Local Defence System in Healthy Lungs
Abstract
:1. Introduction
2. Materials and Methods
2.1. Material Characteristics of Subjects
2.2. Immunohistochemical Analysis
2.3. Statistical Analysis
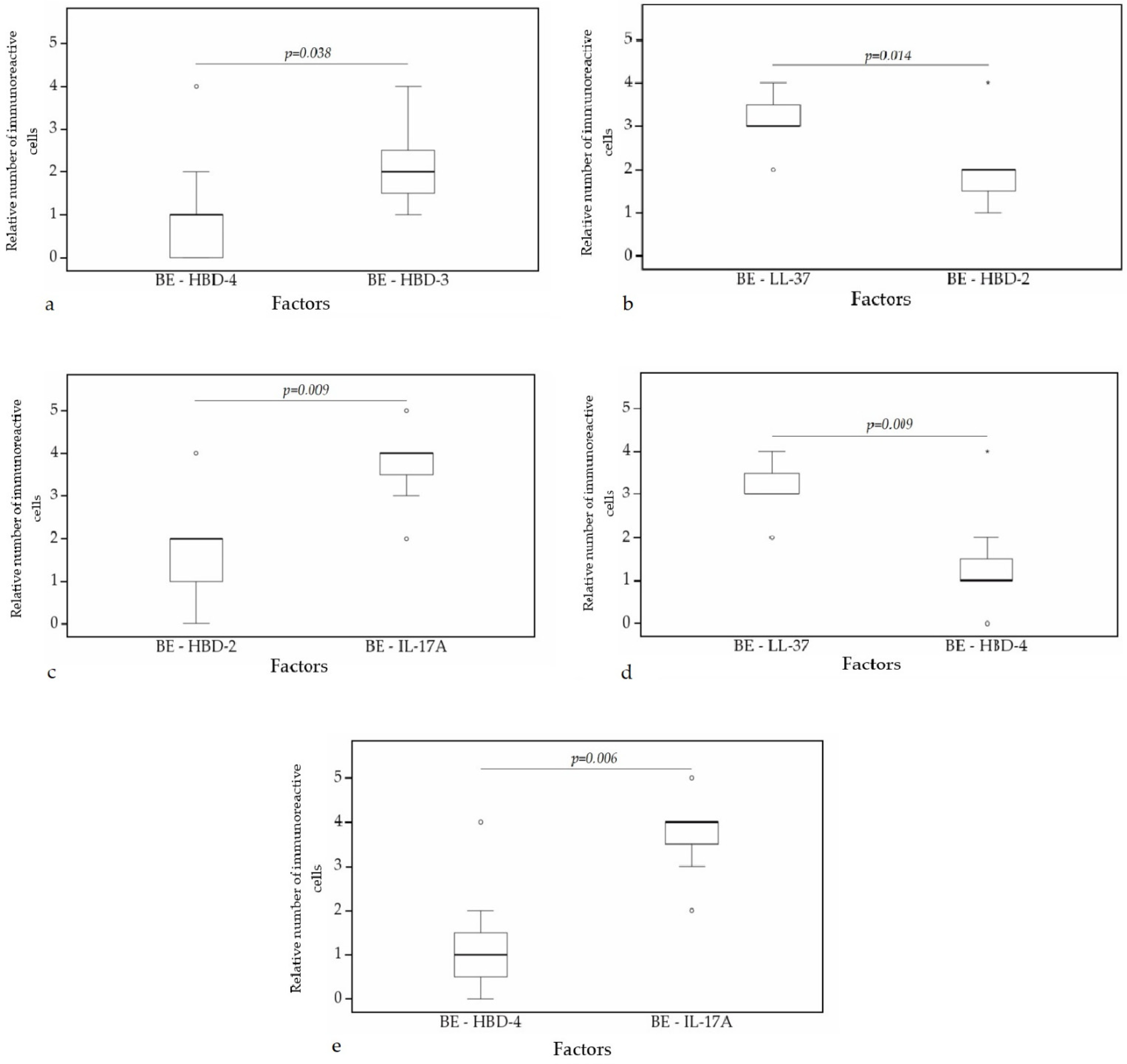
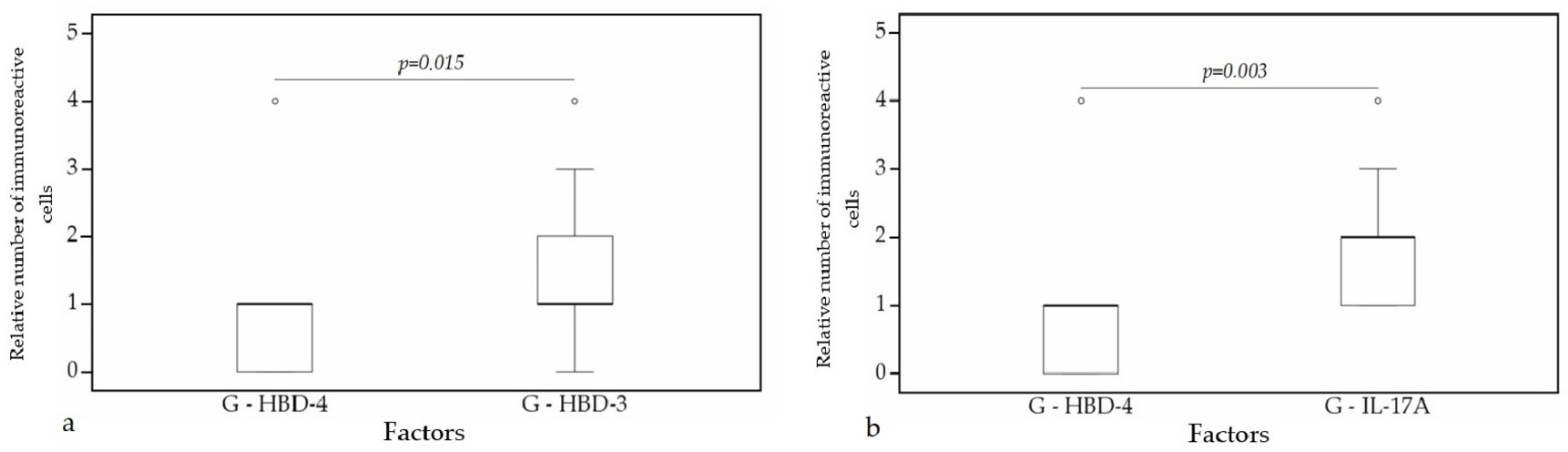
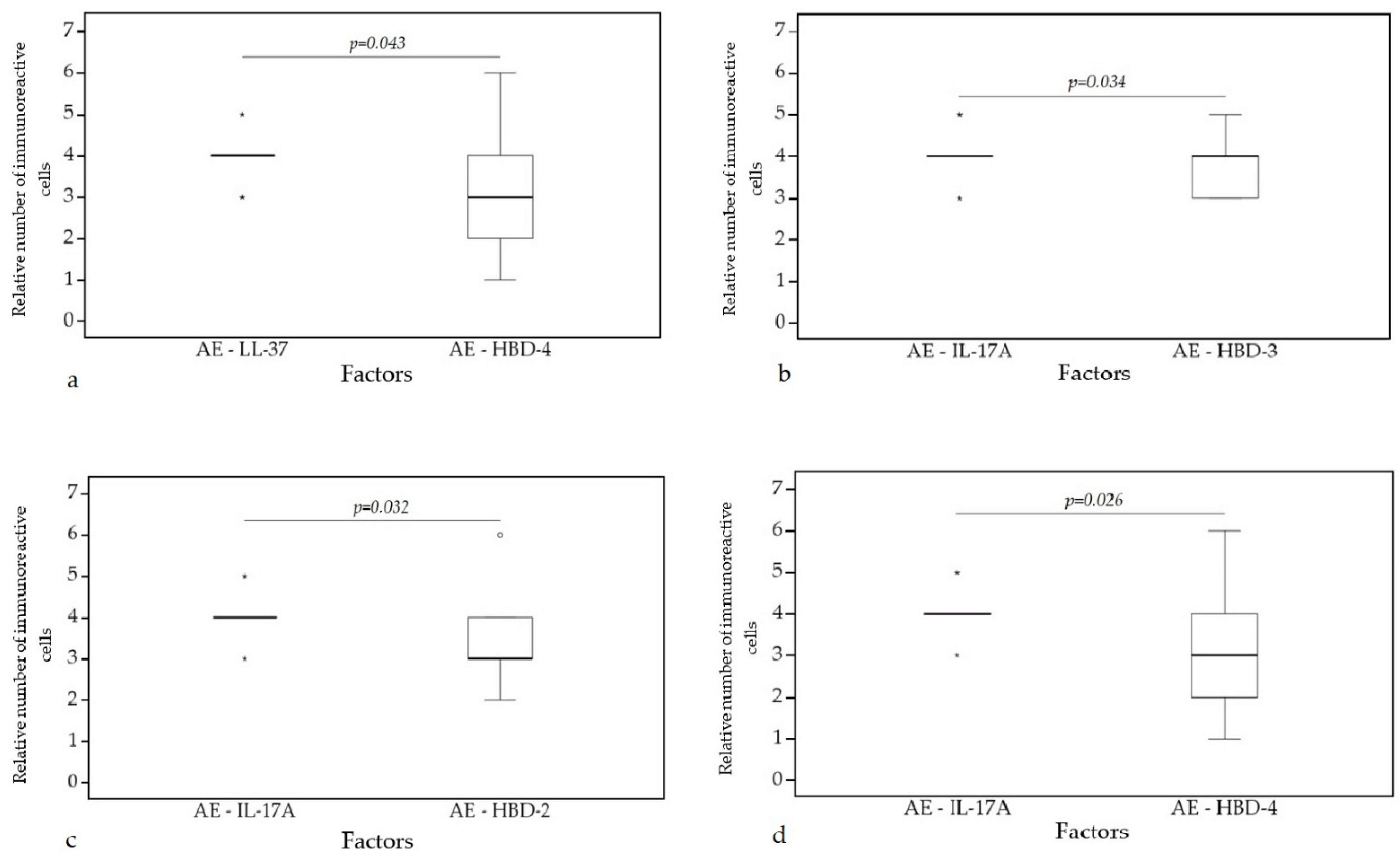
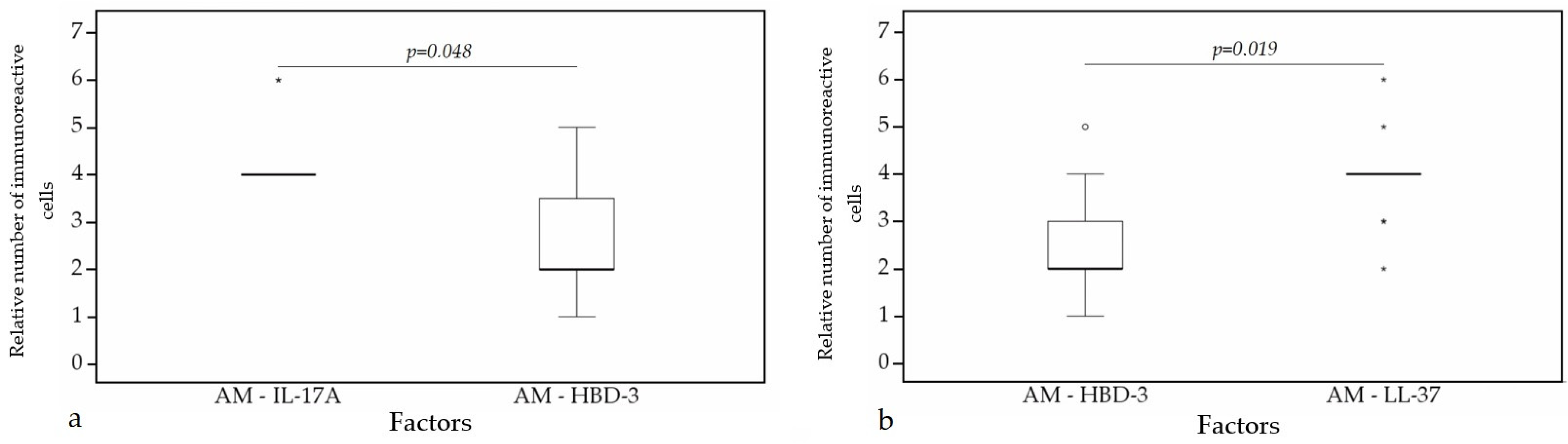
3. Results
3.1. Tissue Review
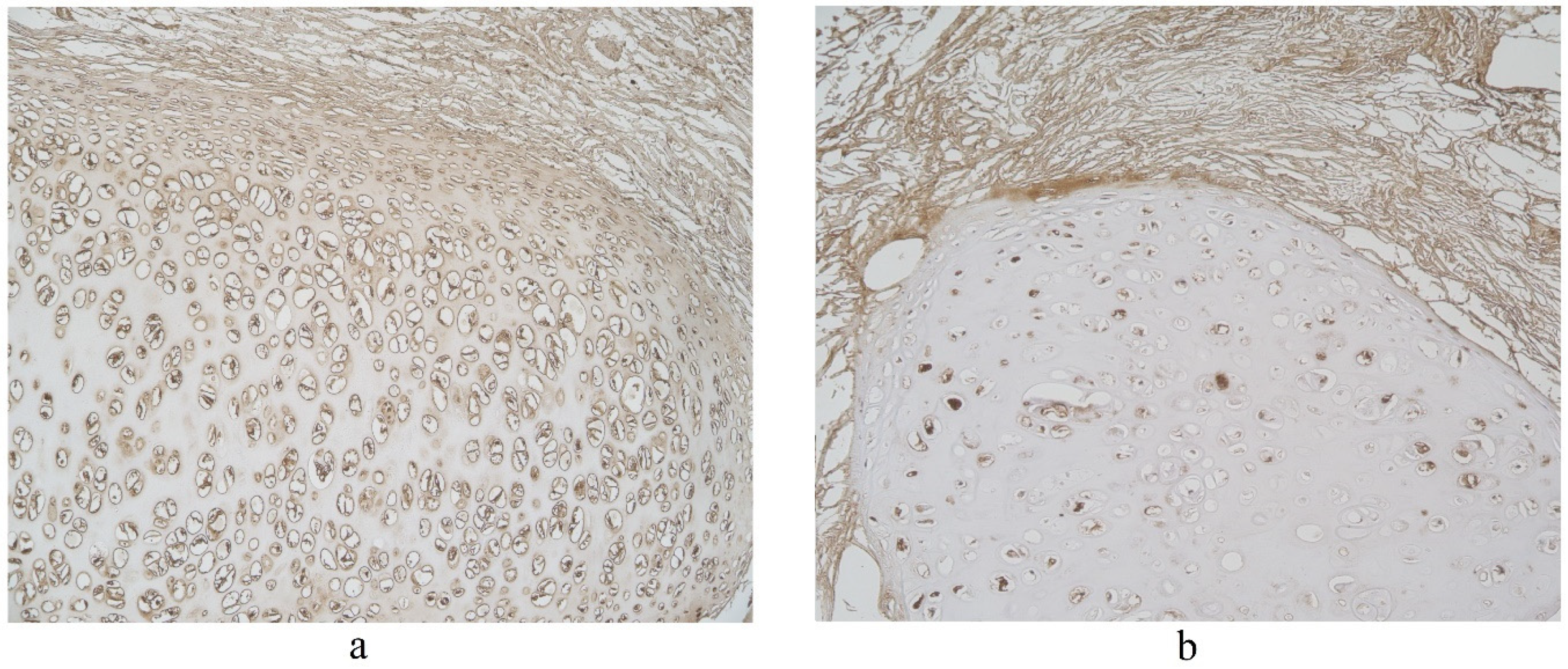
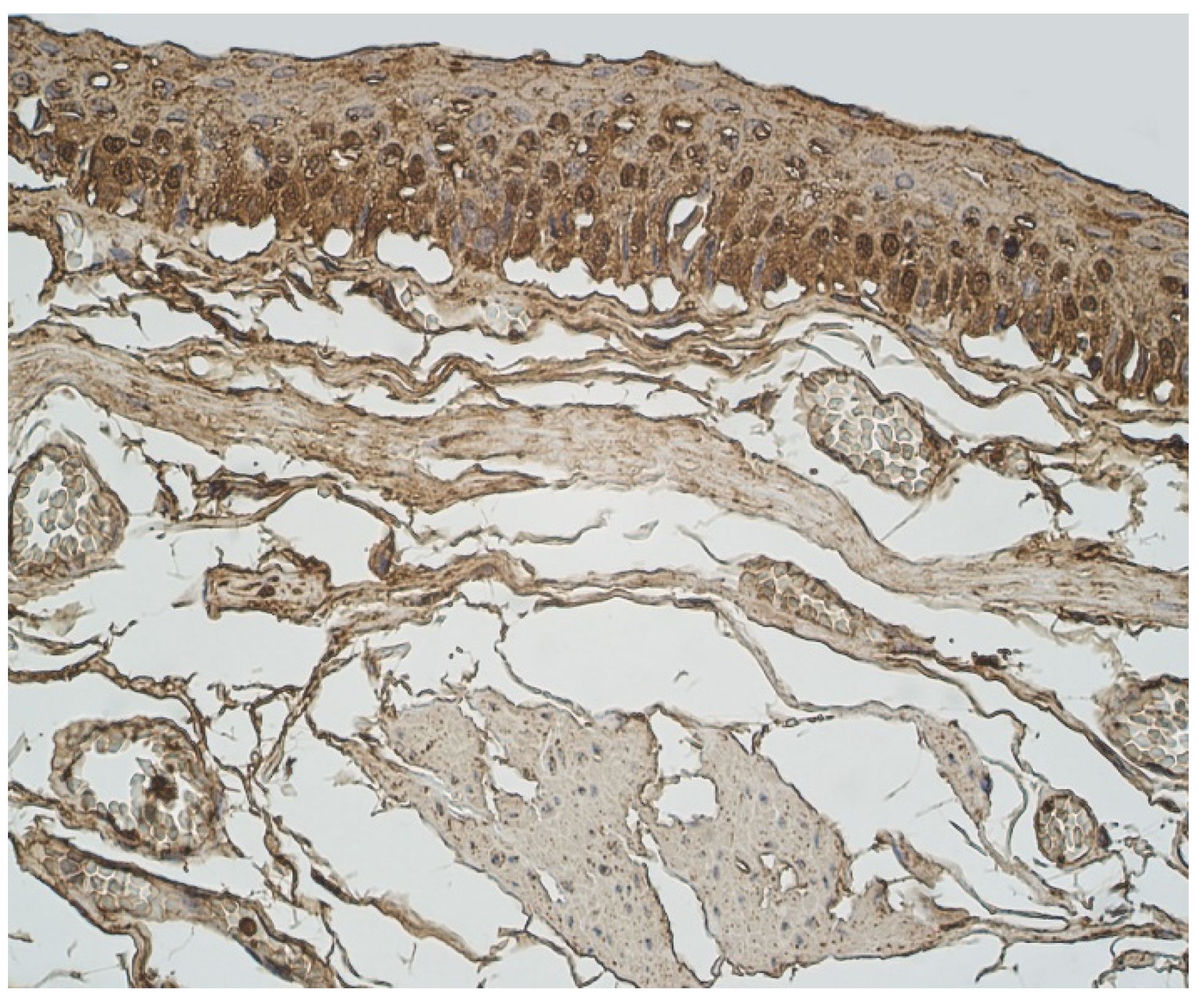
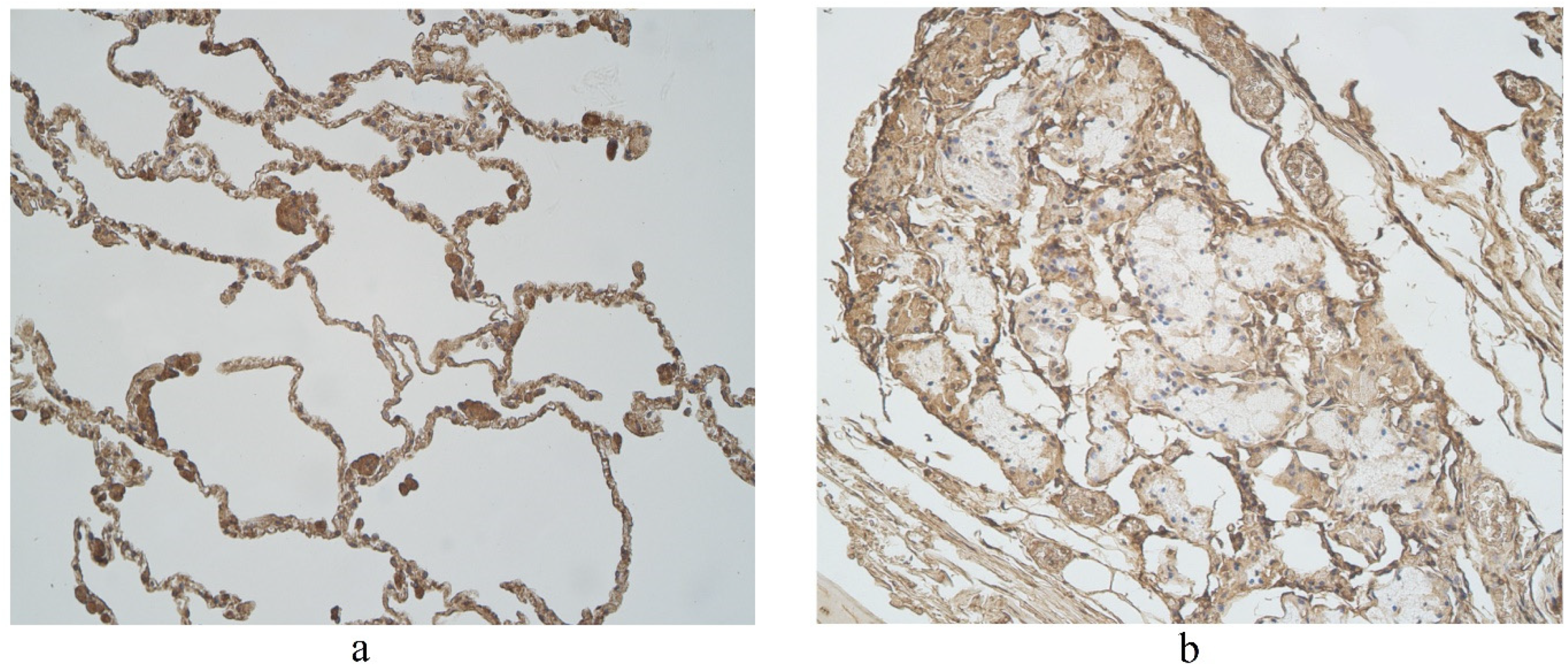
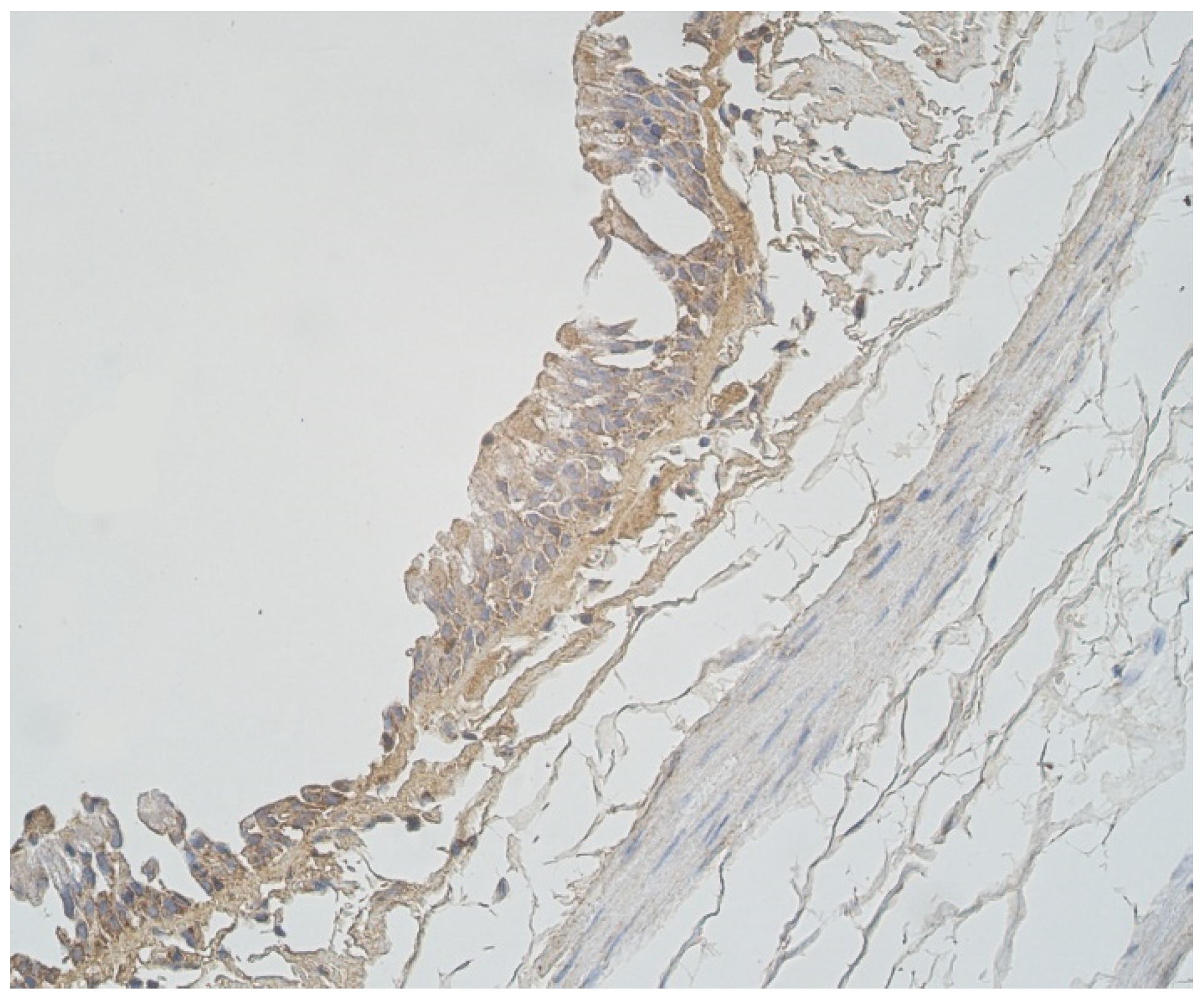
3.2. Immunohistochemical (IMH) Data
3.3. Statistical Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Doss, M.; White, M.R.; Tecle, T.; Hartshorn, K.L. Human defensins and LL-37 in mucosal immunity. J. Leukoc. Biol. 2010, 87, 79–92. [Google Scholar] [CrossRef] [PubMed]
- Bals, R.; Wang, X.; Zasloff, M.; Wilson, J.M. The peptide antibiotic LL-37/hCAP-18 is expressed in epithelia of the human lung where it has broad antimicrobial activity at the airway surface. Proc. Natl. Acad. Sci. USA 1998, 95, 9541–9546. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Von Haussen, J.; Koczulla, R.; Shaykhiev, R.; Herr, C.; Pinkenburg, O.; Reimer, D.; Wiewrodt, R.; Biesterfeld, S.; Aigner, A.; Czubayko, F.; et al. The host defence peptide LL-37/hCAP-18 is a growth factor for lung cancer cells. Lung Cancer 2008, 59, 12–23. [Google Scholar] [CrossRef] [PubMed]
- Wanke, D.; Mauch-Mücke, K.; Holler, E.; Hehlgans, T. Human beta-defensin-2 and-3 enhance pro-inflammatory cytokine expression induced by TLR ligands via ATP-release in a P2X7R dependent manner. Immunobiology 2016, 221, 1259–1265. [Google Scholar] [CrossRef]
- Johansen, C.; Bertelsen, T.; Ljungberg, C.; Mose, M.; Iversen, L. Characterization of TNF-α– and IL-17A–Mediated Synergistic Induction of DEFB4 Gene Expression in Human Keratinocytes through IκBζ. J. Investig. Dermatol. 2016, 136, 1608–1616. [Google Scholar] [CrossRef] [Green Version]
- Ganz, T.; Selsted, M.E.; Szklarek, D.; Harwig, S.S.; Daher, K.; Bainton, D.F.; Lehrer, R.I. Defensins. Natural peptide antibiotics of human neutrophils. J. Clin. Investig. 1985, 76, 1427–1435. [Google Scholar] [CrossRef]
- Diamond, G.; Zasloff, M.A.; Eck, H.S.; Brasseur, M.; Maloy, W.L.; Bevins, C. Tracheal antimicrobial peptide, a cysteine-rich peptide from mammalian tracheal mucosa: Peptide isolation and cloning of a cDNA. Proc. Natl. Acad. Sci. USA 1991, 88, 3952–3956. [Google Scholar] [CrossRef] [Green Version]
- Harder, J.; Meyer-Hoffert, U.; Teran, L.M.; Schwichtenberg, L.; Bartels, J.; Maune, S.; Schroder, J.M. Mucoid Pseudomonas aeruginosa, TNF-α, and IL-1 β, but Not IL-6, induce human β-defensin-2 in respiratory epithelia. Am. J. Respir. Cell Mol. Biol. 2000, 22, 714–721. [Google Scholar] [CrossRef] [PubMed]
