Increased Water-Solubility and Maintained Antioxidant Power of Resveratrol by Its Encapsulation in Vitamin E TPGS Micelles: A Potential Nutritional Supplement for Chronic Liver Disease
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Solubility Studies
2.3. Formulations Preparation
2.3.1. Solvent Diffusion Evaporation Method
2.3.2. Solution Casting Method
2.4. Determination of the Encapsulation Efficiency and Drug Loading Capacity
2.5. Determination of Micelle Size, Polydispersity Index (PDI), and Zeta (ƺ) Potential
Re-Dispersibility Study of Lyophilized RES-TPGS Micelles
2.6. Principal Component Analysis (PCA)-Assisted FTIR Spectroscopy
Chemometric Analysis: PCA
2.7. Differential Scanning Calorimetry (DSC)
2.8. Formulation Stability
2.9. In Vitro Release Studies
2.10. Determination of the Radical Scavenging Activity (RSA) of RES-TPGSs
2.11. Cytotoxicity Evaluations
2.11.1. Cell Culture
2.11.2. Viability Assay
2.12. Statistical Analyses
3. Results and Discussion
3.1. TPGS Suitability for RES Encapsulation
3.2. Preparation and Characterization of RES-TPGS Formulations
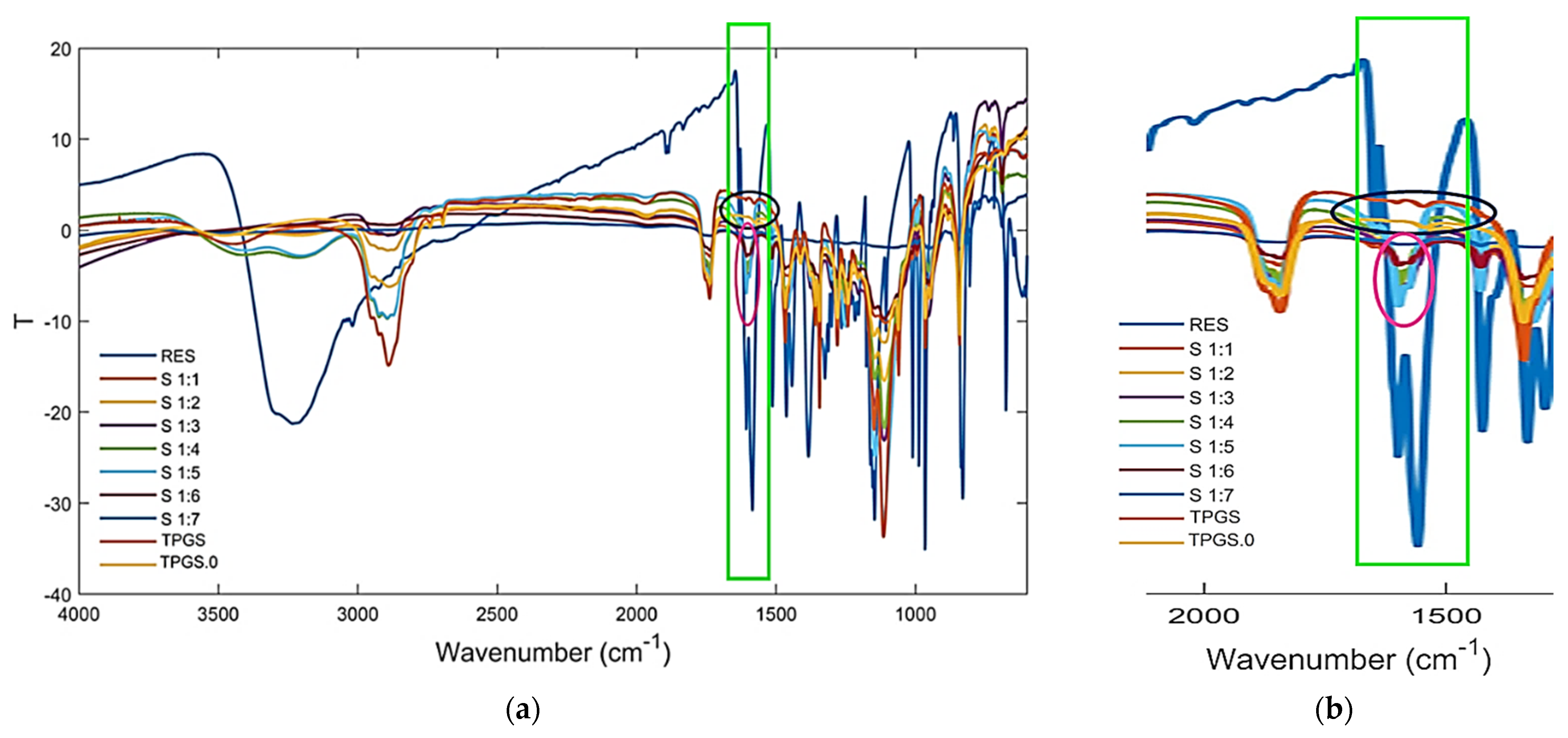
3.3. FTIR Analysis Assisted by PCA
3.3.1. FTIR Spectra
3.3.2. Principal Component Analysis (PCA)
3.4. Thermal Analysis Using Differential Scanning Calorimetry (DSC)
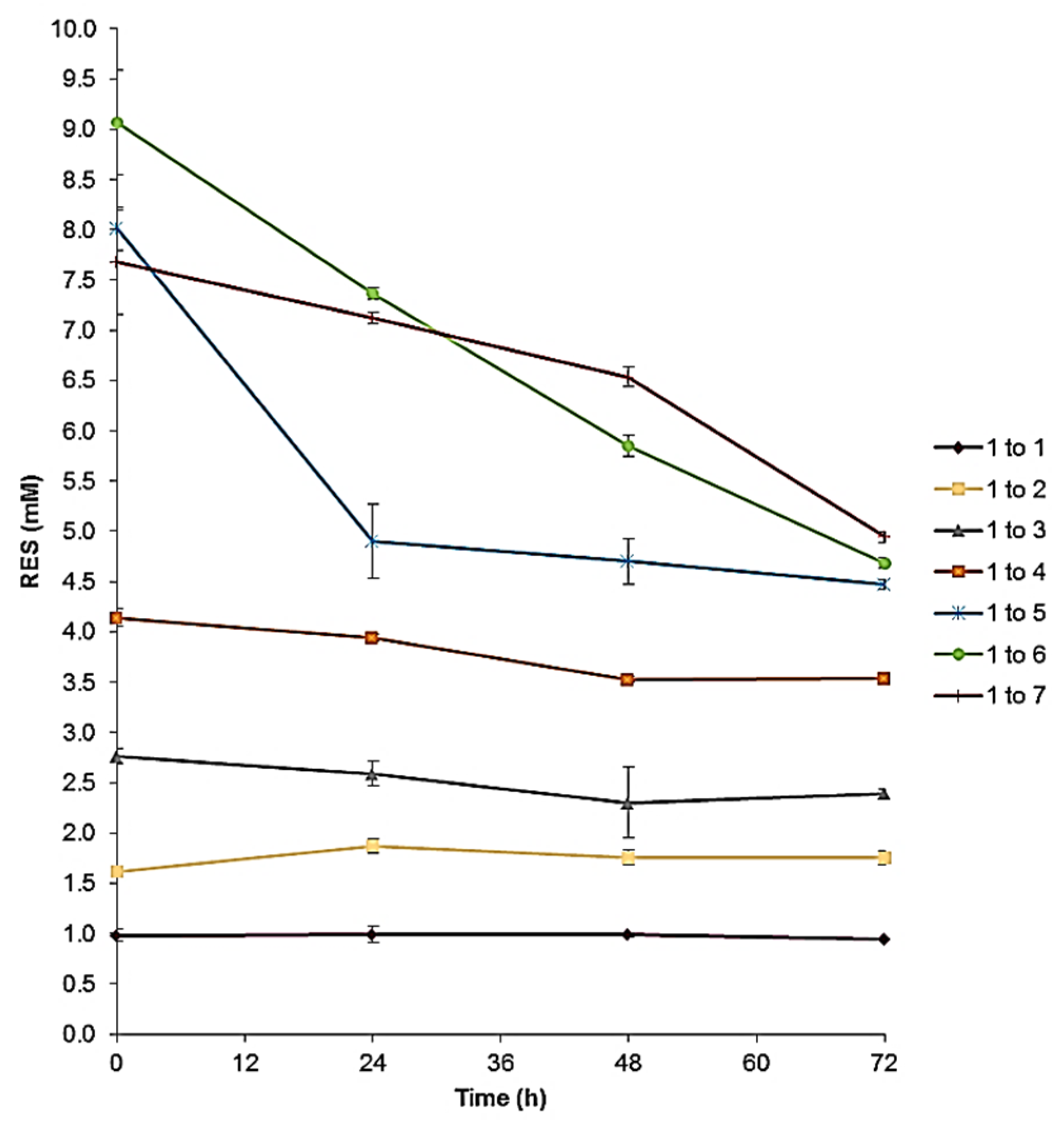
3.5. Stability of RES-TPGSs
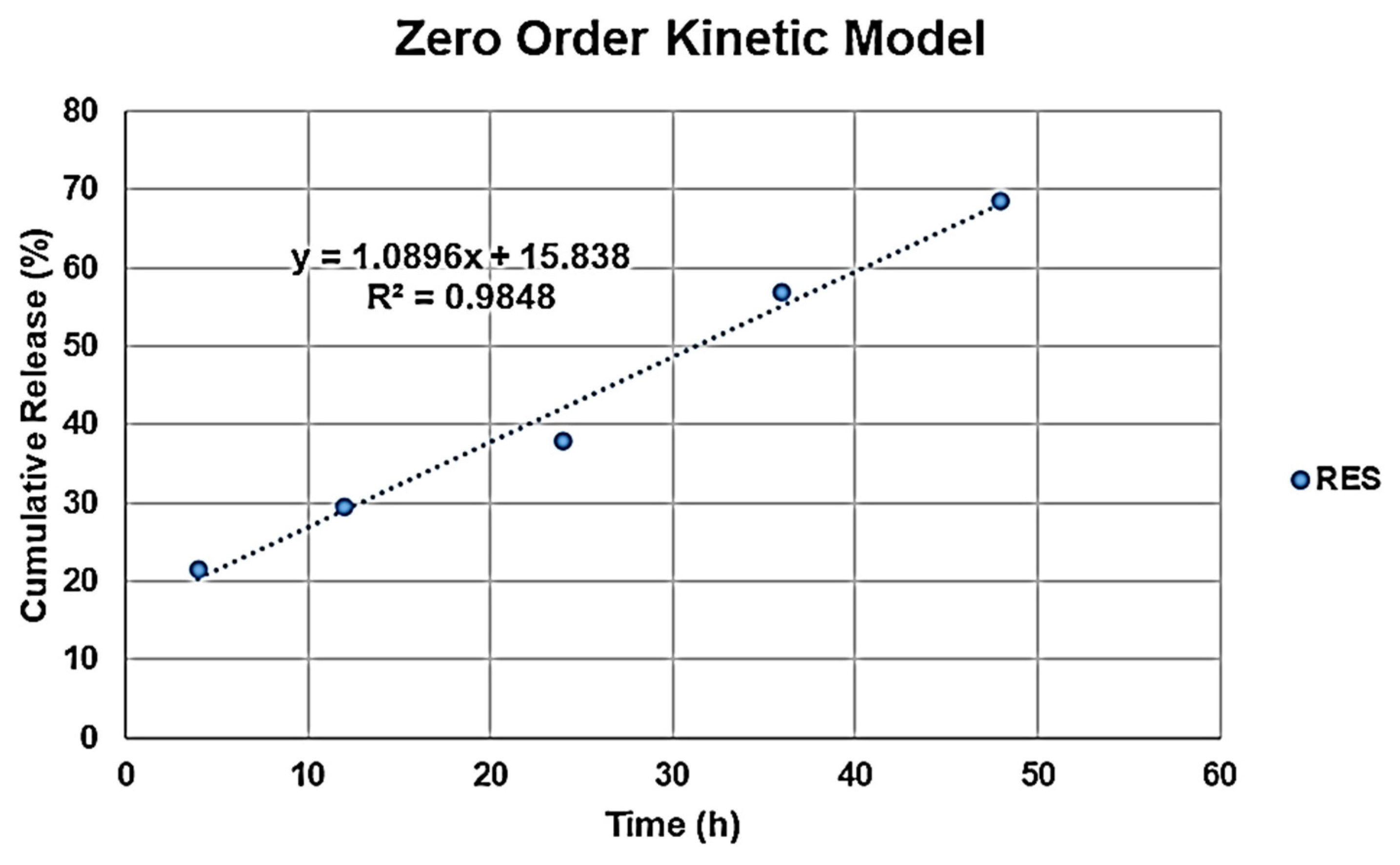
3.6. In Vitro Drug Release Studies
3.7. Antioxidant Activity of RES
3.8. RES-TPGSs’ Effects on HaCaT Cells
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Squires, J.E.; McKiernan, P. Molecular Mechanisms in Pediatric Cholestasis. Gastroenterol. Clin. N. Am. 2018, 47, 921–937. [Google Scholar] [CrossRef] [PubMed]
- Feldman, A.G.; Sokol, R.J. Recent Developments in Diagnostics and Treatment of Neonatal Cholestasis. Semin. Pediatr. Surg. 2020, 29, 150945. [Google Scholar] [CrossRef]
- Santos, J.L.; Choquette, M.; Bezerra, J.A. Cholestatic Liver Disease in Children. Curr. Gastroenterol. Rep. 2010, 12, 30–39. [Google Scholar] [CrossRef] [Green Version]
- Yang, C.H.; Perumpail, B.J.; Yoo, E.R.; Ahmed, A.; Kerner, J.A., Jr. Nutritional Needs and Support for Children with Chronic Liver Disease. Nutrients 2017, 9, 1127. [Google Scholar] [CrossRef] [PubMed]
