Plasma-Engineered PDRN: Surface Charge Neutralization and Nanosizing Enhance Uptake and Regeneration Potential
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
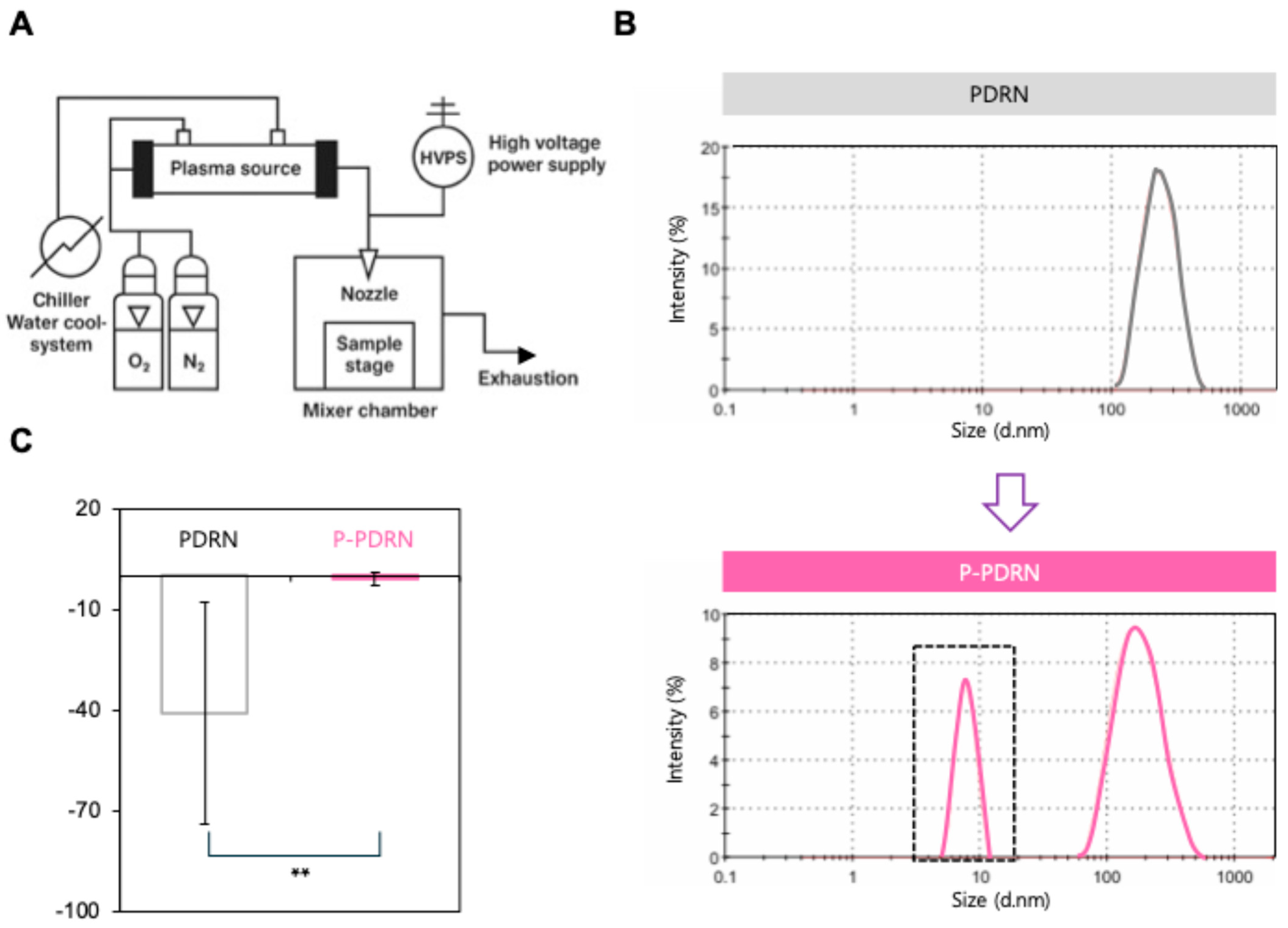
2.2. Plasma Modification of PDRN
2.3. Particle Size
2.4. Zeta Potential Analysis
2.5. Fourier-Transform Infrared (FT-IR) Spectroscopy
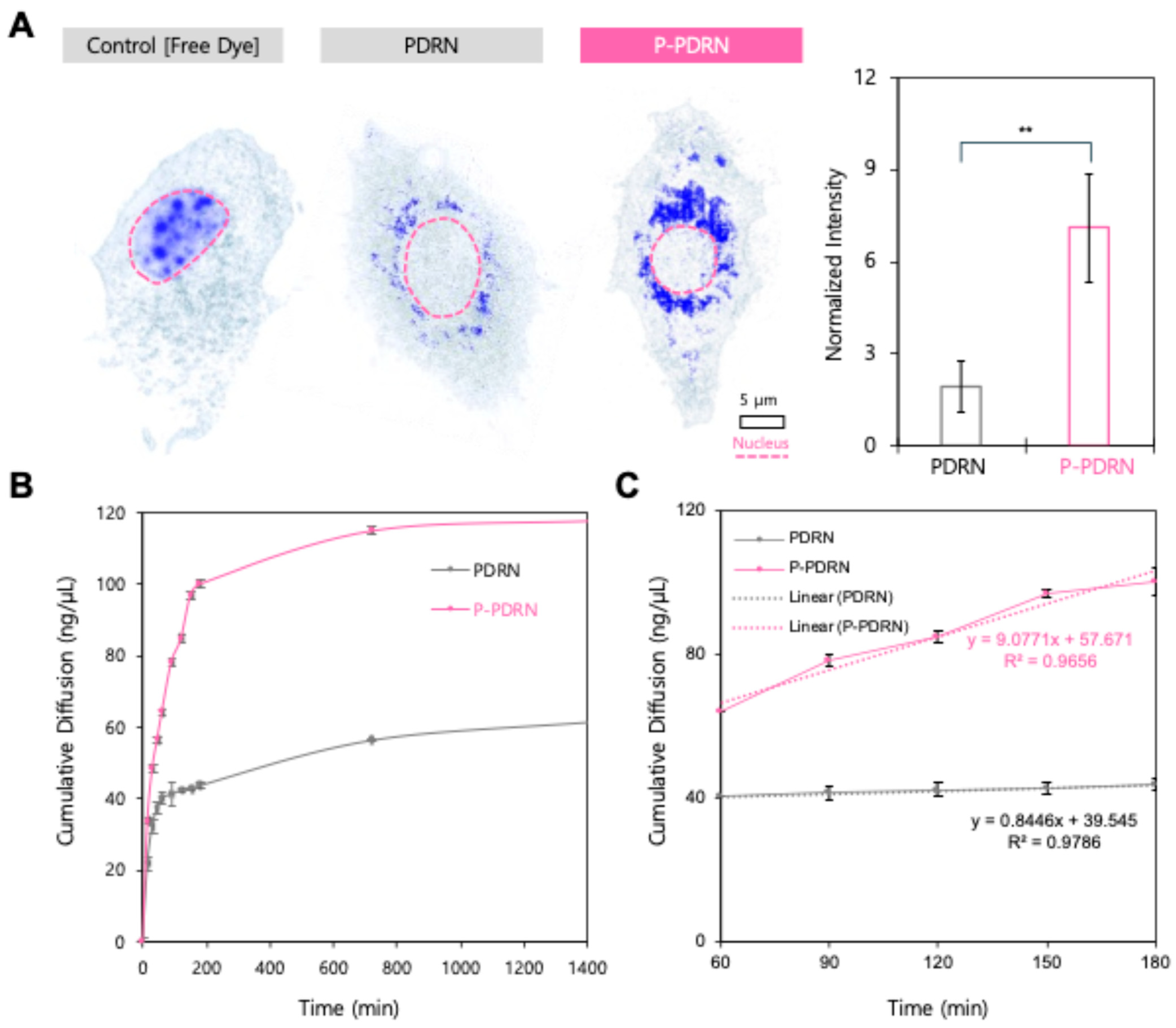
2.6. Cellular Uptake Assay
2.7. In Vitro Skin Permeation Analysis
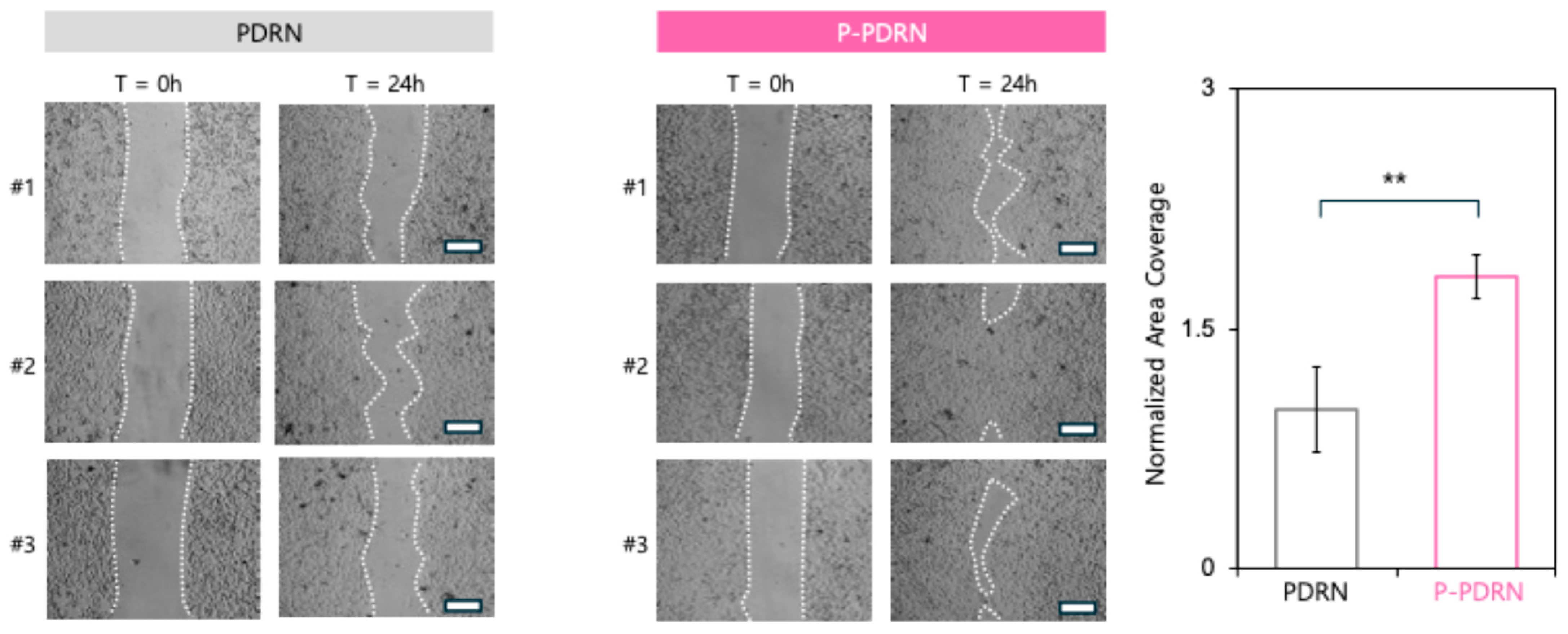
2.8. Cell Migration Assay
2.9. Clinical Evaluation of Cosmetic Formulation Containing P-PDRN
2.10. Statistical Analysis
3. Results
3.1. Plasma-Induced Nanosizing and Surface Charge Modulation of PDRN
3.2. Enhanced Uptake and Permeation of Plasma-Treated PDRN
3.3. Plasma-Treated PDRN Accelerates Fibroblast Migration and Shows Potential as a Skin Regeneration Cosmetic
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| PDRN | Polydeoxyribonucleotide |
| HDFs | Human Dermal Fibroblasts |
| P-PDRN | Plasma-modified PDRN |
| DLS | Dynamic Light Scattering |
| RONS | Reactive oxygen and nitrogen species |
| FT-IR | Fourier-transform infrared |
References
- Picciolo, G.; Mannino, F.; Irrera, N.; Altavilla, D.; Minutoli, L.; Vaccaro, M.; Arcoraci, V.; Squadrito, V.; Picciolo, G.; Squadrito, F. PDRN, a natural bioactive compound, blunts inflammation and positively reprograms healing genes in an “in vitro” model of oral mucositis. Biomed. Pharmacother. 2021, 138, 111538. [Google Scholar] [CrossRef] [PubMed]
- Yi, J.-Y.; Park, S.; Kim, M.; Jeong, Y.; Shin, H.; Cho, Y.; Jeon, M.; Oh, M.-K.; Sung, C. Emerging wound-healing injectable polydeoxyribonucleotide: Potential as a prohibited doping method and its simple detection via CRISPR/Cas12a system. Int. J. Biol. Macromol. 2025, 309, 142999. [Google Scholar] [CrossRef] [PubMed]
- Altavilla, D.; Bitto, A.; Polito, F.; Marini, H.; Minutoli, L.; Stefano, V.D.; Irrera, N.; Cattarini, G.; Squadrito, F. Polydeoxyribonucleotide (PDRN): A safe approach to induce therapeutic angiogenesis in peripheral artery occlusive disease and in diabetic foot ulcers. Cardiovasc. Hematol. Agents Med. Chem. Former. 2009, 7, 313–321. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.; Wang, G.; Zhou, F.; Gong, L.; Zhang, J.; Qi, L.; Cui, H. Polydeoxyribonucleotide: A promising skin anti-aging agent. Chin. J. Plast. Reconstr. Surg. 2022, 4, 187–193. [Google Scholar] [CrossRef]
- Bitto, A.; Polito, F.; Irrera, N.; D’Ascola, A.; Avenoso, A.; Nastasi, G.; Campo, G.M.; Micali, A.; Bagnato, G.; Minutoli, L. Polydeoxyribonucleotide reduces cytokine production and the severity of collagen-induced arthritis by stimulation of adenosine A2A receptor. Arthritis Rheum. 2011, 63, 3364–3371. [Google Scholar] [CrossRef]
- Ko, I.-G.; Jin, J.-J.; Hwang, L.; Kim, S.-H.; Kim, C.-J.; Won, K.Y.; Na, Y.G.; Kim, K.H.; Kim, S.J. Adenosine A2A receptor agonist polydeoxyribonucleotide alleviates interstitial cystitis-induced voiding dysfunction by suppressing inflammation and apoptosis in rats. J. Inflamm. Res. 2021, 14, 367–378. [Google Scholar] [CrossRef] [PubMed]
- Park, G.Y.; Jang, B.-C. Polydeoxyribonucleotide as a Regenerative Agent in Dermatology and Wound Healing: Mechanisms, Clinical Applications, and Safety. Keimyung Med. J. 2025, 44, 9–10. [Google Scholar] [CrossRef]
- Akaberi, S.M.; Sharma, K.; Ahmadi-Ashtiani, H.R.; Hedayati, M. Polydeoxyribonucleotide in Skincare and Cosmetics: Mechanisms, Therapeutic Applications, and Advancements Beyond Wound Healing and Anti-aging. J. Ski. Stem Cell 2025, 12, e159728. [Google Scholar] [CrossRef]
- Noh, T.K.; Chung, B.Y.; Kim, S.Y.; Lee, M.H.; Kim, M.J.; Youn, C.S.; Lee, M.W.; Chang, S.E. Novel anti-melanogenesis properties of polydeoxyribonucleotide, a popular wound healing booster. Int. J. Mol. Sci. 2016, 17, 1448. [Google Scholar] [CrossRef]
- Recalcati, S.; Moltrasio, C.; Nazzaro, G.; Passoni, E.; Gliozzo, J.; Muratori, S.; Berti, E. Effects of Polydeoxyribonucleotide in the treatment of Scleroderma. In Proceedings of the 24th World Congress of Dermatology (WCD 2019), Milan, Italy, 10–15 June 2019. [Google Scholar]
- Squadrito, F.; Bitto, A.; Irrera, N.; Pizzino, G.; Pallio, G.; Minutoli, L.; Altavilla, D. Pharmacological activity and clinical use of PDRN. Front. Pharmacol. 2017, 8, 224. [Google Scholar] [CrossRef]
- Bos, J.D.; Meinardi, M.M. The 500 Dalton rule for the skin penetration of chemical compounds and drugs. Exp. Dermatol. Viewp. 2000, 9, 165–169. [Google Scholar] [CrossRef]
- Mendes, B.B.; Conniot, J.; Avital, A.; Yao, D.; Jiang, X.; Zhou, X.; Sharf-Pauker, N.; Xiao, Y.; Adir, O.; Liang, H. Nanodelivery of nucleic acids. Nat. Rev. Methods Primers 2022, 2, 24. [Google Scholar] [CrossRef]
- Marques, C.; Porcello, A.; Cerrano, M.; Hadjab, F.; Chemali, M.; Lourenço, K.; Hadjab, B.; Raffoul, W.; Applegate, L.A.; Laurent, A.E. From polydeoxyribonucleotides (PDRNs) to polynucleotides (PNs): Bridging the gap between scientific definitions, molecular insights, and clinical applications of multifunctional biomolecules. Biomolecules 2025, 15, 148. [Google Scholar] [CrossRef]
- Braný, D.; Dvorská, D.; Halašová, E.; Škovierová, H. Cold atmospheric plasma: A powerful tool for modern medicine. Int. J. Mol. Sci. 2020, 21, 2932. [Google Scholar] [CrossRef]
- Chen, Z.; Chen, G.; Obenchain, R.; Zhang, R.; Bai, F.; Fang, T.; Wang, H.; Lu, Y.; Wirz, R.E.; Gu, Z. Cold atmospheric plasma delivery for biomedical applications. Mater. Today 2022, 54, 153–188. [Google Scholar] [CrossRef]
- Conrads, H.; Schmidt, M. Plasma generation and plasma sources. Plasma Sources Sci. Technol. 2000, 9, 441. [Google Scholar] [CrossRef]
- Keidar, M. Plasma for cancer treatment. Plasma Sources Sci. Technol. 2015, 24, 033001. [Google Scholar] [CrossRef]
- Laroussi, M. Plasma medicine: A brief introduction. Plasma 2018, 1, 47–60. [Google Scholar] [CrossRef]
- Vasile Nastuta, A.; Pohoata, V.; Topala, I. Atmospheric pressure plasma jet—Living tissue interface: Electrical, optical, and spectral characterization. J. Appl. Phys. 2013, 113, 183302. [Google Scholar] [CrossRef]
- Bhatt, P.; Kumar, V.; Subramaniyan, V.; Nagarajan, K.; Sekar, M.; Chinni, S.V.; Ramachawolran, G. Plasma modification techniques for natural polymer-based drug delivery systems. Pharmaceutics 2023, 15, 2066. [Google Scholar] [CrossRef]
- Oguzlu, H.; Baldelli, A.; Mohammadi, X.; Kong, A.; Bacca, M.; Pratap-Singh, A. Cold Plasma for the Modification of the Surface Roughness of Microparticles. ACS Omega 2024, 9, 35634–35644. [Google Scholar] [CrossRef]
- O’Neill, F.; O’Neill, L.; Bourke, P. Recent developments in the use of plasma in medical applications. Plasma 2024, 7, 284–299. [Google Scholar] [CrossRef]
