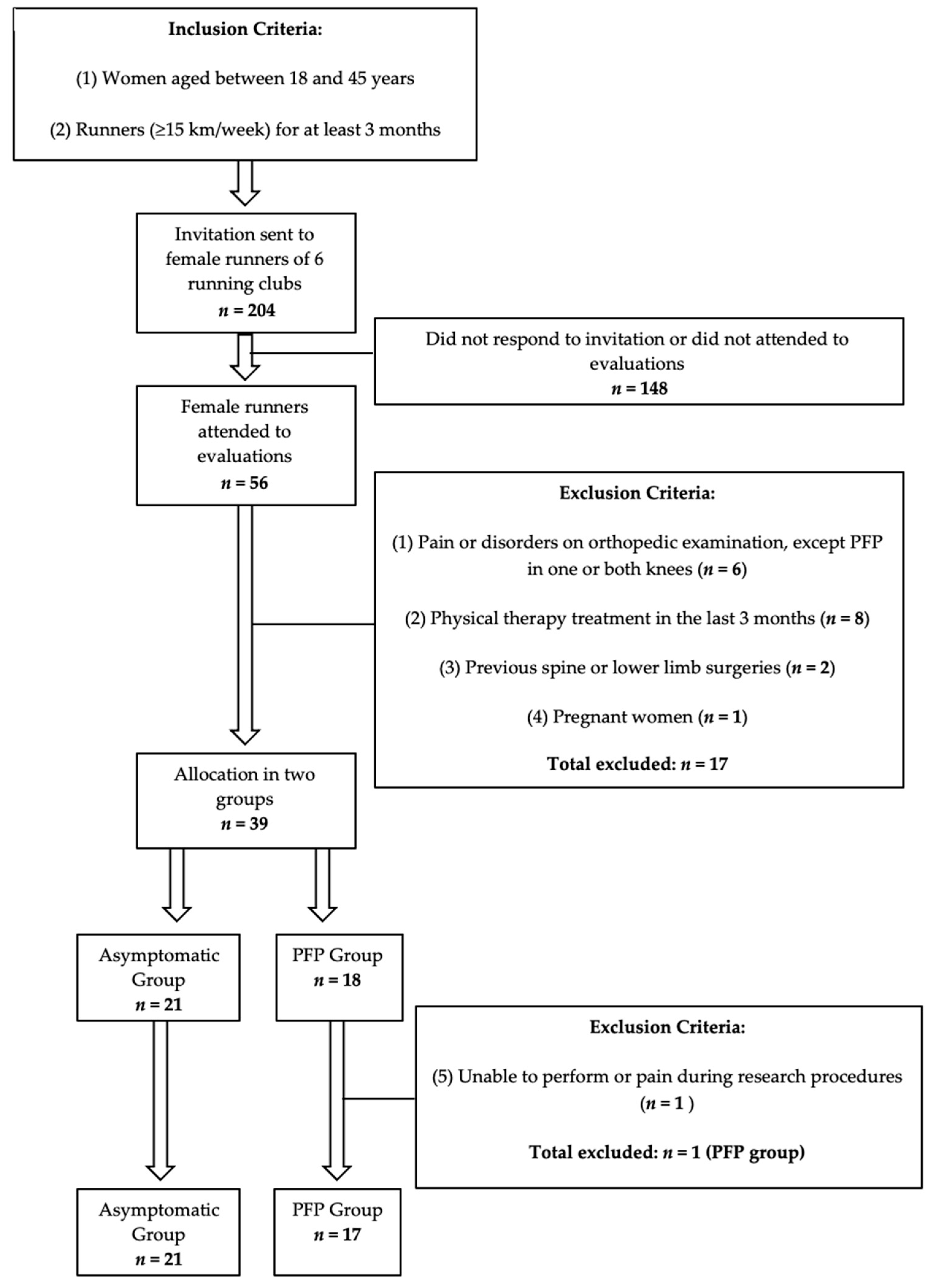
2.1. Participants and Design
2.2. Procedures
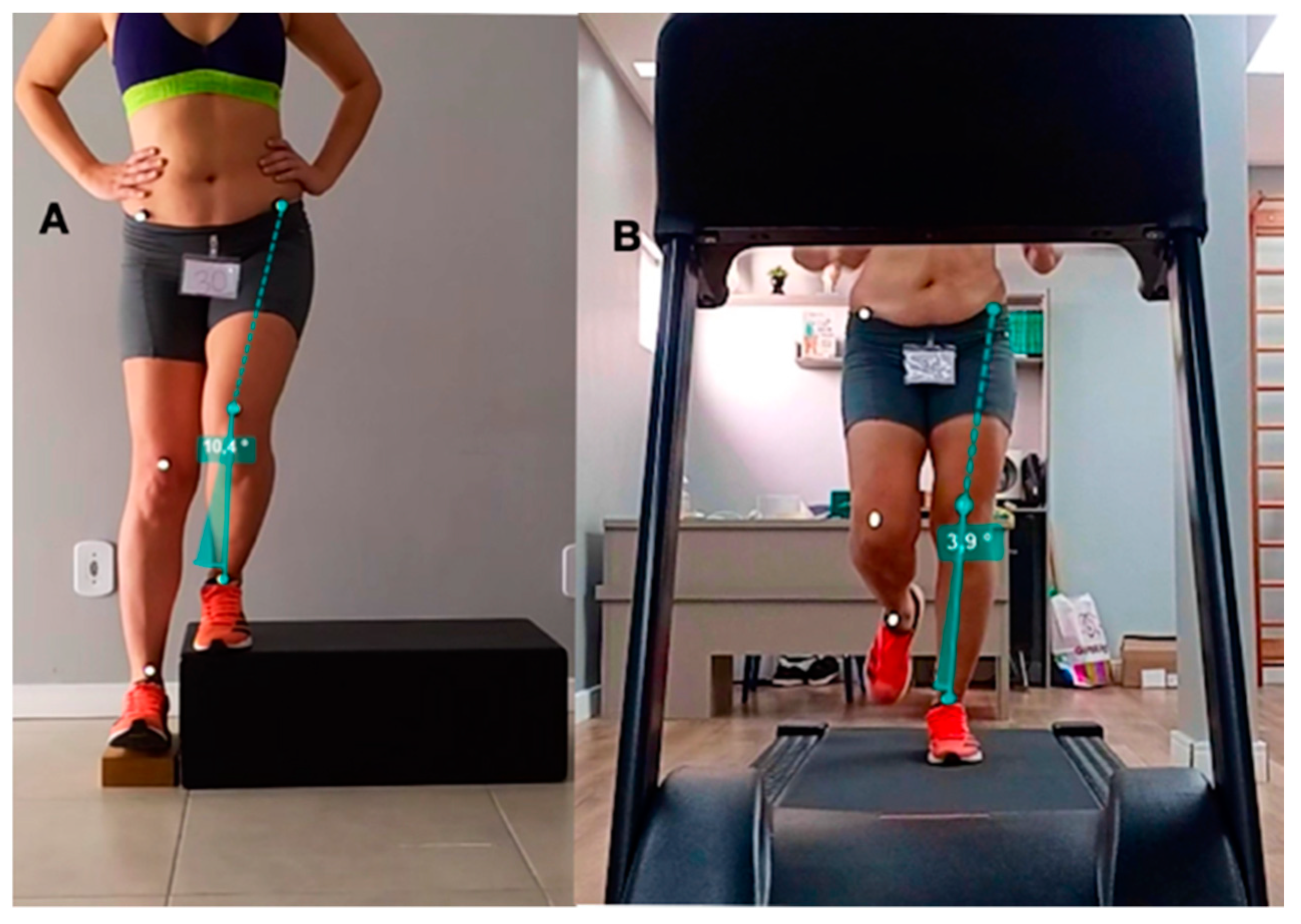
2.2.1. Lateral Step Down (LSD)
2.2.2. Running
2.3. Analysis
2.3.1. Data Processing and Reduction
2.3.2. Statical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Esculier, J.F.; Maggs, K.; Maggs, E.; Dubois, B. A Contemporary Approach to Patellofemoral Pain in Runners. J. Athl. Train. 2020, 55, 1206–1214. [Google Scholar] [CrossRef] [PubMed]
- Witvrouw, E.; Callaghan, M.J.; Stefanik, J.J.; Noehren, B.; Bazett-Jones, D.M.; Willson, J.D.; Earl-Boehm, J.E.; Davis, I.S.; Powers, C.M.; McConnell, J.; et al. Patellofemoral pain: Consensus statement from the 3rd International Patellofemoral Pain Research Retreat held in Vancouver, September 2013. Br. J. Sports Med. 2014, 48, 411–414. [Google Scholar] [CrossRef] [PubMed]
- Boling, M.; Padua, D.; Marshall, S.; Guskiewicz, K.; Pyne, S.; Beutler, A. Gender differences in the incidence and prevalence of patellofemoral pain syndrome. Scand. J. Med. Sci. Sports 2010, 20, 725–730. [Google Scholar] [CrossRef] [PubMed]
- Crossley, K.M.; Stefanik, J.J.; Selfe, J.; Collins, N.J.; Davis, I.S.; Powers, C.M.; McConnell, J.; Vicenzino, B.; Bazett-Jones, D.M.; Esculier, J.F.; et al. 2016 Patellofemoral pain consensus statement from the 4th International Patellofemoral Pain Research Retreat, Manchester. Part 1: Terminology, definitions, clinical examination, natural history, patellofemoral osteoarthritis and patient-reported outcome measures. Br. J. Sports Med. 2016, 50, 839–843. [Google Scholar] [CrossRef] [PubMed]
- Cheung, R.T.; Zhang, Z.; Ngai, S.P. Different relationships between the level of patellofemoral pain and quality of life in professional and amateur athletes. PM R 2013, 5, 568–572. [Google Scholar] [CrossRef]
- Lankhorst, N.E.; Bierma-Zeinstra, S.M.; van Middelkoop, M. Factors associated with patellofemoral pain syndrome: A systematic review. Br. J. Sports Med. 2013, 47, 193–206. [Google Scholar] [CrossRef]
- Powers, C.M.; Witvrouw, E.; Davis, I.S.; Crossley, K.M. Evidence-based framework for a pathomechanical model of patellofemoral pain: 2017 patellofemoral pain consensus statement from the 4th International Patellofemoral Pain Research Retreat, Manchester, UK: Part 3. Br. J. Sports Med. 2017, 51, 1713–1723. [Google Scholar] [CrossRef]