- Li, A.; Gan, Y.; Wang, R.; Liu, Y.; Ma, T.; Huang, M.; Cui, X. IL-22 Up-Regulates β-Defensin-2 Expression in Human Alveolar Epithelium via STAT3 but Not NF-κB Signaling Pathway. Inflammation 2015, 38, 1191–1200. [Google Scholar] [CrossRef]
- Krisanaprakornkit, S.; Jotikasthira, D.; Dale, B.A. Intracellular calcium in signaling human β-defensin-2 expression in oral epithelial cells. J. Dent. Res. 2003, 82, 877–882. [Google Scholar] [CrossRef] [PubMed]
- Scharf, S.; Zahlten, J.; Szymanski, K.; Hippenstiel, S.; Suttorp, N.; N’Guessan, P.D. Streptococcus pneumoniae induces human β-defensin-2 and -3 in human lung epithelium. Exp. Lung Res. 2012, 38, 100–110. [Google Scholar] [CrossRef] [PubMed]
- Bals, R.; Wang, X.; Wu, Z.; Freeman, T.; Bafna, V.; Zasloff, M.; Wilson, J.M. Human beta-defensin 2 is a salt-sensitive peptide antibiotic expressed in human lung. J. Clin. Investig. 1998, 102, 874–880. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schröder, J.M.; Harder, J. Human beta-defensin-2. Int. J. Biochem. Cell Biol. 1999, 31, 645–651. [Google Scholar] [CrossRef]
- Pinkerton, J.W.; Kim, R.Y.; Koeninger, L.; Armbruster, N.S.; Hansbro, N.G.; Brown, A.C.; Jayaraman, R.; Shen, S.; Malek, N.; Cooper, M.A.; et al. Human β-defensin-2 suppresses key features of asthma in murine models of allergic airways disease. Clin. Exp. Allergy 2021, 51, 120–131. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Chen, C.; Di, T.; Yang, J.; Wang, K.; Zhu, Y.; Zhu, R.; Zhou, A.; Qian, Y. Human β-Defensin-2 Improves Hyperoxia-Induced Lung Structural and Functional Injury in Neonatal Rats. Med. Sci. Monit. 2019, 25, 6074–6084. [Google Scholar] [CrossRef]
- Yang, D.; Chertov, O.; Bykovskaia, N.; Chen, Q.; Buffo, M.J.; Shogan, J.; Anderson, M.; Schröder, J.M.; Wang, J.M.; Howard, O.M.Z.; et al. β-defensins: Linking innate and adaptive immunity through dendritic and T cell CCR6. Science 1999, 286, 525–528. [Google Scholar] [CrossRef] [PubMed]
- McGlasson, S.L.; Semple, F.; MacPherson, H.; Gray, M.; Davidson, D.J.; Dorin, J.R. Human β-defensin 3 increases the TLR9-dependent response to bacterial DNA. Eur. J. Immunol. 2017, 47, 658–664. [Google Scholar] [CrossRef] [Green Version]
- Harder, J.; Bartels, J.; Christophers, E.; Schroder, J.M. Isolation and characterization of human beta -defensin-3, a novel human inducible peptide antibiotic. J. Biol. Chem. 2001, 276, 5707–5713. [Google Scholar] [CrossRef] [Green Version]
- Albanesi, C.; Fairchild, H.R.; Madonna, S.; Scarponi, C.; De Pità, O.; Leung, D.Y.; Howell, M.D. IL-4 and IL-13 negatively regulate TNF-alpha- and IFN-gamma-induced beta-defensin expression through STAT-6, suppressor of cytokine signaling (SOCS)-1, and SOCS-3. J. Immunol. 2007, 179, 984–992. [Google Scholar] [CrossRef] [Green Version]
