BALANCE Dietary Index in Patients with Heart Failure, and Its Adherence in Sergipe, Brazil
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Participants
2.3. Sociodemographic, Clinical and Lifestyle Data
2.4. Anthropometric Data
2.5. Food Data
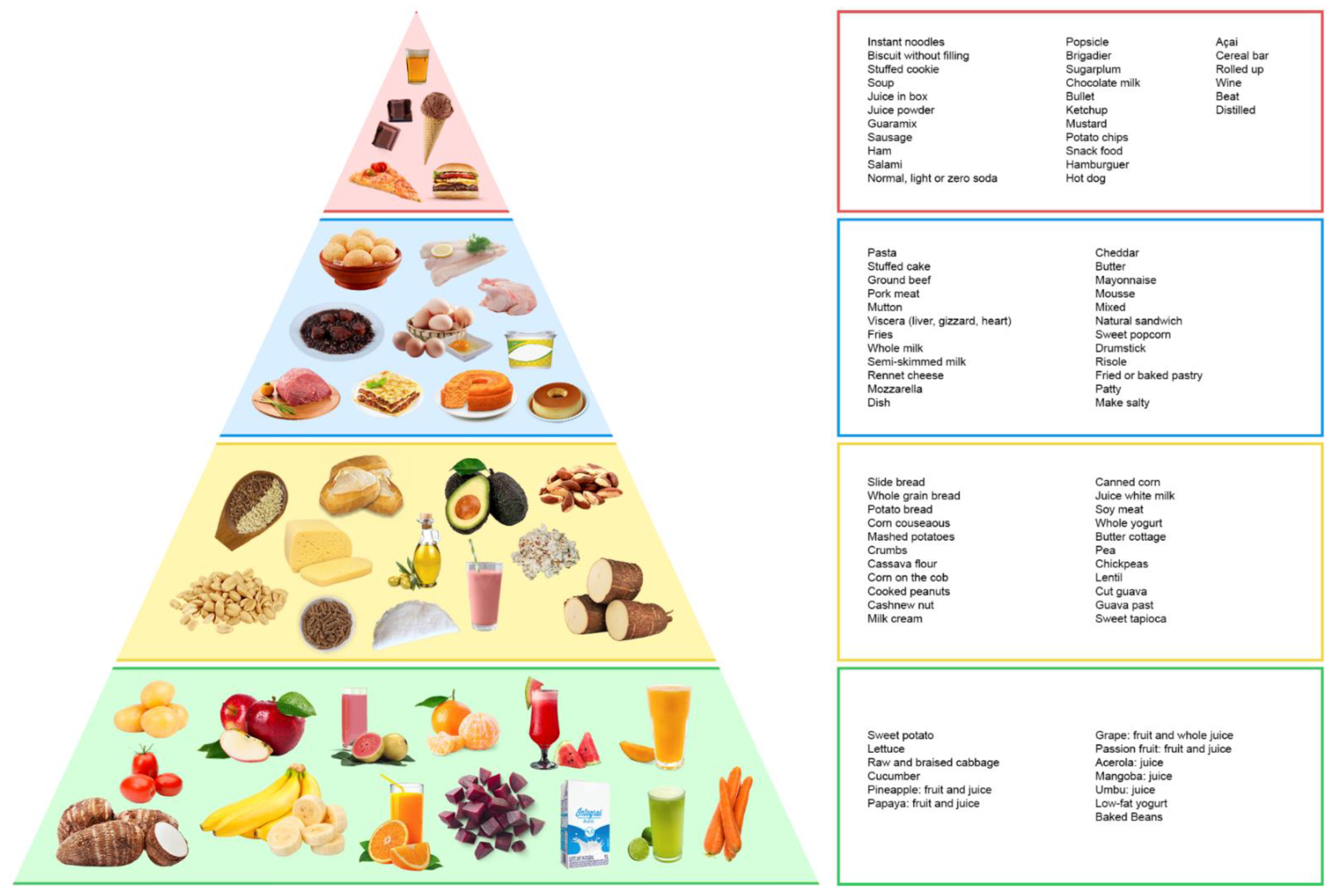
2.6. BALANCE Dietary Index
2.7. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- WHO. World Health Statistics 2020: Monitoring Health for the SDGs, Sustainable Development Goals; World Health Organization: Geneva, Switzerland, 2020. [Google Scholar]
- De Almeida, F.A.; Al, M.J.; Koymans, R.; Riistama, J.; Pauws, S.; Severens, J.L. Impact of hospitalisation on health-related quality of life in patients with chronic heart failure. Health Qual. Life Outcomes 2020, 18, 1–10. [Google Scholar] [CrossRef]
- Virani, S.S.; Alonso, A.; Benjamin, E.J.; Bittencourt, M.S.; Callaway, C.W.; Carson, A.P.; American Heart Association Council on Epidemiology and Prevention Statistics Committee and Stroke Statistics Subcommittee. Heart disease and stroke statistics—2020 update: A report from the American Heart Association. Circulation 2020, 141, e139–e596. [Google Scholar] [CrossRef] [PubMed]
- Benjamin, E.J.; Muntner, P.; Alonso, A.; Bittencourt, M.S.; Callaway, C.W.; Carson, A.P.; American Heart Association Council on Epidemiology and Prevention Statistics Committee and Stroke Statistics Subcommittee. Heart disease and stroke statistics—2019 update: A report from the American Heart Association. Circulation 2019, 139, e56–e528. [Google Scholar] [CrossRef] [PubMed]
- Stevens, B.; Pezzullo, L.; Verdian, L.; Tomlinson, J.; George, A.; Bacal, F. The economic burden of heart conditions in Brazil. Arq. Bras. Cardiol. 2018, 111, 29–36. [Google Scholar] [CrossRef]
- Bocchi, E.A.; Arias, A.; Verdejo, H.; Diez, M.; Gómez, E.; Castro, P. The reality of heart failure in Latin America. J. Am. Coll. Cardiol. 2013, 62, 949–958. [Google Scholar] [CrossRef]
- Savarese, G.; Lund, L.H. Global public health burden of heart failure. Card. Fail. Rev. 2017, 3, 7–11. [Google Scholar] [CrossRef]
- Ponikowski, P.; Anker, S.D.; AlHabib, K.F.; Cowie, M.R.; Force, T.L.; Hu, S.; Jaarsma, T.; Krum, H.; Rastogi, V.; Rohde, L.E.; et al. Heart failure: Preventing disease and death worldwide. ESC Heart Fail. 2014, 1, 4–25. [Google Scholar] [CrossRef]
- Go, A.S.; Mozaffarian, D.; Roger, V.L.; Benjamin, E.J.; Berry, J.D.; Blaha, M.J.; Turner, M.B. Heart disease and stroke statistics—2014 update: A report from the American Heart Association. Circulation 2014, 129, e28–e292. [Google Scholar] [CrossRef]
