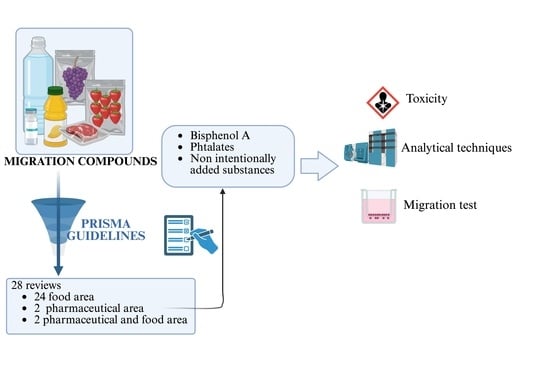
A Systematic Review: Migration of Chemical Compounds from Plastic Material Containers in Food and Pharmaceutical Fields
Abstract
1. Introduction
1.1. Plastic Containers, Advantages and Disadvantages for Their Uses
1.2. Additives Used in Plastic Materials, Risk Assessment of Health Human
1.3. Leaching or Migration Processes, Consequences in Food and Pharmaceutical Fields Area
2. Methods
2.1. Search Strategy
2.2. Inclusion and Exclusion Criteria
2.3. Study Selection
2.4. Data Extraction
2.5. Data Quality Assessment
2.6. Data Analysis/Synthesis
3. Results and Discussion
3.1. Search Results and Included Studies
3.2. Study Characteristic
3.2.1. Toxicity Risk Assessment
- -
- Class I (low toxicity): Encompasses compounds with simple chemical structures and efficient metabolic pathways, which are indicative of a low toxic potential. A TTC threshold value of 30 µg/kg body weight/day is assigned. Typical examples include sugars and amino acids.
- -
- Class II (intermediate toxicity): Includes compounds with moderately complex chemical structures. The TTC threshold is set at 9 µg/kg body weight/day. Alcohols and esters are representative of this category.
- -
- Class III (high toxicity): Comprises compounds whose structural features suggest potential toxicity, including aromatic substances. These are assigned the most conservative TTC threshold value of 1.5 µg/kg body weight/day.
3.2.2. Analytical Techniques
3.2.3. Migration Tests
4. Discussion
4.1. Key Findings
4.2. Future Policy and Research
4.3. Strengths and Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A. PRISMA Abstract and Review Guidelines Checklist
| Section and Topic | Item # | Checklist Item | Reported (Yes/No) |
| TITLE | |||
| Title | 1 | Identify the report as a systematic review. | Yes |
| BACKGROUND | |||
| Objectives | 2 | Provide an explicit statement of the main objective(s) or question(s) the review addresses. | Yes |
| METHODS | |||
| Eligibility criteria | 3 | Specify the inclusion and exclusion criteria for the review. | Yes |
| Information sources | 4 | Specify the information sources (e.g., databases, registers) used to identify studies and the date when each was last searched. | Yes |
| Risk of bias | 5 | Specify the methods used to assess risk of bias in the included studies. | Yes |
| Synthesis of results | 6 | Specify the methods used to present and synthesize results. | Yes |
| RESULTS | |||
| Included studies | 7 | Give the total number of included studies and participants and summarize relevant characteristics of studies. | Yes |
| Synthesis of results | 8 | Present results for main outcomes, preferably indicating the number of included studies and participants for each. If meta-analysis was performed, report the summary estimate and confidence/credible interval. If comparing groups, indicate the direction of the effect (i.e., which group is favoured). | Yes |
| DISCUSSION | |||
| Limitations of evidence | 9 | Provide a brief summary of the limitations of the evidence included in the review (e.g., study risk of bias, inconsistency and imprecision). | No |
| Interpretation | 10 | Provide a general interpretation of the results and important implications. | Yes |
| OTHER | |||
| Funding | 11 | Specify the primary source of funding for the review. | Yes |
| Registration | 12 | Provide the register name and registration number. | Yes |
| Section and Topic | Item # | Checklist Item | Location Where Item Is Reported |
| TITLE | |||
| Title | 1 | Identify the report as a systematic review. | p. 1 |
| ABSTRACT | |||
| Abstract | 2 | See the PRISMA 2020 for Abstracts checklist. | p. 1 |
| INTRODUCTION | |||
| Rationale | 3 | Describe the rationale for the review in the context of existing knowledge. | p. 1–3 |
| Objectives | 4 | Provide an explicit statement of the objective(s) or question(s) the review addresses. | p. 3 |
| METHODS | |||
| Eligibility criteria | 5 | Specify the inclusion and exclusion criteria for the review and how studies were grouped for the syntheses. | 2.2 |
| Information sources | 6 | Specify all databases, registers, websites, organizations, reference lists and other sources searched or consulted to identify studies. Specify the date when each source was last searched or consulted. | 2.1 |
| Search strategy | 7 | Present the full search strategies for all databases, registers and websites, including any filters and limits used. | 2.1 |
| Selection process | 8 | Specify the methods used to decide whether a study met the inclusion criteria of the review, including how many reviewers screened each record and each report retrieved, whether they worked independently, and if applicable, details of automation tools used in the process. | 2.3 |
| Data collection process | 9 | Specify the methods used to collect data from reports, including how many reviewers collected data from each report, whether they worked independently, any processes for obtaining or confirming data from study investigators, and if applicable, details of automation tools used in the process. | 2.4 |
| Data items | 10a | List and define all outcomes for which data were sought. Specify whether all results that were compatible with each outcome domain in each study were sought (e.g., for all measures, time points, analyses), and if not, the methods used to decide which results to collect. | 2.4 |
| 10b | List and define all other variables for which data were sought (e.g., participant and intervention characteristics, funding sources). Describe any assumptions made about any missing or unclear information. | N/A | |
| Study risk of bias assessment | 11 | Specify the methods used to assess risk of bias in the included studies, including details of the tool(s) used, how many reviewers assessed each study and whether they worked independently, and if applicable, details of automation tools used in the process. | 2.5 |