- Sokol, R.J. A New Old Treatment for Vitamin E Deficiency in Cholestasis. J. Pediatr. Gastroenterol. Nutr. 2016, 63, 577–578. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- European Food Safety Agency (EFSA). Opinion of the Scientific Panel on food additives, flavourings, processing aids and materials in contact with food (AFC) related to D-alpha-tocopheryl polyethylene glycol 1000 succinate (TPGS) in use for food for particular nutritional purposes. EFSA J. 2007, 490, 1–20. [Google Scholar] [CrossRef]
- Thébaut, A.; Nemeth, A.; Le Mouhaër, J.; Scheenstra, R.; Baumann, U.; Koot, B.; Gottrand, F.; Houwen, R.; Monard, L.; de Micheaux, S.L.; et al. Oral Tocofersolan Corrects or Prevents Vitamin E Deficiency in Children With Chronic Cholestasis. J. Pediatr. Gastroenterol. Nutr. 2016, 63, 610–615. [Google Scholar] [CrossRef]
- Socha, P.; Koletzko, B.; Pawlowska, J.; Proszynska, K.; Socha, J. Treatment of Cholestatic Children with Water-Soluble Vitamin E (α-Tocopheryl Polyethylene Glycol Succinate): Effects on Serum Vitamin E, Lipid Peroxides, and Polyunsaturated Fatty Acids. J. Pediatr. Gastroenterol. Nutr. 1997, 24, 189–193. [Google Scholar] [CrossRef]
- Novelle, M.G.; Wahl, D.; Diéguez, C.; Bernier, M.; de Cabo, R. Resveratrol Supplementation: Where Are We Now and Where Should We Go? Ageing Res. Rev. 2015, 21, 1–15. [Google Scholar] [CrossRef] [Green Version]
- Wang, T.; Zhou, Z.; Sun, L.; Li, X.; Xu, Z.; Chen, M.; Zhao, G.; Jiang, Z.; Zhang, L. Resveratrol Effectively Attenuates α-Naphthyl-Isothiocyanate-Induced Acute Cholestasis and Liver Injury through Choleretic and Anti-Inflammatory Mechanisms. Acta Pharmacol. Sin. 2014, 35, 1527–1536. [Google Scholar] [CrossRef] [Green Version]
- Ding, L.; Zhang, B.; Li, J.; Yang, L.; Wang, Z. Beneficial effect of resveratrol on α-naphthyl isothiocyanate-induced cholestasis via regulation of the FXR pathway. Mol. Med. Rep. 2018, 17, 1863–1872. [Google Scholar] [CrossRef] [Green Version]
- Amri, A.; Chaumeil, J.C.; Sfar, S.; Charrueau, C. Administration of Resveratrol: What Formulation Solutions to Bioavailability Limitations? J. Control. Release 2012, 158, 182–193. [Google Scholar] [CrossRef]
