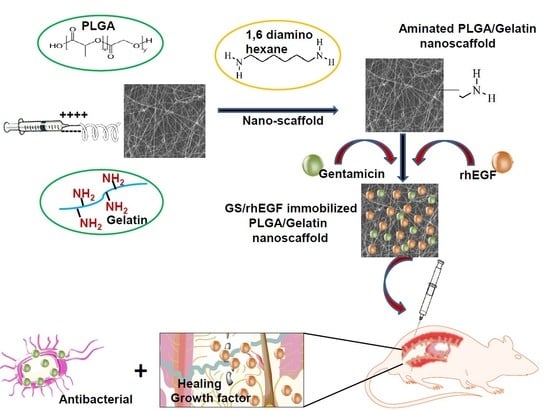
In Vivo Biocompatibility of Electrospun Biodegradable Dual Carrier (Antibiotic + Growth Factor) in a Mouse Model—Implications for Rapid Wound Healing
Abstract
1. Introduction
2. Experimental
2.1. Materials
2.2. Fabrication of GS-Loaded Composite PLGA/Gelatin Nanoscaffolds
2.3. Post-treatment of Composite Nanoscaffolds via Covalent Immobilization of rhEGF
2.4. Surface Topography and Properties
2.5. Quantitative Analysis of rhEGF-Immobilized Composite Nanoscaffolds
2.6. Thermal Analysis
2.7. X-Ray Diffraction
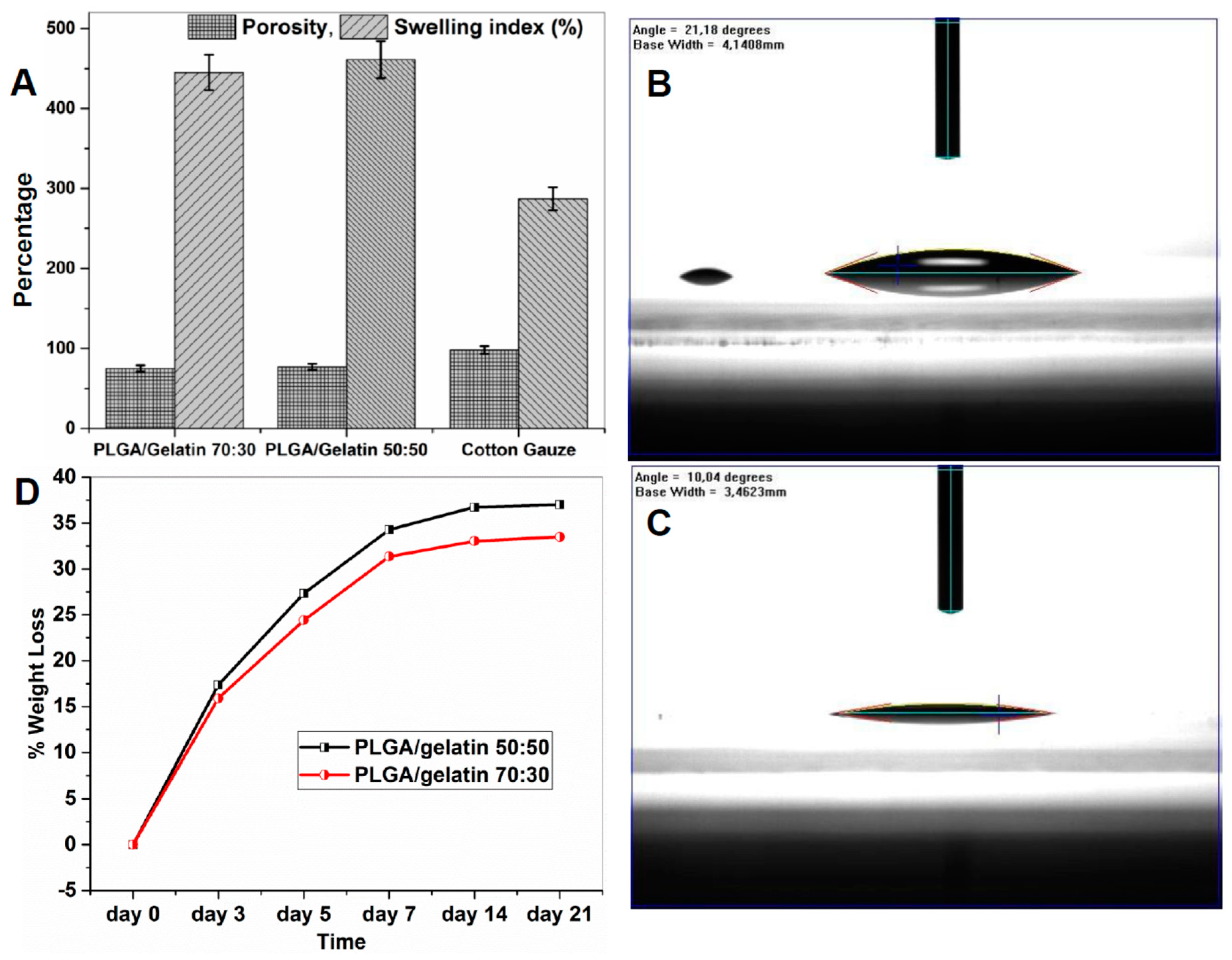
2.8. Degree of Swelling and Porosity.
2.9. Water Contact Angle Measurement
2.10. In Vitro Biodegradability Studies
2.11. Drug-Polymer Profile
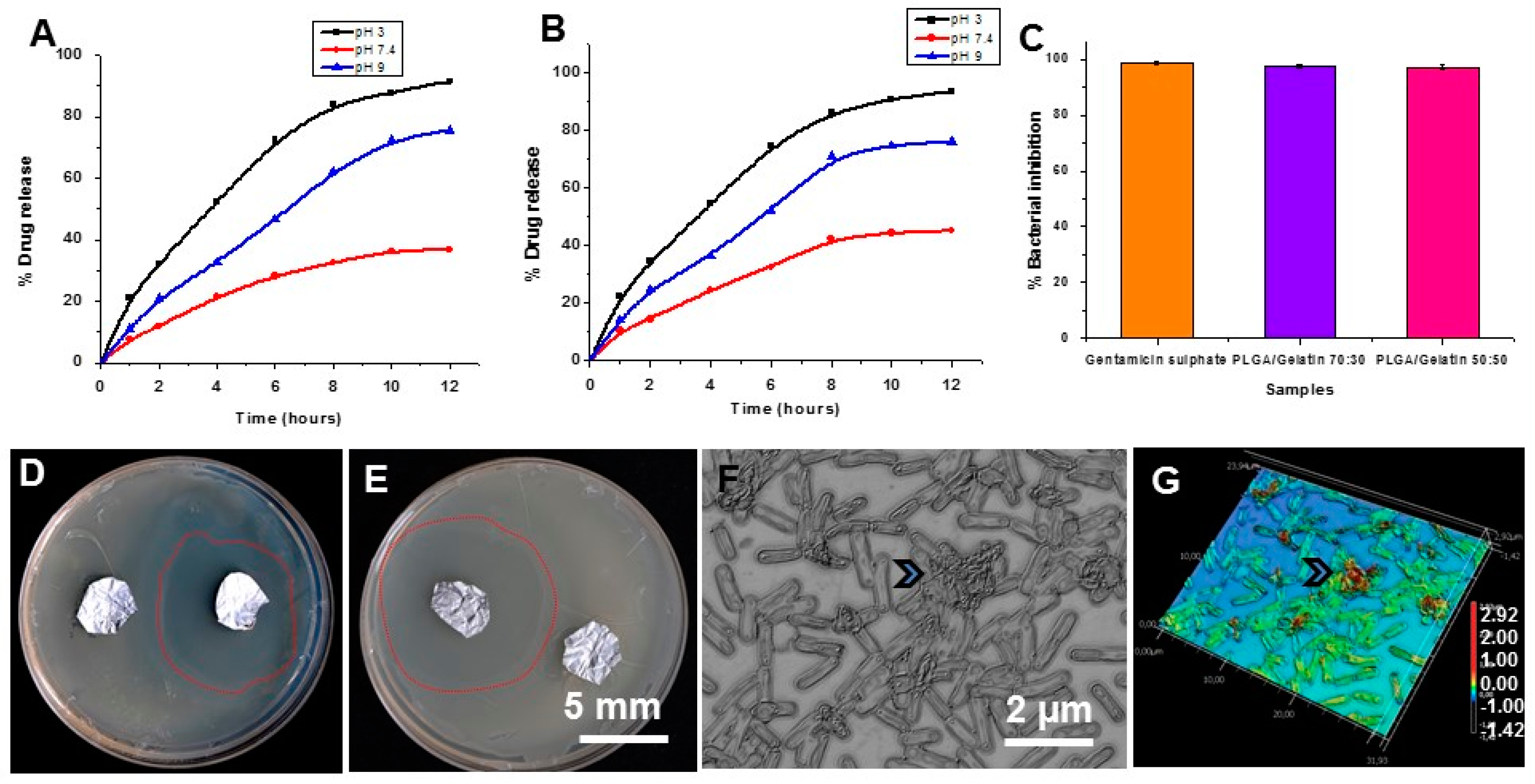
2.12. In Vitro Drug Release Studies
2.13. In Vitro Antibacterial Activity Testing
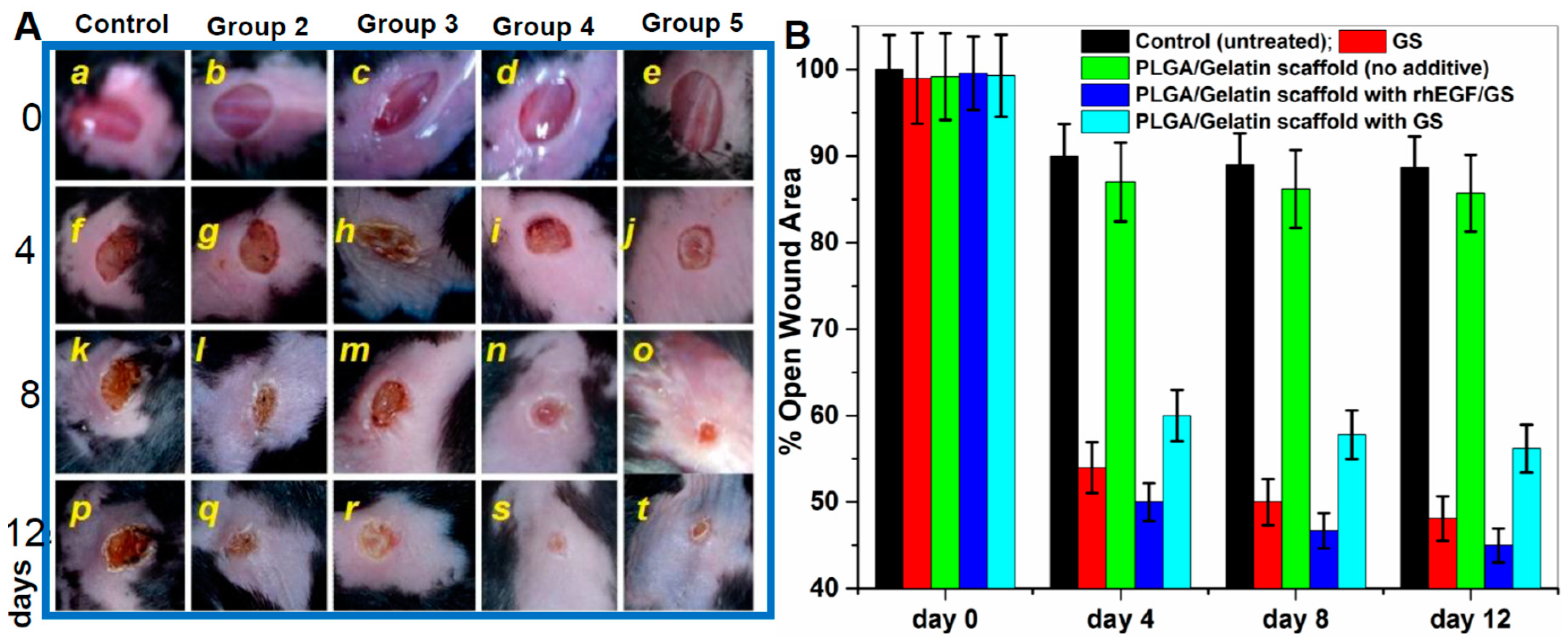
2.14. In Vivo Wound Healing Studies in Diabetic Mice
2.15. Statistical Analysis
3. Results and Discussion
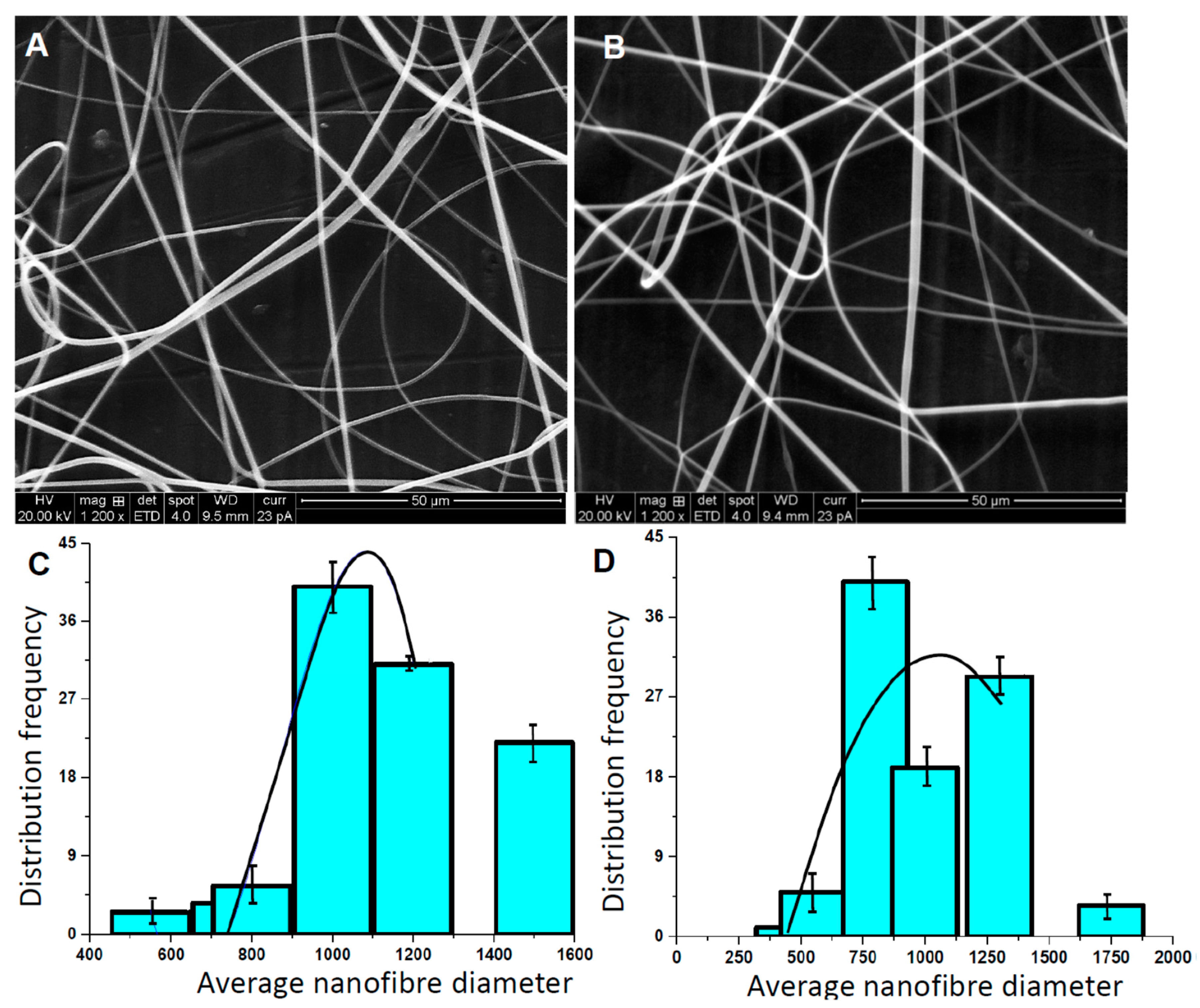
3.1. Surface Topography and Properties of Composite Nanoscaffolds
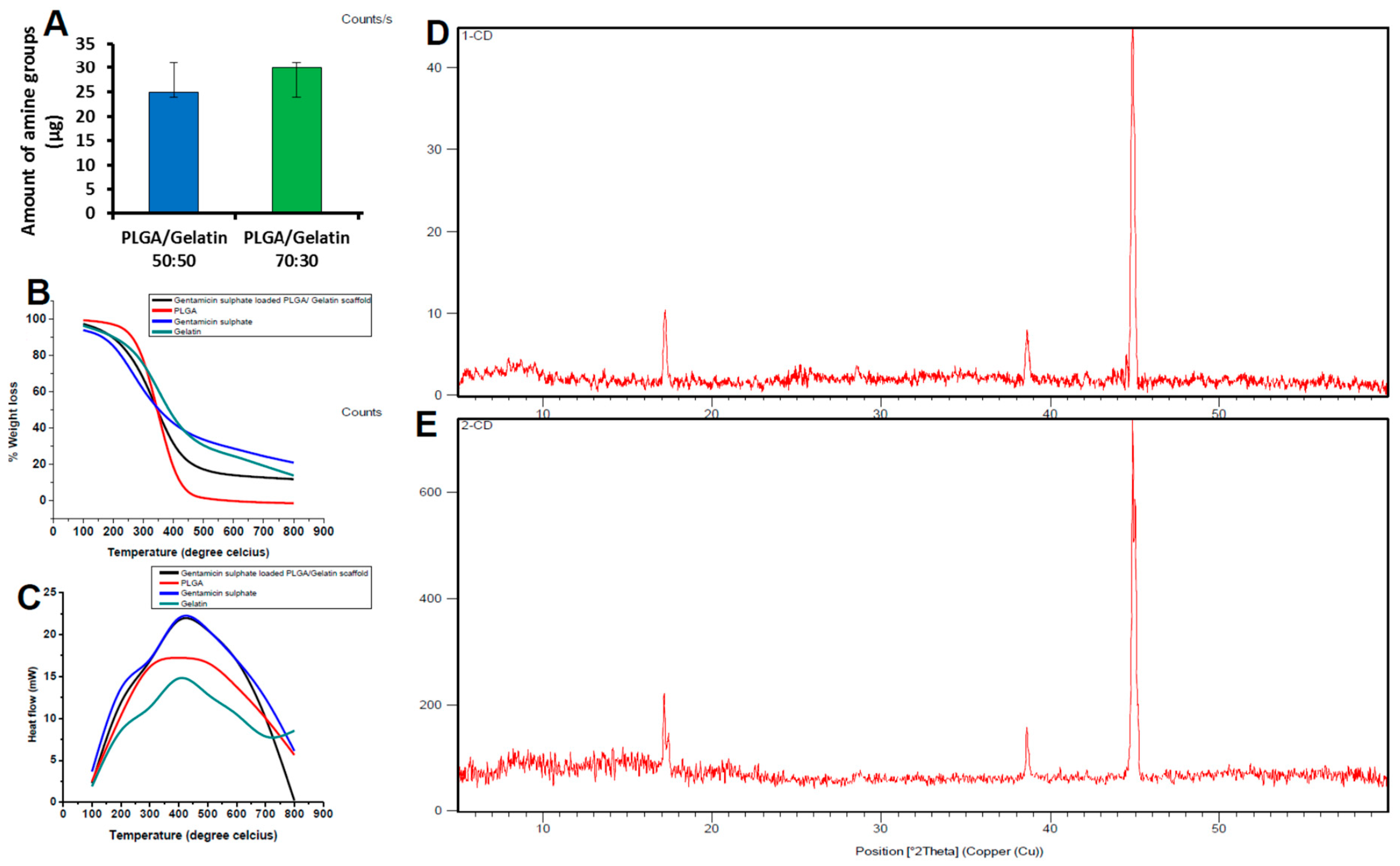
3.2. Quantitative Analysis of rhEGF-Immobilized Nanoscaffolds
3.3. Thermal Analysis
3.4. X-Ray Diffraction Analysis
3.5. Degree of Swelling and Porosity Analysis
3.6. Water Contact Angle Measurements
3.7. In Vitro Biodegradability Studies
3.8. Drug Loading Capacity and Drug Entrapment Efficiency
3.9. In vitro Drug Release Studies
3.10. In Vitro Antibacterial Activity
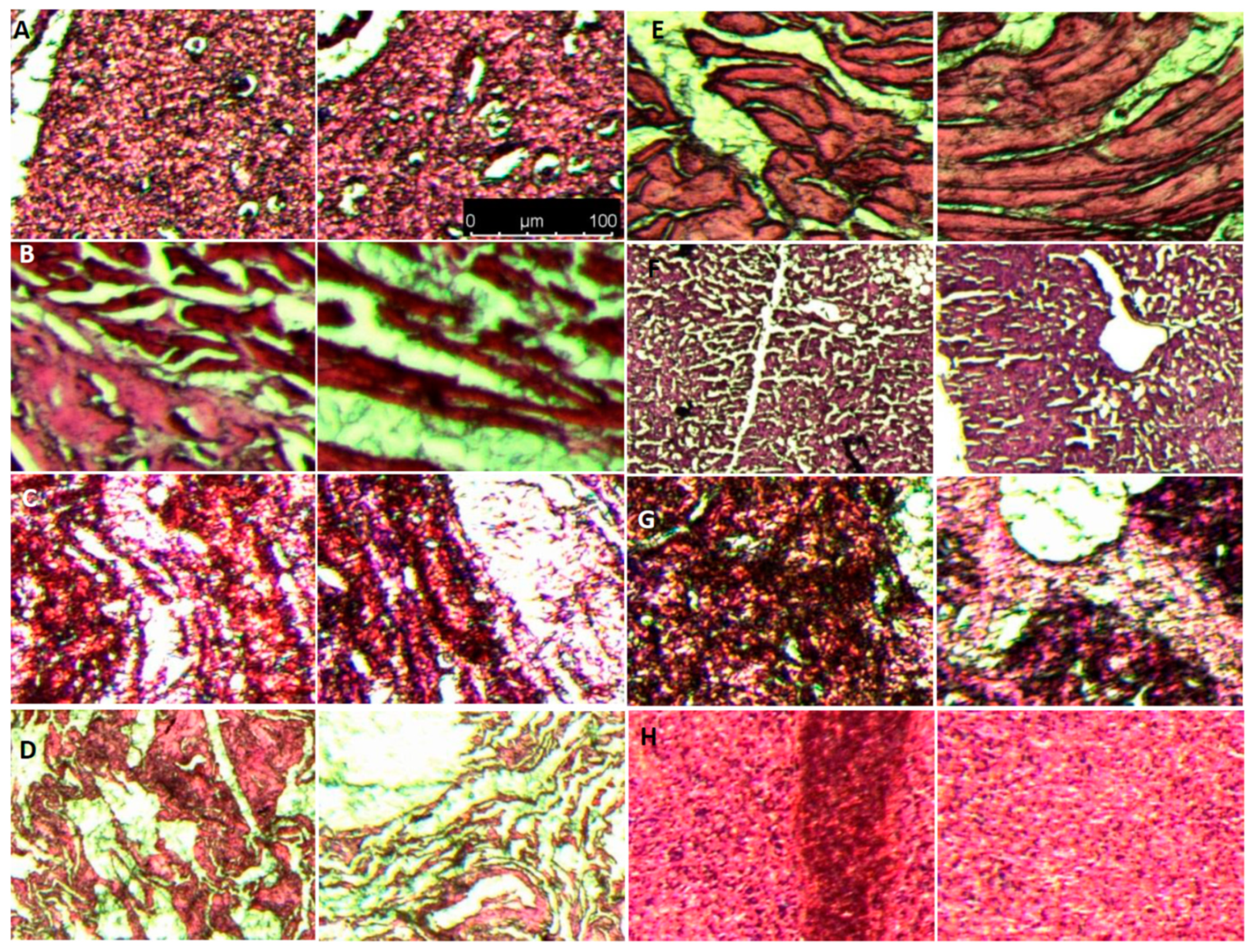
3.11. In Vivo Wound Healing Activity on Diabetic Mice and Organ Toxicity Evaluation
