Composite Drug Delivery System Based on Amorphous Calcium Phosphate–Chitosan: An Efficient Antimicrobial Platform for Extended Release of Tetracycline
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
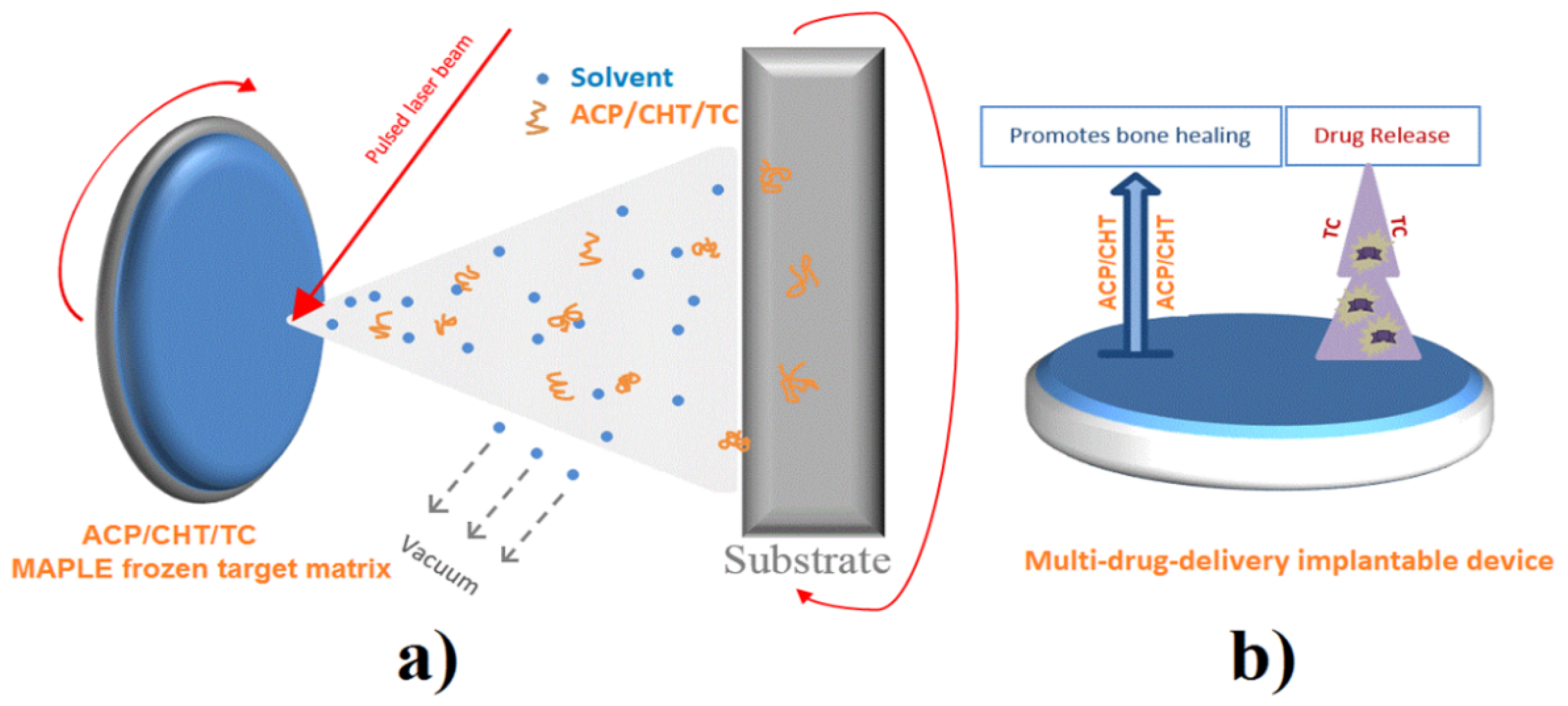
2.2. MAPLE Deposition
2.3. Physico-Chemical Characterization
2.3.1. Coating’s Morphology
2.3.2. IR Active Groups
2.3.3. X-ray Diffraction (XRD)
2.3.4. Static Contact Angle (CA)
2.3.5. Drug Release
2.3.6. Mass Loss Evaluation
2.3.7. pH Evolution of SBF
2.4. Microbiological Assay
2.4.1. Biocompatibility
2.4.2. Antimicrobial Biofilm Activity
3. Results
3.1. Physico-Chemical Investigations
3.1.1. SEM
- Particles of different size, up to several μm, are present on the surface of the deposited films. They are assembled in a rather smooth morphology, characteristic to MAPLE ACP coatings [36].
- Films are quite homogenous and exhibit few microcracks, which do not affect the coating’s toughness or, notably, the adherence to the substrate. In our opinion, this is a beneficial characteristic of this composite, allowing the formation of a CHT continuous matrix where ACP and TC are finely entrapped, favoring a high adherence to the substrate [36,50].
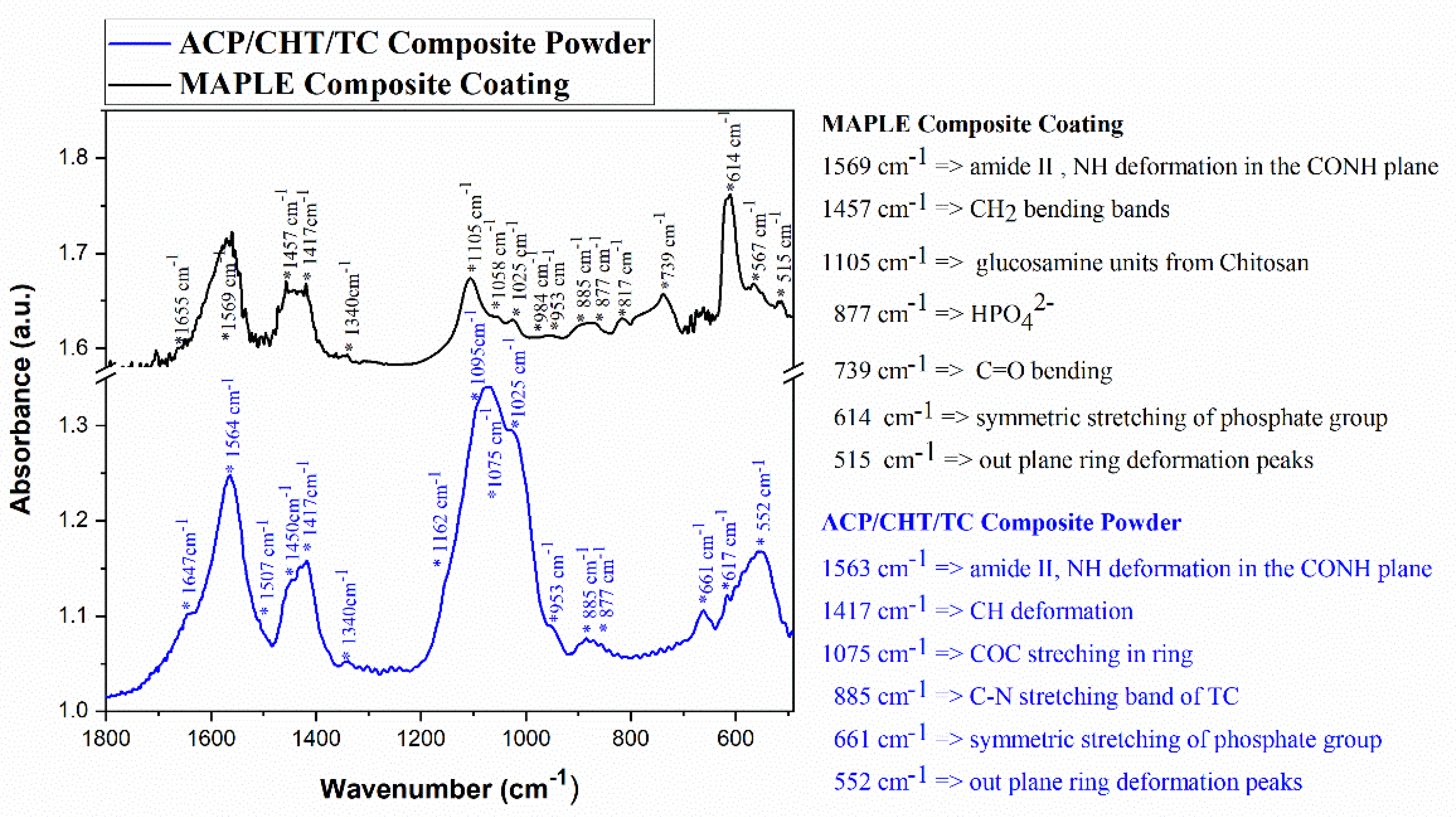
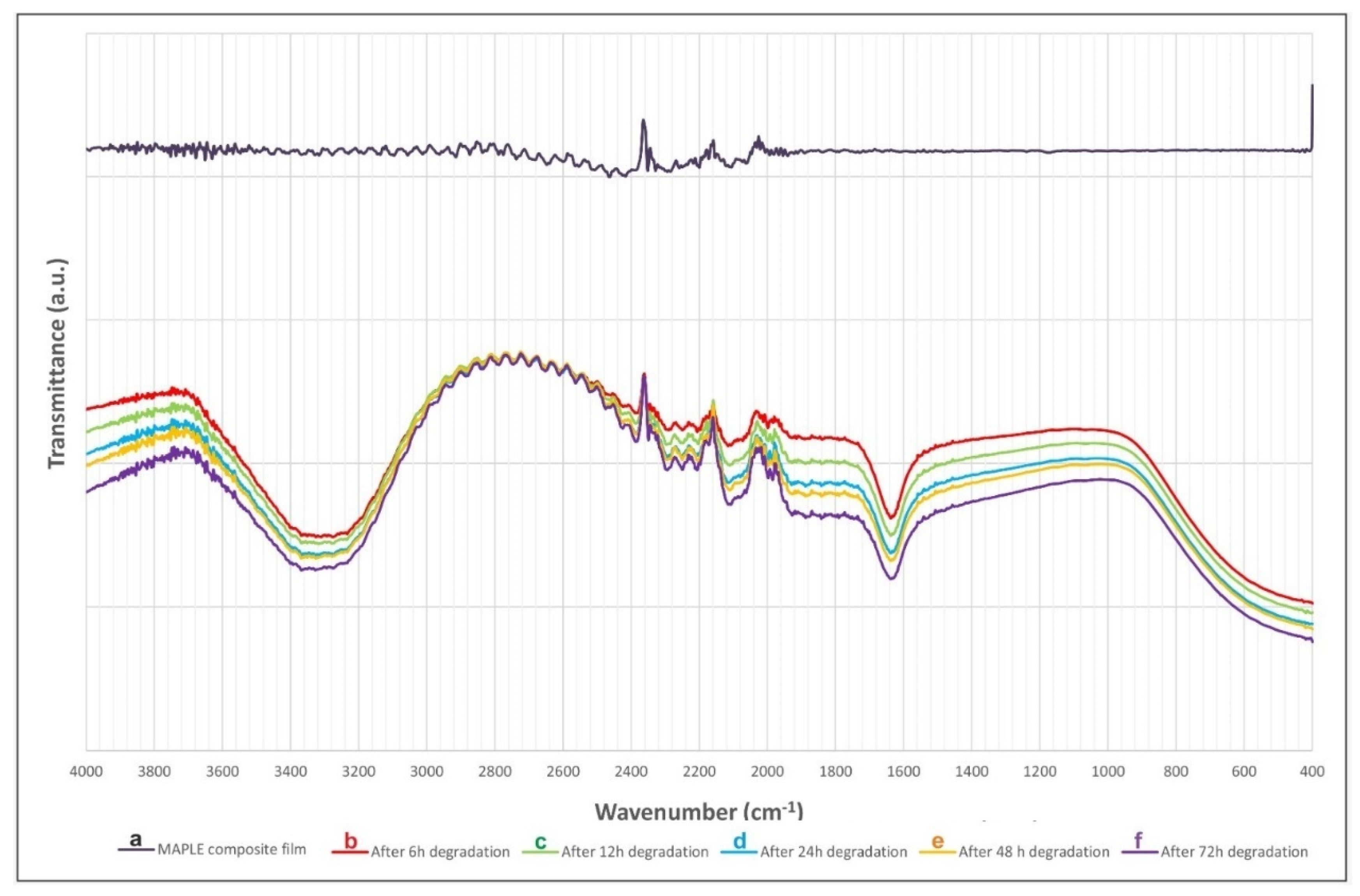
3.1.2. FTIR
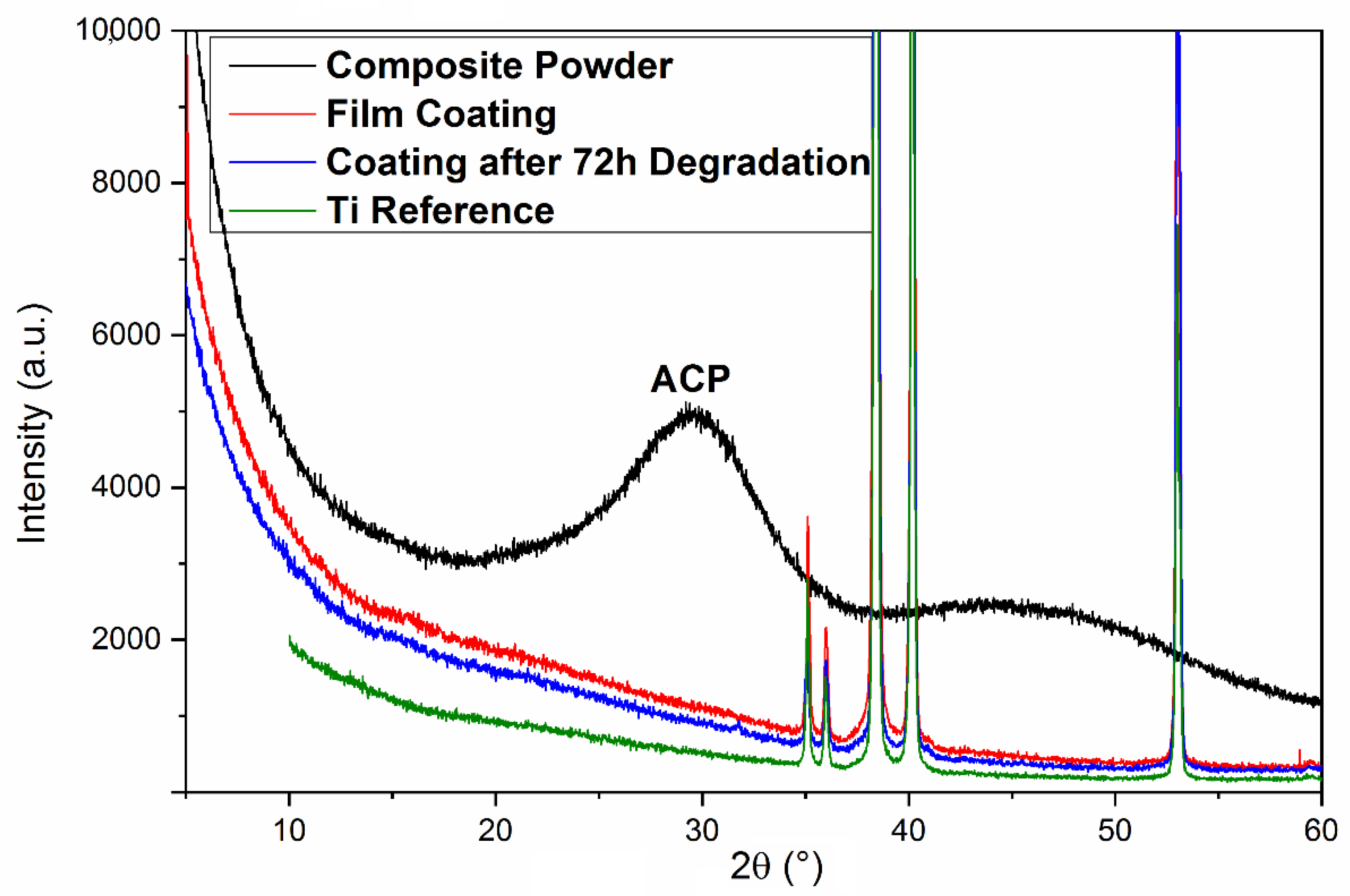
3.1.3. XRD
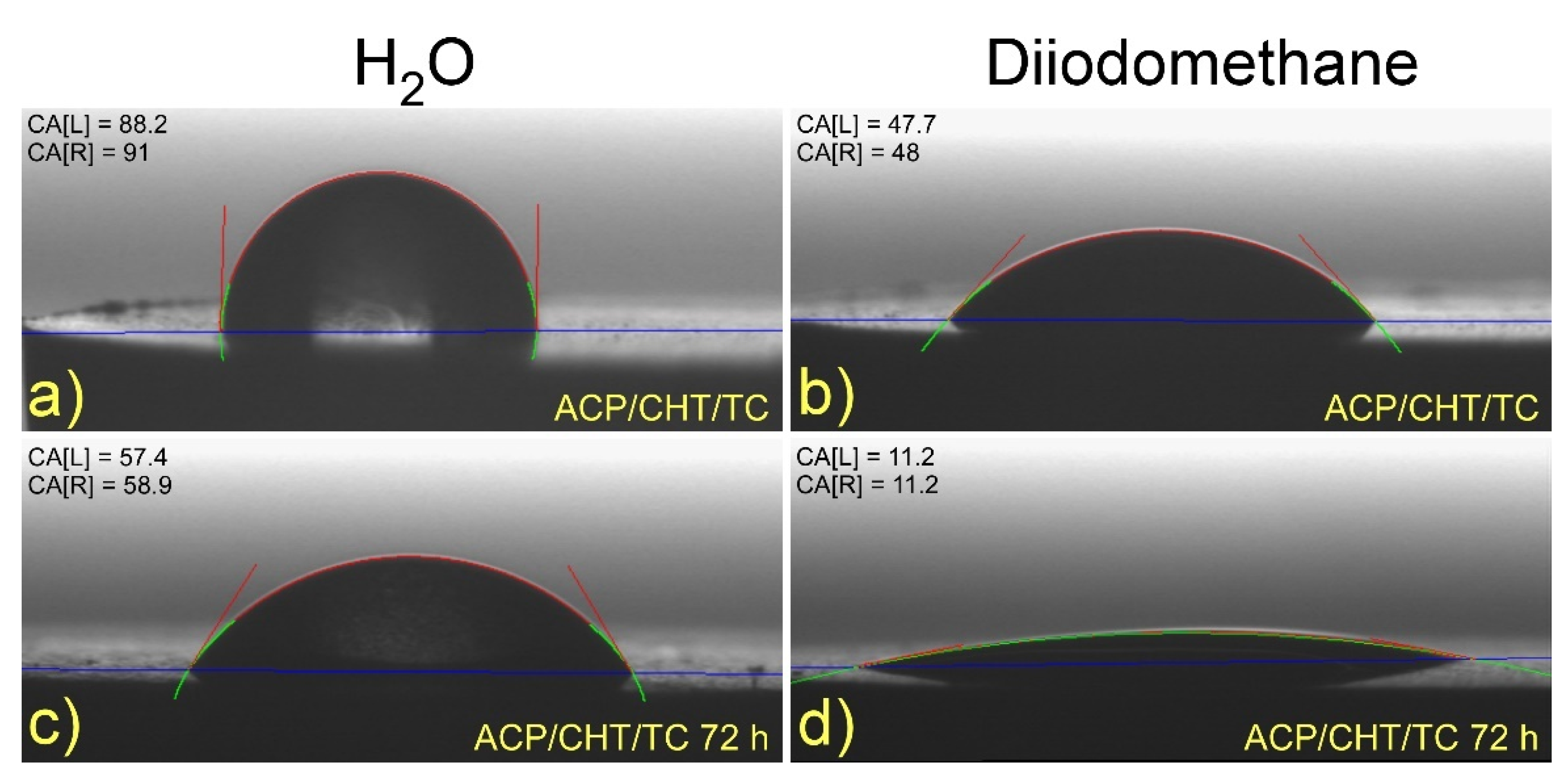
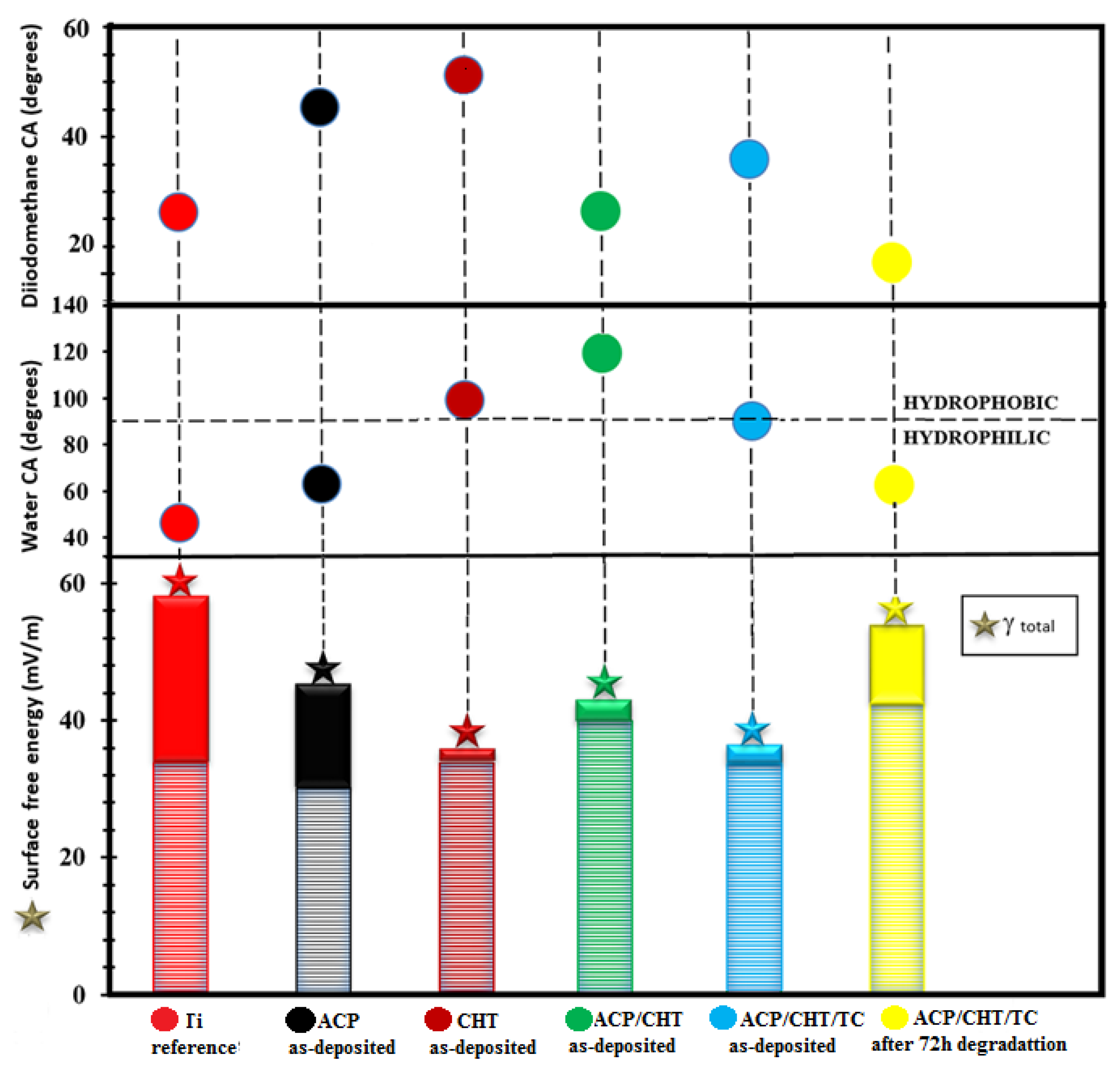
3.1.4. Wettability
- As-deposited films
- Ti is highly hydrophilic: 47° CA in deionized water/28° in diiodomethane and 56.2 mN/m SFE.
- A similar behavior is observed for ACP: 63°/42° and 44 mN/m.
- CHT is slightly hydrophobic: 98°/51° and 34 mN/m.
- ACP/CHT is super hydrophobic: 119°/50° and 42 mN/m.
- 2.
- After 72 h of immersion in SBF
3.2. Composite Mixture (Containing Drug) Delivery
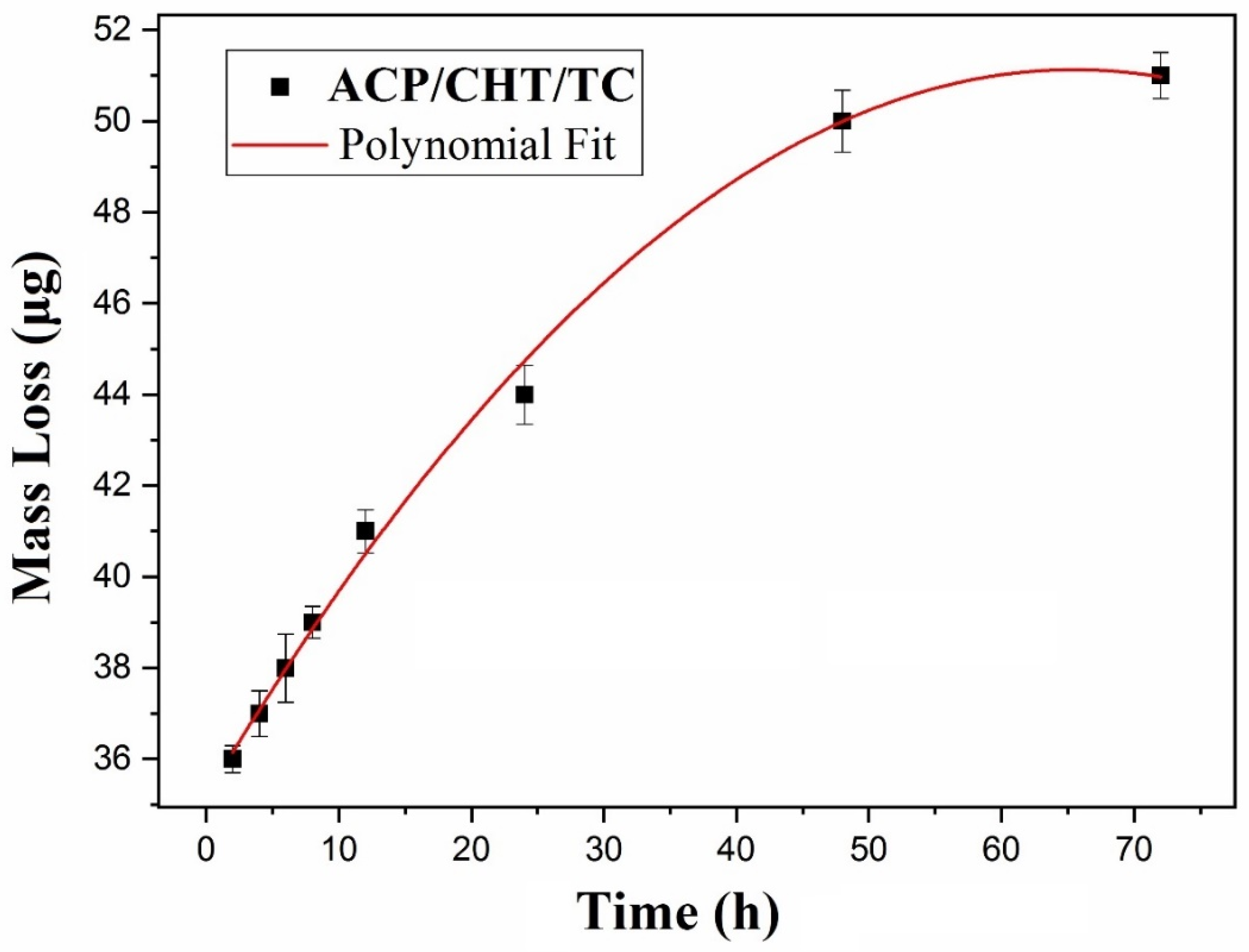
3.2.1. Mass Loss
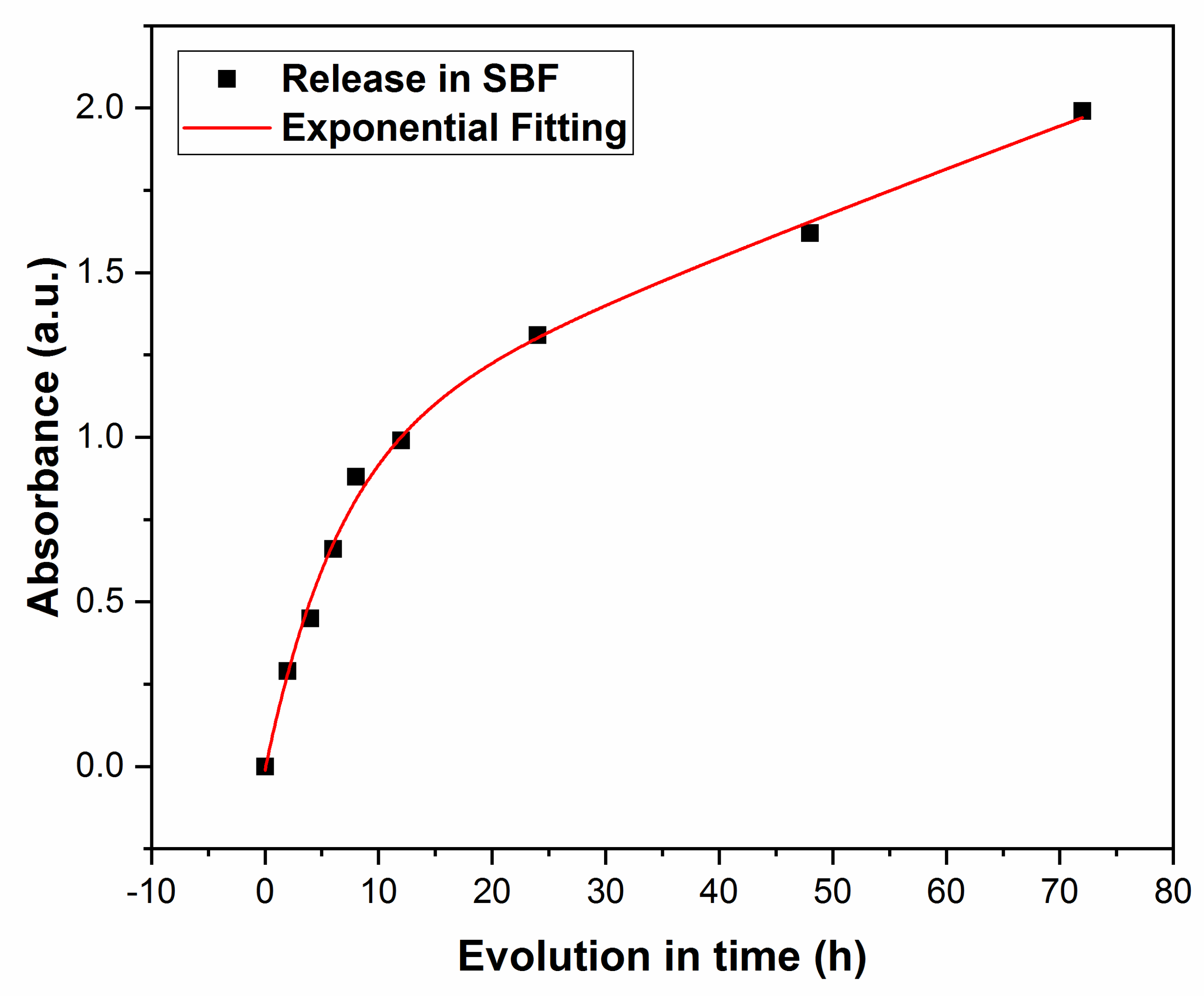
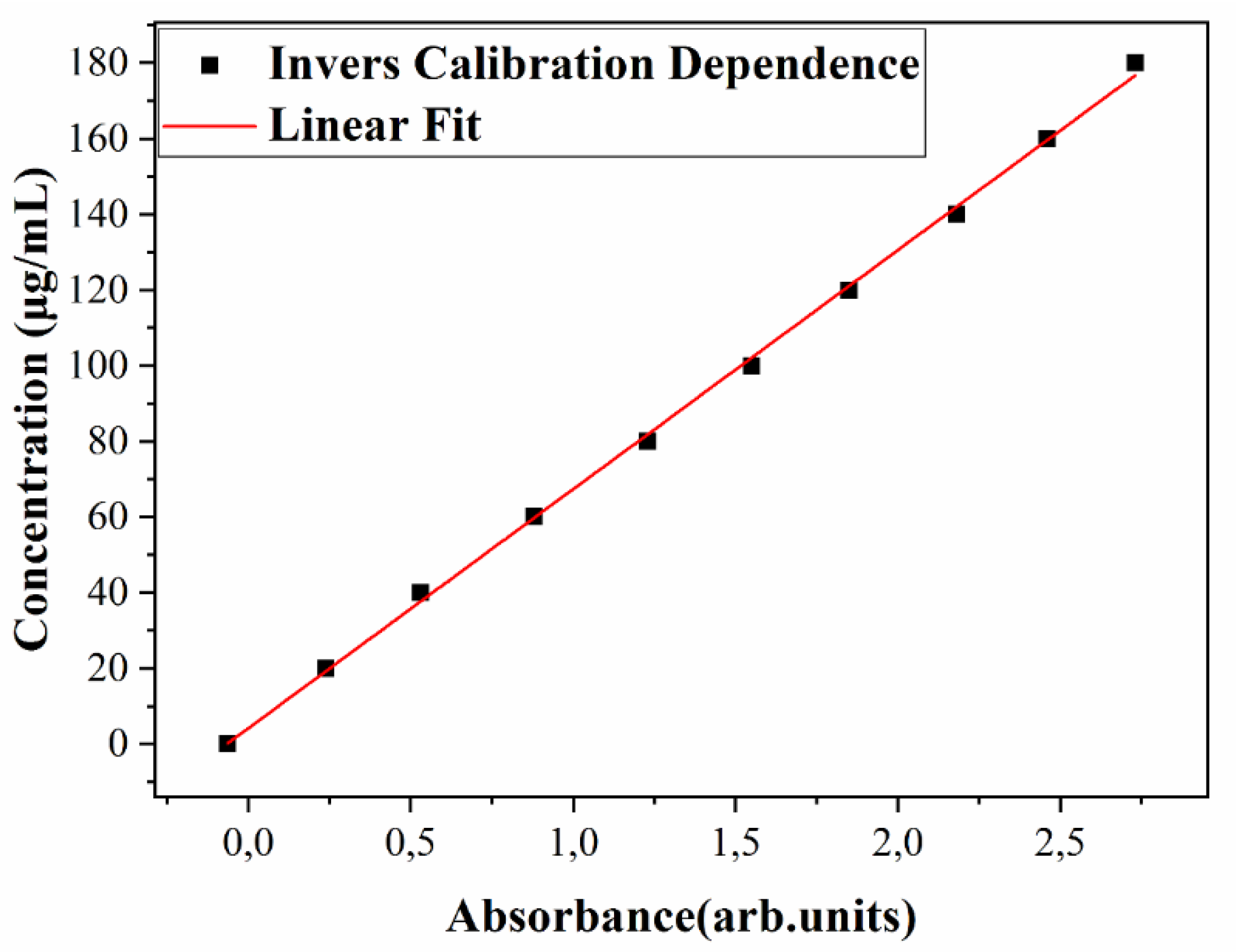
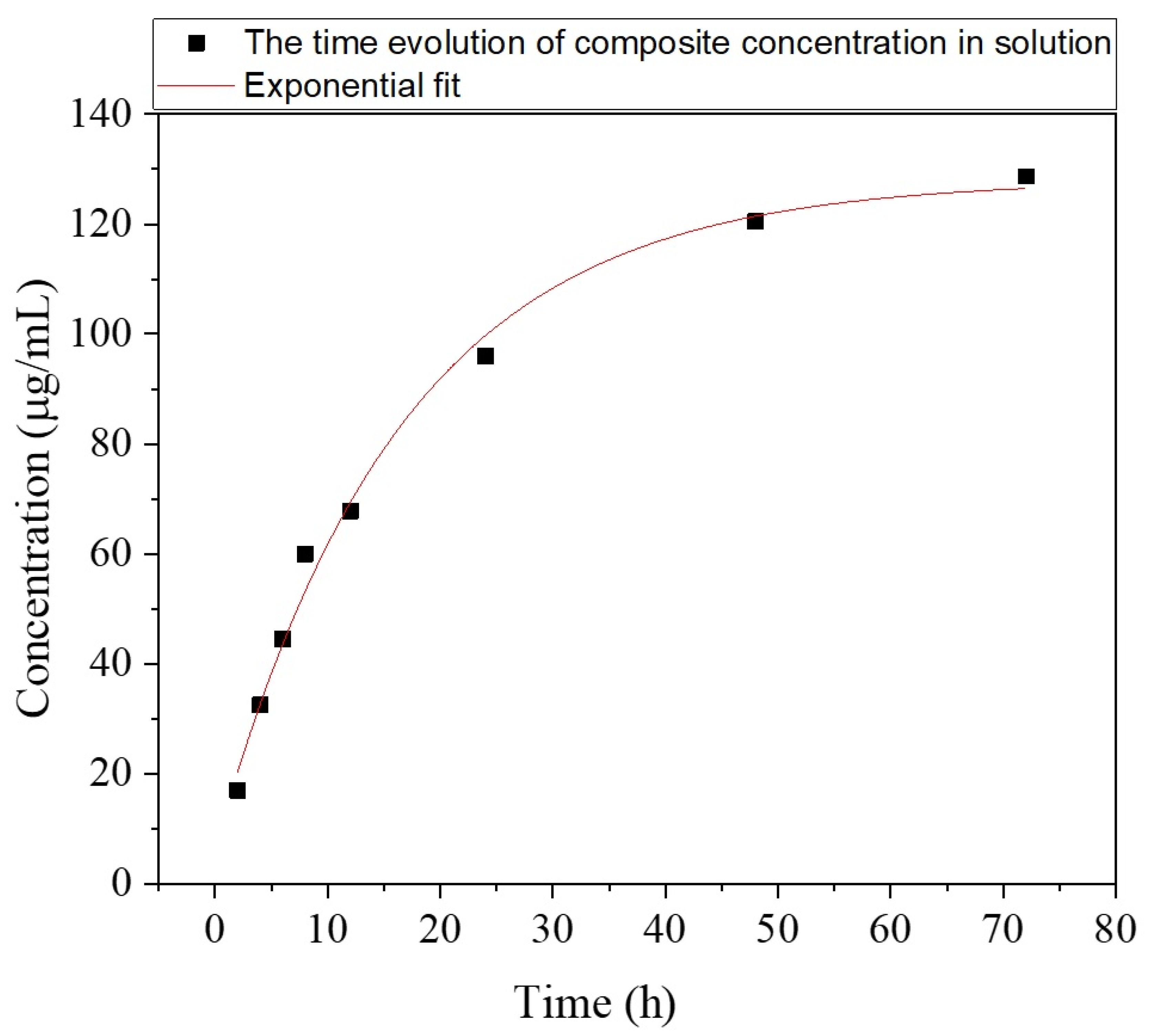
3.2.2. UV-Vis Studies
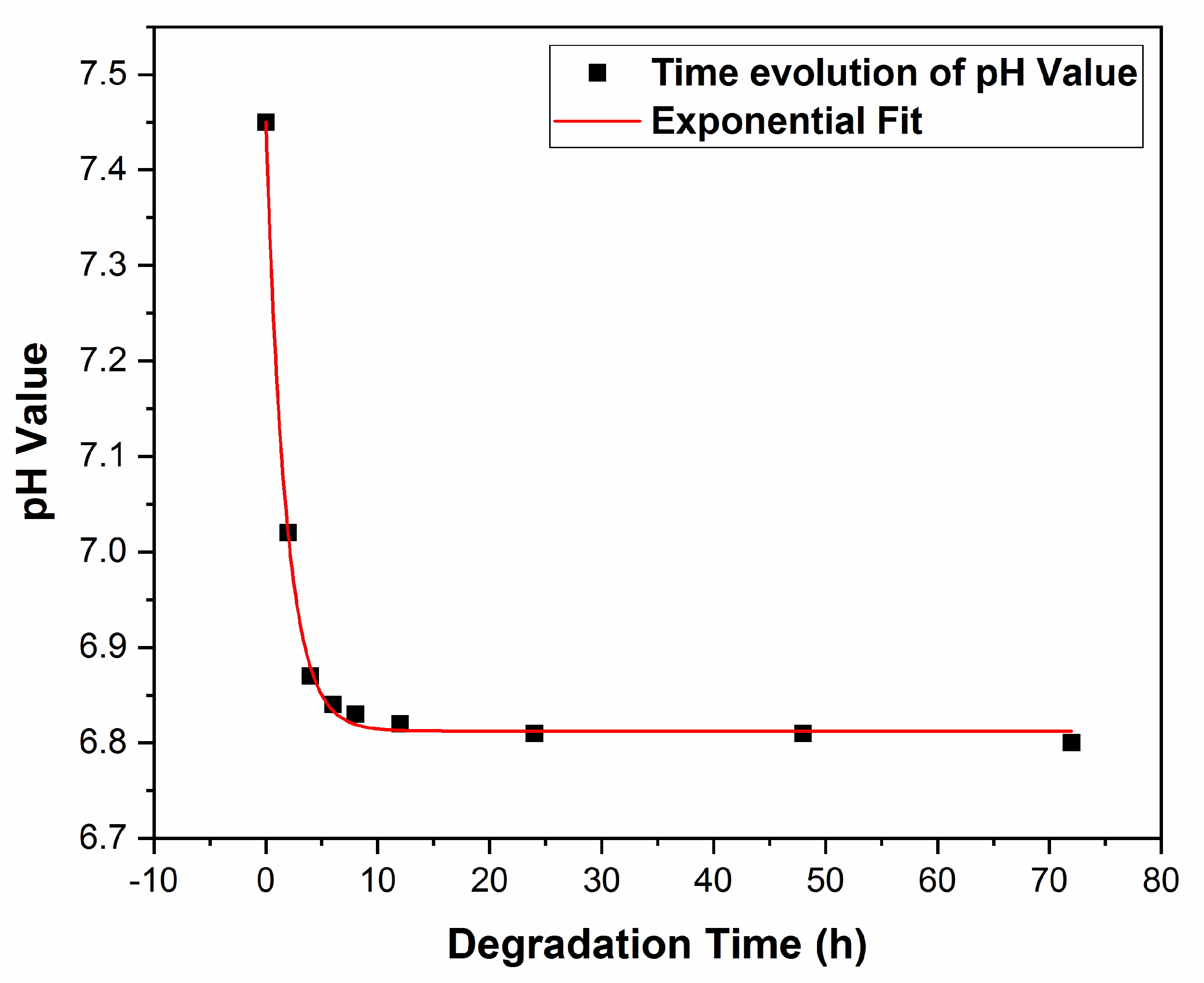
3.2.3. pH Measurements
3.3. Microbiological Assays
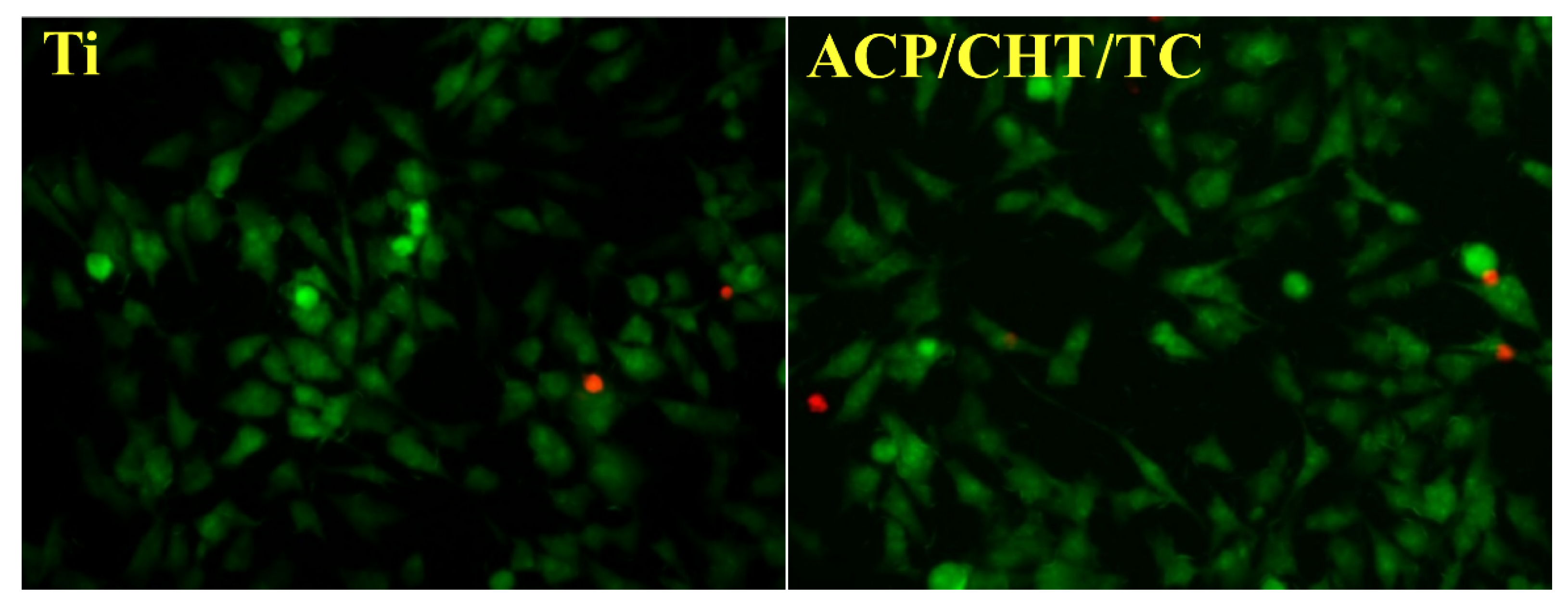
3.3.1. Cellular Morphology
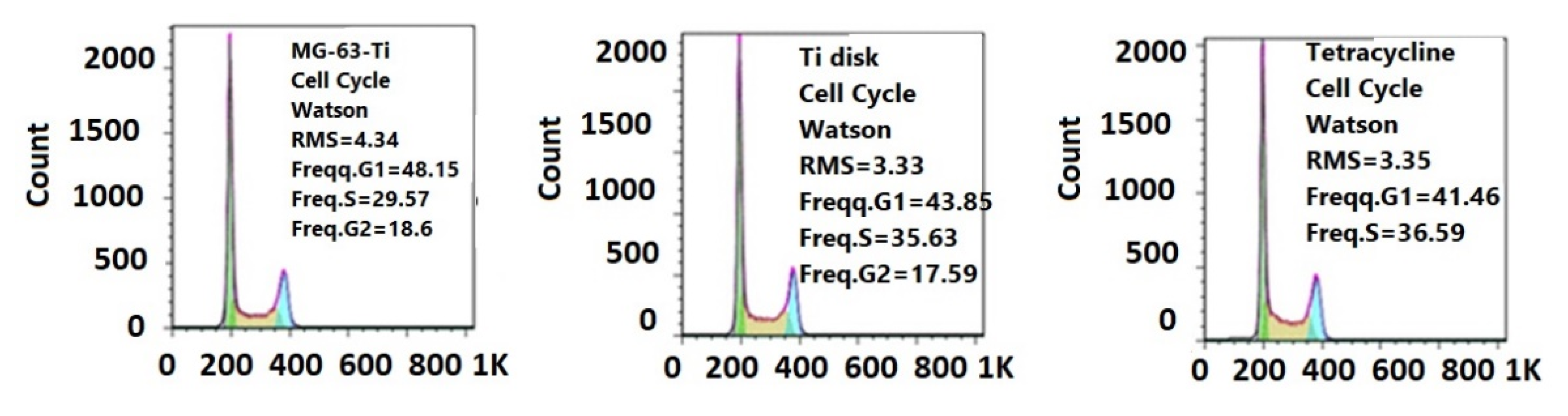
3.3.2. Cellular Cycle Assay
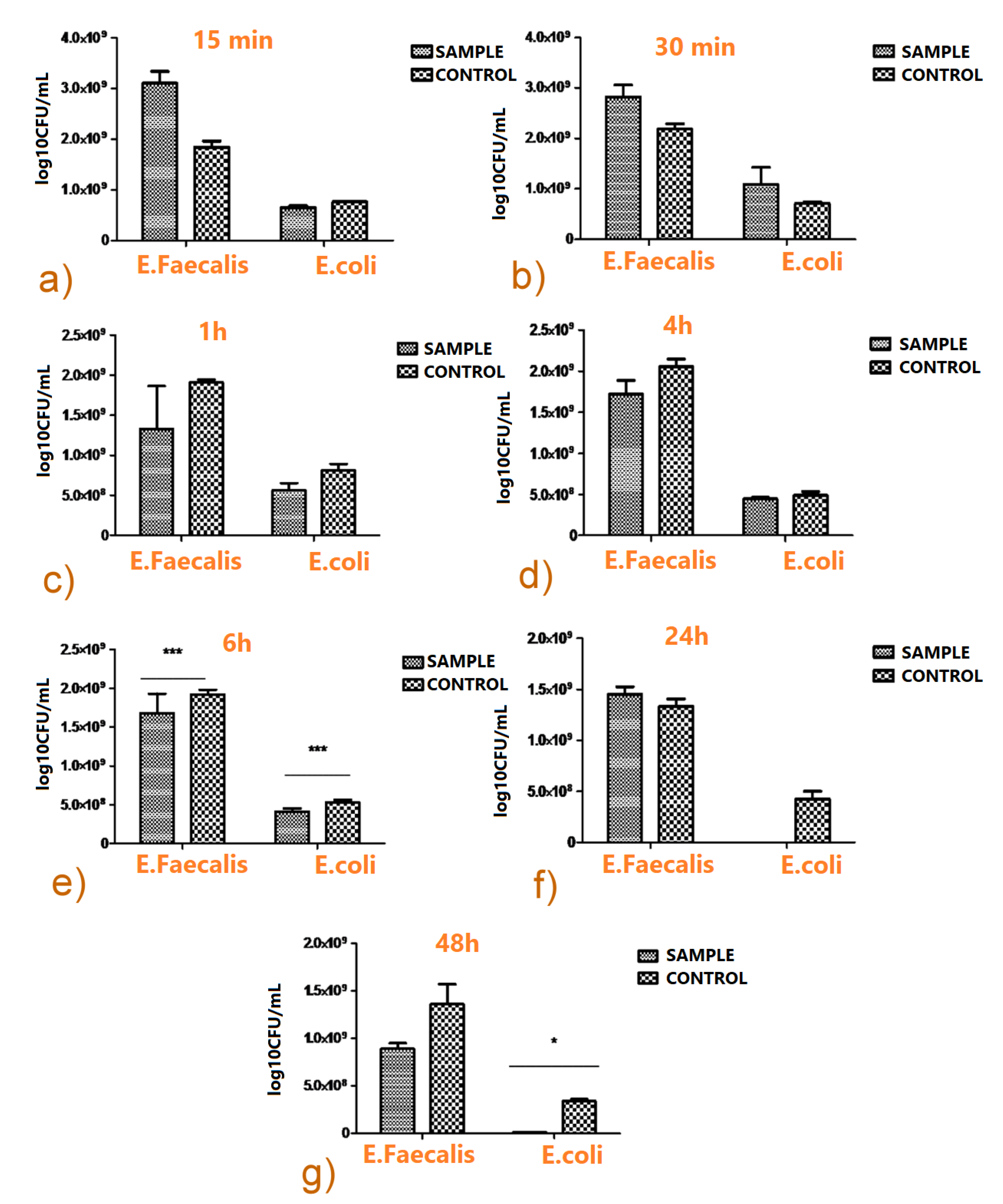
3.3.3. Antimicrobial Biofilm Activity
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Popescu-Pelin, G.; Ristoscu, C.; Duta, L.; Stan, G.E.; Pasuk, I.; Tite, T.; Stan, M.S.; Bleotu, C.; Popa, M.; Chifiriuc, M.C.; et al. Antimicrobial and Cytocompatible Bovine Hydroxyapatite-Alumina-Zeolite Composite Coatings Synthesized by Pulsed Laser Deposition from Low-Cost Sustainable Natural Resources. ACS Sustain. Chem. Eng. 2020, 8, 4026–4036. [Google Scholar] [CrossRef]