- Maisetta, G.; Batoni, G.; Esin, S.; Florio, W.; Bottai, D.; Favilli, F.; Campa, M. In vitro bactericidal activity of human beta-defensin 3 against multidrug-resistant nosocomial strains. Antimicrob. Agents Chemother. 2006, 50, 806–809. [Google Scholar] [CrossRef] [Green Version]
- Jin, G.; Kawsar, H.I.; Hirsch, S.A.; Zeng, C.; Jia, X.; Feng, Z.; Ghosh, S.K.; Zheng, Q.Y.; Zhou, A.; McIntyre, T.M.; et al. An antimicrobial peptide regulates tumor-associated macrophage trafficking via the chemokine receptor CCR2, a model for tumorigenesis. PLoS ONE 2010, 5, e10993. [Google Scholar] [CrossRef] [Green Version]
- Niyonsaba, F.; Ushio, H.; Nakano, N.; Ng, W.; Sayama, K.; Hashimoto, K.; Nagaoka, I.; Okumura, K.; Ogawa, H. Antimicrobial peptides human beta-defensins stimulate epidermal keratinocyte migration, proliferation and production of proinflammatory cytokines and chemokines. J. Investig. Dermatol. 2007, 127, 594–604. [Google Scholar] [CrossRef] [Green Version]
- Lehrer, R.I.; Lu, W. α-Defensins in human innate immunity. Immunol. Rev. 2012, 245, 84–112. [Google Scholar] [CrossRef]
- Schneider, J.J.; Unholzer, A.; Schaller, M.; Schäfer-Korting, M.; Korting, H.C. Human defensins. J. Mol. Med. 2005, 83, 587–595. [Google Scholar] [CrossRef] [PubMed]
- Harder, J.; Meyer-Hoffert, U.; Wehkamp, K.; Schwichtenberg, L.; Schröder, J.M. Differential gene induction of human β-defensins (hBD-1,-2,-3, and-4) in keratinocytes is inhibited by retinoic acid. J. Investig. Dermatol. 2004, 123, 522–529. [Google Scholar] [CrossRef] [Green Version]
- Yanagi, S.; Ashitani, J.; Ishimoto, H.; Date, Y.; Mukae, H.; Chino, N.; Nakazato, M. Isolation of human beta-defensin-4 in lung tissue and its increase in lower respiratory tract infection. Respir. Res. 2005, 6, 130. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, X.; Niyonsaba, F.; Ushio, H.; Hara, M.; Yokoi, H.; Matsumoto, K.; Satio, H.; Nagaoka, I.; Ikeda, S.; Okumura, K.; et al. Antimicrobial peptides human β-defensin (hBD)-3 and hBD-4 activate mast cells and increase skin vascular permeability. Eur. J. Immunol. 2007, 37, 434–444. [Google Scholar] [CrossRef] [PubMed]
- Musumeci, G.; Carnazza, M.L.; Loreto, C.; Leonardi, R.; Loreto, C. β-Defensin-4 (HBD-4) is expressed in chondrocytes derived from normal and osteoarthritic cartilage encapsulated in PEGDA scaffold. Acta Histochem. 2012, 114, 805–812. [Google Scholar] [CrossRef] [PubMed]
- Xhindoli, D.; Pacor, S.; Benincasa, M.; Scocchi, M.; Gennaro, R.; Tossi, A. The human cathelicidin LL-37—A pore-forming antibacterial peptide and host-cell modulator. Biochim. Biophys. Acta 2016, 1858, 546–566. [Google Scholar] [CrossRef]
- Di Nardo, A.; Vitiello, A.; Gallo, R.L. Cutting edge: Mast cell antimicrobial activity is mediated by expression of cathelicidin antimicrobial peptide. J. Immunol. 2003, 170, 2274–2278. [Google Scholar] [CrossRef]