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Xie, Z.; Paras, C.B.; Weng, H.; Punnakitikashem, P.; Su, L.-C.; Vu, K.; Tang, L.; Yang, J.; Nguyen, K.T. Dual growth factor releasing multi-functional nanofibers for wound healing. Acta Biomater. 2013, 9, 9351–9359. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.V.; Subhashree, L.; Milani, P.; Gemmati, D.; Zamboni, P. Interplay of iron metallobiology, metalloproteinases, and fxiii, and role of their gene variants in venous leg ulcer. Int. J. Low. Extrem. Wounds 2010, 9, 166–179. [Google Scholar] [CrossRef]
- Gandhimathi, C.; Venugopal, J.R.; Bhaarathy, V.; Ramakrishna, S.; Kumar, S.D. Biocomposite nanofibrous strategies for the controlled release of biomolecules for skin tissue regeneration. Int. J. Nanomed. 2014, 9, 4709. [Google Scholar]
- Sandhya, S.; Kumar, S.; Vinod, K.; Banji, D.; Kumar, K. Plant as potent anti diabetic and wound healing agent—A review. Hygeia J. Drugs Med. 2011, 3, 11–19. [Google Scholar]
- Tiaka, E.K.; Papanas, N.; Manolakis, A.C.; Maltezos, E. The role of nerve growth factor in the prophylaxis and treatment of diabetic foot ulcers. Int. J. Burn. Trauma 2011, 1, 68. [Google Scholar]
- Djahmi, N.; Messad, N.; Nedjai, S.; Moussaoui, A.; Mazouz, D.; Richard, J.-L.; Sotto, A.; Lavigne, J.-P. Molecular epidemiology of staphylococcus aureus strains isolated from inpatients with infected diabetic foot ulcers in an algerian university hospital. Clin. Microbiol. Infect. 2013, 19, E398–E404. [Google Scholar] [CrossRef] [PubMed]
- Kamtikar, R.; Mitra, N. Clinico microbiological profile of pseudomonas aeruginosa isolated from diabetic foot ulcer. J. Sci. Innov. Res. 2014, 3, 478–481. [Google Scholar]
- Bowen, K. Managing foot infections in patients with diabetes. Aust. Prescr. 2007, 30, 21–24. [Google Scholar] [CrossRef]
- Lipsky, B.A.; Berendt, A.R.; Deery, H.G.; Embil, J.M.; Joseph, W.S.; Karchmer, A.W.; LeFrock, J.L.; Lew, D.P.; Mader, J.T.; Norden, C. Diagnosis and treatment of diabetic foot infections. Clin. Infect. Dis. 2004, 39, 885–910. [Google Scholar] [CrossRef] [PubMed]
- Spichler, A.; Hurwitz, B.L.; Armstrong, D.G.; Lipsky, B.A. Microbiology of diabetic foot infections: From louis pasteur to ‘crime scene investigation’. BMC Med. 2015, 13, 2. [Google Scholar] [CrossRef]
