1. Introduction
Pemetrexed disodium heptahydrate,
N-(4-(2-(2-Amino-3,4-dihydro-4-oxo-7H-pyrrolo(2,3-d)pyrimdin-5-yl)ethyl)benzoyl)glutamic acid·2Na·7H
2O, is a synthetic folate analog metabolic inhibitor which can disrupt folate-dependent metabolic processes that are required for cell replication, and it has been used in the treatment of various cancers [
1]. Alimta
® (Eli Lilly and Company, Indianapolis, IN, USA) containing pemetrexed disodium heptahydrate as an active ingredient was registered by the US Food and Drug Administration (FDA) for the treatment of mesothelioma in 2007 [
2,
3]. It is formulated as a lyophilized cake in a single-dose vial that is reconstituted with 0.9% sodium chloride injection, to make a solution of pemetrexed at a concentration of 25 mg/mL prior to administration. The appropriate volume of reconstituted pemetrexed solution should be diluted further to 100 mL with 0.9% sodium chloride injection and administered as an IV infusion over 10 min. All procedures for the reconstitution and infusion of solution should be performed immediately because there are no antimicrobial preservatives present in the formulation [
4]. In addition, the chemical and physical stability of the reconstituted pemetrexed solution has been demonstrated to last only for up to 24 h after the reconstitution of the original vial, when stored at controlled room temperature [
4,
5]. Owing to the disadvantages of such a lyophilized injection, there has been a requirement for the development of a ready to use (RTU) pemetrexed liquid injection; however, the relatively rapid formation of degradants has been the main challenging factor so far in developing it as a commercially available final drug product [
6,
7,
8,
9].
The mechanism of degradation has been reported previously [
7]. Under acidic conditions, decarboxylation of glutamic acid is observed, and des-glutamate and glutamic acid are generated. Under alkaline conditions, degradation proceeds by hydrolysis of the side-chain amide, followed by deamination. In the presence of oxygen, three oxidative degradants, α-hydroxy lactams, keto-pemetrexed, and oxidative dimers are formed by the oxidation pathway. Although several studies have attempted to improve the instability of pemetrexed in aqueous solution state, none of them have provided useful information that can be utilized in actual product development.
In general, following parameters can affect the stability of injectable drug solutions: temperature, exposure to light, pH of the solution, presence of additives, concentration of API, presence of oxygen, and duration of storage [
10,
11,
12,
13,
14,
15,
16,
17,
18,
19]. In order to stabilize the injectable drug solution, control strategies have been attempted after thorough understanding of the critical quality attributions (CQA), critical material attributes (CMA) and critical process parameters (CPP) in relation to the formulation and manufacturing process, and considering the quality of the intermediate and final products [
20]. Thus, the control strategies can be categorized into (i) control of excipients, (ii) control of formulation, (iii) control of process, and (iv) control of packaging [
21,
22].
The basis for the content criteria of related substances is to establish a validated quantification method and select acceptable criteria range for individual and total impurity in accordance with the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) guidelines [
23,
24,
25]. Extensive stability screening studies that are conducted based on well-designed experiments are required to establish the content criteria of related substances, and to develop a stable formulation that can meet the criteria.
To the best of our knowledge, before this study, there was no systematic study conducted on the stabilization of pemetrexed solution by using multiple approaches in various ways according to the recommendation by the ICH guidelines. Owing to limited data available from previous reports, well-designed control strategies, following a greater understanding of the degradation of pemetrexed, are necessary because they would contribute to the improvement of the stability of the injectable solution formulations of pemetrexed, which in turn may increase the utilization and safety of this drug.
Thus, the aim of this study was to evaluate the stability of pemetrexed in aqueous solution under various control strategies, to acquire beneficial information for the minimization of degradation without color change. First, a forced degradation study was performed to determine the degradation mechanism, including the main pathway responsible for the decomposition of pemetrexed in solution state. Of the four control strategies mentioned above, three were attempted, except for the category (i) control of excipients, which was not attempted because an auto-oxidation susceptible excipient was not used for the preparation of pemetrexed injectable solution in this study. The effects of pH, drug concentration, and addition of antioxidants were evaluated as the formulation controls. In addition, the dissolved oxygen (DO) levels in the solution controlled by nitrogen purging and oxygen level in headspace controlled by nitrogen headspace were evaluated as controls of process and packaging, respectively. The decrease in drug content and the increase in degradation products were monitored by quantitative methods using HPLC, and the change in color due to decomposition was measured by a colorimetric method. In addition, the amounts of antioxidants consumed by oxidation were measured by their quantification methods, and a correlation with the degradation pattern of pemetrexed was investigated to evaluate the potential of antioxidants as sacrificial reductants.
2. Materials and Methods
2.1. Materials
Pemetrexed disodium hemipentahydrate was purchased from Nippon Kayaku (Tokyo, Japan). d-mannitol was obtained from Roquette (Corby, UK). Hydrochloric acid (HCl), sodium hydroxide (NaOH), hydrogen peroxide, potassium dihydrogen phosphate, ortho-phosphoric acid (PA), sodium carbonate, sodium bicarbonate, sodium formaldehyde bisulfite, sodium sulfate, and sodium sulfite were purchased from Merck (Darmstadt, Germany). The pemetrexed-related substances, α-hydroxy lactams, keto-pemetrexed, oxidative dimers, and ring-opened keto-amide were obtained from TRC (Toronto, ON, Canada). N-acetylcysteine (NAC, Kyowa Hakko Bio Co., Ltd., Tokyo, Japan), l-cysteine·HCl, sodium sulfide, and sodium metabisulfite were purchased from Sigma Aldrich (Saint Louis, MO, USA). Hydroxypropyl-β-cyclodextrin (Kleptose® HPB, Roquette-Freres, Lestrem CEDEX, France) and d-α-tocopheryl polyethylene glycol succinate (Vitamin E TPGS, Eastman Chemical Co., Kingsport, TN, USA) were also utilized. Acetonitrile and acetone were obtained from Fisher Scientific (Pittsburgh, PA, USA). All other chemicals were of reagent grade and used without further purification.
