Targeted Development of an Optimised Formulation for 3D-Printing of a Sertraline Hydrochloride-Containing Drug Delivery System with Immediate-Release Characteristics Utilising a Mixture Design
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Screening of Formulation Excipients
2.3. Thermogravimetric Analysis
2.4. Filament Production via Hot Melt Extrusion (HME)
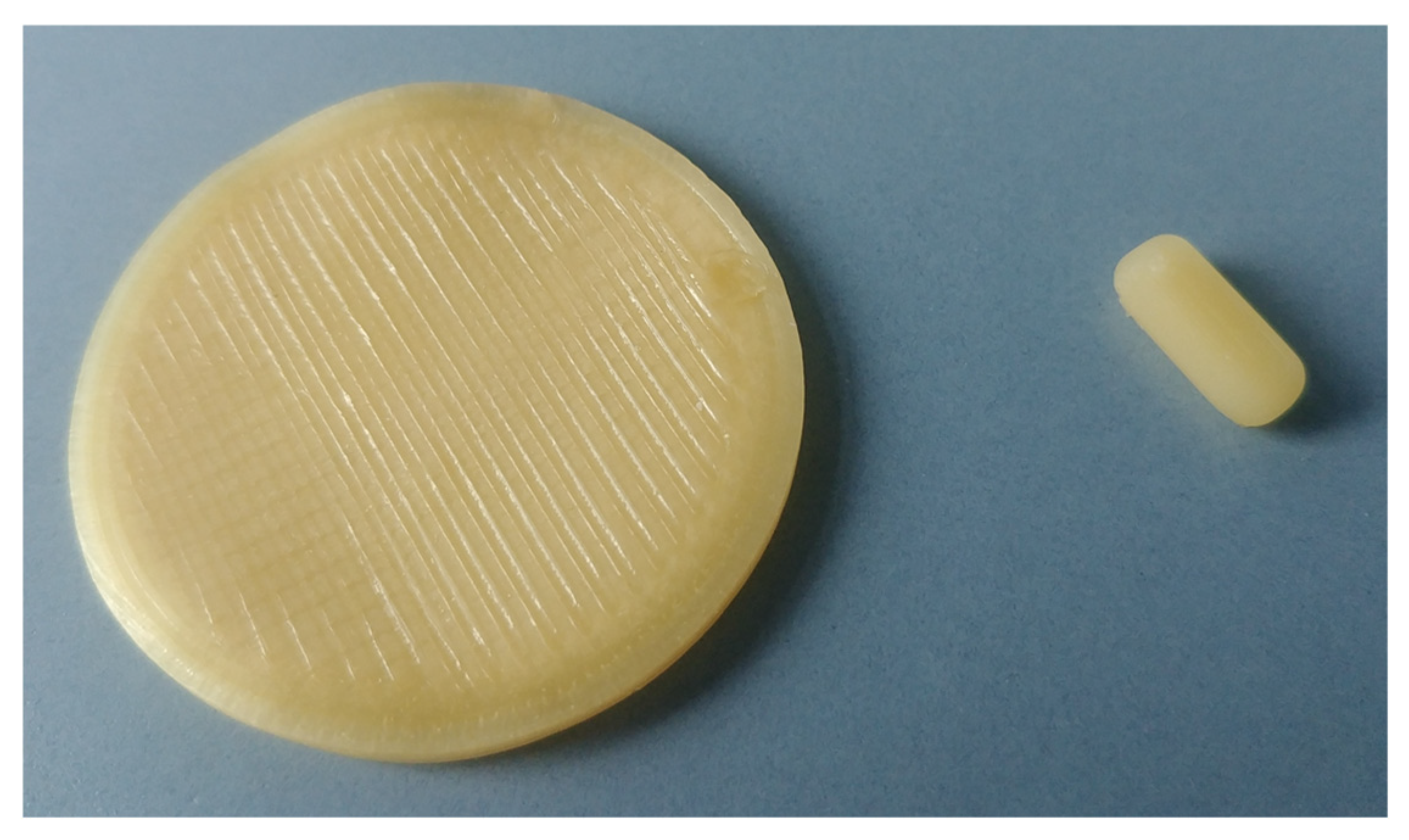
2.5. 3D Printing of Oral Dosage Forms
2.6. Design of Experiment (DoE)
2.7. Three-Point-Bending-Test
2.8. X-Ray Diffraction
2.9. Dissolution Studies
2.10. High Performance Liquid Chromatography (HPLC) Analysis
3. Results
3.1. Screening Observations for the Selection of DoE Limits
3.2. Selecting the Extrusion Temperature
3.3. Setting up the Mixture Design and Sample Preparation
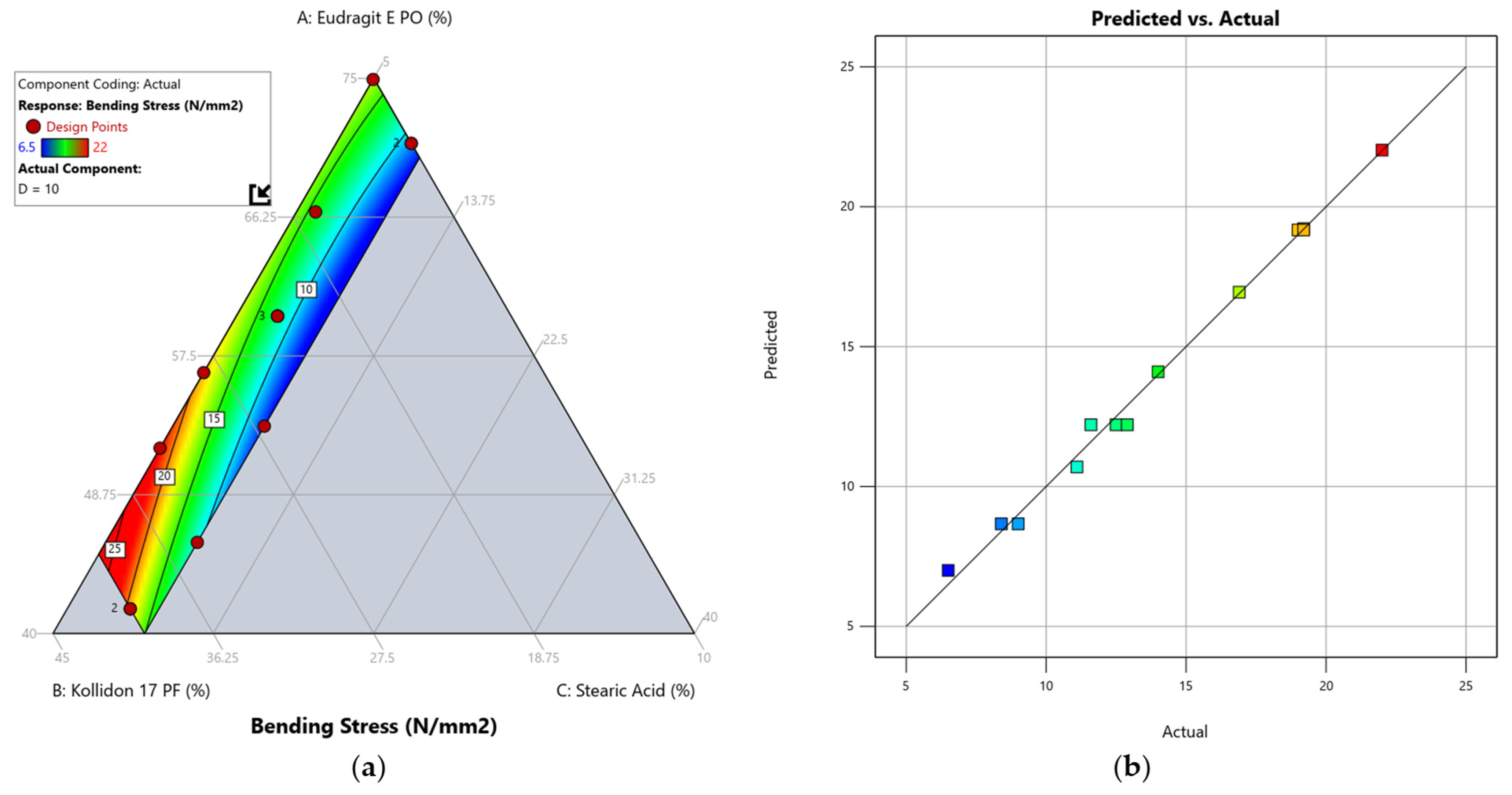
3.4. Effect of the Mixture Composition on the Bending Stress
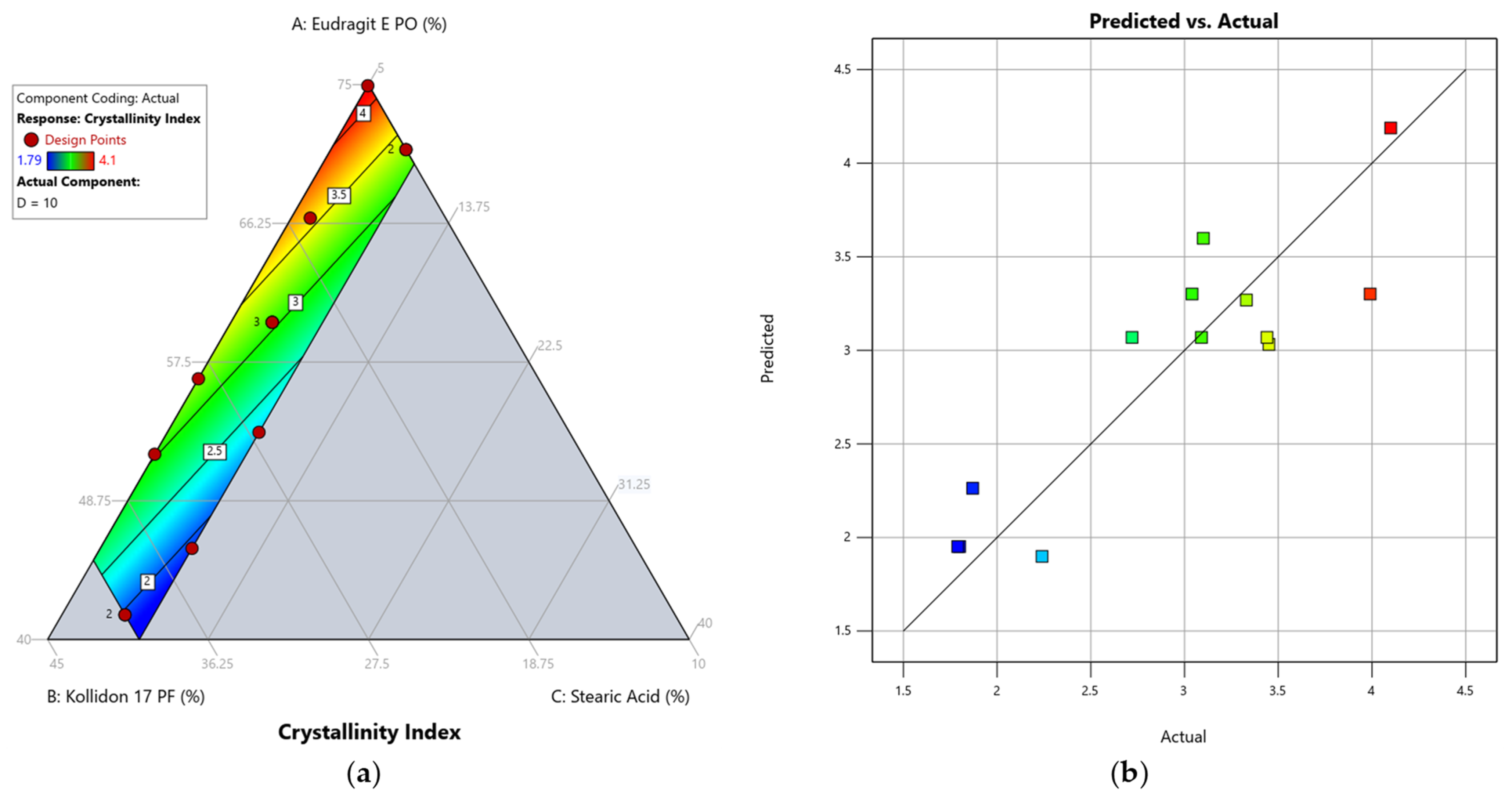
3.5. Effect of the Mixture Composition on the Crystallinity Index After Printing
3.6. Optimisation of the Formulation and Confirmation of the Model
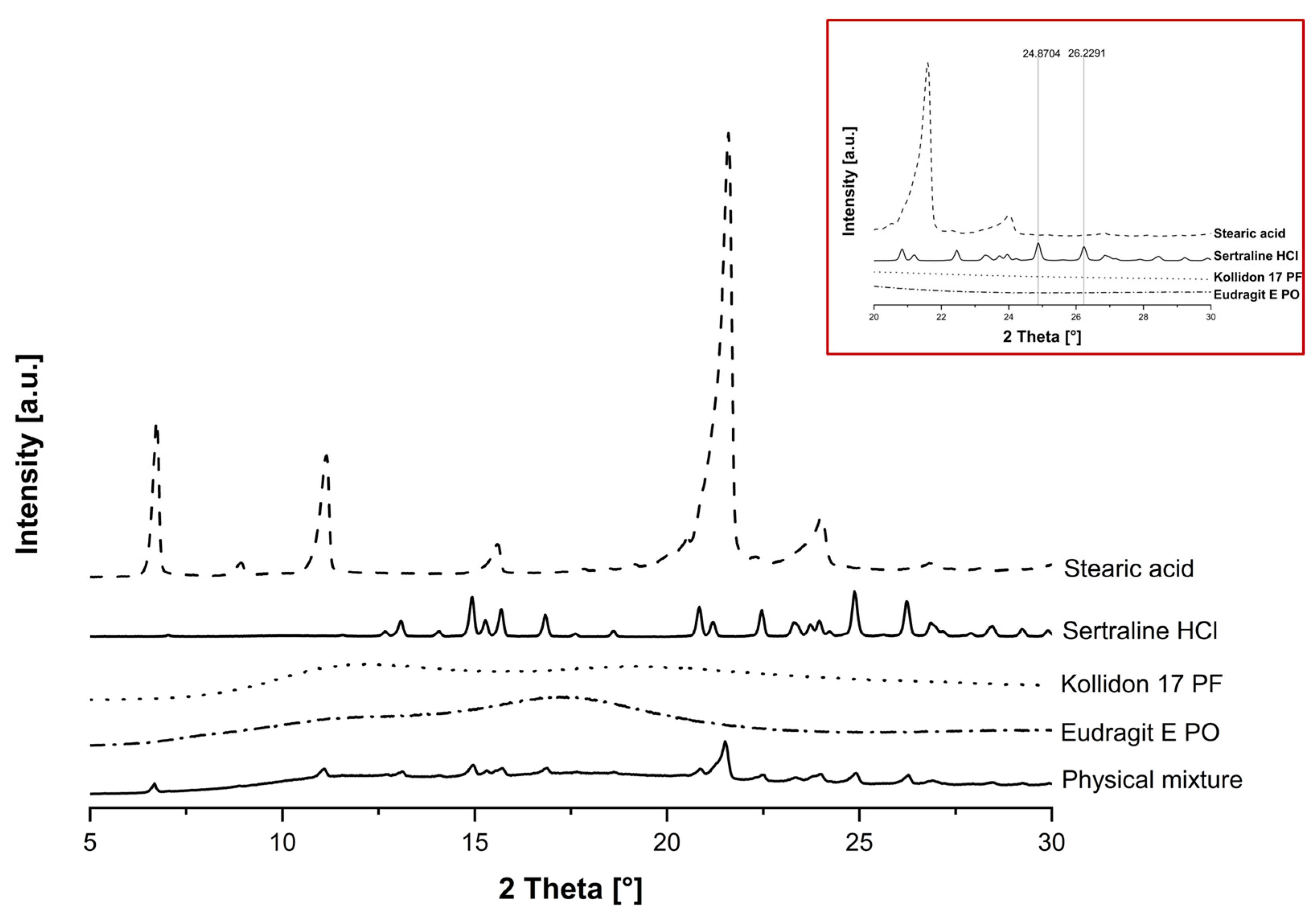
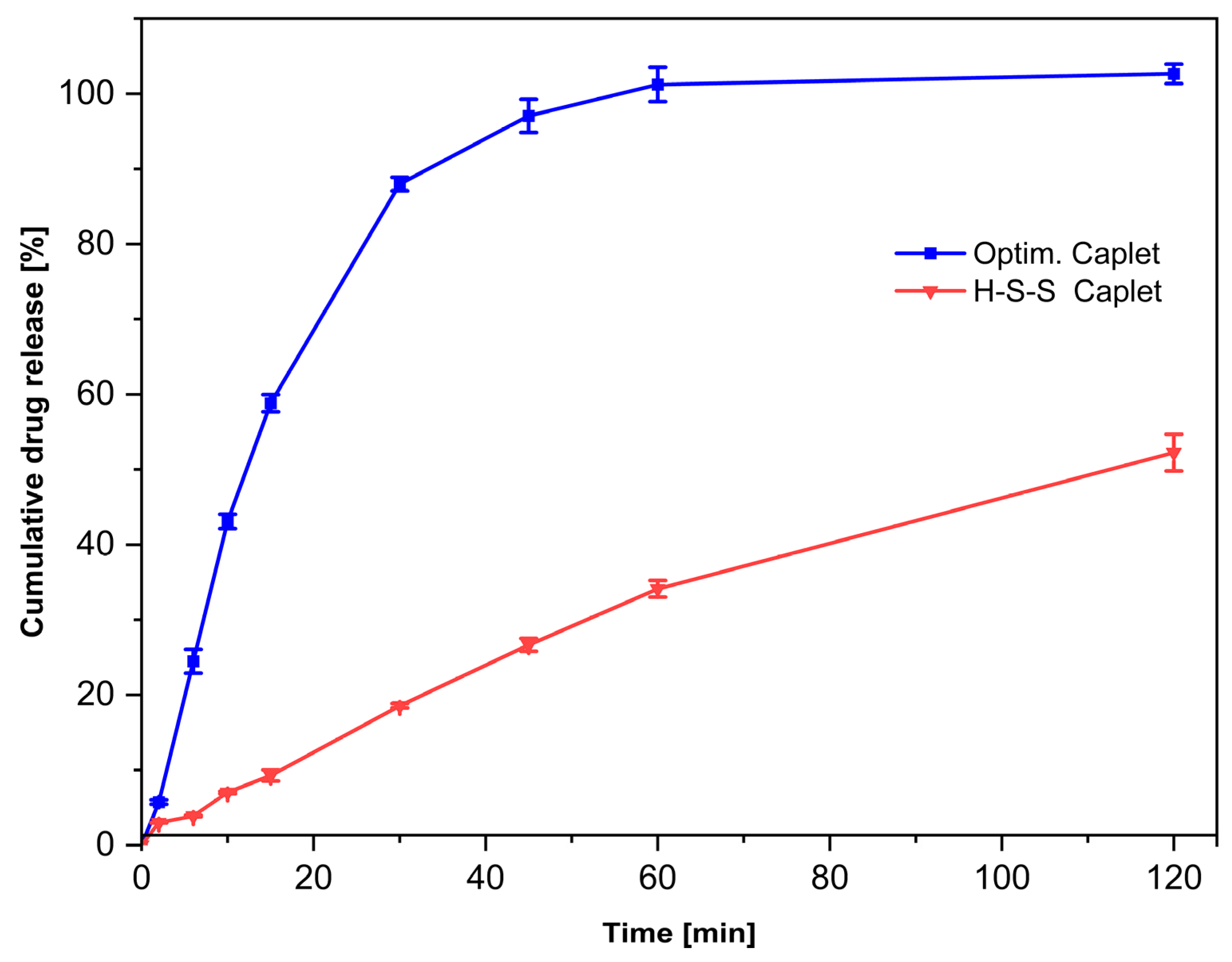
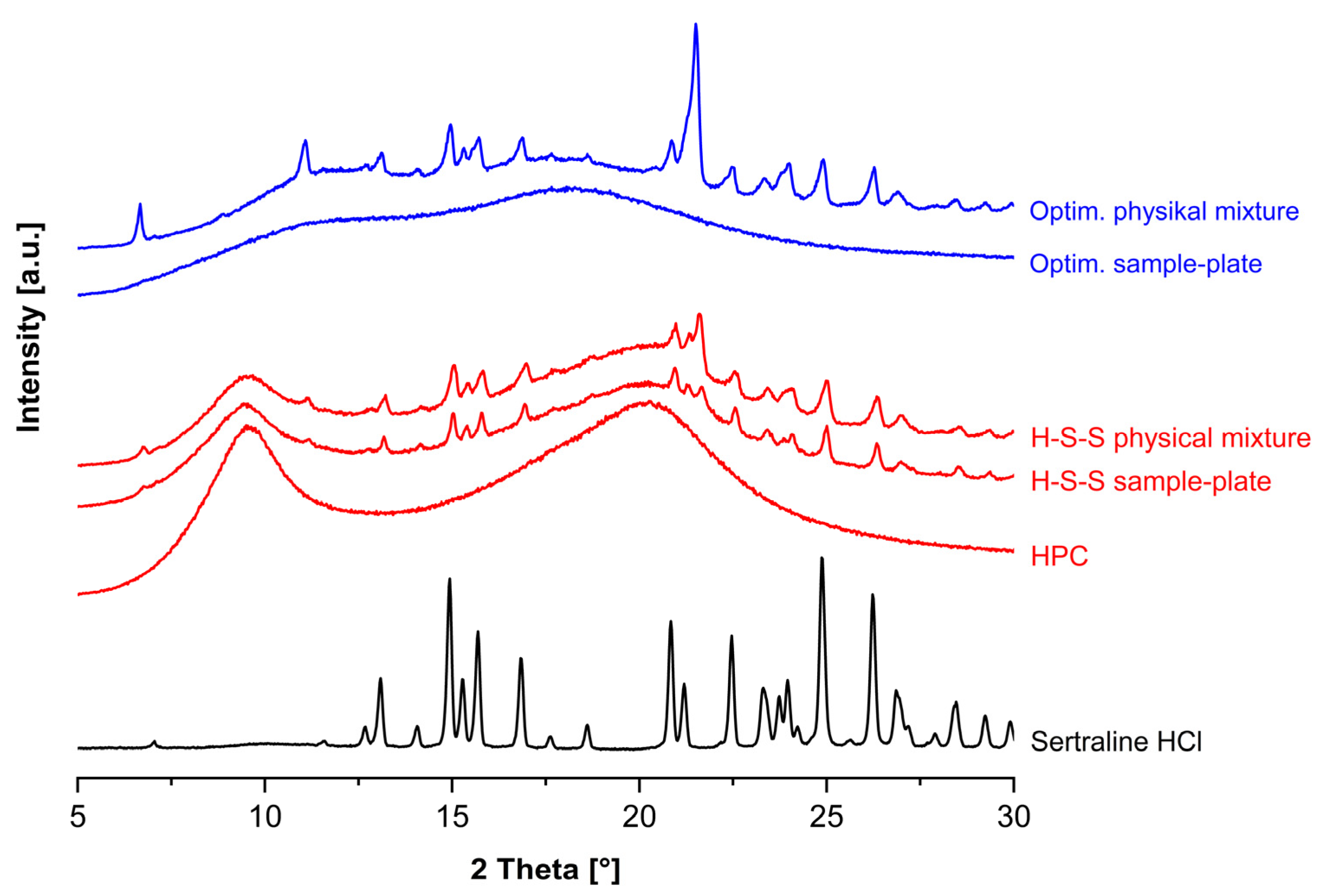
3.7. Dissolution Studies and Solid State Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Alqahtani, M.S.; Kazi, M.; Alsenaidy, M.A.; Ahmad, M.Z. Advances in Oral Drug Delivery. Front. Pharmacol. 2021, 12, 618411. [Google Scholar] [CrossRef]
- Wening, K.; Breitkreutz, J. Oral Drug Delivery in Personalized Medicine: Unmet Needs and Novel Approaches. Int. J. Pharm. 2011, 404, 1–9. [Google Scholar] [CrossRef]
- Alhnan, M.A.; Okwuosa, T.C.; Sadia, M.; Wan, K.-W.; Ahmed, W.; Arafat, B. Emergence of 3D Printed Dosage Forms: Opportunities and Challenges. Pharm. Res. 2016, 33, 1817–1832. [Google Scholar] [CrossRef]
- Lakkala, P.; Munnangi, S.R.; Bandari, S.; Repka, M. Additive Manufacturing Technologies with Emphasis on Stereolithography 3D Printing in Pharmaceutical and Medical Applications: A Review. Int. J. Pharm. X 2023, 5, 100159. [Google Scholar] [CrossRef] [PubMed]
- Cohen, J.S. Ways to Minimize Adverse Drug Reactions: Individualized Doses and Common Sense Are Key. Postgrad. Med. 1999, 106, 163–172. [Google Scholar] [CrossRef] [PubMed]
- Cailleaux, S.; Sanchez-Ballester, N.M.; Gueche, Y.A.; Bataille, B.; Soulairol, I. Fused Deposition Modeling (FDM), the New Asset for the Production of Tailored Medicines. J. Control. Release 2021, 330, 821–841. [Google Scholar] [CrossRef] [PubMed]
- Korte, C.; Quodbach, J. Formulation Development and Process Analysis of Drug-Loaded Filaments Manufactured via Hot-Melt Extrusion for 3D-Printing of Medicines. Pharm. Dev. Technol. 2018, 23, 1117–1127. [Google Scholar] [CrossRef]
