Uncommon Presentation of Granulomatosis with Polyangiitis Mimicking Metastatic Lung Cancer
Abstract
1. Introduction
2. Materials and Methods
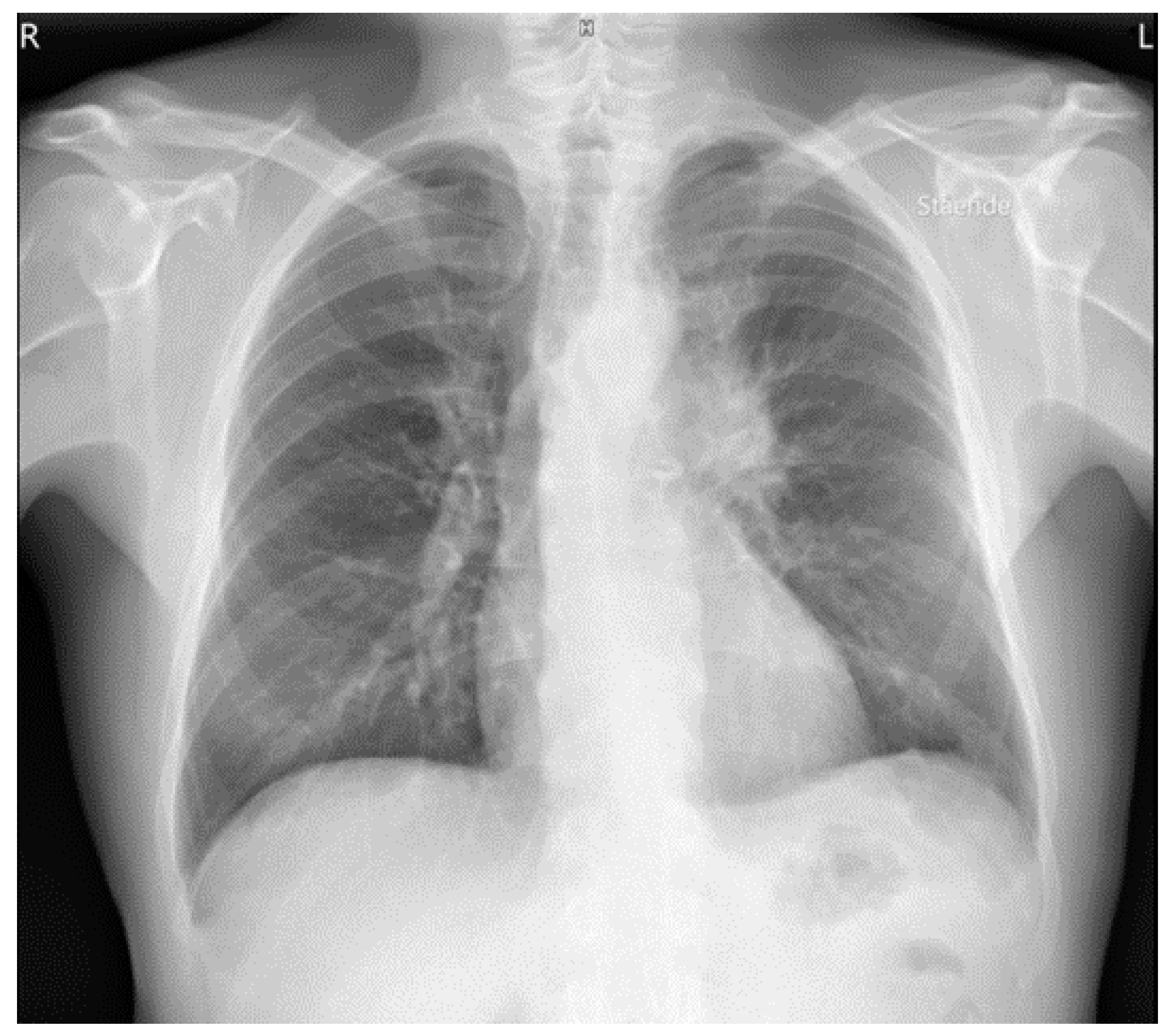
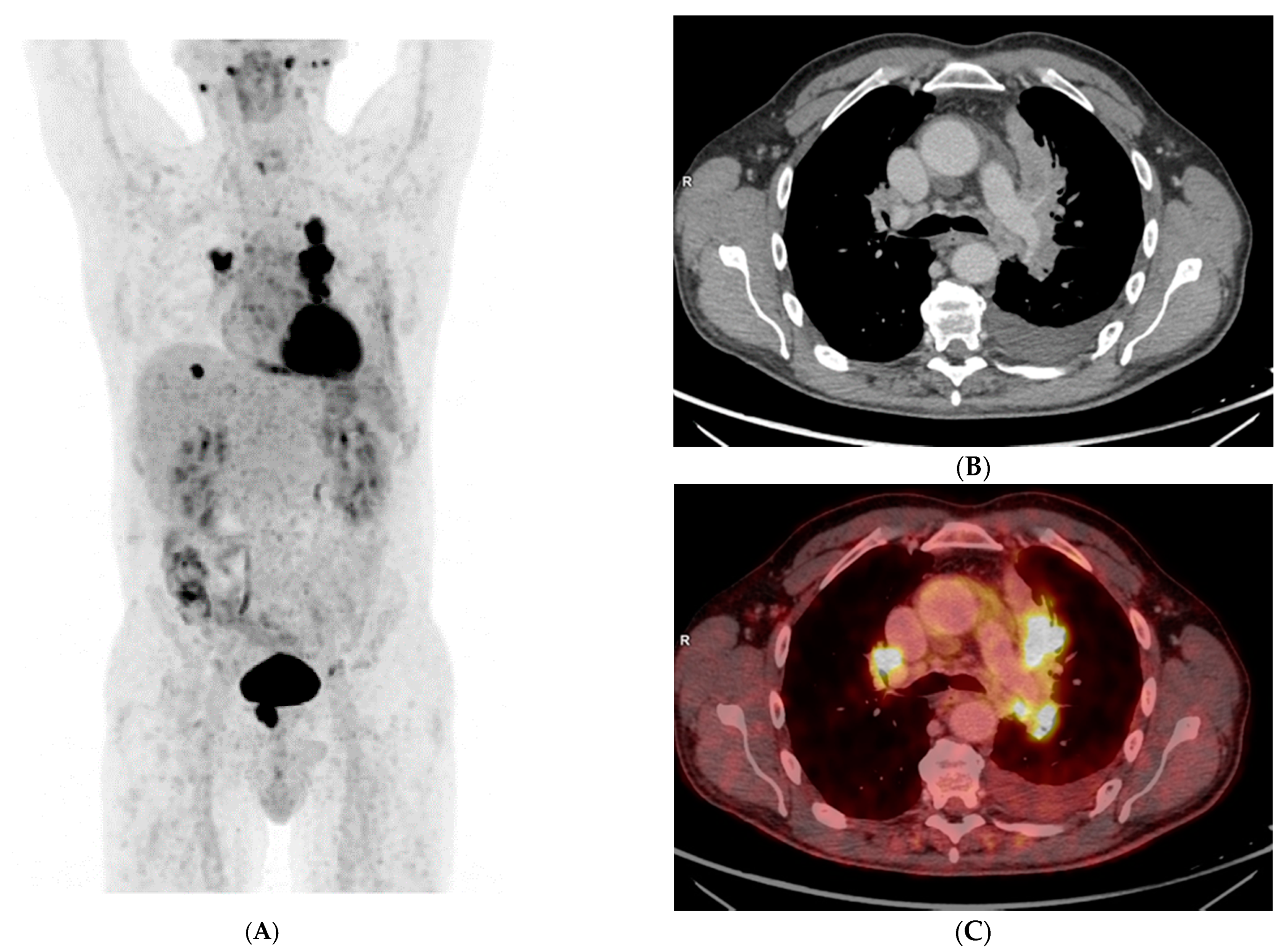
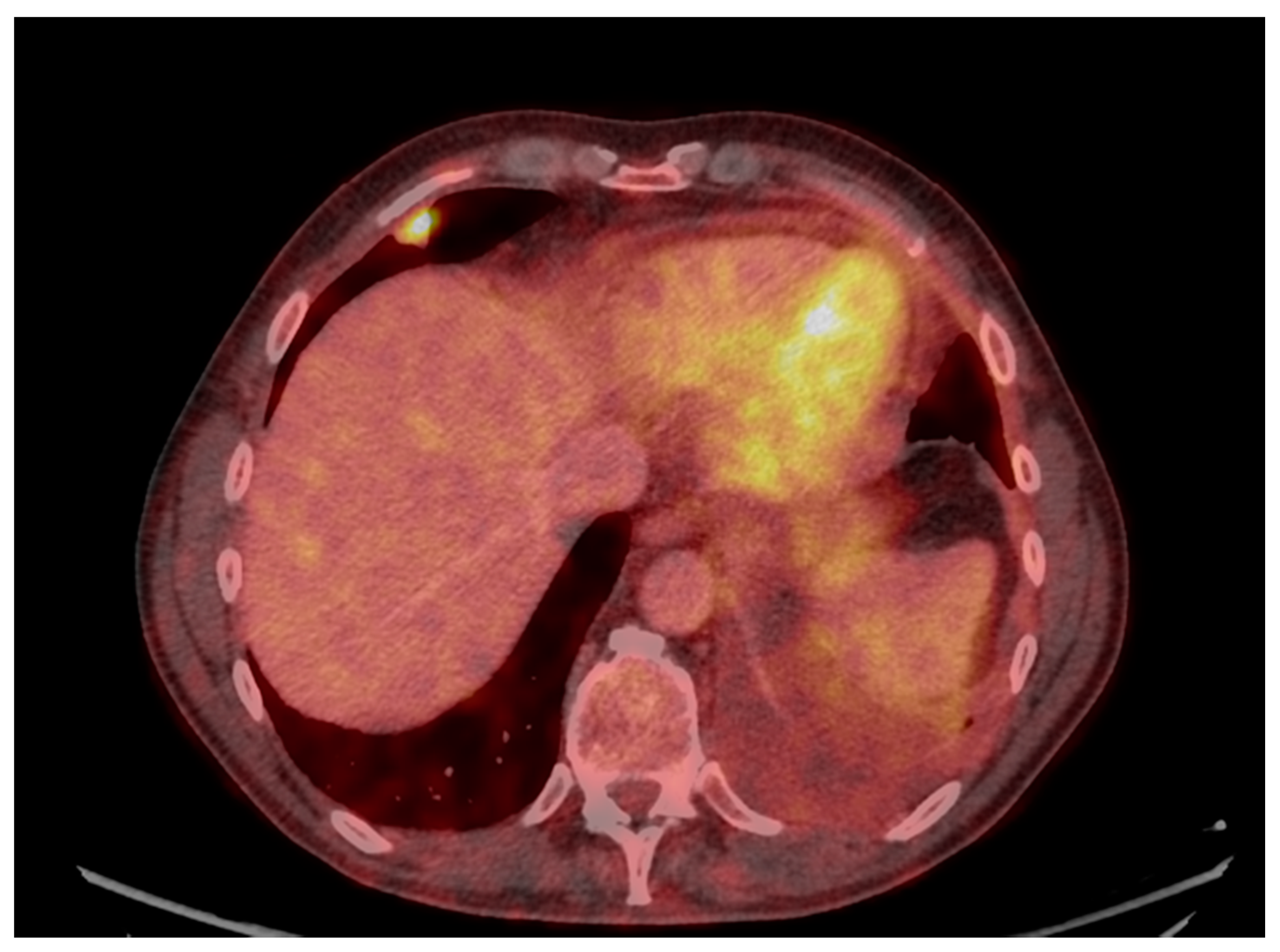
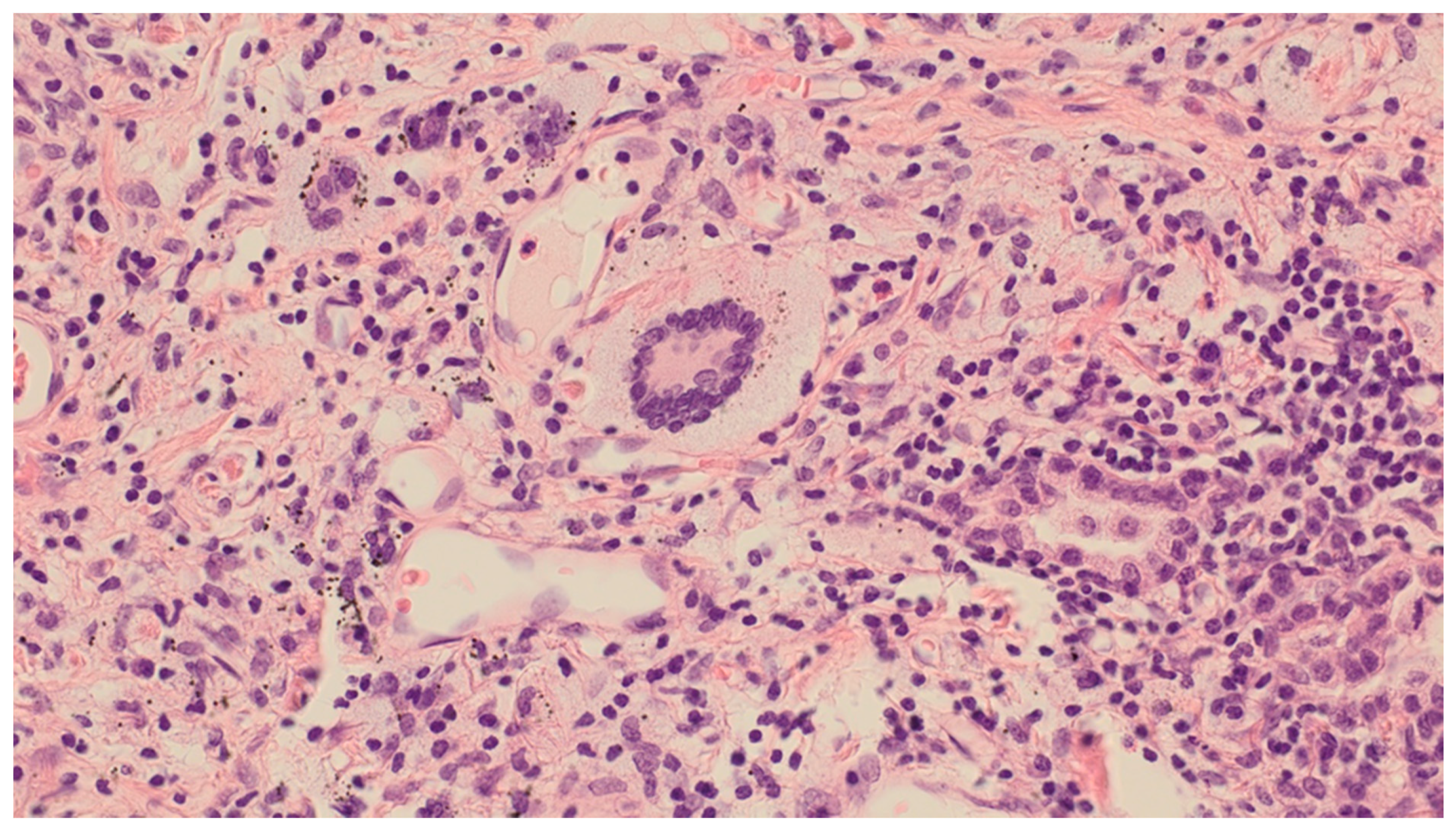
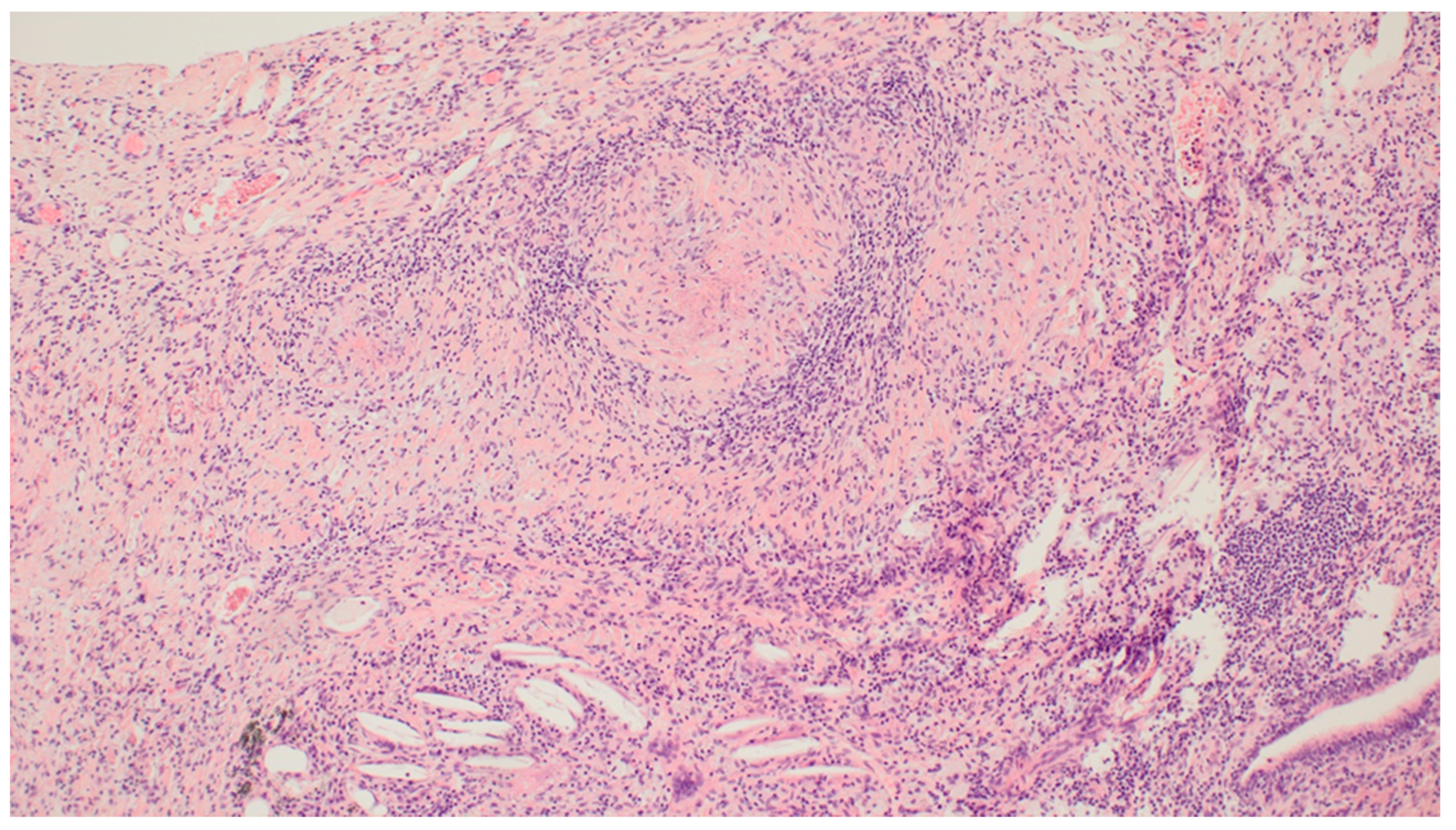
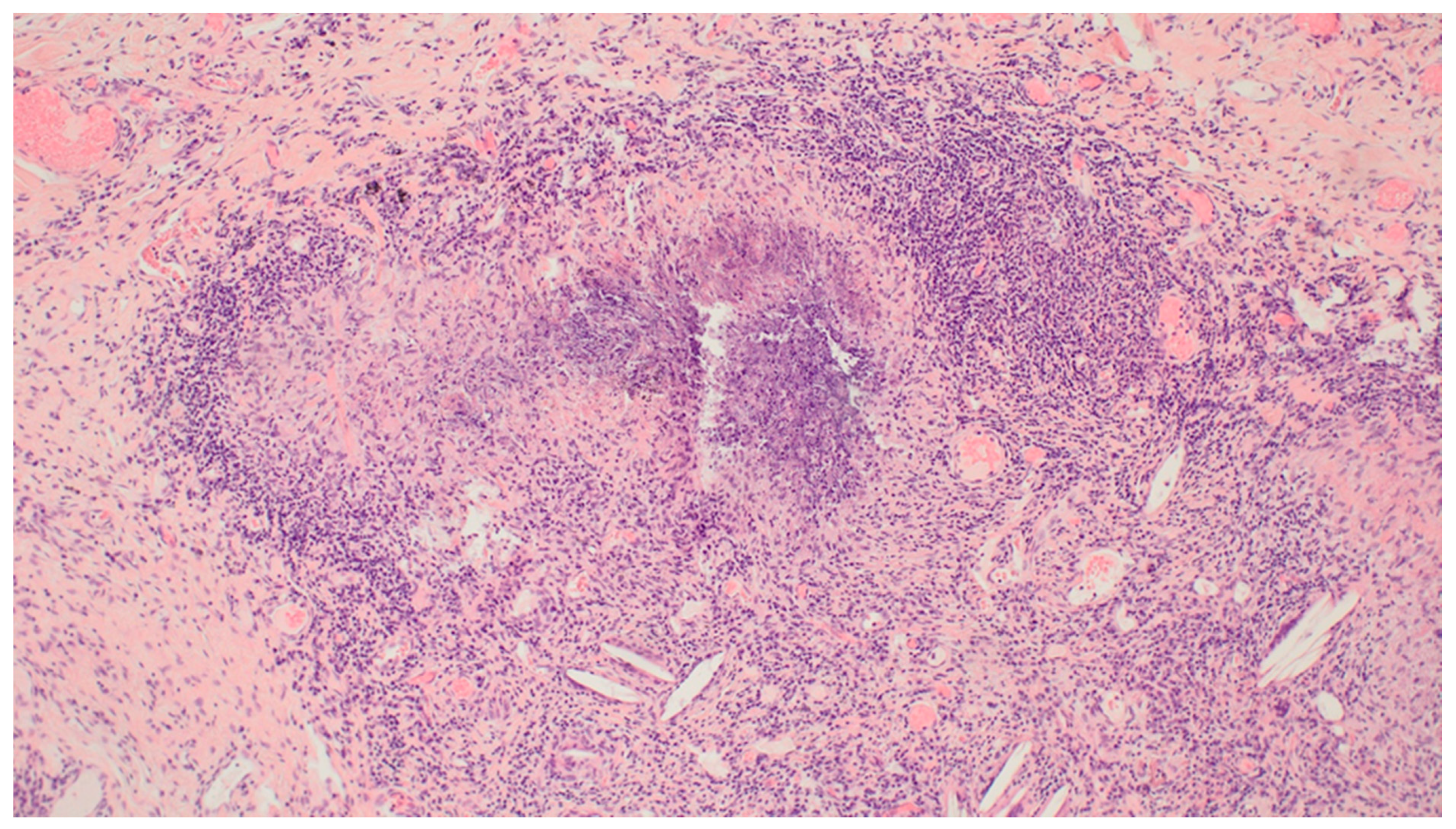
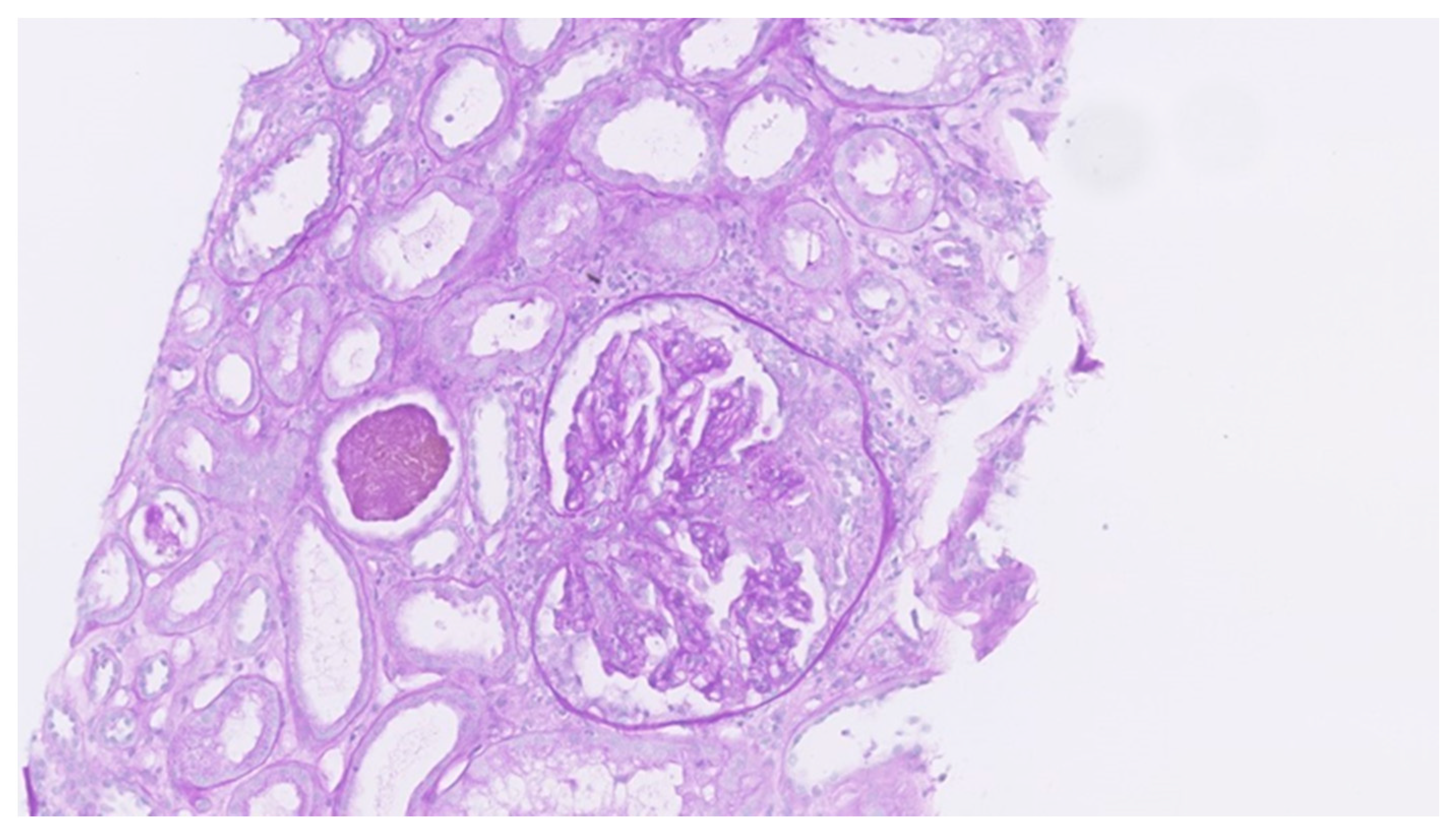
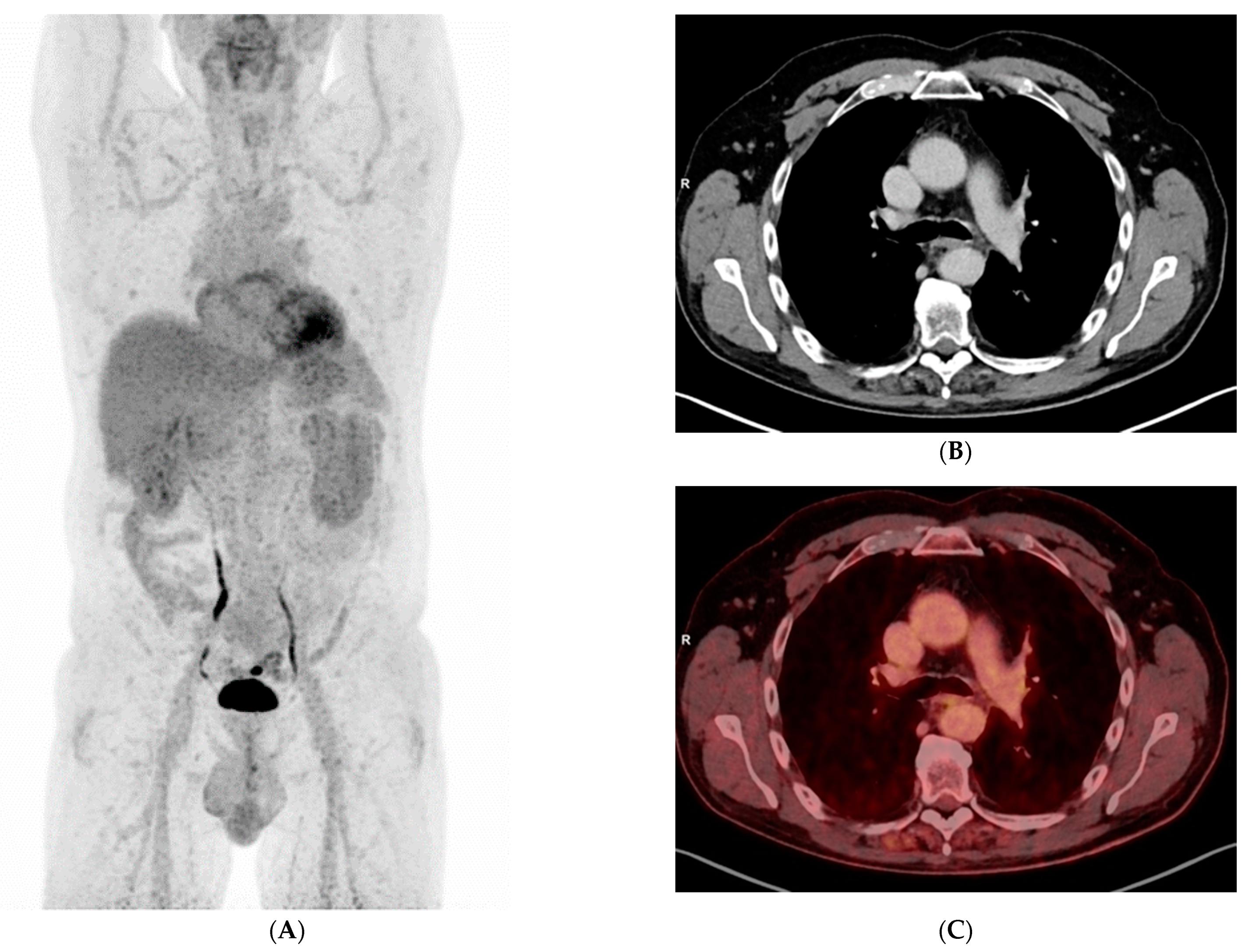
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Jennette, J.C.; Falk, R.J.; Bacon, P.A.; Basu, N.; Cid, M.C.; Ferrario, F.; Flores-Suarez, L.F.; Gross, W.L.; Guillevin, L.; Hagen, E.C.; et al. 2012 Revised International Chapel Hill Consensus Conference Nomenclature of Vasculitides. Arthritis Rheum. 2013, 65, 1–11. [Google Scholar] [CrossRef]
- Watts, A.R. Geoepidemiology of systemic vasculitis: Comparison of the incidence in two regions of Europe. Ann. Rheum. Dis. 2001, 60, 170–172. [Google Scholar] [CrossRef] [PubMed]
- Flossmann, O.; Berden, A.; De Groot, K.; Hagen, C.; Harper, L.; Heijl, C.; Höglund, P.; Jayne, D.; Luqmani, R.; Mahr, A.; et al. Long-term patient survival in ANCA-associated vasculitis. Ann. Rheum. Dis. 2010, 70, 488–494. [Google Scholar] [CrossRef] [PubMed]