- Mozaffarian, D.; Benjamin, E.J.; Go, A.S.; Arnett, D.K.; Blaha, M.J.; Cushman, M.; Das, S.R.; de Ferranti, S.; Després, J.P.; Fullerton, H.J.; et al. Heart disease and stroke statistics-2016 update: A report from the American Heart Association. Circulation 2016, 133, e38–e360. [Google Scholar] [CrossRef]
- Evangelista, L.S.; Kagawa-Singer, M.; Dracup, K. Gender differences in health perception and meaning in person living with heart failure. Heart Lung 2001, 30, 167–176. [Google Scholar] [CrossRef]
- Days, I.A.; Ferreira, L.N. Functional assessment of patients with congestive heart failure using standardized scales. Rev. Saúde Com. 2011, 7, 116–126. [Google Scholar]
- Aquilani, R.; Opasich, C.; Verri, M.; Boschi, F.; Febo, O.; Pasini, E.; Pastoris, O. Is nutritional intake adequate in chronic heart failure patients? J. Am. Coll. Cardiol. 2003, 42, 1218–1223. [Google Scholar] [CrossRef]
- Chess, D.J.; Stanley, W.C. Role of diet and fuel overabundance in the development and progression of heart failure. Cardiovasc. Res. 2008, 79, 269–278. [Google Scholar] [CrossRef]
- Comitê Coordenador da Diretriz de Insuficiência Cardíaca; Rohde, L.E.P.; Montera, M.W.; Bocchi, E.A.; Clausell, N.O.; Albuquerque, D.C.; Rassi, S.; Colafranceschi, A.S.; Freitas, A.F.J.; Ferraz, A.S.; et al. Diretriz Brasileira de Insuficiência Cardíaca Crônica e Aguda. Arq. Bras. De Cardiol. 2018, 111, 436–539. [Google Scholar] [CrossRef]
- Papadaki, A.; Martínez-González, M.Á.; Alonso-Gómez, A.; Rekondo, J.; Salas-Salvadó, J.; Corella, D.; Ros, E.; Fitó, M.; Estruch, R.; Lapetra, J.; et al. Mediterranean diet and risk of heart failure: Results from the PREDIMED randomized controlled trial. Eur. J. Heart Fail. 2017, 19, 1179–1185. [Google Scholar] [CrossRef] [PubMed]
- D’Almeida, K.S.M.; Spillere, S.R.; Zuchinali, P.; Souza, G.C. Mediterranean diet and other dietary patterns in primary prevention of heart failure and changes in cardiac function markers: A systematic review. Nutrients 2018, 10, 58. [Google Scholar] [CrossRef] [PubMed]
- Tuttolomondo, A.; Simonetta, I.; Daidone, M.; Mogavero, A.; Ortello, A.; Pinto, A. Metabolic and Vascular Effect of the Mediterranean Diet. Int. J. Mol. Sci. 2019, 20, 4716. [Google Scholar] [CrossRef]
- Abu-Sawwa, R.; Dunbar, S.B.; Quyyumi, A.A.; Sattler, E.L.P. Nutrition intervention in heart failure: Should consumption of the DASH eating pattern be recommended to improve outcomes? Heart Fail. Rev. 2019, 24, 565–573. [Google Scholar] [CrossRef]
