Neonatal Hearing Rescreening in a Second-Level Hospital: Problems and Solutions
Abstract
1. Introduction
- The rescreening volume and neonates’ origin;
- The performance of rescreening in neonates born in the center’s maternity ward;
- The performance of screening and rescreening in neonates born outside the center’s maternity ward;
- To determine the rescreening times in both groups;
- To know the final ENT diagnosis.
2. Materials and Methods
2.1. Protocol
2.2. Techniques or Equipment
2.2.1. Transient Evoked Otoacoustic Emissions (TEOAE)
2.2.2. Automatic Auditory Brainstem Response (AABR)
2.2.3. Data Analysis
2.3. Study Variables
- TEOAE test result in first and second test (normal/altered);
- ABR test result (normal/altered);
- Hospital of birth (internal/external);
- Test in otorhinolaryngology (ENT) (normal/altered);
- Intervals in days between the different phases of the screening.
3. Results
4. Discussion
4.1. Hospital of Birth of Screened Newborns
4.2. Rescreening Timing
4.3. Quality of Care
4.4. Screening and ENT Results
4.5. Losses (Lost to Follow Up)
5. Limitations
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Trinidad-Ramos, G.; de Aguilar, V.A.; Jaudenes-Casaubón, C.; Núñez-Batalla, F.; Sequí-Canet, J.M. Early hearing detection and intervention: 2010 CODEPEH recommendation. Acta Otorrinolaringol. Esp. 2010, 61, 69–77. [Google Scholar] [CrossRef]
- Sequi Canet, J.M.; Sala Langa, M.J.; Collar Del Castillo, J.I. Análisis crítico de una década de cribado neonatal de hipoacusia en un hospital comarcal [Results from ten years newborn hearing screening in a secondary hospital]. An. Pediatría 2016, 85, 189–196. [Google Scholar] [CrossRef]
- Bussé, A.M.L.; Mackey, A.R.; Hoeve, H.L.J.; Goedegebure, A.; Carr, G.; Uhlén, I.M.; Simonsz, H.J. Assessment of hearing screening programmes across 47 countries or regions I: Provision of newborn hearing screening. Int. J. Audiol. 2021, 60, 821–830. [Google Scholar] [CrossRef]
- Kanji, A.; Khoza-Shangase, K.; Moroe, N. Newborn hearing screening protocols and their outcomes: A systematic review. Int. J. Pediatr. Otorhinolaryngol. 2018, 115, 104–109. [Google Scholar] [CrossRef]
- Organización Panamericana de la Salud. Informe Mundial Sobre la Audición 2021. Available online: https://doi.org/10.37774/9789275324677 (accessed on 4 April 2023).
- Núñez-Batalla, F.; Jaudenes-Casaubón, C.; Sequí-Canet, J.M.; Vivanco-Allende, A.; Zubicaray-Ugarteche, J. Actualización de los programas de detección precoz de la sordera infantil: Recomendaciones CODEPEH 2018 (Nivel 1 Detección). Rev. Española Discapac. 2019, 7, 201–220. [Google Scholar] [CrossRef]
- Trinidad, G.; Jaudenes-Casaubón, C. Sordera Infantil. In Diagnóstico Precoz a la Inclusión Educativa. Guía Práctica para el Abordaje Interdisciplinar, 2nd ed.; Confederación Española de Familias de Personas Sordas-FIAPAS: Madrid, Spain, 2012; Available online: http://www.fiapas.es/sordera-infantil-del-diagnostico-precoz-la-inclusion-educativa (accessed on 29 March 2023).
- Kemp, D.T.; Ryan, S.; Bray, P. A Guide to the Effective Use of Otoacoustic Emissions. Ear Hear. 1990, 11, 93–105. [Google Scholar] [CrossRef]
- Shang, Y.; Hao, W.; Gao, Z.; Xu, C.; Ru, Y.; Ni, D. An effective compromise between cost and referral rate: A sequential hearing screening protocol using TEOAEs and AABRs for healthy newborns. Int. J. Pediatr. Otorhinolaryngol. 2016, 91, 141–145. [Google Scholar] [CrossRef]
- Generalitat Valenciana. Decreto 218/2018, de 30 de Noviembre, del Consell, por el que se Regulan los Programas de Cribados Neonatales en la Comunitat Valenciana, Detección Precoz de la Hipoacusia Neonatal y Cribado Neonatal de Enfermedades Congénitas; DOGV: Valencia, Spain, 2018; Available online: https://dogv.gva.es/es/eli/es-vc/d/2018/11/30/218/ (accessed on 23 March 2023).