- Trivedi, U.; Parameswaran, S.; Armstrong, A.; Burgueno-Vega, D.; Griswold, J.; Dissanaike, S.; Rumbaugh, K.P. Prevalence of multiple antibiotic resistant infections in diabetic versus nondiabetic wounds. J. Pathog. 2014, 2014, 173053. [Google Scholar] [CrossRef] [PubMed]
- Dwivedi, C.; Pandey, H.; Pandey, A.C.; Ramteke, P.W. Nanofiber based smart pharmaceutical scaffolds for wound repair and regenerations. Curr. Pharm. Des. 2016, 22, 1460–1471. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.S.; Leong, K.W.; Yoo, H.S. In vivo wound healing of diabetic ulcers using electrospun nanofibers immobilized with human epidermal growth factor (EGF). Biomaterials 2008, 29, 587–596. [Google Scholar] [CrossRef] [PubMed]
- Herbst, R.S. Review of epidermal growth factor receptor biology. Int. J. Radiat. Oncol. Biol. Phys. 2004, 59, S21–S26. [Google Scholar] [CrossRef]
- Fallon, J.H.; Seroogy, K.B.; Loughlin, S.E.; Morrison, R.S.; Bradshaw, R.A.; Knaver, D.; Cunningham, D.D. Epidermal growth factor immunoreactive material in the central nervous system: Location and development. Science 1984, 224, 1107–1109. [Google Scholar] [CrossRef] [PubMed]
- Vikram Singh, A.; Hasan Dad Ansari, M.; Wang, S.; Laux, P.; Luch, A.; Kumar, A.; Patil, R.; Nussberger, S. The adoption of three-dimensional additive manufacturing from biomedical material design to 3d organ printing. Appl. Sci. 2019, 9, 811. [Google Scholar] [CrossRef]
- Singh, V.; Kashyap, S.; Yadav, U.; Srivastava, A.; Singh, A.V.; Singh, R.K.; Singh, S.K.; Saxena, P.S. Nitrogen doped carbon quantum dots demonstrate no toxicity under in vitro conditions in a cervical cell line and in vivo in swiss albino mice. Toxicol. Res. 2019. [Google Scholar] [CrossRef]