- Rifai, L.; Silver, M.A. A Review of the DASH Diet as an Optimal Dietary Plan for Symptomatic Heart Failure. Prog. Cardiovasc. Dis. 2016, 58, 548–554. [Google Scholar] [CrossRef]
- Salehi-Abargouei, A.; Maghsoudi, Z.; Shirani, F.; Azadbakht, L. Effects of Dietary Approaches to Stop Hypertension (DASH)-style diet on fatal or nonfatal cardiovascular diseases--incidence: A systematic review and meta-analysis on observational prospective studies. Nutrition 2013, 29, 611–618. [Google Scholar] [CrossRef]
- Brazil, Ministry of Health. Cardioprotective Nutrition: Guidebook for Primary Care Health Professionals; Ministry of Health, Hospital do Coração: Brasília, Brazil, 2018; 138p.
- Da Silva, J.T.; Bersch-Ferreira, Â.C.; Torreglosa, C.R.; Weber, B.; Levy, R.B. Development of a dietary index based on the Brazilian Cardioprotective Nutritional Program (BALANCE). Nutr. J. 2018, 17, 49. [Google Scholar] [CrossRef] [PubMed]
- Montera, M.W.; Pereira, S.B.; Colafranceschi, A.S.; de Almeida, D.R.; Tinoco, E.M.; Rocha, R.M.; Moura, L.A.Z.; Réa-Neto, A.; Mangini, S.; Braga, F.G.M.; et al. II Brazilian guideline for acute heart failure. Braz. Arch. Cardiol. 2009, 93, 2–65. [Google Scholar] [CrossRef]
- The Criteria Committee of the New York Heart Association. Nomenclature and Criteria for Diagnosis of Diseases of the Heart and Great Vessels, 9th ed.; Little, Brown & Co.: Boston, MA, USA, 1994; pp. 253–256. [Google Scholar]
- Matsudo, S.M.; Matsudo, V.R.; Araujo, T.; Andrade, D.; Andrade, E.; Oliveira, L.; Braggion, G. Level of physical activity in 56 adults, including and excluding walking, according to the knowledge of Agita São Paulo Program. Med. Sci. Sports Exerc. 2001, 33, S179. [Google Scholar]
- World Health Orgazination—WHO. Obesity: Preventing and Managins the Global Epidemic; World Health Orgazination—WHO: Geneva, Switzerland, 1997. [Google Scholar]
- Lipschitz, D.A. Screening for nutritional status in the elderly. Prim. Care 1994, 21, 55–67. [Google Scholar] [CrossRef]
- Furlan-Viebig, R.; Pastor-Valero, M. Development of a food frequency questionnaire to study diet and non-communicable 65 diseases. J. Public Health 2004, 38, 581–584. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Preparation and Use of Food Basead Dietary Guidelines; WHO 25 Technical Report Series 880; WHO: Geneva, Switzerland, 1998. [Google Scholar]
- BRASIL. M da S. Secretaria de Atenção à Saúde. Departamento de Atenção Básica. Política Nacional de Alimentação e 27 Nutrição. Brasília, 2012. Available online: http://189.28.128.100/nutricao/docs/geral/pnan2011.pdf (accessed on 28 July 2021).
