Norovirus Capsid Protein-Derived Nanoparticles and Polymers as Versatile Platforms for Antigen Presentation and Vaccine Development
Abstract
1. Introduction
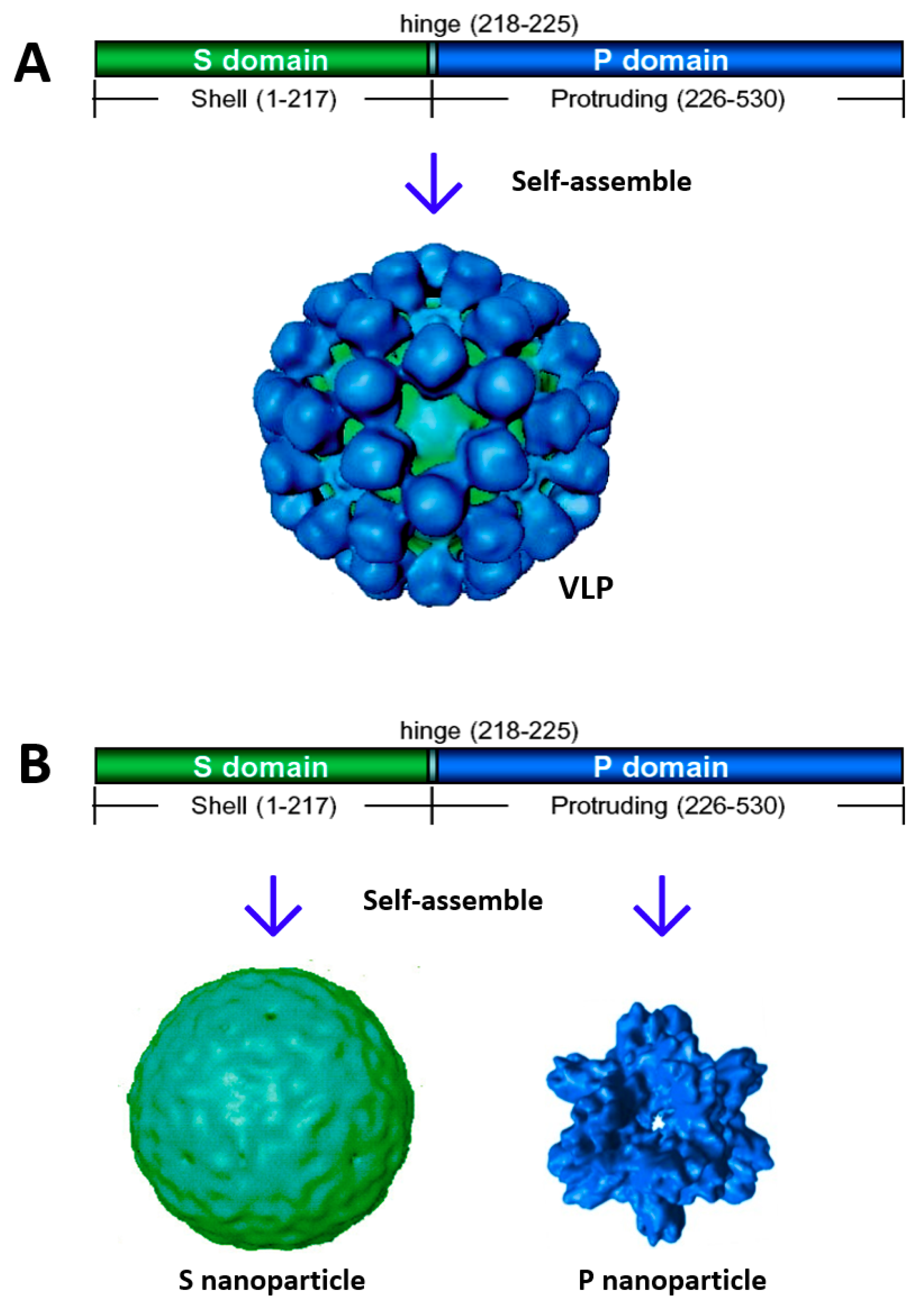
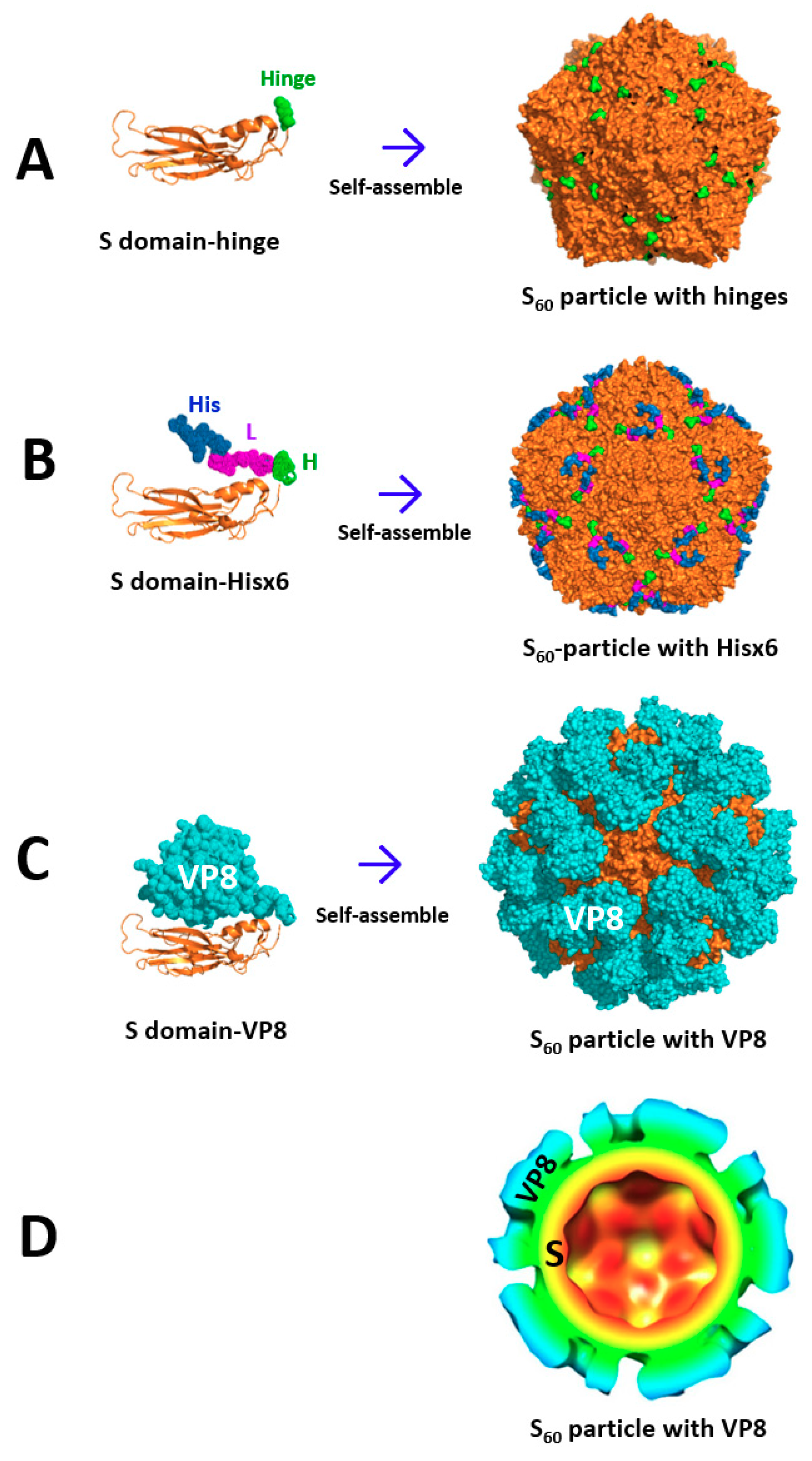
2. Creation of Norovirus S60 Nanoparticles
2.1. Potential of the S60 Nanoparticle as a Norovirus Vaccine Candidate
2.2. The S60 Nanoparticle as a Polyvalent Platform for Antigen Display for Improved Immunogenicity
2.3. Development of S60–VP8* Chimeric Nanoparticles as a Rotavirus Vaccine Candidate
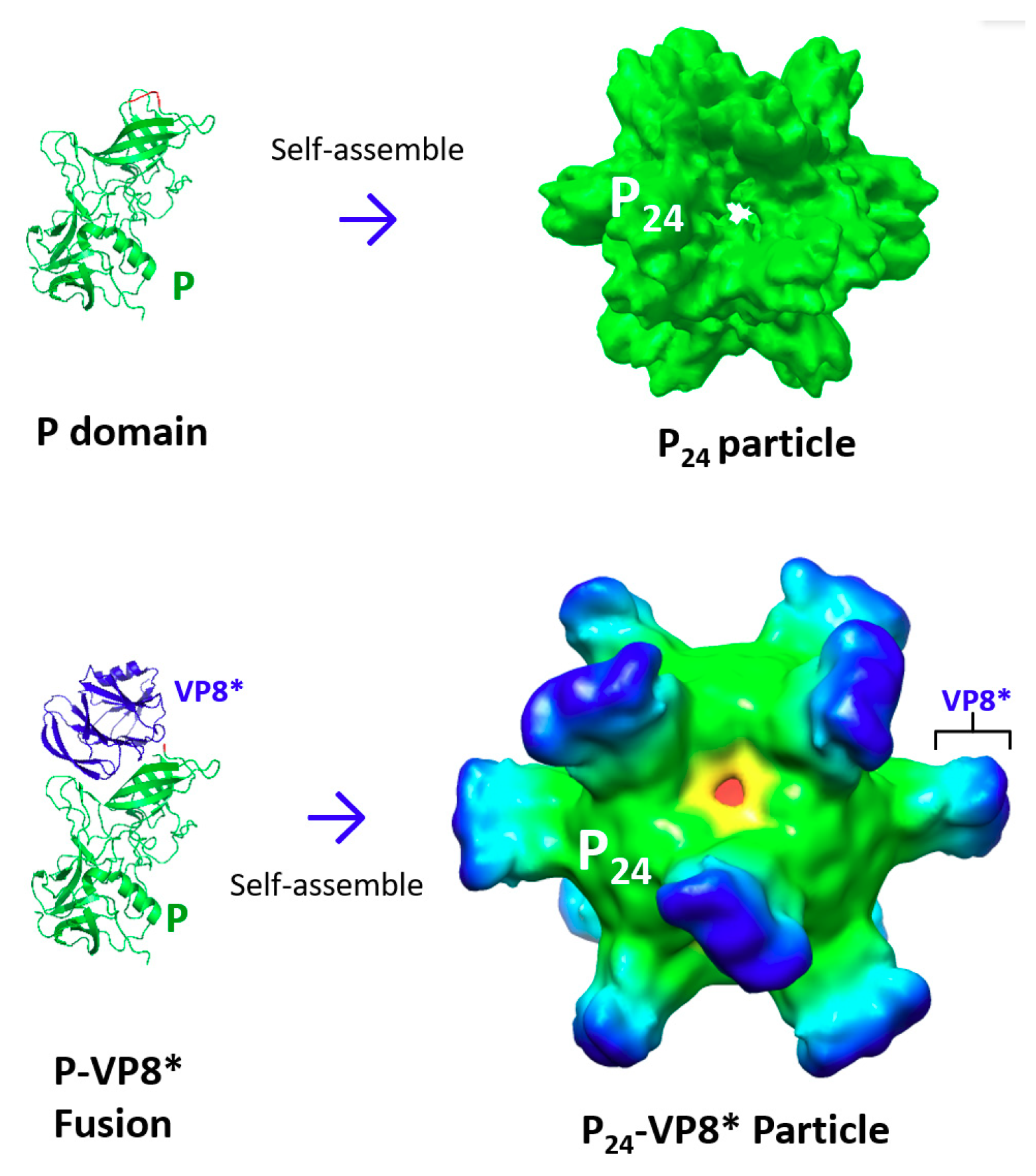
3. Creation of Norovirus P24 Nanoparticles
3.1. The P24 Nanoparticle as a Norovirus Vaccine Candidate
3.2. The P24 Nanoparticle as a Platform to Display Foreign Antigens for Vaccine Development
3.2.1. The P24 nanoparticle for Displaying Rotavirus VP8* Antigens
3.2.2. The P24 nanoparticle as a Nanoplatform to Display Epitopes of other Viral Pathogens
3.2.3. The P24 Nanoparticle as a Platform to Develop a Vaccine Against Noninfectious Disease
4. Norovirus P Domain-Derived Heterologous Polymers as Vaccines and Vaccine Platforms
5. Conclusions
Funding
Conflicts of Interest
References
- Green, K.; Chanock, R.; Kapikian, A. Human Calicivirus. In Fields Virology, 4th ed.; Knipe, D.M., Howley, P.M., Griffin, D.E., Lamb, R.A., Martin, M.A., Roizman, B., Straus, S.E., Eds.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2001; Volume 2, pp. 841–874. [Google Scholar]
- Aliabadi, N.; Lopman, B.A.; Parashar, U.D.; Hall, A.J. Progress toward norovirus vaccines: Considerations for further development and implementation in potential target populations. Expert Rev. vaccines 2015, 14, 1241–1253. [Google Scholar] [CrossRef] [PubMed]
- Kirk, M.D.; Pires, S.M.; Black, R.E.; Caipo, M.; Crump, J.A.; Devleesschauwer, B.; Dopfer, D.; Fazil, A.; Fischer-Walker, C.L.; Hald, T.; et al. World Health Organization Estimates of the Global and Regional Disease Burden of 22 Foodborne Bacterial, Protozoal, and Viral Diseases, 2010: A Data Synthesis. PLoS Med. 2015, 12, e1001921. [Google Scholar] [CrossRef]
- Pires, S.M.; Fischer-Walker, C.L.; Lanata, C.F.; Devleesschauwer, B.; Hall, A.J.; Kirk, M.D.; Duarte, A.S.; Black, R.E.; Angulo, F.J. Aetiology-Specific Estimates of the Global and Regional Incidence and Mortality of Diarrhoeal Diseases Commonly Transmitted through Food. PLoS ONE 2015, 10, e0142927. [Google Scholar] [CrossRef] [PubMed]
- Bartsch, S.M.; Lopman, B.A.; Ozawa, S.; Hall, A.J.; Lee, B.Y. Global Economic Burden of Norovirus Gastroenteritis. PLoS ONE 2016, 11, e0151219. [Google Scholar] [CrossRef] [PubMed]
- Lucero, Y.; Vidal, R.; O’Ryan, G.M. Norovirus vaccines under development. Vaccine 2018, 36, 5435–5441. [Google Scholar] [CrossRef] [PubMed]