- Hansdottir, S.; Monick, M.M.; Hinde, S.L.; Lovan, N.; Look, D.C.; Hunninghake, G.W. Respiratory epithelial cells convert inactive vitamin D to its active form: Potential effects on host defense. J. Immunol. 2008, 181, 7090–7099. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Majewski, K.; Kozłowska, E.; Żelechowska, P.; Brzezińska-Błaszczyk, E. Serum concentrations of antimicrobial peptide cathelicidin LL-37 in patients with bacterial lung infections. Cent. Eur. J. Immunol. 2018, 43, 453–457. [Google Scholar] [CrossRef] [PubMed]
- Nell, M.J.; Sandra Tjabringa, G.; Vonk, M.J.; Hiemstra, P.S.; Grote, J.J. Bacterial products increase expression of the human cathelicidin hCAP-18/LL-37 in cultured human sinus epithelial cells. FEMS Immunol. Med. Microbiol. 2004, 42, 225–231. [Google Scholar] [CrossRef]
- Yang, D.; Chen, Q.; Schmidt, A.P.; Anderson, G.M.; Wang, J.M.; Wooters, J.; Oppenheim, J.J.; Chertov, O. LL-37, the neutrophil granule–and epithelial cell–derived cathelicidin, utilizes formyl peptide receptor–like 1 (FPRL1) as a receptor to chemoattract human peripheral blood neutrophils, monocytes, and T cells. J. Exp. Med. 2000, 192, 1069–1074. [Google Scholar] [CrossRef]
- Bandurska, K.; Berdowska, A.; Barczyńska-Felusiak, R.; Krupa, P. Unique features of human cathelicidin LL-37. Biofactors 2015, 41, 289–300. [Google Scholar] [CrossRef]
- Amatya, N.; Garg, A.V.; Gaffen, S.L. IL-17 signaling: The yin and the yang. Trends Immunol. 2017, 38, 310–322. [Google Scholar] [CrossRef] [Green Version]
- Lorè, N.I.; Bragonzi, A.; Cigana, C. The IL-17A/IL-17RA axis in pulmonary defence and immunopathology. Cytokine Growth Factor Rev. 2016, 30, 19–27. [Google Scholar] [CrossRef]
- Yadava, K.; Pattaroni, C.; Sichelstiel, A.K.; Trompette, A.; Gollwitzer, E.S.; Salami, O.; Von Garnier, C.; Nicod, L.P.; Marsland, B.J. Microbiota promotes chronic pulmonary inflammation by enhancing IL-17A and autoantibodies. Am. J. Respir. Crit. Care Med. 2016, 193, 975–987. [Google Scholar] [CrossRef] [Green Version]
- Brembilla, N.C.; Senra, L.; Boehncke, W.H. The IL-17 Family of Cytokines in Psoriasis: IL-17A and Beyond. Front. Immunol. 2018, 9, 1682. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gouda, M.M.; Bhandary, Y.P. Acute lung injury: IL-17A-mediated inflammatory pathway and its regulation by curcumin. Inflammation 2019, 42, 1160–1169. [Google Scholar] [CrossRef] [PubMed]
- Kao, C.Y.; Chen, Y.; Thai, P.; Wachi, S.; Huang, F.; Kim, C.; Harper, R.W.; Wu, R. IL-17 markedly up-regulates beta-defensin-2 expression in human airway epithelium via JAK and NF-kappaB signaling pathways. J. Immunol. 2004, 173, 3482–3491. [Google Scholar] [CrossRef]
