1. Introduction
Biliary tract malignancies are rare but lethal diseases. Cholangiocarcinoma (CCA) represents the second most prevalent primary hepatic malignancy following hepatocellular carcinoma (HCC), accounting for approximately 15% of all primary liver neoplasms and 3% of all gastrointestinal cancers [
1]. Its incidence and associated mortality have increased globally over the recent decade. Additionally, the other aggressive cancer that can affect the bile duct is pancreatic ductal adenocarcinoma (PDAC), which mostly occurs in the head of the pancreas. Both PDAC and CCA are known for their poor prognosis [
2]. PDAC typically arises from ductal and acinar cells, often developing via precursor pancreatic lesions. In contrast, CCA originates from the epithelial lining of intrahepatic and extrahepatic bile ducts, frequently through biliary intraepithelial precursor lesions. Gallbladder cancers (GBCs) are most commonly adenocarcinoma but can also occur as squamous, adenosquamous, and small-cell subtypes, with no clear prognostic significance. Due to their anatomical site of origin, CCA is classified into intrahepatic (iCCA, 10–20%) and extrahepatic (eCCA) subtypes, with the latter further categorized into perihilar (pCCA, 60%) and distal (dCCA, 20–30%) variations [
3,
4,
5,
6]. Hilar cholangiocarcinoma (HC) is a malignant neoplasm of the proximal common bile duct or distal intrahepatic ducts involving biliary confluence. HC, specifically called Klatskin tumor, is considered an extrahepatic cholangiocarcinoma, as it originates from the extrahepatic bile ducts in the hepatoduodenal ligament and the gallbladder. Klatskin tumors account for 60–70% of all cholangiocarcinoma [
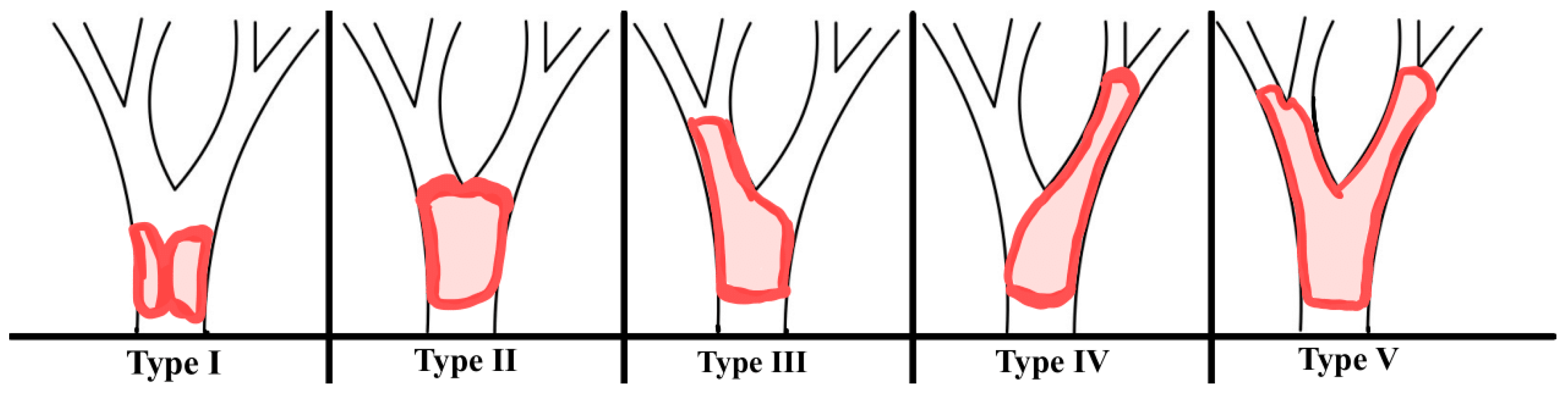
7]. This cancer develops gradually and implicitly, while symptoms appear mostly at the end stage of the disease. To evaluate the degree of ductal infiltration in perihilar cholangiocarcinoma, the 4-type Bismuth-Corlette classification system was developed (
Figure 1).