- Conley, M.J.; McElwee, M.; Azmi, L.; Gabrielsen, M.; Byron, O.; Goodfellow, I.G.; Bhella, D. Calicivirus VP2 forms a portal-like assembly following receptor engagement. Nature 2019, 565, 377–381. [Google Scholar] [CrossRef] [PubMed]
- Jung, J.; Grant, T.; Thomas, D.R.; Diehnelt, C.W.; Grigorieff, N.; Joshua-Tor, L. High-resolution cryo-EM structures of outbreak strain human norovirus shells reveal size variations. Proc. Natl. Acad. Sci. United States Am. 2019, 116, 12828–12832. [Google Scholar] [CrossRef] [PubMed]
- Devant, J.M.; Hofhaus, G.; Bhella, D.; Hansman, G.S. Heterologous expression of human norovirus GII.4 VP1 leads to assembly of T = 4 virus-like particles. Antivir. Res. 2019, 168, 175–182. [Google Scholar] [CrossRef] [PubMed]
- Prasad, B.V.; Hardy, M.E.; Dokland, T.; Bella, J.; Rossmann, M.G.; Estes, M.K. X-ray crystallographic structure of the Norwalk virus capsid. Science 1999, 286, 287–290. [Google Scholar] [CrossRef] [PubMed]
- Tan, M.; Jiang, X. Histo-blood group antigens: A common niche for norovirus and rotavirus. Expert Rev. Mol. Med. 2014, 16, e5. [Google Scholar] [CrossRef]
- Tan, M.; Jiang, X. Norovirus-host interaction: Multi-selections by human histo-blood group antigens. Trends Microbiol. 2011, 19, 382–388. [Google Scholar] [CrossRef] [PubMed]
- Tan, M.; Jiang, X. Norovirus gastroenteritis, carbohydrate receptors, and animal models. PLoS Pathog. 2010, 6, e1000983. [Google Scholar] [CrossRef] [PubMed]
- Tan, M.; Jiang, X. Norovirus-host interaction: Implications for disease control and prevention. Expert Rev. Mol. Med. 2007, 9, 1–22. [Google Scholar] [CrossRef]
- Tan, M.; Jiang, X. Norovirus and its histo-blood group antigen receptors: An answer to a historical puzzle. Trends Microbiol. 2005, 13, 285–293. [Google Scholar] [CrossRef] [PubMed]
- Baclayon, M.; Shoemaker, G.K.; Uetrecht, C.; Crawford, S.E.; Estes, M.K.; Prasad, B.V.; Heck, A.J.; Wuite, G.J.; Roos, W.H. Prestress strengthens the shell of Norwalk virus nanoparticles. Nano. Lett. 2011, 11, 4865–4869. [Google Scholar] [CrossRef]
- Tan, M.; Jiang, X. Nanoparticles of Norovirus. In Viral Nanptechnology; Khudyakov, Y., Pumpens, P., Eds.; CRC Press, Taylor Francis Group: Norwich, UK, 2015; pp. 363–371. [Google Scholar]
- Tan, M.; Jiang, X. Norovirus P particle: A subviral nanoparticle for vaccine development against norovirus, rotavirus and influenza virus. Nanomedicine 2012, 7, 889–897. [Google Scholar] [CrossRef]
- Tan, M.; Jiang, X. Recent advancements in combination subunit vaccine development. Hum. Vaccin. Immunother. 2017, 13, 180–185. [Google Scholar] [CrossRef]