- Dwivedi, C.; Pandey, I.; Misra, V.; Giulbudagian, M.; Jungnickel, H.; Laux, P.; Luch, A.; Ramteke, P.W.; Singh, A.V. The prospective role of nanobiotechnology in food and food packaging products. Integr. Food Nutr. Metab. (IFNM) 2018. [Google Scholar] [CrossRef]
- Abdel-Wahhab, M.A.; Márquez, F. Nanomaterials in biomedicine. Soft Nanosci. Lett. 2015, 5, 53–54. [Google Scholar] [CrossRef]
- Singh, A.V.; Laux, P.; Luch, A.; Sudrik, C.; Wiehr, S.; Wild, A.-M.; Santomauro, G.; Bill, J.; Sitti, M. Review of emerging concepts in nanotoxicology: Opportunities and challenges for safer nanomaterial design. Toxicol. Mech. Methods 2019, 1–10. [Google Scholar] [CrossRef]
- Dwivedi, C.; Pandey, I.; Pandey, H.; Patil, S.; Mishra, S.B.; Pandey, A.C.; Zamboni, P.; Ramteke, P.W.; Singh, A.V. In vivo diabetic wound healing with nanofibrous scaffolds modified with gentamicin and recombinant human epidermal growth factor. J. Biomed. Mater. Res. Part A 2018, 106, 641–651. [Google Scholar] [CrossRef] [PubMed]
- Gupta, K.C.; Haider, A.; Choi, Y.-R.; Kang, I.-K. Nanofibrous scaffolds in biomedical applications. Biomater. Res. 2014, 18, 5. [Google Scholar] [CrossRef] [PubMed]
- Venugopal, J.; Ramakrishna, S. Applications of polymer nanofibers in biomedicine and biotechnology. Appl. Biochem. Biotechnol. 2005, 125, 147–157. [Google Scholar] [CrossRef]
- Wang, S.; Zheng, F.; Huang, Y.; Fang, Y.; Shen, M.; Zhu, M.; Shi, X. Encapsulation of amoxicillin within laponite-doped poly (lactic-co-glycolic acid) nanofibers: Preparation, characterization, and antibacterial activity. ACS Appl. Mater. Interfaces 2012, 4, 6393–6401. [Google Scholar] [CrossRef]
- Hajiali, H.; Shahgasempour, S.; Naimi-Jamal, M.R.; Peirovi, H. Electrospun pga/gelatin nanofibrous scaffolds and their potential application in vascular tissue engineering. Int. J. Nanomed. 2011, 6, 2133. [Google Scholar] [CrossRef] [PubMed]
- Nigam, R.; Mahanta, B. An overview of various biomimetic scaffolds: Challenges and applications in tissue engineering. J. Tissue Sci. Eng. 2014, 5, 1. [Google Scholar]