- Kuensaen, C.; Chomdej, S.; Kongdang, P.; Sirikaew, N.; Jaitham, R.; Thonghoi, S.; Ongchai, S. LL-37 alone and in combination with IL17A enhances proinflammatory cytokine expression in parallel with hyaluronan metabolism in human synovial sarcoma cell line SW982-A step toward understanding the development of inflammatory arthritis. PLoS ONE 2019, 14, e0218736. [Google Scholar] [CrossRef]
- Junga, A.; Pilmane, M.; Ābola, Z.; Volrāts, O. The Distribution of Vascular Endothelial Growth Factor (VEGF), Human Beta-Defensin-2 (HBD-2), and Hepatocyte Growth Factor (HGF) in Intra-Abdominal Adhesions in Children under One Year of Age. Sci. World J. 2018, 2018, 5953095. [Google Scholar] [CrossRef]
- Rivas-Santiago, B.; Hernandez-Pando, R.; Carranza, C.; Juarez, E.; Contreras, J.L.; Aguilar-Leon, D.; Torres, M.; Sada, E. Expression of cathelicidin LL-37 during Mycobacterium tuberculosis infection in human alveolar macrophages, monocytes, neutrophils, and epithelial cells. Infect. Immun. 2008, 76, 935–941. [Google Scholar] [CrossRef] [Green Version]
- Mikacenic, C.; Hansen, E.E.; Radella, F.; Gharib, S.A.; Stapleton, R.D.; Wurfel, M.M. Interleukin-17A Is Associated With Alveolar Inflammation and Poor Outcomes in Acute Respiratory Distress Syndrome. Crit. Care Med. 2016, 44, 496–502. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Duits, L.A.; Ravensbergen, B.; Rademaker, M.; Hiemstra, P.S.; Nibbering, P.H. Expression of β-defensin 1 and 2 mRNA by human monocytes, macrophages and dendritic cells. Immunology 2002, 106, 517–525. [Google Scholar] [CrossRef] [PubMed]
- Nagao, M.; Hamilton, J.L.; Kc, R.; Berendsen, A.D.; Duan, X.; Cheong, C.W.; Li, X.; Im, H.J.; Olsen, B.R. Vascular endothelial growth factor in cartilage development and osteoarthritis. Sci. Rep. 2017, 7, 13027. [Google Scholar] [CrossRef]
- Davidson, D.; Blanc, A.; Filion, D.; Wang, H.; Plut, P.; Pfeffer, G.; Buschmann, M.D.; Henderson, J.E. Fibroblast growth factor (FGF) 18 signals through FGF receptor 3 to promote chondrogenesis. J. Biol. Chem. 2005, 280, 20509–20515. [Google Scholar] [CrossRef] [Green Version]
- Mori, Y.; Saito, T.; Chang, S.H.; Kobayashi, H.; Ladel, C.H.; Guehring, H.; Chung, U.I.; Kawaguchi, H. Identification of fibroblast growth factor-18 as a molecule to protect adult articular cartilage by gene expression profiling. J. Biol. Chem. 2014, 289, 10192–10200. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Varoga, D.; Paulsen, F.P.; Kohrs, S.; Grohmann, S.; Lippross, S.; Mentlein, R.; Tillmann, B.N.; Goldring, M.B.; Besch, L.; Pufe, T. Expression and regulation of human β-defensin-2 in osteoarthritic cartilage. J. Pathol. 2006, 209, 166–173. [Google Scholar] [CrossRef]
- Varoga, D.; Pufe, T.; Harder, J.; Meyer-Hoffert, U.; Mentlein, R.; Schröder, J.M.; Petersen, W.J.; Tillmann, B.N.; Proksch, E.; Goldring, M.B.; et al. Production of endogenous antibiotics in articular cartilage. Arthritis Rheum. 2004, 50, 3526–3534. [Google Scholar] [CrossRef]