- Preda, M.; Mihai, M.M.; Popa, L.I.; Dițu, L.M.; Holban, A.M.; Manolescu, L.S.C.; Popa, G.L.; Muntean, A.A.; Gheorghe, I.; Chifiriuc, C.M.; et al. Phenotypic and genotypic virulence features of staphylococcal strains isolated from difficult-to-treat skin and soft tissue infections. PLoS ONE 2021, 16, e0246478. [Google Scholar] [CrossRef] [PubMed]
- Chaoui, L.; Mhand, R.; Mellouki, F.; Rhallabi, N. Contamination of the Surfaces of a Health Care Environment by Multidrug-Resistant (MDR) Bacteria. Int. J. Microbiol. 2019, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Gallo, J.; Holinka, M.; Moucha, C.S. Antibacterial Surface Treatment for Orthopaedic Implants. Int. J. Mol. Sci. 2014, 15, 13849–13880. [Google Scholar] [CrossRef]
- Popa, M.; Hussien, M.D.; Cirstea, A.; Grigore, R.; Lazar, V.; Bezirtzoglou, E.; Chifiriuc, M.C.; Sakizlian, M.; Stavropoulou, E.; Bertesteanu, S. Insights on Metal Based Dental Implants and their Interaction with the Surrounding Tissues. Curr. Top. Med. Chem. 2015, 15, 1614–1621. [Google Scholar] [CrossRef] [PubMed]
- Romanò, C.L.; Scarponi, S.; Gallazzi, E.; Romanò, D.; Drago, L. Antibacterial coating of implants in orthopaedics and trauma: A classification proposal in an evolving panorama. J. Orthop. Surg. Res. 2015, 10, 157. [Google Scholar] [CrossRef]
- Wang, S.Y.; Hu, H.Z.; Qing, X.C.; Zhang, Z.C.; Shao, Z.W. Recent advances of drug delivery nanocarriers in osteosarcoma treatment. J. Cancer 2020, 11, 69–82. [Google Scholar] [CrossRef]
- Sachan, R.; Jaipan, P.; Zhang, J.Y.; Degan, S.; Erdmann, D.; Tedesco, J.; Vanderwal, L.; Stafslien, S.J.; Negut, I.; Visan, A.; et al. Printing amphotericin B on microneedles using matrix-assisted pulsed laser evaporation. Int. J. Bioprint. 2017, 3, 147–157. [Google Scholar] [CrossRef]
- Li, D.; Gong, Y.; Chen, X.; Zhang, B.; Zhang, H.; Jin, P.; Li, H. Room-temperature deposition of hydroxyapatite/antibiotic composite coatings by vacuum cold spraying for antibacterial applications. Surf. Coat. Technol. 2017, 330, 87–91. [Google Scholar] [CrossRef]
- Visan, A.; Cristescu, R.; Stefan, N.; Miroiu, M.; Nita, C.; Socol, M.; Florica, C.; Rasoga, O.; Zgura, I.; Sima, L.E.; et al. Antimicrobial polycaprolactone/polyethylene glycol embedded lysozyme coatings of Ti implants for osteoblast functional properties in tissue engineering. Appl. Surf. Sci. 2017, 417, 234–243. [Google Scholar] [CrossRef]
- Pattar, V.P.; Nandibewoor, S.T. Electrochemical studies for the determination of an antibiotic drug, d-cycloserine, in pharmaceutical and human biological samples. J. Taibah Univ. Sci. 2016, 10, 92–99. [Google Scholar] [CrossRef][Green Version]
