Ionic Polymethacrylate Based Delivery Systems: Effect of Carrier Topology and Drug Loading
Abstract
1. Introduction
2. Non-Linear Polymers Containing Ionic Groups in Drug Delivery
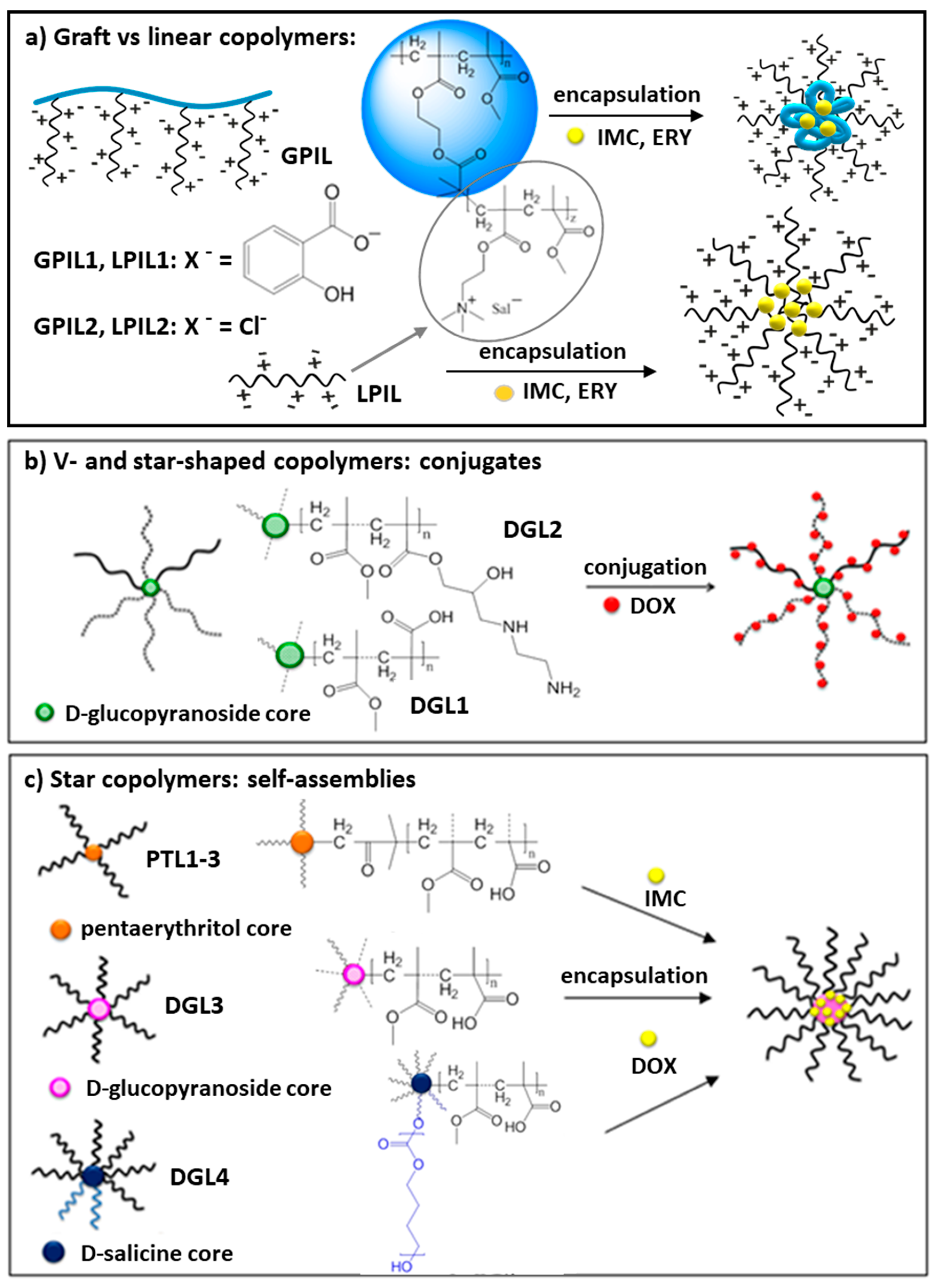
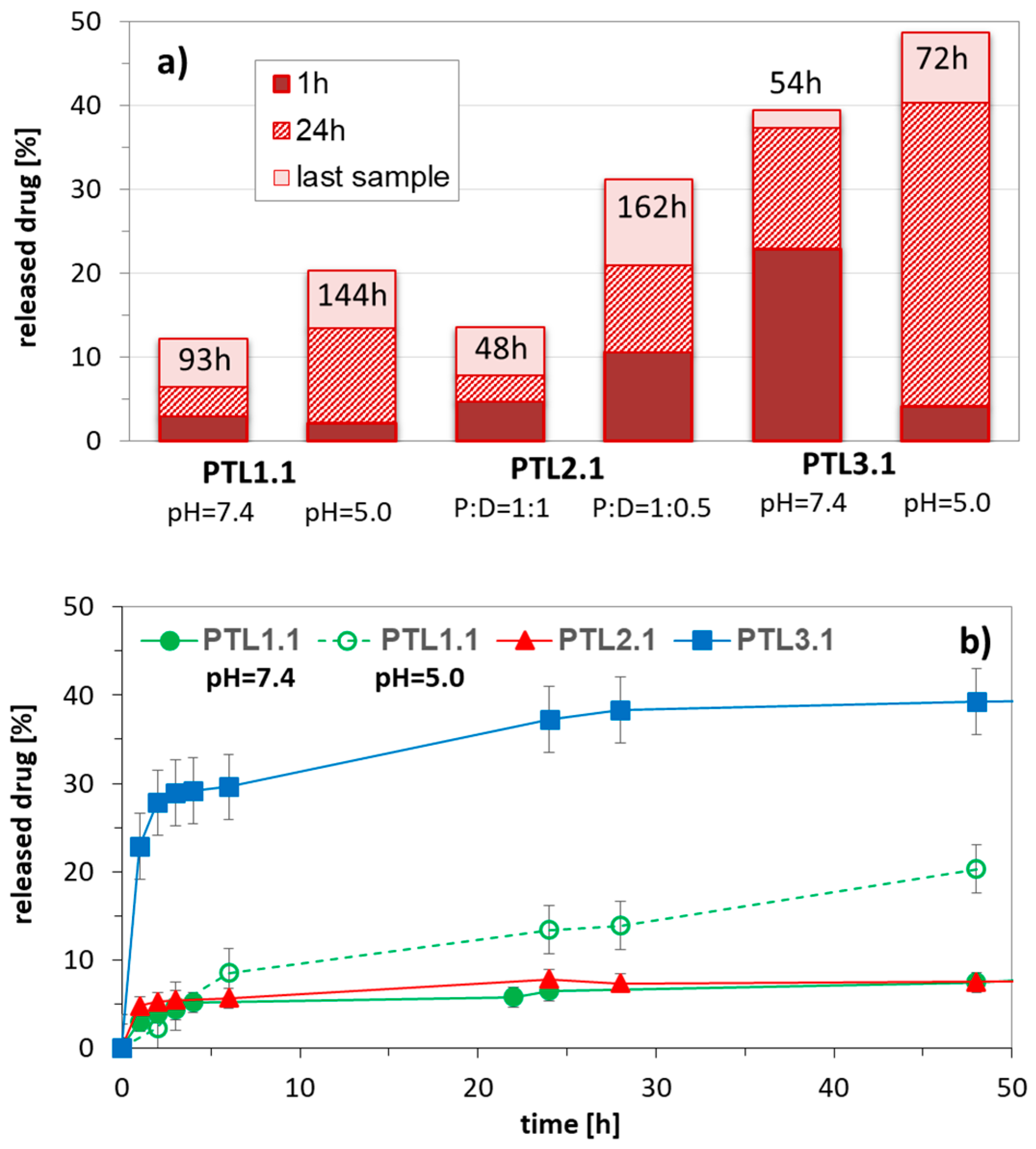
2.1. Poly(Ionic Liquid) Graft Copolymers (DDS Type I)
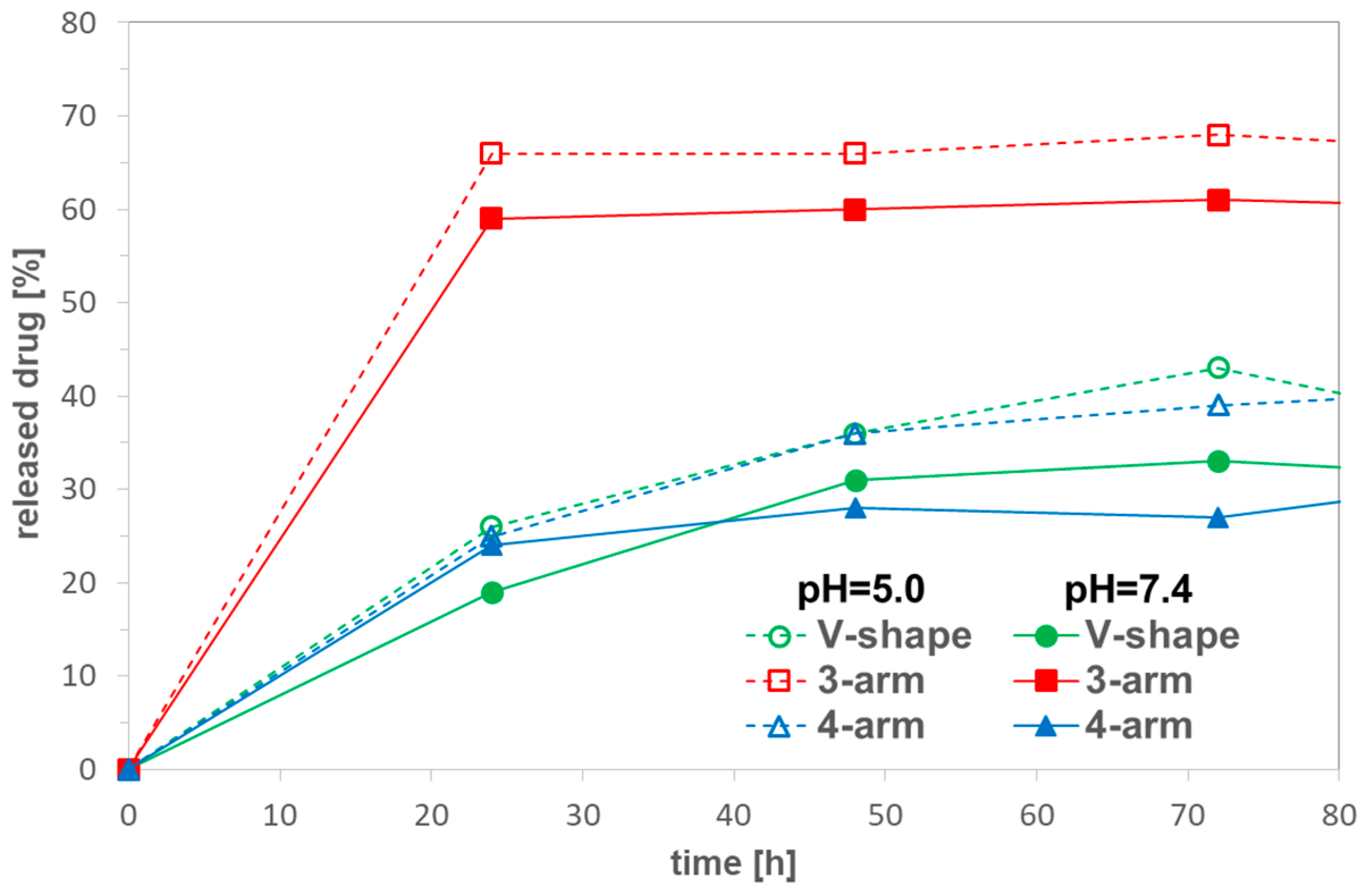
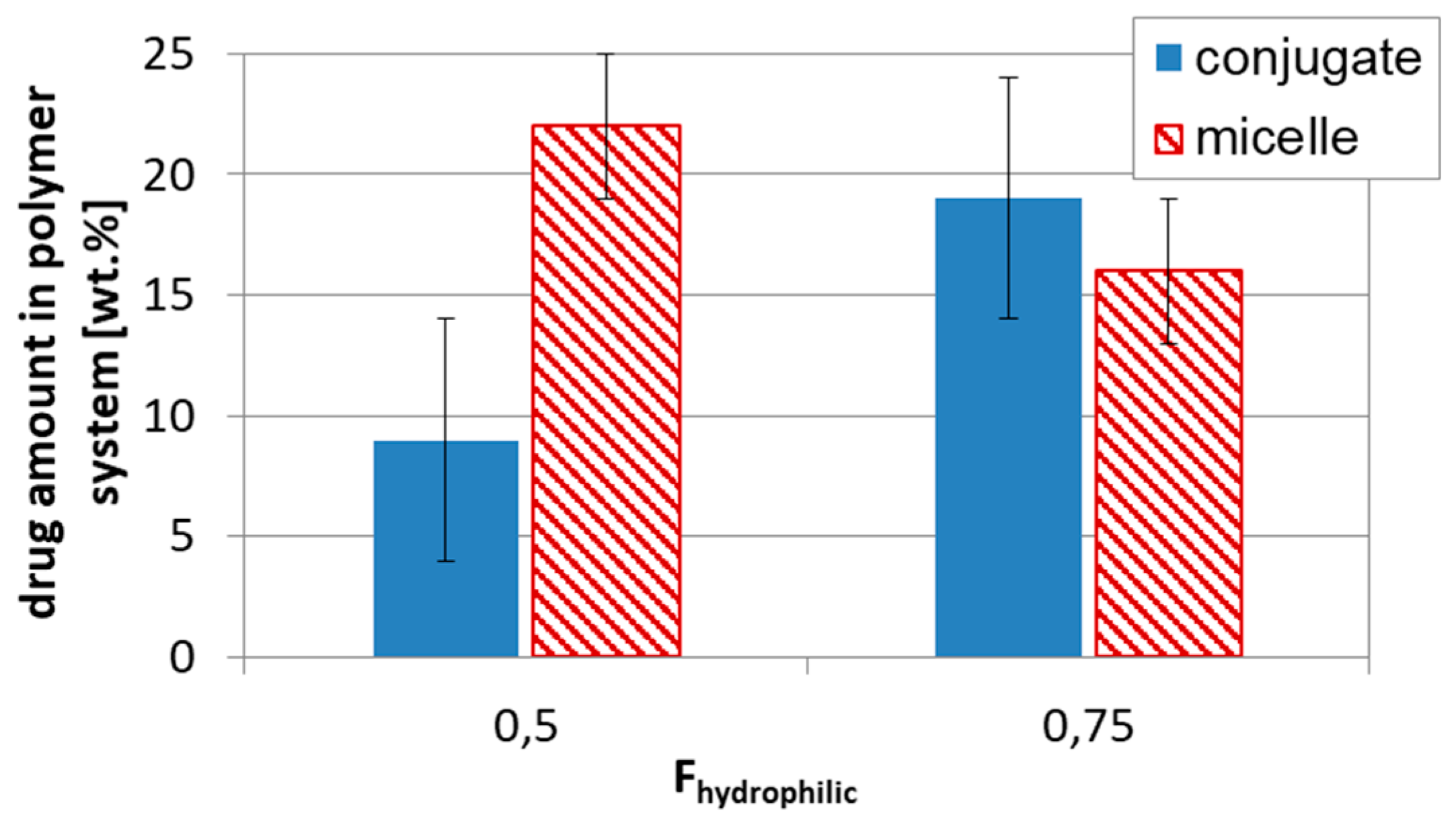
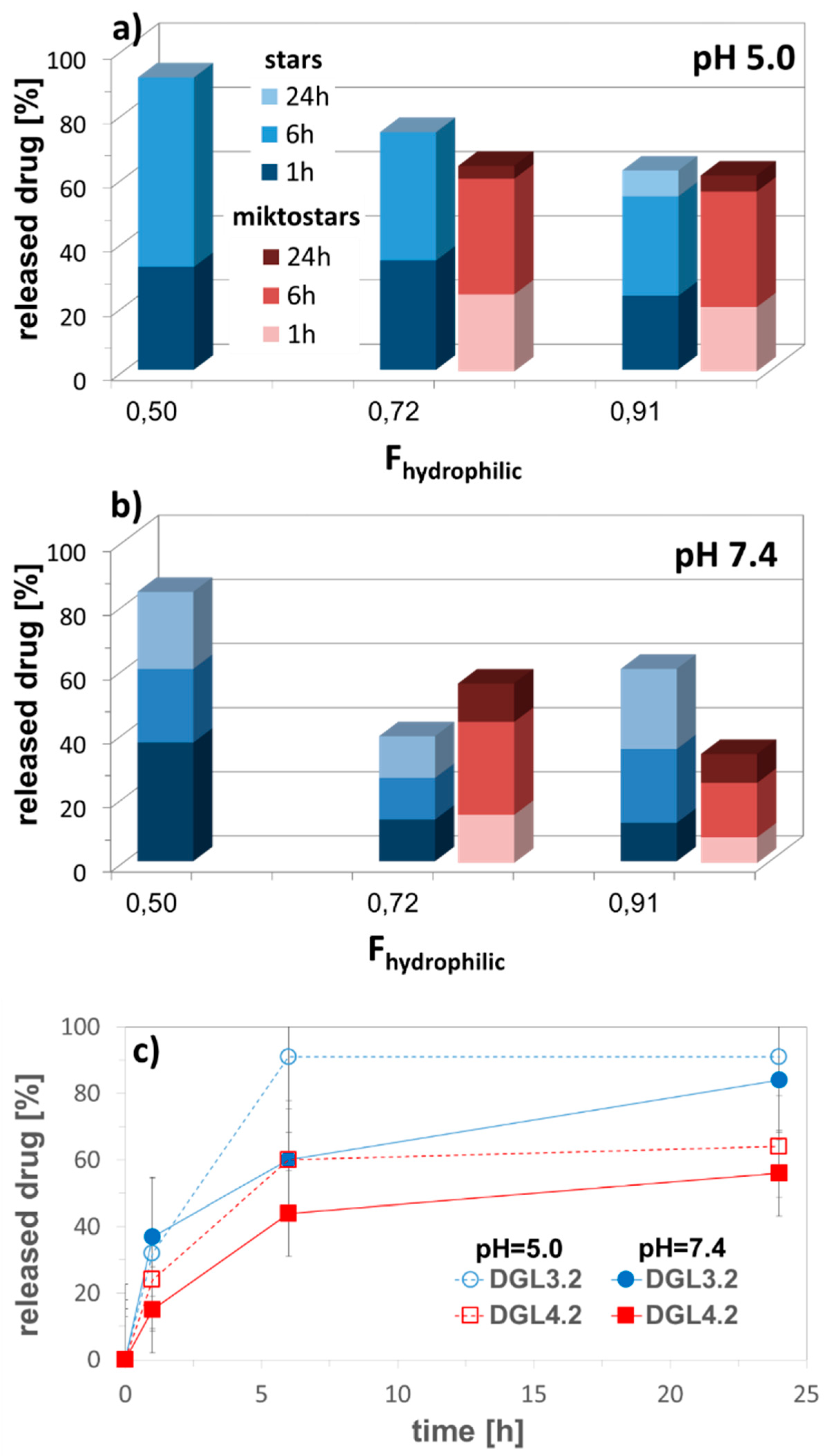
2.2. Conjugates of D-Glucopyranoside Based Star Copolymers (DDS Type II)
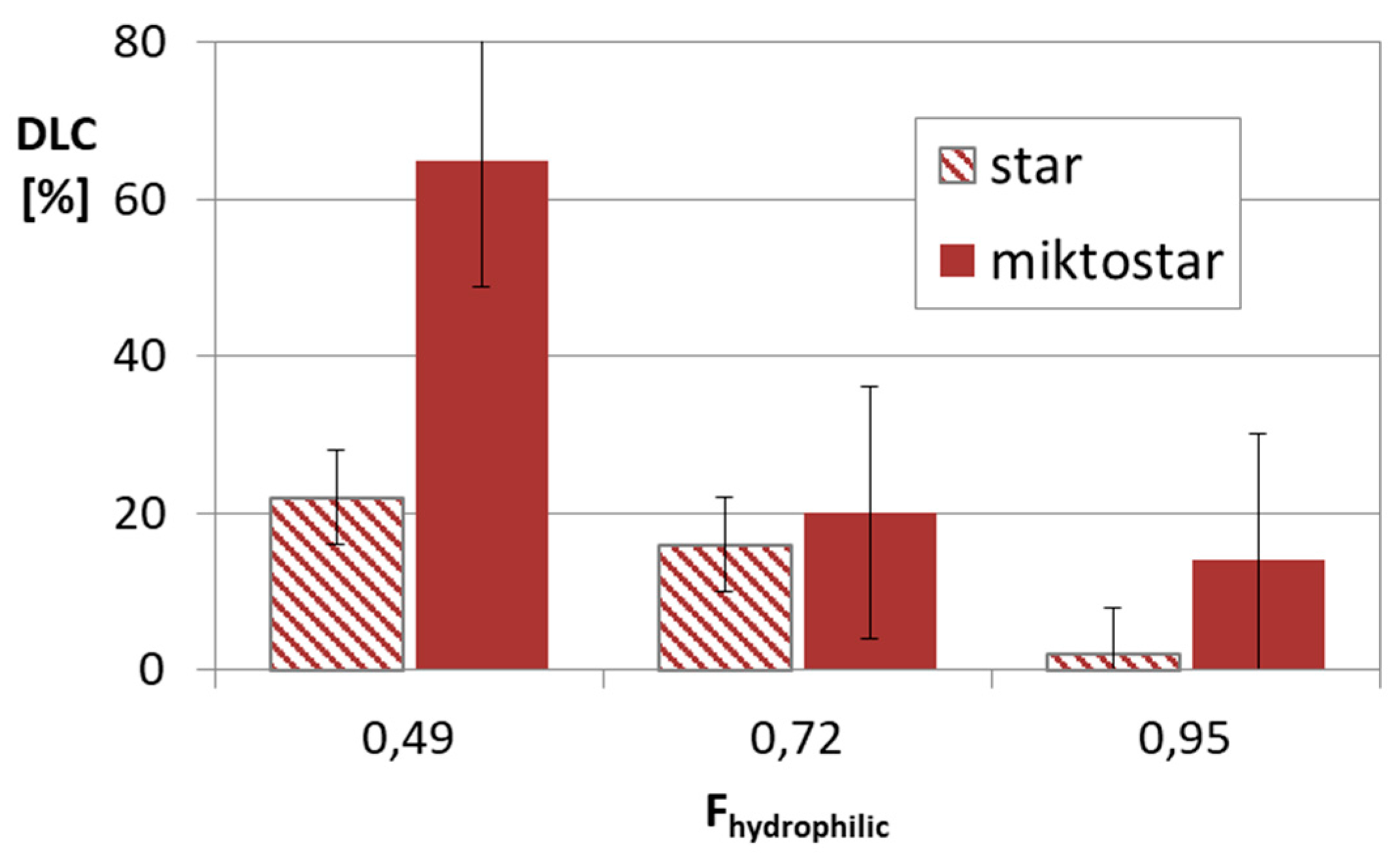
2.3. Self-Assembling Star-Shaped Copolymers (DDS Type III)
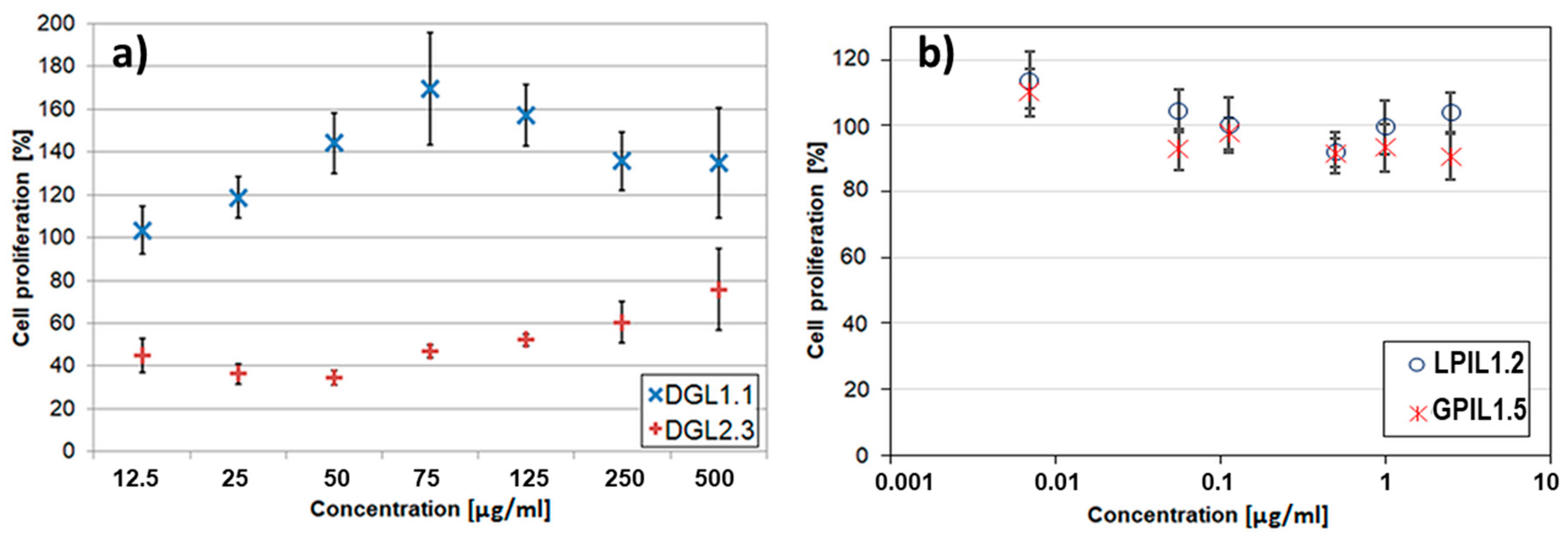
3. Drug Distribution and Cytotoxicity
4. Experimental
4.1. Characterization Techniques
4.2. Cell Viability Assessment
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kadajji, V.G.; Betageri, G.V. Water soluble polymers for pharmaceutical applications. Polymers 2011, 3, 1972–2009. [Google Scholar] [CrossRef]
- Felice, B.; Prabhakaran, M.P.; Rodríguez, A.P.; Ramakrishna, S. Drug delivery vehicles on a nano-engineering perspective. Mater. Sci. Eng. C 2014, 41, 178–195. [Google Scholar] [CrossRef]
- Gao, H.; Matyjaszewski, K. Synthesis of functional polymers with controlled architecture by CRP of monomers in the presence of cross-linkers: From stars to gels. Prog. Polym. Sci. 2009, 34, 317–350. [Google Scholar] [CrossRef]
- Xiao, Y.; Yuan, M.; Zhang, J.; Yan, J.; Lang, M. Functional Poly(ε-caprolactone) Based Materials: Preparation, Self-assembly and Application in Drug Delivery. Curr. Top. Med. Chem. 2014, 14, 781–818. [Google Scholar] [CrossRef]