| Effect measures | 12 | Specify for each outcome the effect measure(s) (e.g., risk ratio, mean difference) used in the synthesis or presentation of results. | N/A |
| Synthesis methods | 13a | Describe the processes used to decide which studies were eligible for each synthesis (e.g., tabulating the study intervention characteristics and comparing against the planned groups for each synthesis (item #5)). | 2.3 |
| 13b | Describe any methods required to prepare the data for presentation or synthesis, such as handling of missing summary statistics, or data conversions. | 2.4 | |
| 13c | Describe any methods used to tabulate or visually display results of individual studies and syntheses. | 2.4 | |
| 13d | Describe any methods used to synthesize results and provide a rationale for the choice(s). If meta-analysis was performed, describe the model(s), method(s) to identify the presence and extent of statistical heterogeneity, and software package(s) used. | N/A | |
| 13e | Describe any methods used to explore possible causes of heterogeneity among study results (e.g., subgroup analysis, meta-regression). | N/A | |
| 13f | Describe any sensitivity analyses conducted to assess robustness of the synthesized results. | N/A | |
| Reporting bias assessment | 14 | Describe any methods used to assess risk of bias due to missing results in a synthesis (arising from reporting biases). | 2.6 |
| Certainty assessment | 15 | Describe any methods used to assess certainty (or confidence) in the body of evidence for an outcome. | N/A |
| RESULTS | |||
| Study selection | 16a | Describe the results of the search and selection process, from the number of records identified in the search to the number of studies included in the review, ideally using a flow diagram. | 3.1 + Figure 1 |
| 16b | Cite studies that might appear to meet the inclusion criteria, but which were excluded, and explain why they were excluded. | 3.1 + Figure 1 | |
| Study characteristics | 17 | Cite each included study and present its characteristics. | 3.2 |
| Risk of bias in studies | 18 | Present assessments of risk of bias for each included study. | N/A |
| Results of individual studies | 19 | For all outcomes, present, for each study: (a) summary statistics for each group (where appropriate) and (b) an effect estimate and its precision (e.g., confidence/credible interval), ideally using structured tables or plots. | N/A |
| Results of syntheses | 20a | For each synthesis, briefly summarize the characteristics and risk of bias among contributing studies. | N/A |
| 20b | Present results of all statistical syntheses conducted. If meta-analysis was performed, present for each the summary estimate and its precision (e.g., confidence/credible interval) and measures of statistical heterogeneity. If comparing groups, describe the direction of the effect. | N/A | |
| 20c | Present results of all investigations of possible causes of heterogeneity among study results. | N/A | |
| 20d | Present results of all sensitivity analyses conducted to assess the robustness of the synthesized results. | N/A | |
| Reporting biases | 21 | Present assessments of risk of bias due to missing results (arising from reporting biases) for each synthesis assessed. | N/A |
| Certainty of evidence | 22 | Present assessments of certainty (or confidence) in the body of evidence for each outcome assessed. | N/A |
| DISCUSSION | |||
| Discussion | 23a | Provide a general interpretation of the results in the context of other evidence. | 4.1 |
| 23b | Discuss any limitations of the evidence included in the review. | N/A | |
| 23c | Discuss any limitations of the review processes used. | 4.3 | |
| 23d | Discuss implications of the results for practice, policy, and future research. | 4.2 | |
| OTHER INFORMATION | |||
| Registration and protocol | 24a | Provide registration information for the review, including register name and registration number, or state that the review was not registered. | 2 |
| 24b | Indicate where the review protocol can be accessed, or state that a protocol was not prepared. | 2 | |
| 24c | Describe and explain any amendments to information provided at registration or in the protocol. | N/A | |
| Support | 25 | Describe sources of financial or non-financial support for the review, and the role of the funders or sponsors in the review. | p. 1 |
| Competing interests | 26 | Declare any competing interests of review authors. | p. 17 |
| Availability of data, code and other materials | 27 | Report which of the following are publicly available and where they can be found: template data collection forms; data extracted from included studies; data used for all analyses; analytic code; any other materials used in the review. | N/A |
| N/A: not available. | |||
References
- Zuccarello, D.J.; Murphy, M.P.; Meyer, R.F.; Winslow, P.A. A Comprehensive Approach for the Determination of Extractable and Leachable Metals in Pharmaceutical Products by Inductively-Coupled Plasma. PDA J. Pharm. Sci. Technol. 2009, 63, 339–352. [Google Scholar]
- Bridson, J.H.; Gaugler, E.C.; Smith, D.A.; Northcott, G.L.; Gaw, S. Leaching and Extraction of Additives from Plastic Pollution to Inform Environmental Risk: A Multidisciplinary Review of Analytical Approaches. J. Hazard. Mater. 2021, 414, 125571. [Google Scholar] [CrossRef]
- Riboni, N.; Bianchi, F.; Cavazza, A.; Piergiovanni, M.; Mattarozzi, M.; Careri, M. Mass Spectrometry-Based Techniques for the Detection of Non-Intentionally Added Substances in Bioplastics. Separations 2023, 10, 222. [Google Scholar] [CrossRef]
- Horodytska, O.; Cabanes, A.; Fullana, A. Non-Intentionally Added Substances (NIAS) in Recycled Plastics. Chemosphere 2020, 251, 126373. [Google Scholar] [CrossRef]
- Bioplastics—European Bioplastics e.V. Available online: https://www.european-bioplastics.org/bioplastics/ (accessed on 3 November 2025).