2.2. Analytical Method of Pemetrexed Impurities
Reverse-phase high-performance liquid chromatography (RP-HPLC) was performed to analyze the contents of pemetrexed and its related substances. RP-HPLC was conducted as previously described, but with slight modifications [
7]. HPLC analysis was performed by using a Dionex Ultimate 3000 RS HPLC system (Dionex Corporation, Sunnyvale, CA, USA) equipped with an Ultimate 3000 RS diode array detector and pump, as well as an RP C8 column (Zobrax RX-C8, 5 µm, 4.6 × 250 mm, Agilent Technologies, Palo Alto, CA, USA). The auto-sampler and column temperature were maintained at 4 and 30 °C, respectively. The injection volume was 10 µL, and the flow rate was maintained at 1.0 mL/min. Two mobile phases were prepared for the gradient elution. Mobile phase A was composed of 1.36 g/L potassium dihydrogen phosphate at pH 2.5, adjusted by 85% ortho-phosphoric acid. Mobile phase B was composed of acetonitrile. The elution program for the binary gradient system is shown in
Table 1. UV detection was performed at 277 nm. The calibration curve of pemetrexed showed a good linearity over the concentration range of 78–121 µg/mL and was used in this study with a correlation coefficient (R
2) of 0.999. The content (%) of impurities was calculated by the following equation based on the United States Pharmacopeia (USP) monograph of pemetrexed for injection [
26,
27]:
where A
U is the peak area of each impurity from the sample solution, A
T is the total peak area from the sample solution, and RRF is the relative response factor for each impurity. The total impurity content was obtained based on the sum of the peak areas corresponding to all impurities, and RRF
i = 1.
2.3. Colorimetric Determination
The color change by degradation was evaluated by color measurement, using a colorimeter (Konica-Minolta CM-3500d spectrocolorimeter, Konica Minolta, Osaka, Japan). The b*, one of the chromaticity coordinates in color space defined by International Commission on Illumination (CIE), was measured by using the D65 illuminant and 10° standard observer, using 2 mL plastic cuvettes. Based on the chromaticity diagram of color space, positive value of b* indicates the change to yellowish colors and negative value of b* indicates the change to bluish colors.
2.4. Forced Degradation Test
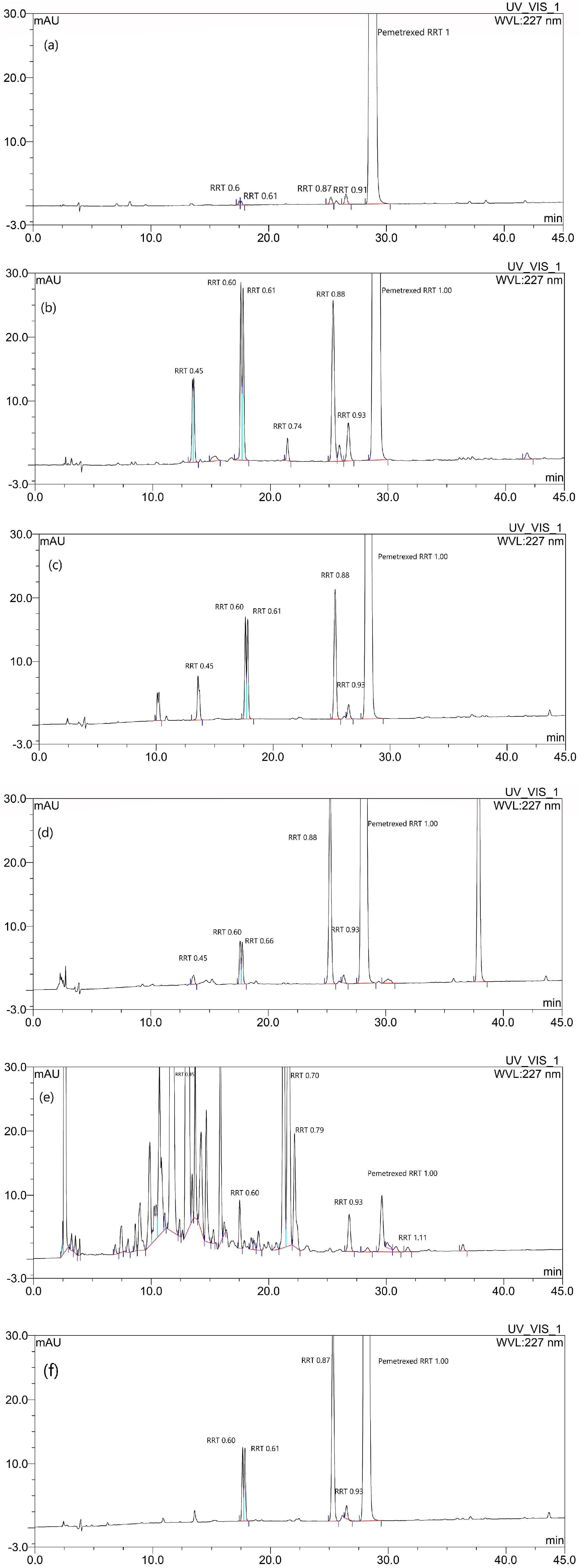
Forced degradation tests using various decomposition pathways were carried out as follows: Mannitol was dissolved in water for injection (WFI) at a concentration of 100 mg/mL. The drug stock solution was prepared by dissolving pemetrexed disodium hemipentahydrate in mannitol solution at a concentration of 100 mg/mL. Each test solution was prepared by following the method described below for each degradation condition and being filtered immediately through 0.2 µm Nalgene filter units (Nalgene, Rochester, NY, USA) for sterilization, followed by filling 20 mL of test solution in a glass vial and sealing with a cap. The vials containing test solution were stored at specific conditions, considering various decomposition mechanisms, and the samples were withdrawn at predetermined time intervals, and then they were diluted suitably and analyzed by HPLC. Each experiment was performed in quadruplicates.