The classification is based on tumor location and the extent of bile duct infiltration, which has significant implications for surgical approaches in clinical practice. Symptoms of bilateral hilar tumors (including jaundice, pruritus, discolored stools, dark urine, and hemobilia) depend on the narrowing of the bile duct, the degree of infiltration into adjacent structures, and the location of the tumor. In the advanced stage of disease, nausea, vomiting, weight reduction, hepatomegaly, and ascites occur [
8]. Cholangiocarcinoma is considered one of the main causes of malignant biliary strictures, which may lead to serious obstructions. Unfortunately, the histopathological diagnosis of the HC is often acquired while the carcinoma is at an advanced, non-resectable stage, which limits possible medical intervention that could result in an improvement of the patients’ condition. Less than one-half of HCs are resectable. The main treatment options encompass surgery, radiation, chemotherapy, and photodynamic therapy. For PDAC localized in the head of the pancreas, surgical resections can be performed, such as a pancreaticoduodenectomy, commonly known as the Whipple procedure, or a pylorus-preserving pancreaticoduodenectomy (Traverso-Longmire procedure). However, only 25% of patients are classified for that surgical treatment. Even with radical treatment, chemotherapy is still obligatory in each case. In the management of GBC, treatment modalities beyond immunotherapy and radiotherapy encompass extensive hepatoduodenal ligament lymphadenectomy as well as resection of the gallbladder fossa, which typically involves partial hepatic resection. Recent studies show that jaundiced patients benefit from surgical treatment in this case [
9]. Endoscopic retrograde cholangiopancreatography (ERCP) and percutaneous transhepatic cholangiography (PTC) are essential techniques for assessing the extent of the tumor in the bile ducts. A method recommended for biliary drainage is the placement of an endoscopic or percutaneous stent with a self-expanding metallic stent (SEMS). Self-expanding metal stents (SEMSs) are mesh-like metallic stents that expand radially after deployment, providing continuous luminal patency by exerting outward force on the bile duct wall. Recent studies have shown that SEMSs tend to have more suitable outcomes than plastic stents, and SEMSs provide a longer patency and improved patient survival after the procedure [
10]. Thus, the insertion of plastic or metal (SEMS) biliary stents during ERCP and combining it with RFA of the tumor mass is one of the minimally invasive methods of treatment for biliary obstruction and palliative treatment of biliary hilar tumors [
11].
RFA is a minimally invasive technique used in the palliative treatment of bile duct cancer. This procedure is typically performed during routine endoscopic retrograde cholangiography (ERCP). RFA works by generating thermocoagulation cell necrosis through ionic excitation using electromagnetic wave frequencies ranging from 104 to 3 × 1012 Hz. Thermal ablation is achieved using an electrode that is inserted into the tumor site, which delivers high-frequency radio waves. This energy heats the diseased tissue, resulting in necrosis and the gradual separation of the tissue from the surrounding healthy area. The depth of necrosis typically extends radially for about 3 to 8 mm and is influenced by the amount of energy applied during treatment. Two types of probes can be used to establish an electrical circuit: a monopolar probe or two bipolar probes. The ions within the cancerous tissue follow the alternating current path, creating frictional heat that leads to protein denaturation and cell dehydration, resulting in cell death. This effect is most potent near the probe, limiting the coagulation of tissues farther away, which are primarily heated by thermal conduction, insufficient to cause necrosis. To achieve effective results, the required intermittent heat must reach a temperature between 60 and 80 °C, maintained for 1–2 min with an output of 7–10 W. Currently, two bipolar endoluminal RFA catheters are available on the market: the Habib™ EndoHPB (EMcision Ltd., London, UK) and the ELRA™ Electrode (STARmed Co., Ltd., Goyang-si, Republic of Korea). This study aims to investigate the impact of using endoluminal RFA via the endoscopic approach for hilar tumors, combined with the placement of bilateral self-expanding metal stents (SEMSs), Amsterdam stents, or double pig-tail stents (PGT), on the clinical prognosis of patients, as well as the safety and occurrence of complications related to the procedure.
Procedural workflow: ERCP was performed under standard cardiorespiratory monitoring with anesthesiology support as per institutional practice. Cholangiography was used to delineate the stricture and guide device positioning. When needed for device/stent passage, biliary cannulation and duct access were facilitated with standard accessories, and balloon dilation was performed at the endoscopist’s discretion. Periprocedural antibiotic prophylaxis and post-procedure observation followed local institutional protocols. The RFA catheter (StarMed ELRA) was positioned across the target stricture segment, and energy was delivered in 90 s applications at 7–10 W, with 1–4 applications per session depending on stricture length and anatomy. A stent (plastic or SEMS; unilateral or bilateral as appropriate) was then deployed to restore drainage.
Although malignant biliary obstruction arises from biologically diverse tumors, the proximate clinical problem is shared: intraductal tumor growth and/or tumor ingrowth/overgrowth leading to recurrent stent dysfunction and cholestasis. Endobiliary RFA is intended to induce local coagulative necrosis within the biliary stricture, potentially reducing intraductal tumor burden and delaying stent occlusion, regardless of the primary tumor type. The magnitude of any benefit is expected to vary with the pattern of obstruction (predominantly intraductal vs. extrinsic compression) and underlying disease trajectory [
12].
