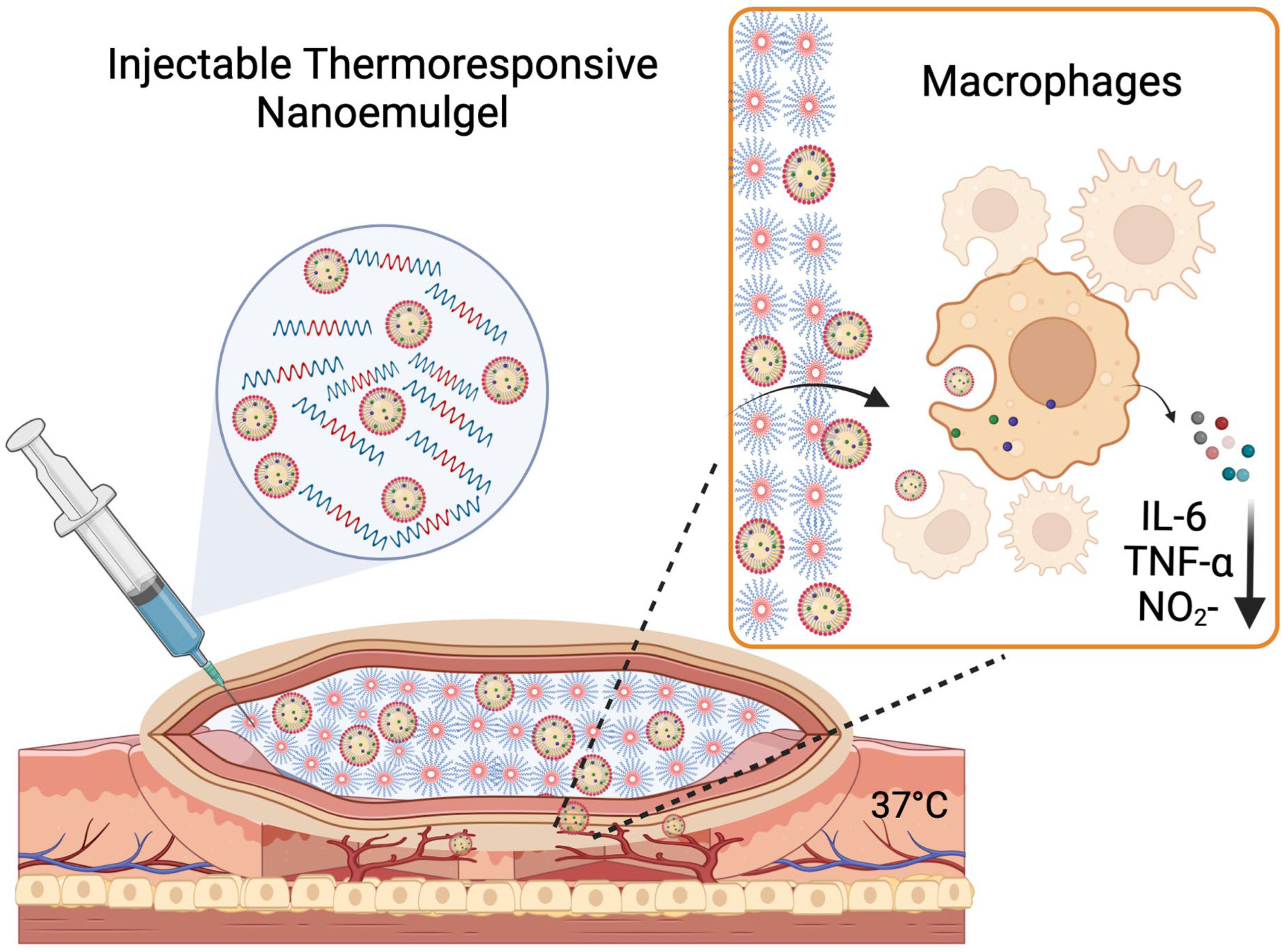
A Reversibly Thermoresponsive, Theranostic Nanoemulgel for Tacrolimus Delivery to Activated Macrophages: Formulation and In Vitro Validation
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. Preparation of Nanoemulsions (NEs)
2.2.2. Preparation of Nanoemulgels (NEGs)
2.2.3. Colloidal Characterization of the NEs and NEGs
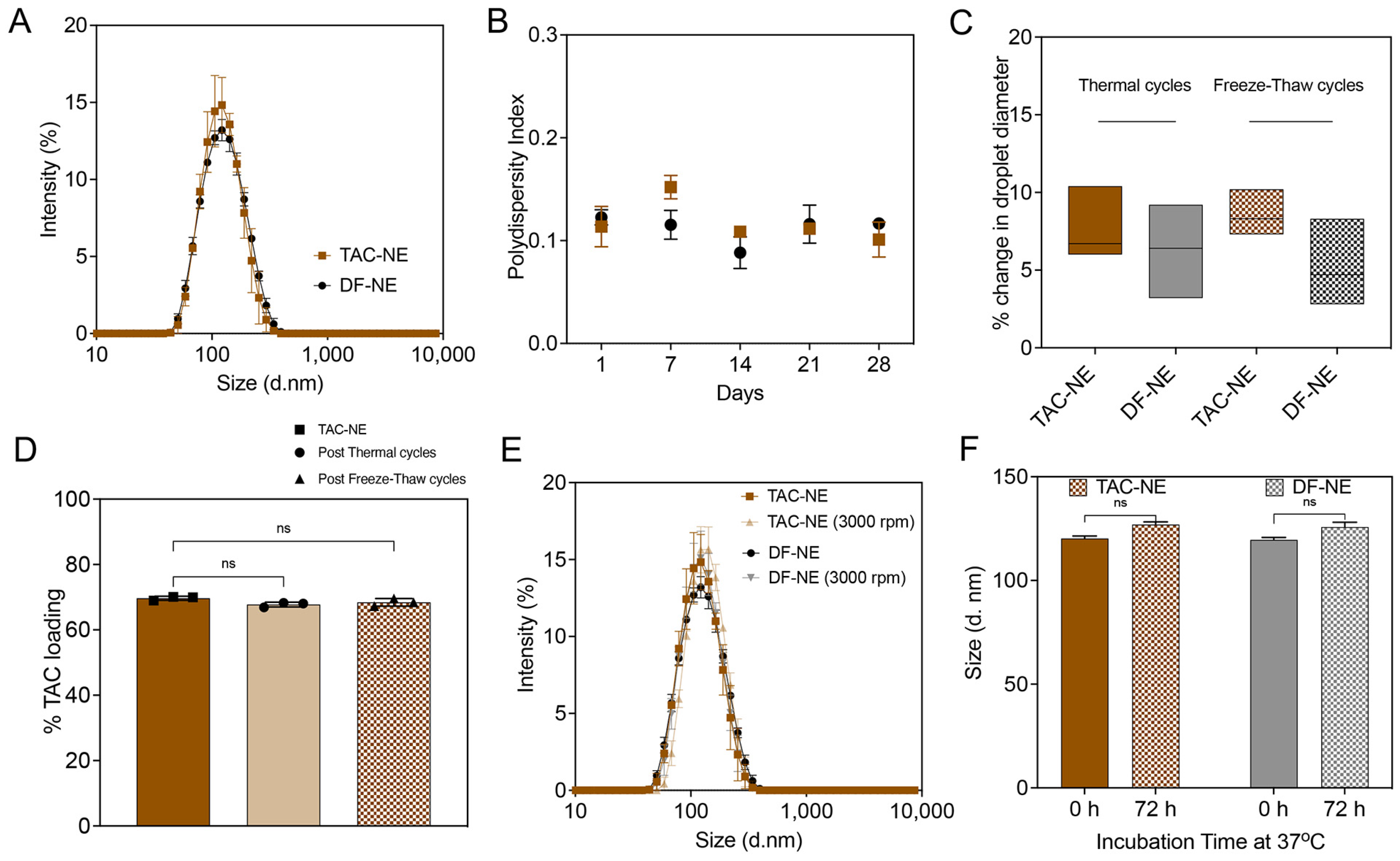
2.2.4. Physical Stress Testing of NEs: Centrifugation and Filtration