- Song, J.; Chen, Q.; Zhang, Y.; Diba, M.; Kolwijck, E.; Shao, J.; Jansen, J.A.; Yang, F.; Boccaccini, A.R.; Leeuwenburgh, S.C.G. Electrophoretic Deposition of Chitosan Coatings Modified with Gelatin Nanospheres to Tune the Release of Antibiotics. ACS Appl. Mater. Interfaces 2016, 8, 13785–13792. [Google Scholar] [CrossRef]
- Besleaga, C.; Stan, G.E.; Galca, A.C.; Ion, L.; Antohe, S. Double layer structure of ZnO thin films deposited by RF-magnetron sputtering on glass substrate. ApSS 2012, 258, 8819–8824. [Google Scholar] [CrossRef]
- Shankaraiah, G.; Saritha, P.; Bhagawan, D.; Himabindu, V.; Vidyavathi, S. Photochemical oxidation of antibiotic gemifloxacin in aqueous solutions—A comparative study. S. Afr. J. Chem. Eng. 2017, 24, 8–16. [Google Scholar] [CrossRef]
- Cristescu, R.; Surdu, A.V.; Grumezescu, A.M.; Oprea, A.E.; Trusca, R.; Vasile, O.; Dorcioman, G.; Visan, A.; Socol, G.; Mihailescu, I.N.; et al. Microbial colonization of biopolymeric thin films containing natural compounds and antibiotics fabricated by MAPLE. ApSS 2015, 336, 234–239. [Google Scholar] [CrossRef]
- Grumezescu, V.; Negut, I.; Grumezescu, A.M.; Ficai, A.; Dorcioman, G.; Socol, G.; Iordache, F.; Truşcă, R.; Vasile, B.S.; Holban, A.M. MAPLE fabricated coatings based on magnetite nanoparticles embedded into biopolymeric spheres resistant to microbial colonization. ApSS 2018, 448, 230–236. [Google Scholar] [CrossRef]
- Popescu-Pelin, G.; Fufă, O.; Popescu, R.C.; Savu, D.; Socol, M.; Zgură, I.; Holban, A.M.; Vasile, B.Ş.; Grumezescu, V.; Socol, G. Lincomycin-embedded PANI-based coatings for biomedical applications. ApSS 2018, 455, 653–666. [Google Scholar] [CrossRef]
- Cristescu, R.; Popescu, C.; Socol, G.; Visan, A.; Mihailescu, I.N.; Gittard, S.D.; Miller, P.R.; Martin, T.N.; Narayan, R.J.; Andronie, A.; et al. Deposition of antibacterial of poly(1,3-bis-(p-carboxyphenoxy propane)-co-(sebacic anhydride)) 20:80/gentamicin sulfate composite coatings by MAPLE. Appl. Surf. Sci. 2011, 257, 5287–5292. [Google Scholar] [CrossRef]
- Farrokhi-Rad, M.; Fateh, A.; Shahrabi, T. Electrophoretic deposition of vancomycin loaded halloysite nanotubes-chitosan nanocomposite coatings. Surf. Coat. Technol. 2018, 349, 144–156. [Google Scholar] [CrossRef]
- Zhou, W.; Jia, Z.; Xiong, P.; Yan, J.; Li, M.; Cheng, Y.; Zheng, Y. Novel pH-responsive tobramycin-embedded micelles in nanostructured multilayer-coatings of chitosan/heparin with efficient and sustained antibacterial properties. Mater. Sci. Eng. C Mater. Biol. Appl. 2018, 90, 693–705. [Google Scholar] [CrossRef]
- Alt, V.; Kirchhof, K.; Seim, F.; Hrubesch, I.; Lips, K.S.; Mannel, H.; Domann, E.; Schnettler, R. Rifampicin–fosfomycin coating for cementless endoprostheses: Antimicrobial effects against methicillin-sensitive Staphylococcus aureus (MSSA) and methicillin-resistant Staphylococcus aureus (MRSA). Acta Biomater. 2014, 10, 4518–4524. [Google Scholar] [CrossRef]