- Brazil, Ministry of Health. Vigitel Brasil 2014: Protection and Risk Factors for Chronic Diseases by Telephone Survey; Ministry of Health: Brasília, Brazil, 2015.
- Benjamin, E.J.; Virani, S.S.; Callaway, C.W.; Chamberlain, A.M.; Chang, A.R.; Cheng, S.; Chiuve, S.E.; Cushman, M.; Delling, F.N.; Deo, R.; et al. Heart Disease and Stroke Statistics-2018 Update: A Report From the American Heart Association. Circulation 2018, 137, e67–e492. [Google Scholar] [CrossRef]
- Weber, B.; Bersch-Ferreira, Â.C.; Torreglosa, C.R.; Marcadenti, A.; Lara, E.S.; da Silva, J.T.; Costa, R.P.; Santos, R.H.; Berwanger, O.; Bosquetti, R.; et al. Implementation of a Brazilian Cardioprotective Nutritional (BALANCE) Program for improvement on quality of diet and secondary prevention of cardiovascular events: A randomized, multicenter trial. Am. Heart J. 2019, 215, 187–197. [Google Scholar] [CrossRef]
- Freitas, T.I.D.; Previdelli, A.N.; Ferreira, M.P.D.N.; Marques, K.M.; Goulart, R.M.M.; Aquino, R.D.C.D. Factors associated with diet quality of older adults. Rev. Nutr. 2017, 30, 297–306. [Google Scholar] [CrossRef]
- Gicevic, S.; Gaskins, A.J.; Fung, T.T.; Rosner, B.; Sabanovic, E.; Milesevic, J.; Kadvan, A.; Kremic, E.; Willett, W. Demographic and socio-economic predictors of diet quality among adults in Bosnia and Herzegovina. Public Health Nutr. 2019, 22, 3107–3117. [Google Scholar] [CrossRef]
- Han, K.T.; Park, E.C.; Kim, J.H.; Kim, S.J.; Park, S. Is marital status associated with quality of life? Health Qual. Life Outcomes 2015, 12, 1–10. [Google Scholar] [CrossRef]
- Donoho, C.J.; Seeman, T.E.; Sloan, R.P.; Crimmins, E.M. Marital status, marital quality, and heart rate variability in the MIDUS cohort. J. Fam. Psychol. 2015, 29, 290. [Google Scholar] [CrossRef] [PubMed]
- Alkerwi, A.A.; Sauvageot, N.; Buckley, J.D.; Donneau, A.-F.; Albert, A.; Guillaume, M.; Crichton, G.E. The potential impact of animal protein intake on global and abdominal obesity: Evidence from the Observation of Cardiovascular Risk Factors in Luxembourg (ORISCAV-LUX) study. Public Health Nutr. 2015, 18, 1831–1838. [Google Scholar] [CrossRef] [PubMed]
- Whitelock, E.; Ensaff, H. On your own: Older adults’ food choice and dietary habits. Nutrients 2018, 10, 413. [Google Scholar] [CrossRef] [PubMed]
- Freeman, A.M.; Morris, P.B.; Barnard, N.; Esselstyn, C.B.; Ros, E.; Agatston, A.; Devries, S.; O’Keefe, J.; Miller, M.; Ornish, D.; et al. Trending Cardiovascular Nutrition Controversies. J. Am. Coll. Cardiol. 2017, 69, 1172–1187. [Google Scholar] [CrossRef] [PubMed]
- Najjar, R.S.; Moore, C.E.; Montgomery, B.D. Consumption of a defined, plant-based diet reduces lipoprotein(a), inflammation, and other atherogenic lipoproteins and particles within 4 weeks. Clin. Cardiol. 2018, 41, 1062–1068. [Google Scholar] [CrossRef] [PubMed]
