Optimized LiFePO4-Based Cathode Production for Lithium-Ion Batteries through Laser- and Convection-Based Hybrid Drying Process
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials and Mixing
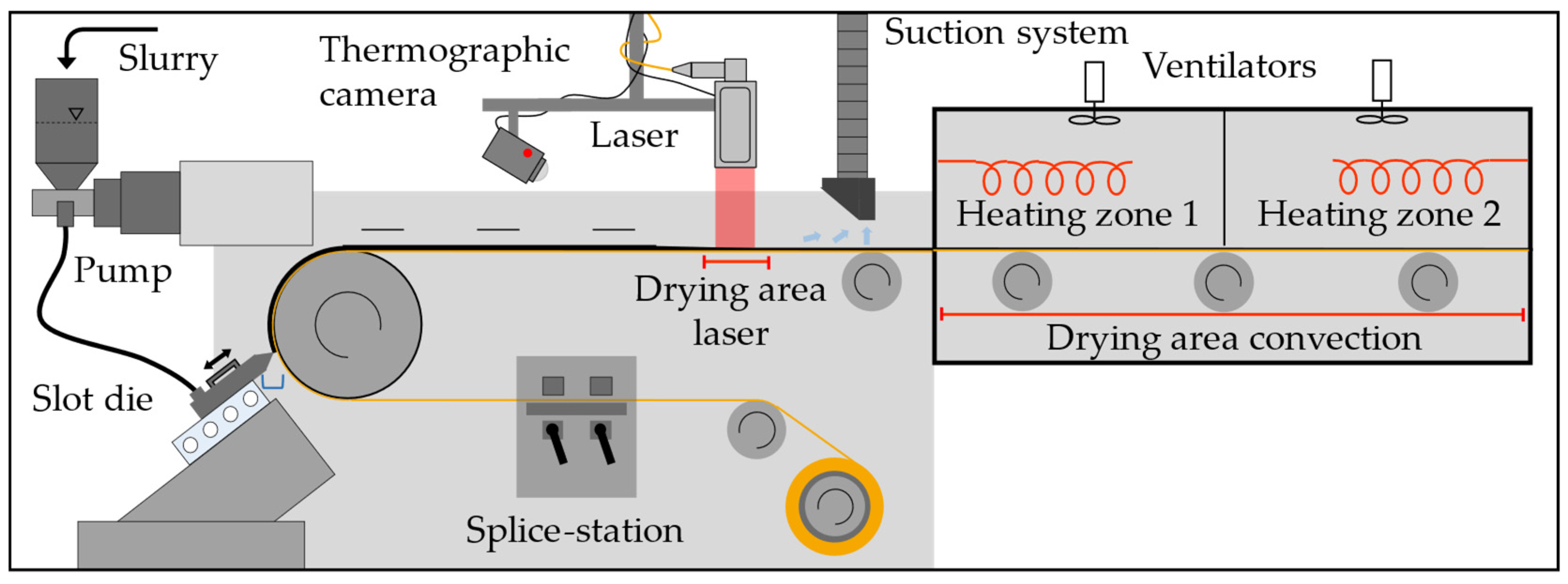
2.2. Experimental Setup and Process Parameters
2.3. Quality Tests
3. Results
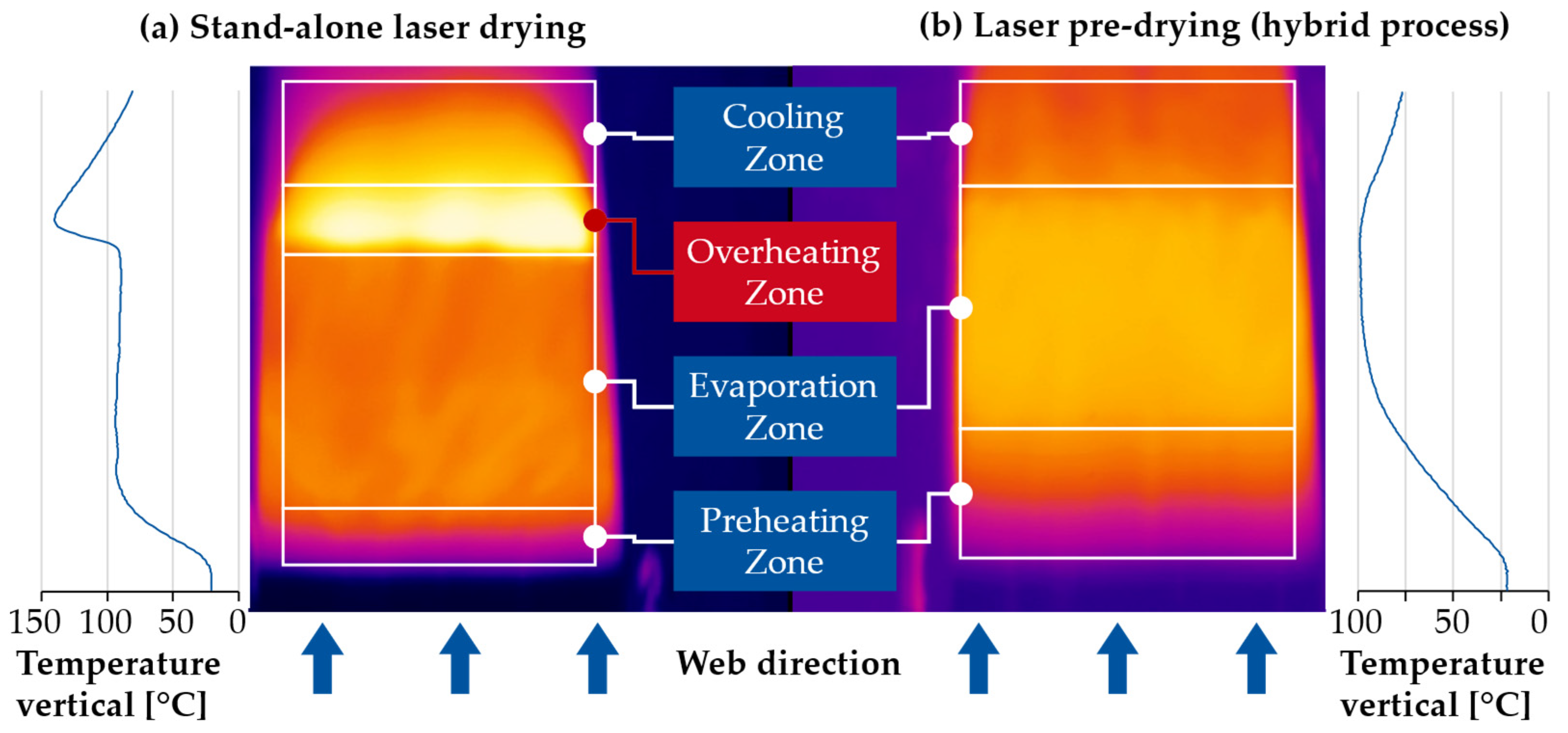
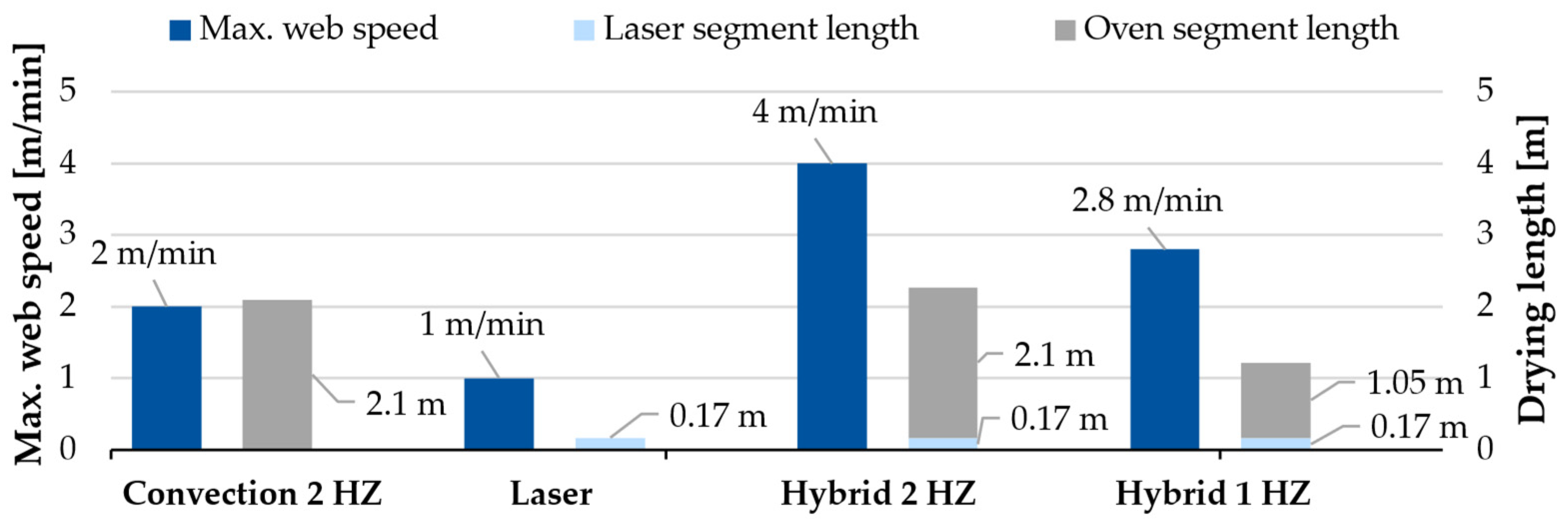
3.1. Production-Related Results
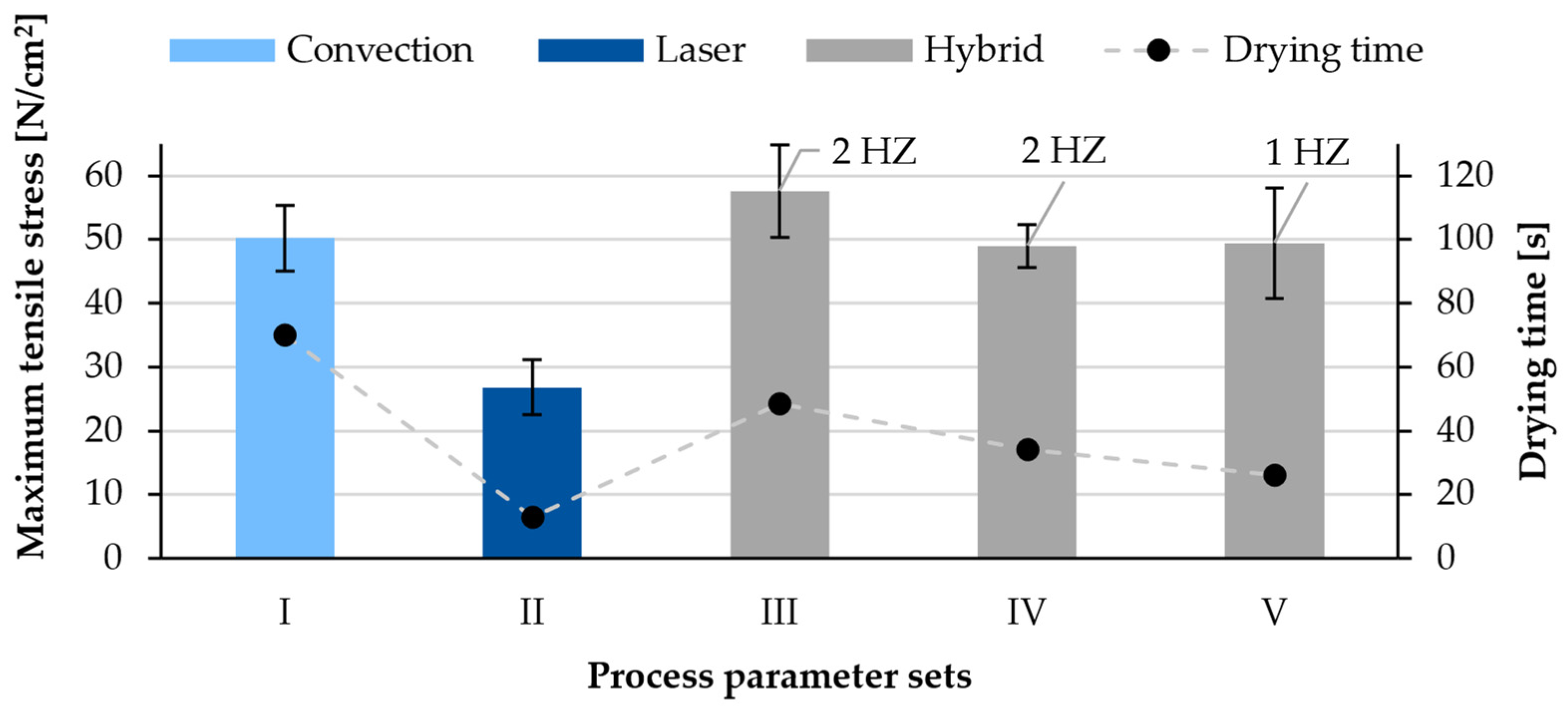