- Booth, A.D.; Almond, M.K.; Burns, A.; Ellis, P.; Gaskin, G.; Neild, G.H.; Plaisance, M.; Pusey, C.D.; Jayne, D.R. Outcome of ANCA-associated renal vasculitis: A 5-year retrospective study. Am. J. Kidney Dis. 2003, 41, 776–784. [Google Scholar] [CrossRef]
- Koldingsnes, W. Predictors of survival and organ damage in Wegener’s granulomatosis. Rheumatology 2002, 41, 572–581. [Google Scholar] [CrossRef] [PubMed]
- Houben, E.; Groenland, S.L.; Van Der Heijden, J.W.; Voskuyl, A.E.; Doodeman, H.J.; Penne, E.L. Relation between duration of the prodromal phase and renal damage in ANCA-associated vasculitis. BMC Nephrol. 2017, 18, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Nelveg-Kristensen, K.E.; Szpirt, W.; Carlson, N.; McClure, M.; Jayne, D.; Dieperink, H.; Gregersen, J.W.; Krarup, E.; Ivarsen, P.; Torp-Pedersen, C.; et al. Increasing incidence and improved survival in ANCA-associated vasculitis—A Danish nationwide study. Nephrol. Dial. Transplant. 2020. [Google Scholar] [CrossRef]