- Shah, B.; Newman, J.; Woolf, K.; Ganguzza, L.; Guo, Y.; Fisher, E.A.; Allen, N.; Hazen, S.L.; Larigakis, J.; Zhong, J.; et al. Anti-inflammatory effect of whole-food plant-based vegan diet vs the American Heart Association-recommended diet in patients with coronary artery disease: The randomized EVADE CAD Trial. Circulation 2017, 136, A23081. [Google Scholar] [CrossRef]
- Razavi, A.C.; Bazzano, L.A.; He, J.; Whelton, S.P.; Fernandez, C.; Ley, S.; Qi, L.; Krousel-Wood, M.; Harlan, T.S.; Kelly, T.N. Consumption of animal and plant foods and risk of left ventricular diastolic dysfunction: The Bogalusa Heart Study. ESC Heart Fail. 2020, 7, 2700–2710. [Google Scholar] [CrossRef]
- Park, K.B.; Park, H.A.; Kang, J.H.; Kim, K.; Cho, Y.G.; Jang, J. Animal and plant protein intake and body mass index and waist circumference in a Korean elderly population. Nutrients 2018, 10, 577. [Google Scholar] [CrossRef]
- IBGE. Pesquisa de Orçamentos Familiares 2017–2018. In Avaliação Nutricional da Disponibilidade Domiciliar de Alimentos no 90 Brasil; IBGE: Rio de Janeiro, Brazil, 2018. [Google Scholar]
- Torreglosa, C.R.; Sarti, F.M.; Bersch-Ferreira, Â.C.; Weber, B.; Santos, R.H.N.; Chiavegatto Filho, A.D.P. Diet quality and daily food expenditure in adults with cardiovascular disease in Brazil. Public Health Noteb. 2020, 36, e00225019. [Google Scholar]
- Fitó, M.; Guxens, M.; Corella, D.; Sáez, G.; Estruch, R.; de la Torre, R.; Francés, F.; Cabezas, C.; López-Sabater, M.D.C.; Marrugat, J.; et al. Effect of a traditional Mediterranean diet on lipoprotein oxidation: A randomized controlled trial. Arch. Intern. Med. 2007, 167, 1195–1203. [Google Scholar] [CrossRef]
- Appel, L.J.; Moore, T.J.; Obarzanek, E.; Vollmer, W.M.; Svetkey, L.P.; Sacks, F.M.; Bray, G.A.; Vogt, T.M.; Cutler, J.A.; Windhauser, M.M.; et al. A clinical trial of the effects of dietary patterns on blood pressure. DASH Collaborative Research Group. N. Engl. J. Med. 1997, 336, 1117–1124. [Google Scholar] [CrossRef] [PubMed]
- Brazilian Diabetes Society. Guidelines of the Brazilian Society of Diabetes 2019–2020; Brazilian Diabetes Society: Sao Paulo, Brazil, 2019. [Google Scholar]
- Izar, M.C.O.; Lottenberg, A.M.; Giraldez, V.Z.R.; Santos Filho, R.D.D.; Machado, R.M.; Bertolami, A.; Assad, M.H.V.; Saraiva, J.F.K.; Faludi, A.A.; Moreira, A.S.B.; et al. Positioning on Fat Consumption and Cardiovascular Health–2021. Braz. Arch. Cardiol. 2021, 116, 160–212. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Huang, M.; Zhuang, P.; Jiao, J.; Chen, X.; Wang, J.; Wu, Y. Exposure to acrylamide and the risk of cardiovascular diseases in the National Health and Nutrition Examination Survey 2003–2006. Environ. Int. 2018, 117, 154–163. [Google Scholar] [CrossRef] [PubMed]
- De Jarnett, N.; Conklin, D.J.; Riggs, D.W.; Myers, J.A.; O’Toole, T.E.; Hamzeh, I.; Wagner, S.; Chugh, A.; Ramos, K.S.; Srivastava, S.; et al. Acrolein exposure is associated with increased cardiovascular disease risk. J. Am. Heart Assoc. 2014, 6, e000934. [Google Scholar] [CrossRef] [PubMed]