2. Materials and Methods
We retrospectively analyzed 24 consecutive patients with unresectable malignancies causing malignant biliary obstruction (MBO) who underwent endobiliary RFA with biliary stenting between May 2020 and May 2024 and were retrospectively identified from electronic medical records. No formal sample size calculation was performed; all eligible consecutive patients treated with RFA during the predefined study period were included. Data were extracted retrospectively from electronic medical records using a standardized data collection spreadsheet (demographics, diagnosis, procedural parameters, laboratory values, and follow-up status). Missing data were not imputed; variables are reported with the number of available observations. Separately, all endoscopic procedures (ERCP with biliary stenting, with or without adjunctive intraductal RFA) were performed as part of routine clinical care; patients provided procedure-specific informed consent according to institutional standards, including discussion of risks and alternatives such as stenting alone.
Patients had malignant biliary obstruction due to biologically diverse unresectable malignancies (including CCA, GBC, HCC with biliary involvement, and metastatic malignancies). Inclusion criteria for RFA treatment included patients with recurrent mechanical jaundice due to cancer progression despite a previous stent. High-power radiofrequency ablation of the narrowed biliary segments, followed by reimplantation of SEMS, Amsterdam stents, or double pigtail (DPT) into their lumen, was performed in 24 patients (19 women and 5 men). All procedures were performed by a dedicated endoscopy team under anesthesiologist-supervised sedation. The procedure was performed under intravenous sedation using 5 mg Midazolam, 100 µg Fentanyl, and 20 mg Hioscin Butyloromite. A PENTAX-ED-34 i10 T2 duodenoscope (HOYA Corporation, Tokyo, Japan) was used for this procedure. After removal of the obstructed stent, selective cannulation of the bile duct was carried out, and a cholangiogram was acquired visualizing the stricture. RFA was carried out with a power setting of 7–10 watts and a temperature of 75–80 °C (depending on the patient physique and the stricture localization: distal bile duct 10–14 watts; hilar region 7–10 watts). Also, depending on the stricture localization and therapy schedule, biliary stenting was performed:
- –
Patients who were qualified for a secondary RFA with the stricture in the distal bile duct received a plastic 10 Fr Amsterdam stent 7–9 cm in length.
- –
Patients who were qualified for a secondary RFA with the stricture in the hilar region received two plastic 7–8.5 Fr Amsterdam stents 12 cm in length.
- –
Patients receiving only one ablation treatment with the stricture in the distal bile duct received a single SEMS (covered or uncovered depending on tumor morphology and the presence of the gallbladder)
- –
Patients receiving only one ablation treatment with the stricture in the hilar region received double uncovered SEMS in the Y (stent-in-stent) or X (side-by-side) configuration. In difficult hepatic duct cannulations, the X configuration was preferred; otherwise the Y configuration was used, as it is considered beneficial during reinterventions.
The following stents were used for the procedure: uncovered SEMS (10 × 80 mm, 10 × 60 mm, 10 × 100 mm), Large Cell D-type stents (LCD) (10 × 60 mm), fully covered SEMS (8 × 80 mm), Amsterdam-type plastic stents (12 cm 10 Fr, 9 cm 10 Fr, 12 cm 8.5 Fr, 12 cm 7 Fr), and self-expandable BIL-0-10-60-RP stents.
Figure 2,
Figure 3 and
Figure 4 present the RFA catheter and stents’ configurations (
Figure 2,
Figure 3 and
Figure 4).
A retrospective study compared serum bilirubin concentration before treatment and 30 days, and 6 months after the procedure. Stent patency was assessed at 30 days and at 6 months. Procedure-related adverse events were recorded only if documented in the available procedural/hospital records; due to the retrospective design, adverse events were not captured prospectively and were not graded using a standardized system.
The primary outcome was 6-month stent patency/maintained biliary drainage, reported both among evaluable survivors and in an intention-to-treat (ITT) framework (counting deaths before 6 months as non-patent). Secondary outcomes included technical success of RFA, need for reintervention due to stent dysfunction, and change in serum bilirubin from baseline to day 30 (and to 6 months among survivors when available), reported as a continuous variable. The following variables were examined:
Categorical variables: gender and type of the stents;
Numerical variables: age, bilirubin level before the procedure, number of stents used, size of the stent, number of RFA series, and RFA power.
Continuous variables were summarized as median (IQR) when distributions were skewed (e.g., bilirubin) and as mean ± SD when approximately normal. Given the small sample size, no hypothesis testing or predictor analyses were performed; results are presented descriptively with available-case denominators.