- Berman, A.Y.; Motechin, R.A.; Wiesenfeld, M.Y.; Holz, K.M. The therapeutic potential of resveratrol: A review of clinical trials. NPJ Precis. Oncol. 2017, 1, 35. [Google Scholar] [CrossRef] [Green Version]
- Löbenberg, R.; Amidon, G.L. Modern bioavailability, bioequivalence and biopharmaceutics classification system. New scientific approaches to international regulatory standards. Eur. J. Pharm. Biopharm. 2000, 50, 3–12. [Google Scholar] [CrossRef]
- Wenzel, E.; Soldo, T.; Erbersdobler, H.; Somoza, V. Bioactivity and Metabolism of trans-Resveratrol Orally Administered to Wistar Rats. Mol. Nutr. Food Res. 2005, 49, 482–494. [Google Scholar] [CrossRef]
- Maier-Salamon, A.; Hagenauer, B.; Wirth, M.; Gabor, F.; Szekeres, T.; Jäger, W. Increased Transport of Resveratrol Across Monolayers of the Human Intestinal Caco-2 Cells Is Mediated by Inhibition and Saturation of Metabolites. Pharm. Res. 2006, 23, 2107–2115. [Google Scholar] [CrossRef]
- Chimento, A.; De Amicis, F.; Sirianni, R.; Sinicropi, M.S.; Puoci, F.; Casaburi, I.; Saturnino, C.; Pezzi, V. Progress to Improve Oral Bioavailability and Beneficial Effects of Resveratrol. Int. J. Mol. Sci. 2019, 20, 1381. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Summerlin, N.; Soo, E.; Thakur, S.; Qu, Z.; Jambhrunkar, S.; Popat, A. Resveratrol Nanoformulations: Challenges and Opportunities. Int. J. Pharm. 2015, 479, 282–290. [Google Scholar] [CrossRef]
- Zuccari, G.; Baldassari, S.; Alfei, S.; Marengo, B.; Valenti, G.E.; Domenicotti, C.; Ailuno, G.; Villa, C.; Marchitto, L.; Caviglioli, G. D-α-Tocopherol-Based Micelles for Successful Encapsulation of Retinoic Acid. Pharmaceuticals 2021, 14, 212. [Google Scholar] [CrossRef]
- Sokol, R.J.; Johnson, K.E.; Karrer, F.M.; Narkewicz, M.R.; Smith, D.; Kam, I. Improvement of Cyclosporin Absorption in Children after Liver Transplantation by Means of Water-Soluble Vitamin E. Lancet 1991, 338, 212–215. [Google Scholar] [CrossRef]
- Yu, L.; Bridgers, A.; Polli, J.; Vickers, A.; Long, S.; Roy, A.; Winnike, R.; Coffin, M. Vitamin E-TPGS increases absorption flux of an HIV protease inhibitor by enhancing its solubility and permeability. Pharm. Res. 1999, 16, 1812–1817. [Google Scholar] [CrossRef]