- Rancière, F.; Lyons, J.G.; Loh, V.H.; Botton, J.; Galloway, T.; Wang, T.; Shaw, J.E.; Magliano, D.J. Bisphenol A and the risk of cardiometabolic disorders: A systematic review with meta-analysis of the epidemiological evidence. Environ. Health 2015, 31, 46. [Google Scholar] [CrossRef]
- Santos, R.D.; Gagliardi, A.C.M.; Xavier, H.T.; Magnoni, C.D.; Cassani, R.; Lottenberg, A.M.P.; Casella Filho, A.; Araújo, D.B.; Cesena, F.Y.; Alves, R.J.; et al. I Diretriz sobre o consumo de gorduras e saúde cardiovascular. Arq. Bras. Cardiol. 2013, 100, 1–40. [Google Scholar] [CrossRef]
- Zhong, G.C.; Gu, H.T.; Peng, Y.; Wang, K.; Wu, Y.Q.; Hu, T.Y.; Jing, F.C.; Hao, F.B. Association of ultra-processed food consumption with cardiovascular mortality in the US population: Long-term results from a large prospective multicenter study. Int. J. Behav. Nutr. Phys. Act. 2021, 3, 21. [Google Scholar] [CrossRef]
- Bonaccio, M.; Di Castelnuovo, A.; Costanzo, S.; De Curtis, A.; Persichillo, M.; Sofi, F.; Cerletti, C.; Donati, M.B.; de Gaetano, G.; Iacoviello, L. Ultra-processed food consumption is associated with increased risk of all-cause and cardiovascular mortality in the Moli-sani Study. Am. J. Clin. Nutr. 2021, 113, 446–455. [Google Scholar] [CrossRef]
- Blanco-Rojo, R.; Sandoval-Insausti, H.; López-Garcia, E.; Graciani, A.; Ordovás, J.M.; Banegas, J.R.; Rodríguez-Artalejo, F.; Guallar-Castillón, P. Consumption of Ultra-Processed Foods and Mortality: A National Prospective Cohort in Spain. Mayo Clin. Proc. 2019, 94, 2178–2188. [Google Scholar] [CrossRef]
- Moreira, P.V.; Hyseni, L.; Moubarac, J.C.; Martins, A.P.B.; Baraldi, L.G.; Capewell, S.; O’Flaherty, M.; Guzman-Castillo, M. Effects of reducing processed culinary ingredients and ultra-processed foods in the Brazilian diet: A cardiovascular modelling study. Public Health Nutr. 2018, 21, 181–188. [Google Scholar] [CrossRef]
- Rassi, S.; Barretto, A.C.; Porto, C.C.; Pereira, C.R.; Calaça, B.W.; Rassi, D.C. Sobrevida e fatores prognósticos na insuficiência cardíaca sistólica com início recente dos sintomas. Arq. Bras. Cardiol. 2005, 84, 309–313. [Google Scholar] [CrossRef] [PubMed]
- Berry, C.; Clark, A.L. Catabolism in chronic heart failure. Eur. Heart J. 2000, 21, 521–532. [Google Scholar] [CrossRef] [PubMed]
- de Araújo, L.A.S.; Firmino, S.M.; Curcelli, E.M.; Martins, M.J.V.; Gusmão, A.A.; Oliveira, M.C.D.; Milan-Mattos, J.C.; dos Santos, P.B.; Marinho, R.S.; Valadão, T.F.C.; et al. Avaliação da função cardiovascular e qualidade de vida em pacientes com insuficiência cardíaca com e sem comportamento sedentário: Sedentarismo e insuficiência cardíaca. Medicina 2021, 54, e173130. [Google Scholar]
- Son, Y.J.; Choi, J.; Lee, H.J. Effectiveness of nurse-led heart failure self-care education on health outcomes of heart failure patients: A systematic review and meta-analysis. Int. J. Environ. Res. Public Health 2020, 17, 6559. [Google Scholar] [CrossRef] [PubMed]