- Ren, J.M.; McKenzie, T.G.; Fu, Q.; Wong, E.H.H.; Xu, J.; An, Z.; Shanmugam, S.; Davis, T.P.; Boyer, C.; Qiao, G.G. Star Polymers. Chem. Rev. 2016, 116, 6743–6836. [Google Scholar] [CrossRef] [PubMed]
- Ito, S.; Goseki, R.; Ishizone, T.; Hirao, A. Synthesis of well-controlled graft polymers by living anionic polymerization towards exact graft polymers. Polym. Chem. 2014, 5, 5523–5534. [Google Scholar] [CrossRef]
- Neugebauer, D. Two decades of molecular brushes by ATRP. Polymer 2015, 72, 413–421. [Google Scholar] [CrossRef]
- Hadjichristidis, N.; Iatrou, H.; Pitsikalis, M.; Mays, J. Macromolecular architectures by living and controlled/living polymerizations. Prog. Polym. Sci. 2006, 31, 1068–1132. [Google Scholar] [CrossRef]
- Chen, W.; Zhang, J.Z.; Hu, J.; Guo, Q.; Yang, D. Preparation of amphiphilic copolymers for covalent loading of paclitaxel for drug delivery system. J. Polym. Sci. Part A Polym. Chem. 2014, 52, 366–374. [Google Scholar] [CrossRef]
- Luo, C.; Sun, J.; Sun, B.; He, Z. Prodrug-based nanoparticulate drug delivery strategies for cancer therapy. Trends Pharmacol. Sci. 2014, 35, 556–566. [Google Scholar] [CrossRef]
- Riess, G. Micellization of block copolymers. Prog. Polym. Sci. 2003, 28, 1107–1170. [Google Scholar] [CrossRef]
- Tang, Z.; He, C.; Tian, H.; Ding, J.; Hsiao, B.S.; Chu, B.; Chen, X. Polymeric nanostructured materials for biomedical applications. Prog. Polym. Sci. 2016, 60, 86–128. [Google Scholar] [CrossRef]