- Tan, M.; Jiang, X. Subviral particle as vaccine and vaccine platform. Curr. Opin. Virol. 2014, 6, 24–33. [Google Scholar] [CrossRef]
- Zhao, Q.; Li, S.; Yu, H.; Xia, N.; Modis, Y. Virus-like particle-based human vaccines: Quality assessment based on structural and functional properties. Trends Biotechnol. 2013, 31, 654–663. [Google Scholar] [CrossRef]
- Jiang, X.; Wang, M.; Graham, D.Y.; Estes, M.K. Expression, self-assembly, and antigenicity of the Norwalk virus capsid protein. J. Virol. 1992, 66, 6527–6532. [Google Scholar]
- Bertolotti-Ciarlet, A.; White, L.J.; Chen, R.; Prasad, B.V.; Estes, M.K. Structural requirements for the assembly of Norwalk virus-like particles. J. virol. 2002, 76, 4044–4055. [Google Scholar] [CrossRef] [PubMed]
- Tan, M.; Hegde, R.S.; Jiang, X. The P domain of norovirus capsid protein forms dimer and binds to histo-blood group antigen receptors. J. virol. 2004, 78, 6233–6242. [Google Scholar] [CrossRef] [PubMed]
- Xia, M.; Huang, P.; Sun, C.; Han, L.; Vago, F.S.; Li, K.; Zhong, W.; Jiang, W.; Klassen, J.S.; Jiang, X.; et al. Bioengineered Norovirus S60 Nanoparticles as a Multifunctional Vaccine Platform. ACS Nano. 2018, 12, 10665–10682. [Google Scholar] [CrossRef] [PubMed]
- Burmeister, W.P.; Buisson, M.; Estrozi, L.F.; Schoehn, G.; Billet, O.; Hannas, Z.; Sigoillot, C.; Poulet, H. Structure determination of feline calicivirus virus-like particles in the context of a pseudo-octahedral arrangement. PLoS ONE 2015, 10, e0119289. [Google Scholar] [CrossRef] [PubMed]
- Tate, J.E.; Burton, A.H.; Boschi-Pinto, C.; Steele, A.D.; Duque, J.; Parashar, U.D.; Network, W.H.-c.G.R.S. 2008 estimate of worldwide rotavirus-associated mortality in children younger than 5 years before the introduction of universal rotavirus vaccination programmes: A systematic review and meta-analysis. Lancet Infect. Dis. 2012, 12, 136–141. [Google Scholar] [CrossRef]
- Parashar, U.D.; Gibson, C.J.; Bresse, J.S.; Glass, R.I. Rotavirus and severe childhood diarrhea. Emerg. Infect. Dis. 2006, 12, 304–306. [Google Scholar] [CrossRef] [PubMed]
- Walker, C.L.; Rudan, I.; Liu, L.; Nair, H.; Theodoratou, E.; Bhutta, Z.A.; O’Brien, K.L.; Campbell, H.; Black, R.E. Global burden of childhood pneumonia and diarrhoea. Lancet 2013, 381, 1405–1416. [Google Scholar] [CrossRef]
- Feng, N.; Hu, L.; Ding, S.; Sanyal, M.; Zhao, B.; Sankaran, B.; Ramani, S.; McNeal, M.; Yasukawa, L.L.; Song, Y.; et al. Human VP8* mAbs neutralize rotavirus selectively in human intestinal epithelial cells. J. Clin. Invest. 2019, 130. [Google Scholar] [CrossRef] [PubMed]
- Nair, N.; Feng, N.; Blum, L.K.; Sanyal, M.; Ding, S.; Jiang, B.; Sen, A.; Morton, J.M.; He, X.S.; Robinson, W.H.; et al. VP4- and VP7-specific antibodies mediate heterotypic immunity to rotavirus in humans. Sci. Transl. Med. 2017, 9. [Google Scholar] [CrossRef] [PubMed]