2.2.5. Thermal Cycling and Freeze-Thaw Cycling of NEs
2.2.6. Serum Stability Testing of NEs and NEGs
2.2.7. Near-Infrared Fluorescence (NIRF) Imaging of NEs and NEGs
2.2.8. Sterility Testing of NEGs
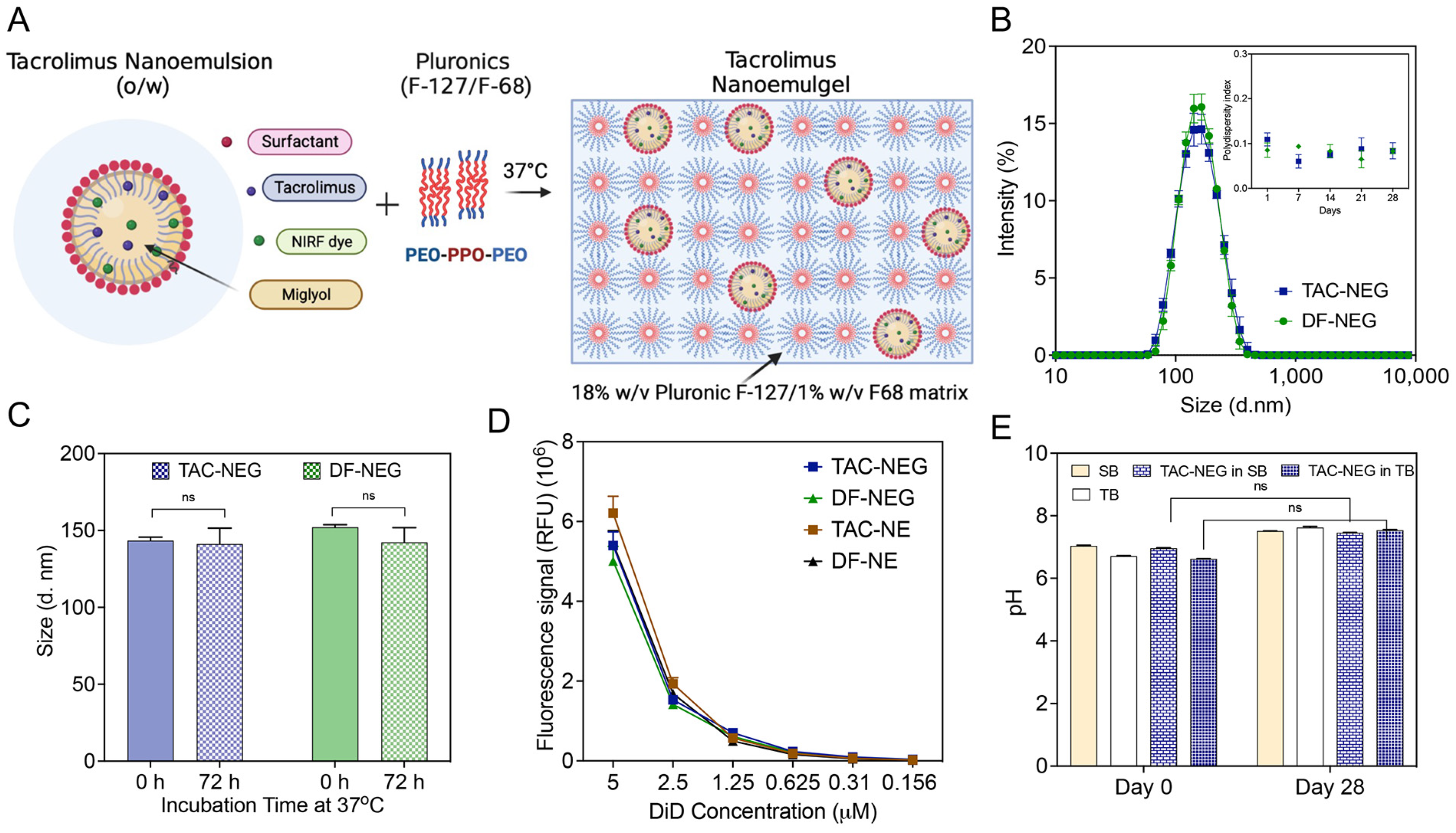
2.2.9. pH Measurements of NEs and NEGs
2.2.10. Reverse Phase High-Performance Liquid Chromatography (RP-HPLC)
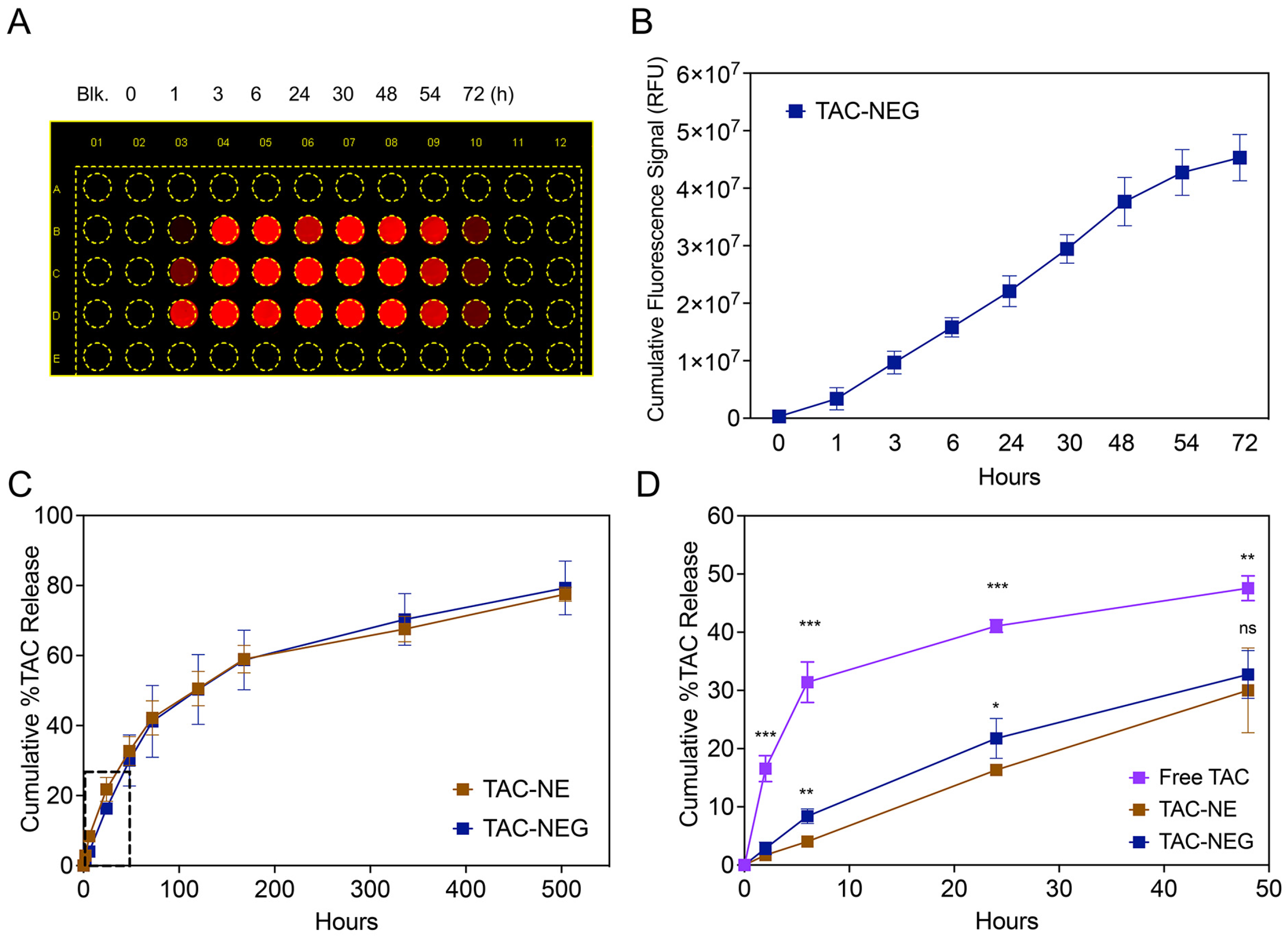
2.2.11. Dissolution Test to Study the Release of TAC-NE from TAC-NEG
