Factors Affecting Posterior Capsule Opacification in the Development of Intraocular Lens Materials
Abstract
1. Introduction
1.1. Pathophysiology of Posterior Capsule Opacification
1.2. Risk Factors for Developing Posterior Capsule Opacification
1.2.1. Patient-Associated Risk Factors
1.2.2. Surgical-Associated Risk Factors
1.2.3. Intraocular Lens-Associated Risk Factors
Lens Material
Lens Design
Lens Function
1.3. Therapeutic Interventions for Posterior Capsule Opacification
1.4. Capsular Devices to Prevent Posterior Capsule Opacification Development
2. Experimental Models to Investigate Posterior Capsule Opacification
2.1. In Vitro Models
2.1.1. Two-Dimensional In Vitro Models
2.1.2. Three-Dimensional In Vitro Models
2.2. In Vivo Models
2.2.1. Murine
2.2.2. Rabbit
2.2.3. Porcine
2.3. Ex Vivo Models
2.4. Clinical Studies
2.4.1. Surgical Technique and Nd:YAG Laser Ablation Rates
2.4.2. Evaluation of Intraocular Lenses
2.4.3. Investigating Potential Therapeutic Interventions
3. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wormstone, I.M.; Wang, L.; Liu, C.S.C. Posterior capsule opacification. Exp. Eye Res. 2009, 88, 257–269. [Google Scholar] [CrossRef] [PubMed]
- Nibourg, M.L.; Gelens, E.; Kuijwe, R.; Hooymans, J.M.M.; Kooten Gvan, T.; Koopmans, A.S. Prevention of posterior capsular opacification. Exp. Eye Res. 2015, 136, 100–115. [Google Scholar] [CrossRef] [PubMed]
- Goshe, J.; Awh, C.; Houser, K. Posterior Capsule Opacification. Am. J. Ophthalmol. 2020, 1. Available online: https://eyewiki.aao.org/w/index.php?title=Posterior_capsule_opacification&oldid=58906 (accessed on 13 September 2020).
- Sinha, R.; Shekhar, H.; Sharma, N.; Titiyal, J.S.; Vajpayee, R.B. Posterior capsular opacification: A review. Indian J. Ophthalmol. 2013, 61, 371–376. [Google Scholar] [CrossRef]
- Ma, B.; Yang, L.; Jing, R.; Liu, J.; Quan, Y.; Hui, Q.; Li, J.; Qin, L.; Pei, C. Effects of Interleukin-6 on posterior capsular opacification. Exp. Eye Res. 2018, 172, 94–103. [Google Scholar] [CrossRef]
- Meacock, W.R.; Spalton, D.J.; Stanford, M.R. Role of cytokines in the pathogenesis of posterior capsule opacification. Br. J. Ophthalmol. 2000, 84, 332–336. [Google Scholar] [CrossRef]
- Awasthi, N.; Guo, S.; Wagner, B.J. Posterior capsular opacification: A problem reduced but not yet eradicated. JAMA Ophthalmol. 2009, 127, 555–562. [Google Scholar] [CrossRef]
- Trent, N. Global intraocular Lens market 2019: Size, share, demand, trends, growth and 2022 forecasts explored in latest research. Wise Guy Rep. 2018, 1–2. [Google Scholar]
- Vasavada, A. Cataract and surgery for cataract. Br. Med. J. 2006, 333, 128–132. [Google Scholar]
- Laser Eye Surgery Hub. Cataract Statistics & Resources. 2018, p. 1. Available online: https://www.lasereyesurgeryhub.co.uk/cataract-statistics/ (accessed on 9 November 2020).
- Ursell, P.G.; Dhariwal, M.; Majirska, K.; Ender, F.; Klason-Ray, S.; Venerus, A.; Miglio, C.; Bouchet, C. Three-year incidence of Nd:YAG capsulotomy and posterior capsule opacification and its relationship to monofocal acrylic IOL biomaterial: A UK real world evidence study. Eye 2018, 32, 1579–1589. [Google Scholar] [CrossRef]
- Sadoughi, M.; Einollahi, B.; Roshandel, D.; Sarimohammadli, M.; Feizi, S. Visual and refractive outcomes of phacoemulsification with implantation of accommodating versus standard monofocal intraocular lenses. J. Ophthalmic Vis. Res. 2015, 10, 370–374. [Google Scholar] [CrossRef]
- Jaffe, N.S. History of cataract surgery. Ophthalmology. 1996, 103, 5–16. [Google Scholar] [CrossRef]
- Raj, M.S.; Vasavada, A.R.; Johar, S.R.V.; Vasavadam, A.V.; Vasavada, A.V. Post-operative capsular opacification: A review. Int. J. Biomed. Sci. 2007, 3, 237–250. [Google Scholar]
- Davidson, M.G.; Morgan, D.K.; McGahan, M.C. Effect of surgical technique on in vitro posterior capsule opacification. J. Cataract Refract. Surg. 2000, 26, 1550–1554. [Google Scholar] [CrossRef]
- Jiang, J.; Shihan, M.H.; Wang, Y.; Duncan, M.K. Lens epithelial cells initiate an inflammatory response following cataract surgery. Investig. Ophthalmol. Vis. Sci. 2018, 59, 4986–4997. [Google Scholar] [CrossRef]
- Wormstone, I.M.; Eldred, J.A. Experimental models for posterior capsule opacification research. Exp. Eye Res. 2016, 142, 2–12. [Google Scholar] [CrossRef]
- Mohammadpour, M.; Jafarinasab, M.R.; Javadi, M.A. Outcomes of acute postoperative inflammation after cataract surgery. Eur. J. Ophthalmol. 2007, 17, 20–28. [Google Scholar] [CrossRef]
- Perez-Vives, C. Biomaterial influence on intraocular lens performance: An overview. J. Ophthalmol. 2018, 15, 2687385. [Google Scholar] [CrossRef]
- Rouillard, A.; Gundersen, G.; Fernandez, N.; Wang, Z.; Monteiro, C.; McDermott, M.; Ma’ayan, A. The harmonizome: A collection of processed datasets gathered to serve and mine knowledge about genes and proteins. Database 2016, 2016, baw100. [Google Scholar] [CrossRef]
- Lewis, A.C. Interleukin-6 in the pathogenesis of posterior capsule opacification and the potential role for interleukin-6 inhibition in the future of cataract surgery. Med. Hypotheses 2013, 80, 466–474. [Google Scholar] [CrossRef]
- Dinarello, C.A. Overview of the IL-1 family in innate inflammation and acquired immunity. Immunol. Rev. 2018, 281, 8–27. [Google Scholar] [CrossRef] [PubMed]
