Italian Case Report with a Double Mutation in PSEN1 (K311R and E318G)
Abstract
:1. Introduction
2. Materials and Methods
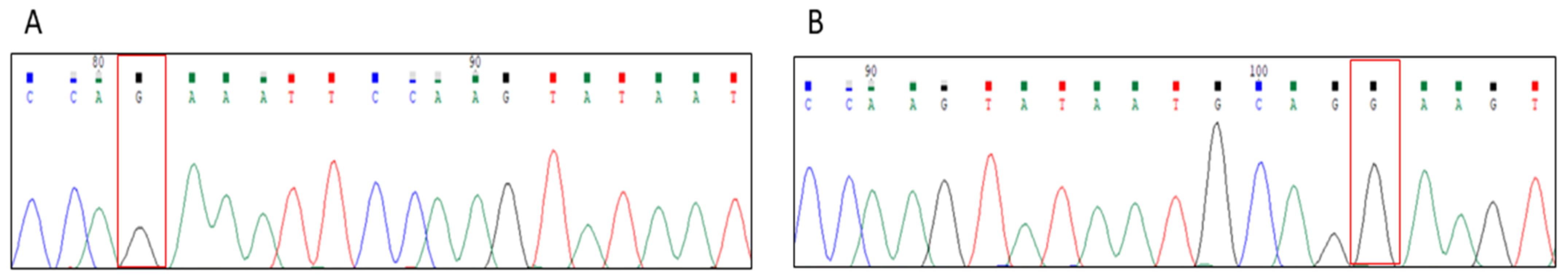
3. Case Presentation
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jack, C.R., Jr.; Albert, M.S.; Knopman, D.S.; McKhann, G.M.; Sperling, R.A.; Carrillo, M.C.; Thies, B.; Phelps, C.H. Introduction to the recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimer’s Dement. 2011, 7, 257–262. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alzheimer’s Association. Alzheimer’s disease facts and figures. Alzheimer’s Dement. 2016, 12, 459–509. [Google Scholar] [CrossRef]
- Goedert, M.; Wischik, C.M.; Crowther, R.A.; Walker, J.E.; Klug, A. Cloning and sequencing of the cDNA encoding a core protein of the paired helical filament of Alzheimer disease: Identification as the microtubule-associated protein tau. Proc. Natl. Acad. Sci. USA 1988, 85, 4051–4055. [Google Scholar] [CrossRef] [Green Version]
- Duyckaerts, C.; Delatour, B.; Potier, M.C. Classification and basic pathology of Alzheimer disease. Acta Neuropathol. 2009, 118, 5–36. [Google Scholar] [CrossRef]
- Anderson, N.D. State of the science on mild cognitive impairment (MCI). CNS Spectr. 2019, 24, 78–87. [Google Scholar] [CrossRef] [Green Version]
- Xiao, X.; Liu, H.; Liu, X.; Zhang, W.; Zhang, S.; Jiao, B. APP, PSEN1, and PSEN2 Variants in Alzheimer’s Disease: Systematic Re-evaluation According to ACMG Guidelines. Front. Aging Neurosci. 2021, 13, 695808. [Google Scholar] [CrossRef] [PubMed]
- Giau, V.V.; Bagyinszky, E.; An, S.S.; Kim, S.Y. Role of apolipoprotein E in neurodegenerative diseases. Neuropsychiatr. Dis. Treat. 2015, 11, 1723–1737. [Google Scholar] [CrossRef] [Green Version]
- Corder, E.H.; Saunders, A.M.; Risch, N.J.; Strittmatter, W.J.; Schmechel, D.E.; Gaskell, P.C., Jr.; Rimmler, J.B.; Locke, P.A.; Conneally, P.M.; Schmader, K.E. Protective effect of apolipoprotein E type 2 allele for late onset Alzheimer disease. Nat. Genet. 1994, 7, 180–184. [Google Scholar] [CrossRef]
- Farrer, L.A.; Cupples, L.A.; Haines, J.L.; Hyman, B.; Kukull, W.A.; Mayeux, R.; Myers, R.H.; Pericak-Vance, M.A.; Risch, N.; van Duijn, C.M. Effects of age, sex, and ethnicity on the association between apolipoprotein E genotype and Alzheimer disease. A meta-analysis. APOE and Alzheimer Disease Meta Analysis Consortium. JAMA 1997, 278, 1349–1356. [Google Scholar]
- Hebert, L.E.; Bienias, J.L.; Aggarwal, N.T.; Wilson, R.S.; Bennett, D.A.; Shah, R.C.; Evans, D.A. Change in risk of Alzheimer disease over time. Neurology 2010, 75, 786–791. [Google Scholar] [CrossRef]
- Hebert, L.E.; Weuve, J.; Scherr, P.A.; Evans, D.A. Alzheimer disease in the United States (2010–2050) estimated using the 2010 census. Neurology 2013, 80, 1778–1783. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Saunders, A.M.; Strittmatter, W.J.; Schmechel, D.; George-Hyslop, P.H.; Pericak-Vance, M.A.; Joo, S.H.; Rosi, B.L.; Gusella, J.F.; Crapper-MacLachlan, D.R.; Alberts, M.J. Association of apolipoprotein E allele epsilon 4 with late-onset familial and sporadic Alzheimer’s disease. Neurology 1993, 43, 1467–1472. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Green, R.C.; Cupples, L.A.; Go, R.; Benke, K.S.; Edeki, T.; Griffith, P.A.; Williams, M.; Hipps, Y.; Graff-Radford, N.; Bachman, D.; et al. Risk of dementia among white and African American relatives of patients with Alzheimer disease. JAMA 2002, 287, 329–336. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Budson, A.E.; Solomon, P.R. New criteria for Alzheimer disease and mild cognitive impairment: Implications for the practicing clinician. Neurologist 2012, 18, 356–363. [Google Scholar] [CrossRef] [PubMed]