- Groh, K.J.; Backhaus, T.; Carney-Almroth, B.; Geueke, B.; Inostroza, P.A.; Lennquist, A.; Leslie, H.A.; Maffini, M.; Slunge, D.; Trasande, L.; et al. Overview of Known Plastic Packaging-Associated Chemicals and Their Hazards. Sci. Total Environ. 2019, 651, 3253–3268. [Google Scholar] [CrossRef]
- Nerin, C.; Alfaro, P.; Aznar, M.; Domeño, C. The Challenge of Identifying Non-Intentionally Added Substances from Food Packaging Materials: A Review. Anal. Chim. Acta 2013, 775, 14–24. [Google Scholar] [CrossRef]
- Kissin, Y.V.; Liu, X.; Pollick, D.J.; Brungard, N.L.; Chang, M. Ziegler-Natta Catalysts for Propylene Polymerization: Chemistry of Reactions Leading to the Formation of Active Centers. J. Mol. Catal. A Chem. 2008, 287, 45–52. [Google Scholar] [CrossRef]
- Bach, C.; Dauchy, X.; Chagnon, M.C.; Etienne, S. Chemical Compounds and Toxicological Assessments of Drinking Water Stored in Polyethylene Terephthalate (PET) Bottles: A Source of Controversy Reviewed. Water Res. 2012, 46, 571–583. [Google Scholar] [CrossRef] [PubMed]
- Kostic, I.; Andjelkovic, T.; Andjelkovic, D.; Cvetkovic, T.; Pavlovic, D. Determination of Di(2-Ethylhexyl) Phthalate in Plastic Medical Devices. Hem. Ind. 2016, 70, 159–164. [Google Scholar] [CrossRef]
- Andjelković, T.; Bogdanović, D.; Kostić, I.; Kocić, G.; Nikolić, G.; Pavlović, R. Phthalates Leaching from Plastic Food and Pharmaceutical Contact Materials by FTIR and GC-MS. Environ. Sci. Pollut. Res. Int. 2021, 28, 31380–31390. [Google Scholar] [CrossRef]
- The European Parliament; The Council of the European Union. Regulation (EC) No 1272/2008: Classification, Labelling and Packaging of Substances and Mixtures, Amending and Repealing Directives 67/548/EEC and 1999/45/EC, and Amending Regulation (EC) No 1907/2006; European Parliament: Strasbourg, France, 2008. [Google Scholar]
- Rowdhwal, S.S.S.; Chen, J. Toxic Effects of Di-2-Ethylhexyl Phthalate: An Overview. Biomed. Res. Int. 2018, 2018, 1750368. [Google Scholar] [CrossRef]
- Hauser, R.; Calafat, A.M. Phthalates and Human Health. Occup. Environ. Med. 2005, 62, 806–818. [Google Scholar] [CrossRef]
- Ramesh Kumar, A.; Sivaperumal, P. Analytical Methods for the Determination of Biomarkers of Exposure to Phthalates in Human Urine Samples. TrAC Trends Anal. Chem. 2016, 75, 151–161. [Google Scholar] [CrossRef]
- Hlisníková, H.; Petrovičová, I.; Kolena, B.; Šidlovská, M.; Sirotkin, A. Effects and Mechanisms of Phthalates’ Action on Neurological Processes and Neural Health: A Literature Review. Pharmacol. Rep. 2021, 73, 386–404. [Google Scholar] [CrossRef]
- Zhang, Y.J.; Guo, J.L.; Xue, J.C.; Bai, C.L.; Guo, Y. Phthalate Metabolites: Characterization, Toxicities, Global Distribution, and Exposure Assessment. Environ. Pollut. 2021, 291, 118106. [Google Scholar] [CrossRef]
- Ventrice, P.; Ventrice, D.; Russo, E.; De Sarro, G. Phthalates: European Regulation, Chemistry, Pharmacokinetic and Related Toxicity. Environ. Toxicol. Pharmacol. 2013, 36, 88–96. [Google Scholar] [CrossRef]
- Wang, Y.; Qian, H. Phthalates and Their Impacts on Human Health. Healthcare 2021, 9, 603. [Google Scholar] [CrossRef]
- Silano, V.; Barat Baviera, J.M.; Bolognesi, C.; Chesson, A.; Cocconcelli, P.S.; Crebelli, R.; Gott, D.M.; Grob, K.; Lampi, E.; Mortensen, A.; et al. Update of the Risk Assessment of Di-Butylphthalate (DBP), Butyl-Benzyl-Phthalate (BBP), Bis(2-Ethylhexyl)Phthalate (DEHP), Di-Isononylphthalate (DINP) and Di-Isodecylphthalate (DIDP) for Use in Food Contact Materials. EFSA J. 2019, 17, e05838. [Google Scholar] [CrossRef] [PubMed]
- Schreier, V.N.; Çörek, E.; Appenzeller-Herzog, C.; Brüschweiler, B.J.; Geueke, B.; Wilks, M.F.; Schilter, B.; Muncke, J.; Simat, T.J.; Smieško, M.; et al. Evaluating the Food Safety and Risk Assessment Evidence-Base of Polyethylene Terephthalate Oligomers: A Systematic Evidence Map. Environ. Int. 2023, 176, 107978. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, L.; Dierkes, G.; Ternes, T.A.; Völker, C.; Wagner, M. Benchmarking the in Vitro Toxicity and Chemical Composition of Plastic Consumer Products. Environ. Sci. Technol. 2019, 53, 11467–11477. [Google Scholar] [CrossRef] [PubMed]
- Hahladakis, J.N.; Velis, C.A.; Weber, R.; Iacovidou, E.; Purnell, P. An Overview of Chemical Additives Present in Plastics: Migration, Release, Fate and Environmental Impact during Their Use, Disposal and Recycling. J. Hazard. Mater. 2018, 344, 179–199. [Google Scholar] [CrossRef]