- Nishi, O.; Nishi, K.; Ohmoto, Y. Synthesis of interleukin-1, interleukin-6, and basic fibroblast growth factor by human cataract lens epithelial cells. J. Cataract Refract. Surg. 1996, 22, 852–858. [Google Scholar] [CrossRef]
- Basu, A.; Krady, J.K.; Levison, S.W. Interleukin-1: A master regulator of neuroinflammation. J. Neurosci. Res. 2004, 78, 151–156. [Google Scholar] [CrossRef] [PubMed]
- Ferrick, M.R.; Thurau, S.R.; Oppenheim, M.H.; Herbort, C.P.; Ni, M.; Zachariae, C.O.C.; Matsushima, K.; Chan, C.C. Ocular inflammation stimulated by intravitreal interleukin-8 and interleukin-1. Investig. Ophthalmol. Vis. Sci. 1991, 32, 1534–1539. [Google Scholar]
- Xiong, W.; Frasch, S.C.; Thomas, S.M.; Bratton, D.L.; Henson, P.M. Induction of TGF-β1 synthesis by macrophages in response to apoptotic cells requires activation of the scavenger receptor CD36. PLoS ONE 2013, 8, e72772. [Google Scholar] [CrossRef]
- Wallentin, N.; Wickström, K.; Lundberg, C. Effect of cataract surgery on aqueous TGF-β and lens epithelial cell proliferation. Investig. Ophthalmol. Vis. Sci. 1998, 39, 1410–1418. [Google Scholar]
- Walton, K.L.; Johnson, K.E.; Harrison, C.A. Targeting TGF-β mediated SMAD signaling for the prevention of fibrosis. Front. Pharmacol. 2017, 8, 461. [Google Scholar] [CrossRef]
- Taiyab, A.; Holms, J.; West-Mays, J.A. β-Catenin/Smad3 interaction regulates transforming growth factor-β-induced epithelial to mesenchymal transition in the lens. Int. J. Mol. Sci. 2019, 20, 2078. [Google Scholar] [CrossRef]
- Nishi, O.; Nishi, K.; Ohmoto, Y. Effect of interleukin 1 receptor antagonist on the blood-aqueous barrier after intraocular lens implantation. Br. J. Ophthalmol. 1994, 78, 917–920. [Google Scholar] [CrossRef][Green Version]
- Wilson, S.E.; Esposito, A. Focus on molecules: Interleukin-1: A master regulator of the corneal response to injury. Exp. Eye Res. 2009, 89, 124–125. [Google Scholar] [CrossRef]
- Ooi, K.G.J.; Galatowicz, G.; Calder, V.L.; Lightman, S.L. Cytokines and chemokines in uveitis—Is there a correlation with clinical phenotype? Clin. Med. Res. 2006, 4, 294–309. [Google Scholar] [CrossRef]
- Kubo, E.; Shibata, S.; Shibata, T.; Kiyokawa, E.; Sasaki, H.; Singh, D.P. FGF2 antagonizes aberrant TGFβ regulation of tropomyosin: Role for posterior capsule opacity. J. Cell Mol. Med. 2017, 21, 916–928. [Google Scholar] [CrossRef]
- Kubo, E.; Shibata, T.; Singh, P.D.; Sasaki, H. Roles of TGFB and FGF signals in the lens: Tropomyosin regulation for posterior capsule opacification. Int. J. Mol. Sci. 2018, 19, 3093. [Google Scholar] [CrossRef]
- Tandon, A.; Tovey, J.C.K.; Sharma, A.; Gupta, R.; Mohan, R.R. Role of transforming growth factor beta in corneal function, biology and pathology. Curr. Mol. Med. 2012, 10, 565–578. [Google Scholar]
- Nishi, O.; Nishi, K.; Wada, K.; Ohmoto, Y. Expression of transforming growth factor (TGF)-α, TGF-β2 and interleukin 8 messenger RNA in postsurgical and cultured lens epithelial cells obtained from patients with senile cataracts. Graefe’s Arch. Clin. Exp. Ophthalmol. 1999, 237, 806–811. [Google Scholar] [CrossRef]
- Saika, S. TGFbeta pathobiology in the eye. Lab. Investig. 2006, 86, 106–115. [Google Scholar] [CrossRef]
- Yamashita, H. Functions of the transforming growth factor-β superfamily in eyes. J. Jpn. Ophthalmol. Soc. 1997, 101, 927–947. [Google Scholar]
- Pei, C.; Ma, B.; Kang, Q.-Y.; Qin, L.; Cui, L.-J. Effects of transforming growth factor beta-2 and connective tissue growth factor on induction of epithelial mesenchymal transition and extracellular matrix synthesis in human lens epithelial cells. Int. J. Ophthalmol. 2013, 6, 752–757. [Google Scholar]
- Cho, H.J.; Baek, K.E.; Saika, S.; Jeong, M.J.; Yoo, J. Snail is required for transforming growth factor-beta-induced epithelial-mesenchymal transition by activating PI3 kinase/Akt signal pathway. Biochem. Biophys. Res. Commun. 2007, 353, 337–343. [Google Scholar] [CrossRef]
- Lois, N.; Taylor, J.; McKinnon, A.D.; Smith, G.C.; Van’t Hof, R.; Forrester, J.V. Effect of TGF-β2 and anti-TGF-β2 antibody in a new in vivo rodent model of posterior capsule opacification. Investig. Ophthalmol. Vis. Sci. 2005, 46, 4260–4266. [Google Scholar] [CrossRef][Green Version]
- Gotoh, N.; Perdue, N.R.; Matsushima, H.; Sage, E.H.; Yan, Q.; Clark, J.I. An in vitro model of posterior capsular opacity: SPARC and TGF-β2 minimize epithelial-to-mesenchymal transition in lens epithelium. Investig. Ophthalmol. Vis. Sci. 2007, 48, 4679–4687. [Google Scholar] [CrossRef]
- Amoozgar, B.; Fitzpatrick, S.D.; Sheardown, H. Effect of anti-TGF-β2 surface modification of polydimethylsiloxane on lens epithelial cell markers of posterior capsule opacification. J. Bioact. Compat. Polym. 2013, 28, 637–651. [Google Scholar] [CrossRef]
- Kurosaka, D.; Kato, K.; Nagamoto, T. Presence of alpha smooth muscle actin in lens epithelial cells of aphakic rabbit eyes. Br. J. Ophthalmol. 1996, 80, 906–910. [Google Scholar] [CrossRef]
- Wormstone, I.M.; Tamiya, S.; Anderson, I.; Duncan, G. TGF-β2-induced matrix modification and cell transdifferentiation in the human lens capsular bag. Investig. Ophthalmol. Vis. Sci. 2002, 43, 2301–2308. [Google Scholar]
- Weizman Institute of Science. S100A9 gene S100 calcium binding protein A9. Hum. Gene Database 2020. Available online: https://www.ncbi.nlm.nih.gov/gene?Db=gene&Cmd=DetailsSearch&Term=6280 (accessed on 9 November 2020).