- Benito-Orejas, J.I.; Poncela-Blanco, M.; García-Vicario, F.; Benito-González, F.; Martín-Sigüenza, G.; San Román-Carbajo, J. ¿Es fácil encargarse de coordinar un programa de hipoacusia infantil? Rev. ORL 2016, 7, 77–90. [Google Scholar] [CrossRef][Green Version]
- Zeng, Q.; Luo, R.; Yan, S.; Tang, Y.; Wen, R.; Liu, W. Screening strategy and time points for newborn hearing re-screening with high risk factors. World J. Otorhinolaryngol. Head Neck Surg. 2022, 8, 257–261. [Google Scholar] [CrossRef]
- Sheng, H.; Zhou, Q.; Wang, Q.; Yu, Y.; Liu, L.; Liang, M.; Zhou, X.; Wu, H.; Tang, X.; Huang, Z. Comparison of Two-Step Transient Evoked Otoacoustic Emissions and One-Step Automated Auditory Brainstem Response for Universal Newborn Hearing Screening Programs in Remote Areas of China. Front. Pediatr. 2021, 9, 655625. [Google Scholar] [CrossRef]
- Jedrzejczak, W.W.; Gos, E.; Pilka, E.; Skarzynski, P.H.; Skarzynski, H.; Hatzopoulos, S. Pitfalls in the Detection of Hearing Loss via Otoacoustic Emissions. Appl. Sci. 2021, 11, 2184. [Google Scholar] [CrossRef]
- Deng, X.; Ema, S.; Mason, C.; Nash, A.D.; Carbone, E.P.; Gaffney, M. Receipt and Timeliness of Newborn Hearing Screening and Diagnostic Services Among Babies Born in 2017 in 9 States. J. Public Health Manag. Pract. 2022, 28, E100–E108. [Google Scholar] [CrossRef] [PubMed]
- Nikolopoulos, T.P. Neonatal hearing screening: What we have achieved and what needs to be improved. Int. J. Pediatr. Otorhinolaryngol. 2015, 79, 635–637. [Google Scholar] [CrossRef]
- Chapman, D.A.; Stampfel, C.C.; Bodurtha, J.N.; Dodson, K.M.; Pandya, A.; Lynch, K.B.; Kirby, R.S. Impact of co-occurring birth defects on the timing of newborn hearing screening and diagnosis. Am. J. Audiol. 2011, 20, 132–139. [Google Scholar] [CrossRef] [PubMed]
- Verstappen, G.; Foulon, I.; Houte, K.V.D.; Heuninck, E.; Van Overmeire, B.; Gordts, F.; Topsakal, V. Analysis of congenital hearing loss after neonatal hearing screening. Front. Pediatr. 2023, 11, 1153123. [Google Scholar] [CrossRef]
- Núñez-Batalla, F.; Jáudenes-Casaubón, C.; Sequí-Canet, J.M.; Vivanco-Allende, A.; Zubicaray-Ugarteche, J.; Lascarro, I.O. Programas de cribado de la hipoacusia congénita en 2020: Recomendaciones CODEPEH. Acta Otorrinolaringol. Esp. 2020, 72, 312–323. [Google Scholar] [CrossRef]
- Sequi-Canet, J.M.; Brines-Solanes, J. Keypoints to Successful Newborn Hearing Screening. Thirty Years of Experience and Innovations. Healthcare 2021, 9, 1436. [Google Scholar] [CrossRef]
- Luz, I.; Ribas, A.; Kozlowski, L.; Willig, M.; Berberian, A.P. Newborn Hearing Screening in a Public Maternity Ward in Curitiba, Brazil: Determining Factors for Not Retesting. Int. Arch. Otorhinolaryngol. 2016, 20, 300–304. [Google Scholar] [CrossRef] [PubMed]
- Davis, A.; Bamford, J.; Wilson, I.; Ramkalawan, T.; Forshaw, M.; Wright, S. A critical review of the role of neonatal hearing screening in the detection of congenital hearing impairment. Heal. Technol. Assess. 1997, 1, 1–176. [Google Scholar] [CrossRef]
- Generalitat Valenciana. Decreto 205/2018, de 16 de Noviembre, del Consell, por el que Aprueba el Mapa Sanitario de la Comunitat Valenciana; DOGV: Valencia, Spain, 2018; Available online: https://dogv.gva.es/portal/es/eli/es-vc/d/2018/11/16/205 (accessed on 24 March 2023).