- Piringer, O.G.; Baner, A.L. (Eds.) Plastic Packaging: Interactions with Food and Pharmaceuticals, 2nd ed.; Completely Revised Edition; Wiley: Hoboken, NJ, USA, 2008; ISBN 978-3-527-62143-9. [Google Scholar]
- Schettler, T. Human Exposure to Phthalates via Consumer Products. Int. J. Androl. 2006, 29, 134–139; discussion 181–185. [Google Scholar] [CrossRef]
- Center for Drug Evaluation and Research (CDER). Guidance for Industry Nasal Spray and Inhalation Solution, Suspension, and Spray Drug Products-Chemistry, Manufacturing, and Controls Documentation; CDER: Silver Spring, MD, USA, 2002. [Google Scholar]
- Cuadros-Rodríguez, L.; Lazúen-Muros, M.; Ruiz-Samblás, C.; Navas-Iglesias, N. Leachables from Plastic Materials in Contact with Drugs. State of the Art and Review of Current Analytical Approaches. Int. J. Pharm. 2020, 583, 119332. [Google Scholar] [CrossRef] [PubMed]
- Giuliani, A.; Zuccarini, M.; Cichelli, A.; Khan, H.; Reale, M. Critical Review on the Presence of Phthalates in Food and Evidence of Their Biological Impact. Int. J. Environ. Res. Public Health 2020, 17, 5655. [Google Scholar] [CrossRef]
- Jenke, D. Identification, Analysis and Safety Assessment of Leachables and Extractables. TrAC Trends Anal. Chem. 2018, 101, 56–65. [Google Scholar] [CrossRef]
- Pack, E.C.; Lee, K.Y.; Jung, J.S.; Jang, D.Y.; Kim, H.S.; Koo, Y.J.; Lee, H.G.; Kim, Y.S.; Lim, K.M.; Lee, S.H.; et al. Determination of the Migration of Plastic Additives and Non-Intentionally Added Substances into Food Simulants and the Assessment of Health Risks from Convenience Food Packaging. Food Packag. Shelf Life 2021, 30, 100736. [Google Scholar] [CrossRef]
- Aznar, M.; Ubeda, S.; Dreolin, N.; Nerín, C. Determination of Non-Volatile Components of a Biodegradable Food Packaging Material Based on Polyester and Polylactic Acid (PLA) and Its Migration to Food Simulants. J. Chromatogr. A 2019, 1583, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Ong, H.-T.; Samsudin, H.; Soto-Valdez, H. Migration of Endocrine-Disrupting Chemicals into Food from Plastic Packaging Materials: An Overview of Chemical Risk Assessment, Techniques to Monitor Migration, and International Regulations. Crit. Rev. Food Sci. Nutr. 2022, 62, 957–979. [Google Scholar] [CrossRef] [PubMed]
- United States Pharmacopeia. <1664> Assessment of Drug Product Leachables Associated with Pharmaceutical Packaging/Delivery Systems; United States Pharmacopeia: North Bethesda, MA, USA, 2024. [Google Scholar]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 Statement: An Updated Guideline for Reporting Systematic Reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- Landrigan, P.J.; Raps, H.; Cropper, M.; Bald, C.; Brunner, M.; Canonizado, E.M.; Charles, D.; Chiles, T.C.; Donohue, M.J.; Enck, J.; et al. The Minderoo-Monaco Commission on Plastics and Human Health. Ann. Glob. Health 2023, 89, 23. [Google Scholar] [CrossRef]
- Sica, V.P.; Krivos, K.L.; Kiehl, D.E.; Pulliam, C.J.; Henry, I.D.; Baker, T.R. The Role of Mass Spectrometry and Related Techniques in the Analysis of Extractable and Leachable Chemicals. Mass Spectrom. Rev. 2020, 39, 212–226. [Google Scholar] [CrossRef]
- Muncke, J. Endocrine Disrupting Chemicals and Other Substances of Concern in Food Contact Materials: An Updated Review of Exposure, Effect and Risk Assessment. J. Steroid Biochem. Mol. Biol. 2011, 127, 118–127. [Google Scholar] [CrossRef]
- Bhogal, S.; Grover, A.; Mohiuddin, I. A Review of the Analysis of Phthalates by Gas Chromatography in Aqueous and Food Matrices. Crit. Rev. Anal. Chem. 2024, 54, 3428–3452. [Google Scholar] [CrossRef]
- Cao, X.L. Phthalate Esters in Foods: Sources, Occurrence, and Analytical Methods. Compr. Rev. Food Sci. Food Saf. 2010, 9, 21–43. [Google Scholar] [CrossRef] [PubMed]
- Vinod, V.; Amritha, P.S.; Harathi, P.B. A Systematic Review on Bisphenols—Sources, Health Impacts, and Analytical Determination of Bisphenol A in Aqueous Samples. Sep. Sci. Plus 2023, 6, 2300072. [Google Scholar] [CrossRef]
- Gugoasa, L.A.D. Review—Electrochemical Sensors for Determination of the Endocrine Disruptor, Bisphenol A. J. Electrochem. Soc. 2020, 167, 037506. [Google Scholar] [CrossRef]