2.2.12. In Vitro Release of TAC from TAC-NE and TAC-NEG
2.2.13. Rheology on NEGs
2.2.14. Macrophage Viability following Exposure of NEs and NEGs
2.2.15. Fluorescence Microscopy
2.2.16. Flow Cytometry
2.2.17. Enzyme-Linked Immunosorbent Assay (ELISA)
2.2.18. Nitric Oxide Assay
2.2.19. Statistical Analysis
3. Results
3.1. Preparation and Characterization of TAC-NE
3.2. Preparation of TAC-NEG
3.3. Release of TAC-NE from TAC-NEG
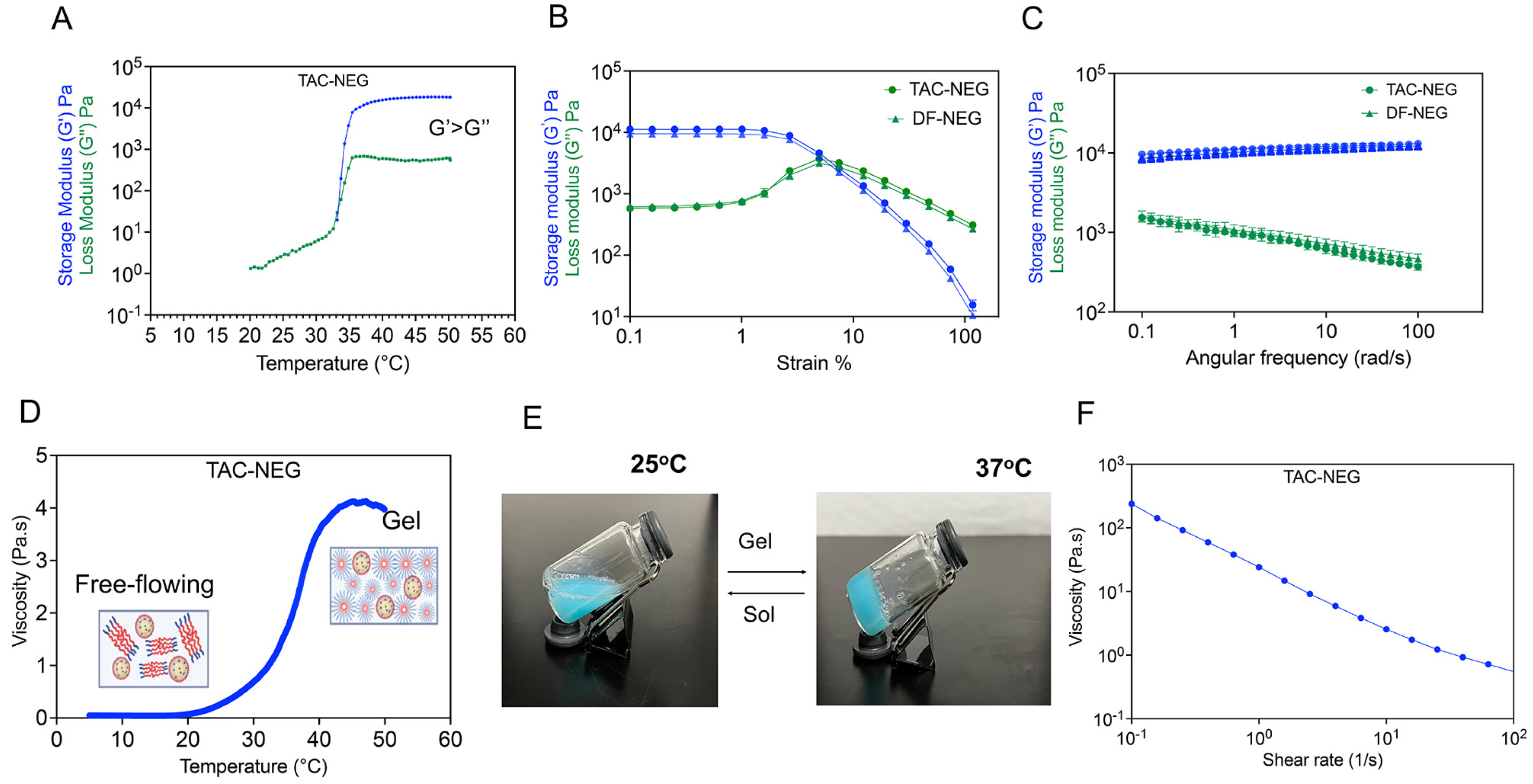