- Zhang, J.; Li, J.; Shi, Z.; Yang, Y.; Xie, X.; Lee, S.M.Y.; Wang, Y.; Leong, K.W.; Chen, M. pH-sensitive polymeric nanoparticles for co-delivery of doxorubicin and curcumin to treat cancer via enhanced pro-apoptotic and anti-angiogenic activities. Acta Biomater. 2017, 58, 349–364. [Google Scholar] [CrossRef] [PubMed]
- Jang, B.; Kwon, H.; Katila, P.; Lee, S.J.; Lee, H. Dual delivery of biological therapeutics for multimodal and synergistic cancer therapies. Adv. Drug Deliv. Rev. 2016, 98, 113–133. [Google Scholar] [CrossRef] [PubMed]
- Egorova, K.S.; Gordeev, E.G.; Ananikov, V.P. Biological Activity of Ionic Liquids and Their Application in Pharmaceutics and Medicine. Chem. Rev. 2017, 117, 7132–7189. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Zhang, S.; Lu, G.; Huang, X. Constructing Well-Defined Star Graft Copolymers. Polym. Chem. 2013, 4, 1289–1299. [Google Scholar] [CrossRef]
- Sheiko, S.S.; Sumerlin, B.S.; Matyjaszewski, K. Cylindrical molecular brushes: Synthesis, characterization, and properties. Prog. Polym. Sci. 2008, 33, 759–785. [Google Scholar] [CrossRef]
- Jie, P.; Venkatraman, S.S.; Min, F.; Freddy, B.Y.C.; Huat, G.L. Micelle-like nanoparticles of star-branched PEO-PLA copolymers as chemotherapeutic carrier. J. Control. Release 2005, 110, 20–33. [Google Scholar] [CrossRef]
- Zhang, Q.; Remsen, E.E.; Wooley, K.L. Shell cross-linked nanoparticles containing hydrolyrically degradable, crystalline core domains. J. Am. Chem. Soc. 2000, 122, 3642–3651. [Google Scholar] [CrossRef]
- Cheng, G.; Böker, A.; Zhang, M.; Krausch, G.; Müller, A.H.E. Amphiphilic cylindrical core-shell brushes via a “grafting from” process using ATRP. Macromolecules 2001, 34, 6883–6888. [Google Scholar] [CrossRef]