- Blanchard, H.; Yu, X.; Coulson, B.S.; von Itzstein, M. Insight into host cell carbohydrate-recognition by human and porcine rotavirus from crystal structures of the virion spike associated carbohydrate-binding domain (VP8*). J. Mol. Biol. 2007, 367, 1215–1226. [Google Scholar] [CrossRef] [PubMed]
- Xia, M.; Huang, P.; Jiang, X.; Tan, M. Immune response and protective efficacy of the S particle presented rotavirus VP8* vaccine in mice. Vaccine 2019, 37, 4103–4110. [Google Scholar] [CrossRef] [PubMed]
- Cao, S.; Lou, Z.; Tan, M.; Chen, Y.; Liu, Y.; Zhang, Z.; Zhang, X.C.; Jiang, X.; Li, X.; Rao, Z. Structural basis for the recognition of blood group trisaccharides by norovirus. J. virol. 2007, 81, 5949–5957. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Tan, M.; Xia, M.; Hao, N.; Zhang, X.C.; Huang, P.; Jiang, X.; Li, X.; Rao, Z. Crystallography of a Lewis-binding norovirus, elucidation of strain-specificity to the polymorphic human histo-blood group antigens. PLoS Pathog. 2011, 7, e1002152. [Google Scholar] [CrossRef] [PubMed]
- Tan, M.; Jiang, X. The p domain of norovirus capsid protein forms a subviral particle that binds to histo-blood group antigen receptors. J. virol. 2005, 79, 14017–14030. [Google Scholar] [CrossRef] [PubMed]
- Tan, M.; Fang, P.; Chachiyo, T.; Xia, M.; Huang, P.; Fang, Z.; Jiang, W.; Jiang, X. Noroviral P particle: Structure, function and applications in virus-host interaction. Virology 2008, 382, 115–123. [Google Scholar] [CrossRef] [PubMed]
- Bereszczak, J.Z.; Barbu, I.M.; Tan, M.; Xia, M.; Jiang, X.; van Duijn, E.; Heck, A.J. Structure, stability and dynamics of norovirus P domain derived protein complexes studied by native mass spectrometry. J. Struct. Biol. 2012, 177, 273–282. [Google Scholar] [CrossRef] [PubMed]
- Fang, H.; Tan, M.; Xia, M.; Wang, L.; Jiang, X. Norovirus P particle efficiently elicits innate, humoral and cellular immunity. PLoS ONE 2013, 8, e63269. [Google Scholar] [CrossRef] [PubMed]
- Tan, M.; Fang, P.A.; Xia, M.; Chachiyo, T.; Jiang, W.; Jiang, X. Terminal modifications of norovirus P domain resulted in a new type of subviral particles, the small P particles. Virology 2011, 410, 345–352. [Google Scholar] [CrossRef] [PubMed]
- Kocher, J.; Bui, T.; Giri-Rachman, E.; Wen, K.; Li, G.; Yang, X.; Liu, F.; Tan, M.; Xia, M.; Zhong, W.; et al. Intranasal P particle vaccine provided partial cross-variant protection against human GII.4 norovirus diarrhea in gnotobiotic pigs. J. Virol. 2014, 88, 9728–9743. [Google Scholar] [CrossRef] [PubMed]
- Tan, M.; Xia, M.; Huang, P.; Wang, L.; Zhong, W.; McNeal, M.; Wei, C.; Jiang, X. Norovirus P Particle as a Platform for Antigen Presentation. Procedia. Vaccinol. 2011, 4, 19–26. [Google Scholar] [CrossRef]
- Tan, M.; Huang, P.; Xia, M.; Fang, P.A.; Zhong, W.; McNeal, M.; Wei, C.; Jiang, W.; Jiang, X. Norovirus P particle, a novel platform for vaccine development and antibody production. J. virol. 2011, 85, 753–764. [Google Scholar] [CrossRef] [PubMed]
- Huang, P.; Xia, M.; Tan, M.; Zhong, W.; Wei, C.; Wang, L.; Morrow, A.; Jiang, X. Spike protein VP8* of human rotavirus recognizes histo-blood group antigens in a type-specific manner. J. virol. 2012, 86, 4833–4843. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Huang, P.; Tan, M.; Liu, Y.; Biesiada, J.; Meller, J.; Castello, A.A.; Jiang, B.; Jiang, X. Rotavirus VP8*: Phylogeny, host range, and interaction with histo-blood group antigens. J. virol. 2012, 86, 9899–9910. [Google Scholar] [CrossRef] [PubMed]
- Reeck, A.; Kavanagh, O.; Estes, M.K.; Opekun, A.R.; Gilger, M.A.; Graham, D.Y.; Atmar, R.L. Serological correlate of protection against norovirus-induced gastroenteritis. J. Infect. Dis. 2010, 202, 1212–1218. [Google Scholar] [CrossRef] [PubMed]
- Atmar, R.L.; Bernstein, D.I.; Lyon, G.M.; Treanor, J.J.; Al-Ibrahim, M.S.; Graham, D.Y.; Vinje, J.; Jiang, X.; Gregoricus, N.; Frenck, R.W.; et al. Serological Correlates of Protection against a GII.4 Norovirus. Clin. vaccine Immunol. CVI 2015, 22. [Google Scholar] [CrossRef] [PubMed]
- Duizer, E.; Schwab, K.J.; Neill, F.H.; Atmar, R.L.; Koopmans, M.P.; Estes, M.K. Laboratory efforts to cultivate noroviruses. J. Gen. Virol. 2004, 85, 79–87. [Google Scholar] [CrossRef] [PubMed]
- Dai, Y.C.; Zhang, X.F.; Tan, M.; Huang, P.; Lei, W.; Fang, H.; Zhong, W.; Jiang, X. A dual chicken IgY against rotavirus and norovirus. Antivir. Res. 2012. [Google Scholar] [CrossRef] [PubMed]
- Dai, Y.C.; Wang, Y.Y.; Zhang, X.F.; Tan, M.; Xia, M.; Wu, X.B.; Jiang, X.; Nie, J. Evaluation of anti-norovirus IgY from egg yolk of chickens immunized with norovirus P particles. J. Virol. methods 2012, 186, 126–131. [Google Scholar] [CrossRef]
- Xia, M.; Tan, M.; Wei, C.; Zhong, W.; Wang, L.; McNeal, M.; Jiang, X. A candidate dual vaccine against influenza and noroviruses. Vaccine 2011, 29, 7670–7677. [Google Scholar] [CrossRef]
- Gong, X.; Yin, H.; Shi, Y.; He, X.; Yu, Y.; Guan, S.; Kuai, Z.; Haji, N.M.; Haji, N.M.; Kong, W.; et al. Evaluation of the immunogenicity and protective effects of a trivalent chimeric norovirus P particle immunogen displaying influenza HA2 from subtypes H1, H3 and B. Emerg. Microbes Infect. 2016, 5, e51. [Google Scholar] [CrossRef]