3.2. Adhesion
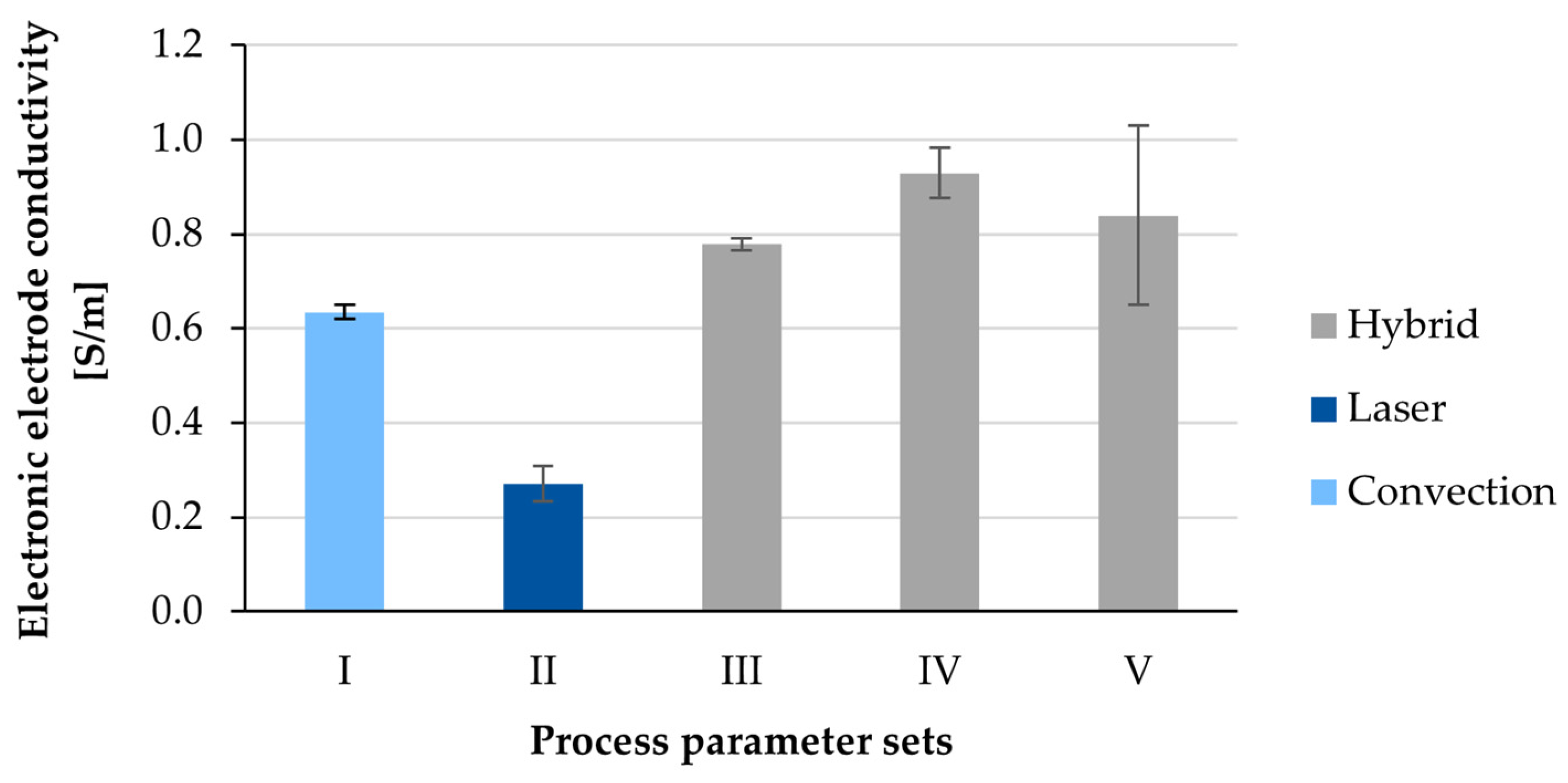
3.3. Electronic Conductivity
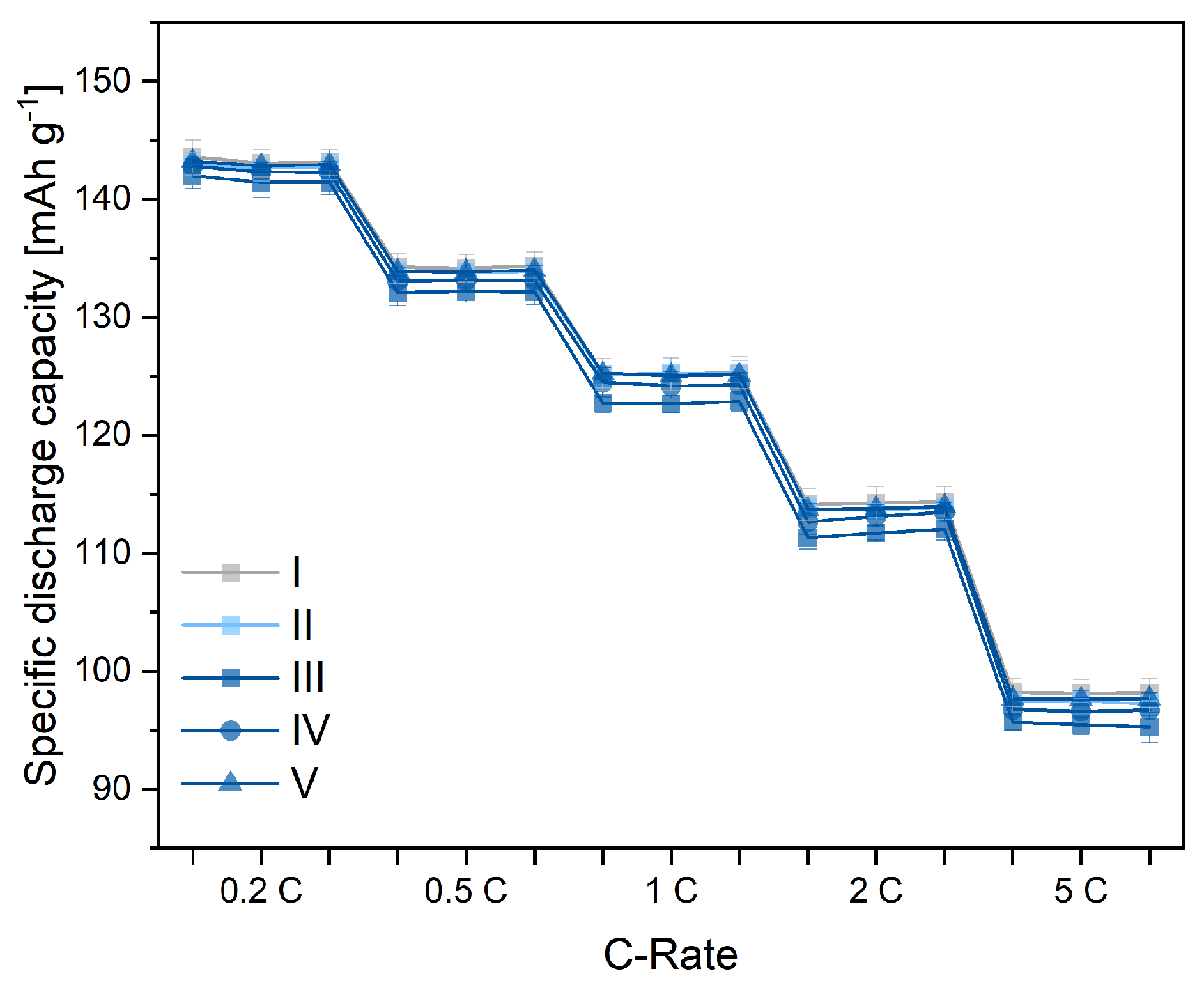
3.4. Electrochemical Performance
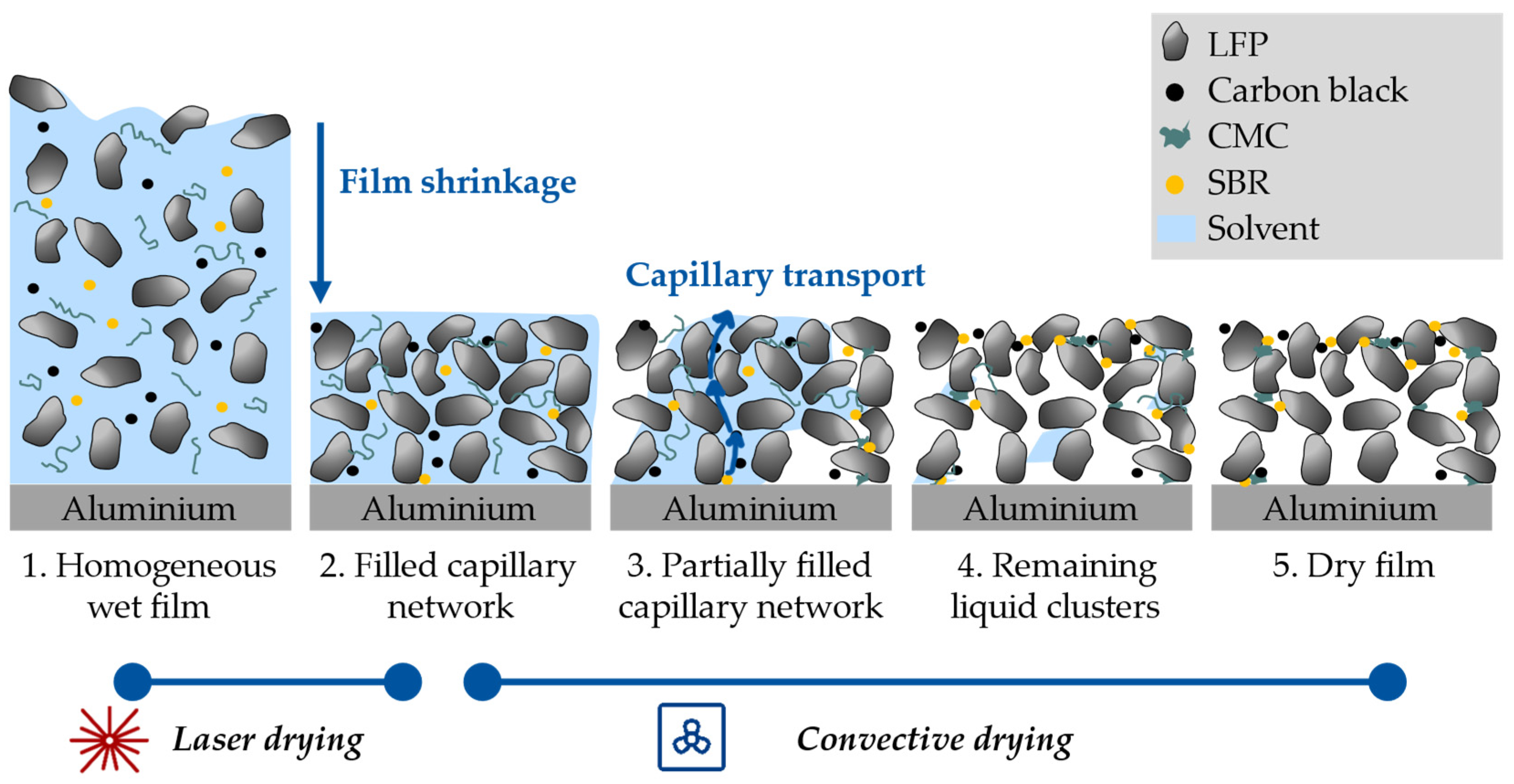
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Degen, F.; Schütte, M. Life cycle assessment of the energy consumption and GHG emissions of state-of-the-art automotive battery cell production. Clean. Prod. 2022, 330, 129798. [Google Scholar] [CrossRef]