- Moughton, A.O.; Sagawa, T.; Gramlich, W.M.; Seo, M.; Lodge, T.P.; Hillmyer, M.A. Synthesis of block polymer miktobrushes. Polym. Chem. 2013, 4, 166–173. [Google Scholar] [CrossRef]
- Djordjevic, J.; Barch, M.; Uhrich, K.E. Polymeric micelles based on amphiphilic scorpion-like macromolecules: Novel carriers for water-insoluble drugs. Pharm. Res. 2005, 22, 24–32. [Google Scholar] [CrossRef] [PubMed]
- Du, J.Z.; Tang, L.Y.; Song, W.J.; Shi, Y.; Wang, J. Evaluation of polymeric micelles from brush polymer with poly(ε-caprolactone)-b-poly(ethylene glycol) side chains as drug carrier. Biomacromolecules 2009, 10, 2169–2174. [Google Scholar] [CrossRef] [PubMed]
- Qiu, L.Y.; Bae, Y.H. Polymer architecture and drug delivery. Pharm. Res. 2006, 23, 1–30. [Google Scholar] [CrossRef] [PubMed]
- Stridsberg, K.M.; Ryner, M.; Albertsson, A.C. Controlled Ring-Opening Polymerization: Polymers with designed Macromolecular Architecture. In Degradable Aliphatic Polyesters. Advances in Polymer Science; Springer: Berlin/Heidelberg, Germany, 2002; pp. 41–66. [Google Scholar]
- Mori, H.; Müller, A.H.E. New polymeric architectures with (meth)acrylic acid segments. Prog. Polym. Sci. 2003, 28, 1403–1439. [Google Scholar] [CrossRef]
- Lee, H.J.; Bae, Y. Brushed block copolymer micelles with pH-sensitive pendant groups for controlled drug delivery. Pharm. Res. 2013, 30, 2077–2086. [Google Scholar] [CrossRef]
- Gou, P.-F.; Zhu, W.-P.; Zhu, N.; Shen, Z.-Q. Synthesis and Characterization of Novel ResorcinareneCentered Amphiphilic Star-Block Copolymers Consisting of Eight ABA Triblock Arms by Combination of ROP, ATRP, and Click Chemistry. J. Polym. Sci. Part A Polym. Chem. 2009, 47, 2905–2916. [Google Scholar] [CrossRef]