- Jiang, L.; Fan, R.; Sun, S.; Fan, P.; Su, W.; Zhou, Y.; Gao, F.; Xu, F.; Kong, W.; Jiang, C. A new EV71 VP3 epitope in norovirus P particle vector displays neutralizing activity and protection in vivo in mice. Vaccine 2015, 33, 6596–6603. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Fu, L.; Shi, Y.; Guan, S.; Yang, L.; Gong, X.; Yin, H.; He, X.; Liu, D.; Kuai, Z.; et al. Elicitation of HIV-1 neutralizing antibodies by presentation of 4E10 and 10E8 epitopes on Norovirus P particles. Immunol. Lett. 2015, 168, 271–278. [Google Scholar] [CrossRef] [PubMed]
- Fu, L.; Li, Y.; Hu, Y.; Zheng, Y.; Yu, B.; Zhang, H.; Wu, J.; Wu, H.; Yu, X.; Kong, W. Norovirus P particle-based active Abeta immunotherapy elicits sufficient immunogenicity and improves cognitive capacity in a mouse model of Alzheimer’s disease. Sci. Rep. 2017, 7, 41041. [Google Scholar] [CrossRef] [PubMed]
- Fu, L.; Li, Y.; Hu, Y.; Yu, B.; Zhang, H.; Wu, J.; Wu, H.; Yu, X.; Kong, W. Norovirus P particle: An excellent vaccine platform for antibody production against Alzheimer’s disease. Immunol. Lett. 2015, 168, 22–30. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Xia, M.; Huang, P.; Fang, H.; Cao, D.; Meng, X.J.; McNeal, M.; Jiang, X.; Tan, M. Branched-linear and agglomerate protein polymers as vaccine platforms. Biomaterials 2014, 35, 8427–8438. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Huang, P.; Fang, H.; Xia, M.; Zhong, W.; McNeal, M.M.; Jiang, X.; Tan, M. Polyvalent complexes for vaccine development. Biomaterials 2013, 34, 4480–4492. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wang, L.; Cao, D.; Wei, C.; Meng, X.J.; Jiang, X.; Tan, M. A dual vaccine candidate against norovirus and hepatitis E virus. Vaccine 2014, 32, 445–452. [Google Scholar] [CrossRef] [PubMed]
- Xia, M.; Wei, C.; Wang, L.; Cao, D.; Meng, X.J.; Jiang, X.; Tan, M. A trivalent vaccine candidate against hepatitis E virus, norovirus, and astrovirus. Vaccine 2016, 34, 905–913. [Google Scholar] [CrossRef] [PubMed]
- Xia, M.; Wei, C.; Wang, L.; Cao, D.; Meng, X.J.; Jiang, X.; Tan, M. Development and evaluation of two subunit vaccine candidates containing antigens of hepatitis E virus, rotavirus, and astrovirus. Sci. Rep. 2016, 6, 25735. [Google Scholar] [CrossRef]
- World Health Organization: Factsheets of Seasonal Influenza. 2016. Available online: http://www.who.int/mediacentre/factsheets/fs211/en/ (accessed on 10 September 2019).
- Pinto, L.H.; Lamb, R.A. Controlling influenza virus replication by inhibiting its proton channel. Mol. Biosyst. 2007, 3, 18–23. [Google Scholar] [CrossRef]
- Du, L.; Zhou, Y.; Jiang, S. Research and development of universal influenza vaccines. Microbes Infect. 2010, 12, 280–286. [Google Scholar] [CrossRef] [PubMed]
- Qiu, J. Enterovirus 71 infection: A new threat to global public health? Lancet Neurol. 2008, 7, 868–869. [Google Scholar] [CrossRef]
- Arthur Huang, K.Y.; Chen, M.F.; Huang, Y.C.; Shih, S.R.; Chiu, C.H.; Lin, J.J.; Wang, J.R.; Tsao, K.C.; Lin, T.Y. Epitope-associated and specificity-focused features of EV71-neutralizing antibody repertoires from plasmablasts of infected children. Nat. Commun. 2017, 8, 762. [Google Scholar] [CrossRef] [PubMed]
- Douek, D.C.; Roederer, M.; Koup, R.A. Emerging concepts in the immunopathogenesis of AIDS. Annu. Rev. Med. 2009, 60, 471–484. [Google Scholar] [CrossRef] [PubMed]
- Weiss, R.A. How Does Hiv Cause Aids. Science 1993, 260, 1273–1279. [Google Scholar] [CrossRef] [PubMed]
- Apostolova, L.G. Alzheimer Disease. Continuum (Minneap Minn) 2016, 22, 419–434. [Google Scholar] [CrossRef] [PubMed]
- Dickson, D.W. The pathogenesis of senile plaques. J. Neuropathol. Exp. Neurol. 1997, 56, 321–339. [Google Scholar] [CrossRef] [PubMed]
- Hardy, J.; Selkoe, D.J. Medicine - The amyloid hypothesis of Alzheimer’s disease: Progress and problems on the road to therapeutics. Science 2002, 297, 353–356. [Google Scholar] [CrossRef] [PubMed]
- Panza, F.; Lozupone, M.; Watling, M.; Imbimbo, B.P. Do BACE inhibitor failures in Alzheimer patients challenge the amyloid hypothesis of the disease? Expert Rev. Neurother. 2019, 19, 599–602. [Google Scholar] [CrossRef] [PubMed]
- Mullard, A. Alzheimer amyloid hypothesis lives on. Nat. Rev. Drug Discov. 2016, 16, 3–5. [Google Scholar] [CrossRef] [PubMed]
- Karran, E.; Hardy, J. A critique of the drug discovery and phase 3 clinical programs targeting the amyloid hypothesis for Alzheimer disease. Ann. Neurol. 2014, 76, 185–205. [Google Scholar] [CrossRef] [PubMed]
- Glass, R.I.; Noel, J.; Mitchell, D.; Herrmann, J.E.; Blacklow, N.R.; Pickering, L.K.; Dennehy, P.; Ruiz-Palacios, G.; de Guerrero, M.L.; Monroe, S.S. The changing epidemiology of astrovirus-associated gastroenteritis: A review. Arch. Virol. Suppl. 1996, 12, 287–300. [Google Scholar] [PubMed]
- Lozano, R.; Naghavi, M.; Foreman, K.; Lim, S.; Shibuya, K.; Aboyans, V.; Abraham, J.; Adair, T.; Aggarwal, R.; Ahn, S.Y.; et al. Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: A systematic analysis for the Global Burden of Disease Study 2010. Lancet 2012, 380, 2095–2128. [Google Scholar] [CrossRef]
- DuBois, R.M.; Freiden, P.; Marvin, S.; Reddivari, M.; Heath, R.J.; White, S.W.; Schultz-Cherry, S. Crystal structure of the avian astrovirus capsid spike. J. Virol. 2013, 87, 7853–7863. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Tang, X.; Seetharaman, J.; Yang, C.; Gu, Y.; Zhang, J.; Du, H.; Shih, J.W.; Hew, C.L.; Sivaraman, J.; et al. Dimerization of hepatitis E virus capsid protein E2s domain is essential for virus-host interaction. PLoS Pathog. 2009, 5, e1000537. [Google Scholar] [CrossRef] [PubMed]