No predictor analysis was pre-specified; due to the limited sample size, inferential testing for factors associated with 6-month outcome was not performed.
3. Results
24 patients (19 females and 5 males) were included in the study. The median age was 69 years (IQR 61–70), with a mean of 67.1 years. All the patients underwent an implantation of a biliary stent in the past due to cholestasis caused by malignancy. Among the investigated cases, 8 were diagnosed with metastatic adenocarcinoma, 6 with cholangiocarcinoma (CCC), 5 with adenocarcinoma of the gallbladder, 4 with hepatocellular carcinoma (HCC), and 1 with neuroendocrine tumor (NET).
Table 1 describes detailed patient characteristics.
The number of RFA series, the energy applied, and the number of stents used varied among patients, reflecting individualized treatment protocols, as summarized in
Table 2.
The cohort and interventions were intentionally heterogeneous (multiple malignancy types and stent strategies with individualized RFA settings), as detailed in
Supplementary Table S1, reflecting real-world practice rather than a standardized protocol.
6 of 24 patients (25%) did not complete the 6-month follow-up. In this group, during the 30-day follow-up, only 1 patient did not achieve a bilirubin biochemical improvement in cholestasis. The rest of the patients died due to the advanced stage of the malignancy.
In the group of patients who completed the 6-month follow-up, 16/18 (88.9%) maintained the patency of biliary stents and did not require reintervention by 6 months. The remaining two patients required endoscopic interventions due to biliary stent obstruction, which were not successful. Among evaluable survivors, 6-month patency was 16/18 (88.9%; 95% CI 67.2–96.9); in an intention-to-treat framework, 6-month patency was 16/24 (66.7%; 95% CI 46.7–82.0). Six-month overall survival was 18/24 (75.0%); six patients (25.0%) died before the 6-month assessment.
Serum bilirubin showed an early decrease at 30 days. In the full cohort (n = 24), median bilirubin decreased from 1.81 mg/dL (IQR 0.87–9.96) at baseline to 1.17 mg/dL (IQR 0.86–1.37) at 30 days, corresponding to a median change of −0.69 mg/dL (IQR −7.09 to 0.13). A reduction in bilirubin at 30 days was observed in 17/24 patients (70.8%). Six patients died before the 6-month assessment; among the 18 patients alive at 6 months, bilirubin remained low with a median of 0.99 mg/dL (IQR 0.89–1.08).
Using available laboratory follow-up, we report baseline, 30-day, and 6-month bilirubin (median, mg/dL) where available, together with early mortality (<6 months):
Metastatic adenocarcinoma (n = 8): deaths < 6 months 1/8; bilirubin median 0.90 → 1.06 → 0.90 (6-month available in 7 patients).
Cholangiocarcinoma (n = 6): deaths < 6 months 2/6; bilirubin median 5.85 → 1.32 → 0.99 (6-month available in 4 patients).
Hepatocellular carcinoma (n = 4): deaths < 6 months 0/4; bilirubin median 1.82 → 1.17 → 0.95 (6-month available in 4 patients).
Gallbladder adenocarcinoma (n = 5): deaths < 6 months 3/5; bilirubin median 2.51 → 1.35 → 6.89 (6-month available in 2 patients).
Metastatic neuroendocrine tumor (n = 1): deaths < 6 months 0/1; bilirubin 11.01 → 0.90 → 1.00.
Given the small number of patients evaluable at 6 months (
n = 18) and the resulting low statistical power, we did not perform formal hypothesis testing to identify predictors of 6-month patency/clinical success. All baseline and procedural variables are therefore presented descriptively. Cohort-level technical success and follow-up outcomes are summarized in
Table 3, whereas detailed individual patient-level procedural parameters and follow-up status are provided in
Supplementary Table S1.
Safety outcomes were retrospectively abstracted from available procedural and hospital records. Because adverse events were not collected prospectively and follow-up documentation was not standardized, minor events may be underreported. The adverse events documented in the available records are mentioned in
Supplementary Table S1.
Adverse events were not collected prospectively and were abstracted only if documented in available records, minor events may be underreported.
4. Discussion
This retrospective single-center case series describes our real-world experience with endoluminal RFA combined with biliary stenting in 24 patients with unresectable malignancies causing MBO. Given the heterogeneity of tumor types and stent strategies and the absence of a comparator arm, the findings should be interpreted descriptively. Among patients who were alive and available for 6-month assessment, stent patency was maintained in 16/18; when early deaths are considered, the corresponding intention-to-treat estimate is 16/24. These data suggest technical feasibility and maintained drainage in a subset of patients, but they do not allow causal inference regarding efficacy versus stenting alone.