- Berden, A.E.; Ferrario, F.; Hagen, E.C.; Jayne, D.R.; Jennette, J.C.; Joh, K.; Neumann, I.; Noël, L.-H.; Pusey, C.D.; Waldherr, R.; et al. Histopathologic Classification of ANCA-Associated Glomerulonephritis. J. Am. Soc. Nephrol. 2010, 21, 1628–1636. [Google Scholar] [CrossRef]
- Jennette, J.C.; Falk, R.J.; Hu, P.; Xiao, H. Pathogenesis of antineutrophil cytoplasmic autoantibody-associated small-vessel vasculitis. Annu. Rev. Pathol. 2013, 8, 139–160. [Google Scholar] [CrossRef]
- Lyons, P.A.; Rayner, T.F.; Trivedi, S.; Holle, J.U.; Watts, R.A.; Jayne, D.R.; Baslund, B.; Brenchley, P.; Bruchfeld, A.; Chaudhry, A.N.; et al. Genetically distinct subsets within ANCA-associated vasculitis. N. Engl. J. Med. 2012, 367, 214–223. [Google Scholar] [CrossRef]
- Lionaki, S.; Blyth, E.R.; Hogan, S.L.; Hu, Y.; Senior, J.B.A.; Jennette, C.E.; Nachman, P.H.; Jennette, J.C.; Falk, R.J. Classification of antineutrophil cytoplasmic autoantibody vasculitides: The role of antineutrophil cytoplasmic autoantibody specificity for myeloperoxidase or proteinase 3 in disease recognition and prognosis. Arthritis Rheum. 2012, 64, 3452–3462. [Google Scholar] [CrossRef]