- Cao, X.L. A Review Recent Development on Analytical Methods for Determination of Bisphenol a in Food and Biological Samples. J. Liq. Chromatogr. Relat. Technol. 2012, 35, 2795–2829. [Google Scholar] [CrossRef]
- Qi, M.; Li, Y.; Zhu, Z.; Du, B.; Chen, D. Current Sample Preparation Methods and Determination Techniques for the Determination of Phthalic Acid Ester Plasticizers in Edible Oils. Molecules 2023, 28, 5106. [Google Scholar] [CrossRef] [PubMed]
- Priovolos, I.; Samanidou, V. Βisphenol A and Its Analogs Migrated from Contact Materials into Food and Beverages: An Updated Review in Sample Preparation Approaches. J. Sep. Sci. 2023, 46, e2300081. [Google Scholar] [CrossRef] [PubMed]
- Krska, R.; Becalski, A.; Braekevelt, E.; Koerner, T.; Cao, X.L.; Dabeka, R.; Godefroy, S.; Lau, B.; Moisey, J.; Rawn, D.F.K.; et al. Challenges and Trends in the Determination of Selected Chemical Contaminants and Allergens in Food. Anal. Bioanal. Chem. 2012, 402, 139–162. [Google Scholar] [CrossRef]
- Martínez-Bueno, M.J.; Gómez Ramos, M.J.; Bauer, A.; Fernández-Alba, A.R. An Overview of Non-Targeted Screening Strategies Based on High Resolution Accurate Mass Spectrometry for the Identification of Migrants Coming from Plastic Food Packaging Materials. TrAC Trends Anal. Chem. 2019, 110, 191–203. [Google Scholar] [CrossRef]
- Salazar-Beltrán, D.; Hinojosa-Reyes, L.; Ruiz-Ruiz, E.; Hernández-Ramírez, A.; Guzmán-Mar, J.L. Phthalates in Beverages and Plastic Bottles: Sample Preparation and Determination. Food Anal. Methods 2018, 11, 48–61. [Google Scholar] [CrossRef]
- González-Sálamo, J.; Socas-Rodríguez, B.; Hernández-Borges, J. Analytical Methods for the Determination of Phthalates in Food. Curr. Opin. Food Sci. 2018, 22, 122–136. [Google Scholar] [CrossRef]
- Zhang, Y.; Lei, Y.; Lu, H.; Shi, L.; Wang, P.; Ali, Z.; Li, J. Electrochemical Detection of Bisphenols in Food: A Review. Food Chem. 2021, 346, 128895. [Google Scholar] [CrossRef] [PubMed]
- Freitas, F.; Cabrita, M.J.; da Silva, M.G. A Critical Review of Analytical Methods for the Quantification of Phthalates Esters in Two Important European Food Products: Olive Oil and Wine. Molecules 2023, 28, 7628. [Google Scholar] [CrossRef]
- S, A.P.; Vinod, V.; Harathi, P.B. A Critical Review on Extraction and Analytical Methods of Phthalates in Water and Beverages. J. Chromatogr. A 2022, 1675, 463175. [Google Scholar] [CrossRef]
- Kato, L.S.; Conte-Junior, C.A. Safety of Plastic Food Packaging: The Challenges about Non-Intentionally Added Substances (NIAS) Discovery, Identification and Risk Assessment. Polymers 2021, 13, 2077. [Google Scholar] [CrossRef]
- Urbelis, J.H.; Cooper, J.R. Migration of Food Contact Substances into Dry Foods: A Review. Food Addit. Contam. Part. A Chem. Anal. Control Expo. Risk Assess. 2021, 38, 1044–1073. [Google Scholar] [CrossRef]
- Kappenstein, O.; Vieth, B.; Luch, A.; Pfaff, K. Toxicologically Relevant Phthalates in Food. Exp. Suppl. 2012, 101, 87–106. [Google Scholar] [CrossRef] [PubMed]
- Maragou, N.C.; Tzachristas, A.; Tsochatzis, E.D.; Thomaidis, N.S. Chemical Migration from Wine Contact Materials. Appl. Sci. 2024, 14, 6507. [Google Scholar] [CrossRef]
- Alberto Lopes, J.; Tsochatzis, E.D. Poly(Ethylene Terephthalate), Poly(Butylene Terephthalate), and Polystyrene Oligomers: Occurrence and Analysis in Food Contact Materials and Food. J. Agric. Food Chem. 2023, 71, 2244–2258. [Google Scholar] [CrossRef]
- Pawlicka, M.; Struciński, P.; Postupolski, J. The Use of Meta-Analysis in Food Contact Materials Risk Assessment. Hum. Ecol. Risk Assess. Int. J. 2020, 26, 1742–1754. [Google Scholar] [CrossRef]
- Hoppe, M.; de Voogt, P.; Franz, R. Identification and Quantification of Oligomers as Potential Migrants in Plastics Food Contact Materials with a Focus in Polycondensates—A Review. Trends Food Sci. Technol. 2016, 50, 118–130. [Google Scholar] [CrossRef]
- Miralles, P.; Fuentes-Ferragud, E.; Socas-Hernández, C.; Coscollà, C. Recent Trends and Challenges on the Non-Targeted Analysis and Risk Assessment of Migrant Non-Intentionally Added Substances from Plastic Food Contact Materials. Toxics 2025, 13, 543. [Google Scholar] [CrossRef]
- Kaya, S.I.; Cetinkaya, A.; Ozkan, S.A. Latest Advances in Determination of Bisphenols with Nanomaterials, Molecularly Imprinted Polymers and Aptamer Based Electrochemical Sensors. Crit. Rev. Anal. Chem. 2022, 52, 1223–1243. [Google Scholar] [CrossRef] [PubMed]