- Topsakal, A.; Ekren, N.; Kilic, O.; Oktar, F.N.; Mahirogullari, M.; Ozkan, O.; Sasmazel, H.T.; Turk, M.; Bogdan, I.M.; Stan, G.E.; et al. Synthesis and characterization of antibacterial drug loaded β-tricalcium phosphate powders for bone engineering applications. J. Mater. Sci. Mater. Med. 2020, 31, 1–17. [Google Scholar] [CrossRef]
- Radda’a, N.S.; Goldmann, W.H.; Detsch, R.; Roether, J.A.; Cordero-Arias, L.; Virtanen, S.; Moskalewicz, T.; Boccaccini, A.R. Electrophoretic deposition of tetracycline hydrochloride loaded halloysite nanotubes chitosan/bioactive glass composite coatings for orthopedic implants. Surf. Coat. Technol. 2017, 327, 146–157. [Google Scholar] [CrossRef]
- Zhang, Z.; Qu, Y.; Li, X.; Zhang, S.; Wei, Q.; Shi, Y.; Chen, L. Electrophoretic deposition of tetracycline modified silk fibroin coatings for functionalization of titanium surfaces. Appl. Surf. Sci. 2014, 303, 255–262. [Google Scholar] [CrossRef]
- Axente, E.; Ghimbeu, C.; Nita, C.; Airoudj, A.; Schrodj, G.; Sima, F.; Sopronyi, M. Matrix-Assisted Pulsed Laser Evaporation: A novel approach to design mesoporous carbon films. Carbon N. Y. 2017, 122, 484–495. [Google Scholar] [CrossRef]
- Miroiu, F.M.; Stefan, N.; Visan, A.I.; Nita, C.; Luculescu, C.R.; Rasoga, O.; Socol, M.; Zgura, I.; Cristescu, R.; Craciun, D.; et al. Composite biodegradable biopolymer coatings of silk fibroin—Poly(3-hydroxybutyric-acid-co-3-hydroxyvaleric-acid) for biomedical applications. ApSS 2015, 355, 1123–1131. [Google Scholar] [CrossRef]
- Mihailescu, I.N.; Bigi, A.; Gyorgy, E.; Ristoscu, C.; Sima, F.; Oner, E.T. Biomaterial thin films by soft pulsed laser technologies for biomedical applications. Springer Ser. Mater. Sci. 2014, 191, 271–294. [Google Scholar] [CrossRef]
- Mihailescu, I.N.; Caricato, A.P. Pulsed Laser Ablation: Advances and Applications in Nanoparticles and Nanostructuring thin Films, 1st ed.; Pan Stanford Publishing Pte. Ltd.: Singapore, 2018; ISBN 9781315185231. [Google Scholar]
- Ringeisen, B.R.; Callahan, J.; Wu, P.K.; Pique, A.; Spargo, B.; McGill, R.A.; Bucaro, M.; Kim, H.; Bubb, D.M.; Chrisey, D.B. Novel Laser-Based Deposition of Active Protein Thin Films. Langmuir 2001, 17, 3472–3479. [Google Scholar] [CrossRef]
- Kyzioł, A.; Khan, W.; Sebastian, V.; Kyzioł, K. Tackling microbial infections and increasing resistance involving formulations based on antimicrobial polymers. Chem. Eng. J. 2020, 385, 123888. [Google Scholar] [CrossRef]
- Eanes, E.D. Amorphous calcium phosphate. Monogr. Oral Sci. 2001, 18, 130–147. [Google Scholar] [CrossRef]
- Drouet, C.; Gomez-Morales, J.; Iafisco, M.; Sarda, S. Calcium phosphate surface tailoring technologies for drug delivering and tissue engineering. In Surface Tailoring of Inorganic Materials for Biomedical Applications; Bentham Science Publishers: Sharjah, UAE, 2012; pp. 43–111. [Google Scholar] [CrossRef]