- Suppiah, R.; Judge, A.; Batra, R.; Flossmann, O.; Harper, L.; Höglund, P.; Javaid, M.K.; Jayne, D.; Mukhtyar, C.; Westman, K.; et al. A model to predict cardiovascular events in patients with newly diagnosed Wegener’s granulomatosis and microscopic polyangiitis. Arthritis Rheum. 2011, 63, 588–596. [Google Scholar] [CrossRef]
- Mukhtyar, C.; Flossmann, O.; Hellmich, B.; Bacon, P.; Cid, M.; Cohen-Tervaert, J.W.; Gross, W.L.; Guillevin, L.; Jayne, D.; Mahr, A.; et al. Outcomes from studies of antineutrophil cytoplasm antibody associated vasculitis: A systematic review by the European League Against Rheumatism systemic vasculitis task force. Ann. Rheum. Dis. 2008, 67, 1004–1010. [Google Scholar] [CrossRef] [PubMed]
- Mahmoud, S.; Ghosh, S.; Farver, C.; Lempel, J.; Azok, J.; Renapurkar, R.D. Pulmonary Vasculitis. Radiol. Clin. N. Am. 2016, 54, 1097–1118. [Google Scholar] [CrossRef] [PubMed]
- Sacoto, G.; Boukhlal, S.; Specks, U.; Flores-Suárez, L.F.; Cornec, D. Lung involvement in ANCA-associated vasculitis. Presse Médicale 2020, 49, 104039. [Google Scholar] [CrossRef]