| Variables | Adherence | Green Group | Yellow Group | Blue Group | Red Group | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| B * | IC95 | B * | IC95 | B * | IC95 | IRR ** | IC95 | B * | IC95 | |
| Age in years (n = 239) | 0.00 | −0.00; 0.01 | 0.00 | −0.00; 0.00 | −0.00 | −0.03; 0.02 | 1.00 | 0.99; 1.01 | 0.00 | −0.00; 0.02 |
| Gender (n = 239) | ||||||||||
| Male (n = 124) | Ref. | Ref. | Ref. | Ref. | Ref. | |||||
| Female (n = 115) | 0.09 | −0.02; 0.22 | 0.06 | −0.02; 0.15 | −0.00 | −0.83; 0.81 | 1.27 | 0.87; 1.01 | −0.02 | −0.36; 0.31 |
| Race (n = 231) | ||||||||||
| Not black (n = 61) | Ref. | Ref. | Ref. | Ref. | Ref. | |||||
| Black (n = 170) | −0.03 | −0.16; 0.10 | 0.01 | −0.08; 0.11 | −0.15 | −1.13; 0.82 | 1.05 | 0.72; 1.52 | −0.06 | −0.45; 0.32 |
| Marital Status (n = 234) | ||||||||||
| Single (n = 108) | Ref. | Ref. | Ref. | Ref. | Ref. | |||||
| With partner (n = 126) | 0.00 | −0.11; 0.12 | 0.09 | 0.00; 0.17 | −0.58 | −1.42; 0.25 | 0.91 | 0.65; 1.25 | 0.09 | −0.24; 0.43 |
| Income (n = 239) | ||||||||||
| <1 MW (n = 164) | Ref. | Ref. | Ref. | Ref. | Ref. | |||||
| ≥1 MW (n = 75) | −0.06 | −0.21; 0,09 | −0.07 | −0.19; 0.04 | −0.09 | −1.20; 1.02 | 0.98 | 0.67; 1.42 | −0.06 | −0.45; 0.32 |
| Type of service (n = 239) | ||||||||||
| Private (n = 77) | Ref. | Ref. | Ref. | Ref. | Ref. | |||||
| Public (n = 162) | −0.05 | −0.22; 0.10 | 0.01 | −0.10; 0.14 | −0.60 | −1.70; 0.49 | 0.90 | 0.56; 1.45 | −0.35 | −0.87; 0.15 |
| Hypertension (n = 237) | ||||||||||
| No (n = 71) | Ref. | Ref. | Ref. | Ref. | Ref. | |||||
| yes (n = 166) | −0.03 | −0.16; 0.10 | −0.08 | −0.18; 0.01 | 0.82 | −0.08; 1.73 | 1.33 | 0.95; 1.86 | −0.04 | −0.42; 0.32 |
| Diebetes Mellitus (n = 237) | ||||||||||
| No (n = 144) | Ref. | Ref. | Ref. | Ref. | Ref. | |||||
| Yes (n = 93) | 0.10 | −0.02; 0.23 | 0.00 | −0.08; 0.10 | −0.35 | −1.20; 0.49 | 1.30 | 0.93; 1.82 | 0.41 | 0.05; 0.77 |
| BMI (n = 239) | ||||||||||
| Not overweight (n = 95) | Ref. | Ref. | Ref. | Ref. | Ref. | |||||
| Overweight (n = 144) | −0.02 | −0.14; 0.10 | 0.01 | −0.07; 0.11 | 0.14 | −0.68; 0.98 | 0.54 | 0.38; 0.78 | 0.09 | −0.26; 0.44 |
| Physical Activity (n = 233) | ||||||||||
| Absent/Mild/Moderate (n = 197) | Ref. | Ref. | Ref. | Ref. | Ref. | |||||
| High Level (n = 36) | 0.02 | −0.14; 0.18 | 0.04 | −0.07; 0.16 | 0.23 | −1.05; 1.52 | 1.21 | 0.81; 1.82 | −0.22 | −0.71; 0.27 |
| Alcoholic beverage (n = 237) | ||||||||||
| No (n = 202) | Ref. | Ref. | Ref. | Ref. | Ref. | |||||
| Yes (n = 35) | −0.04 | −0.21; 0.13 | 0.05 | −0.07; 0.17 | −0.47 | −1.75; 0.80 | 0.58 | 0.29; 1.16 | −0.46 | −0.99; 0.07 |
| Smoking (n = 237) | ||||||||||
| No (n = 226) | Ref. | Ref. | Ref. | Ref. | Ref. | |||||
| Yes (n = 11) | 0.05 | −0.20; 0.32 | 0.02 | −0.16; 0.21 | 0.33 | −1.44; 2.11 | 1.68 | 0.77; 3.65 | −0.17 | −0.97; 0.61 |