- Uhlár, Á.; Ambrus, M.; Kékesi, M.; Fodor, E.; Grand, L.; Szathmáry, G.; Rácz, K.; Lacza, Z. Kinect Azure–Based Accurate Measurement of Dynamic Valgus Position of the Knee—A Corrigible Predisposing Factor of Osteoarthritis. Appl. Sci. 2021, 11, 5536. [Google Scholar] [CrossRef]
- Rabin, A.; Kozol, Z.; Moran, U.; Efergan, A.; Geffen, Y.; Finestone, A.S. Factors associated with visually assessed quality of movement during a lateral step-down test among individuals with patellofemoral pain. J. Orthop. Sports Phys. Ther. 2014, 44, 937–946. [Google Scholar] [CrossRef]
- Lankhorst, N.E.; Bierma-Zeinstra, S.M.; van Middelkoop, M. Risk factors for patellofemoral pain syndrome: A systematic review. J. Orthop. Sports Phys. Ther. 2012, 42, 81–94. [Google Scholar] [CrossRef]
- Hewett, T.E.; Myer, G.D.; Ford, K.R.; Heidt, R.S., Jr.; Colosimo, A.J.; McLean, S.G.; van den Bogert, A.J.; Paterno, M.V.; Succop, P. Biomechanical measures of neuromuscular control and valgus loading of the knee predict anterior cruciate ligament injury risk in female athletes: A prospective study. Am. J. Sports Med. 2005, 33, 492–501. [Google Scholar] [CrossRef] [PubMed]
- Petersen, W.; Rembitzki, I.; Liebau, C. Patellofemoral pain in athletes. Open Access J. Sports Med. 2017, 8, 143–154. [Google Scholar] [CrossRef] [PubMed]
- Larwa, J.; Stoy, C.; Chafetz, R.S.; Boniello, M.; Franklin, C. Stiff Landings, Core Stability, and Dynamic Knee Valgus: A Systematic Review on Documented Anterior Cruciate Ligament Ruptures in Male and Female Athletes. Int. J. Environ. Res. Public Health 2021, 18, 3826. [Google Scholar] [CrossRef] [PubMed]
- Lima, Y.L.; Ferreira, V.M.L.M.; de Paula Lima, P.O.; Bezerra, M.A.; de Oliveira, R.R.; Almeida, G.P.L. The association of ankle dorsiflexion and dynamic knee valgus: A systematic review and meta-analysis. Phys. Ther. Sport 2018, 29, 61–69. [Google Scholar] [CrossRef]
- Wyndow, N.; De Jong, A.; Rial, K.; Tucker, K.; Collins, N.; Vicenzino, B.; Russell, T.; Crossley, K. The relationship of foot and ankle mobility to the frontal plane projection angle in asymptomatic adults. J. Foot Ankle Res. 2016, 9, 3. [Google Scholar] [CrossRef]
- Bittencourt, N.F.; Ocarino, J.M.; Mendonça, L.D.; Hewett, T.E.; Fonseca, S.T. Foot and hip contributions to high frontal plane knee projection angle in athletes: A classification and regression tree approach. J. Orthop. Sports Phys. Ther. 2012, 42, 996–1004. [Google Scholar] [CrossRef]