- Liu, Y.; Zhang, R.; Wang, J.; Wang, Y. Current and future lithium-ion battery manufacturing. iScience 2021, 24, 102332. [Google Scholar] [CrossRef] [PubMed]
- Unlocking the Growth Opportunity in Battery Manufacturing Equipment. 2022. Available online: https://www.mckinsey.com/industries/industrials-and-electronics/our-insights/unlocking-the-growth-opportunity-in-battery-manufacturing-equipment (accessed on 20 September 2023).
- Production Process of a Lithium-Ion Battery Cell. 2023. Available online: https://www.pem.rwth-aachen.de/global/show_document.asp?id=aaaaaaaabwfmnyh (accessed on 27 March 2023).
- Mauler, L.; Duffner, F.; Leker, J. Economies of scale in battery cell manufacturing: The impact of material and process innovations. Appl. Energy 2021, 286, 116499. [Google Scholar] [CrossRef]
- Horstig, M.; Schoo, A.; Loellhoeffel, T.; Mayer, J.; Kwade, A. A Perspective on Innovative Drying Methods for Energy-Efficient Solvent-Based Production of Lithium-Ion Battery Electrodes. Energy Technol. 2022, 10, 2200689. [Google Scholar] [CrossRef]
- Neb, D.; Kim, S.; Clever, H.; Dorn, B.; Kampker, A. Current advances on laser drying of electrodes for lithium-ion battery cells. Procedia CIRP 2022, 107, 1577–1587. [Google Scholar] [CrossRef]
- Vedder, C.; Hawelka, D.; Wolter, M.; Leiva, D.; Stollenwerk, J.; Wissenbach, K. Laser-based drying of electrode layers. In Proceedings of the International Congress on Applications of Lasers & Electro-Optics, San Francisco, CA, USA, 16–20 October 2016. [Google Scholar]
- Fink, S.; Demir, D.; Börner, M.; Göken, V.; Vedder, C. High-Speed Laser Drying of Lithium-Ion Battery Anodes: Challenges and Opportunities. World Electr. Veh. J. 2023, 14, 255. [Google Scholar] [CrossRef]
- Diode Laser Drying of Electrodes for Lithium-Ion Batteries. 2023. Available online: https://www.pem.rwth-aachen.de/global/show_document.asp?id=aaaaaaaabzkmfxo (accessed on 19 July 2023).