2.4.1. Thermal Degradation
The pH of the stock solution was adjusted to 7.0 ± 0.1 by the addition of 0.1 N of NaOH, and the stock solution was then diluted to a final drug concentration of 25 mg/mL, using WFI. The prepared test sample vials were secondary packed, using a box to block the light, and then stored at 60 °C for 4 weeks.
2.4.2. Acidic Hydrolysis
The test solution was prepared by dilution of the stock solution to a final drug concentration of 25 mg/mL, using 0.01 N of HCl. The prepared test sample vials were secondary packed, using a box to block the light, and then stored at 25 °C for a day.
2.4.3. Basic Hydrolysis
The test solution was prepared by dilution of the stock solution to a final drug concentration of 25 mg/mL, using 1 N of NaOH. The prepared test sample vials were secondary packed, using a box to block the light, and then stored at 25 °C for a day.
2.4.4. Oxidation
A 6% solution of hydrogen peroxide, a strong oxidizing agent, in WFI, was added to the two times diluted stock solution, at a volume/volume ratio of 1:1. The final concentration of hydrogen peroxide and pemetrexed was 3% and 25 mg/mL, respectively. The prepared test sample vials were secondary packed, using a box to block the light, and then stored at 25 °C for a day.
2.4.5. Photostability
The test solution was prepared by dilution of the stock solution to a final drug concentration of 25 mg/mL, using WFI, and then the vials containing the test solution samples were stored in a photostability chamber equipped with one ultraviolet lamp with 22 W UV power, at 25 °C, for 1 day, in accordance with the Guidance for Industry Q1B Photostability Testing of New Drug Substances and Products by ICH [
28].
2.5. Effect of Control Strategy Parameters on Pemetrexed Stability
According to the control strategies recommended in the ICH guidelines [
21], the effects of (i) antioxidants and pH in formulation, (ii) solution oxygen levels in process parameters, and (iii) headspace oxygen levels in packaging parameters, on the stability of pemetrexed, were evaluated. The experimental methods for each category are described below in detail. After a certain storage period under each specific condition, samples were withdrawn, and the collected samples were analyzed by colorimeter. For quantification, the collected samples were filtered through 0.2 µm polyvinylidene fluoride (PVDF) syringe filter (Whatman GmbH, Dassel, Germany), and all filtered solution samples were diluted suitably and then analyzed by HPLC. All experiments were performed in quadruplicates.
2.5.1. Antioxidant as a Parameter for Control of Formulation
The stability of pemetrexed solution with or without antioxidants was evaluated at 60 °C for 3 weeks. The used antioxidants and their concentrations are presented in
Table 2.
The experimental methods and ordering are as follows: (i) mannitol was dissolved in WFI, followed by nitrogen purging, using high-tensile 316 stainless-steel tubing (HIP, Erie, PA, USA) and a gas-flow regulator (Chiyoda Seiki, Seoul, Korea) to decrease the oxygen levels to 1.0 ± 0.2 ppm in the solution. The DO levels in the solution were measured by using a portable Orion Star A326 DO meter (Thermo Scientific, Chelmsford, MA, USA); (ii) the antioxidants were dissolved in mannitol solution, and the pH was preadjusted to 5.0 ± 0.1, by addition of 0.5 N of HCl or 0.5 N of NaOH; (iii) pemetrexed was dissolved in the above solution and mixed; (iv) the pH was adjusted to 7.0 ± 0.1 by the addition of 0.5 N of HCl or 0.5 N of NaOH, (v) after pH equilibration, and the final test solutions were prepared by dilution to desired concentrations of the drug and antioxidants, using WFI, as presented in
Table 2. Each test solution was filtered through 0.2 µm Nalgene filter units (Nalgene, Rochester, NY, USA), for sterilization. Then, 20 mL of the filtered test solution was filled in the glass vials, followed by nitrogen headspace substitution by using high-tensile 316 stainless-steel tubing (HIP, Erie, PA, USA) and a gas-flow regulator (Chiyoda Seiki, Seoul, Korea), to reduce the headspace oxygen levels in the vial to 1.0 ± 0.2%. After the nitrogen headspace, the vial was sealed with a cap. The headspace oxygen content was determined by frequency-modulated spectroscopy (FMS-760, Lighthouse Instruments, Charlottesville, VA, USA). The sealed vials containing the test solution were stored at 60 °C for 3 weeks.
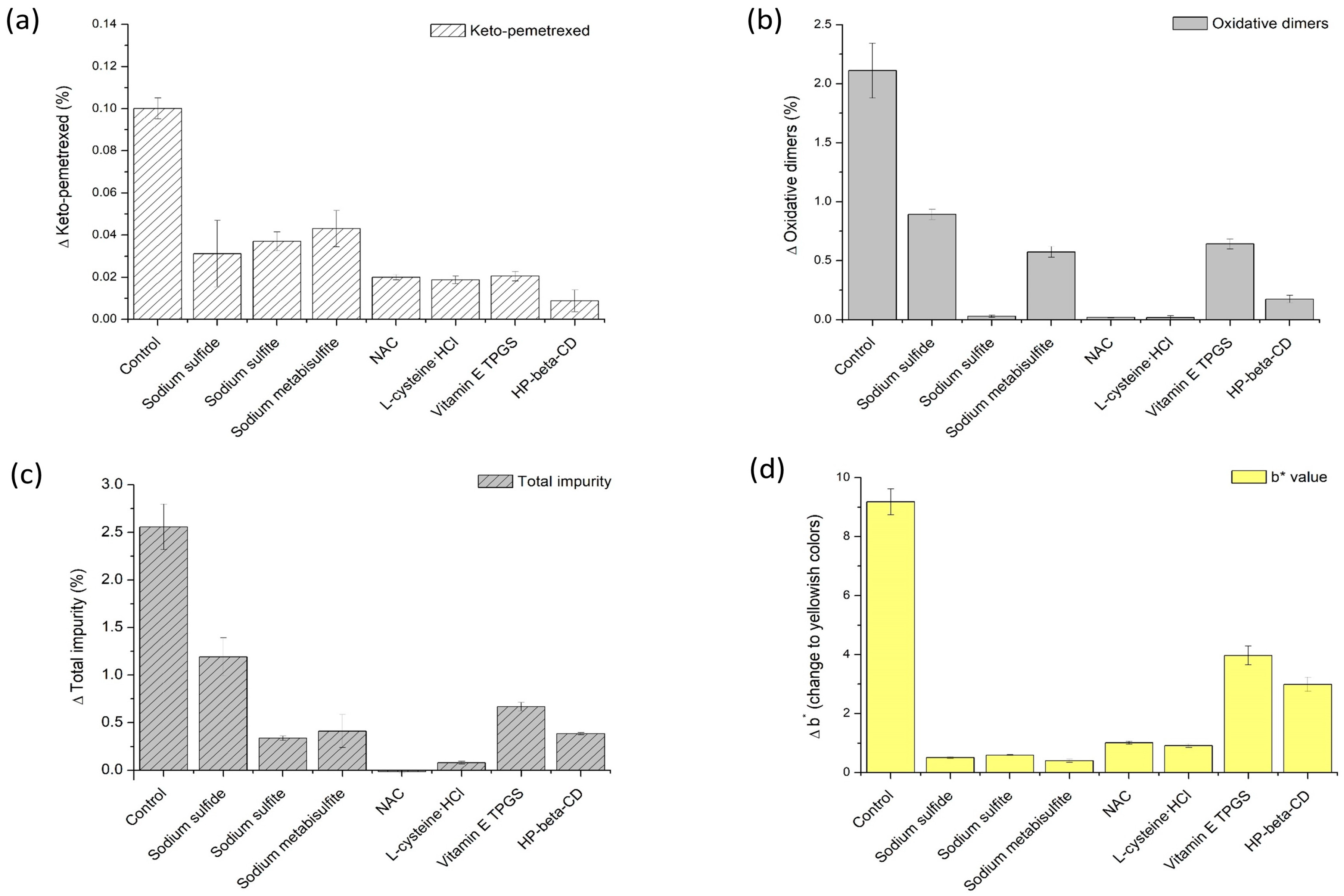
In order to evaluate the effects of various antioxidant types, sulfur, including oxidation-reducing agents, phenolic vitamin derivative, and inclusion complex forming agent were selected (
Table 2). Sodium sulfide was selected as sulfides; sodium sulfite and sodium metabisulfite were selected as sulfides; and NAC and
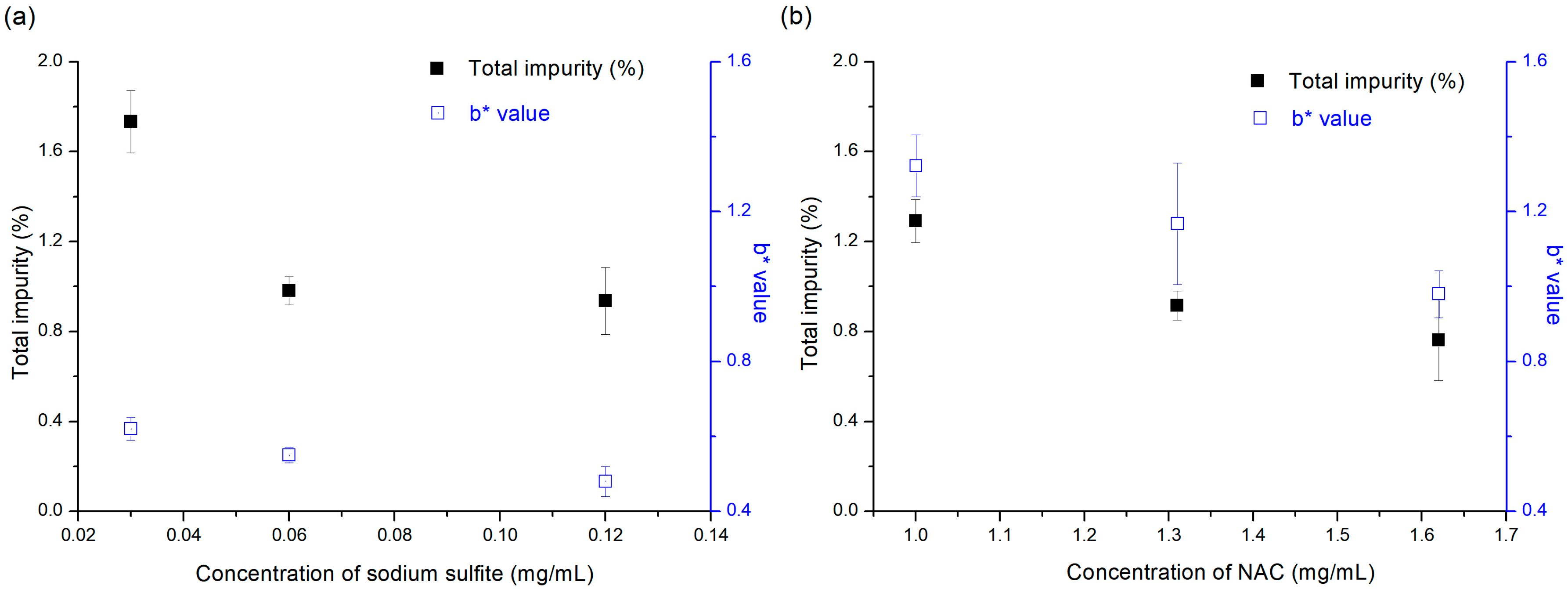
l-cysteine-HCl were selected as sulfur-containing amino acids. In addition, vitamin E TPGS and HP-β-CD were selected as phenolic vitamin derivative and inclusion-complex-forming agent, respectively. In order to evaluate the effect of antioxidant concentration on the stability of the pemetrexed solution, the final concentrations of sodium sulfite and NAC in the sample solution were varied from 0.03 to 0.12 mg/mL and from 1.00 to 1.63 mg/mL. These concentration ranges of various antioxidants were selected based on the clinically prescribed concentration for injection use under FDA approval [
30,
31,
32]. The vials containing the test solutions were stored at 60 °C for 3 weeks.
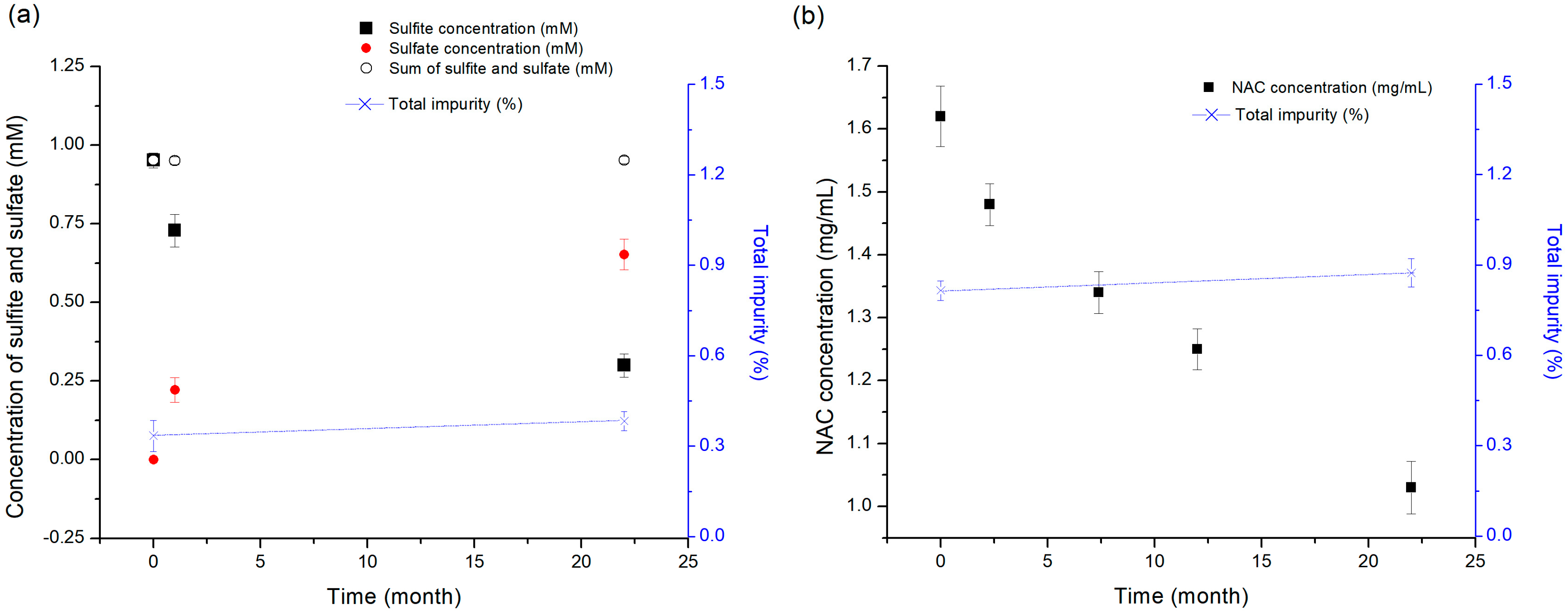
To evaluate the potential of sodium sulfite as a sacrificial reductant, a test solution with a sodium sulfite concentration of 0.12 mg/mL (76.2 mg/mL as a sulfite) was prepared, and then the vial containing the test solution was stored at 25 °C for 22 months, for long-term stability testing. The amounts of sulfite consumed by oxidation and the sulfate generated by oxidation were measured by high-performance ion chromatography (HPIC) [
33]. The test solution for NAC was prepared at a concentration of 1.63 mg/mL, and then the vial containing the test solution was stored at 25 °C for 22 months, for long-term stability testing. The NAC amount consumed by oxidation was determined by the RP-HPLC method. The initial antioxidant concentration before storage was compared to that measured after long-term stability testing. Then, the results were compared with the degradation pattern of pemetrexed and were further analyzed to determine the correlation between them.
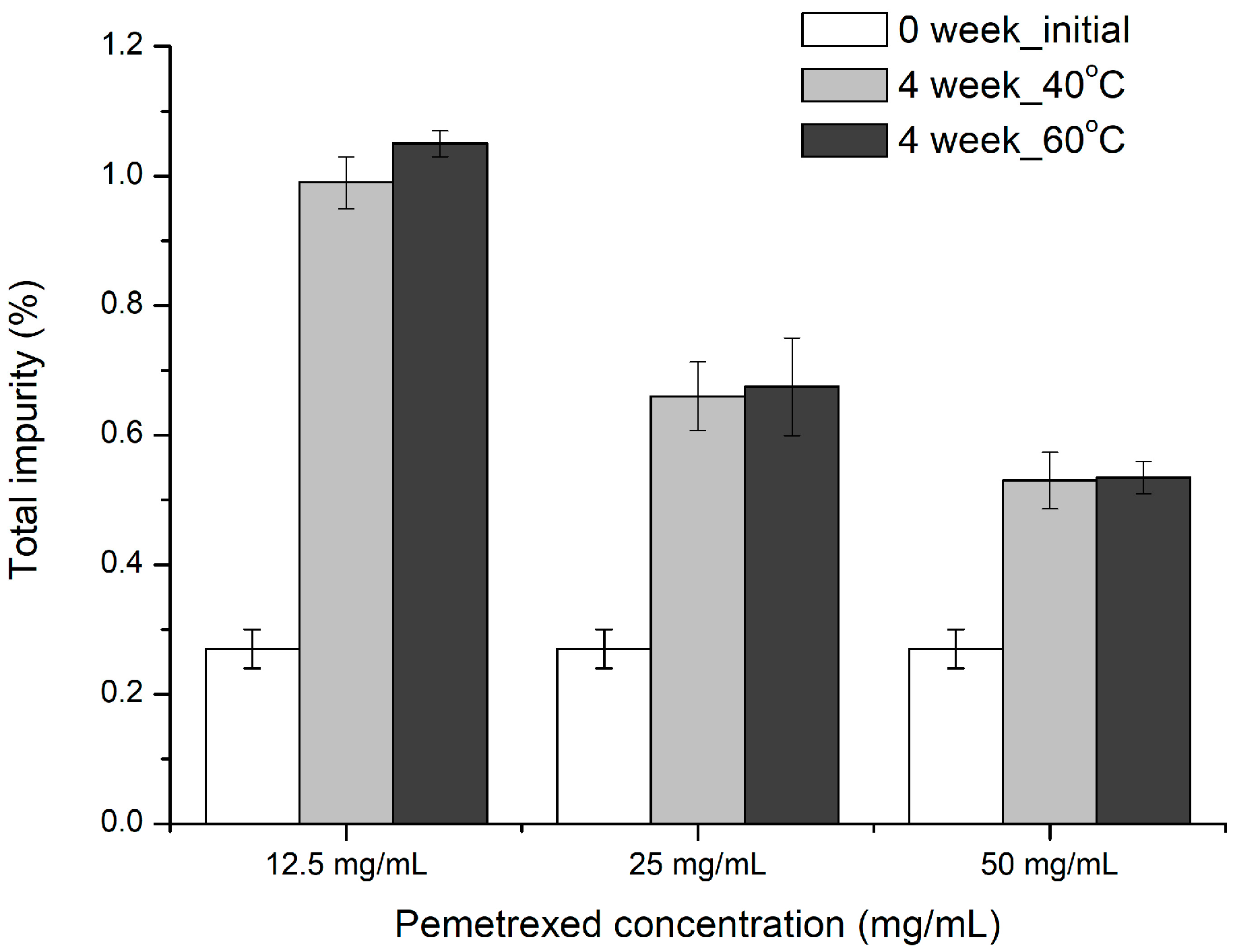
2.5.2. Drug Concentration as a Parameter for Control of Formulation
The effect of drug concentration on the stability of pemetrexed was evaluated by using two samples containing 0.06 mg/mL of sodium sulfite and 1.63 mg/mL of NAC as antioxidants. At pH 7, final drug concentrations of 50, 25, and 12.5 mg/mL were prepared by diluting with WFI (
Table 2). The prepared test sample vials were stored at 40 °C for 4 weeks, and at 60 °C for 4 weeks.
2.5.3. pH as a Parameter for Control of Formulation
The effect of pH on pemetrexed stability was evaluated to determine the optimal pH value for the stability of pemetrexed in aqueous solution. Considering the limited solubility of pemetrexed in acidic aqueous solvents, the evaluation was performed over the pH range of 6–8.5. The pH of the test solutions was adjusted to 6, 6.5, 7, 7.5, 8, and 8.5 in the pH adjustment step iv. The final concentrations of drug, sodium sulfite, and NAC were 50, 0.06, and 1.63 mg/mL, respectively (
Table 2). The prepared sample vials were stored at 40 °C for 4 weeks and at 60 °C for 3 weeks.
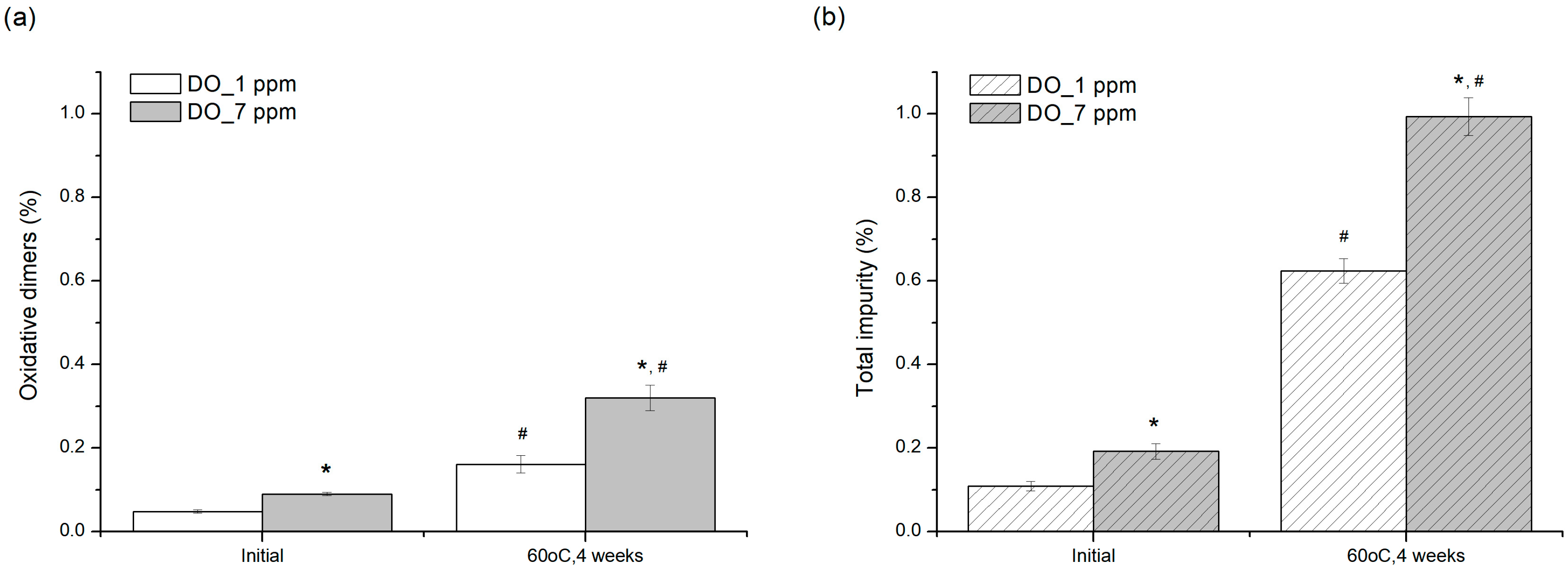
2.5.4. Dissolved Oxygen (DO) Level in Solution as a Parameter for Control of Process
In order to evaluate the effect of oxygen levels in solution on the stability of pemetrexed, two samples with different DO levels, 1 ± 0.2 ppm and 7 ± 0.2 ppm, were prepared by nitrogen purge according to the step (i) in the aforementioned method for evaluating antioxidant effect. The minimum DO that can be controlled accurately by the self-made nitrogen headspace substitution apparatus used in this study was 1 ± 0.2 ppm, so 1 ± 0.2 ppm was set as the lowest DO level. These samples contained 50 mg/mL of pemetrexed, 0.06 mg/mL of sodium sulfite and 1.63 mg/mL of NAC. The prepared sample vials were stored at 60 °C for 4 weeks.
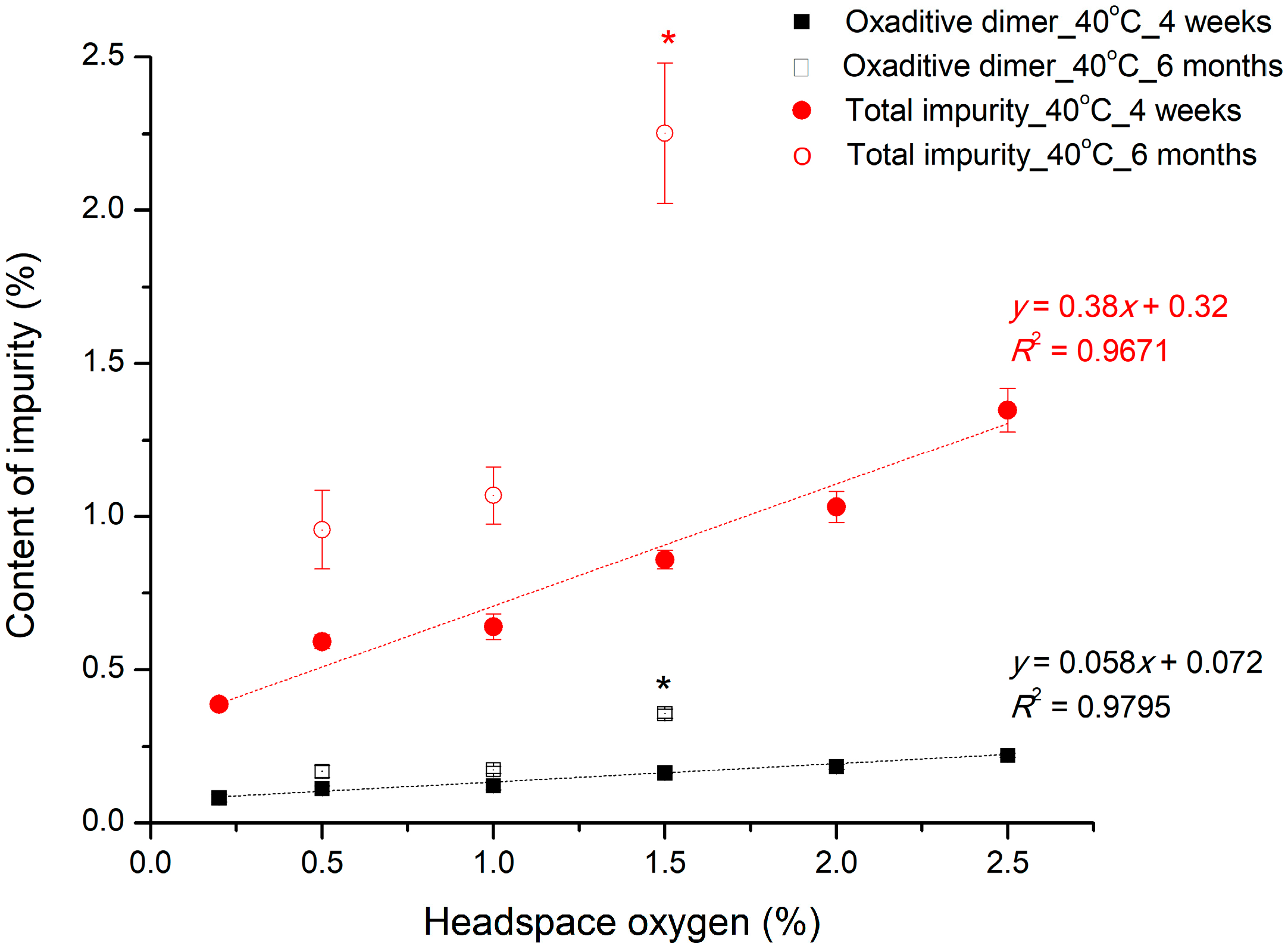
2.5.5. Nitrogen Headspace as a Parameter for Control of Packaging
The effect of oxygen levels in the headspace on the stability of pemetrexed was evaluated. The concentrations of the drug, sodium sulfite, and NAC in the test solution were 50, 0.06, and 1.63 mg/mL, respectively (
Table 2). The oxygen level in the headspace for the test samples varied from 0.2% to 2.5% by nitrogen headspace at step vi. The prepared sample vials were stored at 40 °C for 4 weeks.