- Cronström, A.; Creaby, M.W.; Nae, J.; Ageberg, E. Gender differences in knee abduction during weight-bearing activities: A systematic review and meta-analysis. Gait Posture 2016, 49, 315–328. [Google Scholar] [CrossRef]
- Silva, R.L.E.; Pinheiro, Y.T.; Lins, C.A.A.; de Oliveira, R.R.; Scattone Silva, R. Assessment of quality of movement during a lateral step-down test: Narrative review. J. Bodyw. Mov. Ther. 2019, 23, 835–843. [Google Scholar] [CrossRef]
- Lopes Ferreira, C.; Barton, G.; Delgado Borges, L.; Dos Anjos Rabelo, N.D.; Politti, F.; Garcia Lucareli, P.R. Step down tests are the tasks that most differentiate the kinematics of women with patellofemoral pain compared to asymptomatic controls. Gait Posture 2019, 72, 129–134. [Google Scholar] [CrossRef]
- Willson, J.D.; Davis, I.S. Utility of the frontal plane projection angle in females with patellofemoral pain. J. Orthop. Sports Phys. Ther. 2008, 38, 606–615. [Google Scholar] [CrossRef]
- Mizner, R.L.; Chmielewski, T.L.; Toepke, J.J.; Tofte, K.B. Comparison of 2-dimensional measurement techniques for predicting knee angle and moment during a drop vertical jump. Clin. J. Sport Med. 2012, 22, 221–227. [Google Scholar] [CrossRef] [PubMed]
- Munro, A.; Herrington, L.; Carolan, M. Reliability of 2-dimensional video assessment of frontal-plane dynamic knee valgus during common athletic screening tasks. J. Sport Rehabil. 2012, 21, 7–11. [Google Scholar] [CrossRef] [PubMed]
- Herrington, L. Knee valgus angle during single leg squat and landing in patellofemoral pain patients and controls. Knee 2014, 21, 514–517. [Google Scholar] [CrossRef] [PubMed]
- Holden, S.; Boreham, C.; Doherty, C.; Delahunt, E. Two-dimensional knee valgus displacement as a predictor of patellofemoral pain in adolescent females. Scand. J. Med. Sci. Sports 2017, 27, 188–194. [Google Scholar] [CrossRef] [PubMed]
- Dierks, T.A.; Manal, K.T.; Hamill, J.; Davis, I. Lower extremity kinematics in runners with patellofemoral pain during a prolonged run. Med. Sci. Sports Exerc. 2011, 43, 693–700. [Google Scholar] [CrossRef] [PubMed]
- Glaviano, N.R.; Saliba, S. Association of altered frontal plane kinematics and physical activity levels in females with patellofemoral pain. Gait Posture 2018, 65, 86–88. [Google Scholar] [CrossRef]