- Qiu, L.Y.; Wu, X.L.; Jin, Y. Doxorubicin-loaded polymeric micelles based on amphiphilic polyphosphazenes with poly(N-isopropylacrylamide-co-N,N-dimethylacrylamide) and ethyl glycinate as side groups: Synthesis, preparation and in vitro evaluation. Pharm. Res. 2009, 26, 946–957. [Google Scholar] [CrossRef]
- Neugebauer, D.; Bury, K.; Biela, T. Novel hydroxyl-functionalized caprolactone poly(meth)acrylates decorated with tert-butyl groups. Macromolecules 2012, 45, 4989–4996. [Google Scholar] [CrossRef]
- Neugebauer, D.; Bury, K.; Paprotna, M.; Biela, T. Amphiphilic copolymers with poly(meth)acrylic acid chains “grafted from” caprolactone 2-(methacryloyloxy)ethyl ester-based backbone. Polym. Adv. Technol. 2013, 24, 1094–1101. [Google Scholar] [CrossRef]
- Maksym-Bębenek, P.; Biela, T.; Neugebauer, D. Synthesis and investigation of monomodal hydroxy-functionalized PEG methacrylate based copolymers with high polymerization degrees. Modification by “grafting from”. React. Funct. Polym. 2014, 82, 33–40. [Google Scholar] [CrossRef]
- Maksym-Bębenek, P.; Biela, T.; Neugebauer, D. Water soluble well-defined acidic graft copolymers based on a poly(propylene glycol) macromonomer. RSC Adv. 2015, 5, 3627–3635. [Google Scholar] [CrossRef]
- Bury, K.; Neugebauer, D. Novel self-assembly graft copolymers as carriers for anti-inflammatory drug delivery. Int. J. Pharm. 2014, 460, 150–157. [Google Scholar] [CrossRef]