- Wu, S.; Tong, N.; Pan, L.; Jiang, X.; Li, Y.; Guo, M.; Li, H. Retrospective analyses of potential risk factors for posterior capsule opacification after cataract surgery. J. Ophthalmol. 2018, 2018, 9089285. [Google Scholar] [CrossRef]
- Spalton, D.J. Posterior capsular opacification after cataract surgery. Eye 1999, 13, 489–492. [Google Scholar] [CrossRef]
- Batur, M.; Gül, A.; Seven, E.; Can, E.; Yaşar, T. Posterior capsular opacification in preschool- and school-age patients after pediatric cataract surgery without posterior capsulotomy. Turk. Oftalmoloiji Derg. 2016, 46, 205–208. [Google Scholar] [CrossRef]
- Wormstone, I.M.; Liu, C.S.; Rakic, J.M.; Marcantonio, J.M.; Vrenson, G.F.; Duncan, G. Human lens epithelial cell proliferation in a protein-free medium. Investig. Ophthalmol. Vis. Sci. 1997, 38, 396–404. [Google Scholar]
- Praveen, M.R.; Vasavada, A.R.; Shah, G.D.; Shah, A.R.; Khamar, B.M.; Dave, K.H. A prospective evaluation of posterior capsule opacification in eyes with diabetes mellitus: A case-control study. Eye 2014, 28, 720–727. [Google Scholar] [CrossRef]
- Boureau, C.; Lafuma, A.; Jeanbat, V.; Berdeaux, G.; Smith, A.F. Incidence of Nd:YAG laser capsulotomies after cataract surgery: Comparison of 3 square-edged lenses of different composition. Can. J. Ophthalmol. 2009, 44, 165–170. [Google Scholar] [CrossRef]
- Chen, H.-C.; Lee, C.-Y.; Sun, C.-C.; Huang, J.-Y.; Lin, H.-Y.; Yang, S.-F. Risk factors for the occurrence of visual threatening posterior capsule opacification. J. Transl. Med. 2019, 17, 1. [Google Scholar] [CrossRef] [PubMed]
- Ram, J.; Brar, S.G. Posterior capsule opacification: An overview. DOS Times 2006, 12, 285–288. [Google Scholar]
- Keintzel, M.; Kohnen, T. Anterior Optic Capture. Encycl. Ophthalmol. 2018, 2018. [Google Scholar] [CrossRef]
- Menapace, R. Mastering the posterior capsule and optic capture: Elimination of secondary cataract. Am. J. Ophthalmol. 2020, 2020, 10. [Google Scholar]
- Vasavada, A.; Vasavada, V. Pediatric posterior optic capture. CRST Eur. 2019, 2019, 7–46. [Google Scholar]
- Paik, J.; Shiloach, M.; Macsai-Kaplan, M. The effect of posterior capsule polishing on posterior capsule opacification. Investig. Ophthalmol. Vis Sci. 2012, 53, 6668. [Google Scholar]
- Biswas, P.; Batra, S. Commentary: Anterior capsule polishing: The present perspective. Indian J. Ophthalmol. 2020, 68, 785–786. [Google Scholar] [CrossRef]
- Wang, D.; Yu, X.; Li, Z.; Ding, X.; Lian, H.; Mao, J.; Zhao, Y.; Zhao, Y.-E. The effect of anterior capsule polishing on capsular contraction and lens stability in cataract patients with high myopia. J. Ophthalmol. 2018, 2018, 8676451. [Google Scholar] [CrossRef]
- Joshi, R.S. Incidence of primary peripheral posterior capsular opacification after cataract surgery and posterior capsular opacification in the patients implanted with foldable intraocular lenses. Nepal. J. Ophthalmol. 2017, 9, 149–155. [Google Scholar] [CrossRef]
- Nanavaty, M.A.; Mehta, P.A.; Raj, S.M.; Vasavada, A.R. Diagnosis of pre-existing posterior capsule defect in traumatic white mature cataract with intact anterior capsule. Eye 2006, 20, 949–951. [Google Scholar] [CrossRef]
- Bellucci, R. An introduction to intraocular lenses: Material, optics, haptics, design and aberration. Cataract 2013, 3, 38–55. [Google Scholar]
- Ridley, H. Intra-ocular acrylic lenses—Past, present and future. Trans. Ophthalmol. Soc. UK 1964, 84, 5–14. [Google Scholar]
- Yu, N.; Fang, F.; Wu, B.; Zeng, L.; Cheng, Y. State of the art of intraocular lens manufacturing. Int. J. Adv. Manuf. Technol. 2018, 98, 1103–1130. [Google Scholar] [CrossRef]
- Özyol, P.; Özyol, E.; Karel, F. Biocompatibility of intraocular lenses. Turk Oftalmoloiji Derg. 2017, 47, 221–225. [Google Scholar] [CrossRef]