- INEbase [Internet]. Madrid: Instituto Nacional de Estadística; 2022. Población extranjera por Nacionalidad, Comunidades, Sexo y Año [Comunidad Valenciana, Ambos Sexos, 2022]. Available online: https://www.ine.es/jaxi/Datos.htm?path=/t20/e245/p08/l0/&file=02005.px (accessed on 24 March 2023).
- Findlen, U.M.; Davenport, C.A.; Cadieux, J.; Gehred, A.; Holt, R.F.; Vaughn, L.M.; Houston, D.; Hunter, L.L. Barriers to and Facilitators of Early Hearing Detection and Intervention in the United States: A Systematic Review. Ear Hear. 2023, 44, 448–459. [Google Scholar] [CrossRef]
- Holzinger, D.; Weishaupt, A.; Fellinger, P.; Beitel, C.; Fellinger, J. Prevalence of 2.2 per mille of significant hearing loss at school age suggests rescreening after NHS. Int. J. Pediatr. Otorhinolaryngol. 2016, 87, 121–125. [Google Scholar] [CrossRef] [PubMed]
- Ngui, L.X.; Tang, I.P.; Prepageran, N.; Lai, Z.W. Comparison of distortion product otoacoustic emission (DPOAE) and automated auditory brainstem response (AABR) for neonatal hearing screening in a hospital with high delivery rate. Int. J. Pediatr. Otorhinolaryngol. 2019, 120, 184–188. [Google Scholar] [CrossRef] [PubMed]
- Johnson, J.L.; White, K.R.; Widen, J.E.; Gravel, J.S.; James, M.; Kennalley, T.; Maxon, A.B.; Spivak, L.; Sullivan-Mahoney, M.; Vohr, B.R.; et al. A Multicenter Evaluation of How Many Infants with Permanent Hearing Loss Pass a Two-Stage Otoacoustic Emissions/Automated Auditory Brainstem Response Newborn Hearing Screening Protocol. Pediatrics 2005, 116, 663–672. [Google Scholar] [CrossRef] [PubMed]
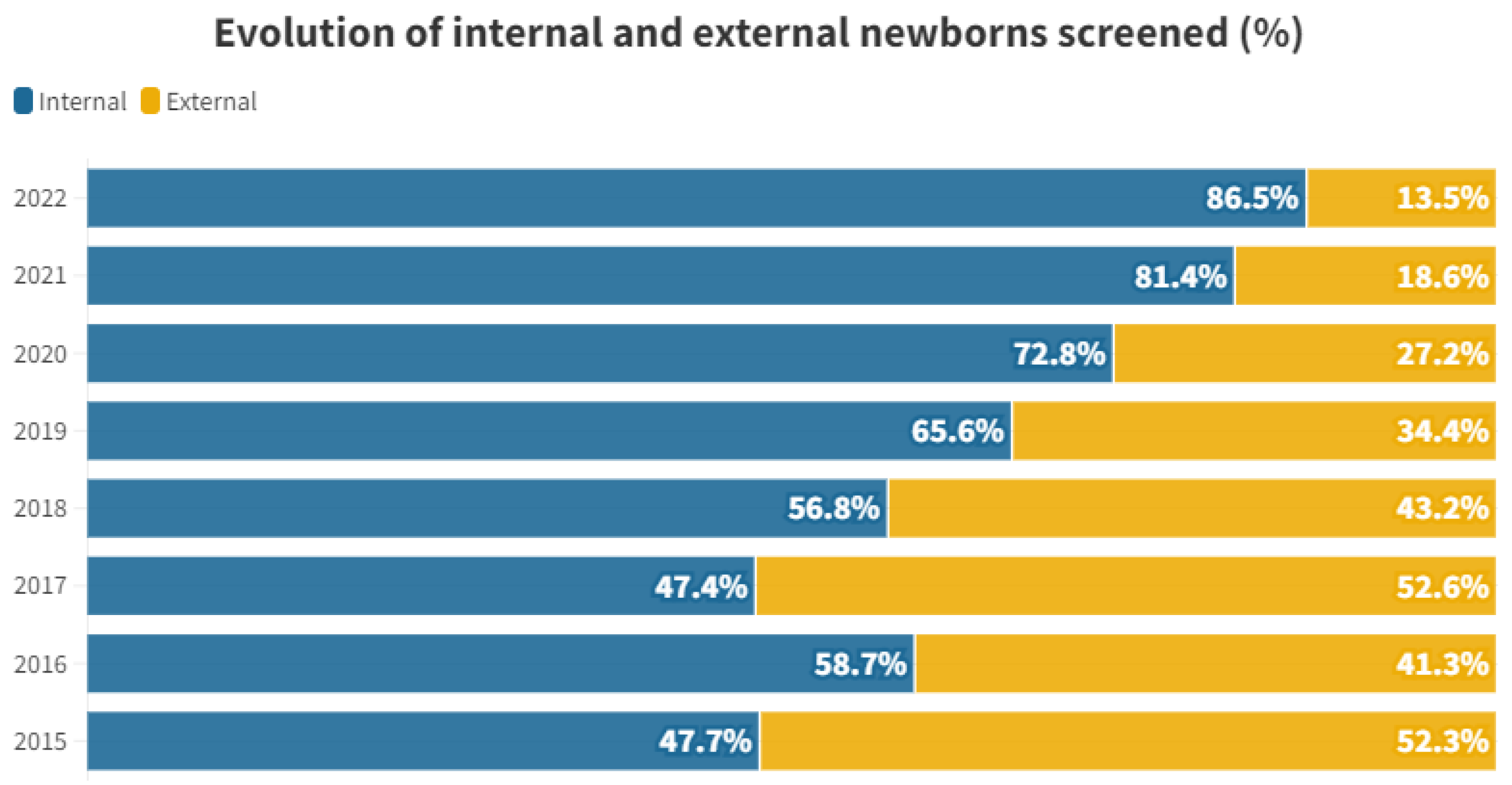
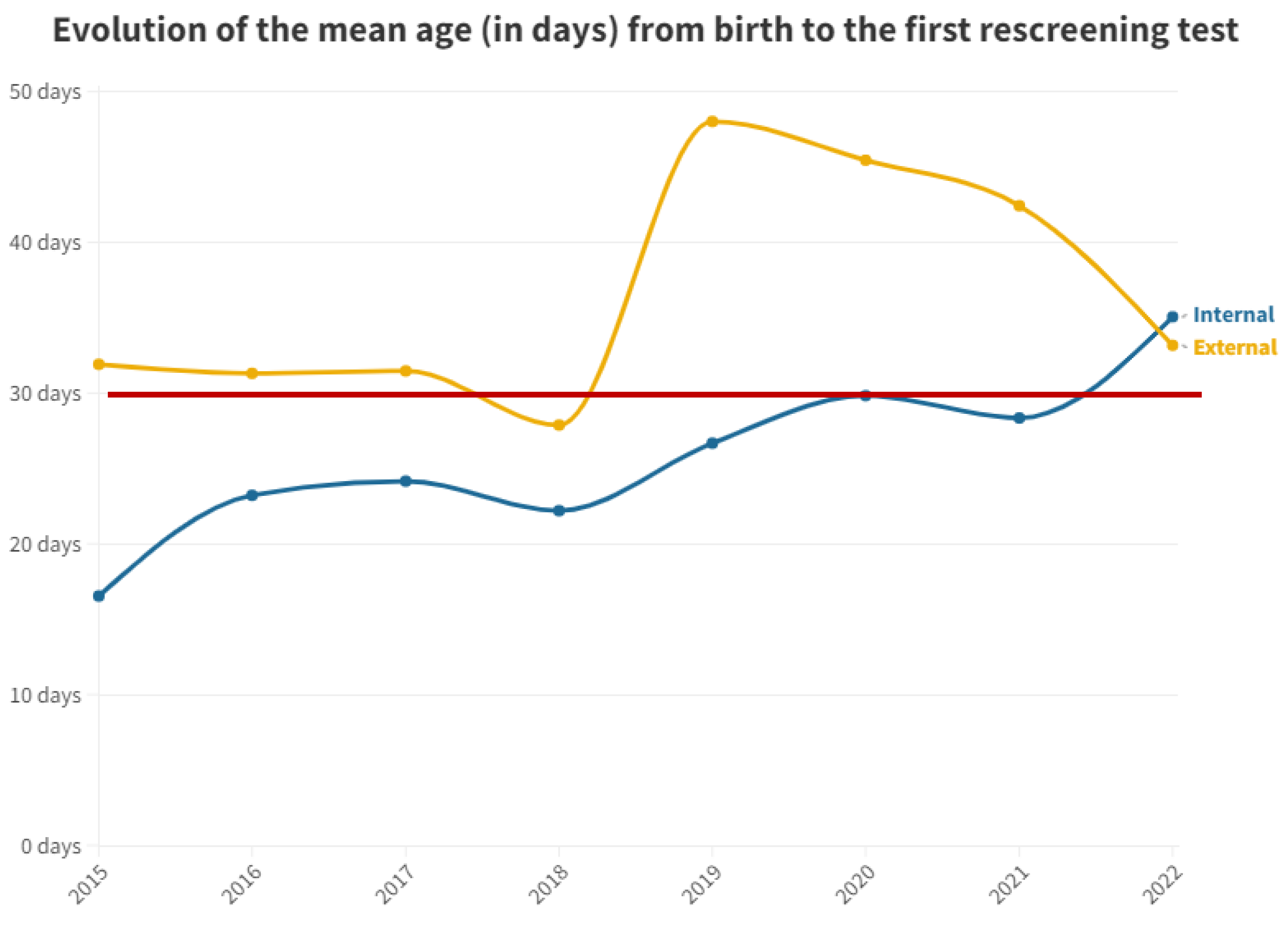
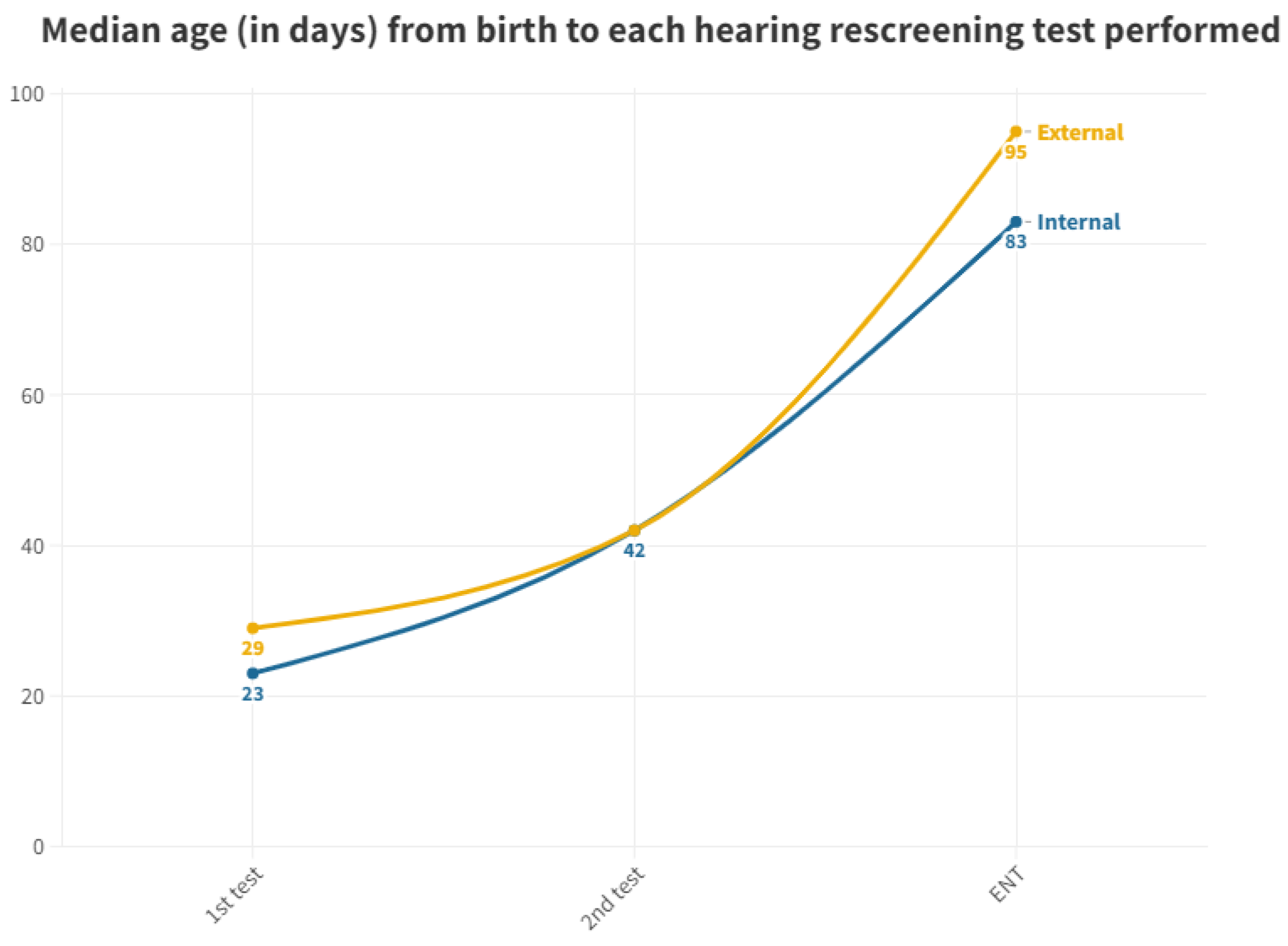