- Varma, M.V.S.; Panchagnula, R. Enhanced Oral Paclitaxel Absorption with Vitamin E-TPGS: Effect on Solubility and Permeability in Vitro, in Situ and in Vivo. Eur. J. Pharm. Sci. 2005, 25, 445–453. [Google Scholar] [CrossRef]
- Mahajan, H.S.; Patil, P.H. Central Composite Design-Based Optimization of Lopinavir Vitamin E-TPGS Micelle: In Vitro Characterization and in Vivo Pharmacokinetic Study. Colloids Surf. B Biointerfaces 2020, 194, 111149. [Google Scholar] [CrossRef]
- Li, H.; Yan, L.; Tang, E.K.Y.; Zhang, Z.; Chen, W.; Liu, G.; Mo, J. Synthesis of TPGS/Curcumin Nanoparticles by Thin-Film Hydration and Evaluation of Their Anti-Colon Cancer Efficacy In Vitro and In Vivo. Front. Pharmacol. 2019, 10, 769. [Google Scholar] [CrossRef] [Green Version]
- Fine-Shamir, N.; Beig, A.; Dahan, A. Adequate Formulation Approach for Oral Chemotherapy: Etoposide Solubility, Permeability, and Overall Bioavailability from Cosolvent- vs. Vitamin E TPGS-Based Delivery Systems. Int. J. Pharm. 2021, 597, 120295. [Google Scholar] [CrossRef]
- Hu, M.; Zhang, J.; Ding, R.; Fu, Y.; Gong, T.; Zhang, Z. Improved oral bioavailability and therapeutic efficacy of dabigatran etexilate via Soluplus-TPGS binary mixed micelles system. Drug Dev. Ind. Pharm. 2017, 43, 687–697. [Google Scholar] [CrossRef] [PubMed]
- Kesharwani, S.S.; Kaur, S.; Tummala, H.; Sangamwar, A.T. Multifunctional Approaches Utilizing Polymeric Micelles to Circumvent Multidrug Resistant Tumors. Colloids Surf. B Biointerfaces 2019, 173, 581–590. [Google Scholar] [CrossRef] [PubMed]
- Holzer, M.; Vogel, V.; Mäntele, W.; Schwartz, D.; Haase, W.; Langer, K. Physico-Chemical Characterisation of PLGA Nanoparticles after Freeze-Drying and Storage. Eur. J. Pharm. Biopharm. 2009, 72, 428–437. [Google Scholar] [CrossRef] [PubMed]
- Ghezzi, M.; Pescina, S.; Padula, C.; Santi, P.; Del Favero, E.; Cantù, L.; Nicoli, S. Polymeric Micelles in Drug Delivery: An Insight of the Techniques for Their Characterization and Assessment in Biorelevant Conditions. J. Control. Release 2021, 332, 312–336. [Google Scholar] [CrossRef]