Randomized trials and meta-analyses evaluating intraductal RFA have reported mixed results, with the strongest evidence coming from disease-specific cohorts, most commonly unresectable extrahepatic cholangiocarcinoma, and with relatively standardized stent strategies [
12,
13]. Therefore, our heterogeneous cohort is not directly comparable to trials such as Gao et al., Yang et al., and Kang et al. [
14,
15,
16]. Instead, we present our results as a descriptive real-world series across malignant biliary obstruction etiologies, which may complement the disease-focused literature by illustrating feasibility in a broader clinical population.
In contrast, Jarosova et al. reported no superiority of RFA plus stenting over stenting alone. Several methodological and clinical factors may contribute to discrepant findings across studies, including differences in patient selection (e.g., inclusion of specific tumor entities and obstruction patterns), baseline disease burden and prognosis, and the relative contribution of systemic therapy [
17]. In addition, RFA technique may vary between studies (catheter type, energy delivery settings, number of applications/sessions, and stent strategy), which could plausibly influence local tumor control and the risk of early dysfunction. Finally, endpoints and follow-up schemes differ (cross-sectional patency assessments versus time-to-event analyses and longer follow-up horizons), limiting direct comparability across trials and observational series. The most recent meta-analysis, including the above-mentioned RCTs, revealed that in patients with unresectable cholangiocarcinoma, the combination of RFA and stenting was associated with improved overall survival and stent patency; however, the statistical power and high heterogeneity of the study group do not allow for clear conclusions to be drawn [
18]. The authors point to the need for further research; hence, our attempt to describe our clinical experience with RFA and stenting.
Biliary drainage is a key element that improves overall survival for malignant biliary obstruction [
19]. Prognosis in nonresectable malignant biliary obstruction is generally poor, but drainage, along with palliative chemotherapy, can increase overall survival, improve quality of life, and relieve pain and pruritus [
20]. Based on the results of available studies, the median overall survival of patients treated with drainage alone, without RFA, remains within a few months (6–9 months). Some studies suggest that adding endobiliary RFA may be associated with longer survival compared with drainage alone; however, estimates vary across trials and meta-analyses and remain heterogeneous, up to 10–12 months. Primary bile duct tumors, such as CCA or ampullary carcinoma, are particularly valuable in terms of median survival, while secondary tumors (PDAC, gallbladder carcinoma, HCC, metastases) derive questionable benefit from the use of RFA [
21]. Complications can also negatively affect survival rates. In retrospective and cohort studies, e.g., among patients with non-operable malignant distal bile duct stricture after stenting, the 6-month survival rate was only 24% (median ≈ 81 days) [
22]. In Yang’s study, patients with CCA were randomly assigned to receive RFA with stenting in combination with a novel 5-fluorouracil compound, S-1, or RFA with biliary stenting alone. The results demonstrated that additional chemotherapy significantly improved overall survival (16 months and 11 months, respectively), and stent patency time was significantly longer (6.6 months and 5.6 months, respectively), and quality of life assessed by the Karnofsky index was also improved [
23].
The study population consisted mostly of females, with a mean age of 67.1 years. The heterogeneity of our study group remains high due to many types of non-resectable cancers and their location within the biliary tract, stent types (plastic, covered, self-expandable), and their number (1 or 2) and dimensions of each stent. Given the small sample size and low power, we did not attempt to identify predictors of 6-month patency/clinical success; potential associations should be explored in larger, more homogeneous cohorts. The main limitation is the small sample size, which does not allow for drawing objective conclusions. Stent type was selected pragmatically based on stricture location and the planned treatment strategy (including whether a repeat RFA session was anticipated), rather than randomized allocation. Therefore, our dataset is not suited to compare outcomes between plastic Amsterdam stents and SEMSs, and any between-stent inferences would be confounded by indication. In line with prior evidence suggesting longer patency with SEMS compared with plastic stents in malignant distal biliary strictures [
24], our findings should be interpreted descriptively. In our series, maintained biliary drainage at follow-up was observed in patients treated with either stent type; however, we did not perform a formal comparative analysis, and the apparent similarity may reflect patient selection and disease trajectory rather than stent performance.