- Miller, S.A.; Dykes, D.D.; Polesky, H.F. A simple salting out procedure for extracting DNA from human nucleated cells. Nucleic Acids Res. 1998, 16, 1215. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dong, J.; Qin, W.; Wei, C.; Tang, Y.; Wang, Q.; Jia, J. A Novel PSEN1 K311R Mutation Discovered in Chinese Families with Late-Onset Alzheimer’s Disease Affects Amyloid-β Production and Tau Phosphorylation. J. Alzheimer’s Dis. 2017, 57, 613–623. [Google Scholar] [CrossRef]
- Abdala, B.B.; dos Santos, J.M.; Gonçalves, A.P.; da Motta, L.B.; Laks, J.; de Borges, M.B.; Gonçalves Pimentel, M.M.; Santos-Rebouças, C.B. Influence of low frequency PSEN1 variants on familial Alzheimer’s disease risk in Brazil. Neurosci. Lett. 2017, 653, 341–345. [Google Scholar] [CrossRef]
- Perrone, F.; Bjerke, M.; Hens, E.; Sieben, A.; Timmers, M.; de Roeck, A.; Vandenberghe, R.; Sleegers, K.; Martin, J.J.; de Deyn, P.P.; et al. Amyloid-β1-43 cerebrospinal fluid levels and the interpretation of APP, PSEN1 and PSEN2 mutations. Alzheimer′s Res. Ther. 2020, 12, 108. [Google Scholar] [CrossRef]
- Artuso, V.; Benussi, L.; Ghidoni, R.; Moradi-Bachiller, S.; Fusco, F.; Curtolo, S.; Roiter, I.; Forloni, G.; Albani, D. Asymptomatic Carriers of Presenilin-1 E318G Variant Show no Cerebrospinal Fluid Biochemical Signs Suggestive of Alzheimer’s disease in a Family with Late-onset Dementia. Curr. Alzheimer Res. 2019, 16, 1–7. [Google Scholar] [CrossRef]
- Hippen, A.A.; Ebbert, M.T.; Norton, M.C.; Tschanz, J.T.; Munger, R.G.; Corcoran, C.D.; Kauwe, J.S. Presenilin E318G variant and Alzheimer’s disease risk: The Cache County study. BMC Genom. 2016, 17, 438. [Google Scholar] [CrossRef] [Green Version]
- Liu, C.-C.; Kanekiyo, T.; Xu, H.; Bu, G. Apolipoprotein E and Alzheimer disease: Risk, mechanisms and therapy. Nat. Rev. Neurol. 2013, 9, 106–118. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Houlden, H.; Crook, R.; Backhovens, H.; Prihar, G.; Baker, M.; Hutton, M.; Rossor, M.; Martin, J.J.; van Broeckhoven, C.; Hardy, J. ApoE genotype is a risk factor in nonpresenilin early-onset Alzheimer’s disease families. Am. J. Med. Genet. 1998, 81, 117–121. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bisceglia, P.; Lo Vecchio, F.; Latino, R.R.; Gravina, C.; Urbano, M.; la Torre, A.; Desina, G.; Greco, A.; Leone, M.; Antonioni, A. Italian Case Report with a Double Mutation in PSEN1 (K311R and E318G). Neurol. Int. 2022, 14, 417-422. https://doi.org/10.3390/neurolint14020034
Bisceglia P, Lo Vecchio F, Latino RR, Gravina C, Urbano M, la Torre A, Desina G, Greco A, Leone M, Antonioni A. Italian Case Report with a Double Mutation in PSEN1 (K311R and E318G). Neurology International. 2022; 14(2):417-422. https://doi.org/10.3390/neurolint14020034
Chicago/Turabian StyleBisceglia, Paola, Filomena Lo Vecchio, Raffaela Rita Latino, Carolina Gravina, Maria Urbano, Annamaria la Torre, Gaetano Desina, Antonio Greco, Maurizio Leone, and Annibale Antonioni. 2022. "Italian Case Report with a Double Mutation in PSEN1 (K311R and E318G)" Neurology International 14, no. 2: 417-422. https://doi.org/10.3390/neurolint14020034
APA StyleBisceglia, P., Lo Vecchio, F., Latino, R. R., Gravina, C., Urbano, M., la Torre, A., Desina, G., Greco, A., Leone, M., & Antonioni, A. (2022). Italian Case Report with a Double Mutation in PSEN1 (K311R and E318G). Neurology International, 14(2), 417-422. https://doi.org/10.3390/neurolint14020034







