Non-Vertical Exposures to HIV, HBV and HCV Infection in Children and Adolescents—Risk of Infection, Standards of Care and Postexposure Prophylaxis
Abstract
1. Introduction
2. Materials and Methods
3. Results
3.1. Estimated Infection Risk after Needlestick Injuries
3.1.1. HIV
3.1.2. HBV
3.1.3. HCV
3.2. Estimated Infection Risk after Sexual Exposition
3.2.1. HIV
3.2.2. HBV
3.2.3. HCV
3.3. Standards of Care
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Centers for Disease Control and Prevention. HIV Surveillance Report. 2019; Volume 32. Available online: http://www.cdc.gov/hiv/library/reports/hiv-surveillance.html (accessed on 2 July 2021).
- Long, S.S.; Prober, C.G.; Fischer, M. Principles and Practise of Pediatric Infectious Diseases, 4th ed.; Elsevier: London, UK, 2012. [Google Scholar]
- Thompson, S.; Boughton, C.R.; Dore, G.J. Blood-borne viruses and their survival in the environment: Is public concern about community needlestick exposures justified? Aust. N. Z. J. Public Health 2003, 27, 602–607. [Google Scholar] [CrossRef] [PubMed]
- Nyiri, P.; Leung, T.; Zuckerman, M.A. Sharps discarded in inner city parks and playgrounds—Risk of bloodborne virus exposure. Commun. Dis. Public Health 2004, 7, 287–288. [Google Scholar] [PubMed]
- Russell, F.M.; Nash, M.C. A prospective study of children with community-acquired needlestick injuries in Melbourne. J. Paediatr. Child Health 2002, 38, 322–323. [Google Scholar] [CrossRef] [PubMed]
- Nourse, C.B.; Charles, C.A.; McKay, M.; Keenan, P.; Butler, K.M. Childhood needlestick injuries in the Dublin metropolitan area. Ir. Med. J. 1997, 90, 66–69. Available online: https://pubmed.ncbi.nlm.nih.gov/9105131 (accessed on 2 July 2021). [PubMed]
- Wyatt, J.P.; Robertson, C.E.; Scobie, W.G. Out of hospital needlestick injuries. Arch. Dis. Child. 1994, 70, 245–246. [Google Scholar] [CrossRef][Green Version]
- Butsashvili, M.; Kamkamidze, G.; Kajaia, M.; Kandelaki, G.; Zhorzholadze, N. Circumstances Surrounding the Community Needle-Stick Injuries in Georgia. J. Community Health 2011, 36, 1050–1052. [Google Scholar] [CrossRef]
- De Waal, N.; Rabie, H.; Bester, R.; Cotton, M.F. Mass Needle Stick Injury in Children from the Western Cape. J. Trop. Pediatr. 2005, 52, 192–196. [Google Scholar] [CrossRef][Green Version]
- Montella, F.; Di Sora, F.; Recchia, O. Can HIV-1 infection be transmitted by a “discarded” syringe? J. Acquir. Immune Defic. Syndr. 1992, 5, 1274–1275. [Google Scholar] [CrossRef]
- Moore, D.; Cps, C.P.S. Infectious Diseases and Immunization Committee Needle stick injuries in the community. Paediatr. Child Health 2008, 13, 205–210. [Google Scholar] [CrossRef]
- Aragón Peña, A.J.; Arrazola Martínez, M.P.; García de Codes, A.; Dávila Alvarez, F.M.; de Juanes Pardo, J.R. Hepatitis B prevention and risk of HIV infection in children injured by discarded needles and/or syringes. Aten Primaria 1996, 17, 138–140. [Google Scholar]
- García-Algar, O.; Vall, O. Hepatitis B virus infection from a needle stick. Pediatr. Infect. Dis. J. 1997, 16, 1099. [Google Scholar] [CrossRef] [PubMed]
- Libois, A.; Fumero, E.; Castro, P.; Nomdedeu, M.; Cruceta, A.; Gatell, J.M.; Garcia, F. Transmission of Hepatitis C Virus by Discarded-Needle Injury. Clin. Infect. Dis. 2005, 41, 129–130. [Google Scholar] [CrossRef] [PubMed]
- American Nurses Association. Needlestick Prevention Guide. Available online: https://www.who.int/occupational_health/activities/2needguid.pdf (accessed on 22 January 2021).
- Post-Exposure Prophylaxis (PEP) to Prevent HIV Infection; NYSDOH AI John Hopkins University: Baltimore, MD, USA, 2020.