An important procedural consideration in our cohort was the selection of biliary prostheses in relation to endoluminal RFA. Self-expanding metal stents (SEMSs) were primarily used in patients undergoing a single planned RFA session because their continuous radial force and larger effective lumen may help maintain ductal patency after ablation, particularly in the presence of post-procedural edema or residual tumor tissue. In contrast, Amsterdam-type plastic stents were mainly selected for patients scheduled for repeat RFA sessions, as their ease of removal and exchange facilitates reintervention despite shorter expected patency. In cases of malignant hilar obstruction, bilateral SEMSs were deployed using either a side-by-side or stent-in-stent configuration based on anatomical feasibility and technical considerations, reflecting a pragmatic, real-world approach to balancing immediate technical success with anticipated future interventions.
Also, it is important to recognize that 25% of patients did not survive 6 months of follow-up due to malignancy progression, which reveals the aggressive nature of the disease and the limited time window to undertake treatment. Nevertheless, the great majority of patients completed the follow-up period and maintained biliary tract patency, which suggests that RFA combined with biliary stenting is a feasible and minimally invasive treatment method. Unfortunately, 2 patients struggled with maintaining patency, requiring multiple interventions, which were not successful. This suggests that when the stent occlusion occurs, the outcomes of treatment may be less favorable, underlining the urge for early detection and the initial effective management.
A major limitation is the marked heterogeneity of both the underlying malignancies and the procedural strategies (stent types/configurations and individualized RFA settings). Consequently, our data do not support inference about which tumor entities, stent strategies, or RFA parameters are associated with better outcomes, nor do they define a single reproducible “RFA + stent” protocol. The findings should therefore be interpreted as descriptive and hypothesis-generating, and future studies should be disease-specific with standardized procedural protocols.
This study has a few more limitations. First, the sample size comprises 24 patients; however, the actual follow-up at 6 months was assessed in only 18 patients. That significantly limits the power of statistical analysis to detect significant associations between variables and treatment outcomes. Although single-institutional reports like ours are encouraging regarding the feasibility of RFA and biliary stenting, this calls for larger, multi-institutional, high-quality randomized controlled studies of RFA with stenting and sole stenting, given the rarity of biliary tract malignancies. Indistinct data in the literature support the need for further research on RFA and biliary stenting. Second, the retrospective design of the study introduces the risk of confounding in patient selection and prosthetic selection. Third, our study assessed only the bilirubin concentration as a success indicator. We did not assess patient-reported outcomes, such as quality of life, pain control, and relief of pruritus. This should be the subject of further research on the impact on quality of life after RFA with biliary stenting. Another crucial limitation is the high heterogeneity of the analyzed group, encompassing several distinct primary malignancies with differing biology, natural history, and treatment responsiveness, making final conclusions not be interpreted as disease-specific.
Importantly, adverse events were not collected prospectively and were not graded using a standardized adverse-event lexicon. Safety outcomes were retrospectively abstracted from available documentation; therefore, minor events and events managed outside our institution may be underreported. Consequently, no definitive safety conclusions can be drawn from this dataset. Because key timestamps (e.g., exact dates of stent dysfunction/occlusion and death/last follow-up) were not consistently documented in the retrospective records, time-to-event analyses (Kaplan–Meier), median survival, and time-to-occlusion estimates could not be reliably performed.
5. Conclusions
In this preliminary retrospective case series, endoluminal RFA combined with biliary stenting was feasible and was associated with maintained biliary drainage in a majority of patients who survived to the 6-month assessment. Given the lack of a comparator arm and the non-standardized retrospective capture of adverse events, these findings are hypothesis-generating and should be confirmed in prospective studies with systematic safety reporting.
Author Contributions
T.K.: Conceptualization, data curation, formal analysis, methodology, software, validation, writing—original draft, writing—review and editing. W.C.: Data curation, formal analysis, methodology, software, validation, writing—original draft, writing—review and editing. W.A.: Data curation, formal analysis, methodology, software, validation, visualization, writing—original draft, writing—review and editing. K.W.: Data curation, formal analysis, methodology, software, validation, writing—original draft, writing—review and editing. A.G.: Data curation, formal analysis, methodology, software, validation, writing—original draft, writing—review and editing. A.D.: Data curation, formal analysis, supervision, validation, writing—review and editing. J.S.: Data curation, formal analysis, supervision, validation, writing—review and editing. P.H.: Data curation, formal analysis, supervision, validation, writing—review and editing. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
This study was a retrospective analysis of anonymized clinical data and did not involve any direct patient intervention or identifiable personal information. According to institutional and national regulations, it does not meet the criteria of a medical experiment and, therefore, was exempt from ethics committee review and informed consent requirements. The study was conducted following the principles of the Declaration of Helsinki and relevant guidelines for research integrity.
Informed Consent Statement
All patients provided procedure-specific informed consent for ERCP and intraductal RFA as part of routine clinical care. The research involved only retrospective evaluation of anonymized clinical information, with no patient contact or use of identifiable personal data. According to institutional and national regulations, the study did not meet the criteria for human subject research requiring informed consent. Therefore, study-specific informed consent was not required. All data used in this study were anonymized prior to analysis.