- Giraudo, C.; Nannini, N.; Balestro, E.; Meneghin, A.; Lunardi, F.; Polverosi, R.; Calabrese, F. Necrotizing Sarcoid Granulo-matosis with uncommon Manifestation. Clinicopathological Features and Review of Literature. Res. Care 2014, 59, 132–136. [Google Scholar] [CrossRef]
- Nelson, D.R.; Johnson, G.B.; Cartin-Ceba, R.; Specks, U. Characterization of F-18 Fluorodeoxyglucose PET/CT in Granulo-matosis with Polyangiitis. Sarcoidosis Vasc. Diffus. Lung Dis. 2016, 18, 342–352. [Google Scholar]
- Travis, W.D.; Hoffman, G.S.; Leavitt, R.Y.; Pass, H.I.; Fauci, A.S. Surgical Pathology of the Lung in Wegenerʼs Granulomatosis. Am. J. Surg. Pathol. 1991, 15, 315–333. [Google Scholar] [CrossRef]
- Toriyama, M.; Tagaya, E.; Yamamoto, T.; Kondo, M.; Nagashima, Y.; Tamaoki, J. Lung cancer development in the patient with granulomatosis with polyangiitis during long term treatment with cyclophosphamide: First documented case. Respirol. Case Rep. 2017, 6, e00284. [Google Scholar] [CrossRef] [PubMed]
- Geetha, D.; Jefferson, J.A. ANCA-Associated Vasculitis: Core Curriculum 2020. Am. J. Kidney Dis. 2020, 75, 124–137. [Google Scholar] [CrossRef] [PubMed]