3.4. Rheology
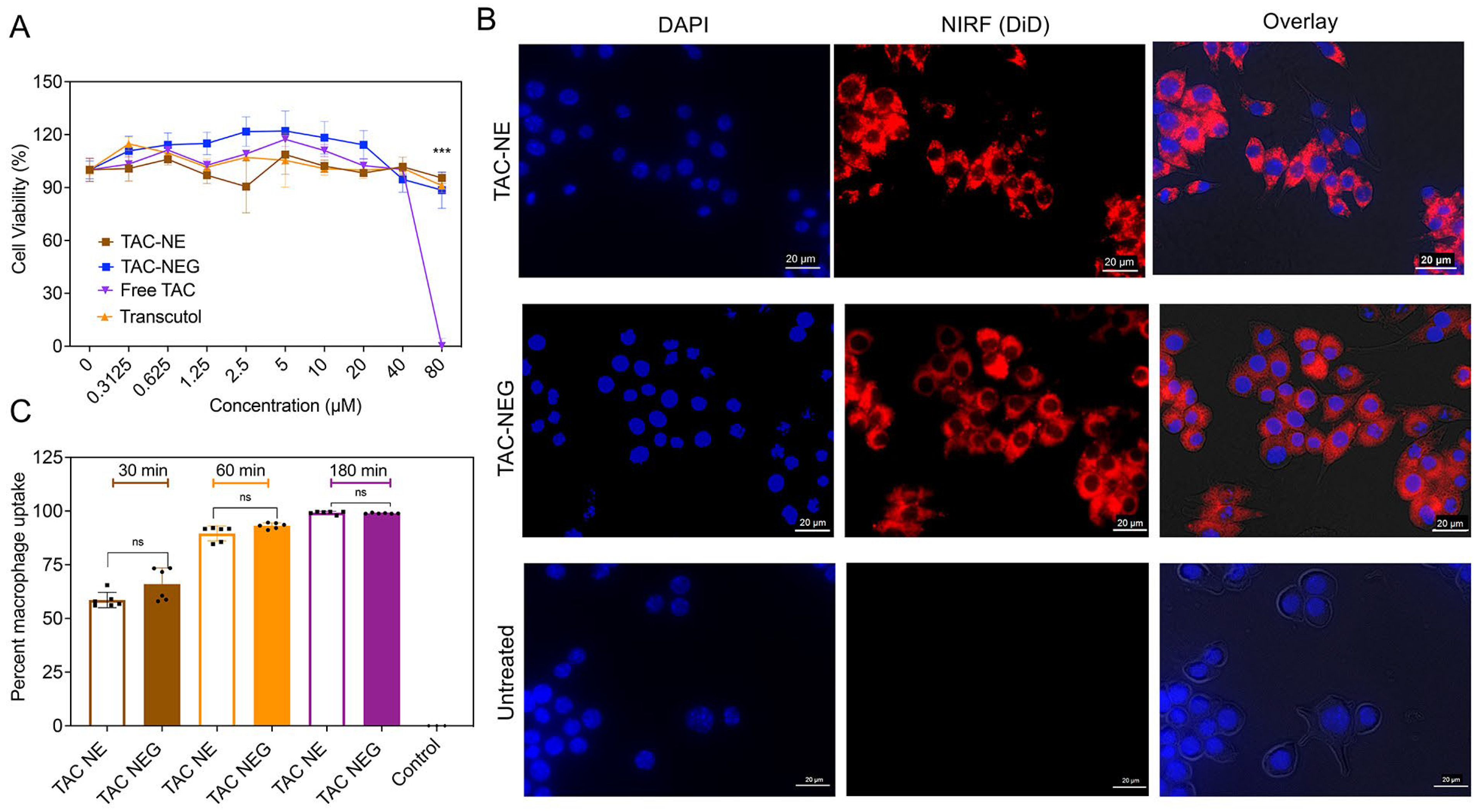
3.5. Macrophage Viability and Cellular Uptake of TAC
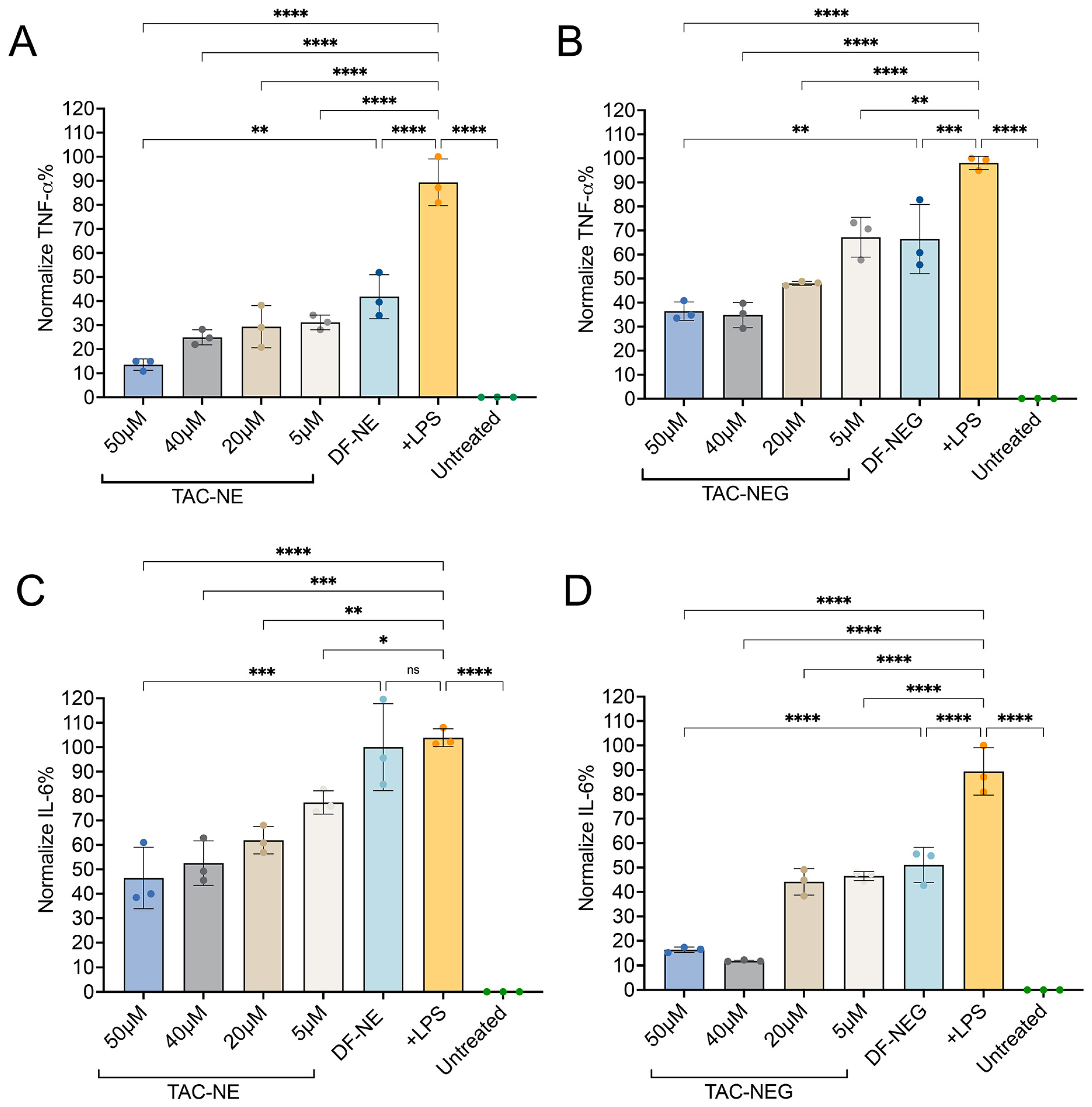
3.6. Enzyme-Linked Immunosorbent Assay
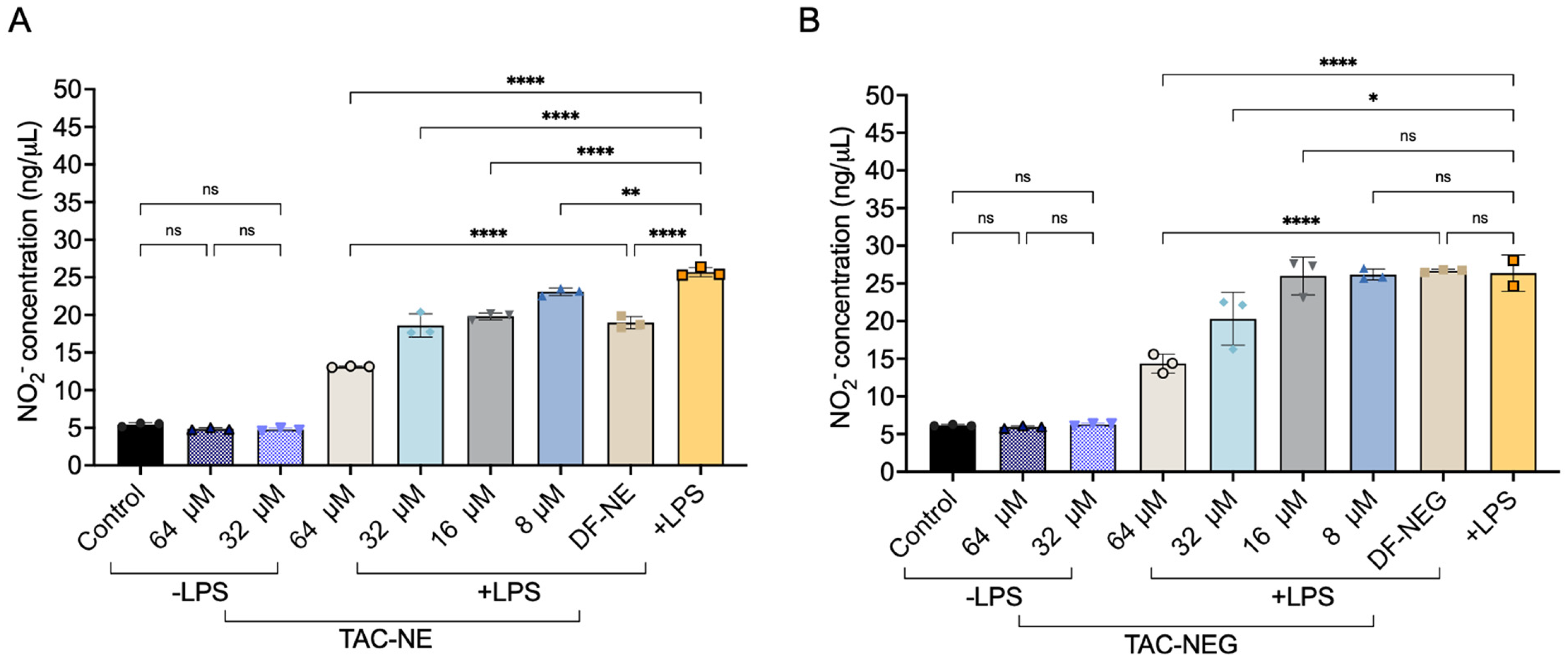
3.7. Nitric Oxide Assay
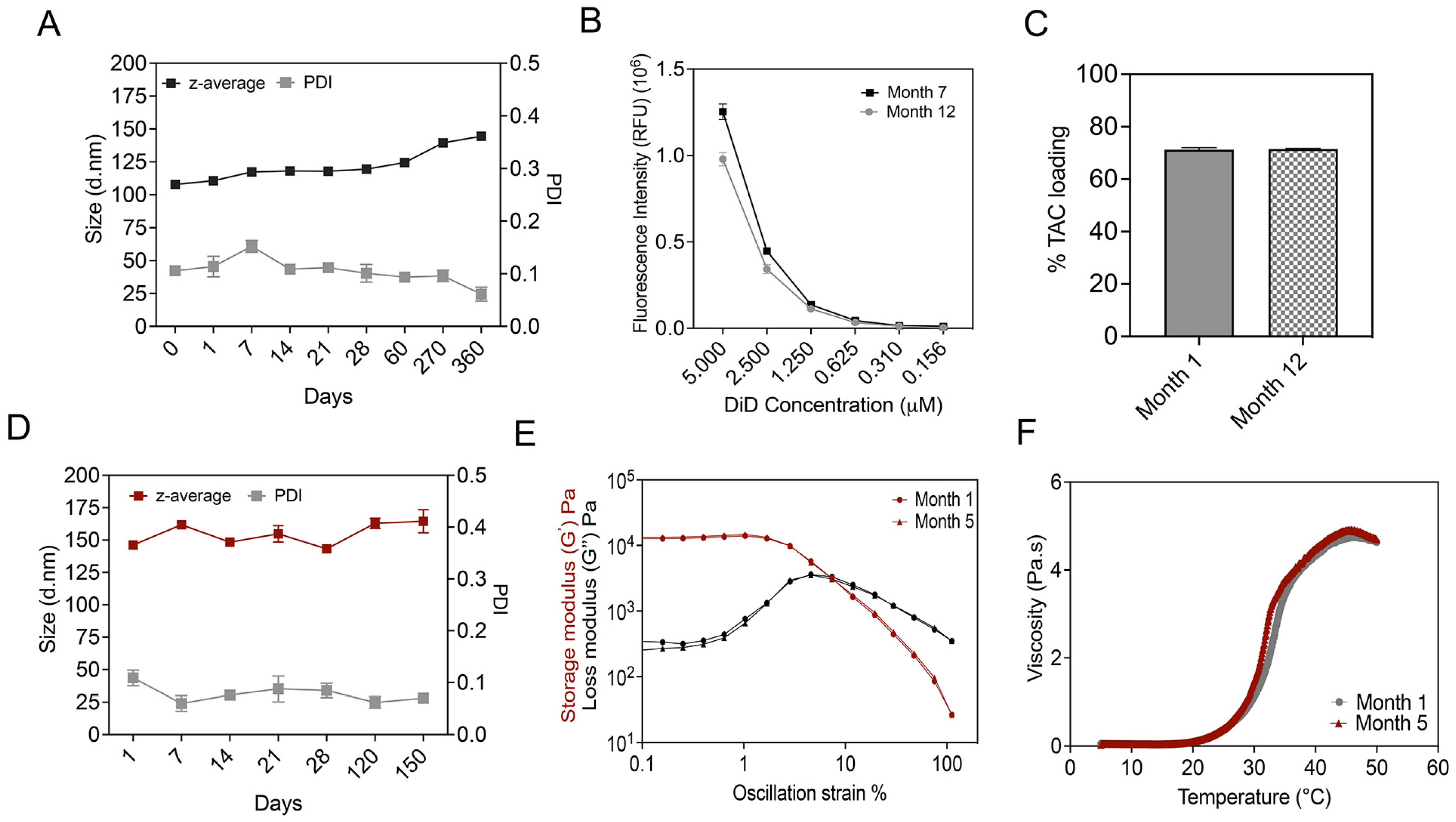
3.8. Long-Term Stability Studies
4. Discussion
5. Conclusions and Translational Relevance
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Giwa, S.; Lewis, J.K.; Alvarez, L.; Langer, R.; Roth, A.E.; Church, G.M.; Markmann, J.F.; Sachs, D.H.; Chandraker, A.; Wertheim, J.A. The promise of organ and tissue preservation to transform medicine. Nat. Biotechnol. 2017, 35, 530–542. [Google Scholar] [CrossRef] [PubMed]
- Kupiec-Weglinski, J.W. Grand challenges in organ transplantation. Front. Transplant. 2022, 1, 3. [Google Scholar] [CrossRef]
- Available online: https://www.organdonor.gov/learn/organ-donation-statistics (accessed on 30 August 2023).