- Turrini, F.; Zunin, P.; Catena, S.; Villa, C.; Alfei, S.; Boggia, R. Traditional or Hydro-Diffusion and Gravity Microwave Coupled with Ultrasound as Green Technologies for the Valorization of Pomegranate External Peels. Food Bioprod. Process. 2019, 117, 30–37. [Google Scholar] [CrossRef]
- Di Paolo, D.; Pastorino, F.; Zuccari, G.; Caffa, I.; Loi, M.; Marimpietri, D.; Brignole, C.; Perri, P.; Cilli, M.; Nico, B.; et al. Enhanced Anti-Tumor and Anti-Angiogenic Efficacy of a Novel Liposomal Fenretinide on Human Neuroblastoma. J. Control. Release 2013, 170, 445–451. [Google Scholar] [CrossRef]
- Wan, S.; Zhang, L.; Quan, Y.; Wei, K. Resveratrol-Loaded PLGA Nanoparticles: Enhanced Stability, Solubility and Bioactivity of Resveratrol for Non-Alcoholic Fatty Liver Disease Therapy. R. Soc. Open Sci. 2018, 5, 181457. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sadoqi, M.; Lau-Cam, C.A.; Wu, S.H. Investigation of the Micellar Properties of the Tocopheryl Polyethylene Glycol Succinate Surfactants TPGS 400 and TPGS 1000 by Steady State Fluorometry. J. Colloid Interface Sci. 2009, 333, 585–589. [Google Scholar] [CrossRef]
- Sheu, M.T.; Chen, S.Y.; Chen, L.C.; Ho, H.O. Influence of micelle solubilization by tocopheryl polyethylene glycol succinate (TPGS) on solubility enhancement and percutaneous penetration of estradiol. J. Control. Release 2003, 88, 355–368. [Google Scholar] [CrossRef]
- Dhakar, N.K.; Matencio, A.; Caldera, F.; Argenziano, M.; Cavalli, R.; Dianzani, C.; Zanetti, M.; López-Nicolás, J.M.; Trotta, F. Comparative Evaluation of Solubility, Cytotoxicity and Photostability Studies of Resveratrol and Oxyresveratrol Loaded Nanosponges. Pharmaceutics 2019, 11, 54. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chang, C.-W.; Wong, C.-Y.; Wu, Y.-T.; Hsu, M.-C. Development of a Solid Dispersion System for Improving the Oral Bioavailability of Resveratrol in Rats. Eur. J. Drug Metab. Pharmacokinet. 2017, 42, 239–249. [Google Scholar] [CrossRef] [PubMed]
- Machado, N.D.; Fernández, M.A.; Díaz, D.D. Recent Strategies in Resveratrol Delivery Systems. Chem. Plus Chem. 2019, 84, 951–973. [Google Scholar] [CrossRef] [PubMed]
- Akbas, S.; Sahin, A.; Calis, S.; Oncel, H.; Capan, Y. Characterization of Bevacizumab by Dynamic Light Scattering While Maintaining Its Native Structure. Pharmazie 2018, 369–374. [Google Scholar] [CrossRef]
- Jain, S.; Pandey, S.; Sola, P.; Pathan, H.; Patil, R.; Ray, D.; Aswal, V.K.; Bahadur, P.; Tiwari, S. Solubilization of Carbamazepine in TPGS Micelles: Effect of Temperature and Electrolyte Addition. AAPS PharmSciTech 2019, 20, 203. [Google Scholar] [CrossRef]
- Bandi, S.P.; Kumbhar, Y.S.; Venuganti, V.V.K. Effect of particle size and surface charge of nanoparticles in penetration through intestinal mucus barrier. J. Nanopart. Res. 2020, 22, 62. [Google Scholar] [CrossRef]
- Saez, A.; Guzmán, M.; Molpeceres, J.; Aberturas, M.R. Freeze-Drying of Polycaprolactone and Poly(d,l-Lactic-Glycolic) Nanoparticles Induce Minor Particle Size Changes Affecting the Oral Pharmacokinetics of Loaded Drugs. Eur. J. Pharm. Biopharm. 2000, 50, 379–387. [Google Scholar] [CrossRef]
- Tiwari, S.; Sarolia, J.; Kansara, V.; Chudasama, N.A.; Prasad, K.; Ray, D.; Aswal, V.K.; Bahadur, P. Synthesis, Colloidal Characterization and Targetability of Phenylboronic Acid Functionalized α-Tocopheryl Polyethylene Glycol Succinate in Cancer Cells. Polymers 2020, 12, 2258. [Google Scholar] [CrossRef]
- Foss. Available online: https://www.fossanalytics.com/it-it/news-articles/technologies/a-short-intro-to-ftir-analysis (accessed on 15 June 2021).
- Alfei, S.; Oliveri, P.; Malegori, C. Assessment of the Efficiency of a Nanospherical Gallic Acid Dendrimer for Long-Term Preservation of Essential Oils: An Integrated Chemometric-Assisted FTIR Study. Chem. Sel. 2019, 4, 8891–8901. [Google Scholar] [CrossRef]
- Alfei, S.; Marengo, B.; Domenicotti, C. Development of a fast, low cost, conservative and eco-friendly method for quantifying gallic acid in polymeric formulations by FTIR spectroscopy in solution. Chem. Sel. 2020, 5, 4381–4388. [Google Scholar] [CrossRef]
- Christiansen, A.; Backensfeld, T.; Kühn, S.; Weitschies, W. Investigating the Stability of the Nonionic Surfactants Tocopheryl Polyethylene Glycol Succinate and Sucrose Laurate by HPLC–MS, DAD, and CAD. J. Pharm. Sci. 2011, 100, 1773–1782. [Google Scholar] [CrossRef]
- Mircioiu, C.; Voicu, V.; Anuta, V.; Tudose, A.; Celia, C.; Paolino, D.; Fresta, M.; Sandulovici, R.; Mircioiu, I. Mathematical Modeling of Release Kinetics from Supramolecular Drug Delivery Systems. Pharmaceutics 2019, 11, 140. [Google Scholar] [CrossRef] [Green Version]
- Bruschi, M.L. (Ed.) Mathematical models of drug release. In Strategies to Modify the Drug Release from Pharmaceutical Systems; Woodhead Publishing: Cambridge, UK, 2015; pp. 63–86. ISBN 978-0-08-100092-2. [Google Scholar]
- Maupas, C.; Moulari, B.; Béduneau, A.; Lamprecht, A.; Pellequer, Y. Surfactant Dependent Toxicity of Lipid Nanocapsules in HaCaT Cells. Int. J. Pharm. 2011, 411, 136–141. [Google Scholar] [CrossRef]
- Zhao, B.; Wang, X.Q.; Wang, X.Y.; Zhang, H.; Dai, W.B.; Wang, J.; Zhong, Z.L.; Wu, H.N.; Zhang, Q. Nanotoxicity comparison of four amphiphilic polymeric micelles with similar hydrophilic or hydrophobic structure. Part. Fibre Toxicol. 2013, 10, 47. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rocha, V.; Marques, C.; Figueiredo, J.L.; Gaio, A.R.; Costa, P.C.; Lobo, J.M.S.; Almeida, I.F. In Vitro Cytotoxicity Evaluation of Resveratrol-Loaded Nanoparticles: Focus on the Challenges of in Vitro Methodologies. Food Chem. Toxicol. 2017, 103, 214–222. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Wu, T.; Qi, Y.; Zhang, Z. Recent Advances in the Application of Vitamin E TPGS for Drug Delivery. Theranostics 2018, 8, 464–485. [Google Scholar] [CrossRef] [PubMed]


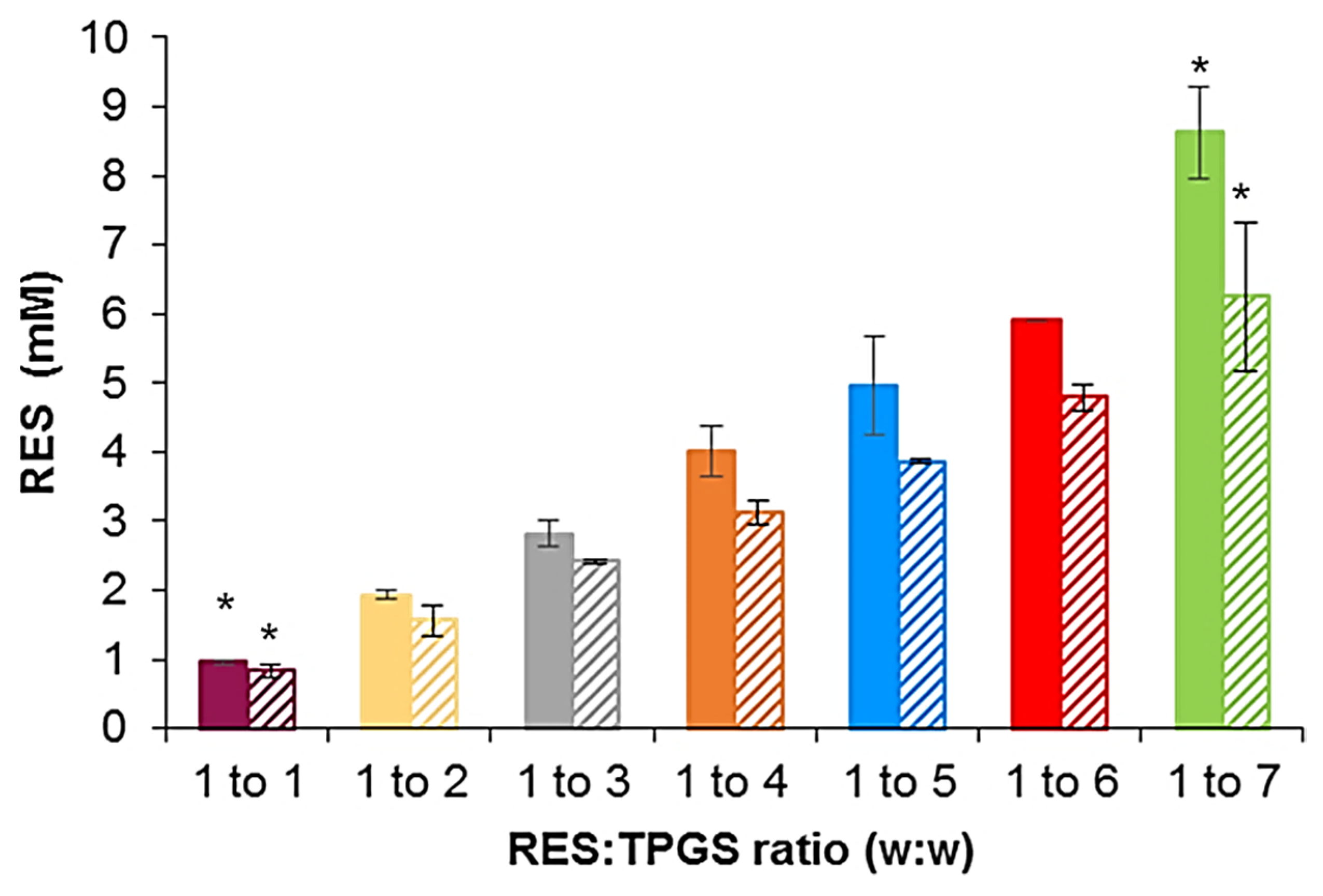




| RES:TPGS (w:w) Ratio in the Preparative Mixture | EE% | DL% |
|---|---|---|
| 1:1 | 10 ± 1 * | 9 ± 1 |
| 1:2 | 20 ± 2 | 10 ± 2 |
| 1:3 | 31 ± 3 | 11 ± 1 |
| 1:4 | 44 ± 5 | 11 ± 2 |
| 1:5 | 56 ± 6 | 11 ± 3 |
| 1:6 | 76 ± 1 * | 13 ± 1 |
| 1:7 | 92 ± 6 * | 15 ± 2 * |
| RES:TPGS (w:w) | Size (nm) Fresh Empty | PDI Fresh Empty | Size (nm) Fresh Loaded | PDI Fresh Loaded | ƺ (mV) Loaded | Size (nm) Lyophilized | PDI Lyophilized |
|---|---|---|---|---|---|---|---|
| 1:1 | 14.1 ± 0.6 | 0.27 ± 0.05 | 12.5 ± 0.2 | 0.26 ± 0.04 * | −2.2 ± 3.6 | 12.2 ± 0.3 | 0.35 ± 0.06 * |
| 1:2 | 20.1 ± 0.7 * | 0.20 ± 0.02 | 12.7 ± 0.8 | 0.24 ± 0.01 | −3.6 ± 4.13 | 13.1 ± 0.6 | 0.27 ± 0.02 |
| 1:3 | 14.7 ± 0.1 | 0.31 ± 0.09 * | 12.0 ± 0.3 | 0.21 ± 0.07 | −3.1 ± 3.3 | 14.0 ± 0.7 | 0.32 ± 0.04 |
| 1:4 | 13.2 ± 0.3 | 0.26 ± 0.03 | 11.9 ± 0.8 | 0.19 ± 0.04 | −4.1 ± 2.4 | 15.0 ± 0.3 * | 0.212 ± 0.001 * |