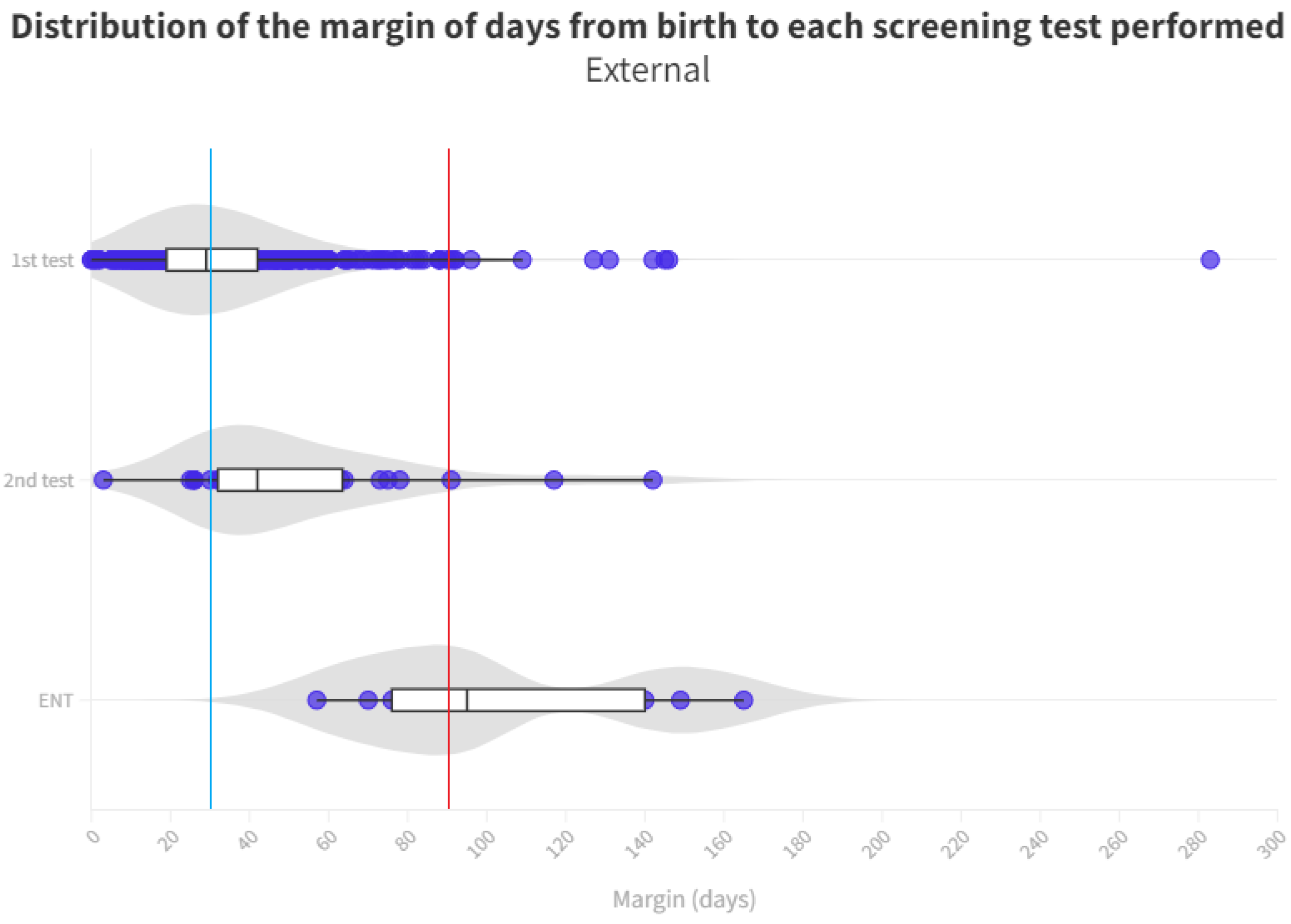
| 2015 | 2016 | 2017 | 2018 | 2019 | 2020 | 2021 | 2022 | Total | |
|---|---|---|---|---|---|---|---|---|---|
| Internal | 82 | 115 | 93 | 88 | 84 | 59 | 92 | 77 | 690 (61.07%) |
| External | 90 | 81 | 103 | 67 | 44 | 22 | 21 | 12 | 440 (38.93%) |
| Total | 172 | 196 | 196 | 155 | 128 | 81 | 113 | 89 | 1130 |
| Internal | External | |
|---|---|---|
| Maternity | 2.57 | |
| 1st test | 25.41 | 33.91 |
| 2nd test | 64.86 | 51.37 |
| ENT | 90.11 | 104.33 |
| Origin | Birth to 1st Outpatient Clinic Test | 1st to 2nd Outpatient Clinic Test | to ENT |
|---|---|---|---|
| Internal | 23 | 19 | 41 |
| External | 29 | 13 | 53 |
| Origin | Normal | Altered | ||
|---|---|---|---|---|
| Internal: n = 53 (85.48%) | 34 | 64.15% | 19 | 35.85% |
| External: n = 9 (14.52%) | 7 | 77.78% | 2 | 22.22% |