- Douwes, J.; Thorne, P.; Pearce, N.; Heederik, D. Bioaerosol health effects and exposure assessment: Progress and prospects. Ann. Occup. Hyg. 2003, 47, 187–200. [Google Scholar] [CrossRef] [Green Version]
- Hernandez, M.L.; Mills, K.; Almond, M.; Todoric, K.; Aleman, M.M.; Zhang, H.; Zhou, H.; Peden, D.B. IL-1 receptor antagonist reduces endotoxin-induced airway inflammation in healthy volunteers. J. Allergy Clin. Immunol. 2015, 135, 379–385. [Google Scholar] [CrossRef] [Green Version]
- Thorne, P.S. Inhalation toxicology models of endotoxin-and bioaerosol-induced inflammation. Toxicology 2000, 152, 13–23. [Google Scholar] [CrossRef]
- Kraus, D.; Deschner, J.; Jäger, A.; Wenghoefer, M.; Bayer, S.; Jepsen, S.; Allam, J.P.; Novak, N.; Meyer, R.; Winter, J. Human β-defensins differently affect proliferation, differentiation, and mineralization of osteoblast-like MG63 cells. J. Cell. Physiol. 2011, 227, 994–1003. [Google Scholar] [CrossRef] [PubMed]
- Sakkas, L.I.; Zafiriou, E.; Bogdanos, D.P. Mini review: New treatments in psoriatic arthritis. Focus on the IL-23/17 axis. Front. Pharmacol. 2019, 10, 872. [Google Scholar] [CrossRef] [Green Version]
- Mookherjee, N.; Brown, K.L.; Bowdish, D.M.; Doria, S.; Falsafi, R.; Hokamp, K.; Roche, F.M.; Mu, R.; Doho, G.H.; Pistolic, J.; et al. Modulation of the TLR-mediated inflammatory response by the endogenous human host defense peptide LL-37. J. Immunol. 2006, 176, 2455–2464. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, J.; Wang, D.; Wang, L.; Wang, S.; Roden, A.C.; Zhao, H.; Li, X.; Prakash, Y.S.; Matteson, E.L.; Tschumperlin, D.J.; et al. Profibrotic effect of IL-17A and elevated IL-17RA in idiopathic pulmonary fibrosis and rheumatoid arthritis-associated lung disease support a direct role for IL-17A/IL-17RA in human fibrotic interstitial lung disease. Am. J. Physiol. Lung Cell. Mol. Physiol. 2019, 316, L487–L497. [Google Scholar] [CrossRef] [PubMed]
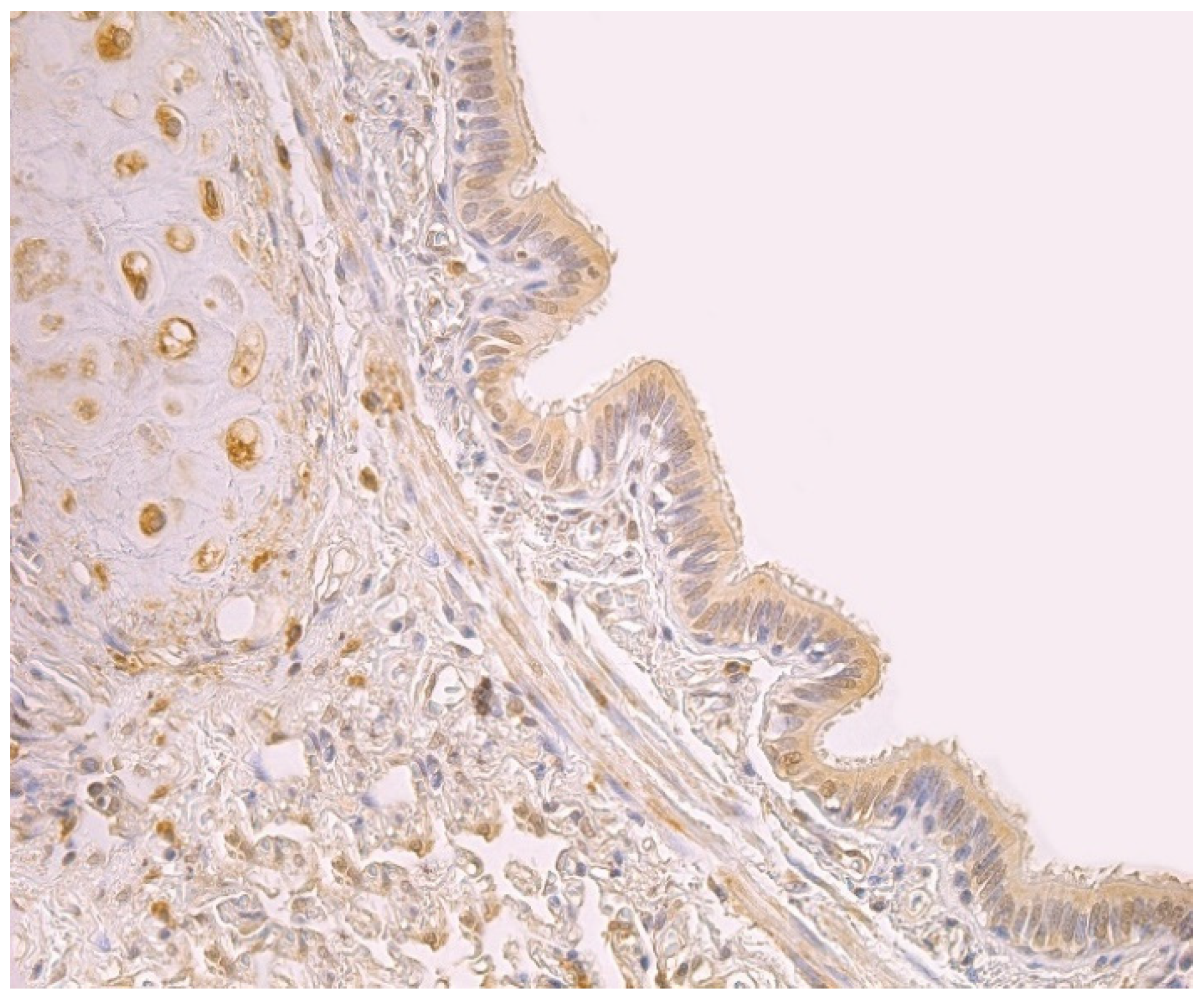
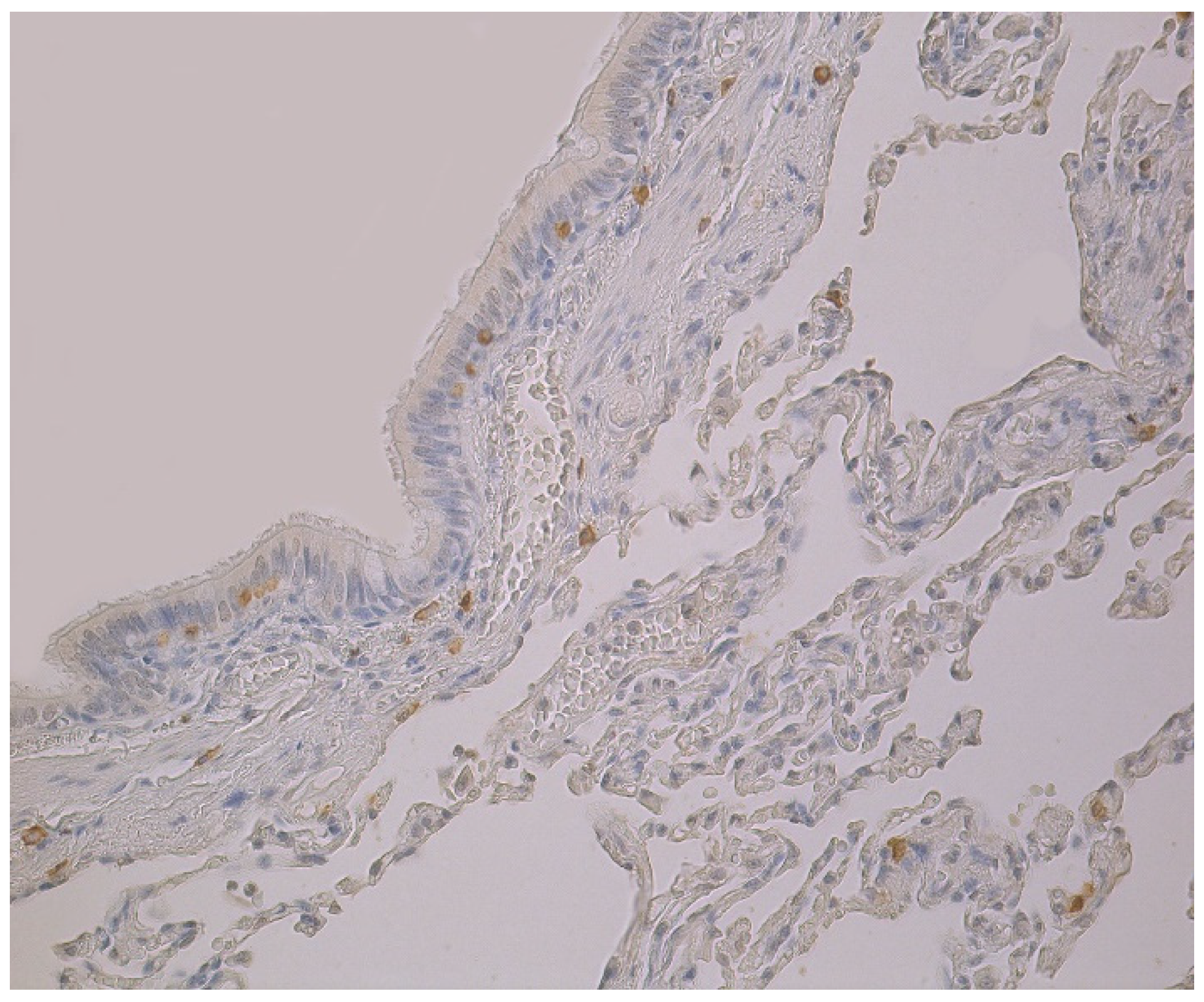
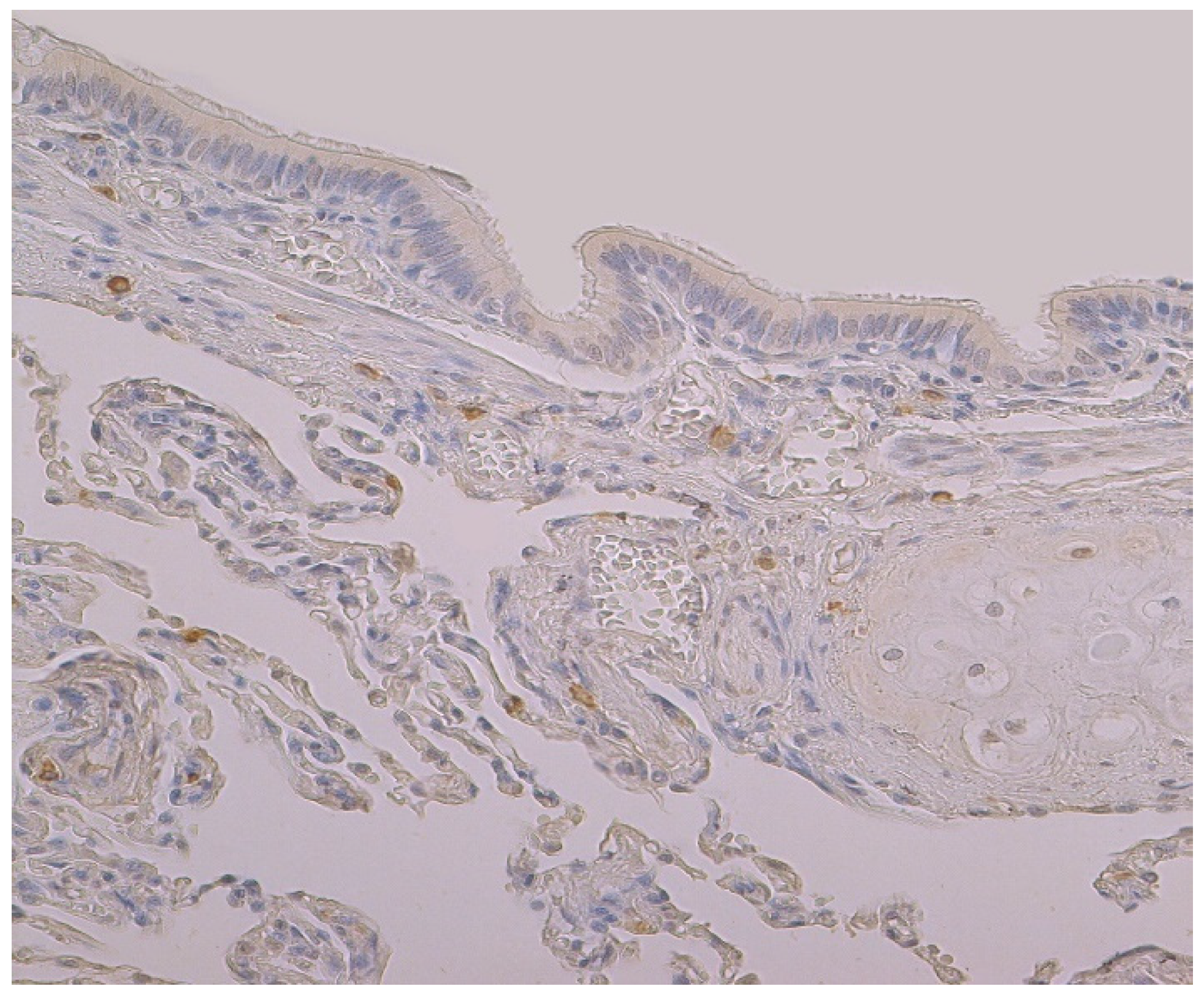
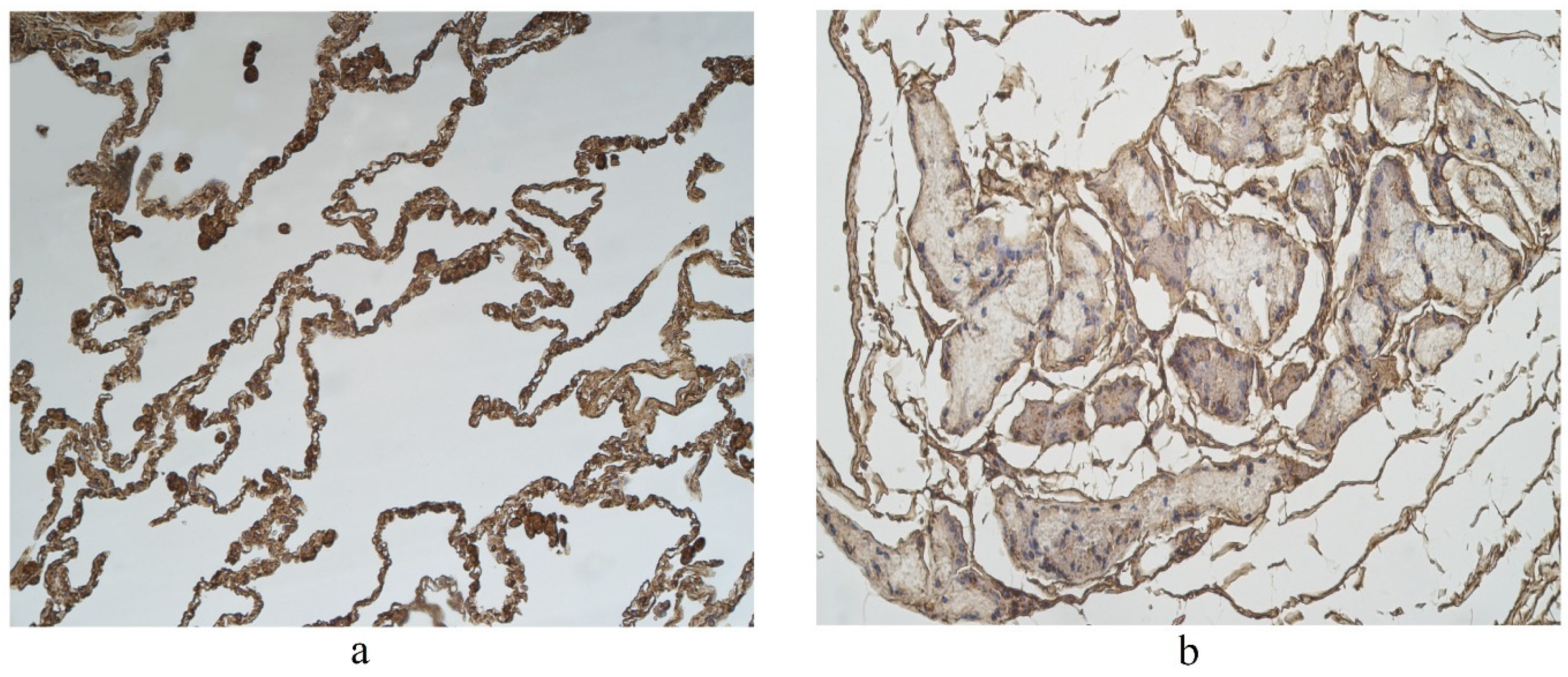


| Nr | Age | HBD-2 | HBD-3 | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| C | CT | G | BE | AE | AM | C | CT | G | BE | AE | AM | ||
| 1 | 18 | +++ | + | + | + | 0 | 0 | ++ | + | +/++ | +/++ | 0 | 0 |
| 2 | 19 | +++ | +/++ | +/++ | ++ | 0 | 0 | +/++ | + | ++ | + | 0 | 0 |
| 3 | 19 | ++ | 0 | 0 | + | + | + | ++ | + | 0/+ | 0/+ | +/++ | + |
| 4 | 25 | +++ | +/++ | ++ | 0 | +/++ | +/++ | +++ | +/++ | + | ++ | +/++ | + |
| 5 | 27 | ++ | ++ | + | 0/+ | +/++ | ++ | ++ | 0/+ | + | + | ++ | ++ |
| 6 | 29 | ++/+++ | 0/+ | 0/+ | 0 | +++ | ++ | ++ | 0/+ | 0/+ | 0 | ++/+++ | ++/+++ |