- United Network for Organ Sharing. OPTN/SRTR Annual Report 2006. 2006. Available online: http://www.unos.org/ (accessed on 30 August 2023).
- Gorantla, V.S.; Barker, J.H.; Jones, J.W., Jr.; Prabhune, K.; Maldonado, C.; Granger, D.K. Immunosuppressive agents in transplantation: Mechanisms of action and current anti-rejection strategies. Microsurg. Off. J. Int. Microsurg. Soc. Eur. Fed. Soc. Microsurg. 2000, 20, 420–429. [Google Scholar] [CrossRef]
- Mannon, R.B. Macrophages: Contributors to allograft dysfunction, repair or Innocent bystanders? Curr. Opin. Organ Transplant. 2012, 17, 20. [Google Scholar] [CrossRef] [PubMed]
- Salehi, S.; Reed, E.F. The divergent roles of macrophages in solid organ transplantation. Curr. Opin. Organ Transplant. 2015, 20, 446. [Google Scholar] [CrossRef]
- Toki, D.; Zhang, W.; Hor, K.; Liuwantara, D.; Alexander, S.; Yi, Z.; Sharma, R.; Chapman, J.; Nankivell, B.; Murphy, B. The role of macrophages in the development of human renal allograft fibrosis in the first year after transplantation. Am. J. Transplant. 2014, 14, 2126–2136. [Google Scholar] [CrossRef]
- Kitchens, W.; Chase, C.; Uehara, S.; Cornell, L.; Colvin, R.; Russell, P.; Madsen, J. Macrophage depletion suppresses cardiac allograft vasculopathy in mice. Am. J. Transplant. 2007, 7, 2675–2682. [Google Scholar] [CrossRef]
- Jose, M.D.; Le Meur, Y.; Atkins, R.C.; Chadban, S.J.; Rajotte, R.V. Blockade of macrophage colony-stimulating factor reduces macrophage proliferation and accumulation in renal allograft rejection. Am. J. Transplant. 2003, 3, 294–300. [Google Scholar] [CrossRef]
- Qi, F.; Adair, A.; Ferenbach, D.; Vass, D.G.; Mylonas, K.J.; Kipari, T.; Clay, M.; Kluth, D.C.; Hughes, J.; Marson, L.P. Depletion of cells of monocyte lineage prevents loss of renal microvasculature in murine kidney transplantation. Transplantation 2008, 86, 1267–1274. [Google Scholar] [CrossRef]
- Zielinski, C.E.; Mele, F.; Aschenbrenner, D.; Jarrossay, D.; Ronchi, F.; Gattorno, M.; Monticelli, S.; Lanzavecchia, A.; Sallusto, F. Pathogen-induced human TH17 cells produce IFN-γ or IL-10 and are regulated by IL-1β. Nature 2012, 484, 514–518. [Google Scholar] [CrossRef]
- Fan, H.; Li, L.-X.; Han, D.-D.; Kou, J.-T.; Li, P.; He, Q. Increase of peripheral Th17 lymphocytes during acute cellular rejection in liver transplant recipients. Hepatobiliary Pancreat. Dis. Int. 2012, 11, 606–611. [Google Scholar] [CrossRef]
- Plosker, G.L.; Foster, R.H. Tacrolimus: A further update of its pharmacology and therapeutic use in the management of organ transplantation. Drugs 2000, 59, 323–389. [Google Scholar] [CrossRef] [PubMed]
- European FK506 Multicentre Liver Study Group. Randomised trial comparing tacrolimus (FK506) and cyclosporin in prevention of liver allograft rejection. Lancet 1994, 344, 423–428. [Google Scholar]
- Brooks, E.; Tett, S.E.; Isbel, N.M.; Staatz, C.E. Population pharmacokinetic modelling and Bayesian estimation of tacrolimus exposure: Is this clinically useful for dosage prediction yet? Clin. Pharmacokinet. 2016, 55, 1295–1335. [Google Scholar] [CrossRef]
- Jusko, W.J.; Piekoszewski, W.; Klintmalm, G.B.; Shaefer, M.S.; Hebert, M.F.; Piergies, A.A.; Lee, C.C.; Schechter, P.; Mekki, Q.A. Pharmacokinetics of tacrolimus in liver transplant patients. Clin. Pharmacol. Ther. 1995, 57, 281–290. [Google Scholar] [CrossRef] [PubMed]
- Yu, M.; Liu, M.; Zhang, W.; Ming, Y. Pharmacokinetics, pharmacodynamics and pharmacogenetics of tacrolimus in kidney transplantation. Curr. Drug Metab. 2018, 19, 513–522. [Google Scholar] [CrossRef]
- Thomson, A.; Bonham, C.; Zeevi, A. Mode of action of tacrolimus (FK506): Molecular and cellular mechanisms. Ther. Drug Monit. 1995, 17, 584–591. [Google Scholar] [CrossRef]
- Jennings, C.; Kusler, B.; Jones, P.P. Calcineurin inactivation leads to decreased responsiveness to LPS in macrophages and dendritic cells and protects against LPS-induced toxicity in vivo. Innate Immun. 2009, 15, 109–120. [Google Scholar] [CrossRef]
- Schultze, J.L. Precision attack on calcineurin in macrophages: A new anti-inflammatory weapon. EMBO J. 2014, 33, 1087–1088. [Google Scholar] [CrossRef][Green Version]
- Van den Bosch, T.P.; Kannegieter, N.M.; Hesselink, D.A.; Baan, C.C.; Rowshani, A.T. Targeting the monocyte–macrophage lineage in solid organ transplantation. Front. Immunol. 2017, 8, 153. [Google Scholar] [CrossRef]
- Kannegieter, N.M.; Hesselink, D.A.; Dieterich, M.; Kraaijeveld, R.; Rowshani, A.T.; Leenen, P.J.; Baan, C.C. The effect of tacrolimus and mycophenolic acid on CD14+ monocyte activation and function. PLoS ONE 2017, 12, e0170806. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y.J.; Kusler, B.; Otsuka, M.; Hughes, M.; Suzuki, N.; Suzuki, S.; Yeh, W.-C.; Akira, S.; Han, J.; Jones, P.P. Calcineurin negatively regulates TLR-mediated activation pathways. J. Immunol. 2007, 179, 4598–4607. [Google Scholar] [CrossRef] [PubMed]
- Denhaerynck, K.; Burkhalter, F.; Schäfer-Keller, P.; Steiger, J.; Bock, A.; De Geest, S. Clinical consequences of non adherence to immunosuppressive medication in kidney transplant patients. Transpl. Int. 2009, 22, 441–446. [Google Scholar] [CrossRef] [PubMed]
- Gordon, E.J.; Prohaska, T.; Gallant, M.; Siminoff, L. Adherence to immunosuppression: A prospective diary study. In Transplantation Proceedings; Elsevier: Amsterdam, The Netherlands, 2007; pp. 3081–3085. [Google Scholar]
- Wang, X.; Alfrey, E.; Posselt, A.; Tafra, L.; Alak, A.; Dafoe, D. Intraportal delivery of immunosuppression to intrahepatic islet allograft recipients. Transpl. Int. 1995, 8, 268–272. [Google Scholar] [CrossRef]
- Ruers, T.; Buurman, W.; Smits, J.; Van Der Linden, C.; Van Dongen, J.; Struyker-Boudier, H.; Kootstra, G. Local treatment of renal allografts, a promising way to reduce the dosage of immunosuppressive drugs: Comparison of various ways of administering prednisolone. Transplantation 1986, 41, 156–160. [Google Scholar] [CrossRef]
- Yano, K.; Fukuda, Y.; Sumimoto, R.; Sumimoto, K.; Ito, H.; Dohi, K. Suppression of liver allograft rejection by administration of 15-deoxyspergualin: Comparison of administration via the hepatic artery, portal vein, or systemic circulation. Transpl. Int. 1994, 7, 149–156. [Google Scholar]
- Stepkowski, S.M.; Goto, S.; Ito, T.; Reynolds, K.; Didlake, R.; Kim, E.K.; Kahan, B.D. Prolongation of heterotopic heart allograft survival by local delivery of continuous low-dose cyclosporine therapy. Transplantation 1989, 47, 17–23. [Google Scholar] [CrossRef]
- Schnider, J.T.; Weinstock, M.; Plock, J.A.; Solari, M.G.; Venkataramanan, R.; Zheng, X.X.; Gorantla, V.S. Site-specific immunosuppression in vascularized composite allotransplantation: Prospects and potential. Clin. Dev. Immunol. 2013, 2013, 495212. [Google Scholar] [CrossRef]
- Sultana, N.; Akhtar, J.; Badruddeen, M.I.K.; Ahmad, U.; Arif, M.; Ahmad, M.; Upadhyay, T. Nanoemulgel: For Promising Topical and Systemic Delivery. In Drug Development Life Cycle; InTechOpen: London, UK, 2022. [Google Scholar]
- Al Khateb, K.; Ozhmukhametova, E.K.; Mussin, M.N.; Seilkhanov, S.K.; Rakhypbekov, T.K.; Lau, W.M.; Khutoryanskiy, V.V. In situ gelling systems based on Pluronic F127/Pluronic F68 formulations for ocular drug delivery. Int. J. Pharm. 2016, 502, 70–79. [Google Scholar] [CrossRef]
- Trong, L.C.P.; Djabourov, M.; Ponton, A. Mechanisms of micellization and rheology of PEO–PPO–PEO triblock copolymers with various architectures. J. Colloid Interface Sci. 2008, 328, 278–287. [Google Scholar] [CrossRef]
- Alexandridis, P.; Hatton, T.A. Poly (ethylene oxide)-poly (propylene oxide)-poly (ethylene oxide) block copolymer surfactants in aqueous solutions and at interfaces: Thermodynamics, structure, dynamics, and modeling. Colloids Surf. A Physicochem. Eng. Asp. 1995, 96, 1–46. [Google Scholar] [CrossRef]
- Hashemnejad, S.M.; Badruddoza, A.Z.M.; Zarket, B.; Ricardo Castaneda, C.; Doyle, P.S. Thermoresponsive nanoemulsion-based gel synthesized through a low-energy process. Nat. Commun. 2019, 10, 2749. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.K.; Zhang, Y.; Pollock, J.A.; Janjic, J.M. Cyclooxgenase-2 inhibiting perfluoropoly (ethylene glycol) ether theranostic nanoemulsions—In vitro study. PLoS ONE 2013, 8, e55802. [Google Scholar] [CrossRef]
- Dumortier, G.; Grossiord, J.L.; Agnely, F.; Chaumeil, J.C. A review of poloxamer 407 pharmaceutical and pharmacological characteristics. Pharm. Res. 2006, 23, 2709–2728. [Google Scholar] [CrossRef] [PubMed]
- US Pharmacopeia. USP 34 Microgiological Tests <71> Sterility Tests; Rockville, MD, USA, 2011; pp. 65–70. [Google Scholar]
- Herneisey, M.; Liu, L.; Lambert, E.; Schmitz, N.; Loftus, S.; Janjic, J.M. Development of theranostic perfluorocarbon nanoemulsions as a model non-opioid pain nanomedicine using a quality by design (QbD) approach. AAPS PharmSciTech 2019, 20, 65. [Google Scholar] [CrossRef]
- Janjic, J.M.; Vasudeva, K.; Saleem, M.; Stevens, A.; Liu, L.; Patel, S.; Pollock, J.A. Low-dose NSAIDs reduce pain via macrophage targeted nanoemulsion delivery to neuroinflammation of the sciatic nerve in rat. J. Neuroimmunol. 2018, 318, 72–79. [Google Scholar] [CrossRef]
- Rowe, R.C.; Sheskey, P.; Quinn, M. Handbook of Pharmaceutical Excipients; Libros Digitales-Pharmaceutical Press: London, UK, 2009. [Google Scholar]
- Rosso, A.; Lollo, G.; Chevalier, Y.; Troung, N.; Bordes, C.; Bourgeois, S.; Maniti, O.; Granjon, T.; Dugas, P.-Y.; Urbaniak, S. Development and structural characterization of a novel nanoemulsion for oral drug delivery. Colloids Surf. A Physicochem. Eng. Asp. 2020, 593, 124614. [Google Scholar] [CrossRef]
- Borhade, V.; Nair, H.; Hegde, D. Design and evaluation of self-microemulsifying drug delivery system (SMEDDS) of tacrolimus. Aaps Pharmscitech. 2008, 9, 13–21. [Google Scholar] [CrossRef]
- Nan, A. Miscellaneous drugs, materials, medical devices and techniques. In Side Effects of Drugs Annual; Elsevier: Amsterdam, The Netherlands, 2015; Volume 37, pp. 603–619. [Google Scholar]
- Sarheed, O.; Dibi, M.; Ramesh, K.V. Studies on the effect of oil and surfactant on the formation of alginate-based O/W lidocaine nanocarriers using nanoemulsion template. Pharmaceutics 2020, 12, 1223. [Google Scholar] [CrossRef]
- Mcclements, D.J. Critical review of techniques and methodologies for characterization of emulsion stability. Crit. Rev. Food Sci. Nutr. 2007, 47, 611–649. [Google Scholar] [CrossRef]