- Klemens, J.; Schneider, L.; Herbst, E.C.; Bohn, N.; Müller, M.; Bauer, W.; Scharfer, P.; Schabel, W. Drying of NCM Cathode Electrodes with Porous, Nanostructured Particles versus Compact Solid Particles: Comparative Study of Binder Migration as a Function of Drying Conditions. Energy Technol. 2022, 10, 2100985. [Google Scholar] [CrossRef]
- Kumberg, J.; Müller, M.; Diehm, R.; Spiegel, S.; Wachsmann, C.; Bauer, W.; Scharfer, P.; Schabel, W. Drying of Lithium-Ion Battery Anodes for Use in High-Energy Cells: Influence of Electrode Thickness on Drying Time, Adhesion, and Crack Formation. Energy Technol. 2019, 7, 1900722. [Google Scholar] [CrossRef]
- Günther, T.; Billot, N.; Schuster, J.; Schnell, J.; Spingler, F.B.; Gasteiger, H.A. The Manufacturing of Electrodes: Key Process for the Future Success of Lithium-Ion Batteries. Adv. Mater. Res. 2016, 1140, 304–311. [Google Scholar] [CrossRef]
- Jaiser, S.; Müller, M.; Baunach, M.; Bauer, W.; Scharfer, P.; Schabel, W. Investigation of film solidification and binder migration during drying of Li-Ion battery anodes. J. Power Sources 2016, 318, 210–219. [Google Scholar] [CrossRef]
- Wolf, S.; Garbade, L.; Göken, V.; Tien, R.; Börner, M.; Neb, D.; Heimes, H.H. Process and Material Analysis of Laser- and Convection-Dried Silicon-Graphite Anodes for Lithium-Ion Batteries. World Electr. Veh. J. 2023, 14, 87. [Google Scholar] [CrossRef]
- Altvater, A.; Heckmann, T.; Eser, J.C.; Spiegel, S.; Scharfer, P.; Schabel, W. (Near-) Infrared Drying of Lithium-Ion Battery Electrodes: Influence of Energy Input on Process Speed and Electrode Adhesion. Energy Technol. 2022, 10, 2200785. [Google Scholar] [CrossRef]
- Jaiser, S.; Kumberg, J.; Klaver, J.; Urai, J.L.; Schabel, W.; Schmatz, J.; Scharfer, P. Micostructures formation of lithium-ion battery electrodes during drying—An ex-situ study using cyrogenic broad ion beam slope-cutting and scanning electron microscopy. J. Power Sources 2017, 345, 97–107. [Google Scholar] [CrossRef]
- Kumberg, J.; Baunach, M.; Eser, J.C.; Altvater, A.; Scharfer, P.; Schabel, W. Investigation of Drying Curves of Lithium-Ion Battery Electrodes with a New Gravimetrical Double-Side Batch Dryer Concept Including Setup Characterization and Model Simulations. Energy Technol. 2021, 9, 2000889. [Google Scholar] [CrossRef]
- Nikpour, M.; Liu, B.; Minson, P.; Hillman, Z.; Mazzeo, B.A.; Wheeler, D.R. Li-ion Electrode Microstructure Evolution during Drying and Calendering. Batteries 2022, 8, 107. [Google Scholar] [CrossRef]
- Font, F.; Protas, B.; Richardson, G.; Foster, J.M. Binder migration during drying of lithium-ion battery electrodes: Modelling and comparison to experiment. J. Power Sources 2018, 393, 177–185. [Google Scholar] [CrossRef]
- Jaiser, S.; Funk, L.; Baunach, M.; Scharfer, P.; Schabel, W. Experimental investigation into battery electrode surfaces: The distribution of liquid at the surface and the emptying of pores during drying. Colloid. Interface Sci. 2017, 494, 22–31. [Google Scholar] [CrossRef] [PubMed]
- Ambrock, K.; Ruttert, M.; Vinograd, A.; Billmann, B.; Yang, X.; Placke, T.; Winter, M.; Börner, M. Optimization of graphite/silicon-based composite electrodes for lithium ion batteries regarding the interdependencies of active and inactive materials. J. Power Sources 2022, 552, 232252. [Google Scholar] [CrossRef]