- Hu, L.; Crawford, S.E.; Czako, R.; Cortes-Penfield, N.W.; Smith, D.F.; Le Pendu, J.; Estes, M.K.; Prasad, B.V. Cell attachment protein VP8* of a human rotavirus specifically interacts with A-type histo-blood group antigen. Nature 2012, 485, 256–259. [Google Scholar] [CrossRef] [PubMed]
| Nanoparticle/Polymer | Antigen/Epitope to be Displayed (Pathogen) | Chimeric Products as Vaccine Candidate | Immunity against Pathogens or Diseases | Reference |
|---|---|---|---|---|
| S60 | VP8* (rotavirus) | S60–VP8* | Rotavirus | [25,33] |
| P24 | P domain (norovirus) | P24 | Norovirus | [39,41] |
| P24 | VP8* (rotavirus) | P24–VP8* | Rotavirus and norovirus | [43] |
| P24 | M2e (influenza virus) | P24–M2e | Influenza virus | [51] |
| P24 | HA2 B cell epitope (influenza virus) | Trivalent HA2-PP (P24-HA2:90-105) | Influenza A virus and influenza B virus | [52] |
| P24 | VP3 B cell epitope (EV71) | PP-71-6 (P24-71-6) | EV71 | [53] |
| P24 | 4E10/10E8 epitopes (HIV-1) | 4E10-PP/10E8-PP | HIV-1 | [54] |
| P24 | Amyloid-beta, Aβ | PP-3copy-Aβ1-6 | Alzheimer’s disease | [55,56] |
| P polymer | P domains (noroviruses) | NoV PGI-NoV PGII GST NoV P+ | Different noroviruses | [57,58] |
| P polymer | P domain (HEV) | NoV P-HEV P | Norovirus and HEV | [59] |
| P polymer | P domain (astrovirus) P domain (HEV) | Ast P-HEV P-NoV P | Norovirus, astrovirus, and HEV | [60] |
| P polymer | P domain (astrovirus) P domain (HEV) VP8* (rotavirus) | Ast P-HEV P-VP8* | Rotavirus, astrovirus, and HEV | [61] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tan, M.; Jiang, X. Norovirus Capsid Protein-Derived Nanoparticles and Polymers as Versatile Platforms for Antigen Presentation and Vaccine Development. Pharmaceutics 2019, 11, 472. https://doi.org/10.3390/pharmaceutics11090472
Tan M, Jiang X. Norovirus Capsid Protein-Derived Nanoparticles and Polymers as Versatile Platforms for Antigen Presentation and Vaccine Development. Pharmaceutics. 2019; 11(9):472. https://doi.org/10.3390/pharmaceutics11090472
Chicago/Turabian StyleTan, Ming, and Xi Jiang. 2019. "Norovirus Capsid Protein-Derived Nanoparticles and Polymers as Versatile Platforms for Antigen Presentation and Vaccine Development" Pharmaceutics 11, no. 9: 472. https://doi.org/10.3390/pharmaceutics11090472
APA StyleTan, M., & Jiang, X. (2019). Norovirus Capsid Protein-Derived Nanoparticles and Polymers as Versatile Platforms for Antigen Presentation and Vaccine Development. Pharmaceutics, 11(9), 472. https://doi.org/10.3390/pharmaceutics11090472