- Saika, S. Relationship between posterior capsule opacification and intraocular lens biocompatibility. Prog. Retin. Eye Res. 2004, 23, 283–305. [Google Scholar] [CrossRef]
- Zhao, Y.; Yang, K.; Li, J.; Huang, Y.; Zhu, S. Comparison of hydrophobic and hydrophilic intraocular lens in preventing posterior capsule opacification after cataract surgery An updated meta-analysis. Medicine 2017, 96, e8301. [Google Scholar] [CrossRef]
- Auffarth, G.U.; Brezin, A.; Caporossi, A.; Lafuma, A.; Mendicute, J.; Bordeaux, G.; Smith, A.F. Comparison of Nd:YAG capsulotomy rates following phacoemulsification with implantation of PMMA, silicone, or acrylic intra-ocular lenses in four European countries. Ophthalmic Epidemiol. 2004, 11, 319–329. [Google Scholar] [CrossRef]
- Mathew, R.G.; Coombes, A.G.A. Reduction of Nd:YAG capsulotomy rates after implantation of a single-piece acrylic hydrophilic intraocular lens with 360° squared optic edge: 24-Month results. Ophthalmic Surg. Lasers Imaging 2010, 41, 651–655. [Google Scholar] [CrossRef]
- Bai, L.; Zhang, J.; Chen, L.; Ma, T.; Liang, H.C. Comparison of posterior capsule opacification at 360-degree square edge hydrophilic and sharp edge hydrophobic acrylic intraocular lens in diabetic patients. Int. J. Ophthalmol. 2015, 8, 725–729. [Google Scholar]
- Werner, L.; Mamalis, N.; Pandey, S.K.; Izak, A.M.; Nilson, C.D.; Davis, B.L.; Weight, C.; Apple, D.J. Posterior capsule opacification in rabbit eyes implanted with hydrophilic acrylic intraocular lenses with enhanced square edge. J. Cataract Refract. Surg. 2004, 30, 2403–2409. [Google Scholar] [CrossRef]
- Haripriya, A.; Chang, D.F.; Vijayakumar, B.; Niraj, A.; Shekhar, M.; Tanpreet, S.; Aravind, S. Long-term posterior capsule opacification reduction with square-edge polymethylmethacrylate intraocular lens: Randomized controlled study. Ophthalmology 2017, 124, 295–302. [Google Scholar] [CrossRef][Green Version]
- Kohnen, T.; Fabian, E.; Gerl, R.; Hunold, W.; Hütz, W.; Strobel, J.; Hoyer, H.; Mester, U. Optic edge design as long-term factor for posterior capsular opacification rates. Ophthalmology 2008, 115, 1308–1314. [Google Scholar] [CrossRef]
- Nishi, O.; Nishi, K.; Akura, J.; Nagata, T. Effect of round-edged acrylic intraocular lenses on preventing posterior capsule opacification. J. Cataract Refract. Surg. 2001, 27, 608–613. [Google Scholar] [CrossRef]
- Mencucci, R.; Favuzza, E.; Boccalini, C.; Gicquel, J.J.; Raimondi, L. Square-edge intraocular lenses and epithelial lens cell proliferation: Implications on posterior capsule opacification in an in vitro model. BMC Ophthalmol. 2015, 15, 1–5. [Google Scholar] [CrossRef]
- Linnola, R.J.; Sund, M.; Ylönen, R.; Pihlajaniemi, T. Adhesion of soluble fibronectin, laminin, and collagen type IV to intraocular lens materials. J. Cataract Refract. Surg. 1999, 25, 1486–1491. [Google Scholar] [CrossRef]
- Linnola, R.J. Sandwich theory: Bioactivity-based explanation for posterior capsule opacification. J. Cataract Refract. Surg. 1997, 23, 1539–1542. [Google Scholar] [CrossRef]
- Vock, L.; Menapace, R.; Stifter, E.; Georgopoulos, M.; Sacu, S.; Buhl, W. Posterior capsule opacification and neodymium:YAG laser caspulotomy rates with a round-edged silicone and a sharp-edged hydrophobic acrylic intraocular lens 10 years after surgery. J. Cataract Refract. Surg. 2009, 35, 459–465. [Google Scholar] [CrossRef]
- Dawes, L.J.; Illingworth, C.D.; Wormstone, I.M. A fully human in vitro capsular bag model to permit intraocular lens evaluation. Invest. Ophthalmol. Vis. Sci. 2012, 53, 23–29. [Google Scholar] [CrossRef]
- Mylonas, G.; Prskavec, M.; Baradaran-Dilmaghani, R.; Karnik, N.; Buehl, W.; Wirtitsch, M. Effect of a single-piece and a three-piece acrylic sharp-edged IOL on posterior capsule opacification. Curr. Eye Res. 2013, 38, 86–90. [Google Scholar] [CrossRef]