- Zamora, A.B.; Rivera, M.O.; García-Algar, Ó.; Buqueras, J.C.; Combelles, O.V.; García-Sáiz, A. Detection of infectious human immunodeficiency type 1 virus in discarded syringes of intravenous drug users. Pediatr. Infect. Dis. J. 1998, 17, 655–657. [Google Scholar] [CrossRef]
- Papenburg, J.; Blais, D.; Moore, D.; Al-Hosni, M.; Laferrière, C.; Tapiero, B.; Quach, C. Pediatric Injuries From Needles Discarded in the Community: Epidemiology and Risk of Seroconversion. Pediatrics 2008, 122, e487–e492. [Google Scholar] [CrossRef]
- Bond, W.W.; Favero, M.S.; Petersen, N.J.; Gravelle, C.R.; Ebert, J.W.; Maynard, J.E. Survival of hepatitis b virus after drying and storage for one week. Lancet 1981, 317, 550–551. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention. CDC Yellow Book 2020: Health Information for International Travel; Oxford University Press: New York, NY, USA, 2017. Available online: https://wwwnc.cdc.gov/travel/yellowbook/2020/travel-related-infectious-diseases/hepatitis-b (accessed on 10 October 2021).
- Davis, L.G.; Weber, D.J.; Lemon, S.M. Horizontal transmission of hepatitis B virus. Lancet 1989, 1, 889–893. [Google Scholar] [CrossRef]
- Krawczyk, P.; Białkowska, J.; Dworniak, D.; Kamerys, J.; Szosland, D.; Jabłkowski, M. Czy ryzyko narażenia zawodowego na zakażenie wirusami HBV, HCV, HIV dotyczy tylko pracowników ochrony zdrowia? Med. Pr. 2010, 61, 15–23. [Google Scholar] [PubMed]
- Bruce, M.G.; Bruden, D.; Hurlburt, D.; Zanis, C.; Thompson, G.; Rea, L.; Toomey, M.; Townshend-Bulson, L.; Rudolph, K.; Bulkow, L.; et al. Antibody Levels and Protection After Hepatitis B Vaccine: Results of a 30-Year Follow-up Study and Response to a Booster Dose. J. Infect. Dis. 2016, 214, 16–22. [Google Scholar] [CrossRef]
- Middleman, A.B.; Baker, C.J.; Kozinetz, C.A.; Kamili, S.; Nguyen, C.; Hu, D.J.; Spradling, P.R. Duration of Protection After Infant Hepatitis B Vaccination Series. Pediatrics 2014, 133, e1500–e1507. [Google Scholar] [CrossRef]
- Webster, D.P.; Klenerman, P.; Dusheiko, G.M. Hepatitis C. Lancet 2015, 385, 1124–1135. [Google Scholar] [CrossRef]
- Paintsil, E.; He, H.; Peters, C.; Lindenbach, B.D.; Heimer, R. Survival of Hepatitis C Virus in Syringes: Implication for Transmission among Injection Drug Users. J. Infect. Dis. 2010, 202, 984–990. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention, U.S. Department of Health and Human Services. Updated Guidelines for Antiretroviral Postexposure Prophylaxis After Sexual, Injection Drug Use, or Other Non-Occupational Exposure To HIV—United States. 2016. Available online: https://www.cdc.gov/hiv/pdf/programresources/cdc-hiv-npep-guidelines.pdf (accessed on 15 April 2021).