| Results | 41 | 66.13% | 21 | 33.87% |
| Results | Internal | |
| Normal | 661 | 97.21% |
| Altered | 19 | 2.79% |
| External | ||
| Normal | 435 | 99.54% |
| Altered | 2 | 0.46% |
| LFU | Cases | Losses | % |
|---|---|---|---|
| Internal | 690 | 10 | 1.45% |
| 1st test | 690 | 4 | 0.58% |
| 2nd test | 65 | 1 | 1.54% |
| ENT | 58 | 5 | 8.62% |
| External | 440 | 3 | 0.68% |
| 1st test | 440 | 2 | 0.45% |
| 2nd test | 29 | 1 | 3.45% |
| ENT | 9 | 0 | 0.00% |
| Total | 1130 | 13 | 1.15% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gómez-Delgado, M.; Sequi-Sabater, J.M.; Marco-Sabater, A.; Lora-Martin, A.; Aparisi-Climent, V.; Sequi-Canet, J.M. Neonatal Hearing Rescreening in a Second-Level Hospital: Problems and Solutions. Audiol. Res. 2023, 13, 655-669. https://doi.org/10.3390/audiolres13040058
Gómez-Delgado M, Sequi-Sabater JM, Marco-Sabater A, Lora-Martin A, Aparisi-Climent V, Sequi-Canet JM. Neonatal Hearing Rescreening in a Second-Level Hospital: Problems and Solutions. Audiology Research. 2023; 13(4):655-669. https://doi.org/10.3390/audiolres13040058
Chicago/Turabian StyleGómez-Delgado, Marta, Jose Miguel Sequi-Sabater, Ana Marco-Sabater, Alberto Lora-Martin, Victor Aparisi-Climent, and Jose Miguel Sequi-Canet. 2023. "Neonatal Hearing Rescreening in a Second-Level Hospital: Problems and Solutions" Audiology Research 13, no. 4: 655-669. https://doi.org/10.3390/audiolres13040058
APA StyleGómez-Delgado, M., Sequi-Sabater, J. M., Marco-Sabater, A., Lora-Martin, A., Aparisi-Climent, V., & Sequi-Canet, J. M. (2023). Neonatal Hearing Rescreening in a Second-Level Hospital: Problems and Solutions. Audiology Research, 13(4), 655-669. https://doi.org/10.3390/audiolres13040058