- Jamróz, W.; Szafraniec, J.; Kurek, M.; Jachowicz, R. 3D Printing in Pharmaceutical and Medical Applications–Recent Achievements and Challenges. Pharm. Res. 2018, 35, 176. [Google Scholar] [CrossRef]
- Muhindo, D.; Elkanayati, R.; Srinivasan, P.; Repka, M.A.; Ashour, E.A. Recent Advances in the Applications of Additive Manufacturing (3D Printing) in Drug Delivery: A Comprehensive Review. AAPS PharmSciTech 2023, 24, 57. [Google Scholar] [CrossRef]
- Azad, M.A.; Olawuni, D.; Kimbell, G.; Badruddoza, A.Z.M.; Hossain, M.S.; Sultana, T. Polymers for Extrusion-Based 3D Printing of Pharmaceuticals: A Holistic Materials–Process Perspective. Pharmaceutics 2020, 12, 124. [Google Scholar] [CrossRef]
- Patil, H.; Tiwari, R.V.; Repka, M.A. Hot-Melt Extrusion: From Theory to Application in Pharmaceutical Formulation. AAPS PharmSciTech 2016, 17, 20–42. [Google Scholar] [CrossRef]
- Paredes, A.J.; McKenna, P.E.; Ramöller, I.K.; Naser, Y.A.; Volpe-Zanutto, F.; Li, M.; Abbate, M.T.A.; Zhao, L.; Zhang, C.; Abu-Ershaid, J.M.; et al. Microarray Patches: Poking a Hole in the Challenges Faced When Delivering Poorly Soluble Drugs. Adv. Funct. Mater. 2021, 31, 2005792. [Google Scholar] [CrossRef]
- Baghel, S.; Cathcart, H.; O’Reilly, N.J. Polymeric Amorphous Solid Dispersions: A Review of Amorphization, Crystallization, Stabilization, Solid-State Characterization, and Aqueous Solubilization of Biopharmaceutical Classification System Class II Drugs. J. Pharm. Sci. 2016, 105, 2527–2544. [Google Scholar] [CrossRef]
- Pereira, G.G.; Figueiredo, S.; Fernandes, A.I.; Pinto, J.F. Polymer Selection for Hot-Melt Extrusion Coupled to Fused Deposition Modelling in Pharmaceutics. Pharmaceutics 2020, 12, 795. [Google Scholar] [CrossRef]
- Alhijjaj, M.; Belton, P.; Qi, S. An Investigation into the Use of Polymer Blends to Improve the Printability of and Regulate Drug Release from Pharmaceutical Solid Dispersions Prepared via Fused Deposition Modeling (FDM) 3D Printing. Eur. J. Pharm. Biopharm. 2016, 108, 111–125. [Google Scholar] [CrossRef]
- Ilyés, K.; Kovács, N.K.; Balogh, A.; Borbás, E.; Farkas, B.; Casian, T.; Marosi, G.; Tomuță, I.; Nagy, Z.K. The Applicability of Pharmaceutical Polymeric Blends for the Fused Deposition Modelling (FDM) 3D Technique: Material Considerations–Printability–Process Modulation, with Consecutive Effects on in Vitro Release, Stability and Degradation. Eur. J. Pharm. Sci. 2019, 129, 110–123. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, C.-M.; Yang, T.-L.; Putri, A.D.; Chen, C.-T. Application of Design of Experiments in the Development of Self-Microemulsifying Drug Delivery Systems. Pharmaceuticals 2023, 16, 283. [Google Scholar] [CrossRef] [PubMed]