| 7 | 33 | ++ | ++ | 0/+ | 0 | ++ | + | ++ | 0/+ | 0 | 0 | ++ | ++ |
| 8 | 35 | +/++ | +/++ | 0/+ | 0 | + | + | + | +/++ | 0/+ | 0/+ | +/++ | 0/+ |
| 9 | 38 | ++/+++ | 0/+ | 0 | 0/+ | +/++ | ++ | ++ | 0 | + | +/++ | +/++ | + |
| 10 | 45 | ++ | 0 | 0/+ | + | ++ | ++ | +/++ | + | + | + | ++ | +/++ |
| 11 | 55 | + | + | 0 | 0 | +/++ | ++/+++ | + | 0/+ | 0/+ | + | ++ | +/++ |
| 12 | 56 | ++ | +/++ | 0/+ | + | +/++ | ++ | +/++ | + | 0/+ | + | +/++ | 0/+ |
| 13 | 59 | +++ | + | 0 | 0/+ | +/++ | 0/+ | ++ | 0 | 0 | 0/+ | ++ | + |
| 14 | 74 | ++ | 0 | + | 0 | ++ | ++ | +/++ | 0/+ | 0/+ | 0 | ++ | +/++ |
| 15 | 86 | +/++ | 0/+ | 0/+ | 0 | +/++ | ++ | +/++ | 0 | 0/+ | 0 | +/++ | + |
| Common | ++ | + | 0/+ | + | +/++ | +/++ | ++ | + | + | + | ++ | +/++ | |
| Nr | Age | HBD-4 | |||||
|---|---|---|---|---|---|---|---|
| C | CT | G | BE | AE | AM | ||
| 1 | 18 | +/++ | 0 | 0/+ | + | 0 | 0 |
| 2 | 19 | ++ | + | ++ | ++ | 0 | 0 |
| 3 | 19 | +/++ | 0/+ | 0/+ | 0/+ | + | + |
| 4 | 25 | ++/+++ | ++ | 0/+ | 0/+ | +++ | ++ |
| 5 | 27 | ++ | 0/+ | 0 | 0 | +/++ | 0/+ |
| 6 | 29 | ++ | + | 0/+ | 0 | ++ | +++ |
| 7 | 33 | +/++ | 0/+ | 0 | 0 | ++ | ++ |
| 8 | 35 | +/++ | 0 | 0 | 0 | +/++ | + |
| 9 | 38 | ++ | + | 0/+ | 0/+ | ++ | ++ |
| 10 | 45 | 0/+ | + | 0 | 0/+ | + | +++ |
| 11 | 55 | +/++ | 0/+ | 0 | 0 | +/++ | ++ |
| 12 | 56 | ++ | 0/+ | 0/+ | 0/+ | +/++ | ++ |
| 13 | 59 | ++ | 0 | 0 | 0 | 0/+ | 0/+ |
| 14 | 74 | ++ | 0/+ | 0/+ | 0 | +/++ | + |
| 15 | 86 | + | 0/+ | 0/+ | 0 | + | 0/+ |
| Common | ++ | 0/+ | 0/+ | 0/+ | +/++ | +/++ | |
| Nr | Age | LL-37 | IL-17A | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| C | CT | G | BE | AE | AM | C | CT | G | BE | AE | AM | ||
| 1 | 18 | ++ | + | + | +/++ | 0 | 0 | +++ | 0/+ | +/++ | +/++ | 0 | 0 |
| 2 | 19 | ++/+++ | + | + | ++ | 0 | 0 | +++ | + | ++ | ++/+++ | 0 | 0 |
| 3 | 19 | ++ | 0/+ | 0 | + | ++v | ++ | ++/+++ | + | + | + | +/++ | ++ |
| 4 | 25 | ++ | 0/+ | 0/+ | 0 | ++ | ++/+++ | ++ | 0 | + | 0 | ++ | ++ |
| 5 | 27 | ++ | + | + | +/++ | ++ | +/++ | +++ | + | + | ++ | ++ | ++ |
| 6 | 29 | ++ | 0/+ | + | + | ++ | ++ | +++ | 0 | + | + | ++v | ++v |
| 7 | 33 | ++ | 0/+ | + | 0 | ++/+++ | ++ | ++/+++ | 0/+ | + | 0 | ++/+++ | ++ |
| 8 | 35 | +/++ | + | 0/+ | 0 | ++ | ++ | ++v | 0/+ | 0/+ | 0 | ++v | +++ |
| 9 | 38 | +/++ | 0/+ | 0/+ | +/++ | +/++ | +/++ | ++ | 0 | + | ++ | ++v | ++ |
| 10 | 45 | ++ | 0 | + | ++ | ++v | ++ | ++/+++ | 0/+ | 0/+ | ++ | ++v | ++v |
| 11 | 55 | ++v | + | 0/+ | 0 | ++ | ++ | ++ | + | + | ++ | ++/+++ | ++ |
| 12 | 56 | + | 0/+ | 0/+ | +/++ | ++ | +++ | ++ | 0/+ | 0/+ | ++ | ++ | +++ |
| 13 | 59 | + | 0/+ | 0/+ | 0 | +/++ | ++ | ++/+++ | 0/+ | +/++ | 0 | ++v | ++ |
| 14 | 74 | ++ | + | + | +/++ | ++v | ++v | ++ | + | 0/+ | ++ | ++v | 0 |