- Bilge, A.H.; Aykan, U.; Akin, T.; Unsal, U. The effects of three-piece or single-piece acrylic intraocular lens implantation on posterior capsule opacification. Eur. J. Ophthalmol. 2004, 14, 375–380. [Google Scholar] [CrossRef]
- Wallin, T.R.; Hinckley, M.; Nilson, C.; Olson, R.J. A clinical comparison of single-piece and three-piece truncated hydrophobic acrylic intraocular lenses. Am. J. Ophthalmol. 2003, 136, 614–619. [Google Scholar] [CrossRef]
- Hayashi, K.; Hayashi, H.; Nakao, F.; Hayashi, F. Comparison of decentration and tilt between one piece and three piece polymethyl methacrylate intraocular lenses. Br. J. Ophthalmol. 1998, 82, 419–422. [Google Scholar] [CrossRef] [PubMed]
- Zvornicanin, J.; Zvornicanin, E. Premium intraocular lenses: The past, present and future. J. Curr. Ophthalmol. 2018, 30, 287–296. [Google Scholar] [CrossRef] [PubMed]
- Buckhurst, P.J.; Naroo, S.A.; Shah, S. Advanced intraocular lens designs. Eur. Ophthalmic Rev. 2010, 4, 82–86. [Google Scholar] [CrossRef]
- Alió, J.L.; Barrio, J.L.A.; Vega-Estrada, A. Accommodative intraocular lenses: Where are we and where we are going. Eye Vis. 2017, 4, 1–12. [Google Scholar] [CrossRef]
- Takakura, A.; Iyer, P.; Adams, J.R.; Pepin, S.M. Functional assessment of accommodating intraocular lenses versus monofocal intraocular lenses in cataract surgery: Metaanalysis. J. Cataract Refract. Surg. 2010, 36, 380–388. [Google Scholar] [CrossRef]
- Elgohary, M.A.; Dowler, J.G. Incidence and risk factors of Nd:YAG capsulotomy after phacoemulsification in non-diabetic and diabetic patients. Clin. Exp. Ophthalmol. 2006, 34, 526–534. [Google Scholar] [CrossRef]
- Burq, M.A.; Taqui, A.M. Frequency of retinal detachment and other complications after neodymium:YAG capsulotomy. J. Cataract Refract. Surg. 2008, 58, 550–552. [Google Scholar]
- Nixon, D.R.; Woodcock, M.G. Pattern of posterior capsule opacification models 2 years postoperatively with 2 single-piece acrylic intraocular lenses. J. Cataract Refract. Surg. 2010, 36, 929–934. [Google Scholar] [CrossRef]
- Shao, J.Z.; Qi, Y.; Du, S.S.; Du, W.W.; Li, F.Z.; Zhang, F.Y. In vitro inhibition of proliferation, migration and epithelial-mesenchymal transition of human lens epithelial cells by fasudil. Int. J. Ophthalmol. 2018, 11, 1253–1257. [Google Scholar]
- Kramer, G.D.; Werner, L.; Mamalis, N. Prevention of postoperative capsular bag opacification using intraocular lenses and endocapsular devices maintaining an open or expanded capsular bag. J. Cataract Refract. Surg. 2016, 42, 469–484. [Google Scholar] [CrossRef]
- Hasanee, K.; Butler, M.; Ahmed, I.I.K. Capsular tension rings and related devices: Current concepts. Curr. Opin. Ophthalmol. 2006, 17, 31–41. [Google Scholar]
- Hara, T.; Hara, T.; Yamada, Y. “Equator ring” for maintenance of the completely circular contour of the capsular bag equator after cataract removal. Ophthalmic Surg. 1991, 22, 358–359. [Google Scholar]
- Hara, T.; Hara, T.; Narita, M. Long-term study of posterior capsular opacification prevention with endocapsular equator rings in humans. Arch Ophthalmol. 2011, 129, 855–863. [Google Scholar] [CrossRef]
- Menapace, R.; Sacu, S.; Georgopoulos, M.; Findl, O.; Rainer, G.; Nishi, O. Efficacy and safety of capsular bending ring implantation to prevent posterior capsule opacification: Three-year results of a randomized clinical trial. J. Cataract Refract. Surg. 2008, 34, 1318–1328. [Google Scholar] [CrossRef]
- Nishi, O.; Nishi, K.; Menapace, R.; Akura, J. Capsular bending ring to prevent posterior capsule opacification: 2 year follow-up. J. Cataract Refract. Surg. 2001, 27, 1359–1365. [Google Scholar] [CrossRef]
- Slutzky, L.; Kleinmann, G. Further enhancement of intraocular open-capsule devices for prevention of posterior capsule opacification. Transl. Vis. Sci. Technol. 2018, 7, 21. [Google Scholar] [CrossRef]