| Logistic Regression | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Adherence | Green Group | Yellow Group | Blue Group | Red Group | ||||||
| OR | CI95 | OR | CI95 | OR | CI95 | OR | CI95 | OR | CI95 | |
| NYHA | ||||||||||
| 1 and 2 | Ref. | Ref. | Ref. | Ref. | Ref. | |||||
| 3 and 4 | 0.98 | 0.89; 1.08 | 1.14 | 0.93; 1.40 | 0.99 | 0.85; 1.15 | 1.01 | 0.84; 1.21 | 0.87 | 0.73; 1.02 |
| EF | ||||||||||
| Changed | Ref. | Ref. | Ref. | Ref. | Ref. | |||||
| Preserved | 1.05 | 0.98; 1.13 | 0.91 | 0.78; 1.05 | 1.09 | 0.98; 1.22 | 1.00 | 0.89; 1.13 | 1.11 | 0.98; 1.26 |
| Outcome in 30 days | ||||||||||
| Uneventful | Ref. | Ref. | Ref. | Ref. | Ref. | |||||
| Intercurring | 0.98 | 0.92; 1.04 | 0.95 | 0.83; 1.09 | 0.95 | 0.86; –1.05 | 1.05 | 0.94; 1.18 | 0.98 | 0.88; 1.10 |
| Linear regression | ||||||||||
| β * | CI95 | β * | CI95 | β * | CI95 | β * | CI95 | β * | CI95 | |
| LVESD | −0.01 | −0.06; 0.03 | 0.05 | −0.05; 0.17 | −0.09 | −0.17; −0.01 | 0.00 | −0.08; 0.96 | 0.03 | −0.06; 0.12 |
| LVEDD | −0.00 | −0.03; 0.02 | 0.01 | −0.06; 0.08 | −0.00 | −0.05; 0.05 | 0.01 | −0.04; 0.07 | −0.03 | −0.10; 0.02 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Costa, J.O.; Aidar, F.J.; Barbosa, J.S.; Alves, L.V.S.; Oliveira, V.B.; Oliveira, L.M.S.M.d.; Rocha, R.M.S.; Vieira, D.A.d.S.; Costa, I.M.N.B.d.C.; Souza, M.F.C.d.; et al. BALANCE Dietary Index in Patients with Heart Failure, and Its Adherence in Sergipe, Brazil. Clin. Pract. 2022, 12, 383-395. https://doi.org/10.3390/clinpract12030043
Costa JO, Aidar FJ, Barbosa JS, Alves LVS, Oliveira VB, Oliveira LMSMd, Rocha RMS, Vieira DAdS, Costa IMNBdC, Souza MFCd, et al. BALANCE Dietary Index in Patients with Heart Failure, and Its Adherence in Sergipe, Brazil. Clinics and Practice. 2022; 12(3):383-395. https://doi.org/10.3390/clinpract12030043
Chicago/Turabian StyleCosta, Jamille Oliveira, Felipe J. Aidar, Juliana Santos Barbosa, Luciana Vieira Sousa Alves, Victor Batista Oliveira, Larissa Marina Santana Mendonça de Oliveira, Raysa Manuelle Santos Rocha, Diva Aliete dos Santos Vieira, Ingrid Maria Novais Barros de Carvalho Costa, Márcia Ferreira Cândido de Souza, and et al. 2022. "BALANCE Dietary Index in Patients with Heart Failure, and Its Adherence in Sergipe, Brazil" Clinics and Practice 12, no. 3: 383-395. https://doi.org/10.3390/clinpract12030043
APA StyleCosta, J. O., Aidar, F. J., Barbosa, J. S., Alves, L. V. S., Oliveira, V. B., Oliveira, L. M. S. M. d., Rocha, R. M. S., Vieira, D. A. d. S., Costa, I. M. N. B. d. C., Souza, M. F. C. d., Oliveira, J. L. M., Baumworcel, L., Neves, E. B., Díaz-de-Durana, A. L., Almeida-Santos, M. A., & Sousa, A. C. S. (2022). BALANCE Dietary Index in Patients with Heart Failure, and Its Adherence in Sergipe, Brazil. Clinics and Practice, 12(3), 383-395. https://doi.org/10.3390/clinpract12030043