- Gulati, S.; Patel, N.P.; Swierczynski, S.L. Vasculitides associated with haematological malignancies: A case-based review. BMJ Case Rep. 2012, 2012, 2012007123. [Google Scholar] [CrossRef]
- Folci, M.; Ramponi, G.; Shiffer, D.; Zumbo, A.; Agosti, M.; Brunetta, E. ANCA-Associated Vasculitides and Hematologic Malignancies: Lessons from the Past and Future Perspectives. J. Immunol. Res. 2019, 2019, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Campainha, S.; Gonçalves, M.; Tavares, V.; Castelões, P.; Marinho, A.; Neves, S. Granulomatose com poliangeíte inicialmente diagnosticada como cancro do pulmão. Rev. Port. Pneumol. 2013, 19, 45–48. [Google Scholar] [CrossRef] [PubMed]
- Chemouny, J.M.; Pagnoux, C.; Caudwell, V.; Karras, A.; Borie, R.; Guillevin, L.; Vrtovsnik, F.; Daugas, E. ANCA-associated diseases and lung carcinomas: A five-case series. Clin. Nephrol. 2014, 81, 132–137. [Google Scholar] [CrossRef]
- Sriskandarajah, S.; Bostad, L.; Myklebust, T.A.; Møller, B.; Skrede, S.; Bjørneklett, R. Cancer in ANCA-Associated Glomerulonephritis: A Registry-Based Cohort Study. Int. J. Nephrol. 2017, 2017, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Shang, W.; Ning, Y.; Xu, X.; Li, M.; Guo, S.; Han, M.; Zeng, R.; Ge, S.; Xu, G. Incidence of Cancer in ANCA-Associated Vasculitis: A Meta-Analysis of Observational Studies. PLoS ONE 2015, 10, e0126016. [Google Scholar] [CrossRef]
- Heijl, C.; Harper, L.; Flossmann, O.; Stucker, I.; Scott, I.D.G.; Watts, R.A.; Hoglund, P.; Westman, K.; Mahr, A.; for the European Vasculitis Study Group (EUVAS). Incidence of malignancy in patients treated for antineutrophil cytoplasm antibody-associated vasculitis: Follow-up data from European Vasculitis Study Group clinical trials. Ann. Rheum. Dis. 2011, 70, 1415–1421. [Google Scholar] [CrossRef]
- Heijl, C.; Westman, K.; Höglund, P.; Mohammad, A.J. Malignancies in Patients with Antineutrophil Cytoplasmic Antibody-associated Vasculitis: A Population-based Cohort Study. J. Rheumatol. 2019, 47, 1229–1237. [Google Scholar] [CrossRef]
- Masiak, A.; Fijałkowska, J.; Nowakowski, S.; Smoleńska, Ż.; Zdrojewski, Z. New lung mass in a patient with granulomatosis with polyangiitis. Rheumatol. Int. 2021, 41, 493–499. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Urbanska, E.M.; Elversang, J.; Colville-Ebeling, B.; Löfgren, J.O.; Nelveg-Kristensen, K.E.; Szpirt, W.M. Uncommon Presentation of Granulomatosis with Polyangiitis Mimicking Metastatic Lung Cancer. Clin. Pract. 2021, 11, 293-302. https://doi.org/10.3390/clinpract11020042
Urbanska EM, Elversang J, Colville-Ebeling B, Löfgren JO, Nelveg-Kristensen KE, Szpirt WM. Uncommon Presentation of Granulomatosis with Polyangiitis Mimicking Metastatic Lung Cancer. Clinics and Practice. 2021; 11(2):293-302. https://doi.org/10.3390/clinpract11020042
Chicago/Turabian StyleUrbanska, Edyta Maria, Johanna Elversang, Bonnie Colville-Ebeling, Johan Olof Löfgren, Karl Emil Nelveg-Kristensen, and Wladimir M. Szpirt. 2021. "Uncommon Presentation of Granulomatosis with Polyangiitis Mimicking Metastatic Lung Cancer" Clinics and Practice 11, no. 2: 293-302. https://doi.org/10.3390/clinpract11020042
APA StyleUrbanska, E. M., Elversang, J., Colville-Ebeling, B., Löfgren, J. O., Nelveg-Kristensen, K. E., & Szpirt, W. M. (2021). Uncommon Presentation of Granulomatosis with Polyangiitis Mimicking Metastatic Lung Cancer. Clinics and Practice, 11(2), 293-302. https://doi.org/10.3390/clinpract11020042