- Maksym-Bębenek, P.; Neugebauer, D. Study on Self-Assembled Well-Defined PEG Graft Copolymers as Efficient Drug-Loaded Nanoparticles for Anti-Inflammatory Therapy. Macromol. Biosci. 2015, 15, 1616–1624. [Google Scholar] [CrossRef] [PubMed]
- Maksym, P.; Neugebauer, D. Self-assembling polyether-b-polymethacrylate graft copolymers loaded with indomethacin. Int. J. Polym. Mater. Polym. Biomater. 2017, 66, 317–325. [Google Scholar] [CrossRef]
- Yuan, J.; Antonietti, M. Poly(ionic liquid)s: Polymers expanding classical property profiles. Polymer 2011, 52, 1469–1482. [Google Scholar] [CrossRef]
- Yuan, J.; Mecerreyes, D.; Antonietti, M. Poly(ionic liquid)s: An update. Prog. Polym. Sci. 2013, 38, 1009–1036. [Google Scholar] [CrossRef]
- Bielas, R.; Łukowiec, D.; Neugebauer, D. Drug delivery via anion exchange of salicylate decorating poly(meth)acrylates based on a pharmaceutical ionic liquid. New J. Chem. 2017, 41, 12801–12807. [Google Scholar] [CrossRef]
- Bielas, R.; Mielańczyk, A.; Siewniak, A.; Neugebauer, D. Trimethylammonium-Based Polymethacrylate Ionic Liquids with Tunable Hydrophilicity and Charge Distribution as Carriers of Salicylate Anions. ACS Sustain. Chem. Eng. 2016, 4, 4181–4191. [Google Scholar] [CrossRef]
- Börner, H.G.; Duran, D.; Matyjaszewski, K.; Da Silva, M.; Sheiko, S.S. Synthesis of molecular brushes with gradient in grafting density by atom transfer polymerization. Macromolecules 2002, 35, 3387–3394. [Google Scholar] [CrossRef]
- Bielas, R.; Mielańczyk, A.; Skonieczna, M.; Mielańczyk, Ł.; Neugebauer, D. Choline supported poly(ionic liquid) graft copolymers as novel delivery systems of anionic pharmaceuticals for anti-inflammatory and anti-coagulant therapy. Unpublished.
- Kania, G.; Kwolek, U.; Nakai, K.; Yusa, S.I.; Bednar, J.; Wójcik, T.; Chłopicki, S.; Skórka, T.; Szuwarzyński, M.; Szczubiałka, K.; et al. Stable polymersomes based on ionic-zwitterionic block copolymers modified with superparamagnetic iron oxide nanoparticles for biomedical applications. J. Mater. Chem. B 2015, 3, 5523–5531. [Google Scholar] [CrossRef]
- Bielas, R.; Siewniak, A.; Skonieczna, M.; Adamiec, M.; Mielańczyk, Ł.; Neugebauer, D. Choline based polymethacrylate matrix with pharmaceutical cations as co-delivery system for antibacterial and anti-inflammatory combined therapy. J. Mol. Liq. 2019, 285, 114–122. [Google Scholar] [CrossRef]
- Neugebauer, D.; Mielańczyk, A.; Waśkiewicz, S.; Biela, T. Epoxy functionalized polymethacrylates based on various multifunctional D-glucopyranoside acetals. J. Polym. Sci. Part A Polym. Chem. 2013, 51, 2483–2494. [Google Scholar] [CrossRef]
- Mielańczyk, A.; Odrobińska, J.; Grządka, S.; Mielańczyk, Ł.; Neugebauer, D. Miktoarm star copolymers from D-(-)-salicin core aggregated into dandelion-like structures as anticancer drug delivery systems: Synthesis, self-assembly and drug release. Int. J. Pharm. 2016, 515, 515–526. [Google Scholar] [CrossRef] [PubMed]
- Neugebauer, D.; Odrobińska, J.; Bielas, R.; Mielańczyk, A. Design of systems based on 4-armed star-shaped polyacids for indomethacin delivery. New J. Chem. 2016, 40, 10002–10011. [Google Scholar] [CrossRef]
- Mielańczyk, A.; Neugebauer, D. Synthesis and characterization of D-(-)-Salicine-based star copolymers containing pendant carboxyl groups with fluorophore dyes. J. Polym. Sci. Part A Polym. Chem. 2014, 52, 2399–2411. [Google Scholar] [CrossRef]
- Mielańczyk, A.; Skonieczna, M.; Bernaczek, K.; Neugebauer, D. Fluorescein nanocarriers based on cationic star copolymers with acetal linked sugar cores. Synthesis and biochemical characterization. RSC Adv. 2014, 4, 31904–31913. [Google Scholar] [CrossRef]
- Mielańczyk, A.; Biela, T.; Neugebauer, D. Synthesis and self-assembly behavior of amphiphilic methyl α-D-glucopyranoside-centered copolymers. J. Polym. Res. 2014, 21. [Google Scholar] [CrossRef]
- Mielańczyk, A.; Neugebauer, D. Designing Drug Conjugates Based on Sugar Decorated V-Shape and Star Polymethacrylates: Influence of Composition and Architecture of Polymeric Carrier. Bioconjug. Chem. 2015, 26, 2303–2310. [Google Scholar] [CrossRef] [PubMed]
- Mielańczyk, A.; Skonieczna, M.; Neugebauer, D. Cellular response to star-shaped polyacids. Solution behavior and conjugation advantages. Toxicol. Lett. 2017, 274, 42–50. [Google Scholar] [CrossRef] [PubMed]
- Mielańczyk, A.; Skonieczna, M.; Mielańczyk, Ł.; Neugebauer, D. In Vitro Evaluation of Doxorubicin Conjugates Based on Sugar Core Nonlinear Polymethacrylates toward Anticancer Drug Delivery. Bioconjug. Chem. 2016, 27, 893–904. [Google Scholar] [CrossRef] [PubMed]
- Muggia, F.M. Doxorubicin-polymer conjugates: Further demonstration of the concept of enhanced permeability and retention. Clin. Cancer Res. 1999, 5, 7–8. [Google Scholar] [PubMed]
- Veronese, F.M.; Schiavon, O.; Pasut, G.; Mendichi, R.; Andersson, L.; Tsirk, A.; Ford, J.; Wu, G.; Kneller, S.; Davies, J.; et al. PEG-doxorubicin conjugates: Influence of polymer structure on drug release, in vitro cytotoxicity, biodistribution, and antitumor activity. Bioconjug. Chem. 2005, 16, 775–784. [Google Scholar] [CrossRef] [PubMed]
- Etrych, T.; Strohalm, J.; Chytil, P.; Černoch, P.; Starovoytova, L.; Pechar, M.; Ulbrich, K. Biodegradable star HPMA polymer conjugates of doxorubicin for passive tumor targeting. Eur. J. Pharm. Sci. 2011, 42, 527–539. [Google Scholar] [CrossRef] [PubMed]
- Gao, A.X.; Liao, L.; Johnson, J.A. Synthesis of acid-labile PEG and PEG-doxorubicin-conjugate nanoparticles via Brush-First ROMP. ACS Macro Lett. 2014, 3, 854–857. [Google Scholar] [CrossRef]
- Sun, Y.; Zhang, J.; Han, J.; Tian, B.; Shi, Y.; Ding, Y.; Wang, L.; Han, J. Galactose-Containing Polymer-DOX Conjugates for Targeting Drug Delivery. AAPS PharmSciTech 2017, 18, 749–758. [Google Scholar] [CrossRef] [PubMed]