| 1:5 | 12.3 ± 0.5 | 0.10 ± 0.02 * | 11.9 ± 0.2 | 0.24 ± 0.05 | −4.6 ± 7.7 * | 13.3 ± 0.4 | 0.298 ± 0.009 |
| 1:6 | 12.3 ± 0.2 | 0.126 ± 0.001 * | 9.13 ± 0.01 * | 0.13 ± 0.01 * | −4.8 ± 3.0 * | 9.7 ± 0.8 * | 0.28 ± 0.04 |
| 1:7 | 13.2 ± 0.4 | 0.21 ± 0.04 | 9.6 ± 0.3 * | 0.148 ± 0.001 * | −1.6 ± 3.9 * | 11.9 ± 0.1 | 0.245 ± 0.001 |
| Sample | RSA% |
|---|---|
| RES pristine powder | 16.37 ± 0.28 |
| Freeze-dried RES pristine powder | 15.71 ± 0.63 |
| RES:TPGS 1:2 | 15.93 ± 0.02 |
| RES:TPGS 1:6 | 17.96 ± 0.26 |
| TPGS | 1.24 ± 0.33 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zuccari, G.; Alfei, S.; Zorzoli, A.; Marimpietri, D.; Turrini, F.; Baldassari, S.; Marchitto, L.; Caviglioli, G. Increased Water-Solubility and Maintained Antioxidant Power of Resveratrol by Its Encapsulation in Vitamin E TPGS Micelles: A Potential Nutritional Supplement for Chronic Liver Disease. Pharmaceutics 2021, 13, 1128. https://doi.org/10.3390/pharmaceutics13081128
Zuccari G, Alfei S, Zorzoli A, Marimpietri D, Turrini F, Baldassari S, Marchitto L, Caviglioli G. Increased Water-Solubility and Maintained Antioxidant Power of Resveratrol by Its Encapsulation in Vitamin E TPGS Micelles: A Potential Nutritional Supplement for Chronic Liver Disease. Pharmaceutics. 2021; 13(8):1128. https://doi.org/10.3390/pharmaceutics13081128
Chicago/Turabian StyleZuccari, Guendalina, Silvana Alfei, Alessia Zorzoli, Danilo Marimpietri, Federica Turrini, Sara Baldassari, Leonardo Marchitto, and Gabriele Caviglioli. 2021. "Increased Water-Solubility and Maintained Antioxidant Power of Resveratrol by Its Encapsulation in Vitamin E TPGS Micelles: A Potential Nutritional Supplement for Chronic Liver Disease" Pharmaceutics 13, no. 8: 1128. https://doi.org/10.3390/pharmaceutics13081128
APA StyleZuccari, G., Alfei, S., Zorzoli, A., Marimpietri, D., Turrini, F., Baldassari, S., Marchitto, L., & Caviglioli, G. (2021). Increased Water-Solubility and Maintained Antioxidant Power of Resveratrol by Its Encapsulation in Vitamin E TPGS Micelles: A Potential Nutritional Supplement for Chronic Liver Disease. Pharmaceutics, 13(8), 1128. https://doi.org/10.3390/pharmaceutics13081128