2.6. Analysis Method of Sulfite and Sulfate
To evaluate the potential of sodium sulfite as the sacrificial reductant, the amount of sulfite consumed by oxidation and the sulfate generated by oxidation was measured and compared before and after the evaluation of
Section 2.5.1. In addition, the results were further compared with the degradation pattern of pemetrexed and analyzed to find a correlation between them. HPIC was used to simultaneously determine sulfite (SO
32−) and its oxidized form, sulfate (SO
42−) [
33]. The HPIC method was slightly modified from the previously described method. HPIC was performed with suppressed conductivity detection, using a Dionex Ultimate 3000 RS HPLC system (Dionex Corporation, Sunnyvale, CA, USA) equipped with an anion micro-membrane suppressor (AMMS, Dionex, Sunnyvale, CA, USA), a conductivity detector (CDM, Sunnyvale, CA, USA), and an anion-separation column (Shodex IC SI-90 4E, 4.0 × 250 mm, Showa Denko KK, Tokyo, Japan). The auto-sampler and column temperature were maintained at 4 and 30 °C, respectively. The injection volume was 10 µL, and the flow rate was maintained at 1.0 mL/min. Isocratic mobile phase was composed of 3 mM of NaHCO
3, 1.5 mM of Na
2CO
3, and 5% acetone in deionized water (DW). Sodium formaldehyde bisulfite and sodium sulfate were used as the standards for sulfite and sulfate, respectively, and the conversion amounts for sulfite and sulfate were calculated by multiplying molecular weight ratio after purity correction. The calibration curve of sulfite and sulfate concentration vs. area showed a good linearity over the concentration range of 18.1–362.0 µg/mL, with a correlation coefficient (R
2) of 1.00.
2.7. Analysis Method of NAC
RP-HPLC was performed to analyze the consumed amount of NAC by oxidation. The results were further compared with the degradation pattern of pemetrexed and analyzed to determine a correlation between them. RP-HPLC analysis was carried out on a Dionex Ultimate 3000 RS HPLC system (Dionex Corporation, Sunnyvale, CA, USA) equipped with an Ultimate 3000 RS diode array detector and RP C18 column (µBondapak C18, 125Å, 10 µm, 3.9 × 300 mm, Waters Ass., Milford, MA, USA). The auto-sampler and column temperature were maintained at 4 and 30 °C, respectively. The injection volume was 5 µL, and the flow rate was maintained at 1.5 mL/min. Two mobile phases were prepared for gradient elution (
Table 3). Mobile phase A was composed of 50 mM of KH
2PO
4, with pH 3.0. Mobile phase B was composed of acetonitrile. The elution program for the binary gradient system is presented in
Table 3. UV detection was performed at 214 nm [
34].
2.8. Statistical Analysis
To evaluate the statistical significance of the differences between groups, Student’s t-test or one-way analysis of variance (ANOVA), followed by Tukey’s HSD test and SNK test, was carried out by using SPSS 25.0 software (SPSS, Chicago, IL, USA).
4. Conclusions
In this study, we investigated the effects of various parameters at each control strategy in drug product degradation on the stability of pemetrexed in injectable aqueous solution. A forced degradation study confirmed that oxidation is the main mechanism responsible for the degradation of pemetrexed in aqueous solutions. As formulation controls, the effects of the addition of antioxidants at various concentrations, drug concentration, and pH were evaluated; it was found that these parameters significantly influence the stability of the pemetrexed solution. In addition, the DO level in the solution controlled by nitrogen purging and oxygen level in headspace controlled by nitrogen headspace also showed significant effects on the stability of the pemetrexed solution; thus, it was confirmed that the molecular oxygen is involved in the rate limiting oxidation step. Based on the results obtained above on various control strategies, the optimal injectable solution formulation of pemetrexed is suggested as follows: sodium sulfite at 0.06 mg/mL, as an antioxidant for prevention of color change; NAC at 1.63 mg/mL, as an antioxidant for prevention of chemical degradation; pH range of 7–8; DO levels below 1 ppm; and headspace oxygen level below 1%. Therefore, it can be concluded that this study, which included well-designed control strategies, can lead to a better understanding of the complex degradation mechanism of pemetrexed; thus, it could contribute to the development of an injectable solution formulation of pemetrexed, with improved stability, which in turn may increase the utilization and safety of this drug. Furthermore, the results of this study can be used to set a standard criterion for degradation products of pemetrexed in injectable solution. In further studies, the optimal selection of container/closure with respect to minimizing the oxygen ingress based on the moisture vapor transmission rate (MVTR) should also be investigated as a key factor, considering a balance between the cost and protection efficiency.