- Rees, D.; Younis, A.; MacRae, S. Is there a correlation in frontal plane knee kinematics between running and performing a single leg squat in runners with patellofemoral pain syndrome and asymptomatic runners? Clin. Biomech. 2019, 61, 227–232. [Google Scholar] [CrossRef]
- Noehren, B.; Sanchez, Z.; Cunningham, T.; McKeon, P.O. The effect of pain on hip and knee kinematics during running in females with chronic patellofemoral pain. Gait Posture 2012, 36, 596–599. [Google Scholar] [CrossRef]
- Alenezi, F.; Herrington, L.; Jones, P.; Jones, R. Relationships between lower limb biomechanics during single leg squat with running and cutting tasks: A preliminary investigation. Br. J. Sports Med. 2014, 48, 560–561. [Google Scholar] [CrossRef][Green Version]
- Mostaed, M.F.; Werner, D.M.; Barrios, J.A. 2D and 3D kinematics during lateral step-down testing in individuals with anterior cruciate ligament reconstruction. Int J. Sports Phys. Ther. 2018, 13, 77–85. [Google Scholar] [CrossRef]
- Luz, B.C.; Dos Santos, A.F.; Serrão, F.V. Are hip and knee kinematics and training load characteristics relate to pain intensity and physical function level in runners with Patellofemoral Pain? Gait Posture 2021, 84, 162–168. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, A.F.; Nakagawa, T.H.; Lessi, G.C.; Luz, B.C.; Matsuo, H.T.M.; Nakashima, G.Y.; Maciel, C.D.; Serrão, F.V. Effects of three gait retraining techniques in runners with patellofemoral pain. Phys. Ther. Sport 2019, 36, 92–100. [Google Scholar] [CrossRef] [PubMed]
- Yamato, T.P.; Saragiotto, B.T.; Lopes, A.D. A consensus definition of running-related injury in recreational runners: A modified Delphi approach. J. Orthop. Sports Phys. Ther. 2015, 45, 375–380. [Google Scholar] [CrossRef] [PubMed]
- Bramah, C.; Preece, S.J.; Gill, N.; Herrington, L.A. 10% Increase in Step Rate Improves Running Kinematics and Clinical Outcomes in Runners with Patellofemoral Pain at 4 Weeks and 3 Months. Am. J. Sports Med. 2019, 47, 3406–3413. [Google Scholar] [CrossRef]
- Queen, R.M.; Gross, M.T.; Liu, H.Y. Repeatability of lower extremity kinetics and kinematics for standardized and self-selected running speeds. Gait Posture 2006, 23, 282–287. [Google Scholar] [CrossRef]
- Ho, K.Y.; Murata, A. Asymmetries in Dynamic Valgus Index after Anterior Cruciate Ligament Reconstruction: A Proof-of-Concept Study. Int. J. Environ. Res. Public Health 2021, 18, 7047. [Google Scholar] [CrossRef]