- Steed, D.L. The role of growth factors in wound healing. Surg. Clin. N. Am. 1997, 77, 575–586. [Google Scholar] [CrossRef]
- Fouad, H.; Elsarnagawy, T.; Almajhdi, F.N.; Khalil, K.A. Preparation and in vitro thermo-mechanical characterization of electrospun plga nanofibers for soft and hard tissue replacement. Int. J. Electrochem. Sci. 2013, 8, 2293–2304. [Google Scholar]
- Ansary, R.H.; Awang, M.B.; Rahman, M.M. Biodegradable poly (d,l-lactic-co-glycolic acid)-based micro/nanoparticles for sustained release of protein drugs-a review. Trop. J. Pharm. Res. 2014, 13, 1179–1190. [Google Scholar] [CrossRef]
- Dwivedi, C.; Pandey, H.; Pandey, A.C.; Ramteke, P.W. Novel gentamicin loaded electrospun nanofibrous scaffolds for wound healing: An in-vitro study. Int. J. Pharm. Sci. Res. 2013, 4, 2224–2227. [Google Scholar]
- Singh, A.V.; Aditi, A.S.; Gade, W.N.; Vats, T.; Lenardi, C.; Milani, P. Nanomaterials: New generation therapeutics in wound healing and tissue repair. Curr. Nanosci. 2010, 6, 577–586. [Google Scholar] [CrossRef]
- Singh, D.; Saraf, S.; Dixit, V.K.; Saraf, S. Formulation optimization of gentamicin loaded eudragit rs100 microspheres using factorial design study. Biol. Pharm. Bull. 2008, 31, 662–667. [Google Scholar] [CrossRef][Green Version]
- Deng, A.; Yang, Y.; Du, S.; Yang, S. Electrospinning of in situ crosslinked recombinant human collagen peptide/chitosan nanofibers for wound healing. Biomater. Sci. 2018, 6, 2197–2208. [Google Scholar] [CrossRef] [PubMed]
- Kurečič, M.; Maver, T.; Virant, N.; Ojstršek, A.; Gradišnik, L.; Hribernik, S.; Kolar, M.; Maver, U.; Kleinschek, K.S. A multifunctional electrospun and dual nano-carrier biobased system for simultaneous detection of ph in the wound bed and controlled release of benzocaine. Cellulose 2018, 25, 7277–7297. [Google Scholar] [CrossRef]