- Kovalchuk, V.I.; Loglio, G.; Bykov, A.G.; Ferrari, M.; Krägel, J.; Liggieri, L.; Miller, R.; Milyaeva, O.Y.; Noskov, B.A.; Ravera, F. Effect of temperature on the dynamic properties of mixed surfactant adsorbed layers at the water/hexane interface under low-gravity conditions. Colloids Interfaces 2020, 4, 27. [Google Scholar] [CrossRef]
- Donsì, F.; Wang, Y.; Huang, Q. Freeze–thaw stability of lecithin and modified starch-based nanoemulsions. Food Hydrocoll. 2011, 25, 1327–1336. [Google Scholar] [CrossRef]
- Alexandridis, P.; Holzwarth, J.F.; Hatton, T.A. Micellization of poly (ethylene oxide)-poly (propylene oxide)-poly (ethylene oxide) triblock copolymers in aqueous solutions: Thermodynamics of copolymer association. Macromolecules 1994, 27, 2414–2425. [Google Scholar] [CrossRef]
- Jeong, B.; Kim, S.W.; Bae, Y.H. Thermosensitive sol–gel reversible hydrogels. Adv. Drug Deliv. Rev. 2012, 64, 154–162. [Google Scholar] [CrossRef]
- Gilbert, J.C.; Richardson, J.L.; Davies, M.C.; Palin, K.J.; Hadgraft, J. The effect of solutes and polymers on the gelation properties of pluronic F-127 solutions for controlled drug delivery. J. Control. Release 1987, 5, 113–118. [Google Scholar] [CrossRef]
- Kim, J.H.; Ahn, K.H. Rheological characteristics of poly (ethylene oxide) aqueous solutions under large amplitude oscillatory squeeze flow. Korea-Aust. Rheol. J. 2012, 24, 257–266. [Google Scholar] [CrossRef]
- Lei, W.; Yu, C.; Lin, H.; Zhou, X. Development of tacrolimus-loaded transfersomes for deeper skin penetration enhancement and therapeutic effect improvement in vivo. Asian J. Pharm. Sci. 2013, 8, 336–345. [Google Scholar] [CrossRef]
- Khan, A.S.; Shah, K.U.; Mohaini, M.A.; Alsalman, A.J.; Hawaj, M.A.A.; Alhashem, Y.N.; Ghazanfar, S.; Khan, K.A.; Niazi, Z.R.; Farid, A. Tacrolimus-loaded solid lipid nanoparticle gel: Formulation development and in vitro assessment for topical applications. Gels 2022, 8, 129. [Google Scholar] [CrossRef]
- Hallsten, J.; Vigneswaran, W.T. Cytokine biomarkers as indicators of primary graft dysfunction, acute rejection, and chronic lung allograft dysfunction in lung transplant recipients: A review. Perioper. Care Organ Transpl. Recip. 2019. [Google Scholar] [CrossRef]
- Mathur, A.; Baz, M.; Staples, E.D.; Bonnell, M.; Speckman, J.M.; Hess Jr, P.J.; Klodell, C.T.; Knauf, D.G.; Moldawer, L.L.; Beaver, T.M. Cytokine profile after lung transplantation: Correlation with allograft injury. Ann. Thorac. Surg. 2006, 81, 1844–1850. [Google Scholar] [CrossRef]
- Grenz, A.; Schenk, M.; Zipfel, A.; Viebahn, R. TNF-α and its receptors mediate graft rejection and loss after liver transplantation. Clin. Chem. Lab. Med. 2000, 38. [Google Scholar] [CrossRef]
- Rizzo, M.; SivaSai, K.S.; Smith, M.A.; Trulock, E.P.; Lynch, J.P.; Patterson, G.A.; Mohanakumar, T. Increased expression of inflammatory cytokines and adhesion molecules by alveolar macrophages of human lung allograft recipients with acute rejection: Decline with resolution of rejection. J. Heart Lung Transplant. 2000, 19, 858–865. [Google Scholar] [CrossRef]
- Yang, Z.F.; Ho, D.W.; Lau, C.K.; Lam, C.T.; Lum, C.T.; Poon, R.T.; Fan, S.T. Allograft inflammatory factor-1 (AIF-1) is crucial for the survival and pro-inflammatory activity of macrophages. Int. Immunol. 2005, 17, 1391–1397. [Google Scholar] [CrossRef] [PubMed]
- Watano, K.; Iwabuchi, K.; Fujii, S.; Ishimori, N.; Mitsuhashi, S.; Ato, M.; Kitabatake, A.; Onoé, K. Allograft inflammatory factor-1 augments production of interleukin-6,-10and-12 by a mouse macrophage line. Immunology 2001, 104, 307–316. [Google Scholar] [CrossRef] [PubMed]
- Uzbay, I.T.; Oglesby, M.W. Nitric oxide and substance dependence. Neurosci. Biobehav. Rev. 2001, 25, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Střeštiková, P.; Otova, B.; Filipec, M.; Farghali, H. Inhibitory effect of FK 506 and cyclosporin A on nitric oxide production by LPS-treated cultured rat macrophages. Physiol. Res. 2000, 49, 725–728. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vichare, R.; Crelli, C.; Liu, L.; Das, A.C.; McCallin, R.; Zor, F.; Kulahci, Y.; Gorantla, V.S.; Janjic, J.M. A Reversibly Thermoresponsive, Theranostic Nanoemulgel for Tacrolimus Delivery to Activated Macrophages: Formulation and In Vitro Validation. Pharmaceutics 2023, 15, 2372. https://doi.org/10.3390/pharmaceutics15102372
Vichare R, Crelli C, Liu L, Das AC, McCallin R, Zor F, Kulahci Y, Gorantla VS, Janjic JM. A Reversibly Thermoresponsive, Theranostic Nanoemulgel for Tacrolimus Delivery to Activated Macrophages: Formulation and In Vitro Validation. Pharmaceutics. 2023; 15(10):2372. https://doi.org/10.3390/pharmaceutics15102372
Chicago/Turabian StyleVichare, Riddhi, Caitlin Crelli, Lu Liu, Amit Chandra Das, Rebecca McCallin, Fatih Zor, Yalcin Kulahci, Vijay S. Gorantla, and Jelena M. Janjic. 2023. "A Reversibly Thermoresponsive, Theranostic Nanoemulgel for Tacrolimus Delivery to Activated Macrophages: Formulation and In Vitro Validation" Pharmaceutics 15, no. 10: 2372. https://doi.org/10.3390/pharmaceutics15102372
APA StyleVichare, R., Crelli, C., Liu, L., Das, A. C., McCallin, R., Zor, F., Kulahci, Y., Gorantla, V. S., & Janjic, J. M. (2023). A Reversibly Thermoresponsive, Theranostic Nanoemulgel for Tacrolimus Delivery to Activated Macrophages: Formulation and In Vitro Validation. Pharmaceutics, 15(10), 2372. https://doi.org/10.3390/pharmaceutics15102372