- Niu, X.; Liu, Z.; Tian, F.; Chen, S.; Lei, L.; Jiang, T.; Feng, Q.; Fan, Y. Sustained delivery of calcium and orthophosphate ions from amorphous calcium phosphate and poly(L-lactic acid)-based electrospinning nanofibrous scaffold. Sci. Rep. 2017, 7, 1–9. [Google Scholar] [CrossRef]
- Combes, C.; Rey, C. Amorphous calcium phosphates: Synthesis, properties and uses in biomaterials. Acta Biomater. 2010, 6, 3362–3378. [Google Scholar] [CrossRef]
- Sun, R.; Åhlén, M.; Tai, C.-W.; Bajnóczi, É.G.; de Kleijne, F.; Ferraz, N.; Persson, I.; Strømme, M.; Cheung, O. Highly Porous Amorphous Calcium Phosphate for Drug Delivery and Bio-Medical Applications. Nanomaterials 2019, 10, 20. [Google Scholar] [CrossRef] [PubMed]
- Visan, A.; Grossin, D.; Stefan, N.; Duta, L.; Miroiu, F.M.; Stan, G.E.; Sopronyi, M.; Luculescu, C.; Freche, M.; Marsan, O.; et al. Biomimetic nanocrystalline apatite coatings synthesized by Matrix Assisted Pulsed Laser Evaporation for medical applications. Mater. Sci. Eng. B 2014, 181, 56–63. [Google Scholar] [CrossRef]
- Iafisco, M.; Palazzo, B.; Falini, G.; Foggia, M.D.; Bonora, S.; Nicolis, S.; Casella, L.; Roveri, N. Adsorption and conformational change of myoglobin on biomimetic hydroxyapatite nanocrystals functionalized with alendronate. Langmuir 2008, 24, 4924–4930. [Google Scholar] [CrossRef] [PubMed]
- Roveri, N.; Palazzo, B.; Iafisco, M. The role of biomimetism in developing nanostructured inorganic matrices for drug delivery. Expert Opin. Drug Deliv. 2008, 5, 861–877. [Google Scholar] [CrossRef] [PubMed]
- Barroug, A.; Kuhn, L.T.; Gerstenfeld, L.C.; Glimcher, M.J. Interactions of cisplatin with calcium phosphate nanoparticles: In vitro controlled adsorption and release. J. Orthop. Res. 2004, 22, 703–708. [Google Scholar] [CrossRef]
- Allegrini, S.; da Silva, A.C.; Tsujita, M.; Salles, M.B.; Gehrke, S.A.; Braga, F.J.C. Amorphous calcium phosphate (ACP) in tissue repair process. Microsc. Res. Tech. 2018, 81, 579–589. [Google Scholar] [CrossRef]
- Cazalbou, S.; Bertrand, G.; Drouet, C. Tetracycline-Loaded Biomimetic Apatite: An Adsorption Study. J. Phys. Chem. B 2015, 119, 3014–3024. [Google Scholar] [CrossRef]
- Gautier, H.; Daculsi, G.; Merle, C. Association of vancomycin and calcium phosphate by dynamic compaction: In vitro characterization and microbiological activity. Biomaterials 2001, 22, 2481–2487. [Google Scholar] [CrossRef]
- Liu, Y.; Enggist, L.; Kuffer, A.F.; Buser, D.; Hunziker, E.B. The influence of BMP-2 and its mode of delivery on the osteoconductivity of implant surfaces during the early phase of osseointegration. Biomaterials 2007, 28, 2677–2686. [Google Scholar] [CrossRef]