- Benn, P.; Sultan, B.; Waters, L. Current perspectives in HIV post-exposure prophylaxis. HIV/AIDS Res. Palliat. Care 2014, 6, 147–158. [Google Scholar] [CrossRef]
- Mastro, T.D.; de Vincenzi, I. Probabilities of sexual HIV-1 transmission. AIDS 1996, 10 (Suppl. A), S75–S82. [Google Scholar] [CrossRef] [PubMed]
- Alter, M.J.; Margolis, H.S. The emergence of hepatitis B as a sexually transmitted disease. Med. Clin. N. Am. 1990, 74, 1529–1541. [Google Scholar] [CrossRef]
- Association of hepatitis B virus infection with other sexually transmitted infections in homosexual men. Omega Study Group. Am. J. Public Health 2000, 90, 1570–1574. [Google Scholar] [CrossRef]
- Inoue, T.; Tanaka, Y. Hepatitis B virus and its sexually transmitted infection—An update. Microb. Cell 2016, 3, 419–436. [Google Scholar] [CrossRef] [PubMed]
- Terrault, N.A. Sexual activity as a risk factor for hepatitis C. Hepatology 2002, 36 (Suppl. 1), S99–S105. [Google Scholar] [CrossRef] [PubMed]
- Leruez-Ville, M.; Kunstmann, J.-M.; De Almeida, M.; Rouzioux, C.; Chaix, M.-L. Detection of hepatitis C virus in the semen of infected men. Lancet 2000, 356, 42–43. [Google Scholar] [CrossRef]
- Terrault, N.A.; Dodge, J.L.; Murphy, E.L.; Tavis, J.E.; Kiss, A.; Levin, T.R.; Gish, R.; Busch, M.P.; Reingold, A.L.; Alter, M.J. Sexual transmission of hepatitis C virus among monogamous heterosexual couples: The HCV partners study. Hepatology 2012, 57, 881–889. [Google Scholar] [CrossRef]
- Polish AIDS Society. Zasady Opieki nad Osobami Zakażonymi HIV. Zalecenia Polskiego Towarzystwa Naukowego AIDS 2021; Polskie Towarzystwo Naukowe AIDS: Warszawa, Polands, 2021; pp. 43–52. [Google Scholar]
- Almeda, J.; Barbarà, J.C.; Simon, B.; Gérard, M.; Rey, D.; Puro, V.; Thomas, T.; on Behalf of the Euro-NONOPEP Project Group. Proposed recommendations for the management of HIV post-exposure prophylaxis after sexual, injecting drug or other exposures in Europe. Eurosurveillance 2004, 9, 5–6. [Google Scholar] [CrossRef]
- Ford, N.; Mayer, K.H.; Barlow, L.; Bagyinszky, F.; Calmy, A.; Chakroun, M.; Casas, E.; Dominguez, K.; Kaplan, J.; Green, K.; et al. World Health Organization Guidelines on Postexposure Prophylaxis for HIV: Recommendations for a Public Health Approach. Clin. Infect. Dis. 2015, 60, S161–S164. [Google Scholar] [CrossRef] [PubMed]
- Daar, E.S.; Pilcher, C.D.; Hecht, F.M. Clinical presentation and diagnosis of primary HIV-1 infection. Curr. Opin. HIV AIDS 2008, 3, 10–15. [Google Scholar] [CrossRef] [PubMed]
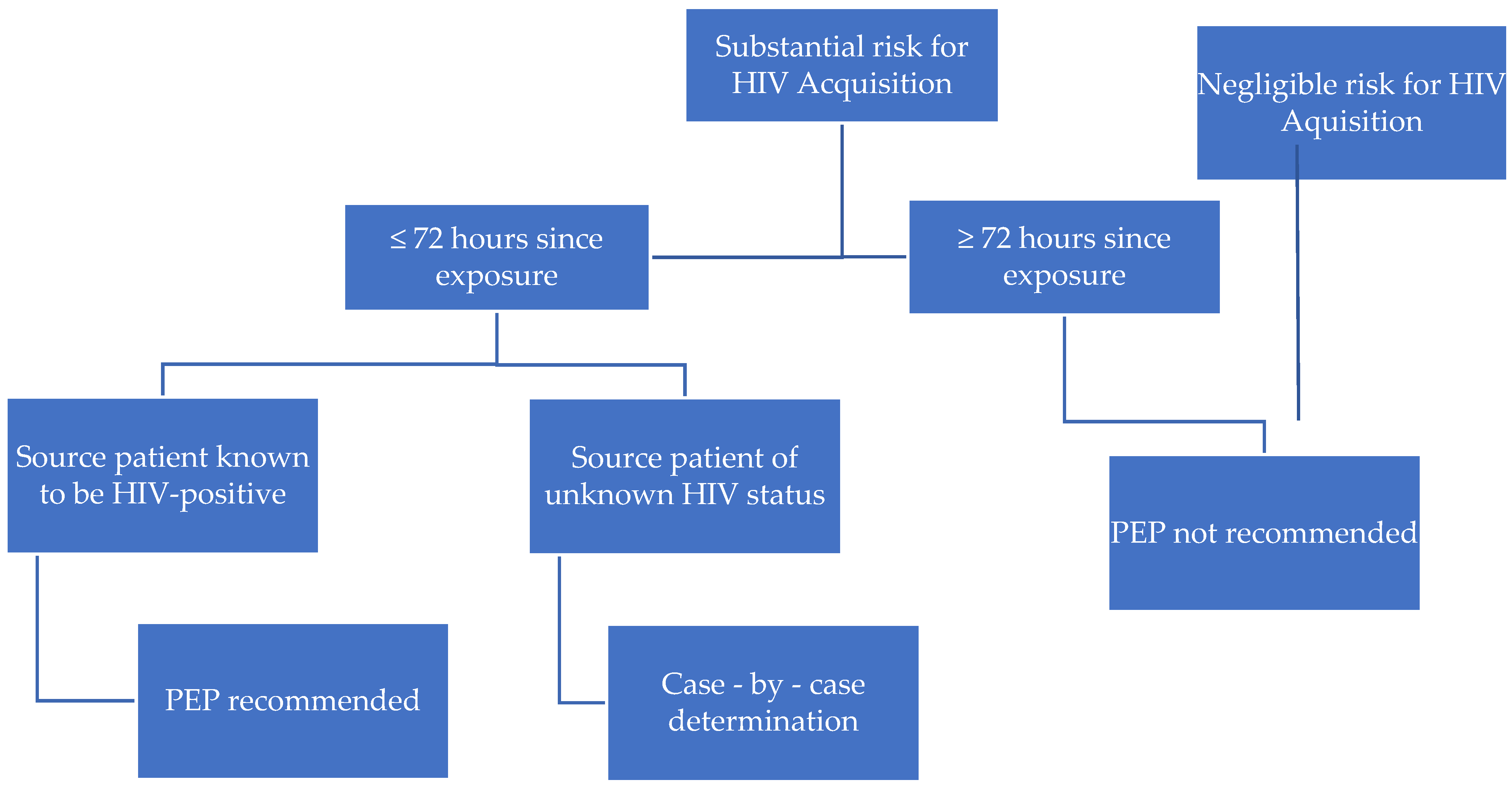
| High Risk of HIV Acquisition | Negligible Risk of HIV Acquisition |
|---|---|
|
|
| Higher Risk of HIV Acquisition | Lower Risk of HIV Acquisition |
|---|---|
|
|
| Exposure | Tests |
|---|---|
| HIV | HIV Ab/Ag (IV generation test) |