| 15 | 86 | ++ | 0/+ | 0/+ | +/++ | ++ | + | +++ | 0 | 0/+ | 0 | ++ | ++v |
| Common | ++ | + | + | +/++ | ++ | ++ | ++/+++ | 0/+ | + | ++ | ++ | ++ | |
| Strength of Correlation | Marker 1 | Marker 2 | Rho (ρ) | p-Value |
|---|---|---|---|---|
| Very strong positive correlation | HBD-3 in alveolar epithelium | HBD-3 in alveolar macrophages | 0.911 | <0.001 |
| HBD-2 in alveolar epithelium | HBD-3 in alveolar epithelium | 0.845 | <0.001 | |
| HBD-2 in alveolar epithelium | HBD-3 in alveolar macrophages | 0.838 | <0.001 | |
| Strong positive correlation | HBD-2 in bronchial epithelium | HBD-4 in bronchial epithelium | 0.781 | 0.001 |
| HBD-3 in glands | HBD-3 in bronchial epithelium | 0.733 | 0.002 | |
| HBD3 in glands | HBD-4 in bronchial epithelium | 0.729 | 0.002 | |
| HBD-2 in alveolar epithelium | HBD-4 in alveolar macrophages | 0.707 | 0.003 | |
| LL-37 in bronchial epithelium | IL-17A in bronchial epithelium | 0.706 | 0.003 | |
| IL-17A in glands | HBD-2 in cartilage | 0.705 | 0.003 | |
| HBD-4 in alveolar macrophages | HBD-4 in alveolar epithelium | 0.692 | 0.004 | |
| HBD-3 in bronchial epithelium | HBD-4 in bronchial epithelium | 0.685 | 0.005 | |
| LL-37 in alveolar epithelium | IL-17A in alveolar epithelium | 0.681 | 0.005 | |
| HBD-2 in cartilage | HBD-3 in cartilage | 0.674 | 0.006 | |
| IL-17A in alveolar epithelium | HBD-3 in alveolar epithelium | 0.670 | 0.006 | |
| IL-17A in alveolar epithelium | HBD-2 in alveolar epithelium | 0.659 | 0.008 | |
| LL-37 in alveolar macrophages | HBD-4 in alveolar macrophages | 0.658 | 0.008 | |
| IL-17A in alveolar epithelium | HBD-3 in alveolar macrophages | 0.645 | 0.009 | |
| LL-37 in bronchial epithelium | HBD-3 in glands | 0.640 | 0.010 | |
| LL-37 in alveolar epithelium | HBD-3 in alveolar macrophages | 0.639 | 0.010 | |
| LL-37 in bronchial epithelium | HBD-2 in bronchial epithelium | 0.630 | 0.012 | |
| IL-17A in alveolar epithelium | HBD-4 in alveolar epithelium | 0.628 | 0.012 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lohova, E.; Vitenberga-Verza, Z.; Kazoka, D.; Pilmane, M. Local Defence System in Healthy Lungs. Clin. Pract. 2021, 11, 728-746. https://doi.org/10.3390/clinpract11040088
Lohova E, Vitenberga-Verza Z, Kazoka D, Pilmane M. Local Defence System in Healthy Lungs. Clinics and Practice. 2021; 11(4):728-746. https://doi.org/10.3390/clinpract11040088
Chicago/Turabian StyleLohova, Elizabeta, Zane Vitenberga-Verza, Dzintra Kazoka, and Mara Pilmane. 2021. "Local Defence System in Healthy Lungs" Clinics and Practice 11, no. 4: 728-746. https://doi.org/10.3390/clinpract11040088
APA StyleLohova, E., Vitenberga-Verza, Z., Kazoka, D., & Pilmane, M. (2021). Local Defence System in Healthy Lungs. Clinics and Practice, 11(4), 728-746. https://doi.org/10.3390/clinpract11040088