- Westphal, B.; Mainusch, N.; Meyer, C.; Haselrieder, W.; Indrikova, M.; Titscher, P.; Bockholt, H.; Viöl, W.; Kwade, A. Influence of high intensive dry mixing and calendering on relative electrode resistivity determined via an advanced two point approach. J. Energy Storage 2017, 11, 76–85. [Google Scholar] [CrossRef]
- Liu, D.; Chen, L.-C.; Liu, T.-J.; Chu, W.-B.; Tiu, C. Improvement of Lithium-Ion Battery Performance by Two-Layered Slot-Die Coating Operation. Energy Technol. 2017, 5, 1235–1241. [Google Scholar] [CrossRef]
- Orikasa, Y.; Gogyo, Y.; Yamashige, H.; Katayama, M.; Chen, K.; Mori, T.; Yamamoto, K.; Masese, T.; Inada, Y.; Ohta, T.; et al. Ionic Conduction in Lithium Ion Battery Composite Electrode Governs Cross-sectional Reaction Distribution. Sci. Rep. 2016, 6, 26382. [Google Scholar] [CrossRef] [PubMed]

| LFP | SBR | CMC | Carbon Black | Solvent | |
|---|---|---|---|---|---|
| wsolid (%) 1 | 86.0 | 2.7 | 2.0 | 9.3 | 0.0 |
| wtotal (%) 2 | 38.0 | 3.0 | 0.9 | 4.1 | 54.0 |
| Process Parameters | Convection | Laser | Hybrid | ||
|---|---|---|---|---|---|
| I | II | III | IV | V | |
| Web speed (m/min) | 1.8 | 0.8 | 2.8 | 4.0 | 2.8 |
| Pump speed (rpm) | 290 | 140 | 440 | 640 | 440 |
| Wet film thickness (µm) | 160 | 160 | 160 | 160 | 160 |
| Drying time laser/oven (s) | 0/70.0 | 12.8/0 | 3.6/45.0 | 2.6/31.5 | 3.6/22.5 |
| Oven heating zone 1 (°C) | 160 | <25 | 160 | 160 | 160 |
| Oven heating zone 2 (°C) | 140 | <25 | 140 | 140 | <25 |
| Laser power (W) | - | 456 | 736 | 1082 | 736 |
| Laser intensity (W/cm2) | - | 1.68 | 2.71 | 3.98 | 2.71 |
| Laser energy input (J/cm2) | - | 21.5 | 9.76 | 14.3 | 9.76 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wolf, S.; Schwenzer, N.; Tratz, T.; Göken, V.; Börner, M.; Neb, D.; Heimes, H.; Winter, M.; Kampker, A. Optimized LiFePO4-Based Cathode Production for Lithium-Ion Batteries through Laser- and Convection-Based Hybrid Drying Process. World Electr. Veh. J. 2023, 14, 281. https://doi.org/10.3390/wevj14100281
Wolf S, Schwenzer N, Tratz T, Göken V, Börner M, Neb D, Heimes H, Winter M, Kampker A. Optimized LiFePO4-Based Cathode Production for Lithium-Ion Batteries through Laser- and Convection-Based Hybrid Drying Process. World Electric Vehicle Journal. 2023; 14(10):281. https://doi.org/10.3390/wevj14100281
Chicago/Turabian StyleWolf, Sebastian, Niklas Schwenzer, Tim Tratz, Vinzenz Göken, Markus Börner, Daniel Neb, Heiner Heimes, Martin Winter, and Achim Kampker. 2023. "Optimized LiFePO4-Based Cathode Production for Lithium-Ion Batteries through Laser- and Convection-Based Hybrid Drying Process" World Electric Vehicle Journal 14, no. 10: 281. https://doi.org/10.3390/wevj14100281
APA StyleWolf, S., Schwenzer, N., Tratz, T., Göken, V., Börner, M., Neb, D., Heimes, H., Winter, M., & Kampker, A. (2023). Optimized LiFePO4-Based Cathode Production for Lithium-Ion Batteries through Laser- and Convection-Based Hybrid Drying Process. World Electric Vehicle Journal, 14(10), 281. https://doi.org/10.3390/wevj14100281