Data Availability Statement
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request. The data are not publicly available due to institutional policy governing access to clinical data.
Acknowledgments
AI-based programs, such as Grammarly and ChatGPT 5.2, were implemented only to improve the readability of the manuscript and grammar. The authors have reviewed and edited the output and take full responsibility for the content of this publication.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Sarcognato, S.; Sacchi, D.; Fassan, M.; Fabris, L.; Cadamuro, M.; Zanus, G.; Cataldo, I.; Capelli, P.; Baciorri, F.; Cacciatore, M.; et al. Cholangiocarcinoma. Pathologica 2021, 113, 158–169. [Google Scholar] [CrossRef]
- Siegel, R.L.; Miller, K.D.; Fuchs, H.E.; Jemal, A. Cancer statistics, 2022. CA Cancer J. Clin. 2022, 72, 7–33. [Google Scholar] [CrossRef] [PubMed]
- Nagtegaal, I.D.; Odze, R.D.; Klimstra, D.; Paradis, V.; Rugge, M.; Schirmacher, P.; Washington, K.M.; Carneiro, F.; Cree, I.A. WHO Classification of Tumours Editorial Board The 2019 WHO classification of tumours of the digestive system. Histopathology 2020, 76, 182–188. [Google Scholar] [CrossRef]
- Banales, J.M.; Marin, J.J.G.; Lamarca, A.; Rodrigues, P.M.; Khan, S.A.; Roberts, L.R.; Cardinale, V.; Carpino, G.; Andersen, J.B.; Braconi, C.; et al. Cholangiocarcinoma 2020, The next horizon in mechanisms and management. Nat. Rev. Gastroenterol. Hepatol. 2020, 17, 557–588. [Google Scholar] [CrossRef] [PubMed]
- Ilyas, S.; Khan, S.A.; Hallemeier, C.L.; Kelley, R.K.; Gores, G.J. Cholangiocarcinoma—Evolving concepts and therapeutic strategies. Nat. Rev. Clin. Oncol. 2018, 15, 95–111. [Google Scholar] [CrossRef]
- Blechacz, B.; Komuta, M.; Roskams, T.; Gores, G.J. Clinical diagnosis and staging of cholangiocarcinoma. Nat. Rev. Gastroenterol. Hepatol. 2011, 8, 512–522. [Google Scholar] [CrossRef]
- Lee, I.C.; Stüben, B.O.; Fard-Aghaie, M.; Giannou, A.; Ghadban, T.; Heumann, A.; Li, J. Individualized surgical approach based on Bismuth-Corlette classification for perihilar cholangiocarcinoma. Clin. Surg. Oncol. 2024, 3, 100057. [Google Scholar] [CrossRef]
- Laimer, G.; Jaschke, N.; Gottardis, M.; Schullian, P.; Putzer, D.; Eberle, G.; Bale, R. Stereotactic radiofrequency ablation of an unresectable intrahepatic cholangiocarcinoma (ICC): Transforming an aggressive disease into a chronic condition. Cardiovasc. Interv. Radiol. 2020, 43, 791–796. [Google Scholar] [CrossRef]
- Weismüller, T.J. Role of Intraductal RFA: A Novel Tool in the Palliative Care of Perihilar Cholangiocarcinoma. Visc. Med. 2021, 37, 39–47. [Google Scholar] [CrossRef]
- Alvarez-Sánchez, M.V.; Napoléon, B. Review of endoscopic radiofrequency in biliopancreatic tumours with emphasis on clinical benefits, controversies, and safety. World J. Gastroenterol. 2016, 22, 8257–8270. [Google Scholar] [CrossRef]
- Jansen, C.; Vilmann, P.; Brink, L.; Olafsson, L.; Kovacevic, B. Endoluminal radiofrequency ablation of malignant biliary obstruction. Ugeskr. Læger 2023, 185, V11220678. [Google Scholar]
- Khizar, H.; Hu, Y.; Wu, Y.; Ali, K.; Iqbal, J.; Zulqarnain, M.; Yang, J. Efficacy and Safety of Radiofrequency Ablation Plus Stent Versus Stent-alone Treatments for Malignant Biliary Strictures: A Systematic Review and Meta-analysis. J. Clin. Gastroenterol. 2023, 57, 335–345. [Google Scholar] [CrossRef] [PubMed]
- Tarar, Z.I.; Farooq, U.; Gandhi, M.; Ghous, G.; Saleem, S.; Kamal, F.; Imam, Z.; Jamil, L. Effect of radiofrequency ablation in addition to biliary stent on overall survival and stent patency in malignant biliary obstruction: An updated systematic review and meta-analysis. Eur. J. Gastroenterol. Hepatol. 2023, 35, 646–653. [Google Scholar] [CrossRef] [PubMed]