- Alon, R.; Assia, E.I.; Kleinmann, G. Prevention of posterior capsule opacification by an intracapsular open capsule device. Lens 2014, 55, 4005–4013. [Google Scholar] [CrossRef][Green Version]
- Chung, K.K.; Schumacher, J.F.; Sampson, E.M.; Burne, R.; Antonelli, P.J.; Brennan, A.B.; Reddy, S.T. Impact of engineered surface microtopography on biofilm formation of Staphylococcus aureus. Biointerphases 2007, 2, 89–94. [Google Scholar] [CrossRef]
- VanSlyke, J.K.; Boswell, B.A.; Musil, L.S. Fibronectin regulates growth factor signaling and cell differentiation in primary lens cells. J. Cell Sci. 2018, 131, 22. [Google Scholar] [CrossRef]
- Wertheimer, C.; Liegl, R.; Kernt, M.; Mayer, W.; Docheva, D.; Kampik, A.; Eibl-Lindner, K.H. EGF receptor inhibitor erlotinib as a potential pharmacological prophylaxis for posterior capsule opacification. Graefe’s Arch. Clin. Exp. Ophthalmol. 2013, 251, 1529–1540. [Google Scholar] [CrossRef]
- Zukin, L.M.; Pedler, M.G.; Groman-Lupa, S.; Pantcheva, M.; Ammar, D.A.; Mark Petrash, J. Aldose reductase inhibition prevents development of posterior capsular opacification in an in vivo model of cataract surgery. Investig. Ophthalmol. Vis. Sci. 2018, 59, 3591–3598. [Google Scholar] [CrossRef] [PubMed]
- Cooke, C.A.; McGimpsey, S.; Mahon, G.; Best, R.M. An in vitro study of human lens epithelial cell adhesion to intraocular lenses with and without a fibronectin coating. Invest. Ophthalmol. Vis. Sci. 2006, 47, 2985–2989. [Google Scholar] [CrossRef] [PubMed]
- Plüss, C.J.; Kustermann, S. A Human Three-Dimensional In Vitro Model of Lens Epithelial Cells as a Model to Study Mechanisms of Drug-Induced Posterior Subcapsular Cataracts. J. Ocul. Pharmacol. Ther. 2019, 36, 56–64. [Google Scholar] [CrossRef] [PubMed]
- Joseph, R.; Bales, K.; Srivastava, K.; Srivastava, O. Lens epithelial cells-induced pluripotent stem cells as a model to study epithelial-mesenchymal transition during posterior capsular opacification. Biochem. Biophys. Rep. 2019, 20, 100696. [Google Scholar] [CrossRef]
- Wertheimer, C.; Kueres, A.; Siedlecki, J.; Braun, C.; Kassumeh, S.; Wolf, A.; Mayer, W.; Priglinger, C.; Priglinger, S.; Eibl-Lindner, K. The intraocular lens as a drug delivery device for an epidermal growth factor–Receptor inhibitor for prophylaxis of posterior capsule opacification. Acta Ophthalmol. 2018, 96, e874–e882. [Google Scholar] [CrossRef]
- Han, Y.; Tang, J.; Xia, J.; Wang, R.; Qin, C.; Liu, S.; Zhao, X.; Chen, H.; Lin, Q. Anti-adhesive and antiproliferative synergistic surface modification of intraocular lens for reduced posterior capsular opacification. Int. J. Nanomed. 2019, 14, 9047–9061. [Google Scholar] [CrossRef]
- Shihan, M.H.; Kanwar, M.; Wang, Y.; Jackson, E.E.; Faranda, A.P.; Duncan, M.K. Fibronectin has multifunctional roles in posterior capsular opacification (PCO). Matrix Biol. 2020, 90, 79–108. [Google Scholar] [CrossRef]
- Gerhart, J.; Withers, C.; Gerhart, C.; Werner, L.; Mamalis, N.; Bravo-Nuevo, A.; Scheinfeld, V.; FitzGerald, P.; Getts, R.; George-Weinstein, M. Myo/Nog cells are present in the ciliary processes, on the zonule of Zinn and posterior capsule of the lens following cataract surgery. Exp. Eye Res. 2018, 171, 101–105. [Google Scholar] [CrossRef]
- Sternberg, K.; Terwee, T.; Stachs, O.; Guthoff, R.; Lobler, M.; Schmitz, K.P. Drug-induced secondary cataract prevention: Experimental ex vivo and in vivo results with disulfiram, methotrexate and actinomycin. D. Ophthalmic Res. 2010, 44, 225–236. [Google Scholar] [CrossRef]
- Ma, B.; Jing, R.; Liu, J.; Qi, T.; Pei, C. Gremlin is a potential target for posterior capsular opacification. Cell Cycle 2019, 18, 1714–1726. [Google Scholar] [CrossRef]
- Kubo, E.; Hasanova, N.; Fatma, N.; Sasaki, H.; Singh, D.P. Elevated tropomyosin expression is associated with epithelial-mesenchymal transition of lens epithelial cells. J. Cell. Mol. Med. 2013, 17, 212–221. [Google Scholar] [CrossRef]