- Llurda-Almuzara, L.; Pérez-Bellmunt, A.; López-de-Celis, C.; Aiguadé, R.; Seijas, R.; Casasayas-Cos, O.; Labata-Lezaun, N.; Alvarez, P. Normative data and correlation between dynamic knee valgus and neuromuscular response among healthy active males: A cross-sectional study. Sci. Rep. 2020, 10, 17206. [Google Scholar] [CrossRef]
- Hopkins, W.G.; Marshall, S.W.; Batterham, A.M.; Hanin, J. Progressive statistics for studies in sports medicine and exercise science. Med. Sci. Sports Exerc. 2009, 41, 3–13. [Google Scholar] [CrossRef]
- Pompeo, K.D.; da Rocha, E.S.; Morales, A.B.; Klein, K.D.; Vaz, M.A. Does forward step-down task frontal kinematics differ in women with and without patellofemoral pain? A cross-sectional study. Sport Sci. Health 2021. [Google Scholar] [CrossRef]
- Choudhary, R.; Malik, M.; Aslam, A.; Khurana, D.; Chauhan, S. Effect of various parameters on Quadriceps angle in adult Indian population. J. Clin. Orthop. Trauma 2019, 10, 149–154. [Google Scholar] [CrossRef]
- Fidai, M.S.; Okoroha, K.R.; Meldau, J.; Meta, F.; Lizzio, V.A.; Borowsky, P.; Redler, L.H.; Moutzouros, V.; Makhni, E.C. Fatigue Increases Dynamic Knee Valgus in Youth Athletes: Results From a Field-Based Drop-Jump Test. Arthroscopy 2020, 36, 214–222. [Google Scholar] [CrossRef] [PubMed]
- Emamvirdi, M.; Letafatkar, A.; Khaleghi Tazji, M. The Effect of Valgus Control Instruction Exercises on Pain, Strength, and Functionality in Active Females with Patellofemoral Pain Syndrome. Sports Health 2019, 11, 223–237. [Google Scholar] [CrossRef] [PubMed]
- Kakouris, N.; Yener, N.; Fong, D.T.P. A systematic review of running-related musculoskeletal injuries in runners. J. Sport Health Sci. 2021, 10, 513–522. [Google Scholar] [CrossRef] [PubMed]
- Mellinger, S.; Neurohr, G.A. Evidence based treatment options for common knee injuries in runners. Ann. Transl. Med. 2019, 7, S249. [Google Scholar] [CrossRef]
| Variables | Group | p | CI 95% | |
|---|---|---|---|---|
| Asymptomatic (n = 21) | PFP (n = 17) | |||
| Age (years) | 35.81 ± 6.32 | 36.00 ± 7.55 | 0.933 | (−4.37; 4.75) |
| Weight (kg) | 61.91 ± 7.10 | 65.21 ± 11.11 | 0.274 | (−2.72; 9.32) |
| Height (cm) | 163.23 ± 6.36 | 163.11 ± 7.40 | 0.957 | (−4.65; 4.40) |
| BMI (kg/cm2) | 23.23 ± 2.21 | 24.48 ± 3.73 | 0.207 | (−0.72; 3.22) |
| Running time (months) | 40.81 ± 30.40 | 56.00 ± 58.78 | 0.311 | (−14.76; 45.14) |
| Weekly training distance (km/week) | 21.19 ± 6.18 | 20.59 ± 5.73 | 0.760 | (−4.56; 3.36) |