- Gao, D.J.; Yang, J.F.; Ma, S.R.; Wu, J.; Wang, T.T.; Jin, H.B.; Xia, M.X.; Zhang, Y.C.; Shen, H.Z.; Ye, X.; et al. Endoscopic radiofrequency ablation plus plastic stent placement versus stent placement alone for unresectable extrahepatic biliary cancer: A multicenter randomized controlled trial. Gastrointest. Endosc. 2021, 94, 91–100.e2. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Wang, J.; Zhou, H.; Zhou, Y.; Wang, Y.; Jin, H.; Lou, Q.; Zhang, X. Efficacy and safety of endoscopic radiofrequency ablation for unresectable extrahepatic cholangiocarcinoma: A randomized trial. Endoscopy 2018, 50, 751–760. [Google Scholar] [CrossRef]
- Kang, H.; Han, S.Y.; Cho, J.H.; Kim, E.J.; Kim, D.U.; Yang, J.K.; Jeon, S.; Park, G.; Lee, T.H. Efficacy and safety of temperature-controlled intraductal radiofrequency ablation in advanced malignant hilar biliary obstruction: A pilot multicenter randomized comparative trial. J. Hepato-Biliary-Pancreat. Sci. 2022, 29, 469–478. [Google Scholar] [CrossRef]
- Jarosova, J.; Zarivnijova, L.; Cibulkova, I.; Mares, J.; Macinga, P.; Hujova, A.; Falt, P.; Urban, O.; Hajer, J.; Spicak, J.; et al. Endoluminal radiofrequency ablation in patients with malignant biliary obstruction: A randomised trial. Gut 2023, 72, 2286–2293. [Google Scholar] [CrossRef]
- Balducci, D.; Montori, M.; Martini, F.; Valvano, M.; De Blasio, F.; Argenziano, M.E.; Tarantino, G.; Benedetti, A.; Bendia, E.; Marzioni, M.; et al. The Impact of Radiofrequency Ablation on Survival Outcomes and Stent Patency in Patients with Unresectable Cholangiocarcinoma: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Cancers 2024, 16, 1372. [Google Scholar] [CrossRef]
- Jagtap, V.K.; Kumar, S.; Harris, C.; Lynser, D.; Raphael, V. Improved Survival With Chemotherapy in Patients With Malignant Biliary Tract Obstruction After Percutaneous Transhepatic Biliary Drainage (PTBD). Cureus 2024, 16, e63218. [Google Scholar] [CrossRef]
- Zhou, Z.; Li, J.; Liu, H.; Wu, D.; Xu, Y.; Xia, Y.; Lu, J.; Guo, C.; Zhou, Y. Quality of life and survival of patients with malignant bile duct obstruction following different ERCP based treatments. Int. J. Clin. Exp. Med. 2016, 9, 8821–8832. [Google Scholar]
- Xia, M.; Qin, W.; Hu, B. Endobiliary radiofrequency ablation for unresectable malignant biliary strictures: Survival benefit perspective. Dig. Endosc. 2023, 35, 584–591. [Google Scholar] [CrossRef] [PubMed]
- Kusumaningtyas, L.; Makmun, D.; Syam, A.F.; Setiati, S. Six-month Survival of Patients with Malignant Distal Biliary Stricture Following Endoscopic Biliary Stent Procedure and Its Associated Factors. Acta Medica Indones. 2020, 52, 31–38. [Google Scholar]
- Yang, J.; Wang, J.; Zhou, H.; Wang, Y.; Huang, H.; Jin, H.; Lou, Q.; Shah, R.J.; Zhang, X. Endoscopic radiofrequency ablation plus a novel oral 5-fluorouracil compound versus radiofrequency ablation alone for unresectable extrahepatic cholangiocarcinoma. Gastrointest. Endosc. 2020, 92, 1204–1212.e1. [Google Scholar] [CrossRef] [PubMed]
- Moole, H.; Jaeger, A.; Cashman, M.; Volmar, F.H.; Dhillon, S.; Bechtold, M.L.; Puli, S.R. Are self-expandable metal stents superior to plastic stents in palliating malignant distal biliary strictures? A meta-analysis and systematic review. Med. J. Armed Forces India 2017, 73, 42–48. [Google Scholar] [CrossRef]
| Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |