Peptide-Based Inorganic Nanoparticles as Efficient Intracellular Delivery Systems
Abstract
1. Introduction
2. Key Physicochemical and Biological Properties of MNPs
2.1. Size of MNPs
2.2. Surface Functionalization
2.3. Surface Charge
2.4. Toxicity Considerations
3. Significance of Combining Peptides with MNPs for Different Applications
4. Peptides in Peptide-Based Inorganic Nanoparticles (PINP) Technology
4.1. Types of PINPs Used According to the Peptide Structure
4.1.1. Linear Peptide Use in PINPs
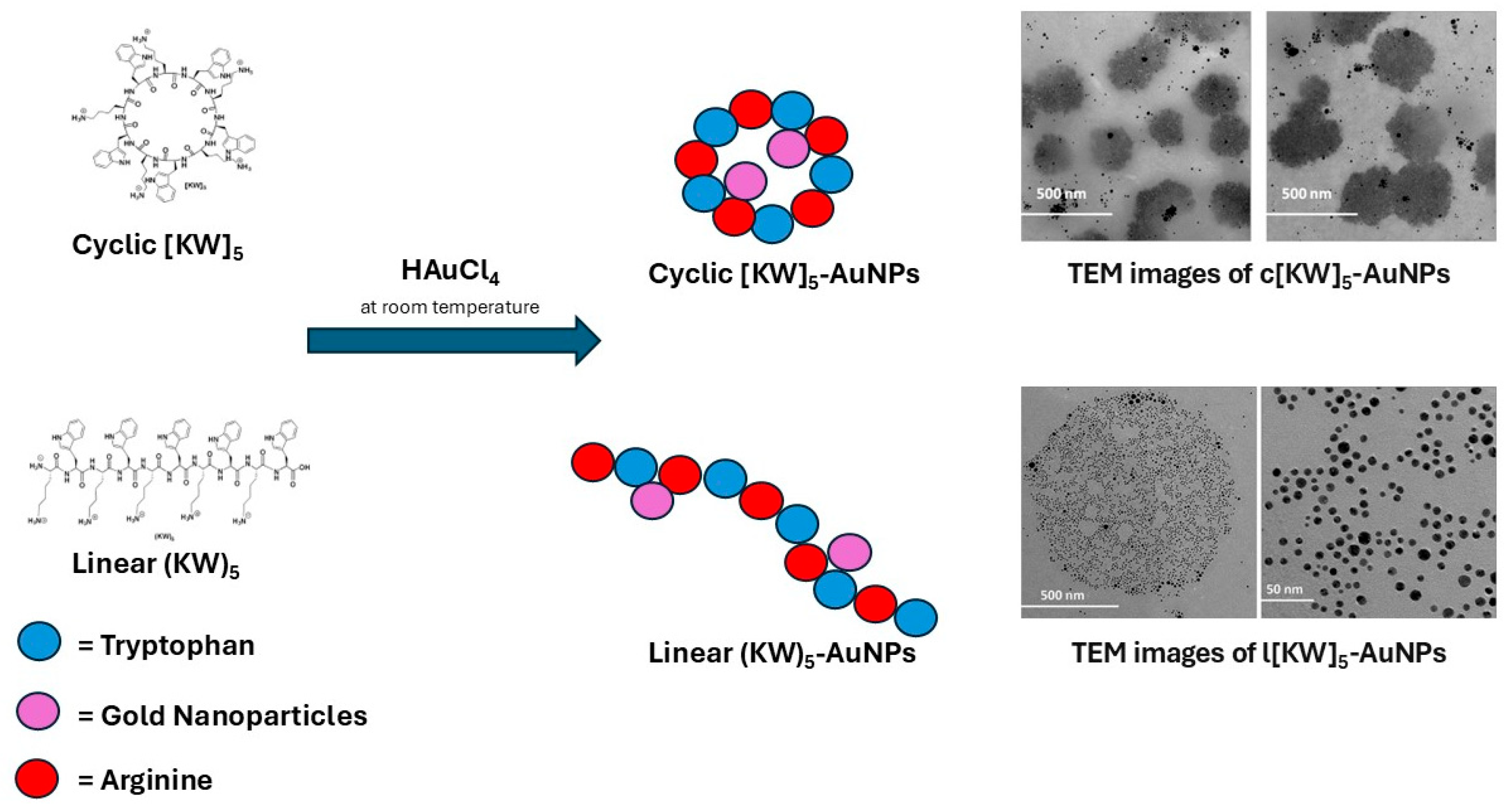
4.1.2. Cyclic Peptide Use in PINPs
4.1.3. Amphipathic Peptide Use in PINPs
4.1.4. Self-Assembling Peptide Use in PINPs
4.2. Classification of Peptides in PINPs
4.2.1. Targeting Ligands
4.2.2. Capping and Stabilizing Agents
4.2.3. Reducing Agents
5. Synthesis of PINPs
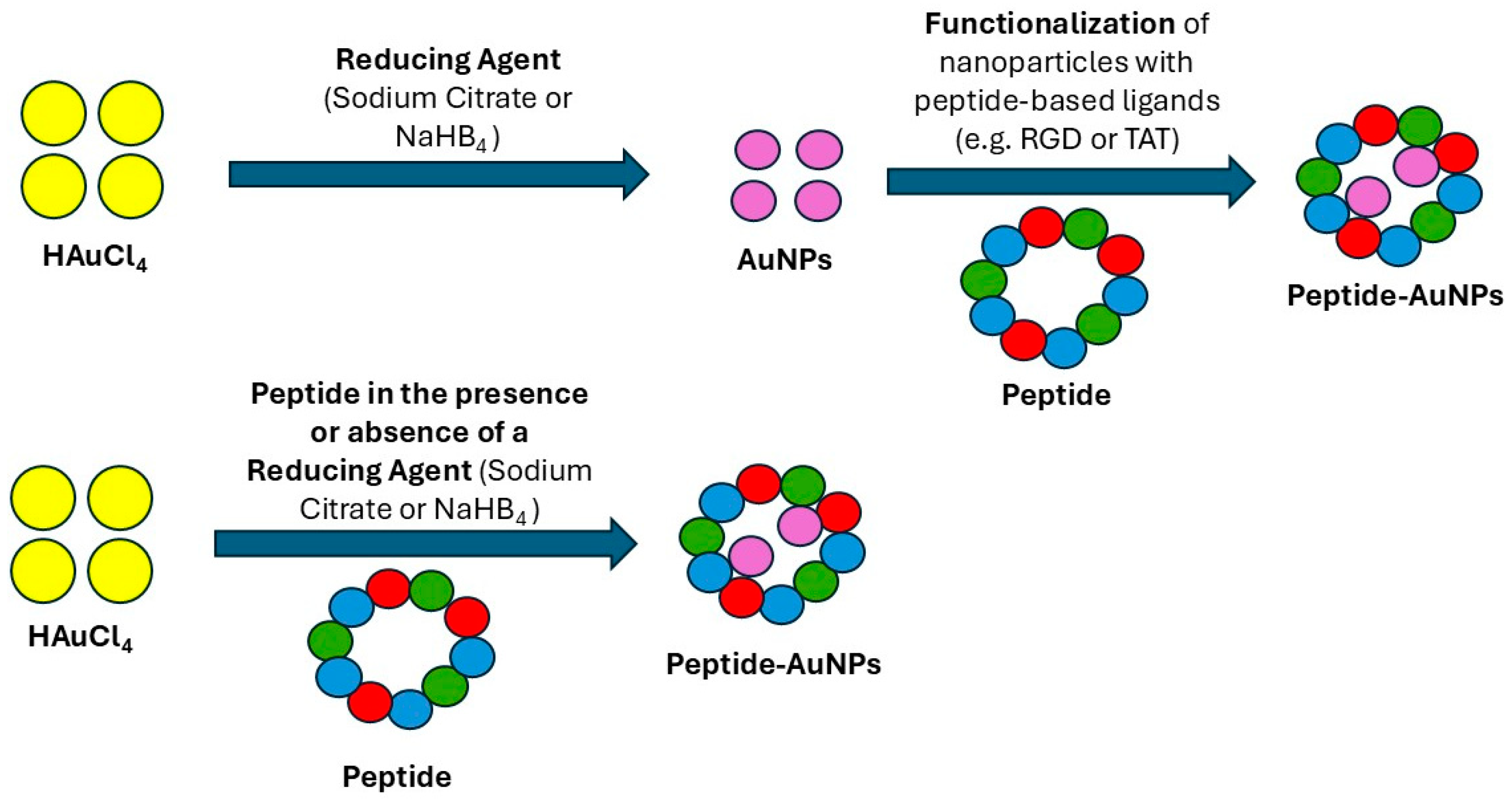
5.1. Peptide-Mediated Synthesis
5.1.1. One-Pot Green Synthesis
5.1.2. Reducing and Stabilizing Activities of Peptides in the Synthesis of PINPs
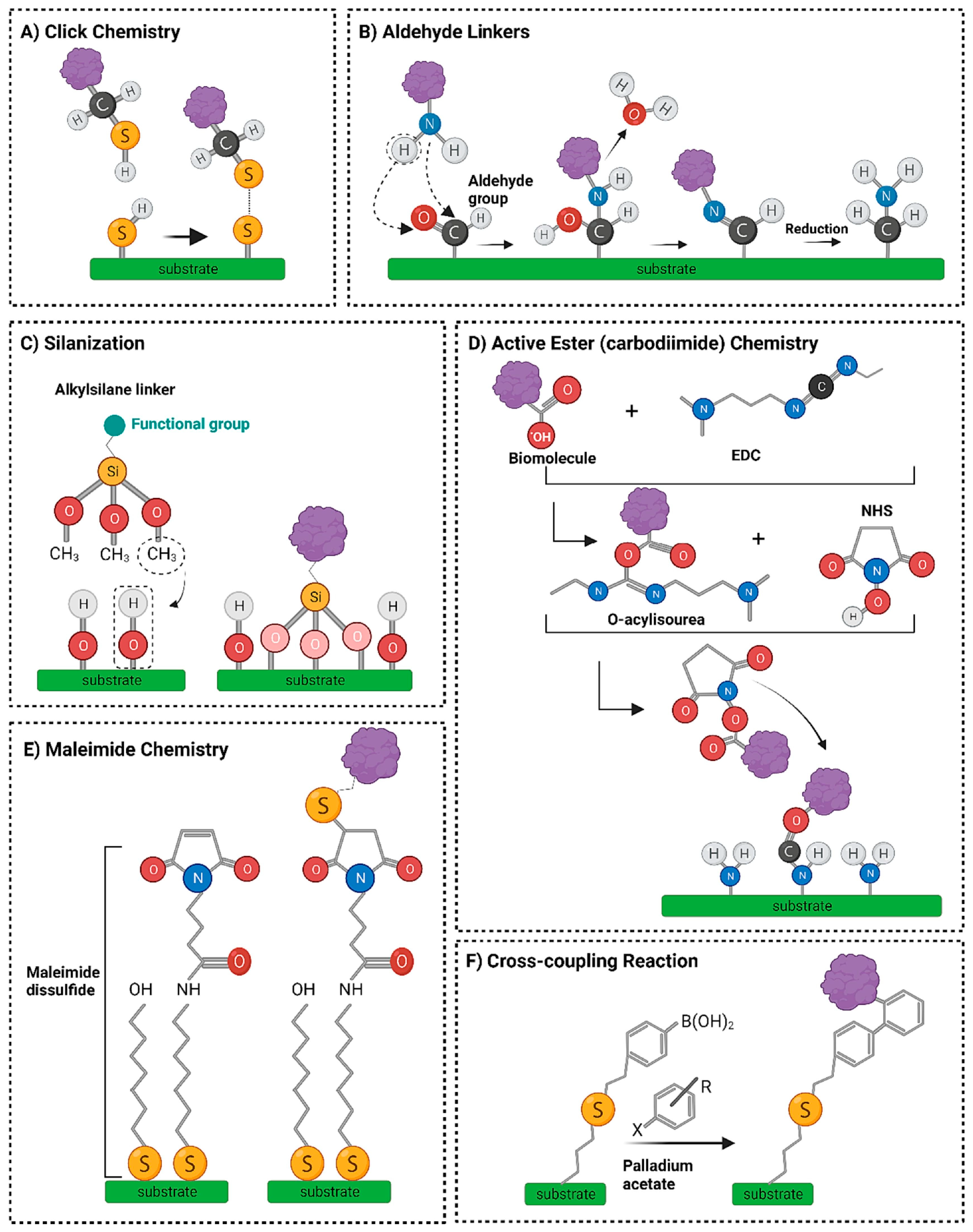
5.1.3. Conjugation Methods Through Surface Functionalization and Self-Assembly
- (a)
- Thiol–Gold Chemistry: Peptides containing cysteine residues are directly conjugated to AuNP surfaces via strong Au–S bonds, forming stable monolayers. This method is widely used due to its simplicity and high binding affinity [86].
- (b)
- EDC/NHS Coupling: Carboxyl-functionalized nanoparticles can be activated using 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide (EDC) and N-hydroxysuccinimide (NHS) (Figure 1) to form an active ester intermediate that reacts with amine groups on peptides, forming stable amide bonds. This allows for site-specific attachment of peptides without disrupting their bioactivity [87].
- (c)
- (d)
- Physical Adsorption: Electrostatic or hydrophobic interactions may also drive non-covalent peptide adsorption onto nanoparticle surfaces. While simpler, this method offers weaker binding and may suffer from desorption under physiological conditions [25].
6. Drug Loading Capacity and Surface Area
7. Pharmacokinetics: Clearance and Circulation Time
7.1. Size-Dependent Clearance Mechanisms
7.2. Cyclic Nature Properties
7.3. Size-Dependent Toxicity
7.4. Incorporation of Non-Natural Amino Acids into the Structure of Peptides
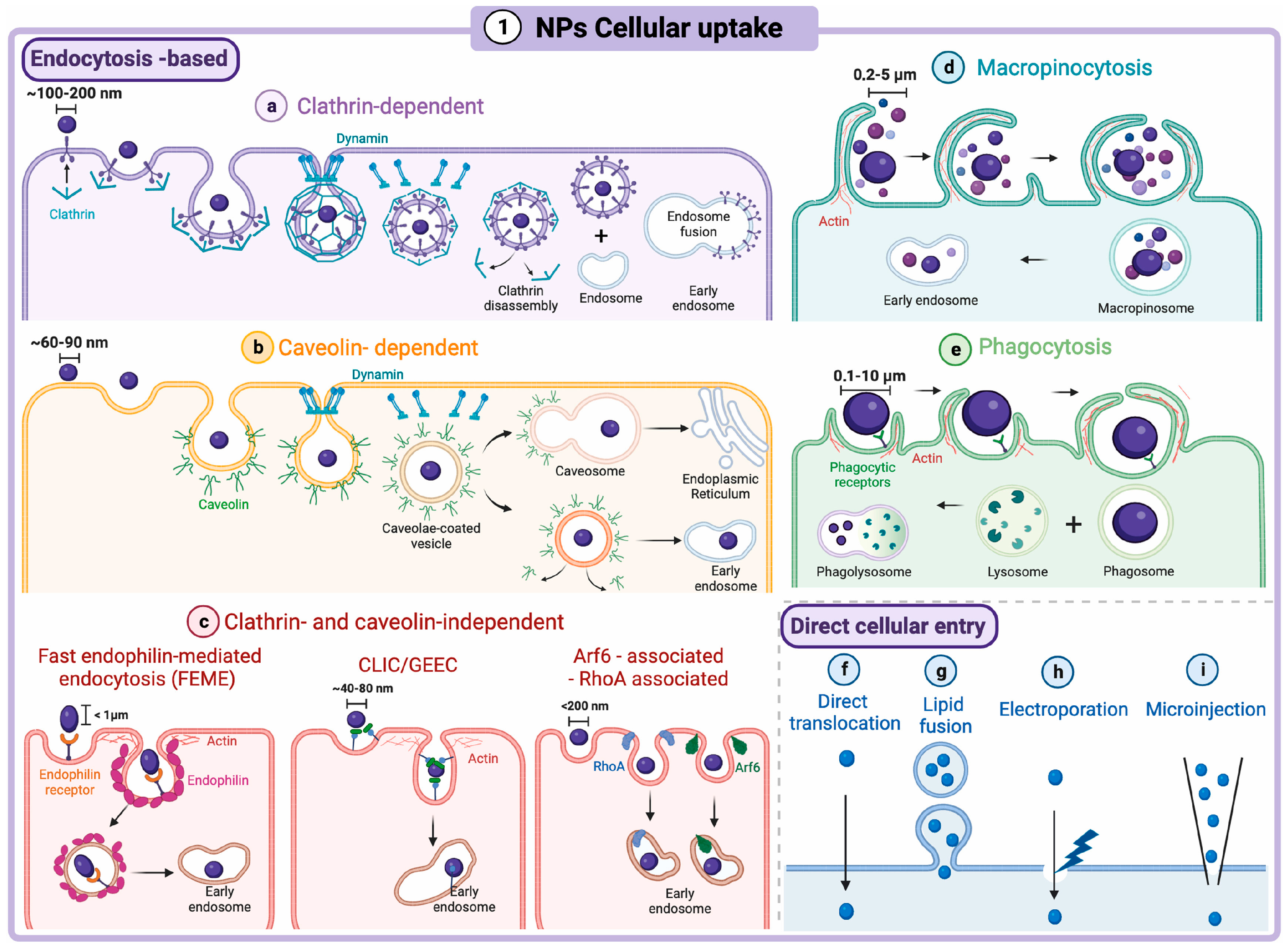
8. Cellular Uptake Mechanisms of Peptide-INPs (PINPs)
9. Delivery of Drugs Using PINPs
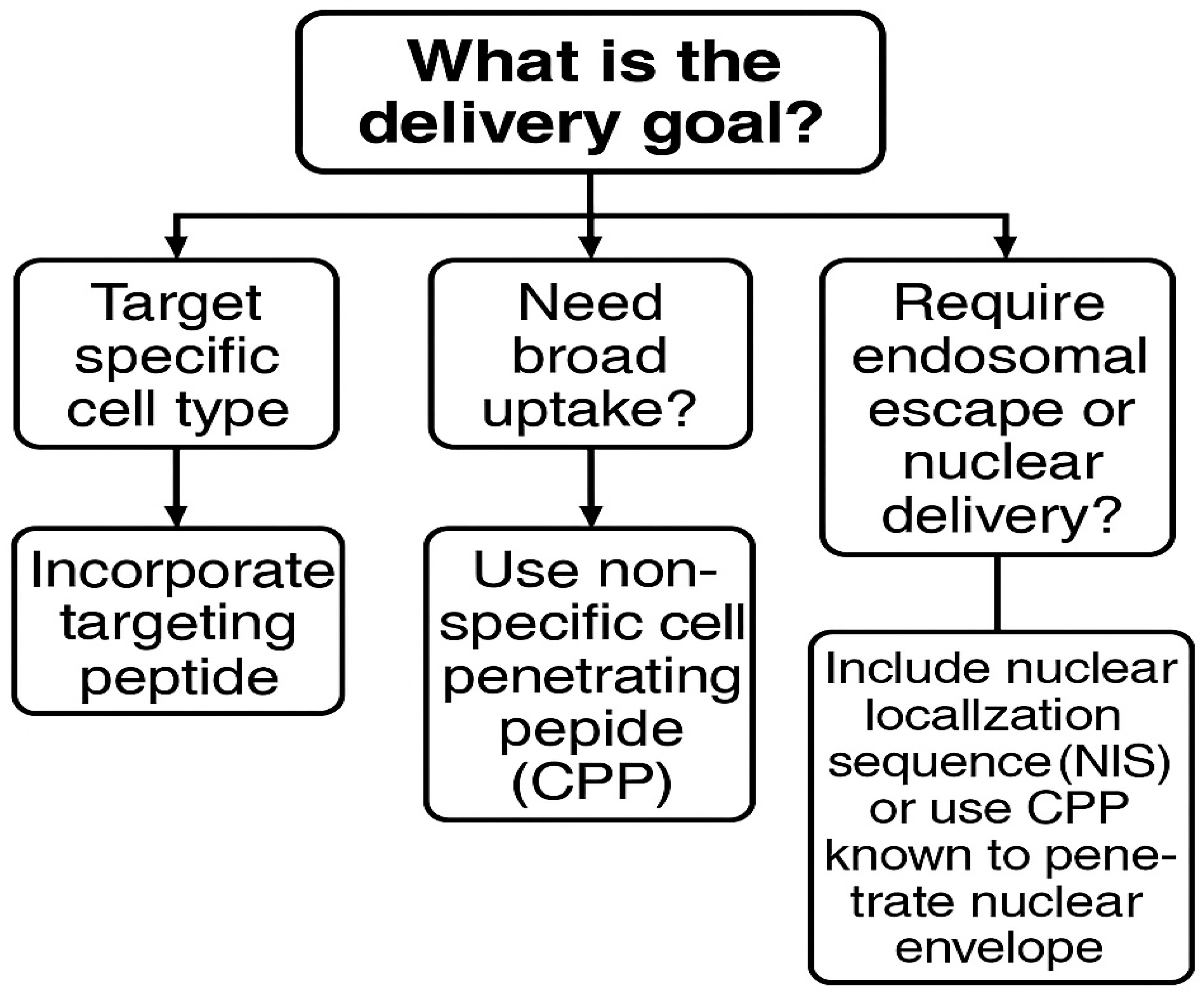
9.1. Targeted Delivery
9.2. Stimuli-Responsive Systems
9.2.1. pH-Responsive Systems
9.2.2. Enzyme-Responsive Systems
9.2.3. Redox-Responsive Systems
9.2.4. Light and Heat Responsiveness
9.2.5. Magnetic Responsiveness
10. Advantages of PINPs
10.1. Biocompatibility and Biodegradability
10.2. Specific Targeting Capabilities
10.3. Versatile Functionalization
11. Challenges and Limitations
11.1. Stability in Biological Environments
11.2. Immunogenicity and Toxicity Concerns
11.3. Scalability and Reproducibility
11.4. Regulatory and Translational Barriers
11.5. In Vivo vs. In Vitro Outcomes
12. Design Rules for Next-Generation PINPs
13. Future Perspectives
14. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| PINPs | Peptide-based inorganic nanoparticles |
| AuNPs | Gold nanoparticles |
| SeNPs | Selenium nanoparticles |
| GdNPs | Gadolinium nanoparticles |
References
- Khan, I.; Saeed, K.; Khan, I. Nanoparticles: Properties, Applications and Toxicities. Arab. J. Chem. 2019, 12, 908–931. [Google Scholar] [CrossRef]
- Roco, M.C. The Long View of Nanotechnology Development: The National Nanotechnology Initiative at 10 Years. J. Nanopart. Res. 2011, 13, 427–445. [Google Scholar] [CrossRef]
- Peer, D.; Karp, J.M.; Hong, S.; Farokhzad, O.C.; Margalit, R.; Langer, R. Nanocarriers as an emerging platform for cancer therapy. Nat. Nanotechnol. 2007, 2, 751–760. [Google Scholar] [CrossRef] [PubMed]
- Fang, J.; Nakamura, H.; Maeda, H. The EPR Effect: Unique Features of Tumor Blood Vessels for Drug Delivery, Factors Involved, and Limitations and Augmentation of the Effect. Adv. Drug Deliv. Rev. 2011, 63, 136–151. [Google Scholar] [CrossRef] [PubMed]
- Iravani, S.; Korbekandi, H.; Mirmohammadi, S.V.; Zolfaghari, B. Synthesis of Silver Nanoparticles: Chemical, Physical and Biological Methods. Res. Pharm. Sci. 2014, 9, 385–406. [Google Scholar]
- Dykman, L.A.; Khlebtsov, N.G. Gold Nanoparticles in Biomedical Applications: Recent Advances and Perspectives. Chem. Soc. Rev. 2012, 41, 2256–2282. [Google Scholar] [CrossRef]
- Liu, D.; Lin, B.; Shao, W.; Zhu, Y. Targeted Cancer Therapy Using Peptide-Functionalized Nanoformulations: Recent Advances and Future Perspectives. Acta Pharm. Sin. B 2021, 11, 2265–2285. [Google Scholar]
- Rai, M.; Yadav, A.; Gade, A. Silver Nanoparticles as a New Generation of Antimicrobials. Biotechnol. Adv. 2009, 27, 76–83. [Google Scholar] [CrossRef]
- Gupta, A.K.; Gupta, M. Synthesis and Surface Engineering of Iron Oxide Nanoparticles for Biomedical Applications. Biomaterials 2005, 26, 3995–4021. [Google Scholar] [CrossRef]
- Genc, S.; Taghizadehghalehjoughi, A.; Yeni, Y.; Jafarizad, A.; Hacimuftuoglu, A.; Nikitovic, D.; Docea, A.O.; Mezhuev, Y.; Tsatsakis, A. Fe3O4 Nanoparticles in Combination with 5-FU Exert Antitumor Effects Superior to Those of the Active Drug in a Colon Cancer Cell Model. Pharmaceutics 2023, 15, 245. [Google Scholar] [CrossRef]
- Premanathan, M.; Karthikeyan, K.; Jeyasubramanian, K.; Manivannan, G. Selective Toxicity of ZnO Nanoparticles toward Gram-Positive Bacteria and Cancer Cells by Apoptosis through Lipid Peroxidation. Nanomedicine 2011, 7, 184–192. [Google Scholar] [CrossRef]
- Huang, Y.; He, L.; Liu, W.; Fan, C.; Zheng, W.; Wong, Y.S.; Chen, T. Selective Cellular Uptake and Induction of Apoptosis of Cancer-Targeted Selenium Nanoparticles. Biomaterials 2013, 34, 7106–7116. [Google Scholar] [CrossRef]
- Wang, H.; Liu, Y.; Zhang, M.; Wang, J.; Wu, Z.; Li, L. Gadolinium-Based Nanoparticles for Theranostic Applications: A Review. ACS Appl. Nano Mater. 2021, 4, 4673–4691. [Google Scholar]
- Kumar, S.; Singh, B.; Mehta, M.; Satija, S.; Gupta, M.; Chellappan, D.K.; Dua, K. Photothermal Effect of Gold Nanoparticles as a Nanomedicine for Diagnostic and Therapeutic Applications. Pharmaceutics 2023, 15, 2349. [Google Scholar] [CrossRef]
- Fahim, M.; Shahzaib, A.; Nishat, N.; Jahan, A.; Bhat, T.A.; Inam, A. Green Synthesis of Silver Nanoparticles: A Comprehensive Review of Methods, Influencing Factors, and Applications. JCIS Open 2024, 7, 100321. [Google Scholar] [CrossRef]
- Rahman, M. Magnetic Resonance Imaging and Iron-oxide Nanoparticles in the Era of Personalized Medicine. Nanotheranostics 2023, 7, 424–449. [Google Scholar] [CrossRef] [PubMed]
- Al-Shehaby, N.; Elshoky, H.A.; Zidan, M.; Salaheldin, T.A.; Gaber, M.H.; Ali, M.A.; El-Sayed, N.M. In Vitro Localization of Modified Zinc Oxide Nanoparticles Showing Selective Anticancer Effects against Colorectal Carcinoma Using Biophysical Techniques. Sci. Rep. 2025, 15, 16811. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Yang, F.; Fan, S.; Jin, H.; Liao, K.; Li, X.; Liu, G.-B.; Liang, J.; Zhang, J.; Xu, J.-F.; et al. Immunomodulatory Roles of Selenium Nanoparticles: Novel Arts for Potential Immunotherapy Strategy Development. Front. Immunol. 2022, 13, 956181. [Google Scholar] [CrossRef]
- Kouri, M.A.; Polychronidou, K.; Loukas, G.; Megapanou, A.; Vagena, I.-A.; Gerardos, A.M.; Spyratou, E.; Eftsathopoulos, E.P. Consolidation of Gold and Gadolinium Nanoparticles: An Extra Step towards Improving Cancer Imaging and Therapy. Nanomaterials 2023, 13, 357. [Google Scholar]
- Conde, J.; Doria, G.; Baptista, P. Noble Metal Nanoparticles Applications in Cancer. Pharmaceutics 2014, 6, 929–954. [Google Scholar] [CrossRef]
- Boisselier, E.; Astruc, D. Gold nanoparticles in nanomedicine: Preparations, imaging, diagnostics, therapies and toxicity. Chem. Soc. Rev. 2009, 38, 1759–1782. [Google Scholar] [CrossRef]
- Kaymaz, S.V.; Nobar, H.M.; Sarıgül, H.; Soylukan, C.; Akyüz, L.; Yüce, M. Nanomaterial surface modification toolkit: Principles, components, recipes, and applications. Adv. Colloid Interface Sci. 2023, 322, 103035. [Google Scholar] [CrossRef]
- Balog, S.; de Almeida, M.S.; Taladriz-Blanco, P.; Rothen-Rutishauser, B.; Petri-Fink, A. Does the Surface Charge of the Nanoparticles Drive Nanoparticle–Cell Membrane Interactions? Curr. Opin. Biotechnol. 2024, 87, 103128. [Google Scholar] [CrossRef]
- Albanese, A.; Tang, P.S.; Chan, W.C.W. The Effect of Nanoparticle Size, Shape, and Surface Chemistry on Biological Systems. Annu. Rev. Biomed. Eng. 2012, 14, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Nasrolahi Shirazi, A.; Tiwari, R.K.; Oh, D.; Sullivan, B.; McCaffrey, K.; Mandal, D.; Parang, K. Surface Decorated Gold Nanoparticles by Linear and Cyclic Peptides as Molecular Transporters. Mol. Pharm. 2013, 10, 3137–3146. [Google Scholar] [CrossRef] [PubMed]
- Nasrolahi Shirazi, A.; Mandal, D.; Tiwari, R.K.; Guo, L.; Lu, W.; Parang, K. Cyclic Peptide-Capped Gold Nanoparticles as Drug Delivery Systems. Mol. Pharm. 2013, 10, 500–508. [Google Scholar] [CrossRef]
- Nasrolahi Shirazi, A.; Tiwari, R.; Oh, D.; Sullivan, B.; Kumar, A.; Beni, Y.; Parang, K. Cyclic Peptide-Selenium Nanoparticles as Drug Transporters. Mol. Pharm. 2014, 11, 3631–3640. [Google Scholar] [CrossRef] [PubMed]
- Nasrolahi Shirazi, A.; Park, S.E.; Rad, S.; Baloyan, L.; Mandal, D.; Sajid, M.I.; Hall, R.; Lohan, S.; Zoghebi, K.; Parang, K.; et al. Cyclic Peptide-Gadolinium Particles for Enhanced Intracellular Delivery. Pharmaceutics 2020, 12, 792. [Google Scholar] [CrossRef]
- Tseng, Y.-L.; Liu, J.-J.; Hong, R.-L. Translocation of liposomes into cancer cells by cell-penetrating peptides penetratin and TAT: A kinetic and efficacy study. Mol. Pharmacol. 2002, 62, 864–872. [Google Scholar] [CrossRef]
- Mandal, D.; Nasrolahi Shirazi, A.; Parang, K. Self-Assembly of Peptides to Nanostructures. Org. Biomol. Chem. 2013, 11, 3544–3551. [Google Scholar] [CrossRef]
- Bechara, C.; Sagan, S. Cell-penetrating peptides: 20 years later, where do we stand? FEBS Lett. 2013, 587, 1693–1702. [Google Scholar] [CrossRef]
- Nasrolahi Shirazi, A.; Mandal, D.; Sajid, M.I.; Stickley, D.; Nagasawa, S.; Long, J.; Parang, K.; Tiwari, R.K. Cyclic Peptide-Gadolinium Nanocomplexes as siRNA Delivery Tools. Pharmaceuticals 2021, 14, 1064. [Google Scholar] [CrossRef] [PubMed]
- Nasrolahi Shirazi, A.; Neira, K.; Howlett, N.; Parang, K. Cyclic Peptide-Capped Gold Nanoparticles for Enhanced siRNA Delivery. Molecules 2014, 19, 13319–13331. [Google Scholar] [CrossRef] [PubMed]
- Binaymotlagh, R.; Del Giudice, A.; Mignardi, S.; Amato, F.; Marrani, A.G.; Sivori, F.; Cavallo, I.; Di Domenico, E.G.; Palocci, C.; Chronopoulou, L. Green In Situ Synthesis of Silver Nanoparticles-Peptide Hydrogel Composites: Investigation of Their Antibacterial Activities. Gels 2022, 8, 700. [Google Scholar] [CrossRef]
- Yin, H.-Q.; Mai, D.-S.; Gan, F.; Chen, X.-J. One-step synthesis of linear and cyclic RGD conjugated gold nanoparticles for tumour targeting and imaging. RSC Adv. 2014, 4, 9078–9085. [Google Scholar] [CrossRef]
- Silva, S.; Almeida, A.J.; Vale, N. Combination of Cell-Penetrating Peptides with Nanoparticles for Therapeutic Application: A Review. Biomolecules 2019, 9, 22. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Trinh, T.H.T.; Yoo, M.; Shin, J.; Lee, H.; Kim, J.; Hwang, E.; Lim, Y.-B.; Ryou, C. Self-Assembling Peptides and Their Application in the Treatment of Diseases. Int. J. Mol. Sci. 2019, 20, 5850. [Google Scholar] [CrossRef]
- Zappavigna, S.; Misso, G.; Falanga, A.; Perillo, E.; Novellino, E.; Galdiero, M.; Grieco, P.; Caraglia, M.; Galdiero, S. Nanocarriers Conjugated with Cell Penetrating Peptides: New Trojan Horses by Modern Ulysses. Curr. Pharm. Biotechnol. 2016, 17, 700–722. [Google Scholar] [CrossRef]
- Martian, P.C.; Tertis, M.; Leonte, D.; Hadade, N.; Cristea, C.; Crisan, O. Cyclic Peptides: A Powerful Instrument for Advancing Biomedical Nanotechnologies and Drug Development. J. Pharm. Biomed. Anal. 2025, 252, 116488. [Google Scholar] [CrossRef]
- Mandal, D.; Nasrolahi Shirazi, A.; Parang, K. Cell-Penetrating Homochiral Cyclic Peptides as Nuclear-Targeting Molecular Transporters. Angew. Chem. Int. Ed. 2011, 50, 9633–9637. [Google Scholar] [CrossRef]
- Bajiya, N.; Najrin, S.; Kumar, P.; Choudhury, S.; Tomer, R.; Raghava, G.P.S. CPPsite3: An Updated Large Repository of Experimentally Validated Cell-Penetrating Peptides. Drug Discov. Today 2025, 30, 104421. [Google Scholar] [CrossRef]
- Qian, Z.; LaRochelle, J.R.; Jiang, B.; Lian, W.; Hard, R.L.; Selner, N.G.; Luechapanichkul, R.; Barrios, A.M.; Pei, D. Early Endosomal Escape of a Cyclic Cell-Penetrating Peptide Allows Effective Cytosolic Cargo Delivery. Biochemistry 2014, 53, 4034–4046. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Ma, H.; Zhang, X.; Huang, K.; Jin, S.; Liu, J.; Wei, T.; Cao, W.; Zou, G.; Liang, X.J. Gold nanoparticles functionalized with therapeutic and targeted peptides for cancer treatment. Biomaterials 2012, 33, 1180–1189. [Google Scholar] [CrossRef] [PubMed]
- Lei, H.; Pei, Z.; Jiang, C.; Cheng, L. Recent progress of metal-based nanomaterials with anti-tumor biological effects for enhanced cancer therapy. Exploration 2023, 3, 20220001. [Google Scholar] [CrossRef] [PubMed]
- Ruks, T.; Loza, K.; Heggen, M.; Prymak, O.; Sehnem, A.L.; Oliveira, C.L.P.; Bayer, P.; Beuck, C.; Epple, M. Peptide-Conjugated Ultrasmall Gold Nanoparticles (2 nm) for Selective Protein Targeting. ACS Appl. Bio Mater. 2021, 4, 945–965. [Google Scholar] [CrossRef]
- Orres-Díaz, M.; Abreu-Takemura, C.; Díaz-Vázquez, L.M. Microalgae Peptide-Stabilized Gold Nanoparticles as a Versatile Material for Biomedical Applications. Life 2022, 12, 831. [Google Scholar] [CrossRef]
- Tiwari, P.M.; Eroglu, E.; Bawage, S.S.; Vig, K.; Miller, M.E.; Pillai, S.; Dennis, V.A.; Singh, S.R. Enhanced Intracellular Translocation and Biodistribution of Gold Nanoparticles Functionalized with a Cell-Penetrating Peptide (VG-21) from Vesicular Stomatitis Virus. Biomaterials 2014, 35, 9484–9494. [Google Scholar] [CrossRef]
- Turkevich, J.; Stevenson, P.C.; Hillier, J. A Study of the Nucleation and Growth Processes in the Synthesis of Colloidal Gold. Discuss. Faraday Soc. 1951, 11, 55–75. [Google Scholar] [CrossRef]
- Thomas, R.G.; Surendran, S.P.; Jeong, Y.Y. Tumor Microenvironment-Stimuli Responsive Nanoparticles for Anticancer Therapy. Front. Mol. Biosci. 2020, 7, 610533. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.; Jia, Y.; Liu, Y.; Chen, Y.; Zhao, P. Tumor Microenvironment-Based Stimuli-Responsive Nanoparticles for Controlled Release of Drugs in Cancer Therapy. Pharmaceutics 2022, 14, 2346. [Google Scholar] [CrossRef]
- Scarì, G.; Porta, F.; Fascio, U.; Avvakumova, S.; Dal Santo, V.; De Simone, M.; Saviano, M.; Leone, M.; Del Gatto, A.; Pedone, C.; et al. Gold Nanoparticles Capped by a GC-Containing Peptide Functionalized with an RGD Motif for Integrin Targeting. Bioconjug. Chem. 2012, 23, 340–349. [Google Scholar] [CrossRef]
- Davani-Davari, D.; Tiwari, R.K.; Parang, K. Future Applications of Cyclic Antimicrobial Peptides in Drug Delivery. Expert Opin. Drug Deliv. 2025, 2, 383–404. [Google Scholar] [CrossRef]
- Nasrolahi Shirazi, A.; Tiwari, R.; Chhikara, B.S.; Mandal, D.; Parang, K. Design and Biological Evaluation of Cell-Penetrating Peptide–Doxorubicin Conjugates as Prodrugs. Mol. Pharmaceutics 2013, 10, 488–499. [Google Scholar] [CrossRef]
- Lee, N.K.; Choi, J.U.; Kim, H.R.; Chung, S.W.; Ko, Y.G.; Cho, Y.S.; Park, S.J.; Lee, E.J.; Kim, S.Y.; Kim, I.-S.; et al. Caspase-cleavable peptide–doxorubicin conjugate in combination with CD47-antagonizing nanocage therapeutics for immune-mediated elimination of colorectal cancer. Pharmaceutics 2021, 277, 121105. [Google Scholar]
- Behzadi, S.; Serpooshan, V.; Tao, W.; Hamaly, M.A.; Alkawareek, M.Y.; Dreaden, E.C.; Brown, D.; Alkilany, A.M.; Farokhzad, O.C.; Mahmoudi, M. Cellular uptake of nanoparticles: Journey inside the cell. Chem. Soc. Rev. 2017, 46, 4218–4244. [Google Scholar] [CrossRef]
- Dutta, A.K.; Dutta, N.K. Protein, Biomimetic Protein, and Designer Peptide-Directed Synthesis of Metal Nanoparticles, Metal Nanoclusters and Nanobioconjugates, and Their Potential Applications. In Biomimetic Protein Based Elastomers: Emerging Materials for the Future; Choudhury, N.R., Liu, J.C., Dutta, N.K., Eds.; The Royal Society of Chemistry: Cambridge, UK, 2022; pp. 306–366. [Google Scholar]
- Tiwari, P.M.; Vig, K.; Dennis, V.A.; Singh, S.R. Functionalized Gold Nanoparticles and Their Biomedical Applications. Nanomaterials 2011, 1, 31–63. [Google Scholar] [CrossRef]
- Li, Y.; Tang, Z.; Prasad, P.N.; Knecht, M.R.; Swihart, M.T. Peptide-mediated synthesis of gold nanoparticles: Effects of peptide sequence and nature of binding on physicochemical properties. Nanoscale 2014, 6, 3165–3172. [Google Scholar] [CrossRef]
- Figat, A.M.; Bartosewicz, B.; Liszewska, M.; Budner, B.; Norek, M.; Jankiewicz, B.J. α-Amino Acids as Reducing and Capping Agents in Gold Nanoparticles Synthesis Using the Turkevich Method. Langmuir 2023, 39, 8646–8657. [Google Scholar] [CrossRef]
- Maruyama, T.; Fujimoto, Y.; Maekawa, T. Synthesis of gold nanoparticles using various amino acids. J. Colloid Interface Sci. 2015, 447, 254–257. [Google Scholar] [CrossRef]
- Reches, M.; Gazit, E. Casting metal nanowires within discrete self-assembled peptide nanotubes. Science 2003, 300, 625–627. [Google Scholar] [CrossRef]
- Cardoso-Avila, P.E.; Patakfalvi, R.; Rodríguez-Pedroza, C.; Aparicio-Fernández, X.; Loza-Cornejo, S.; Villa-Cruz, V.; Martínez-Cano, E. One-pot green synthesis of gold and silver nanoparticles using Rosa canina L. extract. RSC Adv. 2021, 11, 14624–14631. [Google Scholar] [CrossRef]
- Suarasan, S.; Focsan, M.; Soritau, O.; Maniu, D.; Astilean, S. One-pot, green synthesis of gold nanoparticles by gelatin and investigation of their biological effects on osteoblast cells. Colloids Surf. B Biointerfaces 2015, 132, 122–131. [Google Scholar] [CrossRef]
- Sasidharan, S.; Bahadur, D.; Srivastava, R. Rapid, One-Pot, Protein-Mediated Green Synthesis of Gold Nanostars for Computed Tomographic Imaging and Photothermal Therapy of Cancer. ACS Sustain. Chem. Eng. 2017, 5, 10163–10175. [Google Scholar] [CrossRef]
- Patel, R.N.; Kumar, R.; Khan, R. Peptide-capped metal nanoparticles: Green synthesis, characterization, and biomedical applications. J. Mater. Chem. B 2019, 7, 5946–5961. [Google Scholar]
- Kim, J.; Rheem, Y.; Yoo, B.; Chong, Y.; Bozhilov, K.N.; Kim, D.; Sadowsky, M.J.; Hur, H.G.; Myung, N.V. Peptide-mediated shape- and size-tunable synthesis of gold nanostructures. Acta Biomater. 2010, 6, 2681–2689. [Google Scholar] [CrossRef]
- Thongpon, P.; Tang, M.; Cong, Z. Peptide-Based Nanoparticle for Tumor Therapy. Biomedicines 2025, 13, 1415. [Google Scholar] [CrossRef]
- Cheng, Y.; Dai, Q.; Morshed, R.A.; Fan, X.; Wegscheid, M.L.; Wainwright, D.A.; Han, Y.; Zhang, L.; Auffinger, B.; Tobias, A.L.; et al. Blood-brain barrier permeable gold nanoparticles: An efficient delivery platform for enhanced malignant glioma therapy and imaging. Small 2014, 10, 5137–5150. [Google Scholar] [CrossRef]
- Wu, P.H.; Onodera, Y.; Ichikawa, Y.; Rankin, E.B.; Giaccia, A.J.; Watanabe, Y.; Qian, W.; Hashimoto, T.; Shirato, H.; Nam, J.M. Targeting integrins with RGD-conjugated gold nanoparticles in radiotherapy decreases the invasive activity of breast cancer cells. Int. J. Nanomed. 2017, 12, 5069–5085. [Google Scholar] [CrossRef]
- Zhang, Y.; Huang, F.; Ren, C.; Yang, J.; Wang, Y.; Wu, D. Gold nanoparticles conjugated with TAT peptide for enhanced in vitro and in vivo antitumor efficacy. Biomater. Sci. 2020, 8, 5450–5460. [Google Scholar]
- Song, S.; Gui, L.; Feng, Q.; Taledaohan, A.; Li, Y.; Wang, W.; Wang, Y.; Wang, Y. TAT-modified gold nanoparticles enhance the antitumor activity of PAD4 inhibitors. Int. J. Nanomed. 2020, 15, 6659–6671. [Google Scholar] [CrossRef]
- Vaseghi, Z.; Nematollahzadeh, A.; Tavakoli, O. Green methods for the synthesis of metal nanoparticles using biogenic reducing agents: A review. Rev. Chem. Eng. 2018, 34, 529–559. [Google Scholar] [CrossRef]
- Si, S.; Mandal, T.K. Tryptophan-based peptides to synthesize gold and silver nanoparticles: A mechanistic and kinetic study. Chem.–Eur. J. 2007, 13, 3160–3168. [Google Scholar] [CrossRef]
- Tanaka, T.; Urabe, A.; Ohtake, H. Reduction of metal ions by peptides containing tyrosine and tryptophan residues. J. Pept. Sci. 2011, 17, 383–389. [Google Scholar]
- Pujals, S.; Bastús, N.G.; Pereiro, E.; López-Iglesias, C.; Puntes, V.F.; Kogan, M.J.; Giralt, E. Shuttling gold nanoparticles into tumoral cells with an amphipathic proline-rich peptide. ChemBioChem 2009, 10, 1025–1031. [Google Scholar] [CrossRef]
- Bansal, K.; Aqdas, M.; Kumar, M.; Bala, R.; Singh, S.; Agrewala, J.N.; Katare, O.P.; Sharma, R.K.; Wangoo, N. A facile approach for synthesis and intracellular delivery of size-tunable cationic peptide functionalized gold nanohybrids in cancer cells. Bioconjug. Chem. 2018, 29, 1102–1110. [Google Scholar] [CrossRef] [PubMed]
- Tatar, A.S.; Nagy-Simon, T.; Tigu, A.B.; Tomuleasa, C.; Boca, S. Optimization of Tyrosine Kinase Inhibitor-Loaded Gold Nanoparticles for Stimuli-Triggered Antileukemic Drug Release. J. Funct. Biomater. 2023, 14, 399. [Google Scholar] [CrossRef] [PubMed]
- Amina, S.J.; Guo, B. A Review on the Synthesis and Functionalization of Gold Nanoparticles as a Drug Delivery Vehicle. Int. J. Nanomed. 2020, 15, 9823–9857. [Google Scholar] [CrossRef]
- Daima, H.K.; Selvakannan, P.R.; Shukla, R.; Bhargava, S.K.; Bansal, V. Fine-Tuning the Antimicrobial Profile of Biocompatible Gold Nanoparticles by Sequential Surface Functionalization Using Polyoxometalates and Lysine. Langmuir 2013, 29, 15201–15209. [Google Scholar] [CrossRef]
- Nawrocki, G.; Cieplak, M. Interactions of aqueous amino acids and proteins with the (110) surface of ZnS in molecular dynamics simulations. J. Chem. Phys. 2014, 140, 095101. [Google Scholar] [CrossRef]
- Ding, X.; Hua, Y.; Chen, Y.; Zhang, C.; Kong, X. Heavy Metal Complexation of Thiol-Containing Peptides from Soy Glycinin Hydrolysates. Int. J. Mol. Sci. 2015, 16, 8040–8058. [Google Scholar] [CrossRef]
- Chen, C.; Gao, P.; Wang, H.; Cheng, Y.; Lv, J. Histidine-based coordinative polymers for efficient intracellular protein delivery via enhanced protein binding, cellular uptake, and endosomal escape. Biomater. Sci. 2023, 11, 1765–1775. [Google Scholar] [CrossRef]
- Havaldar, D.V.; Patil, R.V.; Moholkar, D.N.; Magdum, P.S.; Vadrale, A.P.; Pawar, K.D. Differently synthesized gold nanoparticles respond differently to functionalization with L-amino acids. Particuology 2020, 52, 97–104. [Google Scholar] [CrossRef]
- Ding, Y.; Sun, Z.; Tong, Z.; Zhang, S.; Min, J.; Xu, Q.; Zhou, L.; Mao, Z.; Xia, H.; Wang, W. Tumor microenvironment-responsive multifunctional peptide coated ultrasmall gold nanoparticles and their application in cancer radiotherapy. Theranostics 2020, 10, 5195–5208. [Google Scholar] [CrossRef]
- Scheibel, T.; Parthasarathy, R.; Sawicki, G.; Lin, X.-M.; Jaeger, H.; Lindquist, S.L. Conducting nanowires built by controlled self-assembly of amyloid fibers and selective metal deposition. Proc. Natl. Acad. Sci. USA 2003, 100, 4527–4532. [Google Scholar] [CrossRef]
- Kasotakis, E.; Mossou, E.; Adler-Abramovich, L.; Mitchell, E.P.; Forsyth, V.T.; Gazit, E.; Mitraki, A. Design of metal-binding sites onto self-assembled peptide fibrils. Biopolymers 2009, 92, 164–172. [Google Scholar] [CrossRef]
- Salamone, T.A.; Rutigliano, L.; Pennacchi, B.; Cerra, S.; Matassa, R.; Nottola, S.; Sciubba, F.; Battocchio, C.; Marsotto, M.; Del Giudice, A.; et al. Thiol Functionalised Gold Nanoparticles Loaded with Methotrexate for Cancer Treatment: From Synthesis to In Vitro Studies on Neuroblastoma Cell Lines. J. Colloid Interface Sci. 2023, 649, 264–278. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Li, R.-X.; Chen, X.; He, X.-X.; Han, A.-L.; Fang, G.-Z.; Liu, J.-F.; Wang, S. Study of Efficiency of Coupling Peptides with Gold Nanoparticles. Chin. J. Anal. Chem. 2017, 45, 662–667. [Google Scholar] [CrossRef]
- Tian, T.; Yang, W.; Wang, X.; Liu, T.; Pan, B.; Guo, W.; Wang, B. Click chemistry-enabled gold nanorods for sensitive detection and viability evaluation of copper (II)-reducing bacteria. Mater. Today Bio 2025, 30, 101453. [Google Scholar] [CrossRef] [PubMed]
- Herizchi, R.; Abbasi, E.; Milani, M.; Akbarzadeh, A. Current Methods for Synthesis of Gold Nanoparticles. Artif. Cells Nanomed. Biotechnol. 2016, 44, 596–602. [Google Scholar] [CrossRef]
- Waragai, A.; Nakagawa, T.; Hinze, W.L.; Takagai, Y. Shape- and Size-Controlled Fabrication of Gold Nano-Urchins via Use of a Mixed Sodium Borohydride and Ascorbic Acid Reductant System. Langmuir 2021, 37, 10702–10707. [Google Scholar] [CrossRef]
- Boruah, S.K.; Medhi, O.K.; Boruah, P.K.; Sarma, P. Green Synthesis of Gold Nanoparticles Using Camellia sinensis and Kinetics of the Reaction. Adv. Mater. Lett. 2012, 3, 481–486. [Google Scholar] [CrossRef]
- Amendola, V.; Meneghetti, M. Laser ablation synthesis in solution and size manipulation of noble metal nanoparticles. Phys. Chem. Chem. Phys. 2009, 11, 3805–3821. [Google Scholar] [CrossRef] [PubMed]
- Longmire, M.; Choyke, P.L.; Kobayashi, H. Clearance Properties of Nano-Sized Particles and Molecules as Imaging Agents: Considerations and Caveats. Nanomedicine 2008, 3, 703–717. [Google Scholar] [CrossRef]
- Blanco, E.; Shen, H.; Ferrari, M. Principles of nanoparticle design for overcoming biological barriers to drug delivery. Nat. Biotechnol. 2015, 33, 941–951. [Google Scholar] [CrossRef]
- Arvizo, R.R.; Miranda, O.R.; Thompson, M.A.; Pabelick, C.M.; Bhattacharya, R.; Robertson, J.D.; Rotello, V.M.; Prakash, Y.S.; Mukherjee, P. Effect of nanoparticle surface charge at the plasma membrane and beyond. Nano Lett. 2010, 10, 2543–2548. [Google Scholar] [CrossRef]
- Han, E.L.; Tang, S.; Kim, D.; Murray, A.M.; Swingle, K.L.; Hamilton, A.G.; Mrksich, K.; Padilla, M.S.; Palanki, R.; Li, J.J.; et al. Peptide-Functionalized Lipid Nanoparticles for Targeted Systemic mRNA Delivery to the Brain. Nano Lett. 2024, in press. [Google Scholar] [CrossRef]
- Zaro, J.L.; Shen, W.-C. Cationic and Amphipathic Cell-Penetrating Peptides (CPPs): Their Structures and In Vivo Studies in Drug Delivery. Front. Chem. Sci. Eng. 2015, 9, 407–427. [Google Scholar] [CrossRef]
- Zhang, N.; Xiong, G.; Liu, Z. Toxicity of metal-based nanoparticles: Challenges in the nano era. Front. Bioeng. Biotechnol. 2022, 10, 1001572. [Google Scholar] [CrossRef]
- Kim, T.H.; Kim, M.; Park, H.S.; Shin, U.S.; Gong, M.S.; Kim, H.W. Size-dependent cellular toxicity of silver nanoparticles. J. Biomed. Mater. Res. A 2012, 100, 1033–1043. [Google Scholar] [CrossRef] [PubMed]
- Karlsson, H.L.; Gustafsson, J.; Cronholm, P.; Möller, L. Size-dependent toxicity of metal oxide particles—A comparison between nano- and micrometer size. Toxicol. Lett. 2009, 188, 112–118. [Google Scholar] [CrossRef]
- Kulkarni, S.A.; Feng, S.S. Effects of particle size and surface modification on cellular uptake and biodistribution of polymeric nanoparticles for drug delivery. Pharm. Res. 2013, 30, 2512–2522. [Google Scholar] [CrossRef]
- Hoshyar, N.; Gray, S.; Han, H.; Bao, G. The effect of nanoparticle size on in vivo pharmacokinetics and cellular interaction. Nanomedicine 2016, 11, 673–692. [Google Scholar] [CrossRef] [PubMed]
- Dobrovolskaia, M.A.; Aggarwal, P.; Hall, J.B.; McNeil, S.E. Preclinical studies to understand nanoparticle interaction with the immune system and its potential effects on nanoparticle biodistribution. Mol. Pharm. 2008, 5, 487–495. [Google Scholar] [CrossRef]
- González-García, L.E.; MacGregor, M.N.; Visalakshan, R.M.; Lazarian, A.; Cavallaro, A.A.; Morsbach, S.; Mierczynska-Vasilev, A.; Mailänder, V.; Landfester, K.; Vasilev, K. Nanoparticles Surface Chemistry Influence on Protein Corona Composition and Inflammatory Responses. Nanomaterials 2022, 12, 682. [Google Scholar] [CrossRef] [PubMed]
- Zhen, J.B.; Yi, J.; Ding, H.H.; Yang, K.W. Self-Assembled Cationic Nanoparticles Combined with Curcumin against Multidrug-Resistant Bacteria. ACS Omega 2022, 7, 29909–29922. [Google Scholar] [CrossRef]
- Fröhlich, E. The Role of Surface Charge in Cellular Uptake and Cytotoxicity of Medical Nanoparticles. Int. J. Nanomed. 2012, 7, 5577–5591. [Google Scholar] [CrossRef]
- Wan, X.; Zhang, J.; Yu, W.; Shen, L.; Ji, S.; Hu, T. Effect of protein immunogenicity and PEG size and branching on the anti-PEG immune response to PEGylated proteins. Process Biochem. 2017, 52, 183–191. [Google Scholar] [CrossRef]
- Fosgerau, K.; Hoffmann, T. Peptide Therapeutics: Current Status and Future Directions. Drug Discov. Today 2015, 20, 122–128. [Google Scholar] [CrossRef] [PubMed]
- Augustine, R.; Hasan, A.; Primavera, R.; Wilson, R.J.; Thakor, A.S.; Kevadiya, B.D. Cellular Uptake and Retention of Nanoparticles: Insights on Particle Properties and Interaction with Cellular Components. Mater. Today Commun. 2020, 25, 101692. [Google Scholar] [CrossRef]
- Allen, J.; Pellois, J.P. Hydrophobicity Is a Key Determinant in the Activity of Arginine-Rich Cell Penetrating Peptides. Sci. Rep. 2022, 12, 15981. [Google Scholar] [CrossRef]
- Li, Z.; Chen, Y.; Yang, Y.; Yu, Y.; Zhang, Y.; Zhu, D.; Yu, X.; Ouyang, X.; Xie, Z.; Zhao, Y.; et al. Recent Advances in Nanomaterials-Based Chemo-Photothermal Combination Therapy for Improving Cancer Treatment. Front. Bioeng. Biotechnol. 2019, 7, 293. [Google Scholar] [CrossRef]
- Vines, J.B.; Yoon, J.H.; Ryu, N.E.; Lim, D.J.; Park, H. Gold Nanoparticles for Photothermal Cancer Therapy. Front. Chem. 2019, 7, 167. [Google Scholar] [CrossRef]
- Zhou, H.; Xu, H.; Li, X.; Lv, Y.; Ma, T.; Guo, S.; Huang, Z.; Wang, X.; Xu, P. Dual targeting hyaluronic acid–RGD mesoporous silica coated gold nanorods for chemo-photothermal cancer therapy. Mater. Sci. Eng. C 2017, 81, 261–270. [Google Scholar] [CrossRef] [PubMed]
- Zhi, D.; Yang, T.; Yang, J.; Fu, S.; Zhang, S. Targeting Strategies for Superparamagnetic Iron Oxide Nanoparticles in Cancer Therapy. Acta Biomater. 2020, 102, 13–34. [Google Scholar] [CrossRef] [PubMed]
- Vangijzegem, T.; Lecomte, V.; Ternad, I.; Van Leuven, L.; Muller, R.N.; Stanicki, D.; Laurent, S. Superparamagnetic Iron Oxide Nanoparticles (SPION): From Fundamentals to State-of-the-Art Innovative Applications for Cancer Therapy. Pharmaceutics 2023, 15, 236. [Google Scholar] [CrossRef]
- Makharadze, D.; del Valle, L.J.; Katsarava, R.; Puiggalí, J. The Art of PEGylation: From Simple Polymer to Sophisticated Drug Delivery System. Int. J. Mol. Sci. 2025, 26, 3102. [Google Scholar] [CrossRef]
- Larson, T.A.; Joshi, P.P.; Sokolov, K. Preventing Protein Adsorption and Macrophage Uptake of Gold Nanoparticles via a Hydrophobic Shield. ACS Nano 2012, 6, 9182–9190. [Google Scholar] [CrossRef] [PubMed]
- Sultana, N.; Arayne, M.S.; Sabri, R. Erythromycin synergism with essential and trace elements. Pak. J. Pharm. Sci. 2005, 18, 35–39. [Google Scholar]
- Javid, A.; Ghani, M.J.; Shahzad, S.; Rezaei-Zarchi, S. Antineoplastic Drug-Loaded Polymer-Modified Magnetite Nanoparticles: Comparative Analysis of EPR-Mediated Drug Delivery. Cell Biol. Int. 2020, 44, 2042–2052. [Google Scholar] [CrossRef]
- Taherian, A.; Esfandiari, N.; Rouhani, S. Breast Cancer Drug Delivery by Novel Drug-Loaded Chitosan-Coated Magnetic Nanoparticles. Cancer Nano 2021, 12, 15. [Google Scholar] [CrossRef]
- Gong, Z.; Liu, X.; Zhou, B.; Wang, G.; Guan, X.; Xu, Y.; Zhang, J.; Hong, Z.; Cao, J.; Sun, X.; et al. Tumor acidic microenvironment-induced drug release of RGD peptide nanoparticles for cellular uptake and cancer therapy. Colloids Surf. B Biointerfaces 2021, 202, 111673. [Google Scholar] [CrossRef]
- Zhang, X.; Wang, X.; Zhong, W.; Ren, X.; Sha, X.; Fang, X. Matrix Metalloproteinases-2/9-Sensitive Peptide-Conjugated Polymer Micelles for Site-Specific Release of Drugs and Enhancing Tumor Accumulation: Preparation and In Vitro and In Vivo Evaluation. Int. J. Nanomed. 2016, 11, 1643–1661. [Google Scholar]
- Zhu, L.; Torchilin, V.P. Stimulus-Responsive Nanopreparations for Tumor Targeting. Integr. Biol. 2013, 5, 96–107. [Google Scholar] [CrossRef]
- Yao, Q.; Kou, L.; Tu, Y.; Zhu, L. MMP-Responsive ‘Smart’ Drug Delivery and Tumor Targeting. Trends Pharmacol. Sci. 2018, 39, 766–781. [Google Scholar] [CrossRef]
- Ding, Y.; Dai, Y.; Wu, M.; Li, L. Glutathione-Mediated Nanomedicines for Cancer Diagnosis and Therapy. Chem. Eng. J. 2021, 426, 128880. [Google Scholar] [CrossRef]
- Liu, Z.; Chen, Y.; Zhang, Q.; Li, W.; He, X. Glutathione-Responsive and -Exhausting Metal Nanomedicines for Robust Synergistic Cancer Therapy. Front. Bioeng. Biotechnol. 2023, 1161472. [Google Scholar] [CrossRef]
- Li, Y.; He, D.; Tu, J.; Wang, R.; Zu, C.; Chen, Y.; Yang, W.; Shi, D.; Webster, T.J.; Shen, Y. The Comparative Effect of Wrapping Solid Gold Nanoparticles and Hollow Gold Nanoparticles with Doxorubicin-Loaded Thermosensitive Liposomes for Cancer Thermo-Chemotherapy. Nanoscale 2018, 10, 8628–8641. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Xing, L.; Hu, Y.; Xiong, Z.; Wang, R.; Xu, X.; Du, L.; Shen, M.; Shi, X. An RGD-modified hollow silica@Au core/shell nanoplatform for tumor combination therapy. Acta Biomater. 2017, 62, 273–283. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.; Liao, R.; Mahmood, A.A.; Xu, H.; Zhou, Q. pH-Responsive pHLIP (pH Low Insertion Peptide) Nanoclusters of Superparamagnetic Iron Oxide Nanoparticles as a Tumor-Selective MRI Contrast Agent. Acta Biomater. 2017, 55, 194–203. [Google Scholar] [CrossRef]
- Lee, D.; Zhao, J.; Yang, H.; Xu, S.; Kim, H.; Pacheco, S.; Keshavjee, S.; Liu, M. Effective Delivery of a Rationally Designed Intracellular Peptide Drug with Gold Nanoparticle–Peptide Hybrids. Nanoscale 2015, 7, 12356–12360. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhang, L.; Liu, C.; Luo, Y.; Chen, D. Peptide-Mediated Nanocarriers for Targeted Drug Delivery: Developments and Strategies. Pharmaceutics 2024, 16, 240. [Google Scholar] [CrossRef]
- Mittal, A.K.; Banerjee, U.C. In vivo Safety, Toxicity, Biocompatibility and Anti-Tumour Efficacy of Bioinspired Silver and Selenium Nanoparticles. Mater. Today Commun. 2021, 26, 102001. [Google Scholar] [CrossRef]
- Chen, Y.; Hou, S. Recent Progress in the Effect of Magnetic Iron Oxide Nanoparticles on Cells and Extracellular Vesicles. Cell Death Discov. 2023, 9, 195. [Google Scholar] [CrossRef]
- Arami, H.; Khandhar, A.; Liggitt, D.; Krishnan, K.M. In Vivo Delivery, Pharmacokinetics, Biodistribution and Toxicity of Iron Oxide Nanoparticles. Chem. Soc. Rev. 2015, 44, 8576–8607. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Zhou, X.; Yu, Q.; Yang, L.; Sun, D.; Zhou, Y.; Liu, J. Epigallocatechin-3-Gallate (EGCG)-Stabilized Selenium Nanoparticles Coated with Tet-1 Peptide to Reduce Amyloid-β Aggregation and Cytotoxicity. ACS Sustain. Chem. Eng. 2014, 2, 932–940. [Google Scholar] [CrossRef] [PubMed]
- Caravan, P. Strategies for Increasing the Sensitivity of Gadolinium-Based MRI Contrast Agents. Chem. Soc. Rev. 2006, 35, 512–523. [Google Scholar] [CrossRef]
- Chauhan, V.; Dhiman, V.K.; Mahajan, G.; Pandey, A.; Kanwar, S.S. Synthesis and characterization of silver nanoparticles developed using a novel lipopeptide biosurfactant and evaluating its antimicrobial and cytotoxic efficacy. Process Biochem. 2023, 124, 51–62. [Google Scholar] [CrossRef]
- Zhou, Q.; Pan, Y.; Liang, Q.; Yang, J.; Zhu, S.; Shi, H.; Li, G. Peptide-Guided Assembly of Silver Nanoparticles for the Diagnosis of HER2-Positive Breast Cancer. Anal. Chem. 2024, 96, 49. [Google Scholar] [CrossRef]
- White, B.E.; White, M.K.; Adhvaryu, H.; Makhoul, I.; Nima, Z.A.; Biris, A.S.; Ali, N. Nanotechnology Approaches to Addressing HER2-Positive Breast Cancer. Cancer Nanotechnol. 2020, 11, 12. [Google Scholar] [CrossRef]
- Song, E.; Gau/din, A.; King, A.; Seo, Y.-E.; Suh, H.W.; Deng, Y.; Yi, Y.; Rotello, V.M. Surface Chemistry Governs Cellular Tropism of Nanoparticles in the Brain. Nat. Commun. 2017, 8, 15322. [Google Scholar] [CrossRef]
- Hoskins, C.; Min, Y.; Gueorguieva, M.; McDougall, C.; Volovick, A.; Prentice, P.; Wang, Z.; Melzer, A.; Cuschieri, A.; Wang, L. Hybrid Gold–Iron Oxide Nanoparticles as a Multifunctional Platform for Biomedical Applications. J. Nanobiotechnol. 2012, 10, 27. [Google Scholar] [CrossRef]
- Fang, C.; Veiseh, O.; Kievit, F.; Bhattarai, N.; Wang, F.; Stephen, Z.; Li, C.; Lee, D.; Ellenbogen, R.G.; Zhang, M. Functionalization of Iron Oxide Magnetic Nanoparticles with Targeting Ligands: Their Physicochemical Properties and In Vivo Behavior. Nanomedicine 2010, 5, 1357–1369. [Google Scholar] [CrossRef]
- Liang, J.; Zhang, X.; Miao, Y.; Li, J.; Gan, Y. Lipid-Coated Iron Oxide Nanoparticles for Dual-Modal Imaging of Hepatocellular Carcinoma. Int. J. Nanomed. 2017, 12, 2033–2044. [Google Scholar] [CrossRef]
- Shahdeo, D.; Roberts, A.; Kesarwani, V.; Horvat, M.; Chouhan, R.S.; Gandhi, S. Polymeric Biocompatible Iron Oxide Nanoparticles Labeled with Peptides for Imaging in Ovarian Cancer. Biosci. Rep. 2022, 42, BSR20212622. [Google Scholar] [CrossRef]
- Biscaglia, F.; Caligiuri, I.; Rizzolio, F.; Ripani, G.; Palleschi, A.; Meneghetti, M.; Gobbo, M. Protection against proteolysis of a targeting peptide on gold nanostructures. Nanoscale 2021, 13, 10544–10554. [Google Scholar] [CrossRef]
- Zhang, D.; Neumann, O.; Wang, H.; Yuwono, V.M.; Barhoumi, A.; Perham, M.; Hartgerink, J.D.; Wittung-Stafshede, P.; Halas, N.J. Gold nanoparticles can induce the formation of protein-based aggregates at physiological pH. Nano Lett. 2009, 9, 666–671. [Google Scholar] [CrossRef]
- Truong, L.; Zaikova, T.; Baldock, B.L.; Balik-Meisner, M.; To, K.; Reif, D.M.; Kennedy, Z.C.; Hutchison, J.E.; Tanguay, R.L. Systematic Determination of the Relationship between Nanoparticle Core Diameter and Toxicity for a Series of Structurally Analogous Gold Nanoparticles in Zebrafish. Nanotoxicology 2019, 13, 879–893. [Google Scholar] [CrossRef] [PubMed]
- Mülhopt, S.; Diabaté, S.; Dilger, M.; Adelhelm, C.; Anderlohr, C.; Bergfeldt, T.; Gómez de la Torre, J.; Jiang, Y.; Valsami-Jones, E.; Langevin, D.; et al. Characterization of Nanoparticle Batch-to-Batch Variability. Nanomaterials 2018, 8, 311. [Google Scholar] [CrossRef] [PubMed]
- Nagarkar, R.P.; Schneider, J.P. Synthesis and Primary Characterization of Self-Assembled Peptide-Based Hydrogels. In Nanostructure Design; Gazit, E., Nussinov, R., Eds.; Methods in Molecular Biology™; Humana Press: Totowa, NJ, USA, 2008; Volume 474, pp. 69–83. [Google Scholar]
- Đorđević, S.; Gonzalez, M.M.; Conejos-Sánchez, I.; Carreira, B.; Pozzi, S.; Acúrcio, R.C.; Satchi-Fainaro, R.; Florindo, H.F.; Vicent, M.J. Current Hurdles to the Translation of Nanomedicines from Bench to the Clinic. Drug Deliv. Transl. Res. 2022, 12, 500–525. [Google Scholar] [CrossRef] [PubMed]
- Wicki, A.; Witzigmann, D.; Balasubramanian, V.; Huwyler, J. Nanomedicine in Cancer Therapy: Challenges, Opportunities, and Clinical Applications. J. Control. Release 2015, 200, 138–157. [Google Scholar] [CrossRef]
- Polderdijk, S.G.I.; Limzerwala, J.F.; Spiess, C. Plasma Membrane Damage Limits Cytoplasmic Delivery by Conventional Cell Penetrating Peptides. PLoS ONE 2024, 19, e0305848. [Google Scholar] [CrossRef]
- Egorova, E.A.; Nikitin, M.P. Delivery of theranostic nanoparticles to various cancers by means of integrin-binding peptides. Int. J. Mol. Sci. 2022, 23, 13735. [Google Scholar] [CrossRef]
- Yang, Y.; Chen, Q.; Li, S.; Ma, W.; Yao, G.; Ren, F.; Cai, Z.; Zhao, P.; Liao, G.; Xiong, J.; et al. iRGD-Mediated and Enzyme-Induced Precise Targeting and Retention of Gold Nanoparticles for the Enhanced Imaging and Treatment of Breast Cancer. J. Biomed. Nanotechnol. 2018, 14, 1396–1408. [Google Scholar] [CrossRef]
- López-Laguna, H.; Sánchez, J.; Unzueta, U.; Mangues, R.; Vázquez, E.; Villaverde, A. Divalent Cations: A Molecular Glue for Protein Materials. Trends Biochem. Sci. 2020, 45, 992–1003. [Google Scholar] [CrossRef]
- Soni, S.; Kori, S.K.; Nema, P.; Iyer, A.K.; Soni, V.; Kashaw, S.K. Cell-Penetrating Peptides as Keys to Endosomal Escape and Intracellular Trafficking in Nanomedicine Delivery. Curr. Med. Chem. 2024, 31, 1–25. [Google Scholar] [CrossRef] [PubMed]
- Luthuli, S.D.; Chili, M.M.; Revaprasadu, N.; Shonhai, A. Cysteine-Capped Gold Nanoparticles Suppress Aggregation of Proteins Exposed to Heat Stress. IUBMB Life 2013, 65, 454–461. [Google Scholar] [CrossRef]
- Basu, A.; Vaskevich, A.; Chuntonov, L. Glutathione Self-Assembles into a Shell of Hydrogen-Bonded Intermolecular Aggregates on “Naked” Silver Nanoparticles. J. Phys. Chem. B 2021, 125, 895–906. [Google Scholar] [CrossRef]
- Kumar, D.; Meenan, B.J.; Dixon, D. Glutathione-Mediated Release of Bodipy® from PEG Cofunctionalized Gold Nanoparticles. Int. J. Nanomed. 2012, 7, 4007–4022. [Google Scholar] [CrossRef] [PubMed]
- Fan, T.; Yu, X.; Shen, B.; Sun, L. Peptide Self-Assembled Nanostructures for Drug Delivery Applications. J. Nanomater. 2017, 2017, 4562474. [Google Scholar] [CrossRef]
- Pentlavalli, S.; Coulter, S.; Laverty, G. Peptide Nanomaterials for Drug Delivery Applications. Curr. Protein Pept. Sci. 2020, 21, 401–412. [Google Scholar] [CrossRef] [PubMed]
- Cui, H.; Webber, M.J.; Stupp, S.I. Self-Assembly of Peptide Amphiphiles: From Molecules to Nanostructures to Biomaterials. Biopolymers 2010, 94, 1–18. [Google Scholar] [CrossRef]
- Lohan, S.; Konshina, A.G.; Mohammed, E.H.M.; Helmy, N.M.; Jha, S.K.; Tiwari, R.K.; Maslennikov, I.; Efremov, R.G.; Parang, K. Impact of Stereochemical Replacement on Activity and Selectivity of Membrane-Active Antibacterial and Antifungal Cyclic Peptides. NPJ Antimicrob. Resist. 2025, 3, 56. [Google Scholar] [CrossRef]
- Salehi, D.; Mohammed, E.H.M.; Helmy, N.M.; Parang, K. Antibacterial and Antifungal Activities of Linear and Cyclic Peptides Containing Arginine, Tryptophan, and Diphenylalanine. Antibiotics 2025, 14, 82. [Google Scholar] [CrossRef] [PubMed]
- Buckton, L.K.; McAlpine, S.R. Improving the Cell Permeability of Polar Cyclic Peptides by Replacing Residues with Alkylated Amino Acids, Asparagines, and d-Amino Acids. Org. Lett. 2018, 20, 506–509. [Google Scholar] [CrossRef] [PubMed]
- Dražić, E.; Jelušić, D.; Janković Bevandić, P.; Mauša, G.; Kalafatović, D. Using Machine Learning to Fast-Track Peptide Nanomaterial Discovery. ACS Nano 2025, 19, 20295–20320. [Google Scholar] [CrossRef]
- Goles, M.; Daza, A.; Cabas-Mora, G.; Sarmiento-Varón, L.; Sepúlveda-Yañez, J.; Anvari-Kazemabad, H.; Davari, M.D.; Uribe-Paredes, R.; Olivera-Nappa, Á.; Navarrete, M.A.; et al. Peptide-Based Drug Discovery through Artificial Intelligence: Towards an Autonomous Design of Therapeutic Peptides. Brief. Bioinform. 2024, 25, bbae275. [Google Scholar] [CrossRef]
- Kopac, T. Leveraging Artificial Intelligence and Machine Learning for Characterizing Protein Corona, Nanobiological Interactions, and Advancing Drug Discovery. Bioengineering 2025, 12, 312. [Google Scholar] [CrossRef]
- Batra, R.; Loeffler, T.D.; Chan, H.; Srinivasan, S.; Cui, H.; Korendovych, I.V.; Nanda, V.; Palmer, L.C.; Solomon, L.A.; Fry, H.C.; et al. Machine Learning Overcomes Human Bias in the Discovery of Self-Assembling Peptides. Nat. Chem. 2022, 14, 1427–1435. [Google Scholar] [CrossRef]
- Saito, S.; Oka, R.; Ikuta, T.; Abe, Y.; Choi, Y.; Ohashi, T.; Critchley, K.; Choi, J.; Evans, S.D.; Tanaka, M.; et al. Integration of Peptide Array Screening and Machine Learning for the Discovery of Peptides Directing Triangular Gold Nanoplate Formation. ACS Appl. Nano Mater. 2025, 8, 13227–13237. [Google Scholar] [CrossRef]
| MNP Type | Unique Properties | Biomedical Applications [Ref] |
|---|---|---|
| Gold (AuNPs) | High stability and biocompatibility; strong SPR (~520 nm) | Photothermal therapy, imaging; easily functionalized for targeted drug delivery [14]. |
| Silver (AgNPs) | Potent antimicrobial and anticancer activity (ROS generation) | Wound healing, antibacterial coatings; co-delivery with antibiotics or anticancer drugs for synergistic effect [15]. |
| Iron Oxide (Fe3O4) | Superparamagnetic; MRI contrast enhancement | Magnetic targeting of drugs; theranostics combining MRI imaging and drug delivery [16]. |
| Zinc Oxide (ZnO) | Dissolves in acidic environments; generates ROS in tumors | Tumor-specific drug release and cytotoxicity due to pH sensitivity [17]. |
| Selenium (SeNPs) | Antioxidant and immunomodulatory; induces apoptosis in cancer cells | Delivery of chemotherapeutics; treatment of cancer, cardiovascular, and inflammatory diseases [18]. |
| Gadolinium (GdNPs) | Paramagnetic, excellent MRI contrast; relatively heavy metal | Theranostics: combined MRI and drug delivery or photothermal therapy [19]. |
| Peptide Class | Structural Features | Roles in PINPs [Ref] |
|---|---|---|
| Linear | Unbranched amino acid chain | Easily modified; act as capping agents or templates (e.g., RGD-peptide for targeting); modulate NP size and functionality [35]. |
| Cyclic | Head-to-tail or side-chain (disulfide/lactam) cyclization | Rigid and protease-resistant; provide high-affinity binding; can reduce and stabilize NPs simultaneously (e.g., [WR] peptides for AuNPs) [26,35]. |
| Amphipathic (CPPs) | Spatially segregated hydrophobic/hydrophilic residues (often α-helical) | Facilitate membrane penetration and endosomal escape; stabilize MNPs in aqueous media; common cell-penetrating motifs (HIV-TAT, penetration) enhance uptake of MNP cargos [36]. |
| Self-assembling | Sequences forming β-sheet or fibrillar structures (often aromatic) | Form nanofibers or hydrogels that template inorganic growth; encapsulate drugs; enable stimuli-responsive release (e.g., fibrils with cysteines nucleate metal nanoparticles) [37]. |
| Peptide Morphology | Nanoparticle Uptake Mechanism | Endosomal Escape Ability | Advantages | Limitations |
|---|---|---|---|---|
| Linear peptide coating | Primarily receptor-independent or nonspecific endocytosis | Low, often leads to entrapment in endo-lysosomal compartments [38] | Simple design; easy synthesis and functionalization | Poor endosomal escape; cargo degradation in lysosomes |
| Cyclic peptide coating | Targeted receptor-mediated endocytosis due to high binding affinity [24] | Low, generally remains in vesicles without membrane-lytic properties [39] | High structural rigidity; protease resistance; selective targeting | Limited membrane-disruptive capacity [39] |
| Amphipathic peptide coating | Endocytosis (often receptor-mediated if targeting domain is present) or direct translocation bypassing endocytosis [40] | High, disrupts endosomal membranes for cytosolic release | Strong membrane interaction; efficient endosomal escape; enables delivery to cytosol/nucleus | May lack targeting specificity; risk of nonspecific membrane disruption [39] |
| Function Category | Description and Examples [Ref] |
|---|---|
| Targeting Ligands | Peptides that bind overexpressed receptors (e.g., RGD for integrins, CRGDK for NRP-1) direct PINPs to tumors, e.g., CRGDK–AuNPs show enhanced tumor accumulation; dual-targeted AuNPs carrying both a homing peptide and drug peptide improve delivery efficiency [43]. |
| Capping/Stabilizing Agents | Peptides that bind metal surfaces (thiol, amine, carboxyl) to prevent aggregation and add biofunctionality. For example, cysteine-containing peptides cap AuNPs via Au–S bonds; peptides embedding RGD sequences provide stability and integrin targeting [51]. |
| Reducing Agents | Redox-active peptides reduce metal ions to NPs under mild conditions. For instance, peptides with multiple Trp reduce Au3+ while binding the NP, yielding stable AuNPs (e.g., [WR] peptides) [26]. |
| Therapeutic/Functional | Peptides that carry bioactivity or responsive elements (e.g., AMPs, enzyme-cleavable linkers) to add functionality (overlapping) [52,53,54]. |
| Approach | Reducing/Capping Agent | Notes/Examples [Refs] |
|---|---|---|
| Chemical (Turkevich) | Sodium citrate (mild reductant + weak capping) | Classic AuNP synthesis (10–20 nm); citrate reduces Au3+ to Au0. Produces stable colloids, but organic solvents or byproducts are possible [89]. |
| Chemical (borohydride) | NaBH4 (strong reductant); surfactant capping | Produces very small Ag/Au NPs (<5 nm); fast reduction but often leaves toxic by-products [90]. |
| Plant extracts (bio) | Polyphenols (e.g., flavonoids) | Green synthesis: leaf/fruit extracts reduce Au/Ag ions to NPs. E.g., Camellia extract yields AuNPs. Eco-friendly and scalable, product variability depends on extract composition [91,92]. |
| Peptide-mediated (green) | Functional peptides (Tyr, Trp, Cys rich) | Peptides reduce and cap in one step, e.g., cyclic [WR] peptides form AuNPs with integrated targeting. Mild conditions yield biocompatible, functionalized NPs [26]. |
| Physical (laser ablation) | None (requires energy input) | Generates pure MNPs in solution without chemical reagents. Can control size by laser parameters; high energy cost, specialized equipment [93]. |
| Peptide Class | Typical Cargo and Role | Delivery Efficiency (Examples) | Key Limitations |
|---|---|---|---|
| Cell-Penetrating Peptides (CPPs), e.g., TAT, penetratin, poly-Arg (R9) | Broad range: small drugs, siRNA, proteins, often used to ferry cargo across cell membranes (non-specific uptake enhancer). | High cellular uptake: For instance, TAT-modified AuNP–drug conjugates significantly delayed tumor growth in vivo compared to free drug [71]. | Non-specific targeting: CPPs enter many cell types, potentially causing off-target effects. Often become sequestered in endosomes (if not combined with endosomolytic element). Highly cationic CPPs can be cytotoxic at high density (membrane disruption, hemolysis) [153]. |
| Targeting peptides (homing ligands), e.g., cRGD, iRGD, LyP-1, F3 peptide | Typically, small molecules or drugs attached to NPs; peptides guide NPs to specific cell-surface receptors (tumor vasculature, cancer cells, etc.). Often used for targeted drug delivery. | Enhanced tissue/cell specificity: Peptide-guided NPs achieve high accumulation at target sites, e.g., doxorubicin-loaded AuNPs functionalized with cRGD showed greater tumor uptake and lower off-target toxicity than free drug [153]. RGD-decorated NPs increased tumor localization in integrin-rich tumors (vs. non-targeted) [154]. Some targeting peptides (e.g., iRGD) also promote deep tumor penetration and enhance chemotherapeutic efficacy in vivo [154,155]. | Dependency on receptor expression: Only effective if target receptors are highly expressed; variability in patients’ tumors can reduce efficacy. Uptake is typically via receptor-mediated endocytosis, and cargo can remain entrapped without an additional escape mechanism. Possible immunogenicity for larger targeting peptides or if the human sequence is foreign. |
| Amphipathic and fusogenic peptides, e.g., INF7 (derived from HA2), GALA, KALA, LK15, histidine-rich peptides | Often used alongside CPPs or targeting ligands to promote endosomal escape of nucleic acids (siRNA/DNA), toxins, or other therapeutics. Some also serve as primary CPPs (transportan, arginine-rich AMPs). | Greatly improved cytosolic delivery: For example, histidine-rich CPPs enhance endosomal escape via proton sponge swelling and membrane disruption, significantly boosting functional delivery efficiency for proteins and nucleic acids [156]. Conjugation of histidine-rich peptides to nanoparticles improved cytosolic access, confirmed by leakage assays and microscopy, without significant toxicity. Fusogenic peptide strategies prevent lysosomal degradation and can increase protein cytosolic delivery several-fold [157]. | Potential cell toxicity: If too potent, they can disrupt plasma membranes or cause global membrane damage. Often pH-dependent; efficacy may vary with endosomal maturation conditions. Usually not cell-specific (need to be targeted to the endosome of specific cells via another ligand). Peptide sequences may risk complement activation or immunogenicity if administered systemically in high doses. |
| Stabilizing/capping peptides, e.g., cysteine-rich peptides, glutathione, di- or tripeptides (CG, YY) | Serve as surface coatings to stabilize metal NPs (prevent aggregation) and can also reduce metal ions during “green” synthesis. Cargo is typically the NP itself (which then carries drugs or imaging agents); sometimes a drug is co-conjugated. | Improved colloidal stability and circulation: Cysteine-capped AuNPs suppress protein aggregation under heat stress and maintain nanoparticle stability [158]. Glutathione self-assembles into dense, ordered shells on metal nanoparticles, preventing aggregation and enhancing biocompatibility [159]. PEG-cofunctionalized gold nanoparticles with glutathione exhibit prolonged circulation times and reduced opsonization in vivo [160]. | May reduce cellular uptake: Highly hydrophilic or “stealth” capping peptides (or PEGylated peptides) can inhibit interaction with target cells (lower uptake). Often no active targeting or uptake function—they may need additional targeting or CPP ligands for efficient delivery. Peptide capping layers must remain intact until reaching target site (risk of premature desorption in blood). |
| Self-assembling peptide systems, e.g., peptide nanofibers, peptide amphiphiles that nucleate metals [161,162] | Peptides form the nanoparticle scaffold or template; they can carry drugs within peptide assemblies or nucleate metal NP growth in situ. Cargo can be drugs co-assembled, or the peptide–metal hybrid itself acts as the therapeutic | One-step construction and high payload: Peptide-driven assembly can create uniform nanostructures with high peptide density, simplifying functionalization. For example, short dipeptides were used to simultaneously reduce gold ions and cap the resulting AuNPs, achieving a high peptide payload per particle [161]. Some cyclic peptide nanospheres can encapsulate drugs and release them in response to stimuli [162]. | Polydispersity and reproducibility issues: Self-assembly is sensitive to conditions; slight changes can alter NP size/shape, making consistency a challenge [163]. These systems may lack in vivo stability, and peptide assemblies might disassemble or be enzymatically degraded before reaching the target. Tuning their biodegradation rate is complex. Also, without further modification, purely peptide-based NPs might have limited target specificity or might trigger immune responses if the assembled structures are recognized as foreign [162]. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shirazi, A.N.; Vadlapatla, R.; Koomer, A.; Nguyen, A.; Khoury, V.; Parang, K. Peptide-Based Inorganic Nanoparticles as Efficient Intracellular Delivery Systems. Pharmaceutics 2025, 17, 1123. https://doi.org/10.3390/pharmaceutics17091123
Shirazi AN, Vadlapatla R, Koomer A, Nguyen A, Khoury V, Parang K. Peptide-Based Inorganic Nanoparticles as Efficient Intracellular Delivery Systems. Pharmaceutics. 2025; 17(9):1123. https://doi.org/10.3390/pharmaceutics17091123
Chicago/Turabian StyleShirazi, Amir Nasrolahi, Rajesh Vadlapatla, Ajoy Koomer, Anthony Nguyen, Vian Khoury, and Keykavous Parang. 2025. "Peptide-Based Inorganic Nanoparticles as Efficient Intracellular Delivery Systems" Pharmaceutics 17, no. 9: 1123. https://doi.org/10.3390/pharmaceutics17091123
APA StyleShirazi, A. N., Vadlapatla, R., Koomer, A., Nguyen, A., Khoury, V., & Parang, K. (2025). Peptide-Based Inorganic Nanoparticles as Efficient Intracellular Delivery Systems. Pharmaceutics, 17(9), 1123. https://doi.org/10.3390/pharmaceutics17091123