- Camacho Vieira, C.; Peltonen, L.; Karttunen, A.P.; Ribeiro, A.J. Is It Advantageous to Use Quality by Design (QbD) to Develop Nanoparticle-Based Dosage Forms for Parenteral Drug Administration? Int. J. Pharm. 2024, 657, 124163. [Google Scholar] [CrossRef]
- Kulkarni, V.R.; Bashyal, S.; Nair, V.V.; Duggal, I.; Maniruzzaman, M. Single-Step Extrusion Process for Formulation Development of Self-Emulsifying Granules for Oral Delivery of a BCS Class IV Drug. Mol. Pharm. 2024, 21, 6123–6136. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.X.; Amidon, G.; Khan, M.A.; Hoag, S.W.; Polli, J.; Raju, G.K.; Woodcock, J. Understanding Pharmaceutical Quality by Design. AAPS J. 2014, 16, 771. [Google Scholar] [CrossRef] [PubMed]
- Okwuosa, T.C.; Stefaniak, D.; Arafat, B.; Isreb, A.; Wan, K.-W.; Alhnan, M.A. A Lower Temperature FDM 3D Printing for the Manufacture of Patient-Specific Immediate Release Tablets. Pharm. Res. 2016, 33, 2704–2712. [Google Scholar] [CrossRef] [PubMed]
- Pietrzak, K.; Isreb, A.; Alhnan, M.A. A Flexible-Dose Dispenser for Immediate and Extended Release 3D Printed Tablets. Eur. J. Pharm. Biopharm. 2015, 96, 380–387. [Google Scholar] [CrossRef] [PubMed]
- Diniz, L.F.; Tenorio, J.C.; Ribeiro, C.; Carvalho, P.d.S. Structural Aspects, Solid-State Properties, and Solubility Performance of Pharmaceutical Sertraline-Based Organic Salts. J. Mol. Struct. 2023, 1273, 134293. [Google Scholar] [CrossRef]
- Amidon, G.L.; Lennernäs, H.; Shah, V.P.; Crison, J.R. A Theoretical Basis for a Biopharmaceutic Drug Classification: The Correlation of in Vitro Drug Product Dissolution and in Vivo Bioavailability. Pharm. Res. 1995, 12, 413–420. [Google Scholar] [CrossRef] [PubMed]
- Holsboer, F. How Can We Realize the Promise of Personalized Antidepressant Medicines? Nat. Rev. Neurosci. 2008, 9, 638–646. [Google Scholar] [CrossRef]
- Haddad, P.M.; Brain, C.; Scott, J. Nonadherence with Antipsychotic Medication in Schizophrenia: Challenges and Management Strategies. Patient Relat. Outcome Meas. 2014, 5, 43–62. [Google Scholar] [CrossRef]
- Kollamaram, G.; Croker, D.M.; Walker, G.M.; Goyanes, A.; Basit, A.W.; Gaisford, S. Low Temperature Fused Deposition Modeling (FDM) 3D Printing of Thermolabile Drugs. Int. J. Pharm. 2018, 545, 144–152. [Google Scholar] [CrossRef]
- Overgaard, A.B.A.; Møller-Sonnergaard, J.; Christrup, L.L.; Højsted, J.; Hansen, R. Patients’ Evaluation of Shape, Size and Colour of Solid Dosage Forms. Pharm. World Sci. 2001, 23, 185–188. [Google Scholar] [CrossRef]