- Bury, K.; Du Prez, F.; Neugebauer, D. Self-assembling linear and star shaped Poly(Ïμ-caprolactone)/poly[(meth)acrylic acid] block copolymers as carriers of indomethacin and quercetin. Macromol. Biosci. 2013, 13, 1520–1530. [Google Scholar] [CrossRef]

| No. | nsc a | FChMAX b (mol %) | DPsc c | DG d (%) | Mn,NMR (g/mol) | Mn,SEC e (g/mol) | Ð e | Dh f (nm) | DC (%) | Ref. |
|---|---|---|---|---|---|---|---|---|---|---|
| GPIL1.1 | 75 | 25 | 24 | 22 | 201,500 | 17,680 | 1.04 | 28 | 19 | [44] |
| 6g | ||||||||||
| GPIL1.2 | 75 | 67 | 12 | 22 | 127,000 | 19,500 | 1.04 | 56 | 32 | |
| GPIL1.3 | 75 | 71 | 28 | 22 | 314,500 | 19,000 | 1.10 | 51 | 36 | |
| GPIL1.4 | 105 | 74 | 39 | 53 | 724,100 | 30,400 | 1.06 | 22 | 38 | |
| GPIL1.5 | 185 | 74 | 43 | 74 | 1,498,500 | 828,700 | 1.28 | 40 | 39 | |
| LPIL1.1 | - | 78 | 187 * | - | 49,200 | 6200 | 1.25 | 293 | 41 | [39,40] |
| 49 g | [42] | |||||||||
| LPIL1.2 | - | 45 | 119 * | - | 23,100 | 8300 | 1.42 | 232 | 32 | [39,40] |
| 51 g | [42] | |||||||||
| GPIL2.1 | 165 | 19 | 26 | 53 | 579,000 | nd | nd | 24 | 20 g | [44] |
| 32 h | ||||||||||
| LPIL2.1 | - | 26 | 233 * | - | 28,000 | 10,400 | 1.36 | 149 | 45 g | [42] |
| 11 h | [42] |
| No. | Polyelectrolytes | DOX Conjugates | ref. | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| f | Fh-philic a | DParm b | Mn,NMR (g/mol) | Mn,SEC c (g/mol) | Đ c | Dh d (nm) | DC (%) | Dh d (nm) | ||
| DGL1.1 | 4 | 0.56 | 58 | 21,800 | 11,800 | 1.17 | 10 | 5 | insoluble | [48,52] |
| DGL1.2 | 4 | 0.74 | 68 | 24,200 | 10,900 | 1.28 | 8 | 14 | insoluble | |
| DGL1.3 | 6 | 0.51 | 62 | 35,600 | 16,300 | 1.20 | 9 | 6 | insoluble | |
| DGL1.4 | 6 | 0.75 | 54 | 30,100 | insoluble | insoluble | 8 | 19 | insoluble | |
| DGL2.1 | 2 | 0.54 | 51 | 15,800 | nd | nd | 8 | 27 | 8 | [49,51] |
| DGL2.2 | 3 | 0.49 | 57 | 26,500 | nd | nd | 7 | 28 | 8 | |
| DGL2.3 | 4 | 0.53 | 65 | 41,100 | nd | nd | 12 | 17 | 11 | |
| DGL2.4 | 4 | 0.77 | 52 | 38,000 | nd | nd | 12 | 24 | 12 | |
| Polyacids | Polyelectrolytes | Drug-Loaded | ref. | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| f | Fh-philic a | DParm b | Mn,NMR (g/mol) | Mn,SEC c (g/mol) | D c | Dh d (nm) | DLE h (%) | Dh d (nm) | ||
| PTL1.1 | 4 | 0.48 | 34 | 15,400 | 11,600 | 1.17 | 147 e | 74 | 571 | [47] |
| PTL2.1 | 0.36 | 63 | 27,800 | 18,200 | 1.32 | 198 f | 48 | 628 | ||
| PTL2.2 | 0.70 | 67 | 31,800 | 16,800 | 1.31 | 161 f | 6 | 463 | ||
| PTL3.1 | 0.55 | 56 | 27,000 | 17,700 | 1.30 | 198 f | 86 | 874 | ||
| PTL3.2 | 0.76 | 39 | 20,200 | 13,600 | 1.28 | 162 f | 9 | 579 | ||
| PTL3.3 | 0.98 | 31 | 17,400 | 11,500 | 1.24 | 114 g | 7 | 731 | ||
| DGL3.1 | 6 | 0.50 | 55 | 32,100 | insoluble | insoluble | 202 | 66 | 531 | [46,48] |
| DGL3.2 | 0.75 | 50 | 28,700 | 6000 | 1.33 | 165 | 47 | 321 | ||
| DGL3.3 | 0.97 | 47 | 25,700 | insoluble | insoluble | 180 | 7 | 1165 | ||
| DGL4.1 | 8 (6 + 2) * | 0.48 | 54/19 * | 28,700 | 8400 | 1.57 | 517 | 48 | 705 | [46] |
| DGL4.2 | 0.69 | 44/10 * | 28,900 | 4000 | 1.67 | 252 | 42 | 1169 | ||
| DGL4.3 | 0.92 | 65/10 * | 41,900 | 6600 | 1.44 | 384 | 60 | > 10,000 | ||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Neugebauer, D.; Mielańczyk, A.; Bielas, R.; Odrobińska, J.; Kupczak, M.; Niesyto, K. Ionic Polymethacrylate Based Delivery Systems: Effect of Carrier Topology and Drug Loading. Pharmaceutics 2019, 11, 337. https://doi.org/10.3390/pharmaceutics11070337
Neugebauer D, Mielańczyk A, Bielas R, Odrobińska J, Kupczak M, Niesyto K. Ionic Polymethacrylate Based Delivery Systems: Effect of Carrier Topology and Drug Loading. Pharmaceutics. 2019; 11(7):337. https://doi.org/10.3390/pharmaceutics11070337
Chicago/Turabian StyleNeugebauer, Dorota, Anna Mielańczyk, Rafał Bielas, Justyna Odrobińska, Maria Kupczak, and Katarzyna Niesyto. 2019. "Ionic Polymethacrylate Based Delivery Systems: Effect of Carrier Topology and Drug Loading" Pharmaceutics 11, no. 7: 337. https://doi.org/10.3390/pharmaceutics11070337
APA StyleNeugebauer, D., Mielańczyk, A., Bielas, R., Odrobińska, J., Kupczak, M., & Niesyto, K. (2019). Ionic Polymethacrylate Based Delivery Systems: Effect of Carrier Topology and Drug Loading. Pharmaceutics, 11(7), 337. https://doi.org/10.3390/pharmaceutics11070337