- Chang, P.; Lin, L.; Zheng, Q.; Yu, F.; Yu, X.; Zhao, Y.; Ding, X.; Zhu, W.; Li, J.; Zhao, Y. An optical section-assisted in vivo rabbit model for capsular bend and posterior capsule opacification investigation. PLoS ONE 2016, 2, e0148553. [Google Scholar] [CrossRef][Green Version]
- Duncan, G.; Wang, L.; Neilson, G.J.; Wormstone, I.M. Lens cell survival after exposure to stress in the closed capsular bag. Investig. Ophthalmol. Vis. Sci. 2007, 48, 2701–2707. [Google Scholar] [CrossRef]
- Dawson, H.D.; Smith, A.D.; Chen, C.; Urban, J.F. An in-depth comparison of the porcine, murine and human inflammasomes; lessons from the porcine genome and transcriptome. Vet. Microbiol. 2017, 202, 2–15. [Google Scholar] [CrossRef]
- Meurens, F.; Summerfield, A.; Nauwynck, H.; Saif, L.; Gerdts, V. The pig: A model for human infectious diseases. Trends Microbiol. 2012, 20, 50–57. [Google Scholar] [CrossRef]
- Walters, E.M.; Prather, R.S. Advancing swine models for human health and diseases. Mo. Med. 2013, 110, 212–215. [Google Scholar]
- Wormstone, I.M. The human capsular bag model of posterior capsule opacification. Eye 2019, 34, 225–231. [Google Scholar] [CrossRef]
- Kassumeh, S.A.; Wertheimer, C.M.; von Studnitz, A.; Hillenmayer, A.; Priglinger, C.; Wolf, A.; Mayer, W.J.; Teupser, D.; Holdt, L.M.; Priglinger, S.G.; et al. Poly(lactic-co-glycolic) Acid as a slow-release drug-carrying matrix for methotrexate coated onto intraocular lenses to conquer posterior capsule opacification. Curr. Eye Res. 2018, 43, 702–708. [Google Scholar] [CrossRef]
- D’Antin, J.C.; Barraquer, R.I.; Tresserra, F.; Michael, R. Prevention of posterior capsule opacification through intracapsular hydrogen peroxide or distilled water treatment in human donor tissue. Sci. Rep. 2018, 8, 1–10. [Google Scholar] [CrossRef]
- Eldred, J.A.; Zheng, J.; Chen, S.; Wormstone, I.M. An in vitro human lens capsular bag model adopting a graded culture regime to assess putative impact of iols on pco formation. Investig. Ophthalmol. Vis. Sci. 2019, 60, 113–122. [Google Scholar] [CrossRef]
- Aronson, J.K. What is a clinical trial? Br. J. Clin. Pharmacol. 2004, 58, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Kelkar, J.; Mehta, H.; Kelkar, A.; Agarwal, A.; Kothari, A.; Kelkar, S. Precision pulse capsulotomy in phacoemulsification: Clinical experience in Indian eyes. Indian J. Ophthalmol. 2018, 66, 1272–1277. [Google Scholar] [CrossRef]
- Ton Van, C.; Tran, T.H.C. Incidence of posterior capsular opacification requiring Nd:YAG capsulotomy after cataract surgery and implantation of enVista ® MX60 IOL. J. Français Ophtalmol. 2018, 41, 899–903. [Google Scholar] [CrossRef]
- Hecht, I.; Karesvuo, P.; Achiron, A.; Elbaz, U.; Laine, I.; Tuuminen, R. Anti-inflammatory Medication After Cataract Surgery and Posterior Capsular Opacification. Am. J. Ophthalmol. 2020, 215, 104–111. [Google Scholar] [CrossRef]
- Rabsilber, T.M.; Limberger, I.J.; Reuland, A.J.; Holzer, M.P.; Auffarth, G.U. Long-term results of sealed capsule irrigation using distilled water to prevent posterior capsule opacification: A prospective clinical randomised trial. Br. J. Ophthalmol. 2007, 91, 912–915. [Google Scholar] [CrossRef][Green Version]
- Jun, J.H.; Sohn, W.-J.; Lee, Y.; Chang, S.D.; Kim, J.-Y. Experimental lens capsular bag model for posterior capsule opacification. Cell Tissue Res. 2014, 357, 101–108. [Google Scholar] [CrossRef]
- van Kooten, T.G.; Koopmans, S.; Terwee, T.; Norrby, S.; Hooymans, J.M.M.; Busscher, H.J. Development of an accommodating intra-ocular lens-In vitro prevention of re-growth of pig and rabbit lens capsule epithelial cells. Biomaterials 2006, 27, 5554–5560. [Google Scholar] [CrossRef]
- Awasthi, N.; Wang-Su, S.T.; Wagner, B.J. Downregulation of MMP-2 and -9 by proteasome inhibition: A possible mechanism to decrease LEC migration and prevent posterior capsular opacification. Invest. Ophthalmol. Vis. Sci. 2008, 49, 1998–2003. [Google Scholar] [CrossRef]