- Sopyan, I.; Gozali, D.; Sriwidodo, D.G.; Guntina, R.K. Design-Expert Software (DOE): An Application Tool for Optimization in Pharmaceutical Preparations Formulation. Int. J. Appl. Pharm. 2022, 14, 55–63. [Google Scholar] [CrossRef]
- Malaquias, L.F.B.; Schulte, H.L.; Chaker, J.A.; Karan, K.; Durig, T.; Marreto, R.N.; Gratieri, T.; Gelfuso, G.M.; Cunha-Filho, M. Hot Melt Extrudates Formulated Using Design Space: One Simple Process for Both Palatability and Dissolution Rate Improvement. J. Pharm. Sci. 2018, 107, 286–296. [Google Scholar] [CrossRef]
- Hartzke, D.; Pössl, A.; Schlupp, P.; Runkel, F.E. Evaluation of Hydroxyethyl Cellulose Grades as the Main Matrix Former to Produce 3D-Printed Controlled-Release Dosage Forms. Pharmaceutics 2022, 14, 2103. [Google Scholar] [CrossRef]
- Mamidi, H.K.; Rohera, B.D. Material-Sparing Approach Using Differential Scanning Calorimeter and Response Surface Methodology for Process Optimization of Hot-Melt Extrusion. J. Pharm. Sci. 2021, 110, 3838–3850. [Google Scholar] [CrossRef]
- Grellmann, W.; Seidler, S. Kunststoffprüfung; Carl Hanser Verlag GmbH Co KG: Munich, Germany, 2024; ISBN 978-3-446-48105-3. [Google Scholar]
- Salem, K.S.; Kasera, N.K.; Rahman, M.A.; Jameel, H.; Habibi, Y.; Eichhorn, S.J.; French, A.D.; Pal, L.; Lucia, L.A. Comparison and Assessment of Methods for Cellulose Crystallinity Determination. Chem. Soc. Rev. 2023, 52, 6417–6446. [Google Scholar] [CrossRef]
- Melocchi, A.; Parietti, F.; Loreti, G.; Maroni, A.; Gazzaniga, A.; Zema, L. 3D Printing by Fused Deposition Modeling (FDM) of a Swellable/Erodible Capsular Device for Oral Pulsatile Release of Drugs. J. Drug Deliv. Sci. Technol. 2015, 30, 360–367. [Google Scholar] [CrossRef]
- Than, Y.M.; Titapiwatanakun, V. Tailoring Immediate Release FDM 3D Printed Tablets Using a Quality by Design (QbD) Approach. Int. J. Pharm. 2021, 599, 120402. [Google Scholar] [CrossRef]
- Zidan, A.; Alayoubi, A.; Coburn, J.; Asfari, S.; Ghammraoui, B.; Cruz, C.N.; Ashraf, M. Extrudability Analysis of Drug Loaded Pastes for 3D Printing of Modified Release Tablets. Int. J. Pharm. 2019, 554, 292–301. [Google Scholar] [CrossRef]
- Macwan, J.S.; Fraczkiewicz, G.; Bertolino, M.; Krüger, P.; Peters, S.-A. Application of Physiologically Based Biopharmaceutics Modeling to Understand the Impact of Dissolution Differences on in Vivo Performance of Immediate Release Products: The Case of Bisoprolol. CPT Pharmacomet. Syst. Pharmacol. 2021, 10, 622–632. [Google Scholar] [CrossRef]
- Kempin, W.; Domsta, V.; Grathoff, G.; Brecht, I.; Semmling, B.; Tillmann, S.; Weitschies, W.; Seidlitz, A. Immediate Release 3D-Printed Tablets Produced Via Fused Deposition Modeling of a Thermo-Sensitive Drug. Pharm. Res. 2018, 35, 124. [Google Scholar] [CrossRef]
- Nasereddin, J.M.; Wellner, N.; Alhijjaj, M.; Belton, P.; Qi, S. Development of a Simple Mechanical Screening Method for Predicting the Feedability of a Pharmaceutical FDM 3D Printing Filament. Pharm. Res. 2018, 35, 151. [Google Scholar] [CrossRef]
- Bruère, V.M.; Lion, A.; Holtmannspötter, J.; Johlitz, M. Under-Extrusion Challenges for Elastic Filaments: The Influence of Moisture on Additive Manufacturing. Prog. Addit. Manuf. 2022, 7, 445–452. [Google Scholar] [CrossRef]
- Li, J.; Li, C.; Zhang, H.; Gao, X.; Wang, T.; Wang, Z.; Zheng, A. Preparation of Azithromycin Amorphous Solid Dispersion by Hot-Melt Extrusion: An Advantageous Technology with Taste Masking and Solubilization Effects. Polymers 2022, 14, 495. [Google Scholar] [CrossRef]
- Alshahrani, S.M.; Morott, J.T.; Alshetaili, A.S.; Tiwari, R.V.; Majumdar, S.; Repka, M.A. Influence of Degassing on Hot-Melt Extrusion Process. Eur. J. Pharm. Sci. 2015, 80, 43–52. [Google Scholar] [CrossRef]
- Zayed, M.A.; Hawash, M.F.; Fahmey, M.A.; El-Habeeb, A.A. Structure Investigation of Sertraline Drug and Its Iodine Product Using Mass Spectrometry, Thermal Analyses and MO-Calculations. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2007, 68, 970–978. [Google Scholar] [CrossRef] [PubMed]
- Uttreja, P.; Youssef, A.A.A.; Karnik, I.; Sanil, K.; Narala, N.; Wang, H.; Elkanayati, R.M.; Vemula, S.K.; Repka, M.A. Formulation Development of Solid Self-Nanoemulsifying Drug Delivery Systems of Quetiapine Fumarate via Hot-Melt Extrusion Technology: Optimization Using Central Composite Design. Pharmaceutics 2024, 16, 324. [Google Scholar] [CrossRef] [PubMed]
- Vemula, S.K.; Narala, S.; Uttreja, P.; Narala, N.; Daravath, B.; Kalla, C.S.A.; Baisa, S.; Munnangi, S.R.; Chella, N.; Repka, M.A. Quality by Design (QbD) Approach to Develop Colon-Specific Ketoprofen Hot-Melt Extruded Pellets: Impact of Eudragit® S 100 Coating on the In Vitro Drug Release. Pharmaceutics 2024, 16, 1265. [Google Scholar] [CrossRef] [PubMed]
- Desai, D.; Sandhu, H.; Shah, N.; Malick, W.; Zia, H.; Phuapradit, W.; Vaka, S.R.K. Selection of Solid-State Plasticizers as Processing Aids for Hot-Melt Extrusion. J. Pharm. Sci. 2018, 107, 372–379. [Google Scholar] [CrossRef]
- Stukalin, E.B.; Douglas, J.F.; Freed, K.F. Plasticization and Antiplasticization of Polymer Melts Diluted by Low Molar Mass Species. J. Chem. Phys. 2010, 132, 084504. [Google Scholar] [CrossRef]
- Van den Mooter, G. The Use of Amorphous Solid Dispersions: A Formulation Strategy to Overcome Poor Solubility and Dissolution Rate. Drug Discov. Today Technol. 2012, 9, e79–e85. [Google Scholar] [CrossRef]