- Maver, T.; Smrke, D.; Kurečič, M.; Gradišnik, L.; Maver, U.; Kleinschek, K.S. Combining 3d printing and electrospinning for preparation of pain-relieving wound-dressing materials. J. Sol-Gel Sci. Technol. 2018, 88, 33–48. [Google Scholar] [CrossRef]
- Shi, R.; Geng, H.; Gong, M.; Ye, J.; Wu, C.; Hu, X.; Zhang, L. Long-acting and broad-spectrum antimicrobial electrospun poly (ε-caprolactone)/gelatin micro/nanofibers for wound dressing. J. Colloid Interface Sci. 2018, 509, 275–284. [Google Scholar] [CrossRef] [PubMed]
- Dwivedi, C.; Pandey, H.; Pandey, A.C.; Ramteke, P.W. Fabrication and assessment of gentamicin loaded electrospun nanofibrous scaffolds as a quick wound healing dressing material. Curr. Nanosci. 2015, 11, 222–228. [Google Scholar] [CrossRef]
- Gentile, P.; Chiono, V.; Carmagnola, I.; Hatton, P. An overview of poly (lactic-co-glycolic) acid (PLGA)-based biomaterials for bone tissue engineering. Int. J. Mol. Sci. 2014, 15, 3640–3659. [Google Scholar] [CrossRef]
- Singh, A.V.; Mehta, K.K.; Worley, K.; Dordick, J.S.; Kane, R.S.; Wan, L.Q. Carbon nanotube-induced loss of multicellular chirality on micropatterned substrate is mediated by oxidative stress. ACS Nano 2014, 8, 2196–2205. [Google Scholar] [CrossRef]
- Singh, A.V.; Hosseinidoust, Z.; Park, B.-W.; Yasa, O.; Sitti, M. Microemulsion-based soft bacteria-driven microswimmers for active cargo delivery. ACS Nano 2017, 11, 9759–9769. [Google Scholar] [CrossRef]
- Pandey, H.; Parashar, V.; Parashar, R.; Prakash, R.; Ramteke, P.W.; Pandey, A.C. Controlled drug release characteristics and enhanced antibacterial effect of graphene nanosheets containing gentamicin sulfate. Nanoscale 2011, 3, 4104–4108. [Google Scholar] [CrossRef] [PubMed]
- Maver, T.; Gradišnik, L.; Smrke, D.M.; Kleinschek, K.S.; Maver, U. Systematic evaluation of a diclofenac-loaded carboxymethyl cellulose-based wound dressing and its release performance with changing ph and temperature. AAPS PharmSciTech 2019, 20, 29. [Google Scholar] [CrossRef] [PubMed]