- Malecaze, F.; Decha, A.; Serre, B.; Penary, M.; Duboue, M.; Berg, D.; Levade, T.; Lubsen, N.H.; Kremer, E.J.; Couderc, B. Prevention of posterior capsule opacification by the induction of therapeutic apoptosis of residual lens cells. Gene Ther. 2006, 13, 440–448. [Google Scholar] [CrossRef] [PubMed]
- Shirai, K.; Tanaka, S.I.; Lovicu, F.J.; Saika, S. The murine lens: A model to investigate in vivo epithelial–mesenchymal transition. Dev. Dyn. 2018, 247, 340–345. [Google Scholar] [CrossRef] [PubMed]
- Yadav, U.C.S.; Ighani-Hosseinabad, F.; van Kuijk, F.J.G.M.; Srivastava, S.K.; Ramana, K. Prevention of posterior capsular opacification through aldose reductase inhibition. Investig. Ophthalmol. Vis. Sci. 2009, 50, 752–759. [Google Scholar] [CrossRef] [PubMed]
- Ongkasin, K.; Masmoudi, Y.; Wertheimer, C.M.; Hillenmayer, A.; Eibl-Lindner, K.H.; Badens, E. Supercritical fluid technology for the development of innovative ophthalmic medical devices: Drug loaded intraocular lenses to mitigate posterior capsule opacification. Eur. J. Pharm. Biopharm. 2020, 149, 248–256. [Google Scholar] [CrossRef] [PubMed]
- Chandler, H.L.; Haeussler, D.J.; Gemensky-Metzler, A.J.; Wilkie, D.A.; Lutz, E.A. Induction of posterior capsule opacification by hyaluronic acid in an ex vivo model. Investig. Ophthalmol. Vis. Sci. 2012, 53, 1835–1845. [Google Scholar] [CrossRef] [PubMed]
- Hillenmayer, A.; Wertheimer, C.; Kassumeh, S.; von Studnitz, A.; Luft, N.; Ohlmann, A.; Priglinger, S.; Mayer, W. Evaluation of posterior capsule opacification of the Alcon Clareon IOL vs the Alcon Acrysof IOL using a human capsular bag model. BMC Ophthalmol. 2020, 20, 1–7. [Google Scholar] [CrossRef]
- Pot, S.A.; Chandler, H.L.; Colitz, C.M.H.; Bentley, E.; Dubielzig, R.R.; Mosley, T.S.; Reid, T.W.; Murphy, C.J. Selenium functionalized intraocular lenses inhibit posterior capsule opacification in an ex vivo canine lens capsular bag assay. Exp. Eye Res. 2009, 89, 728–734. [Google Scholar] [CrossRef]
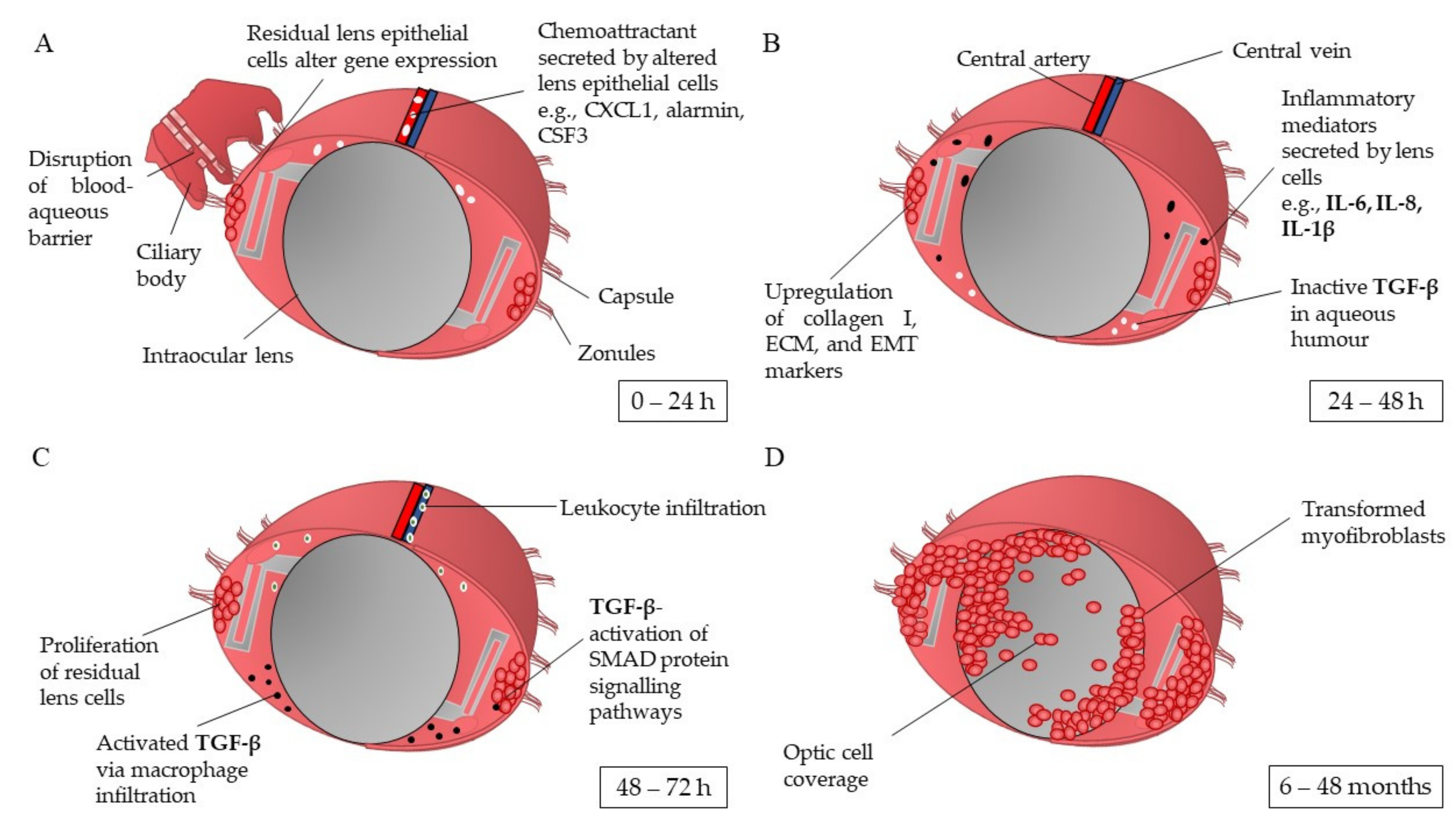
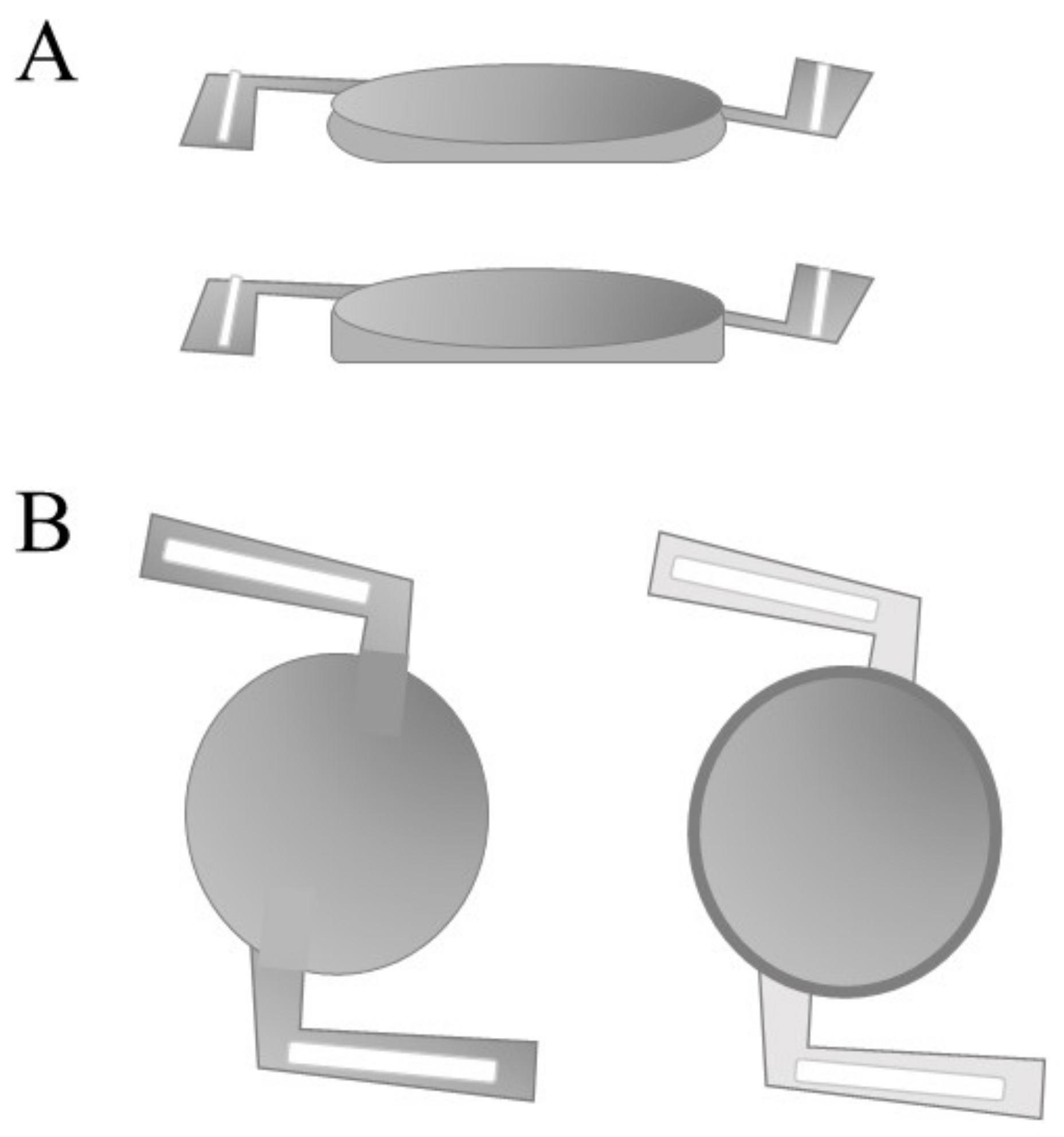
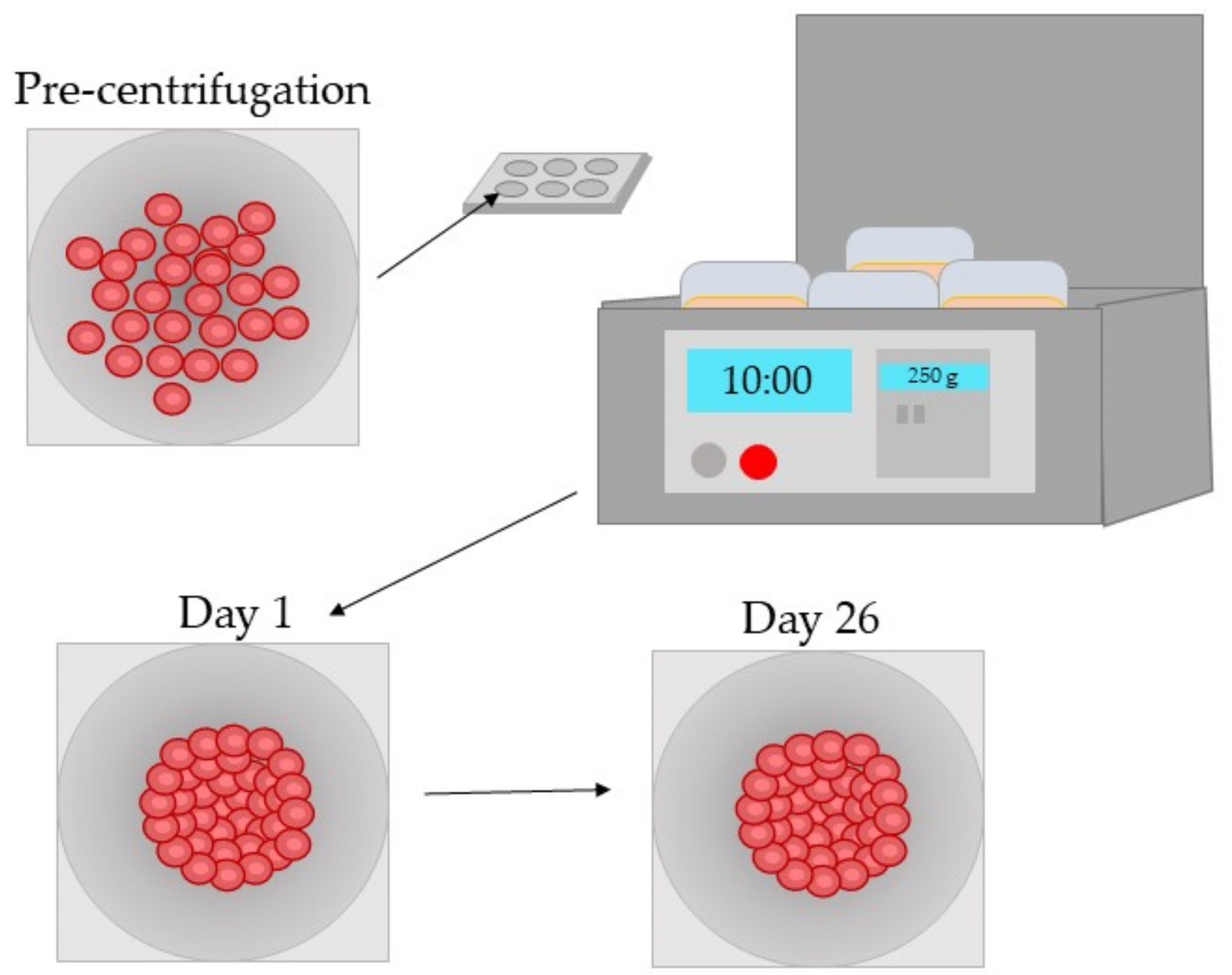
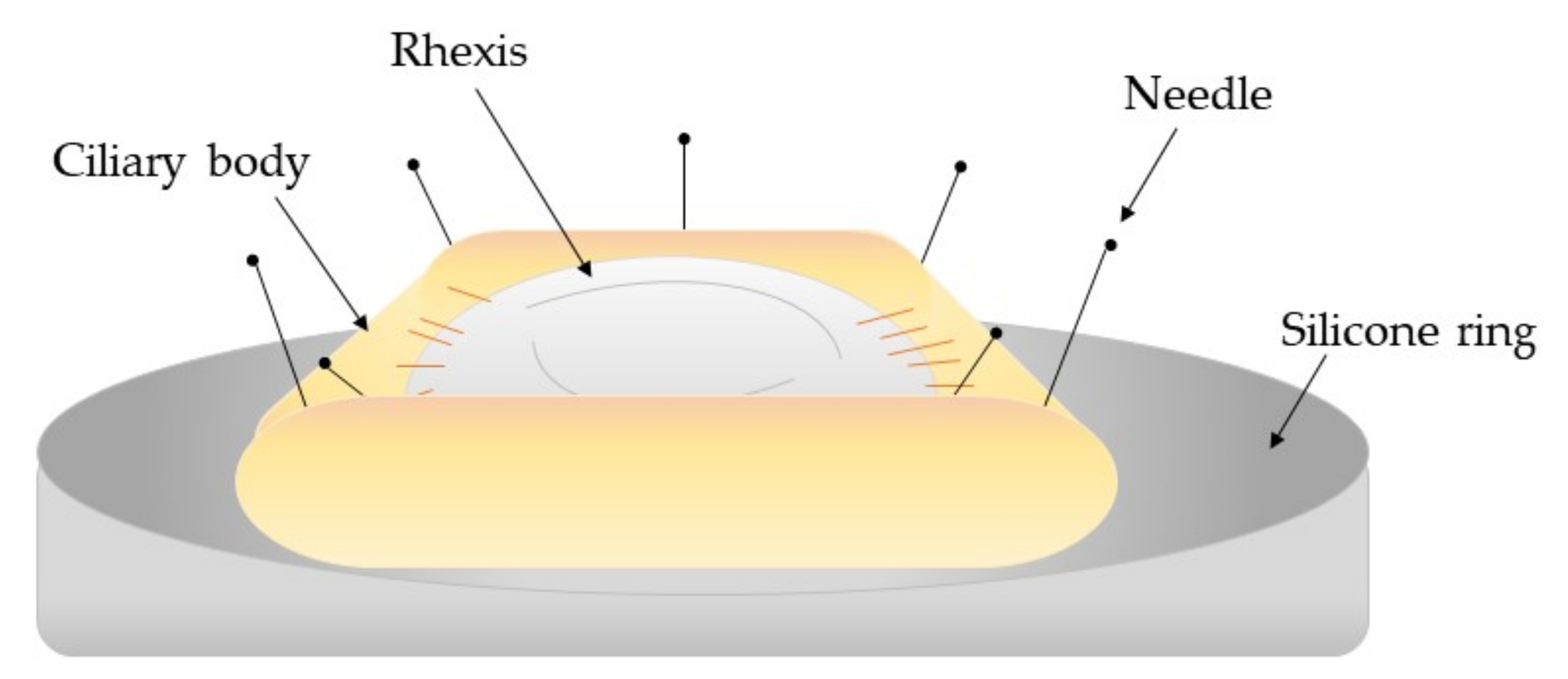
| Model Type | Tissue Donors | Application | References |
|---|---|---|---|
| In vitro | Chick, rat, human, mouse |
| [106,107,123,129] |
| [5,16,102] | ||
| [105,108,109,130] | ||
| [92,103,121,131,132] | ||
| [15] | ||
| In vivo | Porcine, murine, human, rabbit |
| [114,133] |
| [109] | ||
| [110,111] | ||
| [41,112,132,134] | ||
| [25] | ||
| [99,100] | ||
| Ex vivo | Human, rat, porcine, canine, chick |
| [112,121,132,134,135] |
| [29,136] | ||
| [108,137,138] | ||
| Clinical studies | n/a |
| [47,89] |
| [127] | ||
| [11,12,53,68,69,70,121] | ||
| [58,125] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cooksley, G.; Lacey, J.; Dymond, M.K.; Sandeman, S. Factors Affecting Posterior Capsule Opacification in the Development of Intraocular Lens Materials. Pharmaceutics 2021, 13, 860. https://doi.org/10.3390/pharmaceutics13060860
Cooksley G, Lacey J, Dymond MK, Sandeman S. Factors Affecting Posterior Capsule Opacification in the Development of Intraocular Lens Materials. Pharmaceutics. 2021; 13(6):860. https://doi.org/10.3390/pharmaceutics13060860
Chicago/Turabian StyleCooksley, Grace, Joseph Lacey, Marcus K. Dymond, and Susan Sandeman. 2021. "Factors Affecting Posterior Capsule Opacification in the Development of Intraocular Lens Materials" Pharmaceutics 13, no. 6: 860. https://doi.org/10.3390/pharmaceutics13060860
APA StyleCooksley, G., Lacey, J., Dymond, M. K., & Sandeman, S. (2021). Factors Affecting Posterior Capsule Opacification in the Development of Intraocular Lens Materials. Pharmaceutics, 13(6), 860. https://doi.org/10.3390/pharmaceutics13060860