| HBV | HBs Ag HBc Ab HBs Ab (in previously vaccinated patients) |
| HCV | HCV Ab |
| Sexual exposure (additional tests) | Syphilis serology (e.x. VDRL) Pregnancy test |
| Exposure | Initial Visit | 2 Weeks | 4 Weeks | 8 Weeks | 24 Weeks |
|---|---|---|---|---|---|
| All non-vertical exposures | HIV Ab/Ag HBs Ag HBc Ab HBs Ab HCV Ab CBC ALT AST Creatinine | CBC ALT AST Creatinine | HIV Ab/Ag HCV-RNA ALT | HIV Ab/Ag | HBs Ag b HBc Ab b HCV Ab c ALT c |
| Sexual exposure (additional tests) | Syphilis serology (e.x. VDRL) Pregnancy test | Syphilis serology (e.x. VDRL) |
| Exposure | Initial Visit | 4–6 Weeks | 12 Weeks | 24 Weeks |
|---|---|---|---|---|
| All non-vertical exposures | HIV Ab/Ag HBs Ag HBc Ab HBs Ab HCV Ab ALT AST Creatinine | HIV Ab/Ag ALT AST Creatinine | HIV Ab/Ag | HIV Ab/Ag a HBs Ag b HBc Ab b HBs Ab b HCV Ab c |
| Sexual exposure (additional tests) | Syphilis serology (e.x. VDRL) Pregnancy test | Syphilis serology (e.x. VDRL) Pregnancy test |
| Children under 12 Years Old | Children over 12 Years Old |
|---|---|
(maximum 2 × 300 mg)
(maximum 2 × 150 mg)
Lopinavir: 10 mg/kg twice a day Ritonavir: 2.5 mg/kg twice a day (maximum dose 2 × 400/100 mg) |
|
| Children Aged 2–12 Years Old | Adolescents Aged 13 Years Old and Older |
|---|---|
Prefered:
Lopinavir/ritonavir With drugs dosed to age and weight |
Preferred:
Dolutegravir 50 mg once daily Alternative:
|
| Substantial Risk for HIV Acquisition | Negligible Risk for HIV Acquisition |
|---|---|
|
|
| Clinical Signs and Symptoms of Primary HIV Infection |
|---|
|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tomasik, A.; Pokorska-Śpiewak, M.; Marczyńska, M. Non-Vertical Exposures to HIV, HBV and HCV Infection in Children and Adolescents—Risk of Infection, Standards of Care and Postexposure Prophylaxis. Pediatr. Rep. 2021, 13, 566-575. https://doi.org/10.3390/pediatric13040067
Tomasik A, Pokorska-Śpiewak M, Marczyńska M. Non-Vertical Exposures to HIV, HBV and HCV Infection in Children and Adolescents—Risk of Infection, Standards of Care and Postexposure Prophylaxis. Pediatric Reports. 2021; 13(4):566-575. https://doi.org/10.3390/pediatric13040067
Chicago/Turabian StyleTomasik, Anna, Maria Pokorska-Śpiewak, and Magdalena Marczyńska. 2021. "Non-Vertical Exposures to HIV, HBV and HCV Infection in Children and Adolescents—Risk of Infection, Standards of Care and Postexposure Prophylaxis" Pediatric Reports 13, no. 4: 566-575. https://doi.org/10.3390/pediatric13040067
APA StyleTomasik, A., Pokorska-Śpiewak, M., & Marczyńska, M. (2021). Non-Vertical Exposures to HIV, HBV and HCV Infection in Children and Adolescents—Risk of Infection, Standards of Care and Postexposure Prophylaxis. Pediatric Reports, 13(4), 566-575. https://doi.org/10.3390/pediatric13040067