- Bernard, L.; Décaudin, B.; Lecoeur, M.; Richard, D.; Bourdeaux, D.; Cueff, R.; Sautou, V. Analytical Methods for the Determination of DEHP Plasticizer Alternatives Present in Medical Devices: A Review. Talanta 2014, 129, 39–54. [Google Scholar] [CrossRef] [PubMed]
- Elsa, O.; Emmanuelle, B.; Sébastien, H.; Anne-Lise, R.; Fabrice, M.; Hélène, G.; Paul, H.; Gérald, R.; Gaud, D.P.; Cariou, R.; et al. Toward the Characterisation of Non-Intentionally Added Substances Migrating from Polyester-Polyurethane Lacquers by Comprehensive Gas Chromatography-Mass Spectrometry Technologies. J. Chromatogr. A 2019, 1601, 327–334. [Google Scholar] [CrossRef]
- Bschir, K. Risk, Uncertainty and Precaution in Science: The Threshold of the Toxicological Concern Approach in Food Toxicology. Sci. Eng. Ethics 2017, 23, 489–508. [Google Scholar] [CrossRef] [PubMed]
- Félix, J.S.; Isella, F.; Bosetti, O.; Nerín, C. Analytical Tools for Identification of Non-Intentionally Added Substances (NIAS) Coming from Polyurethane Adhesives in Multilayer Packaging Materials and Their Migration into Food Simulants. Anal. Bioanal. Chem. 2012, 403, 2869–2882. [Google Scholar] [CrossRef]
- Alin, J.; Hakkarainen, M. Microwave Heating Causes Rapid Degradation of Antioxidants in Polypropylene Packaging, Leading to Greatly Increased Specific Migration to Food Simulants as Shown by ESI-MS and GC-MS. J. Agric. Food Chem. 2011, 59, 5418–5427. [Google Scholar] [CrossRef]
- Bignardi, C.; Cavazza, A.; Laganà, C.; Salvadeo, P.; Corradini, C. Release of Non-Intentionally Added Substances (NIAS) from Food Contact Polycarbonate: Effect of Ageing. Food Control 2017, 71, 329–335. [Google Scholar] [CrossRef]
- Gelbke, H.P.; Banton, M.; Block, C.; Dawkins, G.; Eisert, R.; Leibold, E.; Pemberton, M.; Puijk, I.M.; Sakoda, A.; Yasukawa, A. Risk Assessment for Migration of Styrene Oligomers into Food from Polystyrene Food Containers. Food Chem. Toxicol. 2019, 124, 151–167. [Google Scholar] [CrossRef]
- Eckardt, M.; Schneider, J.; Simat, T.J. In Vitro Intestinal Digestibility of Cyclic Aromatic Polyester Oligomers from Polyethylene Terephthalate (PET) and Polybutylene Terephthalate (PBT). Food Addit. Contam. Part. A Chem. Anal. Control Expo. Risk Assess. 2019, 36, 1882–1894. [Google Scholar] [CrossRef]
- Mertens, B.; Van Hoeck, E.; Blaude, M.N.; Simon, C.; Onghena, M.; Vandermarken, T.; Van Langenhove, K.; Demaegdt, H.; Vandermeiren, K.; Covaci, A.; et al. Evaluation of the Potential Health Risks of Substances Migrating from Polycarbonate Replacement Baby Bottles. Food Chem. Toxicol. 2016, 97, 108–119. [Google Scholar] [CrossRef]
- Food Packaging Forum Foundation Food Packaging Forum Foundation. FCCmigex Database. Available online: https://foodpackagingforum.org/resources/databases/fccmigex (accessed on 29 October 2024).
- Krishnan, A.V.; Stathis, P.; Permuth, S.F.; Tokes, L.; Feldman, D. Bisphenol-A: An Estrogenic Substance Is Released from Polycarbonate Flasks during Autoclaving. Endocrinology 1993, 132, 2279–2286. [Google Scholar] [CrossRef] [PubMed]
- Sakuragi, Y.; Takada, H.; Sato, H.; Kubota, A.; Terasaki, M.; Takeuchi, S.; Ikeda-Araki, A.; Watanabe, Y.; Kitamura, S.; Kojima, H. An Analytical Survey of Benzotriazole UV Stabilizers in Plastic Products and Their Endocrine-Disrupting Potential via Human Estrogen and Androgen Receptors. Sci. Total Environ. 2021, 800, 149374. [Google Scholar] [CrossRef]
- Loyo-Rosales, J.E.; Rosales-Rivera, G.C.; Lynch, A.M.; Rice, C.P.; Torrents, A. Migration of Nonylphenol from Plastic Containers to Water and a Milk Surrogate. J. Agric. Food Chem. 2004, 52, 2016–2020. [Google Scholar] [CrossRef]
- Maragou, N.C.; Thomaidis, N.S.; Theodoridis, G.A.; Lampi, E.N.; Koupparis, M.A. Determination of Bisphenol A in Canned Food by Microwave Assisted Extraction, Molecularly Imprinted Polymer-Solid Phase Extraction and Liquid Chromatography-Mass Spectrometry. J. Chromatogr. B 2020, 1137, 121938. [Google Scholar] [CrossRef] [PubMed]
- Myint Zaw, M.; Poorahong, S.; Kanatharana, P.; Thavarungkul, P.; Thammakhet-Buranachai, C. Waste Polystyrene Foam-Derived Sorbent for Determining Bisphenol-A from Canned Beverages. Food Chem. 2023, 405, 134834. [Google Scholar] [CrossRef]
- Zhou, Q.; Jin, Z.; Li, J.; Wang, B.; Wei, X.; Chen, J. A Novel Air-Assisted Liquid-Liquid Microextraction Based on in-Situ Phase Separation for the HPLC Determination of Bisphenols Migration from Disposable Lunch Boxes to Contacting Water. Talanta 2018, 189, 116–121. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.J.; Yun, E.S.; Shin, J.M.; Kim, Y.S.; Lee, J.S.; Lee, J.H.; Kim, D.G.; Oh, Y.H.; Jung, K.; Kim, G.H. Concentrations of Bisphenols in Canned Foods and Their Risk Assessment in Korea. J. Food Prot. 2018, 81, 903–916. [Google Scholar] [CrossRef]
- Khan, M.R.; Ouladsmane, M.; Alammari, A.M.; Azam, M. Bisphenol A Leaches from Packaging to Fruit Juice Commercially Available in Markets. Food Packag. Shelf Life 2021, 28, 100678. [Google Scholar] [CrossRef]
- Kalogiouri, N.P.; Samanidou, V. The Use of Molecular Imprinted Polymers Prior to Chromatographic Methods for the Analysis of Bisphenols in Packaged Foods. LCGC N. Am. 2021, 39, 582–587. [Google Scholar]
- Lian, L.; Jiang, X.; Guan, J.; Qiu, Z.; Wang, X.; Lou, D. Dispersive Solid-Phase Extraction of Bisphenols Migrated from Plastic Food Packaging Materials with Cetyltrimethylammonium Bromide-Intercalated Zinc Oxide. J. Chromatogr. A 2020, 1612, 460666. [Google Scholar] [CrossRef] [PubMed]
- Siddique, S.; Zhang, G.; Coleman, K.; Kubwabo, C. Investigation of the Migration of Bisphenols from Baby Bottles and Sippy Cups. Curr. Res. Food Sci. 2021, 4, 619–626. [Google Scholar] [CrossRef]
- Zhang, Y.; Yuan, Z.-L.; Deng, X.-Y.; Wei, H.-D.; Wang, W.-L.; Xu, Z.; Feng, Y.; Shi, X. Metal-Organic Framework Mixed-Matrix Membrane-Based Extraction Combined HPLC for Determination of Bisphenol A in Milk and Milk Packaging. Food Chem. 2022, 386, 132753. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Z.; Liu, J.; Wang, M.; Cao, J.; Li, L.; Wang, C.; Feng, N. Selective Sensing of Bisphenol A and Bisphenol S on Platinum/Poly(Diallyl Dimethyl Ammonium Chloride)-Diamond Powder Hybrid Modified Glassy Carbon Electrode. J. Electrochem. Soc. 2016, 163, B192–B199. [Google Scholar] [CrossRef]
- Zhu, G.; Qian, J.; Sun, H.; Yi, Y.; Wu, X. Enhanced Electrochemical Sensing Based on Perylenetetracarboxylic Acid Functionalized Carbon Nanohorns Hybrids Modified Electrode. J. Electrochem. Soc. 2017, 164, H545–H549. [Google Scholar] [CrossRef]
- Stefan-van Staden, R.-I.; Moscalu-Lungu, A.; van Staden, J.F. Nanostructured Materials Used for Pattern Recognition of Bisphenols in Waste Water Samples. J. Electrochem. Soc. 2019, 166, B903–B907. [Google Scholar] [CrossRef]
- Osorio, J.; Aznar, M.; Nerín, C.; Birse, N.; Elliott, C.; Chevallier, O. Ambient Mass Spectrometry as a Tool for a Rapid and Simultaneous Determination of Migrants Coming from a Bamboo-Based Biopolymer Packaging. J. Hazard. Mater. 2020, 398, 122891. [Google Scholar] [CrossRef]
- Osorio, J.; Aznar, M.; Nerín, C.; Elliott, C.; Chevallier, O. Comparison of LC-ESI, DART, and ASAP for the Analysis of Oligomers Migration from Biopolymer Food Packaging Materials in Food (Simulants). Anal. Bioanal. Chem. 2022, 414, 1335–1345. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Chen, C.; Ouyang, C.; Zeng, X.; Guo, Z.; Lai, F.; Li, J. Analysis of Oligomers in Poly (Butylene Succinate) and Poly (Butylene Adipate-Co-Terephthalate). Polym. Bull. 2023, 80, 4487–4502. [Google Scholar] [CrossRef]
- Wu, P.; Yang, D.; Zhang, L.; Shen, X.; Pan, X.; Wang, L.; Zhang, J.; Tan, Y.; Feng, L.; Ying, Y. Simultaneous Determination of 17 Phthalate Esters in Edible Vegetable Oils by GC-MS with Silica/PSA-Mixed Solid-Phase Extraction. J. Sep. Sci. 2012, 35, 2932–2939. [Google Scholar] [CrossRef]
- Shi, L.K.; Zhang, M.M.; Liu, Y.L. Concentration and Survey of Phthalic Acid Esters in Edible Vegetable Oils and Oilseeds by Gas Chromatography-Mass Spectrometry in China. Food Control 2016, 68, 118–123. [Google Scholar] [CrossRef]
- Zhou, R.-Z.; Jiang, J.; Mao, T.; Zhao, Y.-S.; Lu, Y. Multiresidue Analysis of Environmental Pollutants in Edible Vegetable Oils by Gas Chromatography-Tandem Mass Spectrometry. Food Chem. 2016, 207, 43–50. [Google Scholar] [CrossRef]
- Anastassiades, M.; Lehotay, S.J.; Stajnbaher, D.; Schenck, F.J. Fast and Easy Multiresidue Method Employing Acetonitrile Extraction/Partitioning and “Dispersive Solid-Phase Extraction” for the Determination of Pesticide Residues in Produce. J. AOAC Int. 2003, 86, 412–431. [Google Scholar] [CrossRef]
- Wang, X.; Sun, X.; Wang, X.; Qi, X.; Wang, D.; Jiang, J.; Mao, J.; Ma, F.; Yu, L.; Zhang, L.; et al. Determination of 15 Phthalic Acid Esters Based on GC–MS/MS Coupled with Modified QuEChERS in Edible Oils. Food Chem. X 2022, 16, 100520. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Tian, W.; Fang, Y.; Yang, W.; Hu, Q.; Pei, F. Rapid and Simultaneous Extraction of Phthalates, Polychlorinated Biphenyls and Polycyclic Aromatic Hydrocarbons from Edible Oil for GC–MS Determination. J. Food Compos. Anal. 2022, 114, 104827. [Google Scholar] [CrossRef]
- Wang, X.; Nag, R.; Brunton, N.P.; Siddique, M.A.B.; Harrison, S.M.; Monahan, F.J.; Cummins, E. A Probabilistic Approach to Model Bisphenol A (BPA) Migration from Packaging to Meat Products. Sci. Total Environ. 2023, 854, 158815. [Google Scholar] [CrossRef]
- Park, S.R.; Park, S.J.; Jeong, M.J.; Choi, J.C.; Kim, M.K. Fast and Simple Determination and Exposure Assessment of Bisphenol A, Phenol, p-Tert-Butylphenol, and Diphenylcarbonate Transferred from Polycarbonate Food-Contact Materials to Food Simulants. Chemosphere 2018, 203, 300–306. [Google Scholar] [CrossRef] [PubMed]
- Aznar, M.; Domeño, C.; Osorio, J.; Nerin, C. Release of Volatile Compounds from Cooking Plastic Bags under Different Heating Sources. Food Packag. Shelf Life 2020, 26, 100552. [Google Scholar] [CrossRef]


| Author and Year | Analytes | Food/Drugs | Toxicity Risk Assessment | Analytical Techniques | Migration Tests |
|---|---|---|---|---|---|
| Muncke, 2011 [37] | BPA | Food | YES | YES | YES |
| Bhogal et al., 2023 [38] | Phthalates | Food | YES | ||
| Cao, 2010 [39] | Phthalates | Food | YES | YES | |
| Vinod et al., 2023 [40] | BPA | Food | YES | ||
| Gugoasa, 2019 [41] | BPA | Food | YES | ||
| Cao, 2012 [42] | BPA | Food | YES | ||
| Qi et al., 2023 [43] | Phthalates | Food | YES | ||
| Priovolos et al., 2023 [44] | BPA | Food | YES | YES | YES |
| Krska et al., 2012 [45] | EDCs | Food | YES | YES | |
| Martínez-Bueno et al., 2019 [46] | NIAS | Food | YES | ||
| Salazar-Beltrán et al., 2018 [47] | Phthalates | Food | YES | YES | |
| González-Sálamo et al., 2018 [48] | Phthalates | Food | YES | ||
| Zhang et al., 2021 [49] | BPs | Food | YES | ||
| Freitas et al., 2023 [50] | Phthalates | Food | YES | YES | |
| Amritha et al., 2022 [51] | Phthalates | Food | YES | YES | |
| Kato et al., 2021 [52] | NIAS | Food | YES | YES | YES |
| Urbelis et al., 2021 [53] | Leachable compounds | Food | YES | ||
| Kappenstein et al., 2012 [54] | Phthalates | Food | YES | ||
| Maragou et al., 2024 [55] | Leachable compounds | Food | YES | YES | YES |
| Lopes et al., 2023 [56] | Leachable compounds | Food | YES | YES | |
| Pawlicka et al., 2020 [57] | NIAS | Food | YES | ||
| Hoppe et al., 2016 [58] | Oligomers, NIAS | Food | YES | YES | |
| Riboni et al., 2023 [3] | NIAS | Food | YES | YES | |
| Miralles et al., 2025 [59] | NIAS | Food | YES | YES | YES |
| Sica et al., 2020 [36] | Leachable compounds | Food/Drugs | YES | YES | |
| Landrigan, et al., 2023 [35] | NIAS and others * | Food/Drugs | YES | ||
| Kaya et al., 2022 [60] | BPs | Drug | YES | YES | |
| Bernard et al., 2014 [61] | Phthalates | Drug | YES | YES |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Culleré, L.; Sangüesa, E.; Lomba, L.; Ribate, M.P.; Zuriaga, E.; García, C.B. A Systematic Review: Migration of Chemical Compounds from Plastic Material Containers in Food and Pharmaceutical Fields. J. Xenobiot. 2025, 15, 194. https://doi.org/10.3390/jox15060194
Culleré L, Sangüesa E, Lomba L, Ribate MP, Zuriaga E, García CB. A Systematic Review: Migration of Chemical Compounds from Plastic Material Containers in Food and Pharmaceutical Fields. Journal of Xenobiotics. 2025; 15(6):194. https://doi.org/10.3390/jox15060194
Chicago/Turabian StyleCulleré, Laura, Estela Sangüesa, Laura Lomba, María Pilar Ribate, Estefanía Zuriaga, and Cristina B. García. 2025. "A Systematic Review: Migration of Chemical Compounds from Plastic Material Containers in Food and Pharmaceutical Fields" Journal of Xenobiotics 15, no. 6: 194. https://doi.org/10.3390/jox15060194
APA StyleCulleré, L., Sangüesa, E., Lomba, L., Ribate, M. P., Zuriaga, E., & García, C. B. (2025). A Systematic Review: Migration of Chemical Compounds from Plastic Material Containers in Food and Pharmaceutical Fields. Journal of Xenobiotics, 15(6), 194. https://doi.org/10.3390/jox15060194