- Meng, Z.; Xu, X.; Zheng, W.; Zhou, H.; Li, L.; Zheng, Y.; Lou, X. Preparation and characterization of electrospun plga/gelatin nanofibers as a potential drug delivery system. Colloids Surf. B Biointerfaces 2011, 84, 97–102. [Google Scholar] [CrossRef] [PubMed]
- Meng, Z.; Wang, Y.; Ma, C.; Zheng, W.; Li, L.; Zheng, Y. Electrospinning of plga/gelatin randomly-oriented and aligned nanofibers as potential scaffold in tissue engineering. Mater. Sci. Eng. C 2010, 30, 1204–1210. [Google Scholar] [CrossRef]
- Pal, K.; Banthia, A.K.; Majumdar, D.K. Polyvinyl alcohol–glycine composite membranes: Preparation, characterization, drug release and cytocompatibility studies. Biomed. Mater. 2006, 1, 49. [Google Scholar] [CrossRef]
- Murugan, R.; Ramakrishna, S. Nano-featured scaffolds for tissue engineering: A review of spinning methodologies. Tissue Eng. 2006, 12, 435–447. [Google Scholar] [CrossRef]
- Chong, E.; Phan, T.; Lim, I.; Zhang, Y.; Bay, B.; Ramakrishna, S.; Lim, C. Evaluation of electrospun pcl/gelatin nanofibrous scaffold for wound healing and layered dermal reconstitution. Acta Biomater. 2007, 3, 321–330. [Google Scholar] [CrossRef]
- Gander, B.; Gamazo, C.; Irache, J.M.; Prior, S. Gentamicin-loaded microspheres for treatment of experimental brucella abortus infection in mice. J. Antimicrob. Chemother. 2005, 55, 1032–1036. [Google Scholar]
- Singh, A.V.; Baylan, S.; Park, B.W.; Richter, G.; Sitti, M. Hydrophobic pinning with copper nanowhiskers leads to bactericidal properties. PLoS ONE 2017, 12, e0175428. [Google Scholar] [CrossRef]
- Singh, V.; Kumar, V.; Kashyap, S.; Singh, A.V.; Kishore, V.; Sitti, M.; Saxena, P.S.; Srivastava, A. Graphene oxide synergistically enhances antibiotic efficacy in vancomycin-resistant staphylococcus aureus. ACS Appl. Bio Mater. 2019, 2, 1148–1157. [Google Scholar] [CrossRef]