- Ahvenainen, P.; Kontro, I.; Svedström, K. Comparison of Sample Crystallinity Determination Methods by X-Ray Diffraction for Challenging Cellulose I Materials. Cellulose 2016, 23, 1073–1086. [Google Scholar] [CrossRef]
- Ferreira, A.P.G.; Pinto, B.V.; Cavalheiro, É.T.G. Thermal Decomposition Investigation of Paroxetine and Sertraline. J. Anal. Appl. Pyrolysis 2018, 136, 232–241. [Google Scholar] [CrossRef]
- Jagtap, P.S.; Tagad, R.R.; Shendge, R.S. A Brief Review on Kollidon. J. Drug Deliv. Ther. 2019, 9, 493–500. [Google Scholar] [CrossRef]
- Chen, Z.-Q.; Liu, Y.; Zhao, J.-H.; Wang, L.; Feng, N.-P. Improved Oral Bioavailability of Poorly Water-Soluble Indirubin by a Supersaturatable Self-Microemulsifying Drug Delivery System. Int. J. Nanomed. 2012, 7, 1115–1125. [Google Scholar] [CrossRef]
- Ragonese, R.; Macka, M.; Hughes, J.; Petocz, P. The Use of the Box–Behnken Experimental Design in the Optimisation and Robustness Testing of a Capillary Electrophoresis Method for the Analysis of Ethambutol Hydrochloride in a Pharmaceutical Formulation. J. Pharm. Biomed. Anal. 2002, 27, 995–1007. [Google Scholar] [CrossRef]
- Singh, D.; Tiwary, A.K.; Bedi, N. Canagliflozin Loaded SMEDDS: Formulation Optimization for Improved Solubility, Permeability and Pharmacokinetic Performance. J. Pharm. Investig. 2019, 49, 67–85. [Google Scholar] [CrossRef]
- Xu, P.; Li, J.; Meda, A.; Osei-Yeboah, F.; Peterson, M.L.; Repka, M.; Zhan, X. Development of a Quantitative Method to Evaluate the Printability of Filaments for Fused Deposition Modeling 3D Printing. Int. J. Pharm. 2020, 588, 119760. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Xu, P.; Vo, A.Q.; Bandari, S.; Yang, F.; Durig, T.; Repka, M.A. Development and Evaluation of Pharmaceutical 3D Printability for Hot Melt Extruded Cellulose-Based Filaments. J. Drug Deliv. Sci. Technol. 2019, 52, 292–302. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, J.; da Silva, G.S.; Velho, M.C.; Beck, R.C.R. Eudragit®: A Versatile Family of Polymers for Hot Melt Extrusion and 3D Printing Processes in Pharmaceutics. Pharmaceutics 2021, 13, 1424. [Google Scholar] [CrossRef]
- Claeys, B.; Coen, R.D.; De Geest, B.G.; de la Rosa, V.R.; Hoogenboom, R.; Carleer, R.; Adriaensens, P.; Remon, J.P.; Vervaet, C. Structural Modifications of Polymethacrylates: Impact on Thermal Behavior and Release Characteristics of Glassy Solid Solutions. Eur. J. Pharm. Biopharm. 2013, 85, 1206–1214. [Google Scholar] [CrossRef]
- Hancock, B.C.; Zografi, G. Characteristics and Significance of the Amorphous State in Pharmaceutical Systems. J. Pharm. Sci. 1997, 86, 1–12. [Google Scholar] [CrossRef]
- Saerens, L.; Dierickx, L.; Lenain, B.; Vervaet, C.; Remon, J.P.; Beer, T.D. Raman Spectroscopy for the In-Line Polymer–Drug Quantification and Solid State Characterization during a Pharmaceutical Hot-Melt Extrusion Process. Eur. J. Pharm. Biopharm. 2011, 77, 158–163. [Google Scholar] [CrossRef]

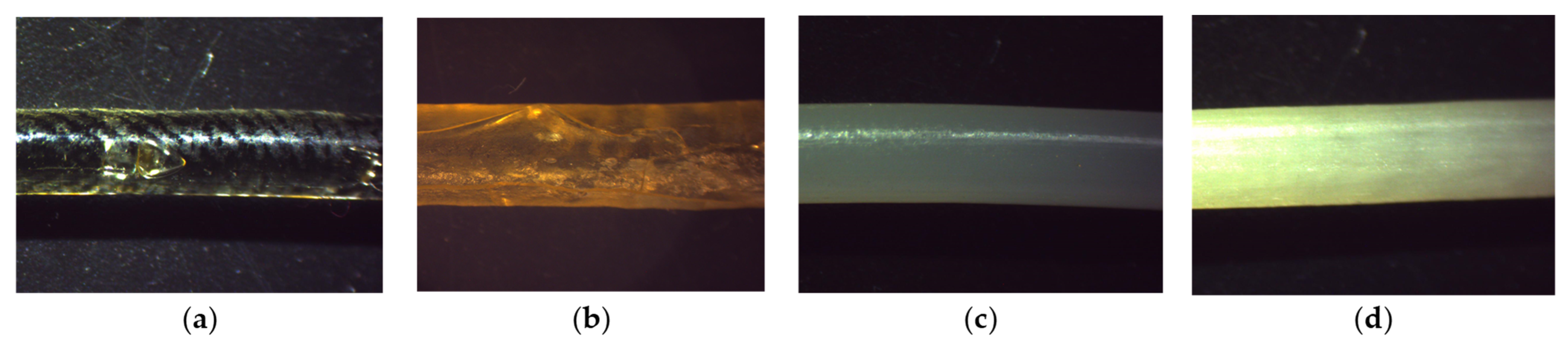


| Blend-Name | Eudragit E PO (% w/w) | Kollidon 17 PF (% w/w) | HPC (% w/w) | Stearic Acid (% w/w) |
|---|---|---|---|---|
| E-S | 95 | - | - | 5 |
| K-S | - | 95 | - | 5 |
| H-S | - | - | 95 | 5 |
| E-K-S | 65 | 30 | - | 5 |
| Printing Parameter | E-S | K-S | H-S | E-K-S | DoE-Runs |
|---|---|---|---|---|---|
| Bed temperature | 80 °C | 70 °C | 70 °C | 70 °C | 70 °C |
| Printing temperature | 170 °C | 170 °C | 160 °C | 165 °C | 160 °C |
| Printing speed | 20 mm/s | 20 mm/s | 20 mm/s | 20 mm/s | 20 mm/s |
| Layer height | 0.8 mm | 0.8 mm | 0.8 mm | 0.8 mm | 0.8 mm |
| Layer width | 0.1 mm | 0.1 mm | 0.1 mm | 0.1 mm | 0.1 mm |
| Mixture Component | Proportion of Powder Blend (% w/w) | |
|---|---|---|
| Low Limit | High Limit | |
| Eudragit E PO | 40 | 75 |
| Kollidon 17 | 10 | 40 |
| Stearic acid | 5 | 10 |
| Sertraline HCl | 10 | 10 |
| Blend-Name | Extrudability | Extrusion Temperature (°C) | Printability | Printing Temperature (°C) | Disintegration Time (min) |
|---|---|---|---|---|---|
| E-S | (+) | 120 | (−) 1 | 170 | 19.27 ± 0.75 |
| K-S | (+) | 170 | (−) | - | - |
| H-S | (+) | 150 | (+) | 160 | >60 |
| E-K-S | (+) | 160 | (+) | 165 | 28.00 ± 1.63 |
| Blend-Name/ DoE-run (R) | Eudragit E PO (% w/w) | Kollidon 17 PF (% w/w) | Stearic Acid (% w/w) | Sertraline HCl (% w/w) | Bending Stress (N/mm2) | Crystallinity Index |
|---|---|---|---|---|---|---|
| R1 | 53.07 | 26.93 | 10 | 10 | 6.54 | 1.87 |
| R2 | 70.9 | 10 | 9.1 | 10 | 9.02 | 3.99 |
| R3 | 45.75 | 34.25 | 10 | 10 | 11.15 | 2.24 |
| R4 | 60.01 | 22.74 | 7.25 | 10 | 12.53 | 3.44 |
| R5 | 41.56 | 40 | 8.44 | 10 | 19.16 | 1.79 |
| R6 | 60.01 | 22.74 | 7.25 | 10 | 12.86 | 2.72 |
| R7 | 66.58 | 17.38 | 6.04 | 10 | 14.03 | 3.10 |
| R8 | 56.45 | 28.55 | 5 | 10 | 19.18 | 3.33 |
| R9 | 60.01 | 22.74 | 7.25 | 10 | 11.63 | 3.09 |
| R10 | 51.69 | 33.31 | 5 | 10 | 21.71 | 3.45 |
| R11 | 74.93 | 10.07 | 5 | 10 | 16.88 | 4.10 |
| R12 | 41.56 | 40 | 8.44 | 10 | 19.43 | 1.80 |
| R13 | 70.9 | 10 | 9.1 | 10 | 8.45 | 3.04 |
| Source | Sum of Squares | df | Mean Square | F-Value | p-Value |
|---|---|---|---|---|---|
| Model | 280.18 | 5 | 56.04 | 253.96 | <0.0001 |
| Linear mixture | 234.40 | 2 | 117.20 | 531.15 | <0.0001 |
| AB | 45.51 | 1 | 45.51 | 206.24 | <0.0001 |
| AC | 0.2184 | 1 | 0.2184 | 0.9896 | 0.3530 |
| BC | 0.0233 | 1 | 0.0233 | 0.1056 | 0.7548 |
| Residual | 1.54 | 7 | 0.2206 | ||
| Lack of fit | 0.4579 | 3 | 0.1526 | 0.5618 | 0.6683 |
| Pure error | 1.09 | 4 | 0.2717 | ||
| Total | 281.72 | 12 | |||
| R2 | 0.9945 | ||||
| Adjusted R2 | 0.9906 | ||||
| Predicted R2 | 0.9839 | ||||
| Adequate precision | 47.0840 |
| Source | Sum of Squares | df | Mean Square | F-Value | p-Value |
|---|---|---|---|---|---|
| Model | 5.91 | 2 | 2.96 | 18.97 | 0.0004 |
| Linear mixture | 5.91 | 2 | 2.96 | 18.97 | 0.0004 |
| Residual | 1.56 | 10 | 0.1558 | ||
| Lack of fit | 0.8471 | 6 | 0.1412 | 0.7948 | 0.6190 |
| Pure error | 0.7106 | 4 | 0.1776 | ||
| Total | 7.47 | 12 | |||
| R2 | 0.7914 | ||||
| Adjusted R2 | 0.7497 | ||||
| Predicted R2 | 0.6243 | ||||
| Adequate precision | 12.0762 |
| Variable | Goal | Lower Limit | Upper Limit | Importance |
|---|---|---|---|---|
| Eudragit E PO (% w/w) | Is in range | 40 | 75 | 3 |
| Kollidon 17 PF (% w/w) | Is in range | 10 | 40 | 3 |
| Stearic acid (% w/w) | Is in range | 5 | 10 | 3 |
| Bending stress (N/mm2) | Minimise | 6.5 | 22.0 | 1 |
| Crystallinity index | Minimise | 1.8 | 4.1 | 5 |
| Response | 95% CI Low | Predicted Mean | Experimental Mean | 95% CI High | Predicted Std. Error | Std. Deviation |
|---|---|---|---|---|---|---|
| Bending Stress | 11.2957 | 12.2199 | 12.9974 | 13.1441 | 0.400794 | 0.442695 |
| Crystallinity Index | 1.08774 | 1.79015 | 1.74145 | 2.49256 | 0.315247 | 0.394677 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bienhaus, M.; Neumann, L.; Müller, C.; Runkel, F.E. Targeted Development of an Optimised Formulation for 3D-Printing of a Sertraline Hydrochloride-Containing Drug Delivery System with Immediate-Release Characteristics Utilising a Mixture Design. Pharmaceutics 2025, 17, 1137. https://doi.org/10.3390/pharmaceutics17091137
Bienhaus M, Neumann L, Müller C, Runkel FE. Targeted Development of an Optimised Formulation for 3D-Printing of a Sertraline Hydrochloride-Containing Drug Delivery System with Immediate-Release Characteristics Utilising a Mixture Design. Pharmaceutics. 2025; 17(9):1137. https://doi.org/10.3390/pharmaceutics17091137
Chicago/Turabian StyleBienhaus, Mirco, Leif Neumann, Charlotte Müller, and Frank E. Runkel. 2025. "Targeted Development of an Optimised Formulation for 3D-Printing of a Sertraline Hydrochloride-Containing Drug Delivery System with Immediate-Release Characteristics Utilising a Mixture Design" Pharmaceutics 17, no. 9: 1137. https://doi.org/10.3390/pharmaceutics17091137
APA StyleBienhaus, M., Neumann, L., Müller, C., & Runkel, F. E. (2025). Targeted Development of an Optimised Formulation for 3D-Printing of a Sertraline Hydrochloride-Containing Drug Delivery System with Immediate-Release Characteristics Utilising a Mixture Design. Pharmaceutics, 17(9), 1137. https://doi.org/10.3390/pharmaceutics17091137