- Thanyaphoo, S.; Kaewsrichan, J. Synthesis and evaluation of novel glass ceramics as drug delivery systems in osteomyelitis. J. Pharm. Sci. 2012, 101, 2870–2882. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Wang, C.; Wang, J.; Qu, Y.; Liu, G. In vivo drug release and antibacterial properties of vancomycin loaded hydroxyapatite/chitosan composite. Drug Deliv. 2012, 19, 264–269. [Google Scholar] [CrossRef] [PubMed]
- Uskoković, V.; Desai, T.A. Phase composition control of calcium phosphate nanoparticles for tunable drug delivery kinetics and treatment of osteomyelitis. I. Preparation and drug release. J. Biomed. Mater. Res. A 2013, 101, 1416–1426. [Google Scholar] [CrossRef] [PubMed]
- Chifiriuc, C.M.; Grumezescu, A.M.; Saviuc, C.; Croitoru, C.; Mihaiescu, D.E.; Lazar, V. Improved antibacterial activity of cephalosporins loaded in magnetic chitosan microspheres. Int. J. Pharm. 2012, 436, 201–205. [Google Scholar] [CrossRef]
- Banerjee, M.; Mallick, S.; Paul, A.; Chattopadhyay, A.; Ghosh, S.S. Heightened reactive oxygen species generation in the antimicrobial activity of a three component iodinated chitosan-silver nanoparticle composite. Langmuir 2010, 26, 5901–5908. [Google Scholar] [CrossRef]
- Costa, F.; Maia, S.; Gomes, J.; Gomes, P.; Martins, M.C. Characterization of hLF1-11 immobilization onto chitosan ultrathin films, and its effects on antimicrobial activity. Acta Biomater. 2014, 10, 3513–3521. [Google Scholar] [CrossRef]
- Anaya-Esparza, L.M.; Ruvalcaba-Gómez, J.M.; Maytorena-Verdugo, C.I.; González-Silva, N.; Romero-Toledo, R.; Aguilera-Aguirre, S.; Pérez-Larios, A.; Montalvo-González, E. Chitosan-TiO2: A Versatile Hybrid Composite. Materials 2020, 13, 811. [Google Scholar] [CrossRef]
- Fan, J.; Bi, L.; Wu, T.; Cao, L.; Wang, D.; Nan, K.; Chen, J.; Jin, D.; Jiang, S.; Pei, G. A combined chitosan/nano-size hydroxyapatite system for the controlled release of icariin. J. Mater. Sci. Mater. Med. 2012, 23, 399–407. [Google Scholar] [CrossRef]
- Visan, A.; Stan, G.E.; Ristoscu, C.; Popescu-Pelin, G.; Sopronyi, M.; Besleaga, C.; Luculescu, C.; Chifiriuc, M.C.; Hussien, M.D.; Marsan, O.; et al. Combinatorial MAPLE deposition of antimicrobial orthopedic maps fabricated from chitosan and biomimetic apatite powders. Int. J. Pharm. 2016, 511, 505–515. [Google Scholar] [CrossRef]
- Dashti, A.; Ready, D.; Salih, V.; Knowles, J.; Barralet, J.E.; Wilson, M.; Donos, N.; Nazhat, S.N. In vitro antibacterial efficacy of tetracycline hydrochloride adsorbed onto Bio-Oss bone graft. J. Biomed. Mater. Res. B Appl. Biomater. 2010, 93, 394–400. [Google Scholar] [CrossRef]
- Le Grill, S.; Soulie, J.; Coppel, Y.; Roblin, P.; Lecante, P.; Marsan, O.; Charvillat, C.; Bertrand, G.; Rey, C.; Brouillet, F. Spray-drying-derived amorphous calcium phosphate: A multi-scale characterization. J. Mater. Sci. 2021, 56, 1189–1202. [Google Scholar] [CrossRef]
- Menzies, K.L.; Jones, L. The impact of contact angle on the biocompatibility of biomaterials. Optom. Vis. Sci. 2010, 87, 387–399. [Google Scholar] [CrossRef]
- Bumgardner, J.D.; Wiser, R.; Elder, S.H.; Jouett, R.; Yang, Y.; Ong, J.L. Contact angle, protein adsorption and osteoblast precursor cell attachment to chitosan coatings bonded to titanium. J. Biomater. Sci. Polym. Ed. 2003, 14, 1401–1409. [Google Scholar] [CrossRef]
- Torchinsky, I.; Rosenman, G. Wettability Modification of Nanomaterials by Low-Energy Electron Flux. Nanoscale Res. Lett. 2009, 4, 1209–1217. [Google Scholar] [CrossRef] [PubMed]
- Kokubo, T.; Kushitani, H.; Sakka, S.; Kitsugi, T.; Yamamuro, T. Solutions able to reproduce in vivo surface-structure changes in bioactive glass-ceramic A-W. J. Biomed. Mater. Res. 1990, 24, 721–734. [Google Scholar] [CrossRef] [PubMed]
- Boyan, B.D.; Lotz, E.M.; Schwartz, Z. * Roughness and Hydrophilicity as Osteogenic Biomimetic Surface Properties. Tissue Eng. Part A 2017, 23, 1479–1489. [Google Scholar] [CrossRef] [PubMed]
- Zhong, W.; Sun, L.; Yu, T.; Zhou, C. Preparation and characterization of calcium phosphate cement with enhanced tissue adhesion for bone defect repair. Ceram. Int. 2021, 47, 1712–1720. [Google Scholar] [CrossRef]
- Malakauskaite-Petruleviciene, M.; Stankeviciute, Z.; Niaura, G.; Garskaite, E.; Beganskiene, A.; Kareiva, A. Characterization of sol-gel processing of calcium phosphate thin films on silicon substrate by FTIR spectroscopy. Vib. Spectrosc. 2016, 85, 16–21. [Google Scholar] [CrossRef]
- Pleshko, N.; Boskey, A.; Mendelsohn, R. Novel infrared spectroscopic method for the determination of crystallinity of hydroxyapatite minerals. Biophys. J. 1991, 60, 786. [Google Scholar] [CrossRef]
- Zeng, H.; Lacefield, W.R. XPS, EDX and FTIR analysis of pulsed laser deposited calcium phosphate bioceramic coatings: The effects of various process parameters. Biomaterials 2000, 21, 23–30. [Google Scholar] [CrossRef]
- Uskoković, V.; Abuna, G.; Ferreira, P.; Wu, V.M.; Gower, L.; Pires-de-Souza, F.C.P.; Murata, R.M.; Sinhoreti, M.A.C.; Geraldeli, S. Synthesis and characterization of nanoparticulate niobium- and zinc-doped bioglass-ceramic/chitosan hybrids for dental applications. J. Sol. Gel. Sci. Technol. 2021, 97, 245–258. [Google Scholar] [CrossRef]
- Ahamad, T.; Naushad, M.; Al-Shahrani, T.; Al-Hokbany, N.; Alshehri, S.M. Preparation of chitosan based magnetic nanocomposite for tetracycline adsorption: Kinetic and thermodynamic studies. Int. J. Biol. Macromol. 2020, 147, 258–267. [Google Scholar] [CrossRef]
- Andrade, Â.L.; Militani, I.A.; de Almeida, K.J.; Belchior, J.C.; Dos Reis, S.C.; Costa e Silva, R.M.F.; Domingues, R.Z. Theoretical and Experimental Studies of the Controlled Release of Tetracycline Incorporated into Bioactive Glasses. AAPS PharmSciTech 2018, 19, 1287–1296. [Google Scholar] [CrossRef] [PubMed]
- Rather, A.M.; Shome, A.; Bhunia, B.K.; Panuganti, A.; Mandal, B.B.; Manna, U. Simultaneous and controlled release of two different bioactive small molecules from nature inspired single material. J. Mater. Chem. B 2018, 6, 7692–7702. [Google Scholar] [CrossRef]
- Mihailescu, I.N.; Hermann, J. Laser–Plasma Interactions. Ser. Mater. Sci. 2010, 139, 49–88. [Google Scholar] [CrossRef]
- Anbazhagan, S.; Thangavelu, K.P. Application of tetracycline hydrochloride loaded-fungal chitosan and Aloe vera extract based composite sponges for wound dressing. J. Adv. Res. 2018, 14, 63–71. [Google Scholar] [CrossRef] [PubMed]
- Corbet, C.; Feron, O. Tumour acidosis: From the passenger to the driver’s seat. Nat. Rev. Cancer 2017, 17, 577–593. [Google Scholar] [CrossRef] [PubMed]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Visan, A.I.; Ristoscu, C.; Popescu-Pelin, G.; Sopronyi, M.; Matei, C.E.; Socol, G.; Chifiriuc, M.C.; Bleotu, C.; Grossin, D.; Brouillet, F.; et al. Composite Drug Delivery System Based on Amorphous Calcium Phosphate–Chitosan: An Efficient Antimicrobial Platform for Extended Release of Tetracycline. Pharmaceutics 2021, 13, 1659. https://doi.org/10.3390/pharmaceutics13101659
Visan AI, Ristoscu C, Popescu-Pelin G, Sopronyi M, Matei CE, Socol G, Chifiriuc MC, Bleotu C, Grossin D, Brouillet F, et al. Composite Drug Delivery System Based on Amorphous Calcium Phosphate–Chitosan: An Efficient Antimicrobial Platform for Extended Release of Tetracycline. Pharmaceutics. 2021; 13(10):1659. https://doi.org/10.3390/pharmaceutics13101659
Chicago/Turabian StyleVisan, Anita Ioana, Carmen Ristoscu, Gianina Popescu-Pelin, Mihai Sopronyi, Consuela Elena Matei, Gabriel Socol, Mariana Carmen Chifiriuc, Coralia Bleotu, David Grossin, Fabien Brouillet, and et al. 2021. "Composite Drug Delivery System Based on Amorphous Calcium Phosphate–Chitosan: An Efficient Antimicrobial Platform for Extended Release of Tetracycline" Pharmaceutics 13, no. 10: 1659. https://doi.org/10.3390/pharmaceutics13101659
APA StyleVisan, A. I., Ristoscu, C., Popescu-Pelin, G., Sopronyi, M., Matei, C. E., Socol, G., Chifiriuc, M. C., Bleotu, C., Grossin, D., Brouillet, F., Grill, S. L., Bertrand, G., Zgura, I., Cristescu, R., & Mihailescu, I. N. (2021). Composite Drug Delivery System Based on Amorphous Calcium Phosphate–Chitosan: An Efficient Antimicrobial Platform for Extended Release of Tetracycline. Pharmaceutics, 13(10), 1659. https://doi.org/10.3390/pharmaceutics13101659