- Karthik, C.; Sarngadharan, S.C.; Thomas, V. Low-temperature plasma techniques in biomedical applications and therapeutics: An overview. Int. J. Mol. Sci. 2023, 25, 524. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.-C.; Rana, J.N.; Choi, E.H.; Han, I. Improvement of transdermal absorption rate by nonthermal biocompatible atmospheric pressure plasma. Drug Metab. Pharmacokinet. 2024, 54, 100536. [Google Scholar] [CrossRef] [PubMed]
- Worldwide, M.I. Dynamic light scattering, common terms defined. Inf. White Paper. Malwern Instrum. Ltd. 2011, 2011, 1–6. [Google Scholar]
- Labouta, H.I.; El-Khordagui, L.K.; Kraus, T.; Schneider, M. Mechanism and determinants of nanoparticle penetration through human skin. Nanoscale 2011, 3, 4989–4999. [Google Scholar] [CrossRef]
- Alvarez-Román, R.; Naik, A.; Kalia, Y.; Guy, R.H.; Fessi, H. Skin penetration and distribution of polymeric nanoparticles. J. Control. Release 2004, 99, 53–62. [Google Scholar] [CrossRef] [PubMed]
- Sabourian, P.; Yazdani, G.; Ashraf, S.S.; Frounchi, M.; Mashayekhan, S.; Kiani, S.; Kakkar, A. Effect of physico-chemical properties of nanoparticles on their intracellular uptake. Int. J. Mol. Sci. 2020, 21, 8019. [Google Scholar] [CrossRef] [PubMed]
- Cai, X.; Woods, A.; Mesquida, P.; Jones, S. Assessing the Potential for Drug–Nanoparticle Surface Interactions To Improve Drug Penetration into the Skin. Mol. Pharm. 2016, 13, 1375–1384. [Google Scholar] [CrossRef]
- Gupta, R.; Rai, B. Effect of size and surface charge of gold nanoparticles on their skin permeability: A molecular dynamics study. Sci. Rep. 2017, 7, 45292. [Google Scholar] [CrossRef]
- Foroozandeh, P.; Aziz, A.A. Insight into cellular uptake and intracellular trafficking of nanoparticles. Nanoscale Res. Lett. 2018, 13, 339. [Google Scholar] [CrossRef] [PubMed]
- Gimondi, S.; Ferreira, H.; Reis, R.L.; Neves, N.M. Intracellular trafficking of size-tuned nanoparticles for drug delivery. Int. J. Mol. Sci. 2023, 25, 312. [Google Scholar] [CrossRef] [PubMed]
- Sahay, G.; Alakhova, D.Y.; Kabanov, A.V. Endocytosis of nanomedicines. J. Control. Release 2010, 145, 182–195. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Park, S.J.; Lee, D.-H.; Yoon, K.B.; Kim, A.; Jung, C.-Y.; Kim, S.T.; Brito, S.; Bin, B.-H. Plasma-Engineered PDRN: Surface Charge Neutralization and Nanosizing Enhance Uptake and Regeneration Potential. Pharmaceutics 2025, 17, 1136. https://doi.org/10.3390/pharmaceutics17091136
Park SJ, Lee D-H, Yoon KB, Kim A, Jung C-Y, Kim ST, Brito S, Bin B-H. Plasma-Engineered PDRN: Surface Charge Neutralization and Nanosizing Enhance Uptake and Regeneration Potential. Pharmaceutics. 2025; 17(9):1136. https://doi.org/10.3390/pharmaceutics17091136
Chicago/Turabian StylePark, Sun Ju, Dong-Hwan Lee, Ki Bok Yoon, AhJin Kim, Chae-Yun Jung, Sung Tae Kim, Sofia Brito, and Bum-Ho Bin. 2025. "Plasma-Engineered PDRN: Surface Charge Neutralization and Nanosizing Enhance Uptake and Regeneration Potential" Pharmaceutics 17, no. 9: 1136. https://doi.org/10.3390/pharmaceutics17091136
APA StylePark, S. J., Lee, D.-H., Yoon, K. B., Kim, A., Jung, C.-Y., Kim, S. T., Brito, S., & Bin, B.-H. (2025). Plasma-Engineered PDRN: Surface Charge Neutralization and Nanosizing Enhance Uptake and Regeneration Potential. Pharmaceutics, 17(9), 1136. https://doi.org/10.3390/pharmaceutics17091136