- Singh, A.V.; Gemmati, D.; Kanase, A.; Pandey, I.; Misra, V.; Kishore, V.; Jahnke, T.; Bill, J. Nanobiomaterials for vascular biology and wound management: A review. Veins Lymphat. 2018, 7. [Google Scholar] [CrossRef]
- Vikram Singh, A. Editorial (thematic issue: Recent trends in nano-biotechnology reinforcing contemporary pharmaceutical design). Curr. Pharm. Des. 2016, 22, 1415–1417. [Google Scholar] [CrossRef]
| Nanofibrous Scaffold | Drug Loading Capacity (µg) | Drug Entrapment Efficiency (%) |
|---|---|---|
| PLGA/Gelatin 50:50 | 161.68 ± 13.2 | 80.26 ± 6.9 |
| PLGA/Gelatin 70:30 | 174.24 ± 19 | 87.12 ± 8.1 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dwivedi, C.; Pandey, H.; Pandey, A.C.; Patil, S.; Ramteke, P.W.; Laux, P.; Luch, A.; Singh, A.V. In Vivo Biocompatibility of Electrospun Biodegradable Dual Carrier (Antibiotic + Growth Factor) in a Mouse Model—Implications for Rapid Wound Healing. Pharmaceutics 2019, 11, 180. https://doi.org/10.3390/pharmaceutics11040180
Dwivedi C, Pandey H, Pandey AC, Patil S, Ramteke PW, Laux P, Luch A, Singh AV. In Vivo Biocompatibility of Electrospun Biodegradable Dual Carrier (Antibiotic + Growth Factor) in a Mouse Model—Implications for Rapid Wound Healing. Pharmaceutics. 2019; 11(4):180. https://doi.org/10.3390/pharmaceutics11040180
Chicago/Turabian StyleDwivedi, Charu, Himanshu Pandey, Avinash C. Pandey, Sandip Patil, Pramod W. Ramteke, Peter Laux, Andreas Luch, and Ajay Vikram Singh. 2019. "In Vivo Biocompatibility of Electrospun Biodegradable Dual Carrier (Antibiotic + Growth Factor) in a Mouse Model—Implications for Rapid Wound Healing" Pharmaceutics 11, no. 4: 180. https://doi.org/10.3390/pharmaceutics11040180
APA StyleDwivedi, C., Pandey, H., Pandey, A. C., Patil, S., Ramteke, P. W., Laux, P., Luch, A., & Singh, A. V. (2019). In Vivo Biocompatibility of Electrospun Biodegradable Dual Carrier (Antibiotic + Growth Factor) in a Mouse Model—Implications for Rapid Wound Healing. Pharmaceutics, 11(4), 180. https://doi.org/10.3390/pharmaceutics11040180