| Running speed (m/s) | 2.42 ± 0.29 | 2.38 ± 0.36 | 0.707 | (−0.17; 0.25) |
| Group | FPPA (°) | p | η2 | CI 95% | r | p | |
|---|---|---|---|---|---|---|---|
| LSD | Running | ||||||
| Asymptomatic | 16.32 ± 5.38 * | 4.02 ± 3.26 | <0.01 * | 0.680 | (9.44; 15.15) | 0.16 | 0.465 |
| PFP | 17.54 ± 7.25 # | 4.64 ± 3.62 | <0.01 # | 0.654 | (9.73; 16.07) | 0.27 | 0.289 |
| FPPA (°) | Group | p | η2 | CI 95% | |
|---|---|---|---|---|---|
| Asymptomatic | PFP | ||||
| LSD | 16.32 ± 5.38 | 17.54 ± 7.25 | 0.555 | 0.010 | (−5.38; 2.93) |
| Running | 4.02 ± 3.26 | 4.64 ± 3.62 | 0.588 | 0.008 | (−2.88; 1.65) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
de Vasconcelos, D.P.; Aidar, F.J.; Lima, T.B.; Filho, F.M.d.N.; Mendonça, I.L.A.; Díaz-de-Durana, A.L.; Garrido, N.D.; Santiago, M.S.; Junior, W.M.d.S. Assessment of Dynamic Knee Valgus between Lateral Step-Down Test and Running in Female Runners with and without Patellofemoral Pain Using Two-Dimensional Video Analysis. Clin. Pract. 2022, 12, 425-435. https://doi.org/10.3390/clinpract12030047
de Vasconcelos DP, Aidar FJ, Lima TB, Filho FMdN, Mendonça ILA, Díaz-de-Durana AL, Garrido ND, Santiago MS, Junior WMdS. Assessment of Dynamic Knee Valgus between Lateral Step-Down Test and Running in Female Runners with and without Patellofemoral Pain Using Two-Dimensional Video Analysis. Clinics and Practice. 2022; 12(3):425-435. https://doi.org/10.3390/clinpract12030047
Chicago/Turabian Stylede Vasconcelos, Diego Protasio, Felipe J. Aidar, Tarcisio Brandao Lima, Flavio Martins do Nascimento Filho, Igor Leonardo Alves Mendonça, Alfonso López Díaz-de-Durana, Nuno Domingos Garrido, Michael Silveira Santiago, and Walderi Monteiro da Silva Junior. 2022. "Assessment of Dynamic Knee Valgus between Lateral Step-Down Test and Running in Female Runners with and without Patellofemoral Pain Using Two-Dimensional Video Analysis" Clinics and Practice 12, no. 3: 425-435. https://doi.org/10.3390/clinpract12030047
APA Stylede Vasconcelos, D. P., Aidar, F. J., Lima, T. B., Filho, F. M. d. N., Mendonça, I. L. A., Díaz-de-Durana, A. L., Garrido, N. D., Santiago, M. S., & Junior, W. M. d. S. (2022). Assessment of Dynamic Knee Valgus between Lateral Step-Down Test and Running in Female Runners with and without Patellofemoral Pain Using Two-Dimensional Video Analysis. Clinics and Practice, 12(3), 425-435. https://doi.org/10.3390/clinpract12030047








